Narrow-Leafed Lupin Main Allergen β-Conglutin (Lup an 1) Detection and Quantification Assessment in Natural and Processed Foods
Abstract
1. Introduction
2. Material and Methods
2.1. Bioinformatics Analysis of β-Conglutin Protein Sequences
2.2. Analysis of the Antigenicity of β-Conglutin Proteins
2.3. β-Conglutin Proteins Structure Modelling
2.4. Construction of the Expression Plasmid
2.5. Overexpression of Conglutin β1
2.6. Purification of the Recombinant Conglutin β1 Protein
2.7. Antibody Production Against β-Conglutin
2.8. Food and Biological Samples Used for Lupin Allergen Detection and Quantification
2.9. Proteins Extraction
2.10. Gel Electrophoresis and Western Blots
2.11. ELISA Test for the Detection and Quantification of β-Conglutin Allergen Proteins in Lupin-Containing Food
3. Results and Discussion
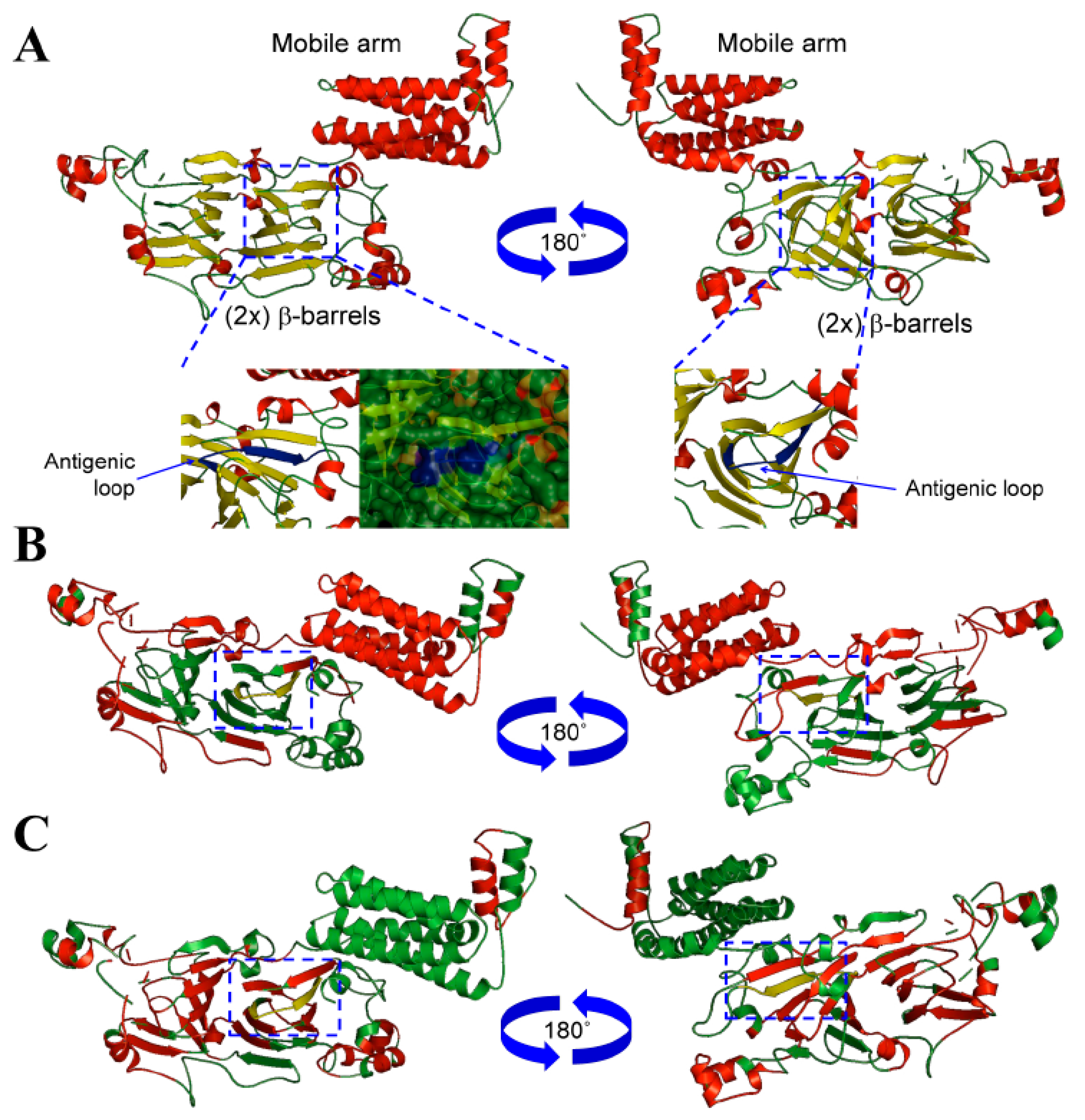
3.1. Conglutin β1 Protein Structure Modelling and Antigenicity Assessment
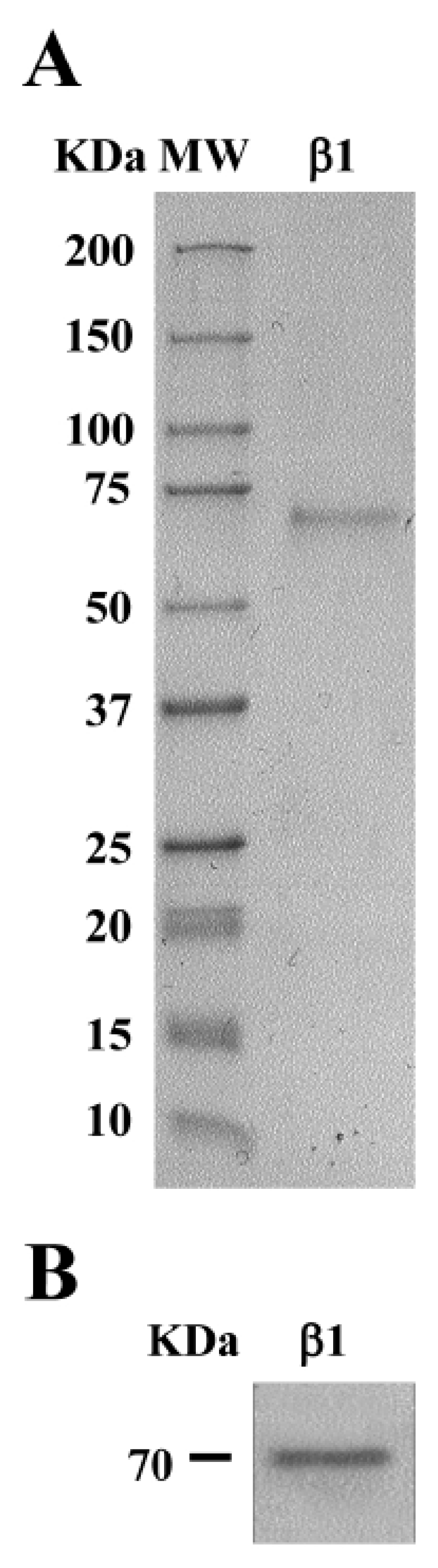
3.2. Purification and Reactivity of Conglutin β1 Protein
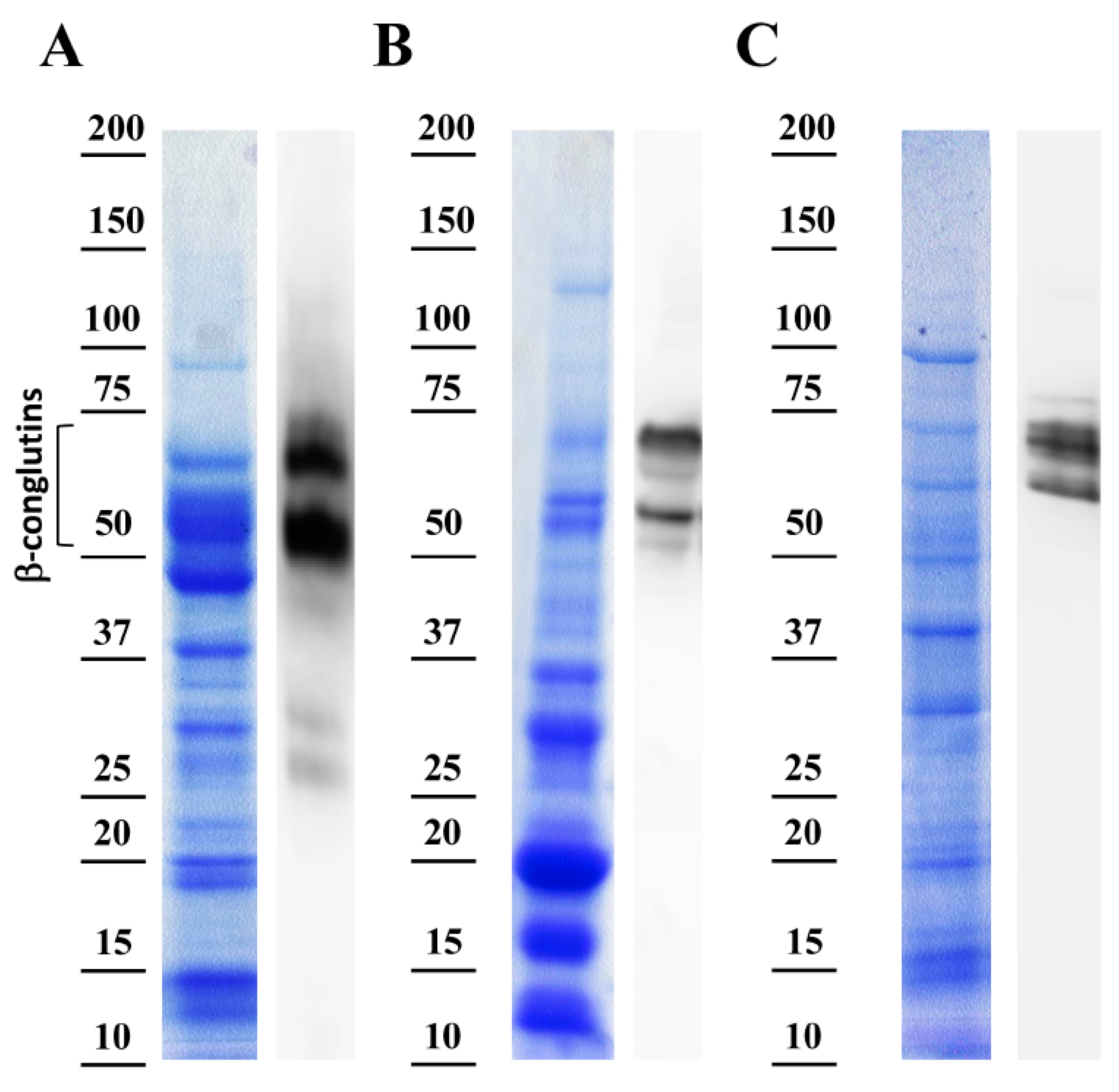
3.3. β-Conglutin Proteins Main Allergen Detection Capability in Lupin—Derived Products
- (1)
- The protein extraction protocol in the current study is highly specific for the extraction of the vicilin family of proteins (β-conglutins), based on the presence of NaCl (0.25M) in the extraction buffer [33]. Previous studies made protein extractions using general protein extraction buffers [4,45,47], or using alternative methods from commercial kits [48] currently no longer available (Abnova, http://www.abnova.com/products/products_detail.asp?catalog_id=KA3310), displaying very limited or no information about: (i) The antibody design; (ii) antibody production and use in the ELISA detection method; (iii) the limited information about protocol for total proteins extraction, which may not be specific for β-conglutin extraction; (iv) no information about lupin species used [46,48] to obtain this protein extract. The last two are the main factors with high impact in the protein extract characteristics, such as a low amount or not of Lup an 1 content. These disadvantages may result in the increase of the number of false positives as a result of the detection of non-allergen proteins from a low specific antibody, or using non-appropriate protein extracts.
- (2)
- A second advantage of the current method compared with previous ones and commercial kits is the design and the production of the antibody (anti-IgG β-conglutin proteins). In the current study, the experimental animal was immunized with a synthetic peptide commonly shared by the seven NLL β-conglutin protein isoforms. This synthetic peptide constitutes a highly antigenic epitope in these proteins probed in the current study, while also exhibits a high specificity to detect the lupin main allergen Lup an 1 in the most frequently used lupin species. On the contrary, previous methods [4,45,46,47] have used the whole crude protein extract from lupin flour to immunize the experimental animal and obtain the antibody. The method to produce this antibody makes this antibody non-specific, and detecting a wide range of proteins, including many non-allergenic proteins, may lead to false positive detections. The detection of false positives might be also enhanced due to the implementation of a buffer inappropriate for vicilin protein extraction from lupin-derived foodstuffs.
- (3)
- The current method exhibited another advantage which was the type of standards (Figure S3) that was made for a specific quantification of Lup an 1. Previous methodological developments of standards for the quantification were based on lupin flour total protein extract [4,45,46,47]. This may induce variable immunization for a complex mix of proteins (allergenic and non-allergenic proteins) from the crude extract leading to an excess or lack of reactivity in the standard samples because a variable representation or content of Lup an 1 (over or under representation, depending on the extraction method).
3.4. Specificity of the Anti-β-Conglutin Antibody Tested in Processed Foodstuffs and Potential Cross-Reactive
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kurlovich, B.S.; Stankevich, A.K.; Stepanova, S.I. The review of the genus Lupinus L. In Lupins (Geography, Classification, Genetic Resources and Breeding); Kurlovich, B.S., Ed.; OY International North Express: St. Petersburg, Russia; Pellosniemi, Finland, 2002; Chapter 2; pp. 11–38. [Google Scholar]
- Uauy, R.; Gattas, V.; Yáñez, E. Sweet lupins in human nutrition. World Rev. Nutr. Diet. 1995, 77, 75–88. [Google Scholar]
- Frick, K.M.; Kamphuis, L.G.; Siddique, K.H.M.; Singh, K.B.; Foley, R.C. Quinolizidine alkaloid biosynthesis in lupins and prospects for grain quality improvement. Front. Plant Sci. 2017, 8, 87. [Google Scholar] [CrossRef]
- Ecker, C.; Cichna-Markl, M. Development and validation of a sandwich ELISA for the determination of potentially allergenic lupine in food. Food. Chem. 2012, 130, 759–766. [Google Scholar] [CrossRef]
- Jappe, U.; Vieths, S. Lupine, a source of new as well as hidden food allergens. Mol. Nutr. Food Res. 2010, 54, 113–126. [Google Scholar] [CrossRef]
- Delgado-Andrade, C.; Olias, R.; Jimenez-Lopez, J.C.; Clemente, A. Nutritional and beneficial effects of grain legumes on human health. ARBOR Ciencia Pensamiento y Cultura 2016, 192, 779. [Google Scholar]
- Lima-Cabello, E.; Alche, V.; Foley, R.C.; Andrikopoulos, S.; Morahan, G.; Singh, K.B.; Alche, J.D.; Jimenez-Lopez, J.C. Narrow-leafed lupin (Lupinus angustifolius L.) β-conglutin proteins modulate the insulin signaling pathway as potential type 2 diabetes treatment and inflammatory-related disease amelioration. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef]
- Lima-Cabello, E.; Morales-Santana, S.; Foley, R.C.; Melser, S.; Alche, V.; Siddique, K.H.M.; Singh, K.B.; Alche, J.D.; Jimenez-Lopez, J.C. Ex vivo and in vitro assessment of anti-inflammatory activity of seed β-conglutin proteins from Lupinus angustifolius. J. Funct. Foods 2018, 40, 510–519. [Google Scholar] [CrossRef]
- Lima-Cabello, E.; Morales-Santana, S.; Leon, J.; Alche, V.; Clemente, A.; Alche, J.D.; Jimenez-Lopez, J.C. Narrow-leafed lupin (Lupinus angustifolius L.) seed beta-conglutins reverse back the induced insulin resistance in pancreatic cells. Food Funct. 2018, 9, 5176–5188. [Google Scholar] [CrossRef]
- Lima-Cabello, E.; Robles-Bolivar, P.; Alche, J.D.; Jimenez-Lopez, J.C. Narrow-leafed lupin Beta-conglutin proteins epitopes identification and molecular features analysis involved in cross-allergenicity to peanut and other legumes. Genom. Comput. Biol. 2016, 2, e29. [Google Scholar] [CrossRef]
- Foley, R.C.; Jimenez-Lopez, J.C.; Kamphuis, L.G.; Hane, J.K.; Melser, S.; Singh, K.B. Analysis of conglutin seed storage proteins across lupin species using transcriptomic, protein and comparative genomic approaches. BMC Plant Biol. 2015, 15, 106. [Google Scholar] [CrossRef]
- Cabello-Hurtado, F.; Keller, J.; Ley, J.; Sanchez-Lucas, R.; Jorrín-Novo, J.V.; Aïnouche, A. Proteomics for exploiting diversity of lupin seed storage proteins and their use as nutraceuticals for health and welfare. J. Proteom. 2016, 143, 57–68. [Google Scholar] [CrossRef]
- De Jong, N.W.; Van Maaren, M.S.; Vlieg-Boersta, B.J.; Dubois, A.E.J.; De Groot, H.; van Wijk, R.G. Sensitization to lupine flour: Is it clinically relevant? Clin. Exp. Allery 2010, 40, 1571–1577. [Google Scholar] [CrossRef]
- Ronchi, A.; Duranti, M.; Scarafoni, A. A real-time PCR method for the detection and quantification of lupin flour in wheat flour-based matrices. Food Chem. 2009, 115, 1088–1093. [Google Scholar]
- Sirtori, E.; Resta, D.; Brambilla, F.; Zacherl, C.; Arnoldi, A. The effects ofvarious processing conditions on a protein isolate from Lupinus angustifolius. Food Chem. 2010, 120, 496–504. [Google Scholar] [CrossRef]
- Sanz, M.L.; De Las Marinas, M.D.; Fernández, J.; Gamboa, P.M. Lupin allergy: A hidden killer in the home. Clin. Exp. Allery 2010, 40, 1461–1466. [Google Scholar] [CrossRef]
- Peeters, K.A.; Koppelman, S.J.; Penninks, A.H.; Lebens, A.; Bruijnzeel-Koomen, C.A.; Hefle, S.L.; Taylor, S.L.; van Hoffen, E.; Knulst, A.C. Clinical relevance of sensitization to lupine in peanut-sensitized adults. Allergy 2009, 64, 549–555. [Google Scholar] [CrossRef]
- Campbell, C.P.; Yates, D.H. Lupin allergy: A hidden killer at home, a menace at work; occupational disease due to lupin allergy. Clin. Exp. Allery 2010, 40, 1467–1472. [Google Scholar] [CrossRef]
- Kaw, C.H.; Hefle, S.L.; Taylor, S.L. Sandwich Enzyme-Linked Immunosorbent Assay (ELISA) for the Detection of Lupine Residues in Foods. Food Sci. 2008, 73, T135–T140. [Google Scholar] [CrossRef]
- Hieta, N.; Hasan, T.; Makinen-Kiljunen, S.; Lammintausta, K. Lupin allergy and lupin sensitization among patients with suspected food allergy. Ann. Allergy Asthma Immunol. 2009, 103, 233–237. [Google Scholar] [CrossRef]
- Jimenez-Lopez, J.C.; Foley, R.C.; Brear, E.; Clarke, V.C.; Lima-Cabello, E.; Florido, J.F.; Singh, K.B.; Alché, J.D.; Smith, P.M.C. Characterization of narrow-leaf lupin (Lupinus angustifolius L.) recombinant major allergen IgE-binding proteins and the natural β-conglutin counterparts in sweet lupin seed species. Food Chem. 2018, 44, 60–70. [Google Scholar] [CrossRef]
- Lima-Cabello, E.; Berral-Hens, J.M.; Florido, J.F.; Alche, J.D.; Jimenez-Lopez, J.C. The function of conglutin proteins from the “sweet lupin” group in food allergy. In Legumes for Global Food Security, 1st ed.; Nova Science Publishers, Inc.: New York, NY, USA, 2017; Chapter 10; pp. 249–275. [Google Scholar]
- Goggin, D.E.; Mir, G.; Smith, W.B.; Stuckey, M.; Smith, P.M.C. Proteomic analysis of lupin seed proteins to identify conglutin β as an allergen, Lup an 1. J. Agric. Food Chem. 2008, 56, 6370–6377. [Google Scholar] [CrossRef]
- Jimenez-Lopez, J.C.; Gachomo, E.W.; Sharma, S.; Kotchoni, S.O. Genetic sequencing and next-generation sequence data analysis: A comprehensive compilation of bioinformatics tools and databases. Am. J. Mol. Biol. 2013, 3, 115–130. [Google Scholar] [CrossRef][Green Version]
- Kyte, J.; Doolittle, R.F. A simple method for displaying the hydropathic character of a protein. J. Mol. Biol. 1982, 157, 105–132. [Google Scholar] [CrossRef]
- Emini, E.A.; Hughes, J.V.; Perlow, D.S.; Boger, J. Induction of hepatitis A virus-neutralizing antibody by a virus-specific synthetic peptide. J. Virol. 1985, 55, 836–839. [Google Scholar]
- Karplus, P.A.; Schulz, G.E. Prediction of chain flexibility in proteins. Naturwissenschaften 1985, 72, 212–213. [Google Scholar] [CrossRef]
- Hopp, T.P.; Woods, K.R. Prediction of protein antigenicdeterminants from amino acid sequences. Proc. Natl. Acad. Sci. USA 1981, 78, 3824–3828. [Google Scholar] [CrossRef]
- Welling, G.W.; Weijer, W.J.; van der Zee, R.; Welling-Wester, S. Prediction of sequential antigenic regions in proteins. FEBS Lett. 1985, 188, 215–218. [Google Scholar] [CrossRef]
- Parker, K.C.; Bednarek, M.A.; Coligan, J.E. Scheme for rankingpotential HLA-A2 binding peptides based on independent binding ofindividual peptide side-chains. J. Immunol. 1994, 152, 163–175. [Google Scholar]
- Kolaskar, A.S.; Tongaonkar, P.C. A semi-empirical method forprediction of antigenic determinants on protein antigens. FEBS Lett. 1990, 276, 172–174. [Google Scholar] [CrossRef]
- Fox, B.G.; Blommel, P.G. Autoinduction of Protein Expression. Curr. Protoc. Protein Sci. 2009, 56, 5–23. [Google Scholar]
- Nadal, P.; Canela, N.; Katakis, I.; O’Sullivan, C.K. Extraction, Isolation, and Characterization of Globulin Proteins from Lupinus albus. J. Agric. Food Chem. 2011, 59, 2752–2758. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, W.; Negre, N.N.; White, K.P.; Li, C.; Shah, P.K. Determinants of antigenicity and specificity in immune response for protein sequences. BMC Bioinform. 2011, 12, 251. [Google Scholar] [CrossRef]
- Palladino, C.; Breiteneder, H. Peanut allergens. Mol. Immunol. 2018, 100, 58–70. [Google Scholar] [CrossRef]
- Jimenez-Lopez, J.C.; Lima-Cabello, E.; Melser, S.; Foley, R.C.; Singh, K.B.; Alché, J.D. Lupin allergy: Uncovering structural features and epitopes of β-conglutin proteins in Lupinus angustifolius L. with a focus on cross-allergenic reactivity to peanut and other legumes. Bioinform. Biomed. Eng. 2015, 9043, 96–107. [Google Scholar]
- Jimenez-Lopez, J.C.; Robles-Bolivar, P.; Lopez-Valverde, F.J.; Lima-Cabello, E.; Kotchoni, S.O.; Alché, J.D. Ole e 13 is the unique food allergen in olive: Structure-functional, substrates docking, and molecular allergenicity comparative analysis. J. Mol. Graph. Model. 2016, 66, 26–40. [Google Scholar] [CrossRef]
- Jimenez-Lopez, J.C.; Kotchoni, S.O.; Hernandez-Soriano, M.C.; Gachomo, E.W.; Alché, J.D. Structural functionality, catalytic mechanism modeling and molecular allergenicity of phenylcoumaran benzylic ether reductase, an olive pollen (Ole e 12) allergen. J. Comput. Aided Mol. Des. 2013, 27, 873–895. [Google Scholar] [CrossRef]
- Jimenez-Lopez, J.C.; Kotchoni, S.O.; Rodríguez-García, M.I.; Alché, J.D. Structure and functional features of olive pollen pectin methylesterase using homology modeling and molecular docking methods. J. Mol. Model. 2012, 18, 4965–4984. [Google Scholar] [CrossRef]
- Jimenez-Lopez, J.C.; Rodríguez-García, M.I.; Alché, J.D. Analysis of the effects of polymorphism on pollen profilin structural functionality and the generation of conformational, T- and B-cell epitopes. PLoS ONE 2013, 17, e76066. [Google Scholar] [CrossRef]
- Jimenez-Lopez, J.C.; Gachomo, E.W.; Ariyo, O.A.; Baba-Moussa, L.; Kotchoni, S.O. Specific conformational epitope features of pathogenesis-related proteins mediating cross-reactivity between pollen and food allergens. Mol. Biol. Rep. 2012, 39, 123–130. [Google Scholar] [CrossRef]
- Jimenez-Lopez, J.C.; Melser, S.; DeBoer, K.; Thatcher, L.F.; Kamphuis, L.G.; Foley, R.C.; Singh, K.B. Narrow-Leafed Lupin (Lupinus angustifolius) β1- and β6-Conglutin Proteins Exhibit Antifungal Activity, Protecting Plants against Necrotrophic Pathogen Induced Damage from Sclerotinia sclerotiorum and Phytophthora nicotianae. Front. Plant Sci. 2016, 9, 1856. [Google Scholar] [CrossRef]
- Magni, C.; Scarafoni, A.; Herndl, A.; Sessa, F.; Prinsi, B.; Espen, L.; Duranti, M. Combined 2D electrophoretic approaches for the study of white lupin mature seed storage proteome. Phytochemistry 2007, 68, 997–1007. [Google Scholar] [CrossRef] [PubMed]
- Duranti, M.; Sessa, F.; Carpen, A. Identification, purification and properties of the precursor of conglutin β, the 7S storage globulin of Lupinus albus L. seeds. J. Exp. Bot. 1992, 43, 1373–1378. [Google Scholar] [CrossRef]
- Holden, L.; Faeste, C.K.; Egaas, E. Quantitative sandwich ELISA for the determination of lupine (Lupinus spp.) in foods. J. Agric. Food Chem. 2005, 53, 5866–5871. [Google Scholar] [CrossRef]
- Taylor, S.L.; Nordlee, J.A.; Niemann, L.M.; Lambrecht, D.M. Allergen immunoassays–considerations for use of naturally incurred standards. Anal. Bioanal. Chem. 2009, 395, 83–92. [Google Scholar]
- Ecker, C.; Ertla, A.; Pulverera, W.; PalSzekely, A.N.; Petraschc, A.; Linsberger-Martin, G.; Cichna-Markl, M. Validation and comparison of a sandwich ELISA, two competitive ELISAs and a real-time PCR method for the detection of lupine in food. Food Chem. 2013, 141, 407–418. [Google Scholar] [CrossRef]
- Koeberl, M.; Sharp, M.F.; Tian, R.; Buddhadasa, S.; Clarke, D.; Roberts, J. Lupine allergen detecting capability and cross-reactivity of related legumes by ELISA. Food Chem. 2018, 256, 105–112. [Google Scholar] [CrossRef]
- European Parliament Regulation (EU). No 1169/2011 of the European Parliament and of the Council of 25 October 2011 on the Provision of Food Information to Consumers, Amending Regulations (EC) No 1924/2006 and (EC) No 1925/2006 of the European Parliament and of the Council, and Repealing Commission Directive 87/250/EEC, Council Directive 90/496/EEC, Commission Directive 1999/10/EC, Directive 2000/13/EC of the European Parliament and of the Council, Commission Directives 2002/67/EC and 2008/5/EC and Commission Regulation (EC) No 608/2004 Text with EEA Relevance L304/18 C.F.R.; EU: Brussel, Belgium, 2011. [Google Scholar]
- EFSA. Directive 2007/68/EC Amending Annex IIIa of Directive 2000/13/EC of the European Parliament and of the Council (2007). Off. J. Eur. Union 2007, 310, 11–14. [Google Scholar]
- Pedersen, M.H.; Holzhauser, T.; Bisson, C.; Conti, A.; Jensen, L.B.; Skov, P.S.; Bindslev-Jensen, C.; Brinch, D.S.; Poulsen, L.K. Soybean allergen detection methods—A comparison study. Mol. Nutr. Food Res. 2008, 52, 1486–1496. [Google Scholar] [CrossRef]
- Taylor, S.L.; Baumert, J.L.; Houben, G.F.; Crevel, R.W.R.; Brooke-Taylor, S.; Allen, K.J.; The Allergen Bureau of Australia & New Zealand; Houben, G. Establishment of reference doses for residues of allergenic foods: Report of the VITAL expert panel. Food Chem. Toxicol. 2014, 63, 9–17. [Google Scholar]
- Alvarez-Alvarez, J.; Guillamon, E.; Crespo, J.F.; Cuadrado, C.; Burbano, C.; Rodriguez, J.; Muzquiz, M. Effects of extrusion, boiling, autoclaving, and microwave heating on lupine allergenicity. J. Agric. Food Chem. 2005, 53, 1294–1298. [Google Scholar] [CrossRef]
- Gupta, R.K.; Gupta, K.; Sharma, A.; Das, M.; Ansari, I.A.; Dwivedi, P.D. Health Risks and Benefits of Chickpea (Cicerarietinum) Consumption. J. Agric. Food Chem. 2017, 65, 6–22. [Google Scholar] [CrossRef]
- Verma, A.K.; Kumar, S.; Das, M.; Dwivedi, P.D. A Comprehensive Review of Legume Allergy. Clin. Rev. Allergy Immunol. 2013, 45, 30–46. [Google Scholar] [CrossRef] [PubMed]
- Török, K.; Hajas, L.; Horváth, V.; Schall, E.; Bugyi, Z.; Kemény, S.; Tömösközi, S. Identification of the factors affecting the analytical results offood allergen ELISA methods. Eur. Food Res. Technol. 2015, 241, 127–136. [Google Scholar] [CrossRef]
- Peeters, K.A.; Nordlee, J.A.; Penninks, A.H.; Chen, L.; Goodman, R.E.; Bruijnzeel-Koomen, C.A.; Knulst, A.C. Lupine allergy: Not simply cross-reactivity with peanut or soy. J. Allery Clin. Immunol. 2007, 120, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Berkner, H.; Neudecker, P.; Mittag, D.; Ballmer-Weber, B.K.; Schweimer, K.; Vieths, S.; Rosch, P. Cross-reactivity of pollen and food allergens: Soybean Gly m 4 is a member of the Bet v 1 superfamily and closely resembles yellow lupine proteins. Biosci. Rep. 2009, 29, 183–192. [Google Scholar] [CrossRef]
- Shaw, J.; Roberts, G.; Grimshaw, K.; White, S.; Hourihane, J. Lupin allergy in peanut-allergic children and teenagers. Allergy 2008, 63, 370–373. [Google Scholar] [CrossRef]
- Hoffmann, B.; Münch, S.; Schwägele, F.; Neusüß, C.; Jira, W. A sensitive HPLC-MS/MS screening method for the simultaneous detection of lupine, pea, and soy proteins in meat products. Food Control 2017, 71, 200–209. [Google Scholar] [CrossRef]
- Holden, L.; Moen, L.H.; Sletten, G.B.; Dooper, M.M. Novel polyclonal-monoclonal-based ELISA utilized to examine lupine (Lupinus species) content in food products. J. Agric. Food Chem. 2007, 55, 2536–2542. [Google Scholar] [CrossRef]
| Natural/Food Samples | Absorbance (450 nm) 1 | Allergen β-Conglutin Proteins Quantity (ng) (Diluted Samples) | Allergen β-Conglutin Proteins Final Quantity (ng) | Allergen β-Conglutin Proteins Quantification (ppm) |
|---|---|---|---|---|
| 1 | 0.0390 | 2.0313 | 8.1250 ± 0.1701 | 0.0406 ± 0.0009 |
| 0.0380 | 1.9792 | |||
| 0.0400 | 2.0833 | |||
| 2 | 0 | 0 | 0 | 0 |
| 0 | 0 | |||
| 0 | 0 | |||
| 3 | 1.8430 | 95.9896 | 391.8753 ± 11.1958 | 1.9594 ± 0.0560 |
| 1.8430 | 95.9896 | |||
| 1.9570 | 101.9271 | |||
| 4 | 0 | 0 | 0 | 0 |
| 0 | 0 | |||
| 0 | 0 | |||
| 5 | 0.3890 | 20.2604 | 80.9028 ± 0.3541 | 0.4045 ± 0.0018 |
| 0.3860 | 20.1042 | |||
| 0.3900 | 20.3125 | |||
| 6 | 1.3430 | 69.9479 | 299.3749 ± 19.7757 | 1.4969 ± 0.0989 |
| 1.5670 | 81.6145 | |||
| 1.4010 | 72.9688 | |||
| 7 | 2.2890 | 119.2188 | 457.8474 ± 15.8272 | 2.2892 ± 0.0791 |
| 2.1030 | 109.5313 | |||
| 2.2010 | 114.6354 | |||
| 8 | 2.8940 | 150.7292 | 574.7918 ± 19.8876 | 2.8740 ± 0.0994 |
| 2.6910 | 140.1563 | |||
| 2.6920 | 140.2083 | |||
| 9 | 0.0190 | 0.9896 | 3.8890 ± 0.2599 | 0.0194 ± 0.0013 |
| 0.0200 | 1.0417 | |||
| 0.0170 | 0.8854 | |||
| 10 | 1.0340 | 53.8542 | 216.1112 ± 0.5197 | 1.0806 ± 0.0026 |
| 1.0380 | 54.0625 | |||
| 1.0400 | 54.1667 | |||
| 11 | 0 | 0 | 0 | 0 |
| 0 | 0 | |||
| 0 | 0 | |||
| 12 | 0.7840 | 40.8334 | 152.9862 ± 7.3353 | 0.7649 ± 0.0367 |
| 0.7060 | 36.7708 | |||
| 0.7130 | 37.1354 | |||
| 13 | 0.6430 | 33.4896 | 129.1667 ± 3.6839 | 0.6458 ± 0.0184 |
| 0.6000 | 31.2500 | |||
| 0.6170 | 32.1354 | |||
| 14 | 1.4020 | 73.2105 | 293.5833 ±11.0853 | 1.4252 ± 0.0554 |
| 1.5140 | 77.5442 | |||
| 1.3860 | 70.8534 | |||
| 15 | 0.2840 | 14.7917 | 59.8660 ± 0.8389 | 0.2993 ± 0.0042 |
| 0.2930 | 15.2604 | |||
| 0.2850 | 14.8438 | |||
| 16 | 0 | 0 | 0 | 0 |
| 0 | 0 | |||
| 0 | 0 | |||
| 17 | 0 | 0 | 0 | 0 |
| 0 | 0 | |||
| 0 | 0 | |||
| 18 | 0 | 0 | 0 | 0 |
| 0 | 0 | |||
| 0 | 0 | |||
| 19 | 0 | 0 | 0 | 0 |
| 0 | 0 | |||
| 0 | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lima-Cabello, E.; Alché, J.D.; Jimenez-Lopez, J.C. Narrow-Leafed Lupin Main Allergen β-Conglutin (Lup an 1) Detection and Quantification Assessment in Natural and Processed Foods. Foods 2019, 8, 513. https://doi.org/10.3390/foods8100513
Lima-Cabello E, Alché JD, Jimenez-Lopez JC. Narrow-Leafed Lupin Main Allergen β-Conglutin (Lup an 1) Detection and Quantification Assessment in Natural and Processed Foods. Foods. 2019; 8(10):513. https://doi.org/10.3390/foods8100513
Chicago/Turabian StyleLima-Cabello, Elena, Juan D. Alché, and Jose C. Jimenez-Lopez. 2019. "Narrow-Leafed Lupin Main Allergen β-Conglutin (Lup an 1) Detection and Quantification Assessment in Natural and Processed Foods" Foods 8, no. 10: 513. https://doi.org/10.3390/foods8100513
APA StyleLima-Cabello, E., Alché, J. D., & Jimenez-Lopez, J. C. (2019). Narrow-Leafed Lupin Main Allergen β-Conglutin (Lup an 1) Detection and Quantification Assessment in Natural and Processed Foods. Foods, 8(10), 513. https://doi.org/10.3390/foods8100513







