Alginate-Based Edible Films and Coatings for Food Packaging Applications
Abstract
1. Introduction
- General information about alginate and gel formation
- Lists of additives incorporated into the alginate-based edible films and coatings in the literature
- Types of film production and coating application
- Sums up the research findings on alginate coated fruits-vegetables, meats, poultry, seafood, cheese
- Transport of the products’ molecular components
- Future trends
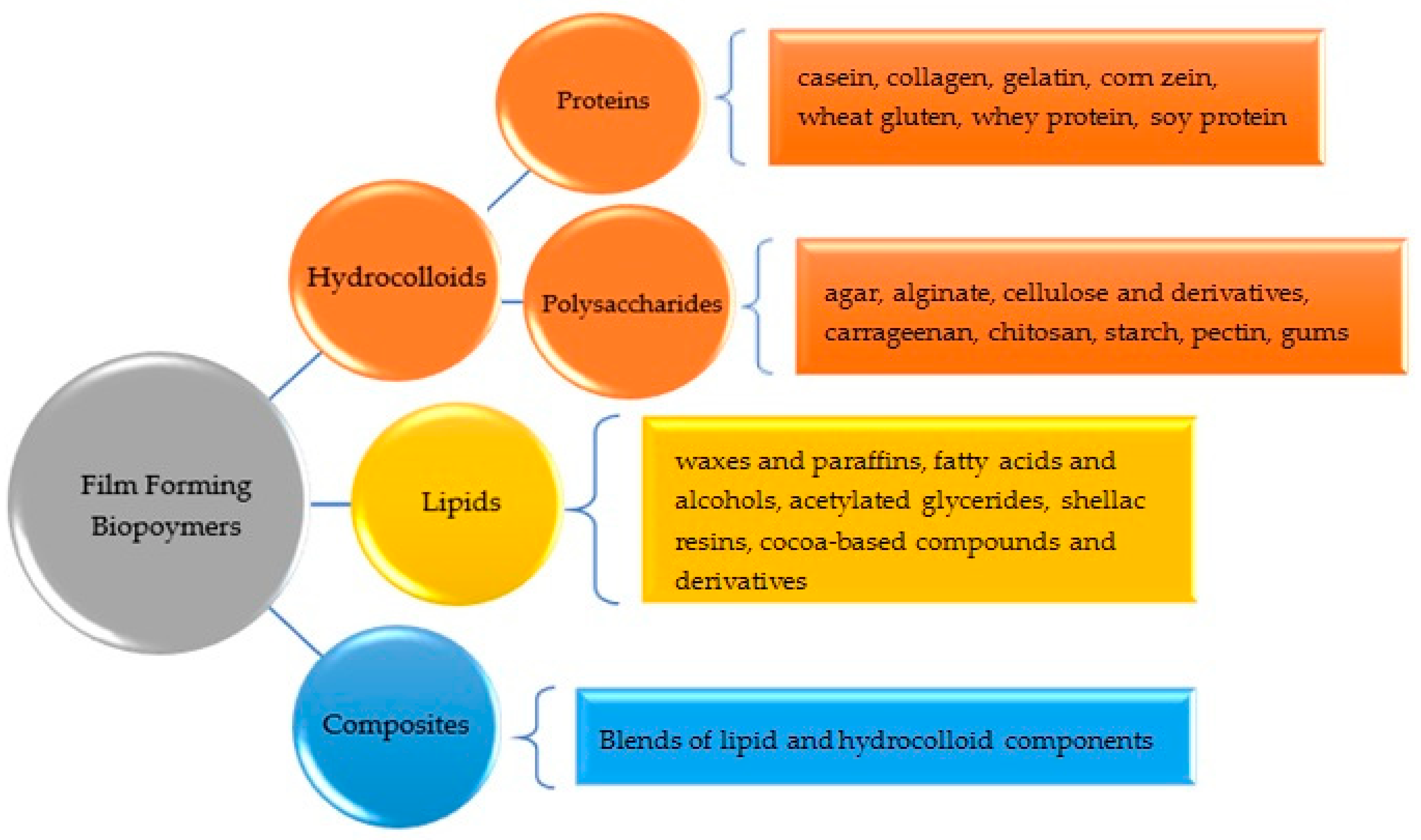
2. Film-Forming Materials
3. Alginate
4. Crosslinking
5. Additives
5.1. Plasticizers
5.2. Surfactants
5.3. Antimicrobials
5.4. Antioxidants
5.5. Antibrowning Agents
5.6. Flavors, Pigments, Nutritional Improvements
6. Application Methods
6.1. Film Formation
- Simple coacervation: The precipitation or phase change of the hydrocolloid, which is dispersed in water, is achieved following to (i) the solvent evaporation process (i.e., drying); (ii) incorporation of hydrosoluble non-electrolyte (in which the hydrocolloid is not soluble, e.g., ethanol); (iii) the pH adjustment with the addition of electrolyte, which impel salting out or cross-linking.
- Complex coacervation: The precipitation of the polymer complex is achieved by mixing two hydrocolloid solutions, which have opposite electron charges.
- Gelation or thermal coagulation: Precipitation or gelation is accomplished by heating of the macromolecule which causes its degradation (e.g., proteins such as ovalbumin) or the cooling of hydrocolloid dispersion (e.g., agar, gelatin).
6.1.1. Solvent Casting
6.1.2. Extrusion
6.2. Coating Application
6.2.1. Dipping
6.2.2. Spraying
6.2.3. Vacuum Impregnation
7. Alginate-Based Coatings and Film Applications
7.1. Fresh-Cut Fruits and Vegetables
7.2. Meats, Poultry, and Seafood
7.3. Cheese
7.4. Only Coating/Film, Without Food Application
8. Transport Mechanisms
8.1. Moisture Barrier Applications
8.2. Gaseous Barrier Applications
8.3. Active Compound Release Applications
9. Future Trends
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Marsh, K.; Bugusu, B. Food packaging—Roles, materials, and environmental issues. J. Food Sci. 2007, 72, R39–R55. [Google Scholar] [CrossRef] [PubMed]
- Coles, R. Introduction. In Food Packaging Technology; Coles, R., McDowell, D., Kirwan, M.J., Eds.; Blackwell Publishing: London, UK, 2003; pp. 1–31. [Google Scholar]
- The World Bank. Chapter 3 Waste Generation. Available online: https://siteresources.worldbank.org/INTURBANDEVELOPMENT/Resources/336387-1334852610766/Chap3.pdf (accessed on 18 September 2018).
- Risch, S.J. New developments in packaging materials. In Food Packaging; American Chemical Society: Washington, DC, USA, 2000; Volume 753, pp. 1–7. [Google Scholar]
- Ramos, Ó.L.; Reinas, I.; Silva, S.I.; Fernandes, J.C.; Cerqueira, M.A.; Pereira, R.N.; Vicente, A.A.; Poças, M.F.; Pintado, M.E.; Malcata, F.X. Effect of whey protein purity and glycerol content upon physical properties of edible films manufactured therefrom. Food Hydrocoll. 2013, 30, 110–122. [Google Scholar] [CrossRef]
- Gontard, N.; Guilbert, S. Bio-packaging: Technology and properties of edible and/or biodegradable material of agricultural origin. In Food Packaging and Preservation; Mathlouthi, M., Ed.; Blackie Academic and Professional: Glasgow, UK, 1994; pp. 159–181. [Google Scholar]
- Diab, T.; Biliaderis, C.G.; Gerasopoulos, D.; Sfakiotakis, E. Physicochemical properties and application of pullulan edible films and coatings in fruit preservation. J. Sci. Food Agric. 2001, 81, 988–1000. [Google Scholar] [CrossRef]
- Guilbert, S.; Gontard, N.; Cuq, B. Technology and applications of edible protective films. Packag. Technol. Sci. 1995, 8, 339–346. [Google Scholar] [CrossRef]
- Pavlath, A.E.; Orts, W. Edible films and coatings: Why, what, and how? In Edible Films and Coatings for Food Applications; Huber, K.C., Embuscado, M.E., Eds.; Springer New York: New York, NY, USA, 2009; pp. 1–23. [Google Scholar]
- Guilbert, S.; Gontard, N.; Gorris, L.G.M. Prolongation of the shelf-life of perishable food products using biodegradable films and coatings. LWT Food Sci. Technol. 1996, 29, 10–17. [Google Scholar] [CrossRef]
- Bourtoom, T. Edible films and coatings: Characteristics and properties. Int. Food Res. J. 2008, 15, 237–248. [Google Scholar]
- Donhowe, I.G.; Fennema, O. Edible films and coatings: Characteristics, formation, definitions, and testing methods. In Edible Coatings and Films to Improve Food Quality; Krochta, J.M., Baldwin, E.A., Nisperos-Carriedo, M.O., Eds.; Technomic Publ Co: Lancaster, NH, USA, 1994; pp. 1–24. [Google Scholar]
- Ncama, K.; Magwaza, L.; Mditshwa, A.; Zeray Tesfay, S. Plant-based edible coatings for managing postharvest quality of fresh horticultural produce: A review. Food Packag. Shelf Life 2018, 16, 157–167. [Google Scholar] [CrossRef]
- Murmu, S.B.; Mishra, H.N. The effect of edible coating based on arabic gum, sodium caseinate and essential oil of cinnamon and lemon grass on guava. Food Chem. 2018, 245, 820–828. [Google Scholar] [CrossRef] [PubMed]
- Shit, S.C.; Shah, P. Edible Polymers: Challenges and Opportunities. J. Polym. 2014, 2014, 1–13. [Google Scholar] [CrossRef]
- Dehghani, S.; Hosseini, S.V.; Regenstein, J.M. Edible films and coatings in seafood preservation: A review. Food Chem. 2018, 240, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Kramer, M.E. Structure and function of starch-based edible films and coatings. In Edible Films and Coatings for Food Applications; Huber, K.C., Embuscado, M.E., Eds.; Springer New York: New York, NY, USA, 2009; pp. 113–134. [Google Scholar]
- Sánchez-Ortega, I.; García-Almendárez, B.E.; Santos-López, E.M.; Amaro-Reyes, A.; Barboza-Corona, J.E.; Regalado, C. Antimicrobial edible films and coatings for meat and meat products preservation. Sci. World J. 2014, 2014, 18. [Google Scholar] [CrossRef] [PubMed]
- Zink, J.; Wyrobnik, T.; Prinz, T.; Schmid, M. Physical, chemical and biochemical modifications of protein-based films and coatings: An extensive review. Int. J. Mol. Sci. 2016, 17, 1376. [Google Scholar] [CrossRef] [PubMed]
- Vilgis, T.A. Hydrocolloids between soft matter and taste: Culinary polymer physics. Int. J. Gastron. Food Sci. 2012, 1, 46–53. [Google Scholar] [CrossRef]
- Mountney, G.J.; Winter, A.R. The use of a calcium alginate film for coating cut-up poultry. Poult. Sci. 1961, 40, 28–34. [Google Scholar] [CrossRef]
- Earle, R.D.; McKee, D.H. Process for Treating Fresh Meats. U.S. Patent 3991218, 9 November 1976. [Google Scholar]
- Lazarus, C.R.; West, R.L.; Oblinger, J.L.; Palmer, A.Z. Evaluation of a calcium alginate coating and a protective plastic wrapping for the control of lamb carcass shrinkage. J. Food Sci. 1976, 41, 639–641. [Google Scholar] [CrossRef]
- West, R.; Lazarus, C.; Oblinger, J.; Palmer, A. In Alginate Coatings for Carcasses. Available online: http://agris.fao.org/agris-search/search.do?recordID=US201303047669 (accessed on 18 September 2018).
- Embuscado, M.; Huber, K.C. Edible Films and Coatings for Food Applications; Springer: New York, NY, USA, 2009. [Google Scholar]
- Skurtys, O.; Acevedo, C.; Pedreschi, F.; Enrione, J.; Osorio, F.; Aguilera, J.M. Food hydrocolloid edible films and coatings. In Food Hydrocolloids Characteristics, Properties and Structures; Hollingworth, C.S., Ed.; Nova Science Publishers, Inc.: New York, NY, USA, 2010; pp. 41–80. [Google Scholar]
- Nisperos-Carriedo, M.O. Edible coatings and films based on polysaccharides. In Edible Coatings and Films to Improve Food Quality; Krochta, J.M., Baldwin, E.A., Nisperos-Carriedo, M.O., Eds.; Technomic Publ. Co.: Lancaster, NH, USA, 1994; Volume 1, pp. 322–323. [Google Scholar]
- Hay, I.D.; Rehman, Z.U.; Moradali, M.F.; Wang, Y.; Rehm, B.H.A. Microbial alginate production, modification and its applications. Microb. Biotechnol. 2013, 6, 637–650. [Google Scholar] [CrossRef] [PubMed]
- Emmerichs, N.; Wingender, J.; Flemming, H.C.; Mayer, C. Interaction between alginates and manganese cations: Identification of preferred cation binding sites. Int. J. Biol. Macromol. 2004, 34, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Smidsrød, O.; Skjåk-Bræk, G. Alginate as immobilization matrix for cells. Trends Biotechnol. 1990, 8, 71–78. [Google Scholar] [CrossRef]
- Evans, L.R.; Linker, A. Production and characterization of the slime polysaccharide of Pseudomonas aeruginosa. J. Bacteriol. 1973, 116, 915–924. [Google Scholar] [PubMed]
- Peteiro, C. Alginate production from marine macroalgae, with emphasis on kelp farming. In Alginates and Their Biomedical Applications; Rehm, B.H.A., Moradali, M.F., Eds.; Springer Singapore: Singapore, 2018; pp. 27–66. [Google Scholar]
- Clementi, F. Alginate production by Azotobacter vinelandii. Crit. Rev. Biotechnol. 1997, 17, 327–361. [Google Scholar] [CrossRef] [PubMed]
- Moe, S.; Skjåk-Bræk, G.; Smidsrød, O.; Ichijo, H. Calcium alginate gel fibers: Influence of alginate source and gel structure on fiber strength. J. Appl. Polym. Sci. 1994, 51, 1771–1775. [Google Scholar] [CrossRef]
- Draget, K.I. Alginates. In Handbook of Hydrocolloids, 2nd ed.; Woodhead Publishing: Sarston, UK, 2009; pp. 807–828. [Google Scholar]
- Anzai, H.; Uchida, N.; Nishide, E. Determination of d-mannuronic to l-guluronic acids ratio in acid hydrolysis of alginate under improved conditions. Nippon Suisan Gakkaishi 1990, 56, 73–81. [Google Scholar] [CrossRef]
- Davidson, I.; Sutherland, I.; Lawson, C. Localization of o-acetyl groups of bacterial alginate. Microbiology 1977, 98, 603–606. [Google Scholar] [CrossRef]
- Lee, P.; Rogers, M.A. Effect of calcium source and exposure-time on basic caviar spherification using sodium alginate. Int. J. Gastron. Food Sci. 2012, 1, 96–100. [Google Scholar] [CrossRef]
- Tapia, M.S.; Rojas-Graü, M.A.; Carmona, A.; Rodríguez, F.J.; Soliva-Fortuny, R.; Martin-Belloso, O. Use of alginate- and gellan-based coatings for improving barrier, texture and nutritional properties of fresh-cut papaya. Food Hydrocoll. 2008, 22, 1493–1503. [Google Scholar] [CrossRef]
- Martinsen, A.; Skjåk-Bræk, G.; Smidsrød, O. Alginate as immobilization material: I. Correlation between chemical and physical properties of alginate gel beads. Biotechnol. Bioeng. 1989, 33, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Younes, M.; Aggett, P.; Aguilar, F.; Crebelli, R.; Filipič, M.; Jose Frutos, M.; Galtier, P.; Gott, D.; Gundert-Remy, U.; Georg Kuhnle, G.; et al. Re-evaluation of alginic acid and its sodium, potassium, ammonium and calcium salts (e 400–e 404) as food additives. EFSA J. 2017, 15, 5049. [Google Scholar]
- Lu, J.-W.; Zhu, Y.-L.; Guo, Z.-X.; Hu, P.; Yu, J. Electrospinning of sodium alginate with poly(ethylene oxide). Polymer 2006, 47, 8026–8031. [Google Scholar] [CrossRef]
- Sime, W.J. Alginates. In Food Gels; Harris, P., Ed.; Springer: Dordrecht, The Netherlands, 1990; pp. 53–78. [Google Scholar]
- United States Department of Agriculture—Agricultural Marketing Service. Alginates Hadling/Processing. Available online: https://www.ams.usda.gov/sites/default/files/media/Alginates%20TR%202015.pdf (accessed on 19 June 2018).
- Kimica Corporation. How to Use Alginates. Available online: https://www.kimica.jp/en/alginate/usage/ (accessed on 19 June 2018).
- U.S. Food & Drug Administration. Code for Federal Regulations Title 21 Part 184—Direct Food Substances Affirmed as Generally Recognized as Safe. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=184.1724 (accessed on 5 April 2018).
- Kim, Y.J.; Yoon, K.J.; Ko, S.W. Preparation and properties of alginate superabsorbent filament fibers crosslinked with glutaraldehyde. J. Appl. Polym. Sci. 2000, 78, 1797–1804. [Google Scholar] [CrossRef]
- Yoo, S.; Krochta, J.M. Whey protein–polysaccharide blended edible film formation and barrier, tensile, thermal and transparency properties. J. Sci. Food Agric. 2011, 91, 2628–2636. [Google Scholar] [CrossRef] [PubMed]
- Kohn, R. Ion binding on polyuronates-alginate and pectin. Pure Appl. Chem. 1975, 42, 371–397. [Google Scholar] [CrossRef]
- Sutherland, I.W. Alginates. In Biomaterials: Novel Materials from Biological Sources, 1st ed.; Byrom, D., Ed.; Palgrave Macmillan: Basingstoke, UK, 1991; pp. 307–331. [Google Scholar]
- Gombotz, W.R.; Wee, S. Protein release from alginate matrices. Adv. Drug Deliv. Rev. 1998, 31, 267–285. [Google Scholar] [CrossRef]
- Soazo, M.; Báez, G.; Barboza, A.; Busti, P.A.; Rubiolo, A.; Verdini, R.; Delorenzi, N.J. Heat treatment of calcium alginate films obtained by ultrasonic atomizing: Physicochemical characterization. Food Hydrocoll. 2015, 51, 193–199. [Google Scholar] [CrossRef]
- Mancini, F.; McHugh, T.H. Fruit-alginate interactions in novel restructured products. Food/Nahrung 2000, 44, 152–157. [Google Scholar] [CrossRef]
- Grant, G.T.; Morris, E.R.; Rees, D.A.; Smith, P.J.C.; Thom, D. Biological interactions between polysaccharides and divalent cations: The egg-box model. FEBS Lett. 1973, 32, 195–198. [Google Scholar] [CrossRef]
- Morris, E.R.; Rees, D.A.; Thom, D. Characterization of polysaccharide structure and interactions by circular dichroism: Order-disorder transition in the calcium alginate system. J. Chem. Soc. Chem. Commun. 1973, 245–246. [Google Scholar] [CrossRef]
- Seifert, D.B.; Phillips, J.A. Production of small, monodispersed alginate beads for cell immobilization. Biotechnol. Prog. 1997, 13, 562–568. [Google Scholar] [CrossRef]
- Kierstan, M.; Darcy, G.; Reilly, J. Studies on the characteristics of alginate gels in relation to their use in separation and immobilized applications. Biotechnol. Bioeng. 1982, 24, 1507–1517. [Google Scholar] [CrossRef] [PubMed]
- Olivas, G.I.; Barbosa-Cánovas, G.V. Alginate–calcium films: Water vapor permeability and mechanical properties as affected by plasticizer and relative humidity. LWT Food Sci. Technol. 2008, 41, 359–366. [Google Scholar] [CrossRef]
- Papajová, E.; Bujdoš, M.; Chorvát, D.; Stach, M.; Lacík, I. Method for preparation of planar alginate hydrogels by external gelling using an aerosol of gelling solution. Carbohydr. Polym. 2012, 90, 472–482. [Google Scholar] [CrossRef] [PubMed]
- Chan, L.W.; Lee, H.Y.; Heng, P.W.S. Mechanisms of external and internal gelation and their impact on the functions of alginate as a coat and delivery system. Carbohydr. Polym. 2006, 63, 176–187. [Google Scholar] [CrossRef]
- Liu, X.D.; Yu, W.Y.; Zhang, Y.; Xue, W.M.; Yu, W.T.; Xiong, Y.; Ma, X.J.; Chen, Y.; Yuan, Q. Characterization of structure and diffusion behaviour of ca-alginate beads prepared with external or internal calcium sources. J. Microencapsul. 2002, 19, 775–782. [Google Scholar] [CrossRef] [PubMed]
- Gennadios, A.; Hanna, M.A.; Kurth, L.B. Application of edible coatings on meats, poultry and seafoods: A review. LWT Food Sci. Technol. 1997, 30, 337–350. [Google Scholar] [CrossRef]
- Ustunol, Z. Edible films and coatings for meat and poultry. In Edible Films and Coatings for Food Applications; Huber, K.C., Embuscado, M.E., Eds.; Springer New York: New York, NY, USA, 2009; pp. 245–268. [Google Scholar]
- Chrastil, J. Gelation of calcium alginate. Influence of rice starch or rice flour on the gelation kinetics and on the final gel structure. J. Agric. Food Chem. 1991, 39, 874–876. [Google Scholar] [CrossRef]
- Draget, K.I.; Østgaard, K.; Smidsrød, O. Alginate-based solid media for plant tissue culture. Appl. Microbiol. Biotechnol. 1989, 31, 79–83. [Google Scholar] [CrossRef]
- Draget, K.I.; Østgaard, K.; Smidsrød, O. Homogeneous alginate gels: A technical approach. Carbohydr. Polym. 1990, 14, 159–178. [Google Scholar] [CrossRef]
- Kuo, C.K.; Ma, P.X. Ionically crosslinked alginate hydrogels as scaffolds for tissue engineering: Part 1. Structure, gelation rate and mechanical properties. Biomaterials 2001, 22, 511–521. [Google Scholar] [CrossRef]
- Kuo, C.K.; Ma, P.X. Maintaining dimensions and mechanical properties of ionically crosslinked alginate hydrogel scaffolds in vitro. J. Biomed. Mater. Res. Part A 2008, 84A, 899–907. [Google Scholar] [CrossRef] [PubMed]
- Pavlath, A.E.; Gossett, C.; Camirand, W.; Robertson, G.H. Ionomeric films of alginic acid. J. Food Sci. 1999, 64, 61–63. [Google Scholar] [CrossRef]
- Rhim, J.-W. Physical and mechanical properties of water resistant sodium alginate films. LWT Food Sci. Technol. 2004, 37, 323–330. [Google Scholar] [CrossRef]
- Zactiti, E.M.; Kieckbusch, T.G. Release of potassium sorbate from active films of sodium alginate crosslinked with calcium chloride. Packag. Technol. Sci. 2009, 22, 349–358. [Google Scholar] [CrossRef]
- Howard, L.R.; Burma, P.; Wagner, A.B. Firmness and cell wall characteristics of pasteurized jalapeño pepper rings affected by calcium chloride and acetic acid. J. Food Sci. 1994, 59, 1184–1186. [Google Scholar] [CrossRef]
- Howard, L.R.; Buescher, R.W. Cell wall characteristics and firmness of fresh pack cucumber pickles affected by pasteurization and calcium chloride. J. Food Biochem. 1990, 14, 31–43. [Google Scholar] [CrossRef]
- Poovaiah, B. Role of calcium in prolonging storage life of fruits and vegetables. Food Technol. 1986, 40, 86–89. [Google Scholar]
- Luna-Guzmán, I.; Cantwell, M.; Barrett, D.M. Fresh-cut cantaloupe: Effects of cacl2 dips and heat treatments on firmness and metabolic activity. Postharvest Biol. Technol. 1999, 17, 201–213. [Google Scholar] [CrossRef]
- Banker, G.S. Film coating theory and practice. J. Pharm. Sci. 1966, 55, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Gontard, N.; Guilbert, S.; Cuq, J.-L. Water and glycerol as plasticizers affect mechanical and water vapor barrier properties of an edible wheat gluten film. J. Food Sci. 1993, 58, 206–211. [Google Scholar] [CrossRef]
- Barreto, P.L.M.; Pires, A.T.N.; Soldi, V. Thermal degradation of edible films based on milk proteins and gelatin in inert atmosphere. Polym. Degrad. Stab. 2003, 79, 147–152. [Google Scholar] [CrossRef]
- Rojas-Graü, M.A.; Tapia, M.S.; Martín-Belloso, O. Using polysaccharide-based edible coatings to maintain quality of fresh-cut fuji apples. LWT Food Sci. Technol. 2008, 41, 139–147. [Google Scholar] [CrossRef]
- Jost, V.; Kobsik, K.; Schmid, M.; Noller, K. Influence of plasticiser on the barrier, mechanical and grease resistance properties of alginate cast films. Carbohydr. Polym. 2014, 110, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Parris, N.; Coffin, D.R.; Joubran, R.F.; Pessen, H. Composition factors affecting the water vapor permeability and tensile properties of hydrophilic films. J. Agric. Food Chem. 1995, 43, 1432–1435. [Google Scholar] [CrossRef]
- Rojas-Graü, M.A.; Tapia, M.S.; Rodríguez, F.J.; Carmona, A.J.; Martin-Belloso, O. Alginate and gellan-based edible coatings as carriers of antibrowning agents applied on fresh-cut fuji apples. Food Hydrocoll. 2007, 21, 118–127. [Google Scholar] [CrossRef]
- Azarakhsh, N.; Osman, A.; Tan, C.P.; Mohd Ghazali, H.; Mohd Adzahan, N. Optimization of alginate and gellan-based edible coating formulations for fresh-cut pineapples. Int. Food Res. J. 2012, 19, 279–285. [Google Scholar]
- Park, H.J. Development of advanced edible coatings for fruits. Trends Food Sci. Technol. 1999, 10, 254–260. [Google Scholar] [CrossRef]
- Souza, B.W.S.; Cerqueira, M.A.; Teixeira, J.A.; Vicente, A.A. The use of electric fields for edible coatings and films development and production: A review. Food Eng. Rev. 2010, 2, 244–255. [Google Scholar] [CrossRef]
- Ribeiro, C.; Vicente, A.A.; Teixeira, J.A.; Miranda, C. Optimization of edible coating composition to retard strawberry fruit senescence. Postharvest Biol. Technol. 2007, 44, 63–70. [Google Scholar] [CrossRef]
- Rodríguez, M.; Osés, J.; Ziani, K.; Maté, J.I. Combined effect of plasticizers and surfactants on the physical properties of starch based edible films. Food Res. Int. 2006, 39, 840–846. [Google Scholar] [CrossRef]
- Senturk Parreidt, T.; Schott, M.; Schmid, M.; Müller, K. Effect of presence and concentration of plasticizers, vegetable oils, and surfactants on the properties of sodium-alginate-based edible coatings. Int. J. Mol. Sci. 2018, 19, 742. [Google Scholar] [CrossRef] [PubMed]
- Zapata, P.J.; Guillén, F.; Martínez-Romero, D.; Castillo, S.; Valero, D.; Serrano, M. Use of alginate or zein as edible coatings to delay postharvest ripening process and to maintain tomato (Solanum lycopersicon Mill) quality. J. Sci. Food Agric. 2008, 88, 1287–1293. [Google Scholar] [CrossRef]
- Díaz-Mula, H.M.; Serrano, M.; Valero, D. Alginate coatings preserve fruit quality and bioactive compounds during storage of sweet cherry fruit. Food Bioprocess Technol. 2012, 5, 2990–2997. [Google Scholar] [CrossRef]
- Valero, D.; Díaz-Mula, H.M.; Zapata, P.J.; Guillén, F.; Martínez-Romero, D.; Castillo, S.; Serrano, M. Effects of alginate edible coating on preserving fruit quality in four plum cultivars during postharvest storage. Postharvest Biol. Technol. 2013, 77, 1–6. [Google Scholar] [CrossRef]
- Duan, J.; Wu, R.; Strik, B.C.; Zhao, Y. Effect of edible coatings on the quality of fresh blueberries (duke and elliott) under commercial storage conditions. Postharvest Biol. Technol. 2011, 59, 71–79. [Google Scholar] [CrossRef]
- Hao, R.; Liu, Y.; Sun, L.; Xia, L.; Jia, H.; Li, Q.; Pan, J. Sodium alginate coating with plant extract affected microbial communities, biogenic amine formation and quality properties of abalone (Haliotis discus hannai ino) during chill storage. LWT Food Sci. Technol. 2017, 81, 1–9. [Google Scholar] [CrossRef]
- Fan, Y.; Xu, Y.; Wang, D.; Zhang, L.; Sun, J.; Sun, L.; Zhang, B. Effect of alginate coating combined with yeast antagonist on strawberry (fragaria × ananassa) preservation quality. Postharvest Biol. Technol. 2009, 53, 84–90. [Google Scholar] [CrossRef]
- Su Cha, D.; Choi, J.H.; Chinnan, M.S.; Park, H.J. Antimicrobial films based on na-alginate and κ-carrageenan. LWT Food Sci. Technol. 2002, 35, 715–719. [Google Scholar] [CrossRef]
- Baldwin, E.; Nisperos-Carriedo, M.; Baker, R. Edible coatings for lightly processed fruits and vegetables. HortScience 1995, 30, 35–38. [Google Scholar]
- Porter, M.R. Handbook of Surfactants; Blackie: Glasgow, Scotland, 1991. [Google Scholar]
- Vojdani, F.; Torres, J.A. Potassium sorbate permeability of methylcellulose and hydroxypropyl methylcellulose coatings: Effect of fatty acids. J. Food Sci. 1990, 55, 841–846. [Google Scholar] [CrossRef]
- Appendini, P.; Hotchkiss, J.H. Review of antimicrobial food packaging. Innov. Food Sci. Emerg. Technol. 2002, 3, 113–126. [Google Scholar] [CrossRef]
- Quintavalla, S.; Vicini, L. Antimicrobial food packaging in meat industry. Meat Sci. 2002, 62, 373–380. [Google Scholar] [CrossRef]
- Burt, S.A.; Reinders, R.D. Antibacterial activity of selected plant essential oils against Escherichia coli o157:H7. Letter. Appl. Microbiol. 2003, 36, 162–167. [Google Scholar] [CrossRef]
- Dorman, H.J.D.; Deans, S.G. Antimicrobial agents from plants: Antibacterial activity of plant volatile oils. J. Appl. Microbiol. 2000, 88, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. Antimicrobial activity of essential oils and other plant extracts. J. Appl. Microbiol. 1999, 86, 985–990. [Google Scholar] [CrossRef] [PubMed]
- Smith-Palmer, A.; Stewart, J.; Fyfe, L. Antimicrobial properties of plant essential oils and essences against five important food-borne pathogens. Lett. Appl. Microbiol. 1998, 26, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Cagri, A.; Ustunol, Z.; Ryser, E.T. Antimicrobial edible films and coatings. J. Food Prot. 2004, 67, 833–848. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Graü, M.A.; Raybaudi-Massilia, R.M.; Soliva-Fortuny, R.C.; Avena-Bustillos, R.J.; McHugh, T.H.; Martín-Belloso, O. Apple puree-alginate edible coating as carrier of antimicrobial agents to prolong shelf-life of fresh-cut apples. Postharvest Biol. Technol. 2007, 45, 254–264. [Google Scholar] [CrossRef]
- Sarengaowa; Hu, W.; Jiang, A.; Xiu, Z.; Feng, K. Effect of thyme oil–alginate-based coating on quality and microbial safety of fresh-cut apples. J. Sci. Food Agric. 2018, 98, 2302–2311. [Google Scholar] [CrossRef] [PubMed]
- Raybaudi-Massilia, R.M.; Mosqueda-Melgar, J.; Martín-Belloso, O. Edible alginate-based coating as carrier of antimicrobials to improve shelf-life and safety of fresh-cut melon. Int. J. Food Microbiol. 2008, 121, 313–327. [Google Scholar] [CrossRef] [PubMed]
- Raybaudi-Massilia, R.M.; Rojas-Graü, M.A.; Mosqueda-Melgar, J.; Martin-Belloso, O. Comparative study on essential oils incorporated into an alginate-based edible coating to assure the safety and quality of fresh-cut fuji apples. J. Food Prot. 2008, 71, 1150–1161. [Google Scholar] [CrossRef] [PubMed]
- Sipahi, R.E.; Castell-Perez, M.E.; Moreira, R.G.; Gomes, C.; Castillo, A. Improved multilayered antimicrobial alginate-based edible coating extends the shelf life of fresh-cut watermelon (Citrullus lanatus). LWT Food Sci. Technol. 2013, 51, 9–15. [Google Scholar] [CrossRef]
- Azarakhsh, N.; Osman, A.; Ghazali, H.M.; Tan, C.P.; Mohd Adzahan, N. Lemongrass essential oil incorporated into alginate-based edible coating for shelf-life extension and quality retention of fresh-cut pineapple. Postharvest Biol. Technol. 2014, 88, 1–7. [Google Scholar] [CrossRef]
- Peretto, G.; Du, W.-X.; Avena-Bustillos, R.J.; Berrios, J.D.J.; Sambo, P.; McHugh, T.H. Optimization of antimicrobial and physical properties of alginate coatings containing carvacrol and methyl cinnamate for strawberry application. J. Agric. Food Chem. 2014, 62, 984–990. [Google Scholar] [CrossRef] [PubMed]
- Nair, M.S.; Saxena, A.; Kaur, C. Characterization and antifungal activity of pomegranate peel extract and its use in polysaccharide-based edible coatings to extend the shelf-life of capsicum (Capsicum annuum L.). Food Bioprocess Technol. 2018, 11, 1317–1327. [Google Scholar] [CrossRef]
- Williams, S.K.; Oblinger, J.L.; West, R.L. Evaluation of a calcium alginate film for use on beef cuts. J. Food Sci. 1978, 43, 292–296. [Google Scholar] [CrossRef]
- Fang, T.J.; Tsai, H.-C. Growth patterns of Escherichia coli o157:H7 in ground beef treated with nisin, chelators, organic acids and their combinations immobilized in calcium alginate gels. Food Microbiol. 2003, 20, 243–253. [Google Scholar] [CrossRef]
- Cutter, C.N.; Siragusa, G.R. Growth of Brochothrix thermosphactain ground beef following treatments with nisin in calcium alginate gels. Food Microbiol. 1997, 14, 425–430. [Google Scholar] [CrossRef]
- Siragusa, G.R.; Dickson, J.S. Inhibition of Listeria monocytogenes on beef tissue by application of organic acids immobilized in a calcium alginate gel. J. Food Sci. 1992, 57, 293–296. [Google Scholar] [CrossRef]
- Siragusa, G.R.; Dickson, J.S. Inhibition of Bisteria monocytogenes, Salmonella typhimurium and Escherichia coli 0157:H7 on beef muscle tissue by lactic or acetic acid contained in calcium alginate gels. J. Food Saf. 1993, 13, 147–158. [Google Scholar] [CrossRef]
- Hamedi, H.; Kargozari, M.; Shotorbani, P.M.; Mogadam, N.B.; Fahimdanesh, M. A novel bioactive edible coating based on sodium alginate and galbanum gum incorporated with essential oil of Ziziphora persica: The antioxidant and antimicrobial activity, and application in food model. Food Hydrocoll. 2017, 72, 35–46. [Google Scholar] [CrossRef]
- Yousefi, M.; Farshidi, M.; Ehsani, A. Effects of lactoperoxidase system-alginate coating on chemical, microbial, and sensory properties of chicken breast fillets during cold storage. J. Food Saf. 2018, 38, e12449. [Google Scholar] [CrossRef]
- Molayi, R.; Ehsani, A.; Yousefi, M. The antibacterial effect of whey protein–alginate coating incorporated with the lactoperoxidase system on chicken thigh meat. Food Sci. Nutr. 2018, 6, 878–883. [Google Scholar] [CrossRef] [PubMed]
- Lu, F.; Liu, D.; Ye, X.; Wei, Y.; Liu, F. Alginate–calcium coating incorporating nisin and edta maintains the quality of fresh northern snakehead (Channa argus) fillets stored at 4 °C. J. Sci. Food Agric. 2009, 89, 848–854. [Google Scholar] [CrossRef]
- Concha-Meyer, A.; Schöbitz, R.; Brito, C.; Fuentes, R. Lactic acid bacteria in an alginate film inhibit Listeria monocytogenes growth on smoked salmon. Food Control 2011, 22, 485–489. [Google Scholar] [CrossRef]
- Neetoo, H.; Ye, M.; Chen, H. Bioactive alginate coatings to control Listeria monocytogenes on cold-smoked salmon slices and fillets. Int. J. Food Microbiol. 2010, 136, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Ye, M.; Neetoo, H.; Chen, H. Prior frozen storage enhances the effect of edible coatings against Listeria monocytogenes on cold-smoked salmon during subsequent refrigerated storage. J. Appl. Microbiol. 2011, 111, 865–876. [Google Scholar] [CrossRef] [PubMed]
- Datta, S.; Janes, M.E.; Xue, Q.G.; Losso, J.; La Peyre, J.F. Control of Listeria monocytogenes and Salmonella anatum on the surface of smoked salmon coated with calcium alginate coating containing oyster lysozyme and nisin. J. Food Sci. 2008, 73, M67–M71. [Google Scholar] [CrossRef] [PubMed]
- Bazargani-Gilani, B. Activating sodium alginate-based edible coating using a dietary supplement for increasing the shelf life of rainbow trout fillet during refrigerated storage (4 ± 1 °C). J. Food Saf. 2018, 38, e12395. [Google Scholar] [CrossRef]
- Jalali, N.; Ariiai, P.; Fattahi, E. Effect of alginate/carboxyl methyl cellulose composite coating incorporated with clove essential oil on the quality of silver carp fillet and Escherichia coli o157:H7 inhibition during refrigerated storage. J. Food Sci. Technol. 2016, 53, 757–765. [Google Scholar] [CrossRef] [PubMed]
- Heydari, R.; Bavandi, S.; Javadian, S.R. Effect of sodium alginate coating enriched with horsemint (Mentha longifolia) essential oil on the quality of bighead carp fillets during storage at 4 °C. Food Sci. Nutr. 2015, 3, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Field, C.E.; Pivarnik, L.F.; Barnett, S.M.; Rand, A.G. Utilization of glucose oxidase for extending the shelf-life of fish. J. Food Sci. 1986, 51, 66–70. [Google Scholar] [CrossRef]
- Nie, X.; Wang, L.; Wang, Q.; Lei, J.; Hong, W.; Huang, B.; Zhang, C. Effect of a sodium alginate coating infused with tea polyphenols on the quality of fresh japanese sea bass (Lateolabrax japonicas) fillets. J. Food Sci. 2018, 83, 1695–1700. [Google Scholar] [CrossRef] [PubMed]
- Cai, L.; Cao, A.; Bai, F.; Li, J. Effect of ε-polylysine in combination with alginate coating treatment on physicochemical and microbial characteristics of japanese sea bass (Lateolabrax japonicas) during refrigerated storage. LWT Food Sci. Technol. 2015, 62, 1053–1059. [Google Scholar] [CrossRef]
- Cai, L.; Wang, Y.; Cao, A.; Lv, Y.; Li, J. Effect of alginate coating enriched with 6-gingerol on the shelf life and quality changes of refrigerated red sea bream (Pagrosomus major) fillets. RSC Adv. 2015, 5, 36882–36889. [Google Scholar] [CrossRef]
- Angiolillo, L.; Conte, A.; Del Nobile, M.A. A new method to bio-preserve sea bass fillets. Int. J. Food Microbiol. 2018, 271, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Angiolillo, L.; Conte, A.; Zambrini, A.V.; Del Nobile, M.A. Biopreservation of fior di latte cheese. J. Dairy Sci. 2014, 97, 5345–5355. [Google Scholar] [CrossRef] [PubMed]
- Kavas, N.; Kavas, G.; Saygili, D. Use of ginger essential oil-fortified edible coatings in kashar cheese and its effects on Escherichia coli o157:H7 and Staphylococcus aureus. CyTA J. Food 2016, 14, 317–323. [Google Scholar] [CrossRef]
- Lucera, A.; Mastromatteo, M.; Conte, A.; Zambrini, A.V.; Faccia, M.; Del Nobile, M.A. Effect of active coating on microbiological and sensory properties of fresh mozzarella cheese. Food Packag. Shelf Life 2014, 1, 25–29. [Google Scholar] [CrossRef]
- Artiga-Artigas, M.; Acevedo-Fani, A.; Martín-Belloso, O. Improving the shelf life of low-fat cut cheese using nanoemulsion-based edible coatings containing oregano essential oil and mandarin fiber. Food Control 2017, 76, 1–12. [Google Scholar] [CrossRef]
- Bustos, R.; Alberti, F.; Matiacevich, S. Edible antimicrobial films based on microencapsulated lemongrass oil. J. Food Sci. Technol. 2016, 53, 832–839. [Google Scholar] [CrossRef] [PubMed]
- Zactiti, E.M.; Kieckbusch, T.G. Potassium sorbate permeability in biodegradable alginate films: Effect of the antimicrobial agent concentration and crosslinking degree. J. Food Eng. 2006, 77, 462–467. [Google Scholar] [CrossRef]
- Alboofetileh, M.; Rezaei, M.; Hosseini, H.; Abdollahi, M. Antimicrobial activity of alginate/clay nanocomposite films enriched with essential oils against three common foodborne pathogens. Food Control 2014, 36, 1–7. [Google Scholar] [CrossRef]
- Pranoto, Y.; Salokhe, V.M.; Rakshit, S.K. Physical and antibacterial properties of alginate-based edible film incorporated with garlic oil. Food Res. Int. 2005, 38, 267–272. [Google Scholar] [CrossRef]
- Han, Y.; Wang, L. Sodium alginate/carboxymethyl cellulose films containing pyrogallic acid: Physical and antibacterial properties. J. Sci. Food Agric. 2017, 97, 1295–1301. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food & Drug Administration. Code of Federal Regulations Title 21 Part 170—Food Additives. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfCFR/CFRSearch.cfm?fr=170.3&SearchTerm=170%2E3 (accessed on 10 April 2018).
- Shahidi, F. Antioxidants in food and food antioxidants. Food/Nahrung 2000, 44, 158–163. [Google Scholar] [CrossRef]
- Nair, M.S.; Saxena, A.; Kaur, C. Effect of chitosan and alginate based coatings enriched with pomegranate peel extract to extend the postharvest quality of guava (Psidium guajava L.). Food Chem. 2018, 240, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Oms-Oliu, G.; Soliva-Fortuny, R.; Martín-Belloso, O. Edible coatings with antibrowning agents to maintain sensory quality and antioxidant properties of fresh-cut pears. Postharvest Biol. Technol. 2008, 50, 87–94. [Google Scholar] [CrossRef]
- Amanatidou, A.; Slump, R.A.; Gorris, L.G.M.; Smid, E.J. High oxygen and high carbon dioxide modified atmospheres for shelf-life extension of minimally processed carrots. J. Food Sci. 2000, 65, 61–66. [Google Scholar] [CrossRef]
- Wu, Y.; Weller, C.L.; Hamouz, F.; Cuppett, S.; Schnepf, M. Moisture loss and lipid oxidation for precooked ground-beef patties packaged in edible starch-alginate-based composite films. J. Food Sci. 2001, 66, 486–493. [Google Scholar] [CrossRef]
- Chidanandaiah; Keshri, R.C.; Sanyal, M.K. Effect of sodium alginate coating with preservatives on the quality of meat patties during refrigerated (4 ± 1c) storage. J. Muscle Foods 2009, 20, 275–292. [Google Scholar] [CrossRef]
- Handley, D.; Ma-Edmonds, M.; Hamouz, F.; Cuppett, S.; Mandigo, R.; Schnepf, M. Controlling oxidation and warmed-over flavor in precooked pork chops with rosemary oleoresin and edible film. In Natural Antioxidants Chemistry, Health Effects, and Applications; Shahidi, F., Ed.; AOCS Press: Champaign, IL, USA, 1997; pp. 311–318. [Google Scholar]
- Song, Y.; Liu, L.; Shen, H.; You, J.; Luo, Y. Effect of sodium alginate-based edible coating containing different anti-oxidants on quality and shelf life of refrigerated bream (Megalobrama amblycephala). Food Control 2011, 22, 608–615. [Google Scholar] [CrossRef]
- Ehsani, A.; Paktarmani, M.; Yousefi, M. Efficiency of dietary sodium alginate coating incorporated with lycopene in preserving rainbow trout. Food Sci. Biotechnol. 2017, 26, 557–562. [Google Scholar] [CrossRef] [PubMed]
- Norajit, K.; Kim, K.M.; Ryu, G.H. Comparative studies on the characterization and antioxidant properties of biodegradable alginate films containing ginseng extract. J. Food Eng. 2010, 98, 377–384. [Google Scholar] [CrossRef]
- Dong, X.; Wrolstad, R.E.; Sugar, D. Extending shelf life of fresh-cut pears. J. Food Sci. 2000, 65, 181–186. [Google Scholar] [CrossRef]
- Rocha, A.M.; Morais, A.M. Polyphenoloxidase activity and total phenolic content as related to browning of minimally processed ‘jonagored’ apple. J. Sci. Food Agric. 2002, 82, 120–126. [Google Scholar] [CrossRef]
- Martinez, M.V.; Whitaker, J.R. The biochemistry and control of enzymatic browning. Trends Food Sci. Technol. 1995, 6, 195–200. [Google Scholar] [CrossRef]
- Montero-Calderón, M.; Rojas-Graü, M.A.; Martín-Belloso, O. Effect of packaging conditions on quality and shelf-life of fresh-cut pineapple (Ananas comosus). Postharvest Biol. Technol. 2008, 50, 182–189. [Google Scholar] [CrossRef]
- Hambleton, A.; Debeaufort, F.; Bonnotte, A.; Voilley, A. Influence of alginate emulsion-based films structure on its barrier properties and on the protection of microencapsulated aroma compound. Food Hydrocoll. 2009, 23, 2116–2124. [Google Scholar] [CrossRef]
- Hambleton, A.; Voilley, A.; Debeaufort, F. Transport parameters for aroma compounds through i-carrageenan and sodium alginate-based edible films. Food Hydrocoll. 2011, 25, 1128–1133. [Google Scholar] [CrossRef]
- Tapia, M.S.; Rojas-Graü, M.A.; Rodríguez, F.J.; Ramírez, J.; Carmona, A.; Martin-Belloso, O. Alginate- and gellan-based edible films for probiotic coatings on fresh-cut fruits. J. Food Sci. 2007, 72, E190–E196. [Google Scholar] [CrossRef] [PubMed]
- The EU Vegetable Oil; Proteinmeal Industry (FEDIOL). Nutrition Factsheet-Composition and Quality of Vegetable Oils and Fats Sunflower Oil. Available online: http://www.fediol.be/data/1327055786Fiche%20sunf_20_01.pdf (accessed on 15 April 2018).
- Centre for the Promotion of Imports from Developing Countries (CBI). Cbi Product Factsheet: Sunflower Oil in Europe. Available online: https://www.cbi.eu/sites/default/files/market_information/researches/product-factsheet-europe-sunflower-oil-2015.pdf (accessed on 15 April 2018).
- EFSA (European Food Safety Authority). Opinion of the scientific panel on dietetic products, nutrition and allergies [nda] related to nutrition claims concerning omega-3 fatty acids, monounsaturated fat, polyunsaturated fat and unsaturated fat. EFSA J. 2005, 3, 29. [Google Scholar]
- Ramana Rao, T.V.; Baraiya, N.S.; Vyas, P.B.; Patel, D.M. Composite coating of alginate-olive oil enriched with antioxidants enhances postharvest quality and shelf life of ber fruit (Ziziphus mauritiana Lamk. Var. Gola). J. Food Sci. Technol. 2016, 53, 748–756. [Google Scholar] [CrossRef] [PubMed]
- Rößle, C.; Brunton, N.; Gormley, R.T.; Wouters, R.; Butler, F. Alginate coating as carrier of oligofructose and inulin and to maintain the quality of fresh-cut apples. J. Food Sci. 2011, 76, H19–H29. [Google Scholar] [CrossRef] [PubMed]
- Dhanapal, A.; Rajamani, L.; Shakila Banu, M. Edible films from polysaccharides. Food Sci. Qual. Manag. 2012, 3, 9–17. [Google Scholar]
- Lin, D.; Zhao, Y. Innovations in the development and application of edible coatings for fresh and minimally processed fruits and vegetables. Compr. Rev. Food Sci. Food Saf. 2007, 6, 60–75. [Google Scholar] [CrossRef]
- Ukai, N.; Ishibashi, S.; Tsutsumi, T.; Marakami, K. Preservation of Agricultural Products. U.S. Patent 3997674, 14 December 1976. [Google Scholar]
- Nussinovitch, A. Hydrocolloids for coatings and adhesives. In Handbook of Hydrocolloids, 2nd ed.; Woodhead Publishing: Shaston, UK, 2009; pp. 760–806. [Google Scholar]
- Cisneros-Zevallos, L.; Krochta, J.M. Dependence of coating thickness on viscosity of coating solution applied to fruits and vegetables by dipping method. J. Food Sci. 2003, 68, 503–510. [Google Scholar] [CrossRef]
- Andrade, R.D.; Skurtys, O.; Osorio, F.A. Atomizing spray systems for application of edible coatings. Compr. Rev. Food Sci. Food Saf. 2012, 11, 323–337. [Google Scholar] [CrossRef]
- Weiss, J.; Takhistov, P.; McClements, D.J. Functional materials in food nanotechnology. J. Food Sci. 2006, 71, R107–R116. [Google Scholar] [CrossRef]
- Poverenov, E.; Danino, S.; Horev, B.; Granit, R.; Vinokur, Y.; Rodov, V. Layer-by-layer electrostatic deposition of edible coating on fresh cut melon model: Anticipated and unexpected effects of alginate–chitosan combination. Food Bioprocess Technol. 2014, 7, 1424–1432. [Google Scholar] [CrossRef]
- Arnon-Rips, H.; Poverenov, E. Improving food products’ quality and storability by using layer by layer edible coatings. Trends Food Sci. Technol. 2018, 75, 81–92. [Google Scholar] [CrossRef]
- Tharanathan, R.N. Biodegradable films and composite coatings: Past, present and future. Trends Food Sci. Technol. 2003, 14, 71–78. [Google Scholar] [CrossRef]
- Norton, T.; Sun, D.-W. Computational fluid dynamics (cfd)—An effective and efficient design and analysis tool for the food industry: A review. Trends Food Sci. Technol. 2006, 17, 600–620. [Google Scholar] [CrossRef]
- Guillemin, A.; Degraeve, P.; Noël, C.; Saurel, R. Influence of impregnation solution viscosity and osmolarity on solute uptake during vacuum impregnation of apple cubes (var. Granny smith). J. Food Eng. 2008, 86, 475–483. [Google Scholar] [CrossRef]
- Vargas, M.; Chiralt, A.; Albors, A.; González-Martínez, C. Effect of chitosan-based edible coatings applied by vacuum impregnation on quality preservation of fresh-cut carrot. Postharvest Biol. Technol. 2009, 51, 263–271. [Google Scholar] [CrossRef]
- Senturk Parreidt, T.; Schmid, M.; Müller, K. Effect of dipping and vacuum impregnation coating techniques with alginate based coating on physical quality parameters of cantaloupe melon. J. Food Sci. 2018, 83, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Del Rosario, B.A.; Beuchat, L.R. Survival and growth of enterohemorrhagic Escherichia coli o157:H7 in cantaloupe and watermelon. J. Food Prot. 1995, 58, 105–107. [Google Scholar] [CrossRef]
- Baldwin, E.A. Edible coatings for fresh fruits and vegetables: Past, present, and future. In Edible Coatings and Films to Improve Food Quality; Krochta, J.M., Baldwin, E.A., Nisperos-Carriedo, M.O., Eds.; Technomic Publ Co: Lancaster, NH, USA, 1994; pp. 25–64. [Google Scholar]
- Olivas, G.I.; Barbosa-Cánovas, G.V. Edible coatings for fresh-cut fruits. Crit. Rev. Food Sci. Nutr. 2005, 45, 657–670. [Google Scholar] [CrossRef] [PubMed]
- Wong, D.W.; Tillin, S.J.; Hudson, J.S.; Pavlath, A.E. Gas exchange in cut apples with bilayer coatings. J. Agric. Food Chem. 1994, 42, 2278–2285. [Google Scholar] [CrossRef]
- Soliva-Fortuny, R.C.; Martin-Belloso, O. New advances in extending the shelf-life of fresh-cut fruits: A review. Trends Food Sci. Technol. 2003, 14, 341–353. [Google Scholar] [CrossRef]
- Forney, C.F.; Mattheis, J.P.; Baldwin, E.A. Effects on flavor. In Modified and Controlled Atmosphere for the Storage, Transpiration and Packaging of Horticultural Commodities; Yahia, E.M., Ed.; CRC Press: Boca Raton, FL, USA, 2009; pp. 119–158. [Google Scholar]
- Watada, A.E.; Qi, L. Quality of fresh-cut produce. Postharvest Biol. Technol. 1999, 15, 201–205. [Google Scholar] [CrossRef]
- Perez-Gago, M.B.; Rojas, C.; Del Río, M.A. Effect of hydroxypropyl methylcellulose-lipid edible composite coatings on plum (cv. Autumn giant) quality during storage. J. Food Sci. 2003, 68, 879–883. [Google Scholar]
- Ghavidel, R.A.; Davoodi, M.G.; Asl, A.F.A.; Tanoori, T.; Sheykholeslami, Z. Effect of selected edible coatings to extend shelf-life of fresh-cut apples. Int. J. Agric. Crop Sci. 2013, 6, 1171. [Google Scholar]
- Moldão-Martins, M.; Beirão-da-Costa, S.M.; Beirão-da-Costa, M.L. The effects of edible coatings on postharvest quality of the “bravo de esmolfe” apple. Eur. Food Res. Technol. 2003, 217, 325–328. [Google Scholar] [CrossRef]
- Oms-Oliu, G.; Soliva-Fortuny, R.; Martín-Belloso, O. Using polysaccharide-based edible coatings to enhance quality and antioxidant properties of fresh-cut melon. LWT Food Sci. Technol. 2008, 41, 1862–1870. [Google Scholar] [CrossRef]
- Li, L.; Sun, J.; Gao, H.; Shen, Y.; Li, C.; Yi, P.; He, X.; Ling, D.; Sheng, J.; Li, J.; et al. Effects of polysaccharide-based edible coatings on quality and antioxidant enzyme system of strawberry during cold storage. Int. J. Polym. Sci. 2017, 2017, 8. [Google Scholar] [CrossRef]
- Ahmed, W.; Butt, M.; Sharif, M.; Shahid, M. Comparative estimation of alginate and soy based coatings on ph and vitamin c contents of strawberry (Fragaria ananassa L.) at controlled climate chamber. J. Food Process. Technol. 2013, 4, 2. [Google Scholar]
- Chiabrando, V.; Giacalone, G. Quality evaluation of blueberries coated with chitosan and sodium alginate during postharvest storage. Int. Food Res. J. 2017, 24, 1553–1561. [Google Scholar]
- Moraes, K.S.D.; Fagundes, C.; Melo, M.C.; Andreani, P.; Monteiro, A.R. Conservation of williams pear using edible coating with alginate and carrageenan. Food Sci. Technol. 2012, 32, 679–684. [Google Scholar] [CrossRef]
- Salinas-Roca, B.; Guerreiro, A.; Welti-Chanes, J.; Antunes, M.D.C.; Martín-Belloso, O. Improving quality of fresh-cut mango using polysaccharide-based edible coatings. Int. J. Food Sci. Technol. 2018, 53, 938–945. [Google Scholar] [CrossRef]
- Maftoonazad, N.; Ramaswamy, H.S.; Marcotte, M. Shelf-life extension of peaches through sodium alginate and methyl cellulose edible coatings. Int. J. Food Sci. Technol. 2008, 43, 951–957. [Google Scholar] [CrossRef]
- Amaral, R.D.A.; Achaerandio, I.; Benedetti, B.C.; Pujolà, M. The influence of edible coatings, blanching and ultrasound treatments on quality attributes and shelf-life of vacuum packaged potato strips. LWT Food Sci. Technol. 2017, 85, 449–455. [Google Scholar] [CrossRef]
- Nussinovitch, A.; Hershko, V. Gellan and alginate vegetable coatings. Carbohydr. Polym. 1996, 30, 185–192. [Google Scholar] [CrossRef]
- Tay, S.L.; Perera, C.O. Effect of 1-methylcyclopropene treatment and edible coatings on the quality of minimally processed lettuce. J. Food Sci. 2004, 69, fct131–fct135. [Google Scholar] [CrossRef]
- Pringle, T.D.; Johnson, L.P.; Bernkopf, D.K.; Williams, S.E. Factors affecting purge losses in portion controlled steaks. Foodserv. Res. Int. 1996, 9, 93–105. [Google Scholar] [CrossRef]
- Ambardekar, A.A. Effects of edible coatings on the moisture content and lipid oxidation of pink salmon (Oncorhynchus gorbuscha) fillets during three months of frozen storage. Asian Fish. Sci. 2007, 20, 395. [Google Scholar]
- Earle, R.D.; Snyder, C.E. Method of Preparing Frozen Seafood. U.S. Patent 3,255,021, 7 June 1966. [Google Scholar]
- Earle, R.D. Method of Preserving Foods by Coating Same. U.S. Patent 3,395,024, 30 July 1968. [Google Scholar]
- Wanstedt, K.G.; Seideman, S.C.; Donnelly, L.S.; Quenzer, N.M. Sensory attributes of precooked, calcium alginate-coated pork patties. J. Food Prot. 1981, 44, 732–735. [Google Scholar] [CrossRef]
- Helgerud, O.; Olsen, A. Block Freezing. U.S. Patent 2,763,557, 18 September 1956. [Google Scholar]
- Koushki, M.R.; Azizi, M.H.; Koohy-Kamaly, P.; Azizkhani, M. Effect of calcium alginate edible coatings on microbial and chemical properties of lamb meat during refrigerated storage. J. Food Qual. Hazards Conrol 2015, 2, 6–10. [Google Scholar]
- Koushki, M.; Azizi, M.H.; Koohy-Kamaly, P.; Amiri, Z.; Azizkhani, M. Effect of Calcium Alginate Coating on Shelf Life of Frozen Lamb Muscle. Available online: http://journals.sbmu.ac.ir/jps/article/view/8115/0 (accessed on 15 April 2018).
- Yu, X.L.; Li, X.B.; Xu, X.L.; Zhou, G.H. Coating with sodium alginate and its effects on the functional properties and structure of frozen pork. J. Muscle Foods 2008, 19, 333–351. [Google Scholar] [CrossRef]
- Liu, L.; Kerry, J.F.; Kerry, J.P. Selection of optimum extrusion technology parameters in the manufacture of edible/biodegradable packaging films derived from food-based polymers. J. Food Agric. Environ. 2005, 3, 51. [Google Scholar]
- Liu, L.; Kerry, J.F.; Kerry, J.P. Effect of food ingredients and selected lipids on the physical properties of extruded edible films/casings. Int. J. Food Sci. Technol. 2006, 41, 295–302. [Google Scholar] [CrossRef]
- Liu, L.; Kerry, J.F.; Kerry, J.P. Application and assessment of extruded edible casings manufactured from pectin and gelatin/sodium alginate blends for use with breakfast pork sausage. Meat Sci. 2007, 75, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Martínez, O.; Salmerón, J.; Epelde, L.; Vicente, M.S.; de Vega, C. Quality enhancement of smoked sea bass (Dicentrarchus labrax) fillets by adding resveratrol and coating with chitosan and alginate edible films. Food Control 2018, 85, 168–176. [Google Scholar] [CrossRef]
- Seyfzadeh, M.; Motalebi, A.; Kakoolaki, S.; Gholipour, H. Chemical, microbiological and sensory evaluation of gutted kilka coated with whey protein based edible film incorporated with sodium alginate during frozen storage. Iran. J. Fish. Sci. 2013, 12, 140–153. [Google Scholar]
- Rockower, R.K.; Deng, J.C.; Otwell, W.S.; Cornell, J.A. Effect of soy flour, soy protein concentrate and sodium alginate on the textural attributes of minced fish patties. J. Food Sci. 1983, 48, 1048–1052. [Google Scholar] [CrossRef]
- Costa, M.J.; Maciel, L.C.; Teixeira, J.A.; Vicente, A.A.; Cerqueira, M.A. Use of edible films and coatings in cheese preservation: Opportunities and challenges. Food Res. Int. 2018, 107, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Cavender, G.; Zhao, Y. Investigation of different coating application methods on the performance of edible coatings on mozzarella cheese. LWT Food Sci. Technol. 2014, 56, 1–8. [Google Scholar] [CrossRef]
- Imeson, A.P.; Ledward, D.A.; Mitchell, J.R. On the nature of the interaction between some anionic polysaccharides and proteins. J. Sci. Food Agric. 1977, 28, 661–668. [Google Scholar] [CrossRef]
- Shih, F.F. Interaction of soy isolate with polysaccharide and its effect on film properties. J. Am. Oil Chem. Soc. 1994, 71, 1281–1285. [Google Scholar] [CrossRef]
- Rhim, J.W.; Wu, Y.; Weller, C.L.; Schnepf, M. Physical characteristics of a composite film of soy protein isolate and propyleneglycol alginate. J. Food Sci. 1999, 64, 149–152. [Google Scholar] [CrossRef]
- Coughlan, K.; Shaw, N.B.; Kerry, J.F.; Kerry, J.P. Combined effects of proteins and polysaccharides on physical properties of whey protein concentrate-based edible films. J. Food Sci. 2004, 69, E271–E275. [Google Scholar] [CrossRef]
- González-Cuello, R.E.; Mogollón, O.F.C.; Berrio-Guzmán, O.J.; Cuevas-Martinez, C.M. Optimization of the Mechanical Properties of Biofilm Based on Alginate-gellan Plasticized with Glycerol. Available online: http://www.m-hikari.com/ces/ces2018/ces17-20-2018/p/rodriguezCES17-20-2018-2.pdf (accessed on 15 April 2018).
- Azeredo, H.M.C.; Magalhães, U.S.; Oliveira, S.A.; Ribeiro, H.L.; Brito, E.S.; De Moura, M.R. Tensile and water vapour properties of calcium-crosslinked alginate-cashew tree gum films. Int. J. Food Sci. Technol. 2012, 47, 710–715. [Google Scholar] [CrossRef]
- Pei, H.N.; Chen, X.G.; Li, Y.; Zhou, H.Y. Characterization and ornidazole release in vitro of a novel composite film prepared with chitosan/poly(vinyl alcohol)/alginate. J. Biomed. Mater. Res. Part A 2008, 85A, 566–572. [Google Scholar] [CrossRef] [PubMed]
- Pauly, J.; Gröger, H.; Patel, A.V. Design, characterisation and application of alginate-based encapsulated pig liver esterase. J. Biotechnol. 2018, 280, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Acevedo, C.A.; López, D.A.; Tapia, M.J.; Enrione, J.; Skurtys, O.; Pedreschi, F.; Brown, D.I.; Creixell, W.; Osorio, F. Using rgb image processing for designing an alginate edible film. Food Bioprocess Technol. 2012, 5, 1511–1520. [Google Scholar] [CrossRef]
- Kokoszka, S.; Lenart, A. Edible coatings-formation, characteristics and use-a review. Pol. J. Food Nutr. Sci. 2007, 57, 399–404. [Google Scholar]
- Buonocore, G.G.; Del Nobile, M.A.; Di Martino, C.; Gambacorta, G.; La Notte, E.; Nicolais, L. Modeling the water transport properties of casein-based edible coating. J. Food Eng. 2003, 60, 99–106. [Google Scholar] [CrossRef]
- Debeaufort, F.; Quezada-Gallo, J.-A.; Voilley, A. Edible barriers: A solution to control water migration in foods. In Food Packaging: Testing Methods and Applications; Risch, S.J., Ed.; American Chemical Society: Washington, DC, USA, 2000; pp. 9–16. [Google Scholar]
- Miller, K.S.; Krochta, J.M. Oxygen and aroma barrier properties of edible films: A review. Trends Food Sci. Technol. 1997, 8, 228–237. [Google Scholar] [CrossRef]
- McHugh, T.H.; Krochta, J.M. Permeability properties of edible films. In Edible Coatings and Films to Improve Food Quality; Krochta, J.M., Baldwin, E.A., Nisperos-Carriedo, M.O., Eds.; Technomic Publ Co: Lancaster, NH, USA, 1994; pp. 139–188. [Google Scholar]
- Yaman, Ö.; Bayoιndιrlι, L. Effects of an edible coating and cold storage on shelf-life and quality of cherries. LWT Food Sci. Technol. 2002, 35, 146–150. [Google Scholar] [CrossRef]
- Rojas-Graü, M.A.; Avena-Bustillos, R.J.; Olsen, C.; Friedman, M.; Henika, P.R.; Martín-Belloso, O.; Pan, Z.; McHugh, T.H. Effects of plant essential oils and oil compounds on mechanical, barrier and antimicrobial properties of alginate–apple puree edible films. J. Food Eng. 2007, 81, 634–641. [Google Scholar] [CrossRef]
- Buonocore, G.G.; Conte, A.; Del Nobile, M.A. Use of a mathematical model to describe the barrier properties of edible films. J. Food Sci. 2005, 70, E142–E147. [Google Scholar] [CrossRef]
- Bonilla, J.; Atarés, L.; Vargas, M.; Chiralt, A. Edible films and coatings to prevent the detrimental effect of oxygen on food quality: Possibilities and limitations. J. Food Eng. 2012, 110, 208–213. [Google Scholar] [CrossRef]
- Earle, R.D.; McKee, D.H. Coated Food Product and Method of Making Same. U.S. Patent 4,504,502, 12 March 1985. [Google Scholar]
- Tanaka, H.; Matsumura, M.; Veliky, I.A. Diffusion characteristics of substrates in ca-alginate gel beads. Biotechnol. Bioeng. 1984, 26, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Gray, C.J.; Dowsett, J. Retention of insulin in alginate gel beads. Biotechnol. Bioeng. 1988, 31, 607–612. [Google Scholar] [CrossRef] [PubMed]
- Mørch, Ý.A.; Donati, I.; Strand, B.L.; Skjåk-Bræk, G. Effect of Ca2+, Ba2+, and Sr2+ on alginate microbeads. Biomacromolecules 2006, 7, 1471–1480. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhang Newby, B.-M. Layer-by-layer polyelectrolyte coating of alginate microgels for sustained release of sodium benzoate and zosteric acid. J. Drug Deliv. Sci. Technol. 2018, 46, 46–54. [Google Scholar] [CrossRef]
- Wong, D.W.S.; Gregorski, K.S.; Hudson, J.S.; Pavlath, A.E. Calcium alginate films: Thermal properties and permeability to sorbate and ascorbate. J. Food Sci. 1996, 61, 337–341. [Google Scholar] [CrossRef]
 ) with Ca2+ (
) with Ca2+ ( ) (Peteiro [32], Lee and Rogers [38]).
) (Peteiro [32], Lee and Rogers [38]).
 ) with Ca2+ (
) with Ca2+ ( ) (Peteiro [32], Lee and Rogers [38]).
) (Peteiro [32], Lee and Rogers [38]).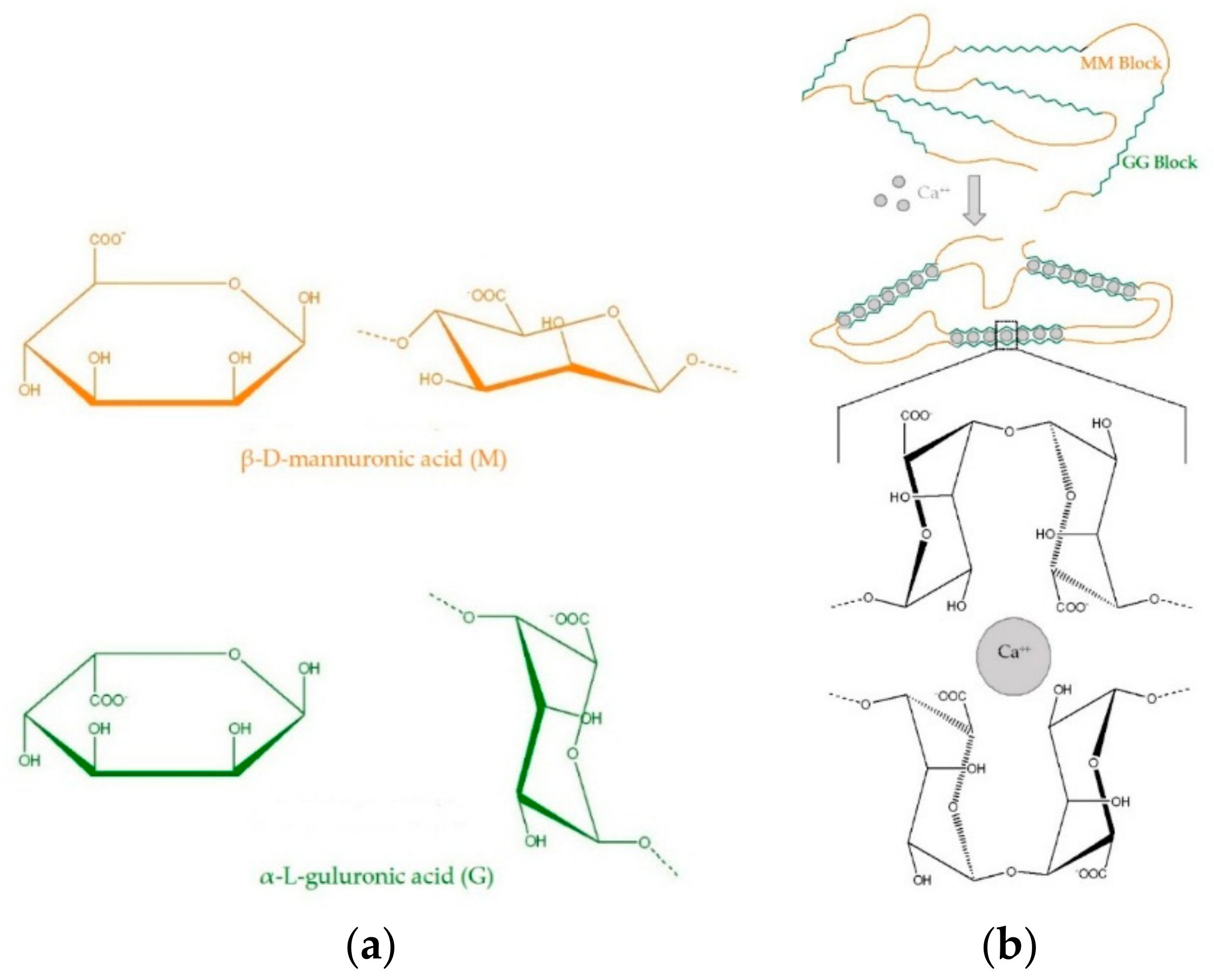
| Food | Coating/Crosslinking | Antimicrobial | Result | Source |
|---|---|---|---|---|
| fresh-cut apple | alginate-apple puree/CaCl2 (EC 1) | oregano, lemongrass, vanillin | high concentrations of Eos 1 inhibited the growth of Listeria innocua, psychrophilic aerobic bacteria, yeasts, and molds. | Rojas-Graü, et al. [106] |
| fresh-cut apple | alginate/CaCl2 (EC) | thyme oil | 15 EOs were evaluated. EC-thyme oil significantly inhibited the TPC 1, total coliform, LAB 1, yeast and mold growth. | Sarengaowa, et al. [107] |
| fresh-cut melon [108], apple [109] | alginate/calcium lactate (EC) | malic acid, cinnamon, palmarosa, lemongrass, clove EOs, and their active compounds | malic acid went through antimicrobial action alone. However, when EOs or their active compounds were incorporated, the effect was increased even further. | Raybaudi-Massilia et al. [108,109] |
| fresh-cut watermelon | alginate/calcium lactate (EC) | trans-cinnamaldehyde | EC-antimicrobial agent was significantly effective against psychrotrophs, coliforms, yeasts, and molds. | Sipahi, et al. [110] |
| fresh-cut pineapple | alginate, sunflower oil/CaCl2 (EC) | lemongrass EO | yeast, mold, and the total plate count were significantly reduced, and the shelf-life was prolonged. | Azarakhsh, et al. [111] |
| strawberry | alginate (EC) | carvacrol, methyl cinnamate | carvacrol was effective against both E. coli and B. cinereal, on the other hand, methyl cinnamate inhibited only B. cinerea. | Peretto, et al. [112] |
| strawberry | alginate/CaCl2 (EF 1) | Cryptococcus laurentii | microbial decay due to psychrotrophs, yeasts, and molds was significantly reduced. | Fan, Xu, Wang, Zhang, Sun, Sun, and Zhang [94] |
| capsicum | alginate/CaCl2 (EC) | pomegranate peel extract | EC-pomegranate peel extract possessed antimicrobial and antifungal activities. | Nair, et al. [113] |
| beef pieces and steak | alginate-maltodextrin/CaCl2-CMC (EC) | hypochlorous acid (HOCl) | EC-HOCl had no inhibitory effect, although HOCl inhibited the bacterial growth when treated alone. | Williams, et al. [114] |
| ground beef | alginate/CaCl2 (EC) | nisin, acetic acid, lactic acid, potassium sorbate chelating agents: EDTA 1, HMP 1 | only acetic and lactic acid inhibited E. coli. Immobilization in EC enhanced the activity of only some of the antimicrobial agent/combination. | Fang and Tsai [115] |
| ground beef | alginate/CaCl2 (EF) | nisin | load of Brohothrix thermosphacta significantly decreased until day 7. | Cutter and Siragusa [116] |
| beef tissue | alginate/CaCl2 (EC) | acetic acid, lactic acid | EC-immobilized acids were more effective in reducing L. monocytogenes compared to their direct application. Lactic acid had a higher inhibitory effect against Gram (−) at the same pH. | Siragusa and Dickson [117,118] |
| chicken fillet | alginate alone or alginate-galbanum gum/CaCl2 (EC) | EO of Ziziphora persica | alginate coating alone had no microbial inhibition effect. Composite coating and addition of EO to formulation had a significant microbial reduction. | Hamedi, et al. [119] |
| chicken breast fillet | alginate-maltodextrin/CaCl2-CMC 1 (EC) | lactoperoxidase enzyme | EC-lactoperoxidase decreased the microbial load of Enterobacteriaceae, P. aeruginosa and aerobic mesophilic bacteria but had no effect on the LAB. | Yousefi, et al. [120] |
| chicken thigh meat | alginate-whey protein/CaCl2 (EC) | lactoperoxidase enzyme | Antimicrobial effect increased with increasing concentration of the lactoperoxidase. | Molayi, et al. [121] |
| northern snakehead fish | alginate/CaCl2 (EC) | nisin, EDTA | EC did not increase the effectiveness of antimicrobials against TVC 1 and TPC. | Lu, et al. [122] |
| smoked salmon | starch-alginate/calcium gluconate (EF) | two strains of LAB, nisin | EF with LAB strains and nisin inhibited L. monocytogenes growth. | Concha-Meyer, et al. [123] |
| smoked salmon | alginate (EF) | sodium lactate, sodium diacetate, commercial formulation consists of both (Opti.Form) | EC-antimicrobials delayed the growth of L. monocytogenes during cold storage [124] and greatly prolonged the microbial shelf life during frozen storage [125]. | Neetoo, Ye, and Chen [124] and Ye, Neetoo, and Chen [125] |
| smoked salmon | alginate/CaCO3 (EC) | oyster lysozyme, hen egg white lysozyme, nisin | both EC-oyster and EC-hen egg white lysozyme inhibited L. monocytogenes and S. anatum. Addition of nisin enhanced the antimicrobial activity. | Datta, et al. [126] |
| abalone | alginate/CaCl2 (EC) | bamboo leaf extract, rosemary extract | EC-rosemary extract enhanced bacterial inhibition. PCA 1 was used to correlate between the microbial count and biogenic amines. | Hao, Liu, Sun, Xia, Jia, Li, and Pan [93] |
| rainbow trout fillet | alginate/CaCl2 (EC) | resveratrol | coating with antimicrobial agent decreased bacterial, yeast, and mold growth. | Bazargani-Gilani [127] |
| silver carp fillet | alginate-CMC/CaCl2 (EC) | clove EO | EC-clove EO has antimicrobial activity against L. monocytogenes, S. aureus and E. coli, in a decreasing order. Gram (+) bacteria were more sensitive then Gram (−). Concentration increase had a significant effect. | Jalali, et al. [128] |
| bighead carp fillet | alginate/CaCl2 (EC) | horsemint EO | combined effect of EC-horsemint EO significantly decreased the growth rate of TVC and TPC. | Heydari, et al. [129] |
| winter flounder (fish) | alginate/CaCl2 (EC) | glucose oxidase (GOx) | enzyme-alginate blankets exhibited very low surface pH values. | Field, et al. [130] |
| sea bass | alginate (EC) | tea polyphenols | EC decreased TVC, the reduction was even higher with the incorporation of tea polyphenols into the coating. | Nie, et al. [131] |
| sea bass [132], red sea bream [133] | alginate/CaCl2 (EC) | ε-polylysine [132], 6-gingerol [133] | EC-ε-polylysine and EC-6-gingerol reduced microbial counts, even more effectively than antimicrobial agent or coating, alone. | Cai et al. [132,133] |
| sea bass [134], Fior di Latte cheese [135] | alginate/CaCl2 (EC) | reuterin produced by Lactobacillus reuteri | EC system containing biopreservative L. reuterin was designed [135]. EC-reuterin was effective in the improvement of microbiological quality [134,135]. | Angiolillo et al. [134,135] |
| kashar cheese | alginate-whey protein isolate (EC) | ginger EO | EC-ginger EO had a bacteriostatic and bactericidal effect on E. coli and S. aureus, respectively. | Kavas, et al. [136] |
| mozzarella | alginate/CaCl2 (EC) | potassium sorbate, sodium benzoate, calcium lactate, calcium ascorbate | active compounds showed a similar effect in terms of the growth of Pseudomonas spp. and Enterobacteriaceae. EC–3% potassium sorbate decreased the growth rate. | Lucera, et al. [137] |
| low-fat cut cheese | alginate-mandarin fiber (EC) | oregano EO | An oregano EO concentration ≥ 2% was effective against S. aureus, psychrophilic bacteria, molds, and yeasts. | Artiga-Artigas, et al. [138] |
| - 2 | alginate/CaCO3 (EF) | microencapsulated lemongrass oil | release kinetics were studied. L. monocytogenes and E. coli were successfully inhibited. | Bustos, et al. [139] |
| - 2 | alginate/CaCl2 (EF) | potassium sorbate | the permeability and release of potassium sorbate were modeled. | Zactiti and Kieckbusch [71,140] |
| - 2 | alginate clay bionanocomposite (EF) | marjoram, clove, cinnamon essential oils | nanocomposite EF-Marjoram was the most effective in controlling foodborne pathogens due to possessing a high content of phenolic compounds. | Alboofetileh, et al. [141] |
| - 2 | alginate/CaCl2 (EF) | garlic oil | the inhibitory effect was dependent on the Gram character and increased in the following order: S. typhimurium < E. coli < S. aureus < B. cereus. | Pranoto, et al. [142] |
| - 2 | alginate (EF) | lysozyme, nisin, grapefruit seed extract, EDTA | EF with grapefruit seed extract alone or in combination with EDTA showed good antimicrobial protection. | Su Cha, Choi, Chinnan, and Park [95] |
| - 2 | alginate-CMC/CaCl2 (EF) | pyrogallic acid | EF-pyrogallic acid had significant inhibitory effect against E. coli and S. aureus. | Han and Wang [143] |
| Food | Coating/Crosslinking | Antioxidant | Result | Source |
|---|---|---|---|---|
| fresh-cut papaya | alginate, sunflower oil/CaCl2 (EC 1) | ascorbic acid | total ascorbic acid content was almost doubled throughout the storage due to oxygen barrier properties. | Tapia, Rojas-Graü, Carmona, Rodríguez, Soliva-Fortuny, and Martin-Belloso [39] |
| guava | alginate/CaCl2 (EC) | pomegranate peel extract | EC increased the antioxidant activity; the effect was even promoted with the addition of pomegranate peel extract. | Nair, et al. [146] |
| fresh-cut pears | alginate, sunflower oil/CaCl2 (EC) | N-acetylcysteine, glutathione | EC-antioxidant agents had significant antioxidant activities, although EC alone did not. | Oms-Oliu, et al. [147] |
| sliced carrots | alginate/CaCl2 (EC) | citric acid | coating process, when applied together with a modified atmosphere, enhanced the shelf life extension effect. | Amanatidou, et al. [148] |
| ground beef patties | alginate, starch, stearic acid (EF 1) | tocopherols | regardless of their incorporation method, tocopherols were effective. Additionally, tocopherols improved the moisture barrier properties. | Wu, et al. [149] |
| buffalo meat patties | alginate/CaCl2 (EC) | sodium ascorbate, citric acid | EC with antioxidants retarded lipid oxidation. | Chidanandaiah, et al. [150] |
| chicken fillet | alginate-galbanum gum/CaCl2 (EC) | EO of Ziziphora persica | both galbaum gum and Ziziphora EO 1 have high antioxidant activities due to the high phenolic and flavonoid content. | Hamedi, Kargozari, Shotorbani, Mogadam, and Fahimdanesh [119] |
| pork chops | alginate, modified starch/CaCl2 (EC) | rosemary oleoresin | lipid oxidation was inhibited. | Handley, et al. [151] |
| bream | alginate/CaCl2 (EC) | vitamin C, tea polyphenols | EC decreased TBA 1 significantly due to being resistant to oxygen diffusion. Vitamin C was more effective in decreasing lipid oxidation. | Song, et al. [152] |
| red sea bream | alginate (EC) | 6-gingerol | EC and antioxidant alone led to an equal inhibition effect; on the other hand, their combination had minimum lipid oxidation values in terms of TBA. | Cai, Wang, Cao, Lv, and Li [133] |
| bighead carp fillet | alginate/CaCl2 (EC) | horsemint EO | EC caused lower oxidation values after the 8th day of storage; the addition of horsemint EO increased this effect even further. | Heydari, Bavandi, and Javadian [129] |
| silver carp fillet | alginate/CaCl2 (EC) | clove EO | EC-clove EO significantly decreased the lipid oxidation probably due to the combined effect of EO and oxygen barrier properties of the alginate coating. | Jalali, Ariiai, and Fattahi [128] |
| rainbow trout fillet | alginate/CaCl2 (EC) | resveratrol | EC-resveratrol coating reduced lipid oxidation significantly. | Bazargani-Gilani [127] |
| rainbow trout fillet | alginate-clay nanoparticles/ CaCl2 (EC) | lycopene | although the EC-lycopene combination helped decrease the FFA 1, other fat oxidation parameters such as peroxide and TBA values could not be significantly decreased. | Ehsani, et al. [153] |
| sea bass | alginate (EC) | tea polyphenols | EC, tea polyphenols inhibited lipid oxidation when they were applied alone, however, the inhibition was higher in their combination due to the synergistic effect. | Nie, Wang, Wang, Lei, Hong, Huang, and Zhang [131] |
| - 2 | alginate/CaCl2 (EF) | white, red, and extruded white ginseng extracts | EC-ginseng extract showed good antioxidant activity, which can be even increased with controlling the extrusion process. | Norajit, et al. [154] |
| Food | Effects | Source |
|---|---|---|
| fresh-cut apples | Optimum composition of alginate-based EC 1 was determined for achieving high water and firmness retention during storage. | Ghavidel, et al. [189] |
| fresh-cut apples | Shelf life of coated apples were prolonged three times compared to uncoated samples. EC maintained firmness, although it increased fermentative metabolites’ (i.e., acetaldehyde and ethanol) production due to MA 1. | Rojas-Graü, Tapia, and Martín-Belloso [79] |
| fresh-cut apples | Base solution was developed with alginate and 26% apple puree. Ethylene, CO2 production, and O2 consumption were reduced. However, solely vanillin incorporated formulations could achieve acceptable test scores in contrast with other EOs 1. | Rojas-Graü, Raybaudi-Massilia, Soliva-Fortuny, Avena-Bustillos, McHugh, and Martín-Belloso [106] |
| fresh-cut apples | The soluble solid content was increased; stable browning index, acidity, and firmness levels were achieved due to coating with prebiotics incorporated EC. | Rößle, Brunton, Gormley, Wouters, and Butler [166] |
| apples | Alginate and gelatin-based coatings not only preserved the freshness of the fruit, but also improved the appearance and attractiveness of the fruit. | Moldão-Martins, et al. [190] |
| apple pieces | Apple pieces were coated with double layers of polysaccharide/lipid (alginate/acetylated monoglyceride) EC to decrease respiratory activity. | Wong, Tillin, Hudson, and Pavlath [184] |
| fresh-cut apples | Thyme oil had the highest antimicrobial activity among the tested 15 EOs. Physical, chemical and microbial qualities of coated (thyme incorporated) samples were assessed. | Sarengaowa, Hu, Jiang, Xiu, and Feng [107] |
| fresh-cut apples [109], fresh-cut melon [108] | The effect of malic acid, EOs and their active compounds on quality characteristics were assessed. Due to the inhibition of microflora, respiration and anaerobic fermentation were decreased. However, physicochemical characteristics of the products were affected differently with respect to the type of EOs and concentrations. | Raybaudi-Massilia et al. [108,109] |
| fresh-cut melon | Sodium alginate-sunflower oil maintained the firmness, however, the coating could not present good barrier properties against O2, CO2, ethylene, and could not reduce the loss of vitamin C and microbial load. | Oms-Oliu, et al. [191] |
| fresh-cut melon | LbL technique with oppositely charged alginate-chitosan presented a superior performance on firmness, gas exchange, and microbial growth. | Poverenov, Danino, Horev, Granit, Vinokur, and Rodov [174] |
| fresh-cut watermelon | LbL coating did not affect the pH and °Brix but preserved the textural firmness and decreased weight loss. | Sipahi, Castell-Perez, Moreira, Gomes, and Castillo [110] |
| ber fruit | The quality was retained with the application of the composite edible coating, consisting of sodium alginate and olive oil, enriched with ascorbic and citric acids. | Ramana Rao, Baraiya, Vyas, and Patel [165] |
| strawberry | The quality of the products was enhanced by implementing a yeast antagonist to the formulation. | Fan, Xu, Wang, Zhang, Sun, Sun, and Zhang [94] |
| strawberry | Effects of alginate, chitosan, pullulan-based EC on antioxidant enzyme system and quality characteristics were compared. All the polysaccharide-based coatings decreased quality losses and extended shelf life. | Li, et al. [192] |
| strawberry | Incorporation of carvacrol and methyl cinnamate changed the physical properties of the alginate coatings such as turbidity, transparency, and viscosity, depending on their concentration. | Peretto, Du, Avena-Bustillos, Berrios, Sambo, and McHugh [112] |
| strawberry | The effectiveness of alginate and soy-based coatings on the pH and vitamin C content of the samples were compared. | Ahmed, et al. [193] |
| blueberry | Numerous ECs (including alginate) were compared in terms of their ability to control quality losses. | Duan, Wu, Strik, and Zhao [92] |
| blueberry | Performances of chitosan and alginate coatings were compared. Although alginate coatings promoted firmness, lightness and total phenolic content; yeast and mold growth in the samples were induced. | Chiabrando and Giacalone [194] |
| cherry | The storability period of the coated products increased from 8 to 16 days with a delay in the post-harvest ripening and maintaining higher amounts of total phenolics and antioxidant activity. | Díaz-Mula, Serrano, and Valero [90] |
| fresh-cut pear | EC with antibrowning agents (N-acetylcysteine and glutathione) reduced microbial growth, increased vitamin C, and the total phenolic content without affecting the firmness of product. | Oms-Oliu, Soliva-Fortuny, and Martín-Belloso [147] |
| pear | Alginate coated samples had a higher tensile strength, elongation, and elasticity; on the other hand, they had a lower water loss, pH increase, metabolic activities with maintained firmness and green color. | Moraes, et al. [195] |
| plums | Particularly 3% alginate coating significantly inhibited ethylene production, softening, acidity and water losses, and slowed down carotenoid and anthocyanin increase (and therefore delayed color change) throughout the storage period of plums. | Valero, Díaz-Mula, Zapata, Guillén, Martínez-Romero, Castillo, and Serrano [91] |
| fresh-cut papaya | The study consisted of two steps: RSM 1 was used to determine the number of ingredients in the formulation in terms of WVR 1; the chosen formulations helped to achieve increased firmness. On the contrary of several previous studies, the alginate coating did not affect the respiratory rate and ethylene production. | Tapia, Rojas-Graü, Carmona, Rodríguez, Soliva-Fortuny, and Martin-Belloso [39] |
| Guava | EC-pomegranate peel extract improved the visual and nutritional parameters with delaying senescence. | Nair, Saxena, and Kaur [146] |
| mango | EC-ascorbic acid retarded firmness loss improved the phenolics and carotenoids content and sensory scores; however, the antimicrobial efficiency was not significant. | Salinas-Roca, et al. [196] |
| fresh-cut pineapples | The concentration of ingredients in EC was formulated with the help of RSM [83]. Incorporation of lemongrass EO and ascorbic-citric acid into EC prolonged the shelf life whilst maintaining quality attributes. | Azarakhsh et al. [83], [111] |
| fresh-cut pineapples | Shelf life of the product was significantly improved. | Montero-Calderón, Rojas-Graü, and Martín-Belloso [158] |
| Peach | Shelf life was increased with maintaining quality. | Maftoonazad, et al. [197] |
| tomato | Reduced ethylene production, respiration rate, weight loss, a diminution rate of hue angle values (indicated that ripening was delayed) as well as a higher fruit firmness, TSS (total soluble solids concentration), titratable acidity (TA), organic acids (citric, malic and, ascorbic acids), sugars (glucose and fructose), and sensory scores of coated products were achieved. | Zapata, Guillén, Martínez-Romero, Castillo, Valero, and Serrano [89] |
| potato strips | Possibility of using the alginate coating and ultrasound process as an alternative to blanching of potato strips were investigated. EC was not effective for diminishing the color changes and microbial load. | Amaral, et al. [198] |
| garlic bulbs | Natural compound isolated from the garlic skin was added into EC. The effects of coating on the mechanical and barrier properties were demonstrated. | Nussinovitch and Hershko [199] |
| carrot | A 5- to 7-day shelf-life extension of the coated samples was achieved. | Amanatidou, Slump, Gorris, and Smid [148] |
| lettuce | 1-Methylcyclopropene incorporated EC reduced the discoloration, respiration rate, ethylene synthesis (therefore senescence) of samples. | Tay and Perera [200] |
| Food | Effects | Source |
|---|---|---|
| ground beef patties | Incorporation of stearic acid into the modified starch-alginate formulation improved the barrier properties against moisture loss and decreased lipid oxidation. Addition of tocopherols increased these effects. | Wu, Weller, Hamouz, Cuppett, and Schnepf [149] |
| buffalo meat patties | EC 1 significantly improved quality attributes such as overall shear force, TBA 1, tyrosine value, and microbial counts, etc. | Chidanandaiah, Keshri, and Sanyal [150] |
| lamb meat | Alginate-maltodextrin coating crosslinked with CaCl2-CMC 1 led to a decrease in the total volatile nitrogen for refrigerated meat, there was no statistical difference for frozen meat. Although a decrease in the total count of refrigerated meat was only due to calcium ions in the crosslinking solution, EC achieved psychrophilic bacterial inhibition during the frozen storage. | Koushki et al. [207,208] |
| pork chops | Composite coating with modified starch-alginate with rosemary oleoresin inhibited lipid oxidation and formation of hexanal, pentane, and total volatiles. | Handley, Ma-Edmonds, Hamouz, Cuppett, Mandigo, and Schnepf [151] |
| pork cuts | Alginate (>1%), helped to decrease the thawing loss; concentration of Ca2+ influenced the tenderness of the meat. Optimum coating conditions were defined as 3% alginate, 7% CaCl2 with 5–7 min crosslinking time to diminish thawing loss, TBARS 1, and an increase in the total protein solubility. | Yu, et al. [209] |
| cut-up poultry parts | Water evaporated from coating instead of meat. One thick coating application was more convenient than repeating number of coats due to preventing residual calcium salts from being transferred into the alginate dipping solution and the easiness of pealing. | Mountney and Winter [21] |
| chicken breast and chicken thigh meat | Lactoperoxidase addition into the alginate-based coating system led to higher bacterial and sensorial quality values of chicken meat. The effect was even increased with the increasing concentration of lactoperoxidase. | Yousefi, Farshidi and Ehsani [120], Molayi, Ehsani, and Yousefi [121] |
| films/casing for breakfast pork sausages | Study assessed the ability of food polymers including gelatin-sodium alginate blends for the formation of stable packaging film. The optimum processing conditions were presented during the extrusion process [210]. The effects of different oil additions on quality parameters of the films/casings [211] and their usage in the manufacturing of sausages were determined [212]. | Liu et al. [210,211,212] |
| bream | EC reduced the rate of quality losses of bream in terms of water loss, pH, TVB-N 1, and K-value. A 5% vitamin C content incorporated coating maintained the best quality and sensory results. | Song, Liu, Shen, You, and Luo [152] |
| red sea bream | EC-6-gingerol coated products obtained a 20-day shelf life extension. | Cai, Wang, Cao, Lv, and Li [133] |
| japanese sea bass | The synergistic effect of EC and ε-polylysine helped products to maintain a fresh color and tissue hardness, reduce lipid oxidation, protein degradation, and nucleotide breakdown. | Cai, Cao, Bai and Li [132] |
| japanese sea bass | EC-tea polyphenols provided the greatest effect on quality (TVB-N, lipid oxidation, protein decomposition) and sensory results compared to their effects alone. | Nie, Wang, Wang, Lei, Hong, Huang, and Zhang [131] |
| sea bass | New biopreservation coating with the addition of food supplement Lactobacillus reuteri and its substrate glycerol to EC was developed. The production and antimicrobial effects of reuterin material were evaluated after 2 different fermentation periods. | Angiolillo, Conte, and Del Nobile [134] |
| sea bass | Two protective processes: salting application of liquid smoke suspension containing resveratrol and alginate coating were used to enhance the quality. Although the treatment combination was effective in reducing oxidation, it could not inhibit bacterial growth. | Martínez, et al. [213] |
| rainbow trout | 0.2% resveratrol improved the effect of EC with the highest inhibition of chemical changes and microbial growth. | Bazargani-Gilani [127] |
| rainbow trout | Effects of EC with or without lycopene were investigated in terms of various quality parameters. | Ehsani, Paktarmani, and Yousefi [153] |
| silver carp fillet | Fillets were coated with alginate-CMC. With the help of the controlled release of clove oil, the coating lead to an 8-day shelf life extension without affecting the sensorial properties. | Jalali, Ariiai, and Fattahi [128] |
| bighead carp fillet | Lower microbial deterioration and auto-oxidation of fish fillets throughout storage were achieved. | Heydari, Bavandi, and Javadian [129] |
| kilka fish | Shelf life extension was achieved with an alginate-whey protein coating. | Seyfzadeh, et al. [214] |
| northern snakehead fillets | Contrary to the previous findings of EC-nisin [115,116], researchers did not find any significant evidence that calcium alginate containing nisin increased the effectiveness of the antimicrobial agent. Nevertheless, inhibition of lipid oxidation, TMA-N 1, TVB-N, promoting water barrier properties and sensory scores were achieved. | Lu, Liu, Ye, Wei, and Liu [122] |
| minced fish patties | EC was applied in a different manner: All the ingredients such as minced fish patties, soy protein concentrate, onions, celery, as well as sodium alginate, were blended. The patties were pre-coated initially with soybean oil and afterward dipped in the CaCl2 solution for film formation, which prevented the patties from sticking to surfaces during processing. | Rockower, et al. [215] |
| abalone | An EC-3.5% rosemary extract was successful for the preservation of the product due to reducing TVB-N, controlling biogenic amines, and maintaining better sensory scores. | Hao, Liu, Sun, Xia, Jia, Li, and Pan [93] |
| Study | Source |
|---|---|
| The effects of soy isolate-sodium alginate and soy isolate-PGA 1 interactions on the functional properties of the film formation were investigated and it was found that protein-polysaccharide interactions enhanced the film-forming properties. | Shih [219] |
| ≈10% PGA addition to the soy protein increased the whiteness and tensile strength and decreased the yellowness, percentage elongation at break, WVP 1, and water solubility of the multicomponent EF 1. | Rhim, et al. [220] |
| Films formed from alginate-whey protein complexes had higher tensile strength, elastic modulus, and elongation than whey protein alone. | Coughlan, et al. [221] |
| Different protein-polysaccharide films were compared in terms of oxygen and WVP, tensile strength, transparency, etc. | Yoo and Krochta [48] |
| By using a Box-Behnken experimental design and RSM 1 (for 3 factors; sodium alginate, low acyl gellan, glycerol concentration), a biofilm formulation was designed in terms of mechanical properties. | González-Cuello, et al. [222] |
| EF formulation was prepared by mixing sodium alginate with a variable quantity of cashew tree gum. However, tensile strength and water barrier properties were weakened due to the competition between two gel-forming polysaccharides for the interaction with calcium ions in the crosslinking step. | Azeredo, et al. [223] |
| Gelation of calcium alginate with rice starch and/or rice flour was examined. Coarser and more rigid gel structures with an increased diffusion coefficient, a heterogeneous structural resistance constant, and a decreased gelation rate constant were obtained. | Chrastil [64] |
| Incorporation of garlic oil as a natural antibacterial agent caused darker, yellowish color formation, reduced tensile strength and elongation at break while garlic oil interfered with the calcium ion interactions due to being incorporated before crosslinking. | Pranoto, Salokhe, and Rakshit [142] |
| Ginseng extract (white, red and extruded white extract) was inserted into the film formulation and a slight decrease in moisture content, and increase in water solubility, transparency, and alteration in the mechanical properties of EF was observed. | Norajit, Kim and Ryu [154] |
| A new multilayer film, which contained chitosan in the top layer, ornidazole-incorporated polyvinyl alcohol middle and sodium alginate sublayer, with an enhanced swelling rate, water absorption capacity, control of water vapor transmission, and light transmittance, was developed. | Pei, et al. [224] |
| Incorporation of pyrogallic acid into the sodium alginate-CMC 1 matrix increased the gas, vapor, and UV barrier properties of EF. | Han and Wang [143] |
| Interactions between encapsulated n-hexanal (aroma compound) and alginate matrix affected the barrier, permeability, and surface properties of emulsified alginate EF. | Hambleton, Debeaufort, Bonnotte, and Voilley [159] |
| Enzyme activity of pig liver esterase with enhanced encapsulation efficiency (i.e., chitosan coating of alginate beads) was studied. | Pauly, et al. [225] |
| With a novel approach, the alginate film was designed based on the RGB image analysis and color changes. Alginate surface concentration and surface color were modeled to predict the physical properties of the film with a non-destructive method. | Acevedo, et al. [226] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Senturk Parreidt, T.; Müller, K.; Schmid, M. Alginate-Based Edible Films and Coatings for Food Packaging Applications. Foods 2018, 7, 170. https://doi.org/10.3390/foods7100170
Senturk Parreidt T, Müller K, Schmid M. Alginate-Based Edible Films and Coatings for Food Packaging Applications. Foods. 2018; 7(10):170. https://doi.org/10.3390/foods7100170
Chicago/Turabian StyleSenturk Parreidt, Tugce, Kajetan Müller, and Markus Schmid. 2018. "Alginate-Based Edible Films and Coatings for Food Packaging Applications" Foods 7, no. 10: 170. https://doi.org/10.3390/foods7100170
APA StyleSenturk Parreidt, T., Müller, K., & Schmid, M. (2018). Alginate-Based Edible Films and Coatings for Food Packaging Applications. Foods, 7(10), 170. https://doi.org/10.3390/foods7100170






