Phenolic Profiling and Antioxidant Capacity of Eugenia uniflora L. (Pitanga) Samples Collected in Different Uruguayan Locations
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Samples
2.3. Extraction
2.4. Identification of Phenolic Compounds Via HPLC-DAD-ESI/MSn and Quantification Via RP-HPLC-DAD
2.5. DPPH Antioxidant Activity Measurements
2.6. ORAC Antioxidant Activity Measurements
2.7. Statistical Analysis
3. Results and Discussion
3.1. Water Content
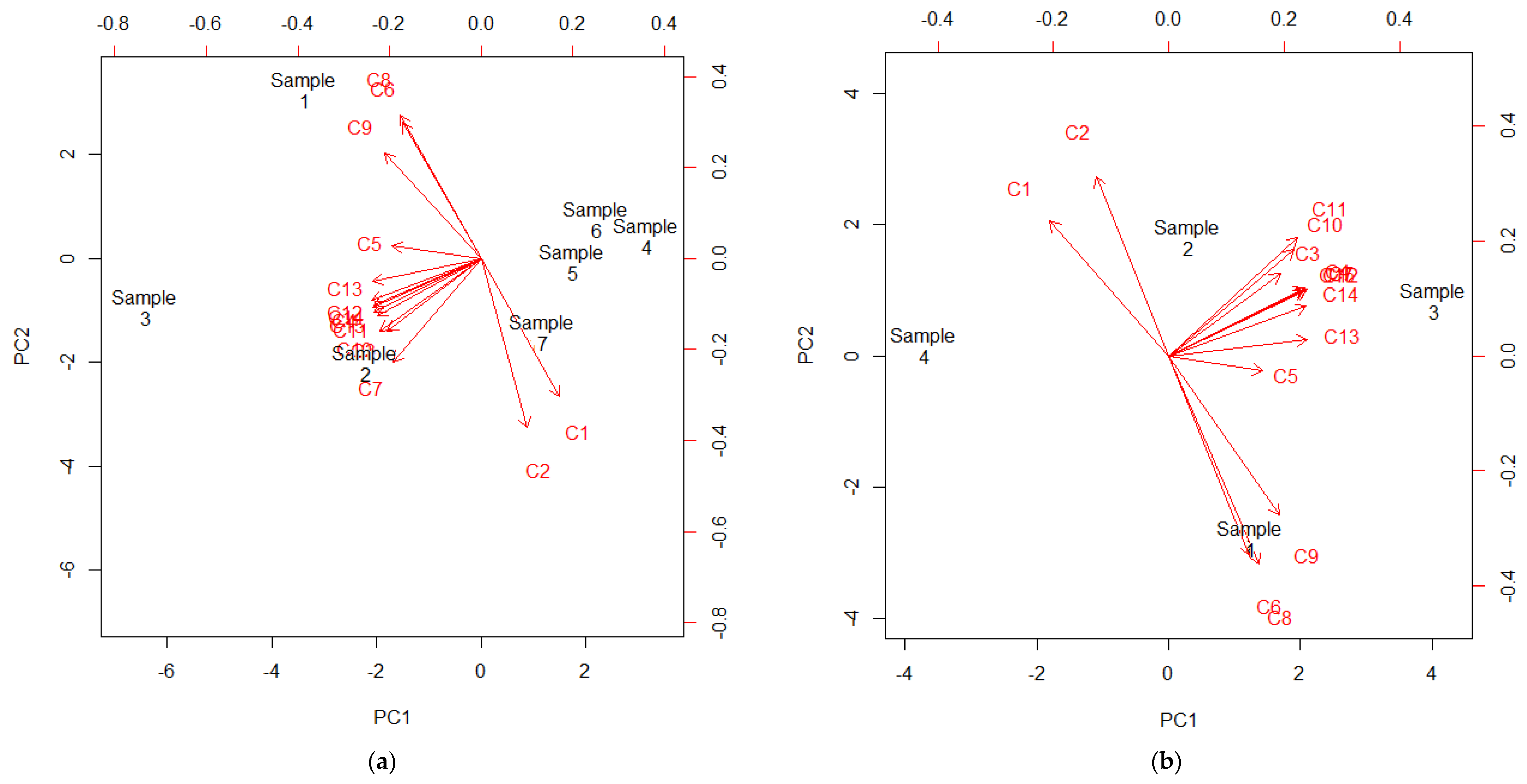
3.2. Identification and Quantification of Phenolic Compounds
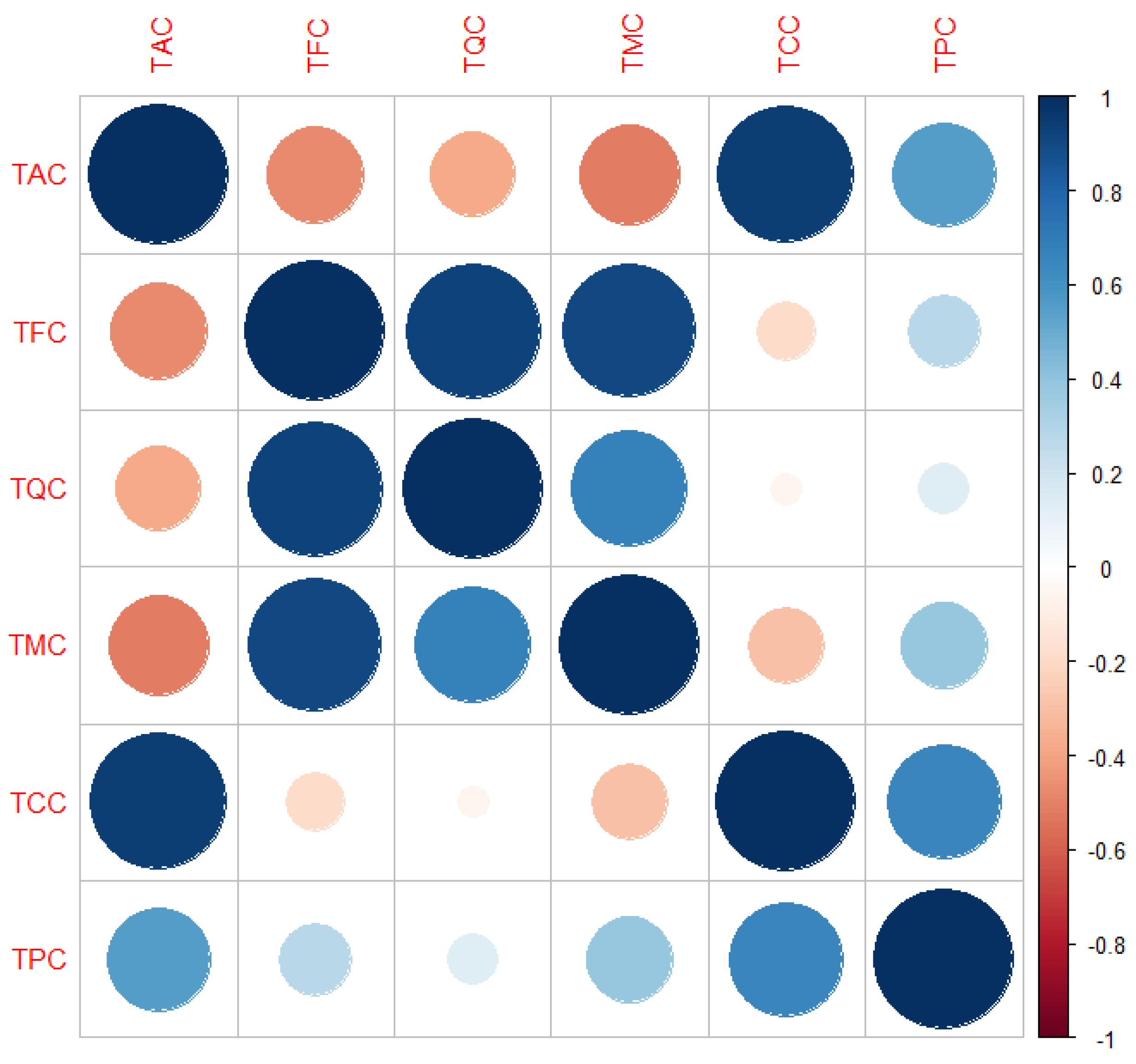
3.3. Antioxidant Activity
(R2 = 0.7345, p-value = 1.876 × 10−5, RSE = 9050 on 15 Degrees of Freedom)
(R2 = 0.9995, p-value = 8.487 × 10−12, RSE = 238.3 on 7 Degrees of Freedom)
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Bicas, J.L.; Molina, G.; Dionísio, A.P.; Barros, F.F.C.; Wagner, R.; Maróstica, M.R.; Pastore, G.M. Volatile constituents of exotic fruits from Brazil. Food Res. Int. 2011, 44, 1843–1855. [Google Scholar] [CrossRef]
- Consolini, A.E.; Sarubbio, M.G. Pharmacological effects of Eugenia uniflora (Myrtaceae) aqueous crude extract on rat’s heart. J. Ethnopharmacol. 2002, 81, 57–63. [Google Scholar] [CrossRef]
- Amoo, A.; Adebayo, O.; Oyeleye, A. Chemical evaluation of winged beans (Psophocarpus Tetragonolobus), Pitanga cherries (Eugenia uniflora) and orchid fruit (Orchid fruit myristica). Afr. J. Food Agric. Nutr. Dev. 2006, 6, 3–12. [Google Scholar]
- Vasconcelos Costa, A.G.; Garcia-Diaz, D.F.; Jimenez, P.; Ibrahim Silva, P. Bioactive compounds and health benefits of exotic tropical red–blackberries. J. Funct. Foods 2013, 5, 539–549. [Google Scholar] [CrossRef]
- Celli, G.B.; Pereira-Netto, A.B.; Beta, T. Comparative analysis of total phenolic content, antioxidant activity, and flavonoids profile of fruits from two varieties of Brazilian cherry (Eugenia uniflora L.) throughout the fruit developmental stages. Food Res. Int. 2011, 44, 2442–2451. [Google Scholar] [CrossRef]
- Galvão de Lima, V.L.; Almeida Mélo, E.; da Silva Lima, D.E. Fenólicos e carotenóides totais em pitanga. Sci. Agric. 2002, 59, 447–450. [Google Scholar] [CrossRef]
- Corrêa, P. Dicionário das Plantas Uteis do Brasil e das Exóticas Cultivadas; Imprensa National-Ministerio da Agricultural: Rio de Janeiro, Brazil, 1984; Volume 5.
- Rücker, G.; Brasil e Silva, G.A.A.; Bauer, L.; Schikarski, M. Neu Inhaltstoffe von Stenoealyx michelii (New constituents of Stenoealyx michelii). Planta Med. 1977, 31, 322–327. [Google Scholar] [CrossRef]
- Ferro, E.; Schinini, A.; Maldonado, M.; Rosner, J.; Schmeda-Hirschmann, G. Eugenia unliflora leaf extract and lipid metabolism in Cebus apella monkeys. J. Ethnopharmacol. 1988, 24, 321–325. [Google Scholar] [CrossRef]
- Schapoval, E.E.S.; Silveira, S.M.; Miranda, M.L.; Alice, C.B.; Henriques, A.T. Evaluation of some pharmacological activities of Eugenia uniflora L. J. Ethnopharmacol. 1994, 44, 137–142. [Google Scholar] [CrossRef]
- Vizzotto, M.; Cabral, L.; Santos Lopes, S. Pitanga (Eugenia uniflora L.). In Postharvest Biology and Technology of Tropical and Subtropical Fruits. Mangosteen to White Sapote; Yahia, E., Ed.; Woodhead Publishing: Cambridge, UK, 2011; Volume 4, pp. 272–286. [Google Scholar]
- Soares, D.J.; Walker, J.; Pignitter, M.; Walker, J.M.; Imboeck, J.M.; Ehrnhoefer-Ressler, M.M.; Montenegro Brasil, I.; Veronika, S. Pitanga (Eugenia uniflora L.) fruit juice and two major constituents thereof exhibit anti-inflammatory properties in human gingival and oral gum epithelial cells. Food Funct. 2014, 5, 2981–2988. [Google Scholar] [CrossRef] [PubMed]
- Soares, D.J.; Pignitter, M.; Ehrnhöfer-Ressler, M.M.; Walker, J.; Montenegro, I.; Somoza, V. Identification and Quantification of Oxidoselina-1,3,7-Trien-8-One and Cyanidin-3-Glucoside as One of the Major Volatile and Non-Volatile Low-Molecular- Weight Constituents in Pitanga Pulp. PLoS ONE 2015, 10, e0138809. [Google Scholar] [CrossRef]
- DeFelice, S.L. The nutraceutical revolution: Its impact on food industry R&D. Trends Food Sci. Technol. 1995, 6, 59–61. [Google Scholar]
- Tai, A.; Sawano, T.; Yazama, F.; Ito, H. Evaluation of antioxidant activity of vanillin by using multiple antioxidant assays. Biochim. Biophys. Acta Gen. Subj. 2011, 1810, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.; Acquaviva, A.; Dennis, G.R.; Shalliker, R.A.; Soliven, A. High throughput screening of phenolic constituents in a complex sample matrix using post-column derivatisations employing reaction flow HPLC columns. Microchem. J. 2018. [Google Scholar] [CrossRef]
- Acquaviva, A.; Jones, A.; Dennis, G.R.; Shalliker, R.A.; Soliven, A. Phenolic profiling of complex tea samples via simultaneous multiplexed detection employing reaction flow HPLC columns and colorimetric post column derivatisation. Microchem. J. 2018, 138, 533–539. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Prior, R.L. The Chemistry behind Antioxidant Capacity Assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef] [PubMed]
- Acquaviva, A.; Jones, A.; Dennis, G.R.; Shalliker, R.A.; Soliven, A. Detection selectivity in the analysis of “reactive” chemical compounds derived from natural samples via reaction flow chromatography. Microchem. J. 2018, 139, 315–321. [Google Scholar] [CrossRef]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized Methods for the Determination of Antioxidant Capacity and Phenolics in Foods and Dietary Supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Schaich, K.M. Re-evaluation of the 2,2-diphenyl-1-picrylhydrazyl Free Radical (DPPH) Assay for Antioxidant Activity. J. Agric. Food Chem. 2014, 62, 4251–4260. [Google Scholar] [CrossRef] [PubMed]
- Bagetti, M.; Facco, E.M.P.; Piccolo, J.; Hirsch, G.E.; Rodriguez-Amaya, D.; Kobori, C.N.; Vizzotto, M.; Emanuelli, T. Physicochemical characterization and antioxidant capacity of pitanga fruits (Eugenia uniflora L.). Ciênc. Tecnol. Aliment. 2011, 31, 147–154. [Google Scholar] [CrossRef]
- Lim, T.K. Edible Medicinal and Non-Medicinal Plants; Springer: Dordrecht, The Netherlands, 2012; pp. 620–630. ISBN 978-94-007-1764-0. [Google Scholar]
- Di Rienzo, J.; Casanoves, F.; Balzarini, M.G.; Gonzalez, L.; Tablada, M.; Robledo, C.W. InfoStat Versión 2017; InfoStat: Córdoba, Argentina, 2017. [Google Scholar]
- Romero, P.; Gil-Muñoz, R.; del Amor, F.M.; Valdés, E.; Fernández, J.I.; Martinez-Cutillas, A. Regulated Deficit Irrigation based upon optimum water status improves phenolic composition in Monastrell grapes and wines. Agric. Water Manag. 2013, 121, 85–101. [Google Scholar] [CrossRef]
- Mielke, M.S.; Schaffer, B. Photosynthetic and growth responses of Eugenia uniflora L. seedlings to soil flooding and light intensity. Environ. Exp. Bot. 2010, 68, 113–121. [Google Scholar] [CrossRef]
- INUMET ANOMALIAS CLIMATICAS Durante 2014 en Uruguay. Available online: http://www.meteorologia.com.uy/reportes/noticias/docs/pdf/rrpp/168_53868.pdf (accessed on 15 October 2016).
- Einbond, L.S.; Reynertson, K.A.; Luo, X.-D.; Basile, M.J.; Kennelly, E.J. Anthocyanin antioxidants from edible fruits. Food Chem. 2004, 84, 23–28. [Google Scholar] [CrossRef]
- Gironés-Vilaplana, A.; Baenas, N.; Villaño, D.; Speisky, H.; García-Viguera, C.; Moreno, D.A. Evaluation of Latin-American fruits rich in phytochemicals with biological effects. J. Funct. Foods 2014, 7, 599–608. [Google Scholar] [CrossRef]
- Jaakola, L.; Määttä, K.; Pirttilä, A.M.; Törrönen, R.; Kärenlampi, S.; Hohtola, A. Expression of Genes Involved in Anthocyanin Biosynthesis in Relation to Anthocyanin, Proanthocyanidin, and Flavonol Levels during Bilberry Fruit Development. Plant Physiol. 2002, 130, 729–739. [Google Scholar] [CrossRef] [PubMed]
- Peyrat-Maillard, M.N.; Cuvelier, M.E.; Berset, C. Antioxidant activity of phenolic compounds in 2,2′-azobis (2-amidinopropane) dihydrochloride (AAPH)-induced oxidation: Synergistic and antagonistic effects. J. Am. Oil Chem. Soc. 2003, 80, 1007–1012. [Google Scholar] [CrossRef]
| Sample | Location/Sampling Date | % Water (% w/w) 1 |
|---|---|---|
| 1 | North of Montevideo/December 2014 | 68.1 c,d ± 0.9 |
| 2 | South of Montevideo/December 2014 | 63.6 b,c ± 1.7 |
| 3 | Paysandú/December 2014 | 66.1 b,c,d ± 0.9 |
| 4 | Canelones/December 2014 | 46.4 a ± 6.4 |
| 5 | North of Montevideo/November 2015 | 66.5 b,c,d ± 4.1 |
| 6 | North of Montevideo/December 2015 | 58.3 b ± 0.6 |
| 7 | North of Montevideo/April 2016 | 73.0 d ± 2.7 |
| Compound 2 | Rt | [M − H]+ | MSn | Sample 1 | Sample 2 | Sample 3 | Sample 4 | Sample 5 | Sample 6 | Sample 7 |
|---|---|---|---|---|---|---|---|---|---|---|
| C1 | 29.8 | 465 | 303 | 19.99 a ± 3.34 | 373.24 c ± 3.98 | ND 3,a | 381.38 c ± 31.81 | 282.99 b ± 24.49 | 215.53 b ± 19.42 | 485.81 d ± 26.25 |
| C2 | 33.8 | 449 | 287 | 115.01 a ± 8.56 | 1757.29 d ± 18.34 | 150.45 a ± 0.25 | 987.77 c ± 49.63 | 916.35 b,c ± 85.15 | 706.78 b ± 68.87 | 2248.68 e ± 98.76 |
| [M − H]− | ||||||||||
| C3 | 48.4 | 631 | 479, 317, 271 | 8.07 d ± 1.15 | 14.18 f ± 0.34 | 10.67 e ± 0.34 | ND 3,a | 3.63 b,c ± 0.08 | 2.41 b ± 0.57 | 4.94 c ± 0.36 |
| C4 | 53.6 | 479 | 317, 271 | 34.33 c ± 1.28 | 46.41 d ± 0.43 | 56.53 e ± 1.11 | 6.11 a ± 0.18 | 12.97 b ± 1.52 | 8.77 a ± 0.86 | 17.05 b ± 2.14 |
| C5 | 54.7 | 479 | 317, 271 | 32.37 d ± 2.52 | 35.82 d ± 0.34 | 22.59 c ± 1.49 | 6.53 a,b ± 0.50 | 4.85 a ± 1.76 | 7.57 a,b ± 0.78 | 10.25 b ± 2.45 |
| C6 | 56.8 | 449 | 317, 271, 179 | 23.44 d ± 0.26 | 6.79 b,c ± 0.51 | 9.39 c ± 4.91 | 1.22 a ± 1.46 | 2.15 a,b ± 0.24 | 1.52 a ± 0.18 | 3.33 a,b ± 0.35 |
| C7 | 58.0 | 615 | 463, 301, 271 | 8.45 c ± 0.09 | 11.10 d ± 0.64 | 16.87 e ± 0.43 | 0.92 a ± 0.81 | 7.61 b,c ± 0.65 | 5.27 b ± 0.75 | 11.59 d ± 1.19 |
| C8 | 61.0 | 449 | 317, 271, 179 | 49.91 d ± 3.46 | 4.57 a,b ± 0.94 | 26.30 c ± 0.85 | 2.09 a ± 0.20 | 5.33 a,b ± 1.10 | 3.96 a ± 0.78 | 8.34 b ± 1.11 |
| C9 | 61.9 | 463 | 317, 271, 179 | 378.69 f ± 10.72 | 138.43 d ± 3.37 | 243.25 e ± 1.20 | 16.81 a ± 1.04 | 41.63 b ± 3.49 | 29.16 a,b ± 3.76 | 56.62 c ± 3.33 |
| C10 | 62.9 | 463 | 301 | 29.25 c ± 2.13 | 64.65 d ± 2.39 | 167.25 e ± 0.00 | 4.11 a ± 1.50 | 14.97 b ± 0.59 | 8.17 a ± 0.66 | 18.48 b ± 1.05 |
| C11 | 63.5 | 463 | 301 | 38.30 b ± 2.65 | 71.26 c ± 4.27 | 101.54 d ± 3.59 | 13.11 a ± 3.81 | 8.22 a ± 0.80 | 8.32 a ± 1.02 | 10.61 a ± 0.20 |
| C12 | 64.5 | 433 | 301 | 8.97 c ± 1.24 | 10.59 c ± 0.04 | 19.64 d ± 0.30 | 2.57 a,b ± 0.14 | 2.41 a,b ± 0.48 | 1.75 a ± 0.42 | 4.17 b ± 1.35 |
| C13 | 64.9 | 433 | 301 | 28.44 d ± 1.75 | 8.71 c ± 2.43 | 55.81 e ± 0.38 | 6.55 a ± 1.35 | 11.36 a,b ± 2.30 | 6.79 a ± 0.96 | 16.48 b,c ± 1.55 |
| C14 | 65.4 | 433 | 301 | 36.12 d ± 0.43 | 36.21 d ± 0.13 | 92.95 e ± 0.17 | 3.37 a ± 0.51 | 15.12 b ± 2.32 | 9.98 b ± 2.93 | 22.92 c ± 2.90 |
| C15 | 65.7 | 447 | 301 | 80.74 d ± 0.13 | 93.13 e ± 1.54 | 224.17 f ± 0.04 | 15.21 a ± 0.33 | 34.62 b ± 3.82 | 26.44 b ± 4.27 | 53.22 c ± 3.34 |
| Sample 1 | Sample 2 | Sample 3 | Sample 4 | Sample 5 | Sample 6 | Sample 7 | |
|---|---|---|---|---|---|---|---|
| TAC | 134.98 a ± 11.90 | 2130.53 d ± 14.35 | 150.45 a ± 0.24 | 1369.15 c ± 79.26 | 1199.35 b,c ± 109.47 | 922.32 b ± 87.78 | 2734.50 e ± 125.00 |
| TQC | 230.26 d ± 1.61 | 308.45 e ± 8.03 | 678.25 f ± 4.14 | 45.84 a ± 2.98 | 94.33 b ± 8.56 | 66.70 a ± 7.42 | 137.44 c ± 11.21 |
| TMC | 526.83 f ± 17.08 | 246.20 d ± 3.43 | 368.79 e ± 6.24 | 32.76 a ± 1.65 | 70.54 b ± 8.14 | 53.39 a,b ± 6.56 | 100.50 c ± 9.57 |
| TFC | 757.09 f ± 18.69 | 554.65 e ± 11.47 | 1047.04 g ± 10.38 | 78.60 a ± 2.50 | 164.87 c ± 14.57 | 120.08 b ± 7.39 | 237.94 d ± 17.48 |
| TCC | 345.25 a ± 10.18 | 2065.75 d ± 10.30 | 828.70 b,c ± 4.38 | 1033.62 c ± 49.24 | 1010.68 c ± 92.65 | 773.48 b ± 65.14 | 2386.12 e ± 100.13 |
| TPC | 546.82 c ± 20.42 | 619.43 c ± 7.42 | 368.79 b ± 6.28 | 414.13 b ± 31.28 | 353.54 a,b ± 32.23 | 268.92 a ± 25.93 | 586.32 c ± 35.30 |
| TPPC | 892.07 a ± 30.59 | 2685.18 d ± 2.89 | 1197.49 b,c ± 10.62 | 1447.75 c ± 77.27 | 1364.21 c ± 124.02 | 1042.40 a,b ± 90.94 | 2972.43 d ± 135.41 |
| Assay | Sample 1 | Sample 2 | Sample 3 | Sample 4 | Sample 5 | Sample 6 | Sample 7 |
|---|---|---|---|---|---|---|---|
| ORAC | 57,440 b,c ± 1090 | 44,510 a,b ± 460 | 22,800 a ± 300 | 57,560 b,c ± 10,040 | 57,400 b,c ± 16,380 | 82,340 c ± 9070 | 72,550 c ± 15,280 |
| DPPH | 44,170 b ± 4480 | 11,940 a ± 2200 | 10,070 a ± 1200 | 12,200 a ± 650 | 15,430 a ± 70 | 12,960 a ± 1250 | 13,950 a ± 560 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Migues, I.; Baenas, N.; Gironés-Vilaplana, A.; Cesio, M.V.; Heinzen, H.; Moreno, D.A. Phenolic Profiling and Antioxidant Capacity of Eugenia uniflora L. (Pitanga) Samples Collected in Different Uruguayan Locations. Foods 2018, 7, 67. https://doi.org/10.3390/foods7050067
Migues I, Baenas N, Gironés-Vilaplana A, Cesio MV, Heinzen H, Moreno DA. Phenolic Profiling and Antioxidant Capacity of Eugenia uniflora L. (Pitanga) Samples Collected in Different Uruguayan Locations. Foods. 2018; 7(5):67. https://doi.org/10.3390/foods7050067
Chicago/Turabian StyleMigues, Ignacio, Nieves Baenas, Amadeo Gironés-Vilaplana, María Verónica Cesio, Horacio Heinzen, and Diego A. Moreno. 2018. "Phenolic Profiling and Antioxidant Capacity of Eugenia uniflora L. (Pitanga) Samples Collected in Different Uruguayan Locations" Foods 7, no. 5: 67. https://doi.org/10.3390/foods7050067
APA StyleMigues, I., Baenas, N., Gironés-Vilaplana, A., Cesio, M. V., Heinzen, H., & Moreno, D. A. (2018). Phenolic Profiling and Antioxidant Capacity of Eugenia uniflora L. (Pitanga) Samples Collected in Different Uruguayan Locations. Foods, 7(5), 67. https://doi.org/10.3390/foods7050067







