Effect of Soaking and Roasting on the Physicochemical and Pasting Properties of Soybean Flour
Abstract
:1. Introduction
2. Materials and Methods
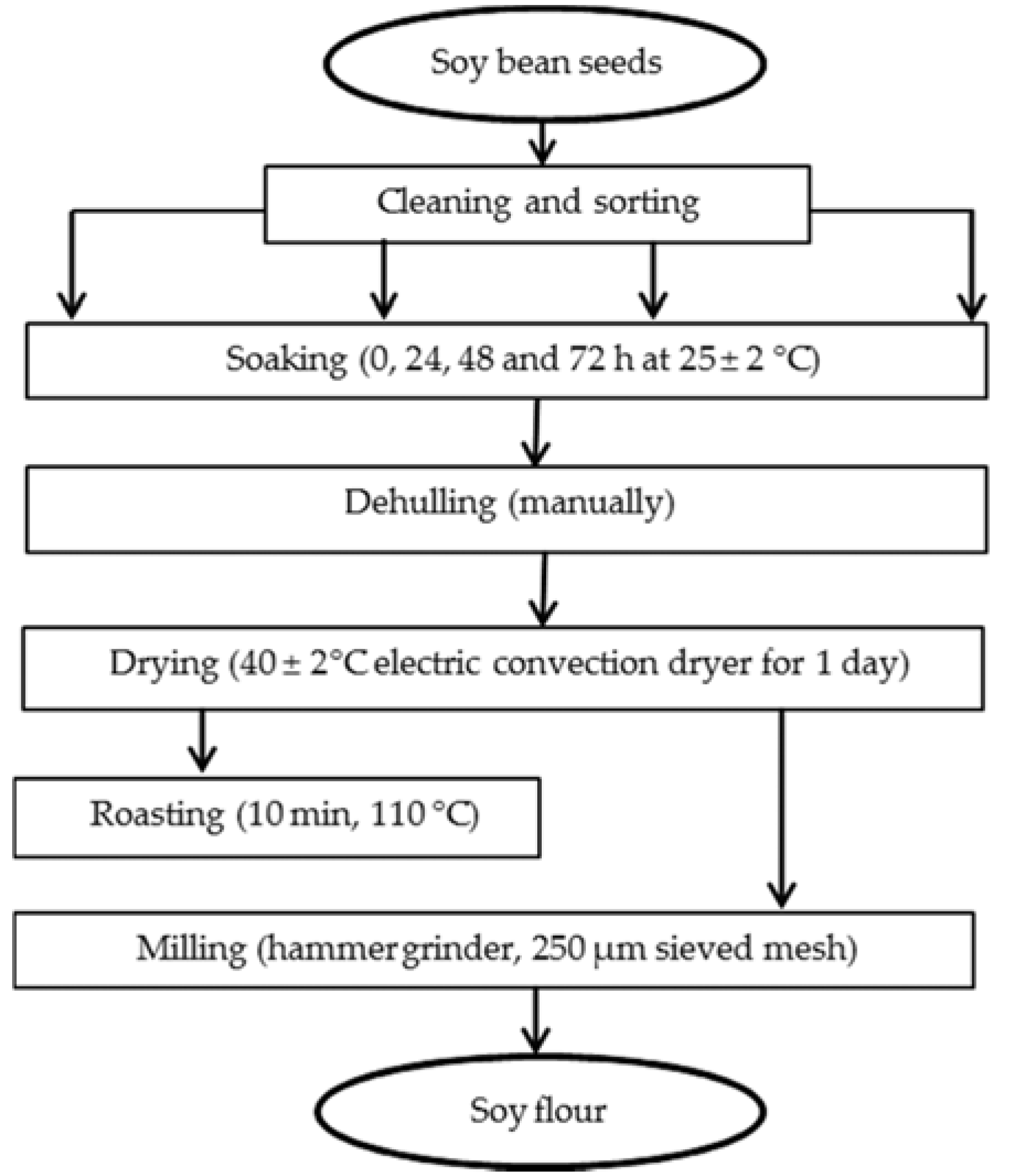
2.1. Sampling of Soybean and Production of Soybean Flour
2.2. Chemical Analysis of Soybean Flours
2.2.1. Determination of the Proximate Composition and Some Phytochemicals
2.2.2. Determination of Some Physicochemical and Pasting Properties
2.3. Statistical Analysis
3. Results
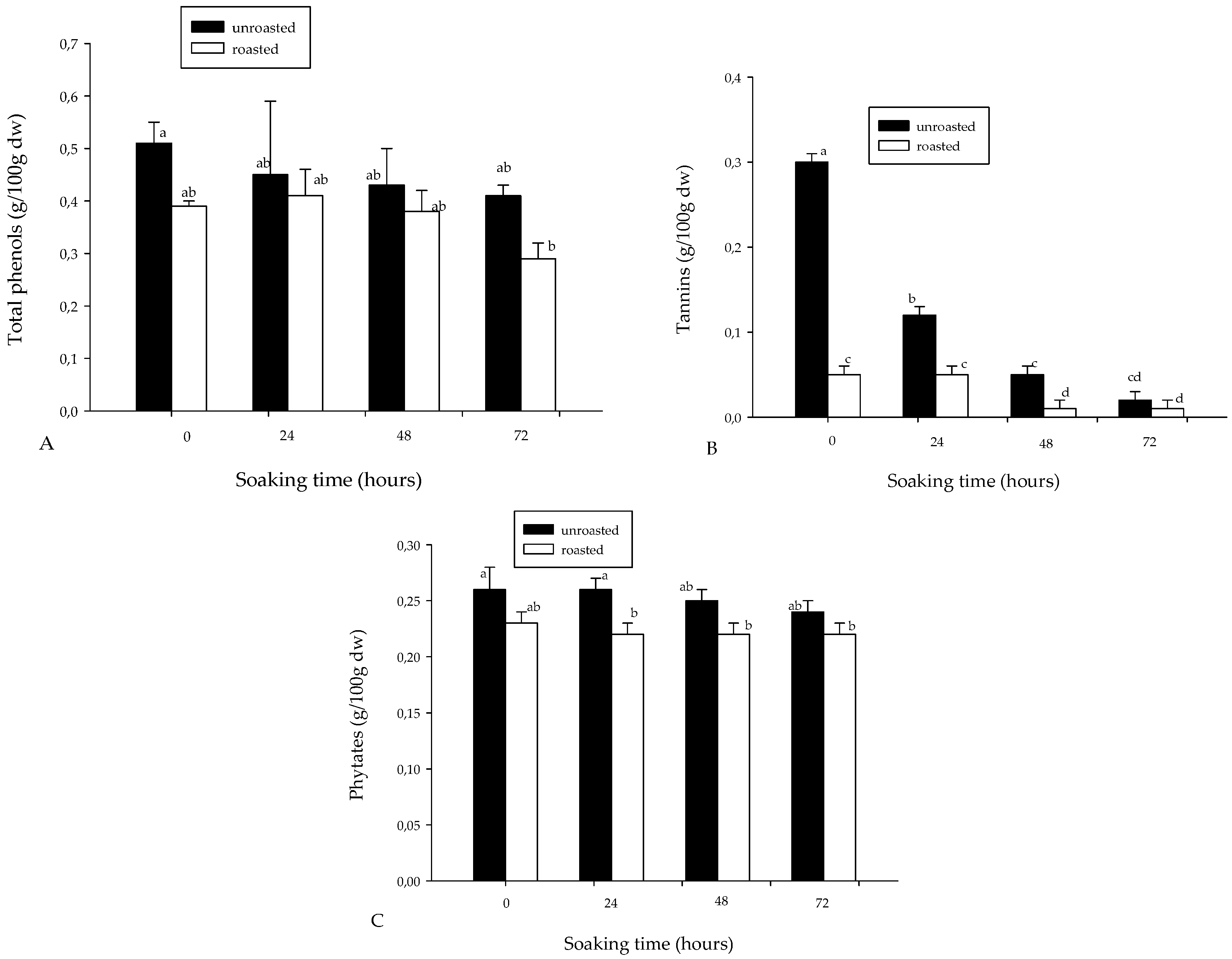
3.1. Proximate Composition and Some Phytochemicals of Soybean Flour
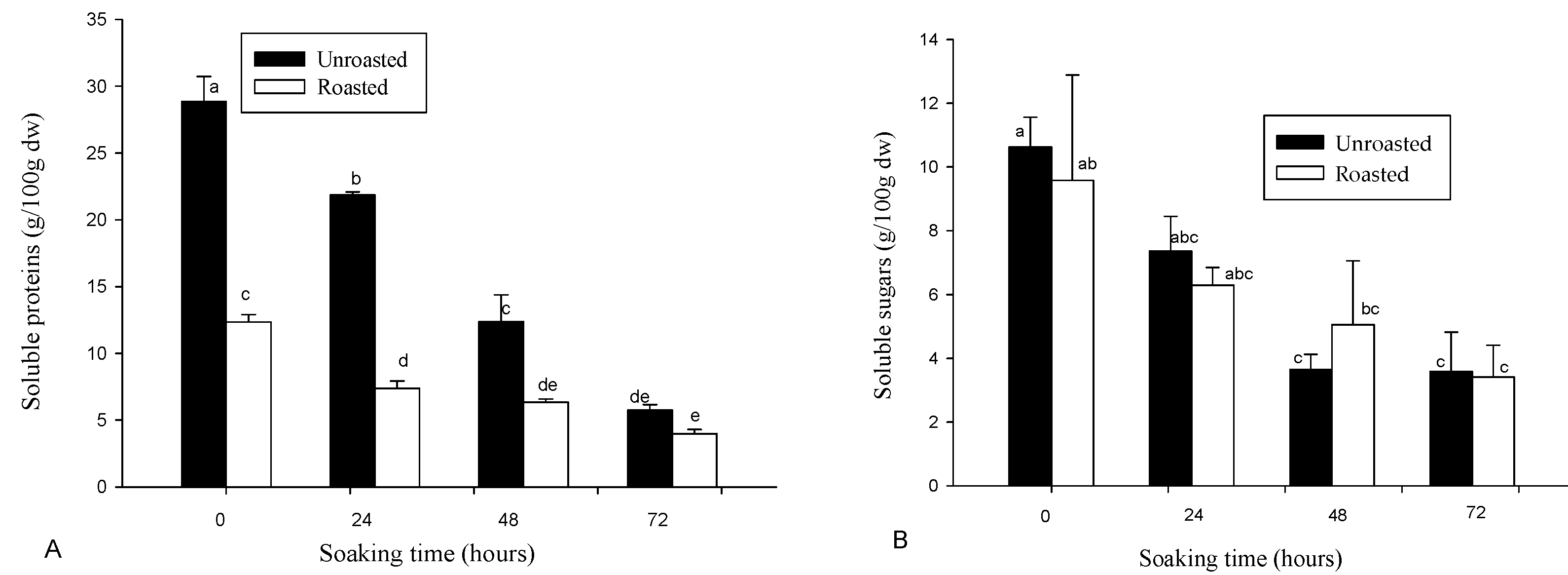
3.2. Variation in Soluble Proteins and Sugars
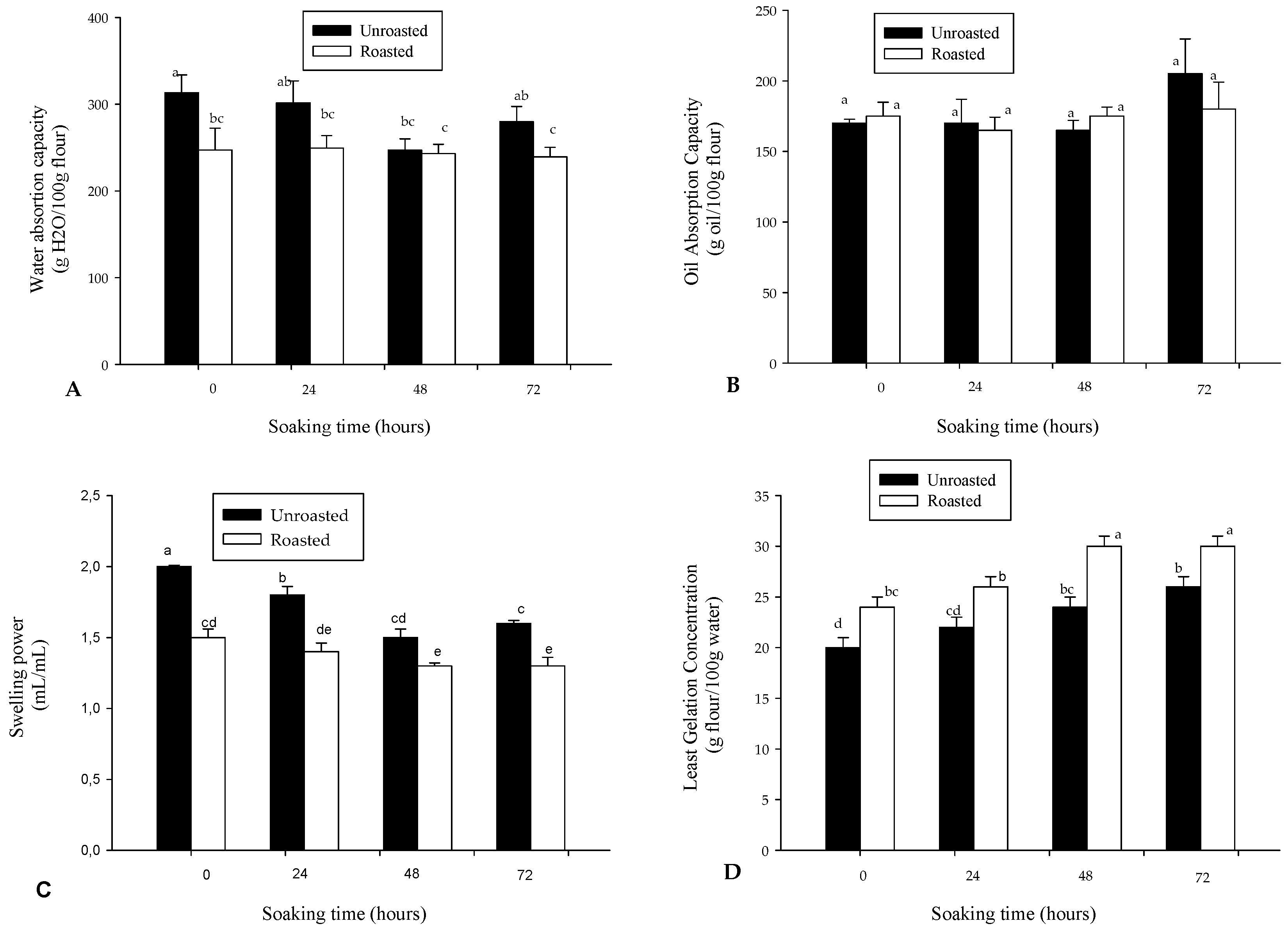
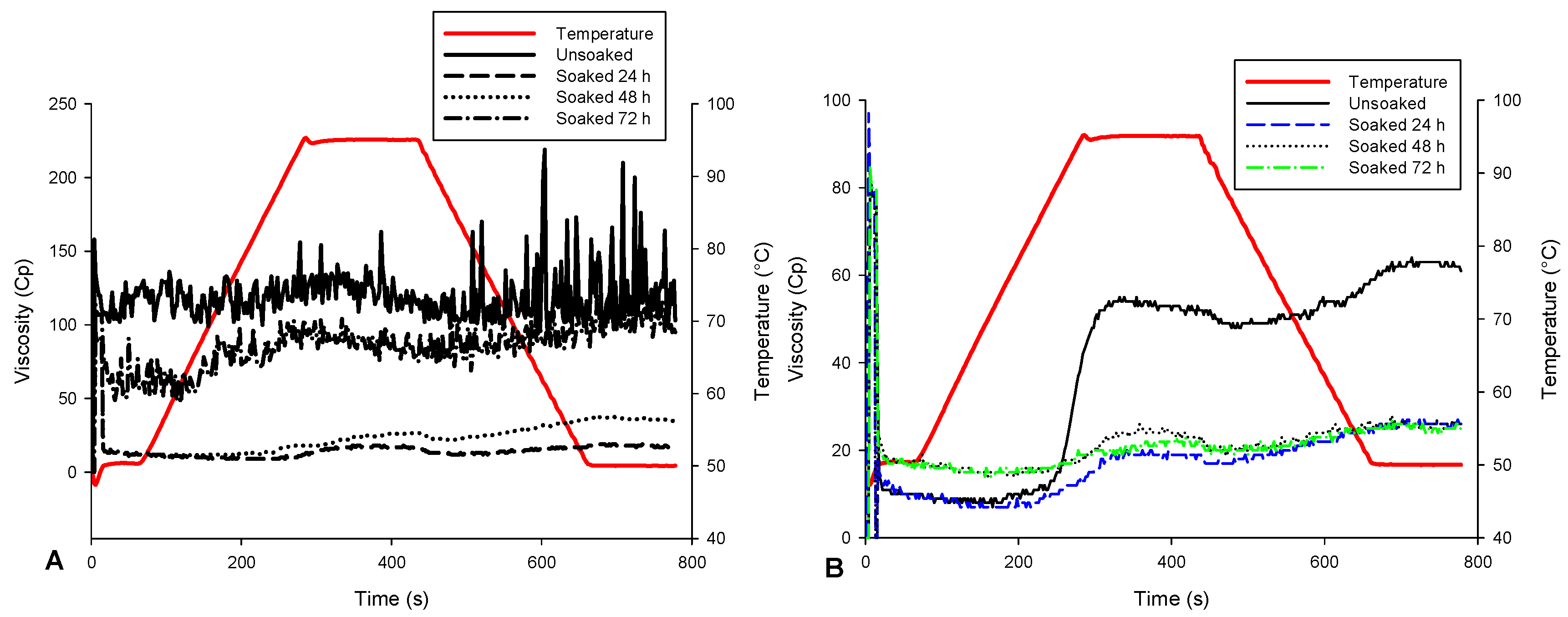
3.3. Some Physicochemical and Pasting Properties of Soybean Flour
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lee, G.-J.; Wu, X.; Shannon, G.J.; Sleper, A.D.; Nguyen, T.H. Soybean, Chapter 1 in Genome Mapping and Molecular Breeding in Plants; Oilseeds Kole, C., Ed.; Springer: Berlin/Heidelberg, Germany, 2007; Volume 2, pp. 1–53. [Google Scholar]
- Knight, D.C.; Eden, J.A. A review of the clinical effects of phytoestrogens. Obs. Gynecol. 1996, 87, 897–904. [Google Scholar]
- Ekor, M.; Emerole, O.G.; Farombi, O.E. Phenolic extract of soybean (Glycine max) attenuates cisplatin-induced nephrotoxicity in rats. Food Chem. Toxicol. 2010, 48, 1005–1012. [Google Scholar] [CrossRef] [PubMed]
- United States Department of Agriculture (USDA). World Agricultural Production; Foreign Agricultural Service Circular Series; Washington, DC, USA, 2016; pp. 6–16.
- Plahar, W.A.; Nti, C.A.; Annan, N.T. Effect of soy-fortification method on the fermentation characteristics and nutritional quality of fermented maize meal. Plant Food Hum. Nutr. 1997, 51, 365–380. [Google Scholar] [CrossRef]
- Tchango Tchango, J. The nutritive quality of maize-soybean (70:30) tempe flour. Plant Food Hum. Nutr. 1995, 47, 319–326. [Google Scholar] [CrossRef]
- Abubakar, M.U.; Sopade, A.P.; Yelmi, M.B. Flow behavior of akamu from different maize varieties and fortified with soybean flour. Food Control 1997, 8, 105–111. [Google Scholar] [CrossRef]
- Akubor, P.I.; Onimawo, A.I. Functional properties and performance of soybean and maize flour blends in cookies. Plant Food Hum. Nutr. 2003, 58, 1–12. [Google Scholar] [CrossRef]
- Egounlety, M.; Syarief, R. Study on the supplementation of ogi with tempe. Nigerian Food J. 1992, 10, 92–102. [Google Scholar]
- Yadav, S.; Khetarpaul, N. Indigenous bean fermentation: Effect on some antinutrients and in vitro digestibility of starch and protein. Food Chem. 1994, 50, 403–406. [Google Scholar] [CrossRef]
- Baik, B.-K.; Han, H.I. Cooking, Roasting, and Fermentation of Chickpeas, Lentils, Peas, and Soybeans for Fortification of Leavened Bread. Cereal Chem. 2012, 89, 269–275. [Google Scholar] [CrossRef]
- Son, S.-J.; Lee, S.-P. Physicochemical and Functional Properties of Roasted Soybean Flour, Barley, and Carrot Juice Mixture Fermented by Solid-state Fermentation Using Bacillus subtilis HA. Food Sci. Biotechnol. 2011, 20, 1509–1515. [Google Scholar] [CrossRef]
- Shin, D.-J.; Kim, W.; Kim, Y. Physicochemical and sensory properties of soy bread made with germinated, steamed, and roasted soy flour. Food Chem. 2013, 141, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Agume, N.A.S.; Njintang, Y.N.; Mbofung, C.M.F. Physicochemical and pasting properties of maize flour as a function of the interactive effect of natural-fermentation and roasting. Food Meas. 2016. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemists (AOAC). International Official Methods of Analysis; AOAC: Arlington, VA, USA, 1998; Volume 1–2. [Google Scholar]
- American Association of Cereal Chemists (AACC). Approved Methods of the AACC, 10th ed.; The Association: St. Paul, MN, USA, 2000. [Google Scholar]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Roberts, P.A.; Smith, F. Colorimetric method for determination of sugar and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Ngatchic, M.J.T.; Sokeng, D.S.; Njintang, Y.N.; Maoundombaye, T.; Oben, J.; Mbofung, C.M.F. Evaluation of some selected blood parameters and histopathology of liver and kidney of rats fed protein-substituted mucuna flour and derived protein rich product. Food Chem. Toxicol. 2013, 57, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Kaur, M.; Singh, N.; Sandhu, S.K. Relationships between selected properties of black gram seeds and their composition. Int. J. Food Prop. 2004, 7, 541–552. [Google Scholar] [CrossRef]
- Coffman, C.W.; Garcia, V.V. Functional properties and amino acid content of a protein isolate from mung bean flour. J. Food Technol. 1977, 12, 473–484. [Google Scholar] [CrossRef]
- Gujral, H.S.; Rosell, C.M. Functionality of rice flour modified with a microbial transglutaminase. J. Cereal Sci. 2004, 39, 225–230. [Google Scholar] [CrossRef]
- Hama, F.; Savadogo, A.; Ouattara, C.A.T.; Traore, A. Biochemical, microbial and processing study of Dèguè a fermented food (from pearl millet dough) from Burkina Faso. Pak. J. Nutr. 2009, 8, 759–764. [Google Scholar] [CrossRef]
- Tchikoua, R. Application des Bactéries Lactiques Isolées du mais en Fermentation à L’amélioration de la Qualité Sanitaire et Physicochimique du Kutukutu. Ph.D. Thesis, National School of Agroindustrial Sciences, University of Ngaoundere, Ngaoundere, Cameroon, 2016; p. 233. [Google Scholar]
- Rojan, P.J.; Nampoothiri, K.M.; Pandey, A. Solid-state fermentation for lactic acid production from agro wastes using Lactobacillus delbrueckii. Process Biochem. 2006, 41, 759–763. [Google Scholar]
- Sade, O.F. Proximate, antinutritional factors and functional properties of processed pearl millet (Pennisetum glaucoma). J. Food Technol. 2009, 7, 92–97. [Google Scholar]
- Oboh, G.; Ademiluyi, O.A.; Akindahunsi, A.A. The effect of roasting on the nutritional and antioxidant properties of yellow and white maize varieties Int. J. Food Sci. Technol. 2010, 45, 1236–1242. [Google Scholar] [CrossRef]
- Makinde, M.F.; Akinoso, R. Comparison between the nutritional quality of flour obtained from raw, roasted and fermented sesame (Sesamum indicum L.) seed grown in Nigeria. Acta Sci. Pol. Technol. Aliment. 2014, 13, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Cuevas-Rodriguez, E.O.; MiIan-Carrillo, J.; Mora-Escobedo, R.; Cardenas-Valenzuela, O.G.; Reyes-Moreno, C. Quality protein maize (Zea mays L.) tempeh flour through solid state fermentation process. LWT Food Sci. Technol. 2004, 37, 59–67. [Google Scholar] [CrossRef]
- Lin, C.H.; Wei, Y.T.; Chou, C.C. Enhanced antioxidative activity of soybean koji prepared with various filamentous fungi. Food Microbiol. 2006, 23, 628–633. [Google Scholar] [CrossRef] [PubMed]
- Georgetti, S.R.; Vicentini, F.T.M.C.; Yokoyama, C.Y.; Borin, M.F.; Spadaro, A.C.C.; Fonseca, M.J.V. Enhanced in vitro and in vivo antioxidant activity and mobilization of free phenolic compounds of soybean flour fermented with different β-glucosidase-producing fungi. J. Appl. Microbiol. 2009, 106, 459–466. [Google Scholar] [CrossRef] [PubMed]
- McCue, P.; Horii, A.; Shetty, K. Mobilization of phenolic antioxidants from defatted soybean powders by Lentinus edobes during solid-state bioprocessing is associated with enhanced production of laccase. Innov. Food Sci. Emerg. Technol. 2004, 5, 385–392. [Google Scholar] [CrossRef]
- Egounlety, M.; Aworh, O.C. Effect of soaking, dehulling, cooking and fermentation with Rhizopus oligosporus on the oligosaccharides, trypsin inhibitor, phytic acid and tannins of soybean (Glycine max Merr.), cowpea (Vigna unguiculata L. Walp) and ground bean (Macrotyloma geocarpa Harms). J. Food Eng. 2003, 56, 249–254. [Google Scholar]
- Taylor, J.; Taylor, J.R.N. Alleviation of the adverse effects of cooking on protein digestibility in sorghum through fermentation in traditional African porridges. Int. J. Food Sci. Technol. 2002, 37, 129–138. [Google Scholar] [CrossRef]
- Gujral, H.S.; Sharma, P.; Rachna, S. Effect of sand roasting on beta glucan extractability, physicochemical and antioxidant properties of oats. LWT Food Sci. Technol. 2011, 44, 2223–2230. [Google Scholar] [CrossRef]
- Griffith, L.D.; Castell-Perez, M.E.; Griffith, M.E. Effects of Blend and Processing Method on the Nutritional Quality of Weaning Foods Made from Select Cereals and Legumes. Cereal Chem. 1998, 75, 105–112. [Google Scholar] [CrossRef]
- Wanink, J.F.; Wanink, T.; Nout, M.J.R. Effect of roasting and fermentation on viscosity of cereal-legume based food formulas. Plant Food Hum. Nutr. 1994, 46, 117–126. [Google Scholar] [CrossRef]
| Parameters (%) | Roasted Soybean Flour | Unroasted Soybean Flour | ||||||
|---|---|---|---|---|---|---|---|---|
| Unsoaked | Soaked 24 h | Soaked 48 h | Soaked 72 h | Unsoaked | Soaked 24 h | Soaked 48 h | Soaked 72 h | |
| Moisture * | 6.9 ± 0.5 a,b | 5.4 ± 0.5 b | 5.3 ± 0.3 b | 5.1 ± 0.5 b | 8.8 ± 1.6 a | 6.8 ± 0.3 a | 6.9 ± 0.4 a | 6.7 ± 0.4 a |
| Total carbohydrates | 27.0 ± 1.1 a | 26.9 ± 1.8 a | 25.1 ± 1.4 a | 24.3 ± 1.8 a | 27.8 ± 1.8 a | 22.8 ± 1.6 a | 27.9 ± 1.8 a | 24.2 ± 1.3 a |
| Total proteins | 44.1 ± 2.1 a,b | 35.8 ± 3.3 b | 37.0 ± 2.3 a,b | 35.5 ± 1.1 b | 46.0 ± 2.1 a | 43.9 ± 3.3 a,b | 37.9 ± 4.3 a,b | 35.8 ± 3.3 b |
| Crude lipids | 21.4 ± 1.4 c | 25.7 ± 1.3 a,b,c | 28.0 ± 1.6 a,b | 29.5 ± 1.2 a | 22.6 ± 1.1 c | 25.5 ± 1.5 a,b,c | 25.1 ± 1.8 b,c | 27.2 ± 1.2 a,b |
| Ash | 3.5 ± 0.2 a | 3.5 ± 0.1 a | 3.5 ± 0.1 a | 3.6 ± 0.1 a | 3.6 ± 0.1 a | 3.6 ± 0.1 a | 3.5 ± 0.1 a | 3.5 ± 0.1 a |
| Treatments | Parameters | |||||
|---|---|---|---|---|---|---|
| Roasting | Time of Soaking (h) | Peak Viscosity (cP) | Hold Viscosity (cP) | Final Viscosity (cP) | Breakdown Viscosity (cP) | Set back Viscosity (cP) |
| Unroasted | ||||||
| 0 | 210 ± 1 a | 91 ± 1 a | 103 ± 3 a | 119 ± 1 a | 12 ± 1 b | |
| 24 | 18 ± 2 e | 12 ± 3 e | 18 ± 2 f | 6 ± 1 c,d | 6 ± 1 c | |
| 48 | 27 ± 2 d | 22 ± 2 d | 35 ± 1 d | 5 ± 1 c,d | 13 ± 1 b | |
| 72 | 104 ± 3 b | 69 ± 3 b | 95 ± 1 b | 35 ± 1 b | 26 ± 1 a | |
| Roasted | ||||||
| 0 | 55 ± 4 c | 48 ± 2 c | 61 ± 2 c | 7 ± 1 c | 13 ± 1 b | |
| 24 | 20 ± 3 d,e | 17 ± 2 d,e | 26 ± 1 e | 3 ± 1 d | 9 ± 1 c | |
| 48 | 26 ± 2 d | 19 ± 3 d,e | 26 ± 1 e | 7 ± 1 c | 7 ± 1 c | |
| 72 | 23 ± 1 d | 19 ± 2 d | 25 ± 1 e | 6 ± 1 c,d | 6 ± 1 c | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agume, A.S.N.; Njintang, N.Y.; Mbofung, C.M.F. Effect of Soaking and Roasting on the Physicochemical and Pasting Properties of Soybean Flour. Foods 2017, 6, 12. https://doi.org/10.3390/foods6020012
Agume ASN, Njintang NY, Mbofung CMF. Effect of Soaking and Roasting on the Physicochemical and Pasting Properties of Soybean Flour. Foods. 2017; 6(2):12. https://doi.org/10.3390/foods6020012
Chicago/Turabian StyleAgume, Aurelie Solange Ntso, Nicolas Yanou Njintang, and Carl Moses F. Mbofung. 2017. "Effect of Soaking and Roasting on the Physicochemical and Pasting Properties of Soybean Flour" Foods 6, no. 2: 12. https://doi.org/10.3390/foods6020012
APA StyleAgume, A. S. N., Njintang, N. Y., & Mbofung, C. M. F. (2017). Effect of Soaking and Roasting on the Physicochemical and Pasting Properties of Soybean Flour. Foods, 6(2), 12. https://doi.org/10.3390/foods6020012





