Butyrylation of Maize and Potato Starches and Characterization of the Products by Nuclear Magnetic Resonance and In Vitro Fermentation
Abstract
1. Introduction
2. Materials and Methods
2.1. Starches
2.2. Organocatalytic Butyrylation of Starches
2.3. DS Determined by Heterogeneous Saponification and Back Titration
2.4. Characterization by Solid-State 13C MAS NMR Spectroscopy
2.5. In Vitro Fermentation
2.6. Statistical Analysis
3. Results
3.1. Starch Composition
3.2. Esterification of Starches
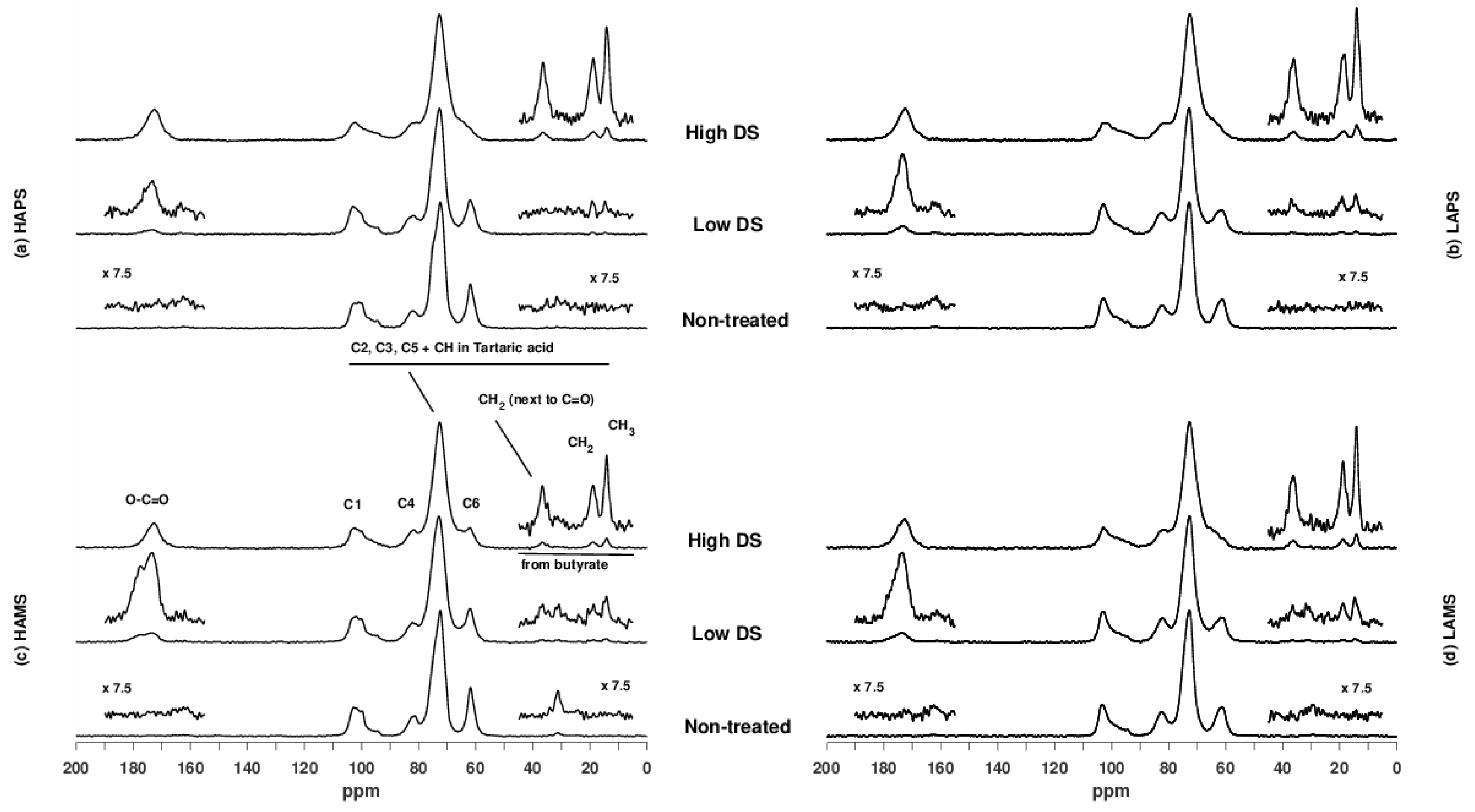
3.3. Characterization of Starches by NMR Spectroscopy
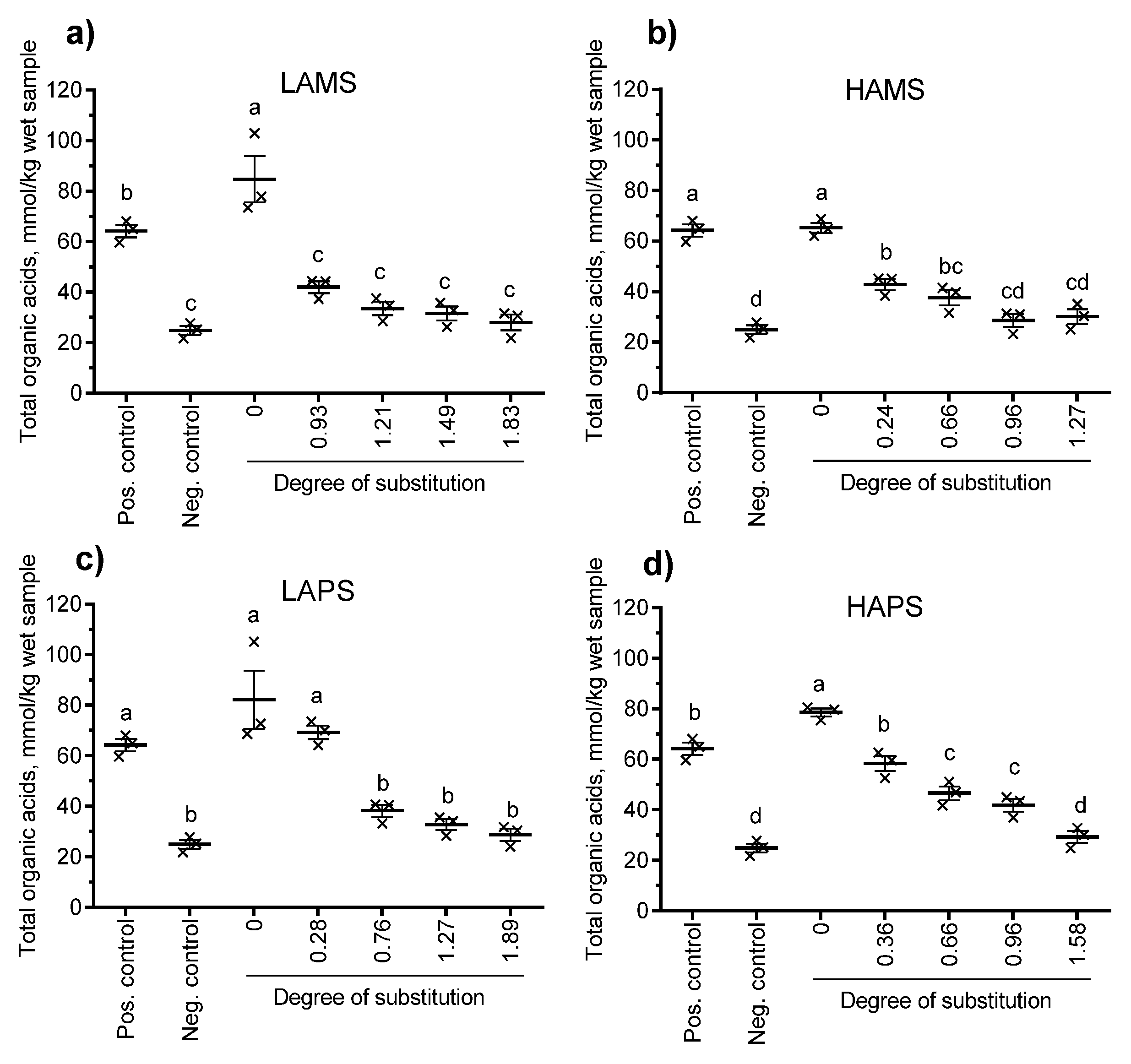
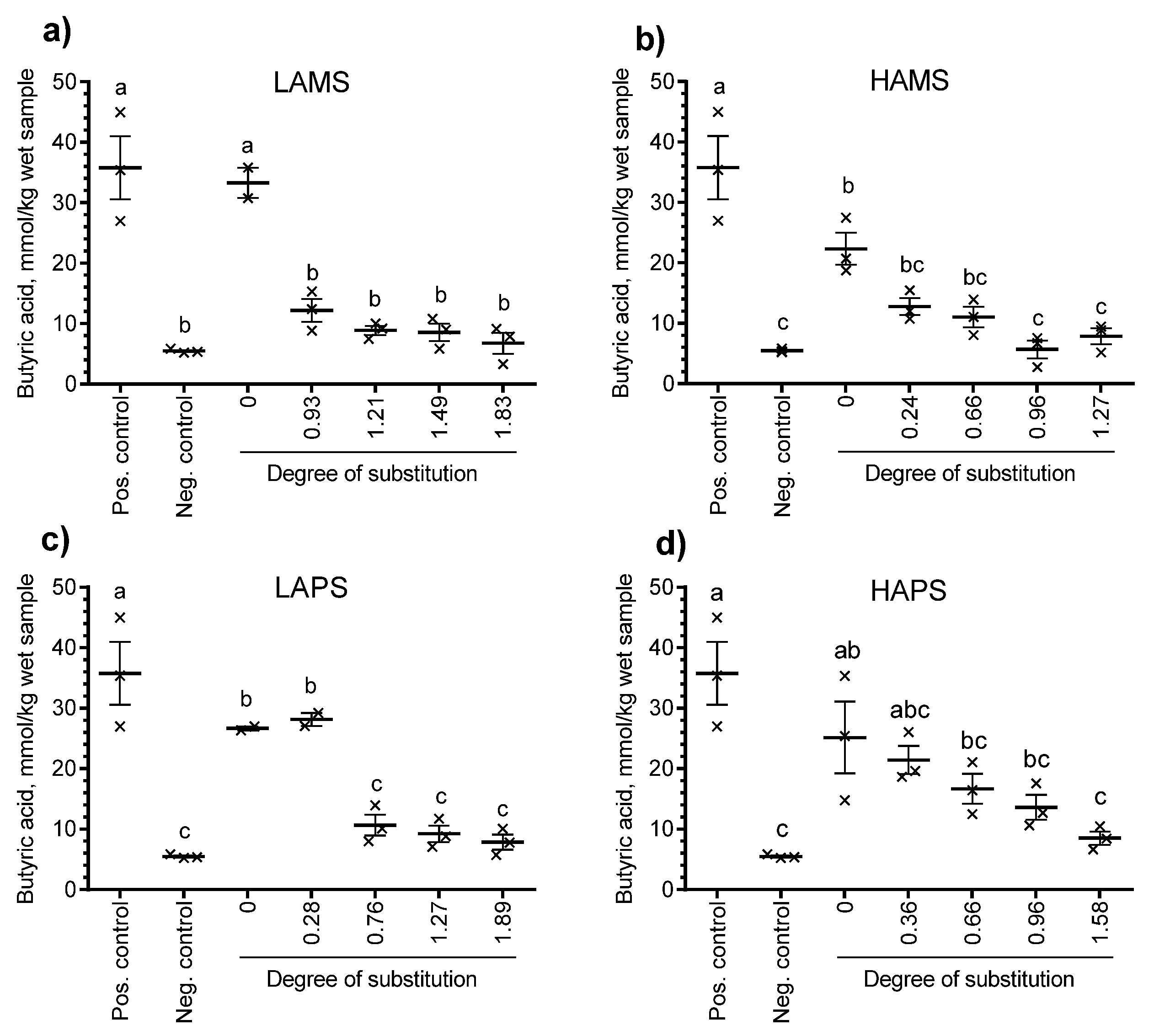
3.4. In Vitro Fermentation of Esterified Starches
4. Discussion
5. Conclusions
Author Contributions
Founding
Acknowledgments
Conflicts of Interest
References
- Van der Beek, C.M.; Dejong, C.H.C.; Troost, F.J.; Masclee, A.A.M.; Lenaerts, K. Role of short-chain fatty acids in colonic inflammation, carcinogenesis, and mucosal protection and healing. Nutr. Rev. 2017, 75, 286–305. [Google Scholar] [CrossRef] [PubMed]
- Guilloteau, P.; Martin, L.; Eeckhaut, V.; Ducatelle, R.; Zabielski, R.; Van Immerseel, F. From the gut to the peripheral tissues: The multiple effects of butyrate. Nutr. Res. Rev. 2010, 23, 366–384. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.M.; de Souza, R.; Kendall, C.W.; Emam, A.; Jenkins, D.J. Colonic health: Fermentation and short chain fatty acids. J. Clin. Gastroenterol. 2006, 40, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Fung, K.Y.; Cosgrove, L.; Lockett, T.; Head, R.; Topping, D.L. A review of the potential mechanisms for the lowering of colorectal oncogenesis by butyrate. Br. J. Nutr. 2012, 108, 820–831. [Google Scholar] [CrossRef] [PubMed]
- Russo, I.; Luciani, A.; De, C.P.; Troncone, E.; Ciacci, C. Butyrate attenuates lipopolysaccharide-induced inflammation in intestinal cells and crohn’s mucosa through modulation of antioxidant defense machinery. PLoS ONE 2012, 7, e32841. [Google Scholar] [CrossRef] [PubMed]
- Correa-Oliveira, R.; Fachi, J.L.; Vieira, A.; Sato, F.T.; Vinolo, M.A. Regulation of immune cell function by short-chain fatty acids. Clin. Trans. Immunol. 2016, 5, e73. [Google Scholar] [CrossRef] [PubMed]
- Canani, R.B.; Di Costanzo, M.; Leone, L. The epigenetic effects of butyrate: Potential therapeutic implications for clinical practice. Clin. Epigen. 2012, 4, 4. [Google Scholar] [CrossRef] [PubMed]
- Belobrajdic, D.P.; Bird, A.R.; Conlon, M.A.; Williams, B.A.; Kang, S.; McSweeney, C.S.; Zhang, D.; Bryden, W.L.; Gidley, M.J.; Topping, D.L. An arabinoxylan-rich fraction from wheat enhances caecal fermentation and protects colonocyte DNA against diet-induced damage in pigs. Br. J. Nutr. 2012, 107, 1274–1282. [Google Scholar] [CrossRef] [PubMed]
- Annison, G.; Topping, D.L. Nutritional role of resistant starch: Chemical structure vs physiological function. Annu. Rev. Nutr. 1994, 14, 297–320. [Google Scholar] [CrossRef] [PubMed]
- McOrist, A.L.; Miller, R.B.; Bird, A.R.; Keogh, J.B.; Noakes, M.; Topping, D.L.; Conlon, M.A. Fecal butyrate levels vary widely among individuals but are usually increased by a diet high in resistant starch. J. Nutr. 2011, 141, 883–889. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, T.S.; Laerke, H.N.; Theil, P.K.; Sorensen, J.F.; Saarinen, M.; Forssten, S.; Bach Knudsen, K.E. Diets high in resistant starch and arabinoxylan modulate digestion processes and scfa pool size in the large intestine and faecal microbial composition in pigs. Br. J. Nutr. 2014, 112, 1837–1849. [Google Scholar] [CrossRef] [PubMed]
- Clarke, J.M.; Topping, D.L.; Christophersen, C.T.; Bird, A.R.; Lange, K.; Saunders, I.; Cobiac, L. Butyrate esterified to starch is released in the human gastrointestinal tract. Am. J. Clin. Nutr. 2011, 94, 1276–1283. [Google Scholar] [CrossRef] [PubMed]
- Clarke, J.M.; Bird, A.R.; Topping, D.L.; Cobiac, L. Excretion of starch and esterified short-chain fatty acids by ileostomy subjects after the ingestion of acylated starches. Am. J. Clin. Nutr. 2007, 86, 1146–1151. [Google Scholar] [CrossRef] [PubMed]
- Annison, G.; Illman, R.J.; Topping, D.L. Acetylated, propionylated or butyrylated starches raise large bowel short-chain fatty acids preferentially when fed to rats. J. Nutr. 2003, 133, 3523–3528. [Google Scholar] [CrossRef] [PubMed]
- Bajka, B.H.; Clarke, J.M.; Cobiac, L.; Topping, D.L. Butyrylated starch protects colonocyte DNA against dietary protein-induced damage in rats. Carcinogenesis 2008, 29, 2169–2174. [Google Scholar] [CrossRef] [PubMed]
- Toden, S.; Lockett, T.J.; Topping, D.L.; Scherer, B.L.; Watson, E.J.; Southwood, J.G.; Clarke, J.M. Butyrylated starch affects colorectal cancer markers beneficially and dose-dependently in genotoxin-treated rats. Cancer Biol. Ther. 2014, 15, 1515–1523. [Google Scholar] [CrossRef] [PubMed]
- Le Leu, R.K.; Winter, J.M.; Christophersen, C.T.; Young, G.P.; Humphreys, K.J.; Hu, Y.; Gratz, S.W.; Miller, R.B.; Topping, D.L.; Bird, A.R.; et al. Butyrylated starch intake can prevent red meat-induced o6-methyl-2-deoxyguanosine adducts in human rectal tissue: A randomised clinical trial. Br. J. Nutr. 2015, 114, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Tupa, M.; Maldonado, L.; Vazquez, A.; Foresti, M.L. Simple organocatalytic route for the synthesis of starch esters. Carbohydr. Polym. 2013, 98, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Rubio, A.; Clarke, J.M.; Scherer, B.; Topping, D.L.; Gilbert, E.P. Structural modifications of granular starch upon acylation with short-chain fatty acids. Food Hydrocol. 2009, 23, 1940–1946. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemists. Official Methods of Analysis of AOAC International, 15th ed.; AOAC International: Arlington, VA, USA, 1990. [Google Scholar]
- Englyst, H.N.; Kingman, S.M.; Cummings, J.H. Classification and measurement of nutritionally important starch fractions. Eur. J. Clin. Nutr. 1992, 46, S33–S50. [Google Scholar] [PubMed]
- Peersen, O.B.; Wu, X.L.; Kustanovich, I.; Smith, S.O. Variable-amplituede cross-polarization mas NMR. J. Magn. Reson. A 1993, 104, 334–339. [Google Scholar] [CrossRef]
- Bennett, A.E.; Rienstra, C.M.; Auger, M.; Lakshmi, K.V.; Griffin, R.G. Heteronuclear decoupling in rotating solids. J. Chem. Phys. 1995, 103, 6951–6958. [Google Scholar] [CrossRef]
- Edwards, C.A.; Gibson, G.; Champ, M.; Jensen, B.B.; Mathers, J.C.; Nagengast, F.; Rumney, C.; Quehl, A. In vitro method for quantification of the fermentation of starch by human faecal bacteria. J. Sci. Food Agric. 1996, 71, 209–217. [Google Scholar] [CrossRef]
- Canibe, N.; Hojberg, O.; Badsberg, J.H.; Jensen, B.B. Effect of feeding fermented liquid feed and fermented grain on gastrointestinal ecology and growth performance in piglets. J. Anim. Sci. 2007, 85, 2959–2971. [Google Scholar] [CrossRef] [PubMed]
- Bach-Knudsen, K.E. Carbohydrate and lignin contents of plant materials used in animal feeding. Anim. Feed Sci. Technol. 1997, 67, 319–338. [Google Scholar] [CrossRef]
- Di Filippo, S.; Tupa, M.V.; Vazquez, A.; Foresti, M.L. Organocatalytic route for the synthesis of propionylated starch. Carbohydr. Polym. 2016, 137, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Bertoft, E. Understanding starch structure: Recent progress. Agronomy 2017, 7, 29. [Google Scholar] [CrossRef]
- Jobling, S. Improving starch for food and industrial applications. Curr. Opi. Plant Biol. 2004, 7, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Tupa, M.V.; Avila Ramirez, J.A.; Vazquez, A.; Foresti, M.L. Organocatalytic acetylation of starch: Effect of reaction conditions on ds and characterisation of esterified granules. Food Chem. 2015, 170, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.-M.; Barnes, M.B.; Gras, S.L.; McSweeney, C.; Lockett, T.; Augustin, M.A.; Gooley, P.R. Esterification of high amylose starch with short chain fatty acids modulates degradation by bifidobacterium spp. J. Func. Foods 2014, 6, 137–146. [Google Scholar] [CrossRef]
- Erickson, J.M.; Carlson, J.L.; Stewart, M.L.; Slavin, J.L. Fermentability of novel type-4 resistant starches in in vitro system. Foods 2018, 7. [Google Scholar] [CrossRef] [PubMed]
- Bajka, B.H.; Topping, D.L.; Cobiac, L.; Clarke, J.M. Butyrylated starch is less susceptible to enzymic hydrolysis and increases large-bowel butyrate more than high-amylose maize starch in the rat. Br. J. Nutr. 2006, 96, 276–282. [Google Scholar] [CrossRef] [PubMed]

| Chemical Composition (% of DM) | LAMS | HAMS | LAPS | HAPS |
|---|---|---|---|---|
| DM (%) | 91.7 | 87.8 | 96.3 | 86.9 |
| Ash | 0.19 | 0.15 | 0.34 | 0.85 |
| Total starch | 97.7 | 97.9 4 | 97.0 | 93.7 |
| Amylose 1 | 30.2 | 79.6 | 19.3 | 41.5 |
| Amylopectin 1 | 69.8 | 20.4 | 80.7 | 58.5 |
| RS 2 | 0.8 | 39.9 | 0.6 | 25.3 |
| RS 3 (% of total starch) | 0.8 | 40.8 | 0.6 | 27.0 |
| Sample | DS 1, Butyrate (NMR) | DS 1, Tartarate (NMR) | DS 1 (Chemical Method) |
|---|---|---|---|
| HAMS-low DS | 0.038 | 0.31 (31) 2 | 0.35 |
| LAMS-low DS | 0.037 | 0.28 (28) 2 | 0.44 |
| HAPS-low DS | 0.022 | 0.08 (8) 2 | 0.17 |
| LAPS-low DS | 0.035 | 0.16 (16) 2 | 0.30 |
| HAMS-high DS | 0.11 | 0.92 (92) 2 | 1.49 |
| LAMS-high DS | 0.23 | 1.16 (116) 2 | 2.48 |
| HAPS-high DS | 0.27 | 0.92 (92) 2 | 2.27 |
| LAPS-high DS | 0.31 | 0.93 (93) 2 | 2.26 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nielsen, T.S.; Canibe, N.; Larsen, F.H. Butyrylation of Maize and Potato Starches and Characterization of the Products by Nuclear Magnetic Resonance and In Vitro Fermentation. Foods 2018, 7, 79. https://doi.org/10.3390/foods7050079
Nielsen TS, Canibe N, Larsen FH. Butyrylation of Maize and Potato Starches and Characterization of the Products by Nuclear Magnetic Resonance and In Vitro Fermentation. Foods. 2018; 7(5):79. https://doi.org/10.3390/foods7050079
Chicago/Turabian StyleNielsen, Tina Skau, Nuria Canibe, and Flemming Hofmann Larsen. 2018. "Butyrylation of Maize and Potato Starches and Characterization of the Products by Nuclear Magnetic Resonance and In Vitro Fermentation" Foods 7, no. 5: 79. https://doi.org/10.3390/foods7050079
APA StyleNielsen, T. S., Canibe, N., & Larsen, F. H. (2018). Butyrylation of Maize and Potato Starches and Characterization of the Products by Nuclear Magnetic Resonance and In Vitro Fermentation. Foods, 7(5), 79. https://doi.org/10.3390/foods7050079