Aromatic Profiles of Essential Oils from Five Commonly Used Thai Basils
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Essential Oil Extraction
2.3. Physical Characteristics of Essential Oil
2.4. Gas Chromatography–Mass Spectrometry (GC-MS)
2.5. Descriptive Analysis
2.6. Statistical Analysis
3. Results and Discussion
3.1. Plant Identification and Physiochemical Characteristics of the Essential Oils
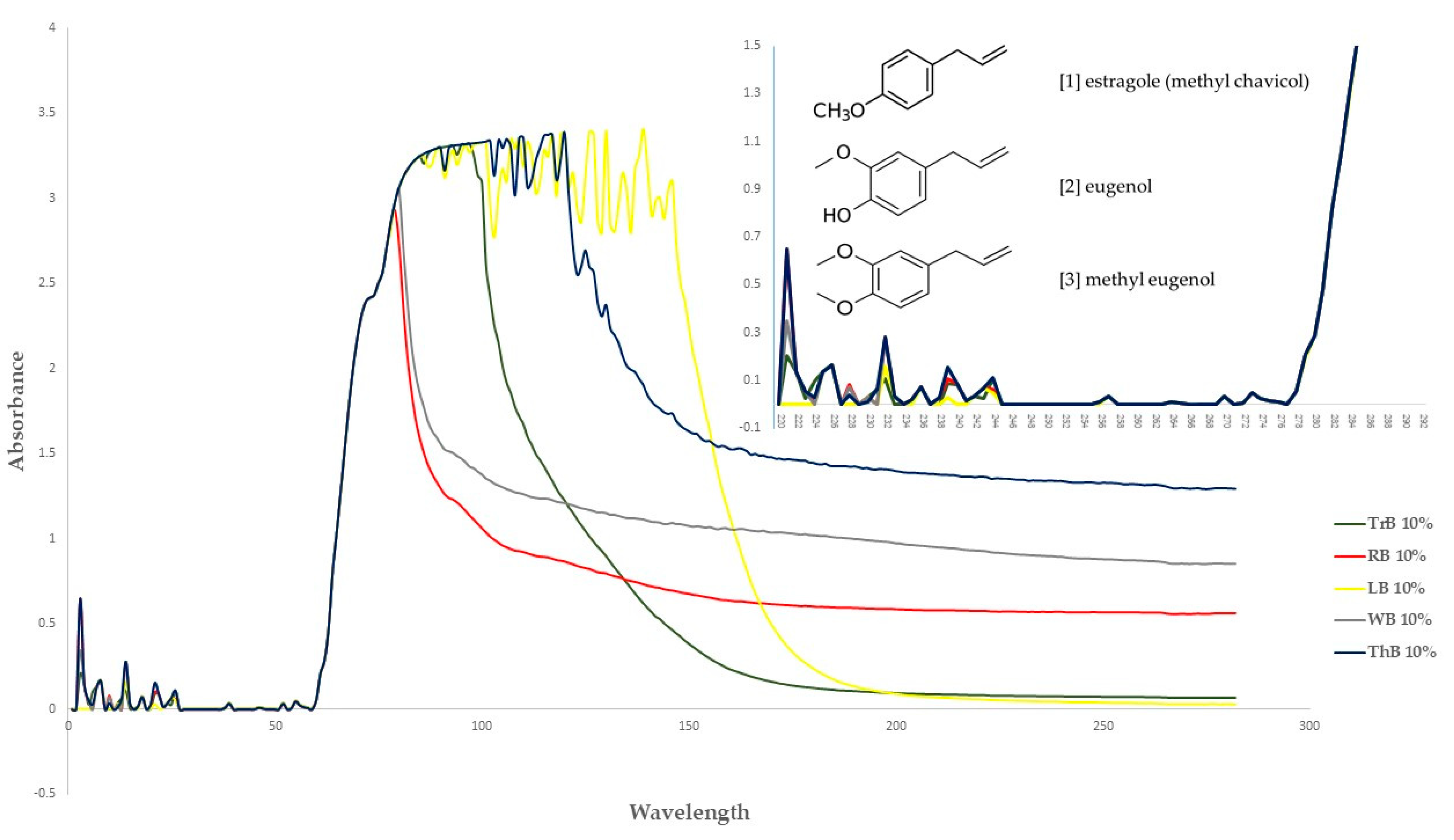
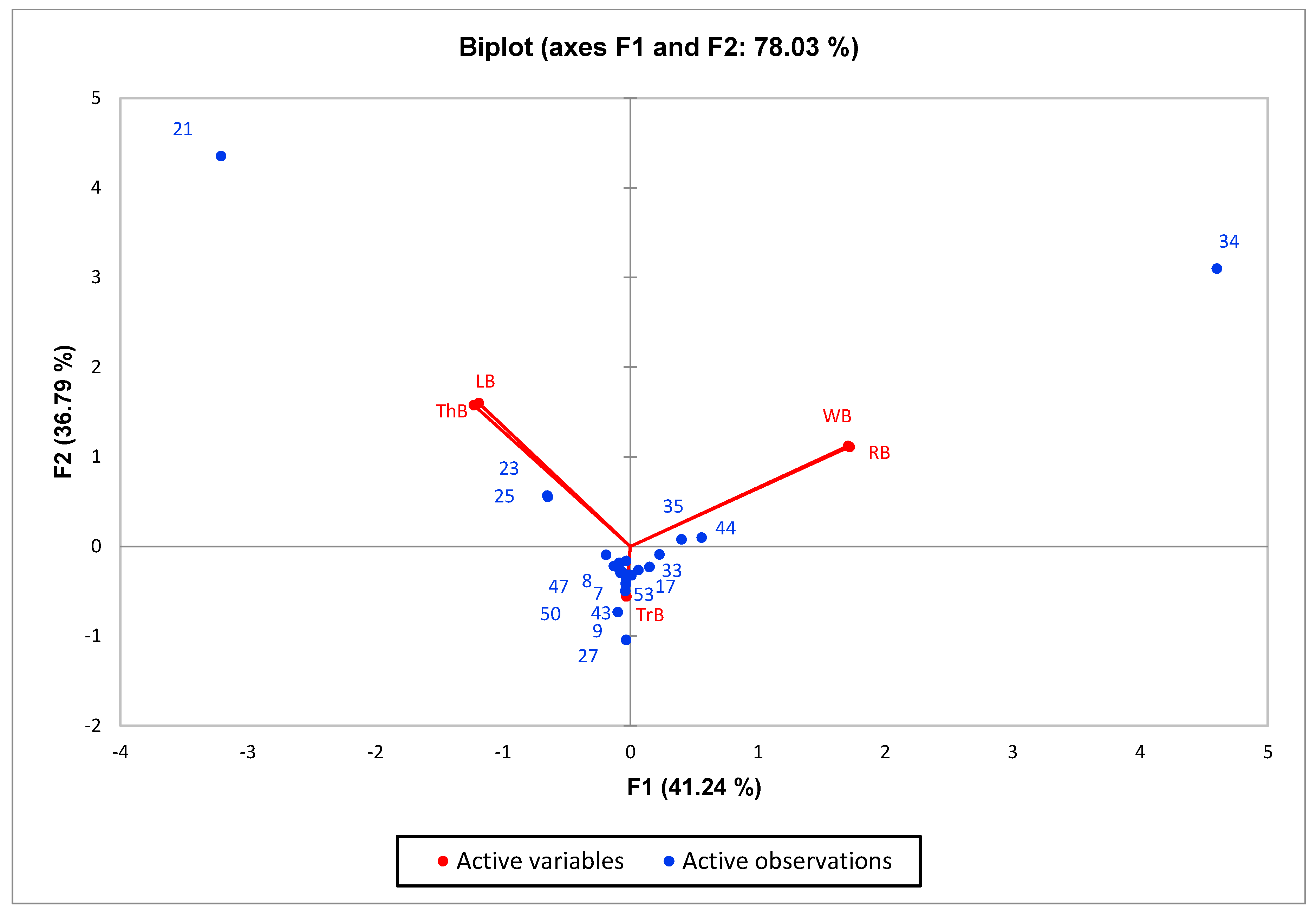
3.2. Chemical Profile of the Essential Oil
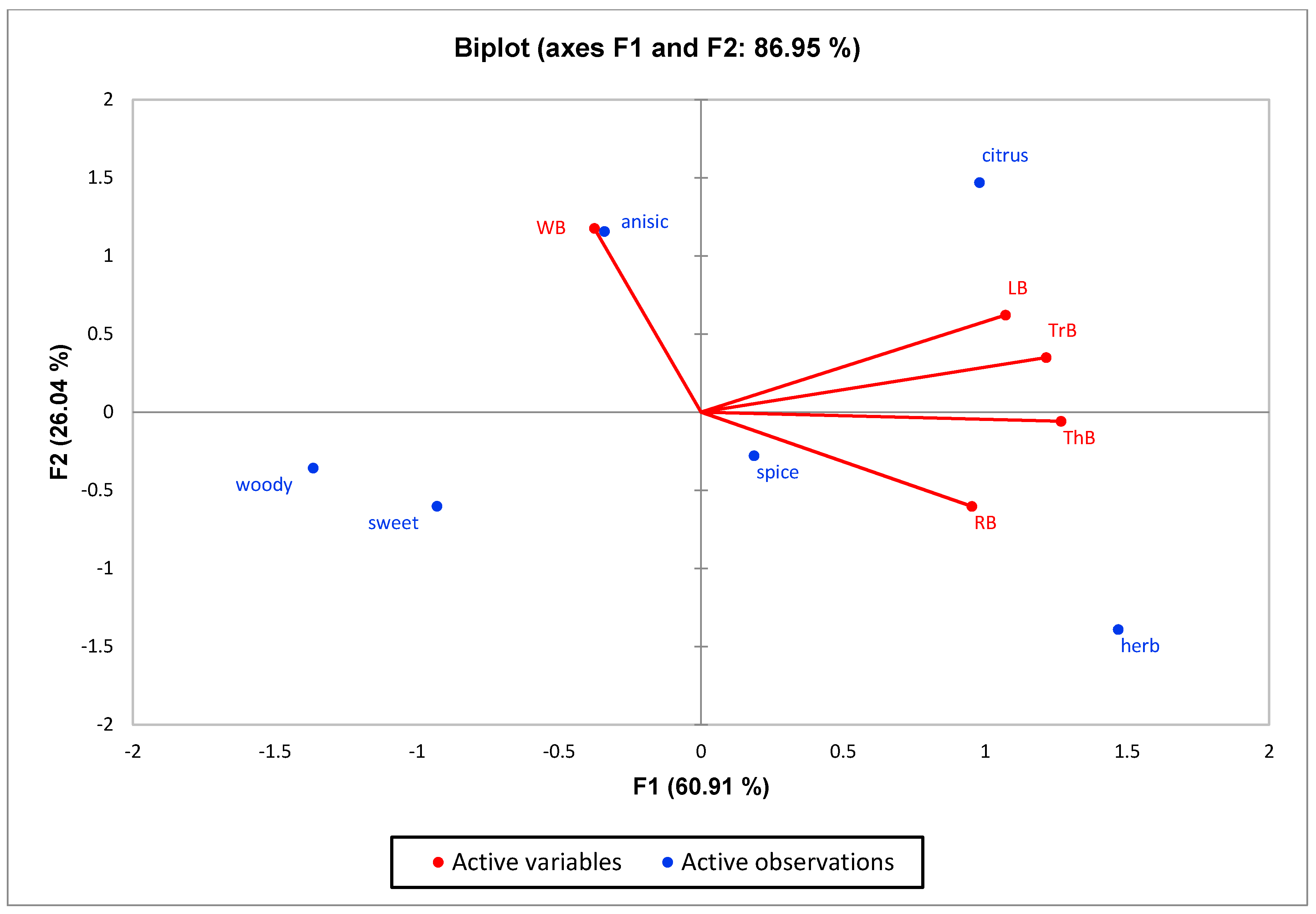
3.3. Sensory Profile of the Essential Oil
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pandey, A.K.; Singh, P.; Tripathi, N.N. Chemistry and bioactivities of essential oils of some Ocimum species: An overview. Asian Pac. J. Trop. Biomed. 2014, 4, 682–694. [Google Scholar] [CrossRef]
- Patel, R.P.; Singh, R.; Rao, B.R.R.; Singh, R.R.; Srivastava, A.; Lal, R.K. Differential response of genotype×environment on phenology, essential oil yield and quality of natural aroma chemicals of five Ocimum species. Ind. Crops Prod. 2016, 87, 210–217. [Google Scholar] [CrossRef]
- Juntachote, T.; Berghofer, E.; Siebenhandl, S.; Bauer, F. The antioxidative properties of holy basil and galangal in cooked ground pork. Meat Sci. 2006, 72, 446–456. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, N.; Rawal, S.; Verma, M.; Poddar, M.; Alok, S. A phytopharmacological overview on Ocimum species with special emphasis on Ocimum sanctum. Biomed. Prev. Nutr. 2013, 3, 185–192. [Google Scholar] [CrossRef]
- Siripongvutikorn, S.; Thummaratwasik, P.; Huang, Y.-W. Antimicrobial and antioxidation effects of thai seasoning, tom-yum. LWT—Food Sci. Technol. 2005, 38, 347–352. [Google Scholar] [CrossRef]
- Wandersleben, T.; Morales, E.; Burgos-Díaz, C.; Barahona, T.; Labra, E.; Rubilar, M.; Salvo-Garrido, H. Enhancement of functional and nutritional properties of bread using a mix of natural ingredients from novel varieties of flaxseed and lupine. LWT—Food Sci. Technol. 2018, 91, 48–54. [Google Scholar] [CrossRef]
- Burdock, G.A.; Wang, W. Our unrequited love for natural ingredients. Food Chem. Toxicol. 2017, 107, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Imen, T.; Olfa, B.; Jamel, H.; Luigi, C.P.; Mokhtar, L.; Guido, F.; Zeineb, O. Essential oil and volatile emissions of basil (Ocimum basilicum) leaves exposed to nacl or Na2SO4 salinity. J. Plant Nutr. Soil Sci. 2013, 176, 748–755. [Google Scholar]
- Kwee, E.M.; Niemeyer, E.D. Variations in phenolic composition and antioxidant properties among 15 basil (Ocimum basilicum L.) cultivars. Food Chem. 2011, 128, 1044–1050. [Google Scholar] [CrossRef]
- Jordán, M.J.; Quílez, M.; Luna, M.C.; Bekhradi, F.; Sotomayor, J.A.; Sánchez-Gómez, P.; Gil, M.I. Influence of water stress and storage time on preservation of the fresh volatile profile of three basil genotypes. Food Chem. 2017, 221, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, B.; Sipos, L.; Kókai, Z.; Gere, A.; Szabó, K.; Bernáth, J.; Sárosi, S. Comparison of different Ocimum basilicum L. Gene bank accessions analyzed by GC-MS and sensory profile. Ind. Crops Prod. 2015, 67, 498–508. [Google Scholar] [CrossRef]
- Muráriková, A.; Ťažký, A.; Neugebauerová, J.; Planková, A.; Jampílek, J.; Mučaji, P.; Mikuš, P. Characterization of essential oil composition in different basil species and pot cultures by a GC-MS method. Molecules 2017, 22, 1221. [Google Scholar] [CrossRef] [PubMed]
- Nurzyñska-Wierdak, R. Morphological variability and essential oil composition of four Ocimum basilicum L. Cultivars. J. Essent. Oil Bear. Pl. 2014, 17, 112–119. [Google Scholar] [CrossRef]
- Zheljazkov, V.D.; Cantrell, C.L.; Evans, W.B.; Ebelhar, M.W.; Coker, C. Yield and composition of Ocimum basilicum L. and Ocimum sanctum L. grown at four locations. HortScience 2008, 43, 737–741. [Google Scholar]
- Jofré Barud, F.; López, S.; Tapia, A.; Feresin, G.E.; López, M.L. Attractant, sexual competitiveness enhancing and toxic activities of the essential oils from Baccharis spartioides and Schinus polygama on Ceratitis capitata wiedemann. Ind. Crops Prod. 2014, 62, 299–304. [Google Scholar] [CrossRef]
- Barkatullah;I.M.;Rauf, A.; Inyat-Ur-Rahman, K. Physicochemical characterization of essential and fixed oils of Skimmia laureola and Zanthoxylum armatum. J. Med. Plant Res. 2012, 1, 51–58. [Google Scholar]
- Taraj, K.; Delibashi, A.; Andoni, A.; Lazo, P.; Kokalari, E.; Lame, A.; Xhaxhiu, K.; Çomo, A. Extraction of chamomile essential oil by subcritical CO2 and its analysis by UV-Vis spectrophotometer. Asian J. Chem. 2013, 25, 7361–7364. [Google Scholar]
- De Lira, C.S.; Pontual, E.V.; de Albuquerque, L.P.; Paiva, L.M.; Paiva, P.M.G.; de Oliveira, J.V.; Napoleão, T.H.; Navarro, D.M.d.A.F. Evaluation of the toxicity of essential oil from Alpinia purpurata inflorescences to Sitophilus zeamais (maize weevil). Crop Prot. 2015, 71, 95–100. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Quadrupole Mass Spectroscopy; Allured Pub. Corporation: Carol Stream, IL, USA, 2001. [Google Scholar]
- Pimentel, T.C.; Gomes da Cruz, A.; Deliza, R. Sensory evaluation: Sensory rating and scoring methods. In Encyclopedia of Food and Health; Caballero, B., Finglas, P.M., Toldrá, F., Eds.; Academic Press: Oxford, UK, 2016; pp. 744–749. [Google Scholar]
- Lee, J.; Chambers, D. A lexicon for flavor descriptive analysis of green tea. J. Sens. Stud. 2007, 22, 256–272. [Google Scholar] [CrossRef]
- Ledeker, C.N.; Suwonsichon, S.; Chambers, D.H.; Adhikari, K. Comparison of sensory attributes in fresh mangoes and heat-treated mango purées prepared from thai cultivars. LWT—Food Sci. Technol. 2014, 56, 138–144. [Google Scholar] [CrossRef]
- Chambers, E.; Sanchez, K.; Phan, U.X.T.; Miller, R.; Civille, G.V.; Di Donfrancesco, B. Development of a “living” lexicon for descriptive sensory analysis of brewed coffee. J. Sens. Stud. 2016, 31, 465–480. [Google Scholar] [CrossRef]
- Chowdhury, T.; Mandal, A.; Roy, S.C.; De Sarker, D. Diversity of the genus Ocimum (lamiaceae) through morpho-molecular (RAPD) and chemical (GC–MS) analysis. J. Genet. Eng. Biotechnol. 2017, 15, 275–286. [Google Scholar] [CrossRef]
- Özcan, M.; Chalchat, J.-C. Essential oil composition of Ocimum basilicum L. and Ocimum minimum L. in turkey. Czech J. Food Sci. 2002, 20, 223–228. [Google Scholar] [CrossRef]
- Sims, C.A.; Juliani, H.R.; Mentreddy, S.R.; Simon, J.E. Essential oils in holy basil (Ocimum tenuiflorum L.) as influenced by planting dates and harvest times in north alabama. J. Med. Active Pl. 2014, 2, 33–41. [Google Scholar]
- Siddique, A.B.; Mizanur Rahman, S.M.; Hossain, M.A. Chemical composition of essential oil by different extraction methods and fatty acid analysis of the leaves of Stevia rebaudiana bertoni. Arab. J. Chem. 2016, 9, S1185–S1189. [Google Scholar] [CrossRef]
- Turek, C.; Stintzing, F.C. Stability of essential oils: A review. Compr. Rev. Food Sci. Food Saf. 2013, 12, 40–53. [Google Scholar] [CrossRef]
- Hădărugă, D.I.; Hădărugă, N.G.; Costescu, C.I.; David, I.; Gruia, A.T. Thermal and oxidative stability of the Ocimum basilicum L. Essential oil/β-cyclodextrin supramolecular system. Beilstein J. Org. Chem. 2014, 10, 2809–2820. [Google Scholar] [CrossRef] [PubMed]
- Beltramea, J.M.; Angnesa, R.A.; Chiavellib, L.U.R.; Costab, W.F.d.; Rosac, M.F.d.; Loboa, V.d.S.; Pominib, A.M. Photodegradation of essential oil from marjoram (Origanum majorana L.) studied by GC-MS and UV-Vis spectroscopy. Rev. Latinoam. Quím. 2013, 41, 81–88. [Google Scholar]
- Le Borgne, E.; Cicchetti, E.; Bertrand, T. HPTLC methods for qualitative and quantitative analysis of selected furocoumarins in essential oils. Flavour Fragr. J. 2017, 32, 330–339. [Google Scholar] [CrossRef]
- Simon, J.E.; Morales, M.R.; Phippen, W.B.; Vieira, R.F.; Hao, Z. Basil: A source of aroma compounds and a popular culinary and ornamental herb. In Perspectives on New Crops and New Uses; Janick, J., Ed.; ASHS Press: Alexandria, VA, USA, 1999; pp. 499–505. [Google Scholar]
- Singh, D.; Chaudhuri, P.K. A review on phytochemical and pharmacological properties of holy basil (Ocimum sanctum L.). Ind. Crops Prod. 2018, 118, 367–382. [Google Scholar] [CrossRef]
- Dighe, V.V.; Gursale, A.A.; Sane, R.T.; Menon, S.; Patel, P.H. Quantitative determination of eugenol from Cinnamomum tamala nees and eberm. leaf powder and polyherbal formulation using reverse phase liquid chromatography. Chromatographia 2005, 61, 443–446. [Google Scholar] [CrossRef]
- Sajjadi, S.E. Analysis of the essential oils of two cultivated basil (Ocimum basilicum L.) from iran. DARU J. Pharm. Sci. 2006, 14, 128–130. [Google Scholar]
- Mondawi, B.; Duprey, R.; Magboul, A.; Satti, A. Constituents of essential oil of Ocimum basilicum var. Thyrsiflorum. Fitoterapia 1984, 55, 60–61. [Google Scholar]
- Khatri, L.; Nasir, M.; Saleem, R.; Noor, F. Evaluation of pakistani sweet basil oil for commercial exploitation. Pak. J. Sci. Ind. Res. 1995, 38, 281–282. [Google Scholar]
- Verma, R.S.; Bisht, P.S.; Padalia, R.C.; Saikia, D.; Chauhan, A. Chemical composition and antibacterial activity of essential oil from two Ocimum spp. grown in sub-tropical india during spring-summer cropping season. Asian J. Tradit. Med. 2011, 6, 211–217. [Google Scholar]
- Avetisyan, A.; Markosian, A.; Petrosyan, M.; Sahakyan, N.; Babayan, A.; Aloyan, S.; Trchounian, A. Chemical composition and some biological activities of the essential oils from basil Ocimum different cultivars. BMC Complement Altern. Med. 2017, 17, 60. [Google Scholar] [CrossRef] [PubMed]
- Mondello, L.; Zappia, G.; Cotroneo, A.; Bonaccorsi, I.; Chowdhury, J.U.; Yusuf, M.; Dugo, G. Studies on the essential oil-bearing plants of bangladesh. Part viii. Composition of some Ocimum oils O. basilicum L. Var. Purpurascens; O. sanctum L. Green; O. sanctum L. Purple; O. americanum L., citral type; O. americanum L., camphor type. Flavour Fragr. J. 2002, 17, 335–340. [Google Scholar] [CrossRef]
- Silva, M.G.V.; Matos, F.J.A.; Lopes, P.R.O.; Silva, F.O.; Holanda, M.T. Composition of essential oils from three Ocimum species obtained by steam and microwave distillation and supercritical CO2 extraction. Arkivoc 2004, 6, 66–71. [Google Scholar]
- Sastry, K.P.; Kumar, R.R.; Arigar Kumar, N.; Sneha, G.; Elizabeth, M. Morpho-chemical description and antimicrobial activity of different Ocimum species. J. Plant Dev. Sci. 2012, 19, 53–64. [Google Scholar]
- Awasthi, P.K.; Dixit, S.C. Chemical compositions of Ocimum sanctum shyama and Ocimum sanctum rama oils from the plains of northern india. J. Essent. Oil Bear. Pl. 2007, 10, 292–296. [Google Scholar] [CrossRef]
- Janssen, A.M.; Scheffer, J.J.C.; Ntezurubanza, L.; Svendsen, A.B. Antimicrobial activities of some Ocimum species grown in rwanda. J. Ethnopharmacol. 1989, 26, 57–63. [Google Scholar] [CrossRef]
- Zoghbi, M.D.G.B.; Oliveira, J.; Andrade, E.H.A.; Trigo, J.R.; Fonseca, R.C.M.; Rocha, A.E.S. Variation in volatiles of Ocimum campechianum mill. And Ocimum gratissimum L. cultivated in the north of brazil. J. Essent. Oil Bear. Pl. 2007, 10, 229–240. [Google Scholar] [CrossRef]
- Joshi, R.K.; Si, H. Chemical composition of the essential oil of Ocimum tenuiflorum L. (krishna tulsi) from north west karnataka, india. Plant Science Today 2014, 1, 99–102. [Google Scholar] [CrossRef]
- Rajabi, Z.; Ebrahimi, M.; Farajpour, M.; Mirza, M.; Ramshini, H. Compositions and yield variation of essential oils among and within nine Salvia species from various areas of Iran. Ind. Crops Prod. 2014, 61, 233–239. [Google Scholar] [CrossRef]
- Agostini, F.; Santos, A.C.A.d.; Rossato, M.; Pansera, M.R.; Santos, P.L.d.; Serafini, L.A.; Molon, R.; Moyna, P. Essential oil yield and composition of Lamiaceae species growing in southern Brazil. Braz. Arch. Biol. Technol. 2009, 52, 473–478. [Google Scholar] [CrossRef]
- Zouari, N.; Ayadi, I.; Fakhfakh, N.; Rebai, A.; Zouari, S. Variation of chemical composition of essential oils in wild populations of Thymus algeriensis boiss. Et reut., a North African endemic species. Lipids Health Dis. 2012, 11, 28. [Google Scholar] [CrossRef] [PubMed]
- Bagci, E.; Bekci, F. Variation in essential oil composition of Hypericum scabrum L. and H. scabroides N. Robson & Poulter (Hypericaceae) aerial parts during its phenological cycle. Acta Bot. Gall. 2010, 157, 247–254. [Google Scholar]
- Lan Phi, N.T.; Minh Tu, N.T.; Nishiyama, C.; Sawamura, M. Characterisation of the odour volatiles in Citrus aurantifolia persa lime oil from Vietnam. In Developments in Food Science; Bredie, W.L.P., Petersen, M.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2006; Volume 43, pp. 193–196. [Google Scholar]
- Xiao, Z.; Chen, J.; Niu, Y.; Chen, F. Characterization of the key odorants of fennel essential oils of different regions using GC–MS and GC–O combined with partial least squares regression. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2017, 1063, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Calín-Sánchez, Á.; Lech, K.; Szumny, A.; Figiel, A.; Carbonell-Barrachina, Á.A. Volatile composition of sweet basil essential oil (Ocimum basilicum L.) as affected by drying method. Food Res. Int. 2012, 48, 217–225. [Google Scholar] [CrossRef]
- Al-Kateb, H.; Mottram, D.S. The relationship between growth stages and aroma composition of lemon basil Ocimum citriodorum vis. Food Chem. 2014, 152, 440–446. [Google Scholar] [CrossRef] [PubMed]
| Odour Attributes | Reference Standard | n/15 | Reference |
|---|---|---|---|
| Anise | anise powder, 2 g | 10/15 | * |
| Citrus | lemon extract (McCormick), 200 µL | 8/15 | [21] |
| Herb | thyme (McCormick), 0.5 g | 10/15 | * |
| Spice | ground allspice (McCormick), 0.5 g | 8/15 | [22] |
| Sweet | vanilla flavour (McCormick), 200 µL | 10/15 | [23] |
| Woody | peanut peel 2 g with 100 µL DI water | 7/15 | * |
| Characteristics | LB | RB | ThB | TrB | WB |
|---|---|---|---|---|---|
| Plant morphology | |||||
| General | 50–105 cm tall | 70–150 cm tall | 45–100 cm tall | 140–200 cm tall | 70–160 cm tall |
| Leaf structure | leaf size ~3.5 × 1 cm, leaf elliptic-broadly obovate, glabrous except hairy midrib, veinlets and margin | leaf size ~4 × 1.5 cm, ovate-obovate, elliptic-oblong, surface patently hairy to clothed with soft spreading hair, Purple leaf | leaf size ~5.5 × 2 cm, leaf ovate-lanceolate to oblong-lanceolate, glabrous except hairy midrib, veinlets and margin | leaf size ~9 × 5 cm, leaf lanceolate, ovate or ovate-lanceolate, glabrous except hairy midrib | leaf size ~4 × 1.5 cm, leaf ovate-obovate, elliptic-oblong, surface patently hairy to clothed with soft spreading hair, green leaf |
| Inflorescenceand floral structure | inflorescence greenish, flowers white, calyx green, long hairy | inflorescence purple, flowers purplish, calyx purple, patently hairy to densely pubescent | inflorescence greenish, flowers whitish pink, calyx green, long hairy | inflorescence greenish purple, flowers yellowish white, calyx greenish purple, hairy | inflorescence green-greenish purple, flowers purplish, calyx green, patently hairy to densely pubescent |
| Seed characteristics | seed brownish black, ellipsoid, mucilaginous | seed brown, globose, non-mucilaginous | seed brownish black, ellipsoid, mucilaginous | seed brown, subglobose, non-mucilaginous | seed brown, globose, non-mucilaginous |
| Physiochemical Characteristics of essential oils | |||||
| Yield *,** | 0.37% ± 0.12 b | 0.33% ± 0.06 b | 0.43% ± 0.09 c | 0.19% ± 0.05 a | 0.47% ± 0.09 c |
| Colourunder day light | yellow | clear | yellow | orange | clear |
| Colourunder UV light*** | + | +++ | ++ | ++ | +++ |
| No. | Compounds | Retention Index | Retention Index * | Amount of Chemicals | ||||
|---|---|---|---|---|---|---|---|---|
| (µg/mL Essential Oils) ** | ||||||||
| LB | RB | ThB | TrB | WB | ||||
| 1 | methyl 2-methylbutanoate | - | - | nd | nd | nd | 0.89 | nd |
| 2 | 3-hexen-1-ol | 850 | - | nd | nd | nd | 0.79 | nd |
| 3 | α-pinene | 930 | 938 | nd | nd | nd | nd | 0.32 |
| 4 | camphene | 944 | - | nd | nd | nd | nd | 0.42 |
| 5 | β-pinene | 973 | 981 | 1.31 | nd | 2.58 | nd | 0.52 |
| 6 | 1-octen-3-ol | 979 | - | nd | nd | nd | 2.77 | nd |
| 7 | myrcene | 989 | 995 | nd | nd | nd | 5.13 | nd |
| 8 | 1,8-cineole | 1028 | 1034 | 2.82 | nd | 5.64 | nd | nd |
| 9 | α-ocimene | 1049 | 1046 | nd | nd | 13.0 | 257 | nd |
| 10 | z-ocimene | 1051 | - | 5.83 | nd | nd | 17.7 | nd |
| 11 | γ-terpinene | 1058 | - | 0.36 | nd | 2.39 | nd | nd |
| 12 | 3-carene | 1101 | - | nd | 1.76 | 14.61 | 22.3 | nd |
| 13 | linalool | 1104 | 1097 | 1.12 | nd | nd | nd | 1.09 |
| 14 | (4e,6z)-allo-ocimene | 1132 | - | nd | nd | nd | 14.9 | nd |
| 15 | d-camphor | 1145 | 1144 | 1.34 | nd | 3.54 | nd | nd |
| 16 | trans-chrysanthemal | 1152 | - | nd | nd | 2.20 | nd | nd |
| 17 | borneol | 1168 | - | nd | 7.79 | nd | nd | 2.80 |
| 18 | 1,4-heptadiene, 3-methyl- | 1169 | - | nd | nd | 5.54 | nd | nd |
| 19 | terpinen-4-ol | 1180 | - | 1.78 | nd | 15.4 | nd | nd |
| 20 | cyclohexane, ethenyl- | 1189 | - | nd | nd | 6.78 | nd | nd |
| 21 | estragole | 1211 | 1196 | 98.2 | nd | 453 | nd | nd |
| 22 | (r)-α-pinene | 1236 | - | nd | nd | 8.60 | nd | nd |
| 23 | neral | 1251 | 1238 | 6.32 | nd | 151 | nd | nd |
| 24 | (+) -(−)-3-carene | 1264 | - | nd | nd | 6.30 | nd | nd |
| 25 | geranial | 1282 | 1268 | nd | nd | 181 | nd | nd |
| 26 | citral | 1283 | - | 9.55 | nd | nd | nd | nd |
| 27 | eugenol | 1371 | 1361 | nd | nd | nd | 408 | 1.50 |
| 28 | α-copaene | 1379 | - | nd | 20.7 | nd | 16.1 | 4.74 |
| 29 | β-bourbonene | 1388 | 1383 | nd | 7.68 | nd | 1.88 | nd |
| 30 | 4-methylpyrazole | 1389 | - | nd | nd | 1.82 | nd | nd |
| 31 | n-butylpyrrole | 1391 | - | nd | nd | nd | nd | 2.25 |
| 32 | β-cubebene | 1396 | 1389 | nd | 1.98 | nd | 7.11 | nd |
| 33 | β-elemene | 1396 | 1391 | 0.77 | 65.7 | 4.30 | 3.65 | 2.79 |
| 34 | methyl eugenol | 1409 | 1411 | 0.77 | 684 | 2.87 | nd | 98.4 |
| 35 | β-caryophyllene | 1424 | 1420 | 1.34 | 146 | 10.6 | 17.3 | nd |
| 36 | α-bergamotene | 1439 | 1437 | 1.00 | nd | 4.49 | 0.69 | nd |
| 37 | (z,e)-α-farnesene | 1442 | - | nd | nd | nd | 51.6 | nd |
| 38 | α-guaiene | 1443 | 1439 | 0.19 | nd | nd | nd | nd |
| 39 | β-sesquiphellandrene | 1447 | - | nd | nd | nd | 0.59 | nd |
| 40 | α-humulene | 1458 | 1454 | 3.42 | 13.4 | 6.88 | 3.36 | 1.42 |
| 41 | bicyclo sesquiphellandrene | 1468 | - | 2.43 | nd | 10.8 | 0.59 | nd |
| 42 | germacrene d | 1469 | 1482 | 2.26 | 0.55 | 14.3 | 0.89 | 1.23 |
| 43 | γ-muurolene | 1490 | - | nd | nd | nd | 91.6 | nd |
| 44 | α-cubebene | 1490 | - | nd | 105 | nd | nd | 9.94 |
| 45 | bicyclo [3.1.1] hept-3-ene-spiro-2,4′-(1′,3′-dioxane), 7,7-dimethyl- | 1493 | - | nd | 1.87 | nd | nd | nd |
| 46 | bicyclogermacrene | 1500 | 1497 | 0.81 | nd | 3.63 | 3.55 | nd |
| 47 | β-gurjunene | 1501 | - | nd | 6.36 | nd | nd | nd |
| 48 | α-selinene | 1505 | - | nd | nd | nd | nd | 0.40 |
| 49 | α-bulnesene | 1509 | 1506 | 1.06 | nd | 3.92 | nd | nd |
| 50 | α-farnesene | 1511 | - | nd | nd | nd | 42.3 | nd |
| 51 | α-amorphene | 1518 | - | nd | nd | 2.58 | nd | nd |
| 52 | phenylethanolamine | 1519 | - | nd | nd | nd | 0.69 | nd |
| 53 | δ-cadinene | 1525 | 1524 | nd | 8.12 | nd | 11.1 | 0.56 |
| 54 | 1-bromo-8-heptadecyne | 1539 | - | 0.44 | nd | nd | nd | nd |
| 55 | (z)-4-decen-1-ol | 1539 | - | nd | 3.84 | nd | nd | nd |
| 56 | (z)-α-bisabolene | 1546 | 1544 | nd | nd | 11.1 | nd | nd |
| 57 | eremophilene | 1556 | - | nd | 2.52 | nd | nd | nd |
| 58 | elemol | 1557 | - | nd | nd | nd | nd | 0.23 |
| 59 | 4-ethylphenethylamine | 1582 | - | nd | nd | nd | 0.59 | nd |
| 60 | ethyl trichloroacetate | 1582 | - | nd | 1.75 | nd | nd | nd |
| 61 | benzofuran, 7-(2,4-dinitrophenoxy)-3-ethoxy-2,3-dihydro-2,2-dimethyl- | 1590 | - | nd | 0.99 | nd | nd | nd |
| 62 | 1,3-diisopropyl-1,3-cyclopentadiene | 1602 | - | nd | nd | nd | 0.99 | nd |
| 63 | cadina-1,4-diene | 1622 | - | nd | nd | 1.24 | nd | nd |
| 64 | naphthalene, 1,2,3,4,4a,7-hexahydro-1,6-dimethyl-4-(1-methylethyl)- | 1622 | - | 0.31 | nd | nd | nd | nd |
| 65 | bromoacetonitrile | 1650 | - | nd | nd | nd | 0.50 | nd |
| 66 | α-muurolene | 1663 | - | nd | 2.41 | nd | nd | nd |
| 67 | β-bisabolene | 1690 | - | 0.27 | nd | nd | nd | nd |
| Basils | Sweet | Herb | Woody | Anise | Citrus | Spice |
|---|---|---|---|---|---|---|
| LB | 3.4 ± 0.24 a | 7.2 ± 0.49 bc | 0.98 ± 0.02 a | 4.72 ± 0.70 bc | 12.8 ± 0.58 d | 5.2 ± 0.66 b |
| RB | 2.6 ± 0.51 a | 5.68 ± 1.14 b | 2.26 ± 0.55 bc | 3.6 ± 0.51 ab | 2.7 ± 0.37 a | 3.5 ± 0.67 a |
| ThB | 2.4 ± 0.24 a | 11.2 ± 0.86 d | 1.6 ± 0.33 ab | 2.34 ± 0.52 a | 10.62 ± 0.69 c | 6.4 ± 0.24 bc |
| TrB | 2.46 ± 0.63 a | 9.2 ± 0.80 cd | 1.8 ± 0.20 ab | 7.4 ± 0.40 d | 8.9 ± 0.56 b | 7.28 ± 0.42 c |
| WB | 2.42 ± 0.50 a | 1.1 ± 0.10 a | 3.34 ± 0.50 c | 5.96 ± 0.87 cd | 4.2 ± 0.37 a | 2.7 ± 0.44 a |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tangpao, T.; Chung, H.-H.; Sommano, S.R. Aromatic Profiles of Essential Oils from Five Commonly Used Thai Basils. Foods 2018, 7, 175. https://doi.org/10.3390/foods7110175
Tangpao T, Chung H-H, Sommano SR. Aromatic Profiles of Essential Oils from Five Commonly Used Thai Basils. Foods. 2018; 7(11):175. https://doi.org/10.3390/foods7110175
Chicago/Turabian StyleTangpao, Tibet, Hsiao-Hang Chung, and Sarana Rose Sommano. 2018. "Aromatic Profiles of Essential Oils from Five Commonly Used Thai Basils" Foods 7, no. 11: 175. https://doi.org/10.3390/foods7110175
APA StyleTangpao, T., Chung, H.-H., & Sommano, S. R. (2018). Aromatic Profiles of Essential Oils from Five Commonly Used Thai Basils. Foods, 7(11), 175. https://doi.org/10.3390/foods7110175






