Systematic Review and Meta-Analysis of a Proprietary Alpha-Amylase Inhibitor from White Bean (Phaseolus vulgaris L.) on Weight and Fat Loss in Humans
Abstract
:1. Introduction
2. Methods
2.1. Literature Search
2.2. Study Selection Criteria
2.3. Assessment of Risk of Bias
2.4. Data Extraction and Quality Assessment
2.5. Statistical Analysis
3. Results
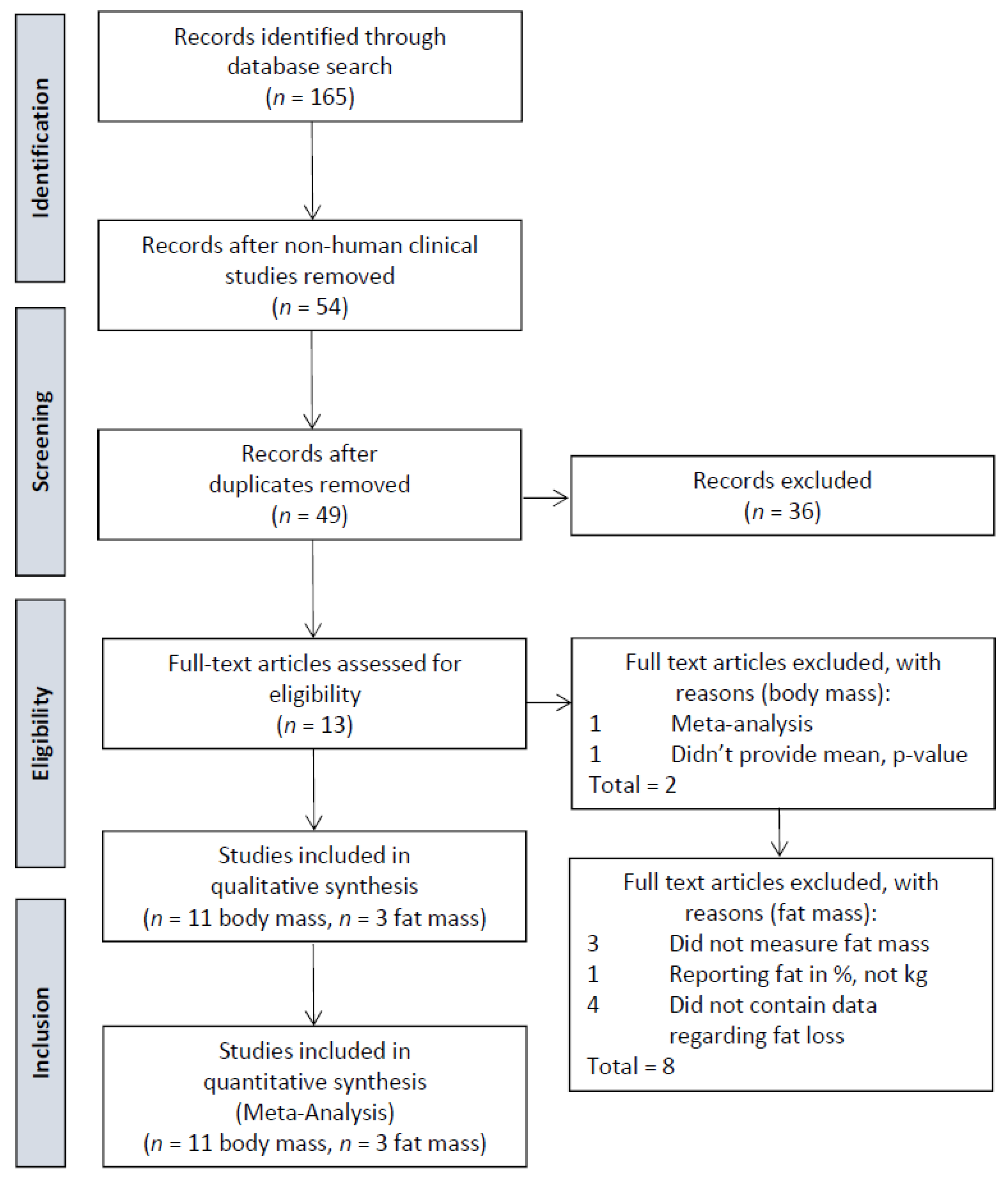
3.1. Article Selection
3.2. Phaseolus vulgaris Doses and Duration of Supplementation
3.3. Control Groups
3.4. Effects on Body Weight
3.5. Effects on Fat Loss
3.6. Risk of Bias and Publication Bias
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- De Toro-Martín, J.; Arsenault, B.J.; Després, J.P.; Vohl, M.C. Precision Nutrition: A Review of Personalized Nutritional Approaches for the Prevention and Management of Metabolic Syndrome. Nutrients 2017, 9, 913. [Google Scholar] [CrossRef] [PubMed]
- Preuss, H.G.; Bagchi, D. Nutritional therapy of impaired glucose tolerance and diabetes mellitus. In Nutritional Aspects and Clinical Management of Chronic Disorders and Diseases; Bronner, F., Ed.; CRC Press: Boca Raton, FL, USA, 2002; pp. 69–91. ISBN 9781420041286. [Google Scholar]
- Bell, S.J.; Sears, B. Low-Glycemic-Load Diets: Impact on Obesity and Chronic Diseases. Crit. Rev. Food Sci. Nutr. 2003, 43, 357–377. [Google Scholar] [CrossRef] [PubMed]
- Fukagawa, N.K.; Anderson, J.W.; Hageman, G.; Young, V.R.; Ninaker, K.L. High-carbohydrate, high-fiber diets increase peripheral insulin sensitivity in healthy young and old adults. Am. J. Clin. Nutr. 1990, 52, 524–528. [Google Scholar] [CrossRef] [PubMed]
- Bowman, D.E. Amylase inhibitor of navy bean. Science 1945, 102, 358–359. [Google Scholar] [CrossRef] [PubMed]
- Santimone, M.; Koukiekolo, R.; Moreau, Y.; Le Berre, V.; Rouge, P.; Marchis-Mouren, G.; Desseaux, V. Porcine pancreatic a-amylase inhibition by the kidney bean (Phaseolus vulgaris) inhibitor (α-AI1) and structural changes in the α-amylase inhibitor complex. Biochim. Biophys. Acta 2004, 1696, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Bompard-Gilles, C.; Rousseau, P.; Rouge, P.; Payan, F. Substrate mimicry in the active center of a mammalian α-amylase: Structural analysis of an enzyme-inhibitor complex. Structure 1996, 4, 1441–1452. [Google Scholar] [CrossRef]
- Payan, F. Structural basis for the inhibition of mammalian and insect α-amylases by plant protein inhibitors. Biochim. Biophys. Acta 2004, 1696, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Le Berre-Anton, V.; Bompard-Gilles, C.; Payan, F.; Rouge, P. Characterization and functional properties of the α-amylase inhibitor (α-AI) from kidney bean (Phaseolus vulgaris) seeds. Biochim. Biophys. Acta 1997, 1343, 31–40. [Google Scholar] [CrossRef]
- Lajolo, F.M.; Finardi-Filho, F. Partial characterization of the amylase inhibitor of black beans (Phaseolus vulgaris), variety Rico 23. J. Agric. Food Chem. 1985, 33, 132–138. [Google Scholar] [CrossRef]
- Udani, J.; Hardy, M.; Kavoussi, B. Dietary supplement carbohydrate digestion inhibitors: A review of the literature. In Obesity. Epidmiology, Pathophysiology, and Prevention; Bagchi, D., Preuss, H.G., Eds.; CRC Press: Boca Raton, FL, USA, 2007; pp. 279–298. [Google Scholar]
- Udani, K. The Mighty Bean; European Baker: Bucharest, Romania, 2005. [Google Scholar]
- Phase 2®/StarchLite. Available online: www.phase2info.com/pdf/Phase2_Study13.pdf (accessed on 5 January 2018).
- Phase 2®/StarchLite in Chewing Gum. Available online: http://www.phase2info.com/pdf/Phase2_Study14.pdf (accessed on 5 January 2018).
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; the PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Verhagen, A.P.; de Vet, H.C.W.; de Bie, R.A.; Kessels, A.G.; Boers, M.; Bouter, L.M.; Knipschild, P.G. The Delphi list: A criteria list for quality assessment of randomized clinical trials for conducting systematic reviews developed by Delphi consensus. J. Clin. Epidemiol. 1998, 51, 1235–1241. [Google Scholar] [CrossRef]
- Grube, B.; Chong, W.; Chong, P.; Riede, L. Weight Reduction and Maintenance with IQP-PV-101: A 12-Week Randomized Controlled Study with a 24-Week Open Label Period. Obesity 2014, 22, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Koike, T.; Koizumi, Y.; Tang, L.; Takahara, K.; Saitou, Y. The antiobesity effect and the safety of taking “Phaseolamin(TM) 1600 diet”. J. New Rem. Clin. 2005, 54, 1–16. [Google Scholar]
- Erner, S.; Meiss, D. The Effect of Thera-Slim on Weight, Body Composition and Select Laboratory Parameters in Adults with Overweight and Mild-Moderate Obesity. Unpublished work. 2003. [Google Scholar]
- Celleno, L.; Tolaini, M.V.; D’Amore, A.; Perricone, N.V.; Preuss, H.G. A dietary supplement containing standardized Phaseolus vulgaris extract influences body composition of overweight men and women. Int. J. Med. Sci. 2007, 49, 45–52. [Google Scholar] [CrossRef]
- Thom, E. A randomized, double-blind, placebo-controlled trial of a new weight-reducing agent of natural origin. J. Int. Med. Res. 2000, 28, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Udani, J.; Singh, B. Blocking Carbohydrate Absorption and Weight Loss: A Clinical Trial Using a Proprietary Fractionated White Bean Extract. Altern. Ther. Health Med. 2007, 13, 32–37. [Google Scholar] [PubMed]
- Osorio, L.; Gamboa, J. Random Multi-Center Evaluation to Test the Efficacy of Phaseolus vulgaris (Precarb) in Obese and Overweight Individuals. Unpublished work. 2005. [Google Scholar]
- Wu, X.; Xiaofeng, X.; Shen, J.; Perricone, N.; Preuss, H. Enhanced Weight Loss From a Dietary Supplement Containing Standardized Phaseolus vulgaris Extract in Overweight Men and Women. J. Appl. Res. 2010, 10, 73–79. [Google Scholar]
- Yamada, J.; Yamamoto, T.; Hideyo, Y. Effects of Combination of Functional Food Materials on Body Weight, Body Fat Percentage, Serum Triglyceride and Blood Glucose. Unpublished work. 2007. [Google Scholar]
- Asano, N. The Report of the Test Regarding the Efficacy of Phaseolamine on Weight Loss. Unpublished work. 2009. [Google Scholar]
- Rothacker, D. Reduction in body weight with a starch blocking diet aid: StarchAway comparison with placebo. Leiner Health Prod. 2003. Unpublished work. [Google Scholar]
- Udani, J.; Hardy, M.; Madsen, D.C. Blocking carbohydrate absorption and weight loss: A clinical trial using Phase 2 brand proprietary fractionated white bean extract. Altern. Med. Rev. 2004, 99, 63–69. [Google Scholar]
- Barrett, M.L.; Udani, J.K. A proprietary alpha-amylase inhibitor from white bean (Phaseolus vulgaris): A review of clinical studies on weight loss and glycemic control. Nutr. J. 2011, 10, 24. [Google Scholar] [CrossRef] [PubMed]
- Vinson, J.; Al Kharrat, H.; Shuta, D. Investigation of an amylase inhibitor on human glucose absorption after starch consumption. Open Nutraceuticals J. 2009, 2, 88–91. [Google Scholar] [CrossRef]
- Onakpoya, I.; Aldaas, S.; Terry, R.; Ernst, E. The efficacy of Phaseolus vulgaris as a weight-loss supplement: A systematic review and meta-analysis of randomised clinical trials. Br. J. Nutr. 2011, 106, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Harvey, C.J.D.C.; Schofield, G.M.; Williden, M. The use of nutritional supplements to induce ketosis and reduce symptoms associated with keto-induction: A narrative review. PeerJ 2018, 16, e4488. [Google Scholar] [CrossRef] [PubMed]
- Gardner, C.D.; Trepanowski, J.F.; Del Gobbo, L.C.; Hauser, M.E.; Rigdon, J.; Ioannidis, J.P.A.; Desai, M.; King, A.C. Effect of Low-Fat vs Low-Carbohydrate Diet on 12-Month Weight Loss in Overweight Adults and the Association With Genotype Pattern or Insulin Secretion. JAMA 2018, 319, 667–679. [Google Scholar] [CrossRef] [PubMed]
| Study | Treatment Group | Control Group | p | Weight | Effect (d) | Weighted Mean Difference (Fixed) 95% CI | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| n | Mean | SD | n | Mean | SD | Lower | Upper | ||||
| Udani et al. 2007 [11] | 13 | −6.0 | 12 | −4.7 | 0.424 | 4% | −0.33 | −0.46 | 1.12 | ||
| Asano [26] | 9 | −2.9 | 3% | −0.19 | −1.06 | 0.67 | |||||
| Koike et al. 2005 [18] | 10 | −1.8 | 0.002 | 3% | −1.61 | −0.61 | −2.62 | ||||
| Grube et al. 2014 [17] | 60 | −2.9 | 2.6 | 60 | −0.9 | 2.0 | 0.001 | 19% | −0.85 | −0.48 | −1.12 |
| Osorio et al. 2009 [23] | 49 | −2.3 | 49 | 2.21 | 0.001 | 15% | −1.00 | −0.41 | −1.60 | ||
| Rothacker (week 12) 2003 [27] | 30 | −6.9 | 60 | 0.8 | 0.029 | 9% | −0.58 | −0.62 | −1.09 | ||
| Wu et al. 2010 [24] | 50 | −1.9 | −0.2 | 51 | −0.4 | −0.1 | 0.049 | 15% | −0.40 | -0.00 | −0.79 |
| Celleno et al. 2007 [20] | 20 | −2.9 | −1.2 | 30 | −0.4 | 0.4 | 9% | −2.99 | −2.25 | −3.73 | |
| Thom et al. 2000 [21] | 20 | −3.5 | 20 | 2.0 | 0.001 | 6% | −1.13 | −0.46 | −0.12 | ||
| Udani et al. 2004 [28] | 20 | −3.8 | 19 | −1.65 | 0.35 | 6% | −0.30 | −0.33 | −0.93 | ||
| Yamada et al. [25] | 33 | −0.8 | 0.2 | 33 | 0.01 | 10% | −0.97 | −0.24 | −1.68 | ||
| Total | 314 | 259 | 0.001 | 100% | −1.08 | −0.43 | −1.16 | ||||
| Study | Treatment Group | Control Group | p | Weight | Effect (d) | Weighted Mean Difference (Fixed) 95% CI | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| n | Mean | SD | n | Mean | SD | Lower | Upper | ||||
| Koike et al. 2005 [18] | 10 | −1.2 | −0.4 | 0.001 | 17% | −1.58 | −2.58 | −0.57 | |||
| Celleno et al. 2007 [20] | 30 | −2.4 | −0.67 | 30 | −0.16 | −0.33 | 0.001 | 50% | −4.24 | −5.15 | −3.33 |
| Thom et al. 2000 [21] | 20 | −2.3 | −1.5 | 20 | 0.7 | −0.6 | 0.01 | 33% | −2.63 | −3.47 | −1.78 |
| Total | 60 | 50 | 0.02 | 100% | −3.26 | −4.16 | −2.35 | ||||
| Study | Participants | Intervention | Comparison | Methods | Study Design | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Country | Subjects Information | Dose | Diet Intervention | Duration of Intervention | n Phase 2 | n Control | Weight | Fat Mass | Design | Delphi-Score | |
| Asano et al. [26] | Japan | 5:1 female to male ratio; average age 36.3 + 12.7; BMI > 25; average BMI = 31.6 | 3000 mg per day (1000 mg per meal) | no caloric restriction | 2 months | 9 | 0 | Scale | n/a | Open-Label | 2 |
| Udani et al. 2007 [22] | USA | 0.3:1 female to male ratio; age 18-40; average BMI = 26 | 2000 mg per day (1000 mg at breakfast & lunch) | maintain a caloric intake of 1800 per day | 4 weeks | 13 | 12 | Scale | - | RDBPC | 8 |
| Koike et al. 2005 [18] | Japan | 1:1 female to male ratio; mean age 41.1 and BMI range 23–30 | 2× per day 1500 mg Phase 2, 400 mg Clove, 40 mg Lysine 40 mg, 40 mg Arginine, 40 mg Alanine | no caloric restriction | 8 weeks | 10 | 0 | Scale | n/a | Open-Label | 1 |
| Grube et al. 2014 [17] | Germany | 3:1 female to male ratio; mean age 46; BMI range 25–35 | 3000 mg per day (1000 mg per meal) | hypocaloric (500 kcal), providing 40% of energy as carbohydrates | 12 weeks | 60 | 57 | Scale | BIA | RDBPC | 9 |
| Osorio et al. 2009 [23] | Mexico | obese and overweight (age range 18–75 years) | 3000 mg per day (1000 mg per meal) | no caloric restriction besides carbohydrate-rich meals | 30 days | 37 | 0 | Scale | - | Open-Label | 1 |
| Rothacker 2003 [27] | USA | 24 male; 36 female; mean age 33.2; BMI range 24–32 | 3000 mg per day (1000 mg per meal) | no caloric restriction | 12 weeks | 34 | 26 | Scale | BIA | RDBPC | 8 |
| Wu et al. 2010 [24] | China | 1:1 female to male ratio; age 20-50; BMI range 25–40 | 3000 mg per day (1000 mg per meal) | no caloric restriction | 8 weeks | 51 | 50 | Scale | - | RDBPC | 8 |
| Celleno et al. 2007 [20] | Italy | 2.5:1 female to male ratio; mean age 34; average BMI = 26 | 3× per day 445 mg of Phase 2, 56 mg vitamin B3, and 0.5 mg chromium | carbohydrate-rich meals (100–200g) | 30 days | 30 | 29 | Scale | BIA | RDBPC | 8 |
| Thom et al. 2000 [21] | Norway | 9:1 female to male ratio; mean age 45.6; average BMI = 31 | 3× per day 400 mg Phase 2, 400 mg inulin, and 100 mg Garcinia cambogia | no caloric restriction | 12 weeks | 20 | 20 | Scale | BIA | RDBPC | 8 |
| Udani et al. 2004 [28] | USA | 9:1 female to male ratio; mean age 36.5; average weight of 193.1 pounds | 3000 mg per day (1000 mg per meal) | no caloric restriction | 8 weeks | 20 | 19 | Scale | BIA | RDBPC | 8 |
| Yamada et al. [25] | Japan | 1:1 female to male ratio; age 25–60; no BMI information | Twice a day proprietary functional food containing Phase 2 | no caloric restriction | 8 weeks | 23 | 24 | Scale | n/a | Open-Label | 4 |
| Delphi-Scores/Studies | Asano et al. [26] | Udani et al. 2007 [11] | Grube et al. 2014 [17] | Rothacker et al. 2003 [27] | Wu et al. 2010 [24] | Celleno et al. 2007 [20] | Thom et al. 2000 [21] | Udani et al. 2004 [28] | Koike et al. 2005 [18] | Osorio et al. 2009 [23] | Yamada et al. [25] |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. Treatment allocation (a) Was a method of randomization performed? | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 1 |
| (b) Was the treatment allocation concealed? | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 0 |
| 2. Were the groups similar at baseline regarding the most important prognostic indicators? | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 1 |
| 3. Where the eligibility criteria specified? | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 1 |
| 4. Was the outcome assessor blinded? | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 0 |
| 5. Was the care provider blinded? | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 0 |
| 6. Was the patient blinded? | 0 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 0 | 0 |
| 7. Were point estimates and measures of variability presented for the primary outcome measures? | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 8. Did the analysis include an intention-to-treat analysis? | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Total Delphi Score | 2 | 8 | 9 | 8 | 8 | 8 | 8 | 8 | 1 | 1 | 4 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Udani, J.; Tan, O.; Molina, J. Systematic Review and Meta-Analysis of a Proprietary Alpha-Amylase Inhibitor from White Bean (Phaseolus vulgaris L.) on Weight and Fat Loss in Humans. Foods 2018, 7, 63. https://doi.org/10.3390/foods7040063
Udani J, Tan O, Molina J. Systematic Review and Meta-Analysis of a Proprietary Alpha-Amylase Inhibitor from White Bean (Phaseolus vulgaris L.) on Weight and Fat Loss in Humans. Foods. 2018; 7(4):63. https://doi.org/10.3390/foods7040063
Chicago/Turabian StyleUdani, Jay, Ollie Tan, and Jhanna Molina. 2018. "Systematic Review and Meta-Analysis of a Proprietary Alpha-Amylase Inhibitor from White Bean (Phaseolus vulgaris L.) on Weight and Fat Loss in Humans" Foods 7, no. 4: 63. https://doi.org/10.3390/foods7040063
APA StyleUdani, J., Tan, O., & Molina, J. (2018). Systematic Review and Meta-Analysis of a Proprietary Alpha-Amylase Inhibitor from White Bean (Phaseolus vulgaris L.) on Weight and Fat Loss in Humans. Foods, 7(4), 63. https://doi.org/10.3390/foods7040063




