Microencapsulation of Camelina sativa Oil Using Selected Soluble Fractions of Dietary Fiber as the Wall Material
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Emulsions and Microcapsules
2.3. Emulsions Stability
2.4. Measurement of Emulsion Particle Size
2.5. Density and Humidity of Microcapsules
2.6. Colorimetric Measurement
2.7. The Particle Size of the Microcapsules
2.8. Determination of Microencapsulation Efficiency
2.8.1. Measurement of Surface Oil
2.8.2. Measurement of Total Oil
2.9. Fatty Acids Profile
2.10. Peroxidase Value
2.11. SEM Analysis
2.12. Statistical Analysis
3. Results and Discussion
3.1. Emulsion Stability
3.2. Measurement of Emulsion Particle Size
3.3. Density and Humidity of Microcapsules
3.4. Colorimetric Measurement
3.5. The Particle Size of the Microcapsules
3.6. Microencapsulation Efficiency
3.7. Fatty Acids Profile
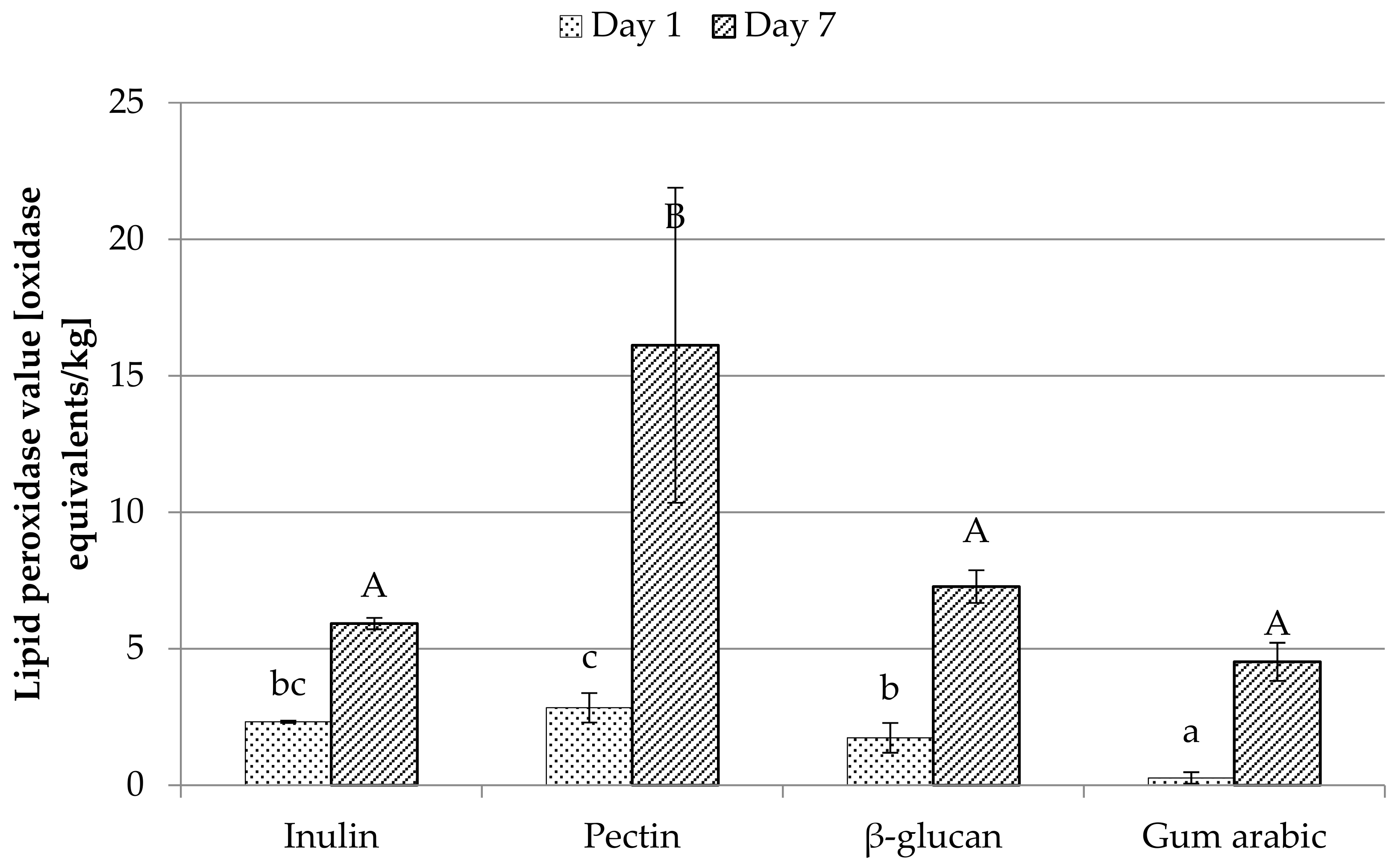
3.8. Peroxidase Value
3.9. SEM Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bakry, A.M.; Abbas, S.; Ali, B.; Majeed, H.; Abouelwafa, M.Y.; Mousa, A.; Liang, L. Microencapsulation of Oils: A Comprehensive Review of Benefits, Techniques, and Applications. Compr. Rev. Food Sci. Food Saf. 2016, 15, 143–182. [Google Scholar] [CrossRef]
- Poshardi, A.; Aparna, K. Microencapsulation technology: A review. J. Res. ANGRAU 2010, 38, 86–102. [Google Scholar]
- Singh, M.; Dua, J.S.; Menra, M.; Soni, M.; Prasad, D.N. Microencapsulation and its various aspects: A review. Int. J. Adv. Res. 2016, 4, 2094–2108. [Google Scholar] [CrossRef]
- Abramovič, H.; Abram, V. Physico-Chemical Properties, Composition and Oxidative Stability of Camelina sativa Oil. Food Technol. Biotechnol. 2005, 43, 63–70. [Google Scholar]
- O’Dwyer, S.P.; O’Beirne, D.; Eidhin, D.N.; O’Kennedy, B.T. Effects of emulsification and microencapsulation on the oxidative stability of camelina and sunflowers oils. J. Microencapsul. 2013, 30, 451–459. [Google Scholar] [CrossRef]
- Belayneh, H.D.; Wehling, R.L.; Zhang, Y.; Ciftci, O.N. Development of omega-3-rich Camelina sativa seed oil emulsions. Food Sci. Nutr. 2017, 6, 440–449. [Google Scholar] [CrossRef]
- Mudgil, D.; Barak, S. Composition, properties and health benefits of indigestible carbohydrate polymers as dietary fiber: A review. Int. J. Biol. Macromol. 2013, 61, 1–6. [Google Scholar] [CrossRef]
- Kurek, M.A.; Karp, S.; Stelmasiak, A.; Pieczykolan, E.; Juszczyk, K.; Rieder, A. Effect of natural flocculants on purity and properties of β-glucan extracted from barley and oat. Carbohydr. Polym. 2018, 188, 60–67. [Google Scholar] [CrossRef]
- Desplaques, S.; Renou, F.; Grisel, M.; Malhiac, C. Impact of chemical composition of xanthan and acacia gums on the emulsification and stability of oil-in-water emulsions. Food Hydrocoll. 2012, 27, 401–410. [Google Scholar] [CrossRef]
- Piwińska, M.; Wyrwisz, J.; Kurek, M.; Wierzbicka, A. Hydration and physical properties of vacuum-dried durum wheat semolina pasta with high-fiber oat powder. LWT-Food Sci. Technol. 2015, 63, 647–653. [Google Scholar] [CrossRef]
- Shantha, N.C.; Decker, E.A. Rapid, sensitive, iron-based spectrophotometric methods for determination of perorlride values of food lipids. J. AOAC Int. 1994, 77, 421–424. [Google Scholar] [PubMed]
- Al-Shannaq, R.; Farid, M.; Al-Muhtaseb, S.; Kurdi, J. Emulsion stability and cross-linking of PMMA microcapsules containing phase change materials. Sol. Energy Mater. Sol. Cells 2015, 132, 311–318. [Google Scholar] [CrossRef]
- Temelli, F. Extraction and Functional Properties of Barley β-Glucan as Affected by Temperature and pH. J. Food Sci. 1997, 62, 1194–1201. [Google Scholar] [CrossRef]
- Böger, B.R.; Georgetti, S.R.; Kurozawa, L.E. Microencapsulation of grapeseed oil by spray drying. Food Sci. Technol. 2018, 38, 263–270. [Google Scholar] [CrossRef]
- Li, J.; Xiong, S.; Wang, F.; Regenstein, J.M.; Liu, R. Optimization of Microencapsulation of Fish Oil with Gum Arabic/Casein/Beta-Cyclodextrin Mixtures by Spray Drying. J. Food Sci. 2015, 80, 1445–1452. [Google Scholar] [CrossRef]
- Ahmad, M.; Ashraf, B.; Gani, A.; Gani, A. Microencapsulation of saffron anthocyanins using β-glucan and β-cyclodextrin: Microencapsule characterization, release behaviour and antioxidant potential during in-vitro digestion. Int. J. Biol. Macromol. 2018, 109, 435–442. [Google Scholar] [CrossRef]
- Fernandes, R.V.D.B.; Borges, S.V.; Botrel, D.A. Gum Arabic/starch/maltodextrin/inulin as wall materials on the microencapsulation of rosemary essential oil. Carbohydr. Polym. 2014, 101, 524–532. [Google Scholar] [CrossRef]
- Park, J.-S.; Lim, Y.-M.; Baik, J.; Jeong, J.-O.; An, S.-J.; Jeong, S.-I.; Gwon, H.-J.; Khil, M.-S. Preparation and evaluation of β-glucan hydrogel prepared by the radiation technique for drug carrier application. Int. J. Biol. Macromol. 2018, 118, 333–339. [Google Scholar] [CrossRef]
- Chew, S.C.; Tan, C.P.; Nyam, K.L. Microencapsulation of refined kenaf (Hibiscus cannabinus L.) seed oil by spray drying using β-cyclodextrin/ / sodium caseinate. J. Food Eng. 2018, 237, 78–85. [Google Scholar] [CrossRef]
- Kurek, M.A.; Moczkowska, M.; Pieczykolan, E.; Sobieralska, M. Barley β-d-glucan–modified starch complex as potential encapsulation agent for fish oil. Int. J. Biol. Macromol. 2018, 120, 596–602. [Google Scholar] [CrossRef]
- Fernandes, R.V.D.B.; Borges, S.V.; Botrel, D.A.; Silva, E.K.; Costa, J.M.G.D.; Queiroz, F. Microencapsulation of rosemary essential oil: Characterization of particles. Dry. Technol. 2013, 31, 1245–1254. [Google Scholar] [CrossRef]
- Jafari, S.M.; Mahdavee Khazaei, K.; Ghorbani, M.; Hemmati Kakhki, A. Application of maltodextrin and gum Arabic in microencapsulation of saffron petal’s anthocyanins and evaluating their storage stability and color. Carbohydr. Polym. 2014, 105, 57–62. [Google Scholar]
- Eidhin, D.N.; Burke, J.; O’Beirne, D. Oxidative Stability of omega-3-rich Camelina Oil and Camelina Oil-based Spread Compared with Plant and Fish Oils and Sunflower Spread. J. Food Sci. 2003, 68, 345–353. [Google Scholar] [CrossRef]
- Charuwat, P.; Boardman, G.; Bott, C.; Novak, J.T. Thermal degradation of long-chain fatty acids. Water Environ. Res. 2018, 90, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, R.V.D.B.; Botrel, D.A.; Silva, E.K.; Borges, S.V.; de Oliveira, C.R.; Yoshida, M.I.; de Andrade Feitosa, J.P.; de Paula, R.C.M. Cashew gum and inulin: New alternative for ginger essential oil microencapsulation. Carbohydr. Polym. 2016, 153, 133–142. [Google Scholar] [CrossRef] [PubMed]

| Emulsion Stability [%] | Emulsion Particle Size [µm] | Microcapsules Density [g/cm3] | Microcapsules Humidity [%] | Microcapsulation Efficiency [%] | L * | a * | b * | Span | Color Bar | |
|---|---|---|---|---|---|---|---|---|---|---|
| Inulin | 75.93 ± 0.21 b | 18.83 ± 0.11 b | 0.367 ± 0.012 a | 2.83 ± 0.12 a | 91.74 ± 0.12 c | 93.657 ± 0.244 d | −1.927 ± 0.074 a | 7.420 ± 0.300 a | 0.972 ± 0.019 b |  |
| Pectin | 84.11 ± 0.19 c | 14.67 ± 0.19 a | 0.517 ± 0.009 b | 3.04 ± 0.17 a | 61.36 ± 0.24 a | 89.177 ± 0.390 c | −1.383 ± 0.047d c | 8.477 ± 0.095 b | 0.806 ± 0.012 a |  |
| β-glucan | 30.00 ± 0.13 a | 44.85 ± 0.21 c | 0.553 ± 0.013 b | 4.97 ± 0.09 c | 79.26 ± 0.11 b | 86.700 ± 0.130 a | −1.860 ± 0.108 a | 9.437 ± 0.534 c | 1.090 ± 0.022 d |  |
| Gum arabic | 75.00 ± 0.11 b | 44.84 ± 0.12 c | 0.554 ± 0.003 b | 3.54 ± 0.11 b | 83.93 ± 0.09 b | 87.923 ± 0.191 b | −1.663 ± 0.050 b | 7.447 ± 0.172 a | 1.012 ± 0.029 c |  |
| Fatty Acids | Inulin | Pectin | β-Glucan | Gum Arabic | Oil |
|---|---|---|---|---|---|
| Percentage of Fatty Acid [%] | |||||
| Palmitc (C16:0) | 5.34 ± 0.06 c | 5.28 ± 0.02 ab | 5.29 ± 0.01 bc | 5.28 ± 0.02 ab | 5.23 ± 0.03 a |
| Stearic (C18:0) | 2.38 ± 0.02 b | 2.40 ± 0.01 b | 2.38 ± 0.06 b | 2.39 ± 0.07 b | 2.33 ± 0.01 a |
| Oleic (C18:1 n9c) | 16.59 ± 0.03 d | 16.02 ± 0.01 bc | 16.15 ± 0.01 c | 15.84 ± 0.03 ab | 15.58 ± 0.01 a |
| α-linolenic (C18:3 n3) | 34.05 ± 0.03 a | 34.17 ± 0.09 a | 34.17 ± 0.07 a | 34.25 ± 0.06 a | 34.73 ± 0.01 b |
| Linolenic (C18:2 n6c) | 17.82 ± 0.07 b | 17.92 ± 0.06 a | 17.95 ± 0.02 a | 17.96 ± 0.01 a | 18.07 ± 0.02 c |
| Eicosanoic (C20:1 n9) | 14.33 ± 0.01 a | 14.54 ± 0.01 c | 14.36 ± 0.08 ab | 14.49 ± 0.02 bc | 14.31 ± 0.01 a |
| Erucic (C22:1 n9) | 3.16 ± 0.05 abc | 3.21 ± 0.04 c | 3.15 ± 0.02 ab | 3.18 ± 0.08 bc | 3.12 ± 0.01 a |
| Nervonic (C24:1 n9) | 1.03 ± 0.02 a | 1.09 ± 0.01 a | 1.07 ± 0.02 a | 0.54 ± 0.01 b | 1.21 ± 0. 04 c |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kanclerz, A.; Drozińska, E.; Kurek, M.A. Microencapsulation of Camelina sativa Oil Using Selected Soluble Fractions of Dietary Fiber as the Wall Material. Foods 2019, 8, 681. https://doi.org/10.3390/foods8120681
Kanclerz A, Drozińska E, Kurek MA. Microencapsulation of Camelina sativa Oil Using Selected Soluble Fractions of Dietary Fiber as the Wall Material. Foods. 2019; 8(12):681. https://doi.org/10.3390/foods8120681
Chicago/Turabian StyleKanclerz, Aleksandra, Ewelina Drozińska, and Marcin Andrzej Kurek. 2019. "Microencapsulation of Camelina sativa Oil Using Selected Soluble Fractions of Dietary Fiber as the Wall Material" Foods 8, no. 12: 681. https://doi.org/10.3390/foods8120681
APA StyleKanclerz, A., Drozińska, E., & Kurek, M. A. (2019). Microencapsulation of Camelina sativa Oil Using Selected Soluble Fractions of Dietary Fiber as the Wall Material. Foods, 8(12), 681. https://doi.org/10.3390/foods8120681






