Evaluation of Electrolytically-Generated Hypochlorous Acid (‘Electrolyzed Water’) for Sanitation of Meat and Meat-Contact Surfaces
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains
2.2. Electrolyzed Water Generation
2.3. Water Analysis
2.4. Effect of Electrolyzed Water on Bacterial Pathogens
2.5. Spray Systems
2.6. Efficacy of Electrolyzed Water in the Presence of Various Protein Levels
2.7. Efficacy of Electrolyzed Water on Contaminated Slicing Blades
2.8. Effect of EW on L. monocytogenes Biofilms
2.9. Statistics
3. Results and Discussion
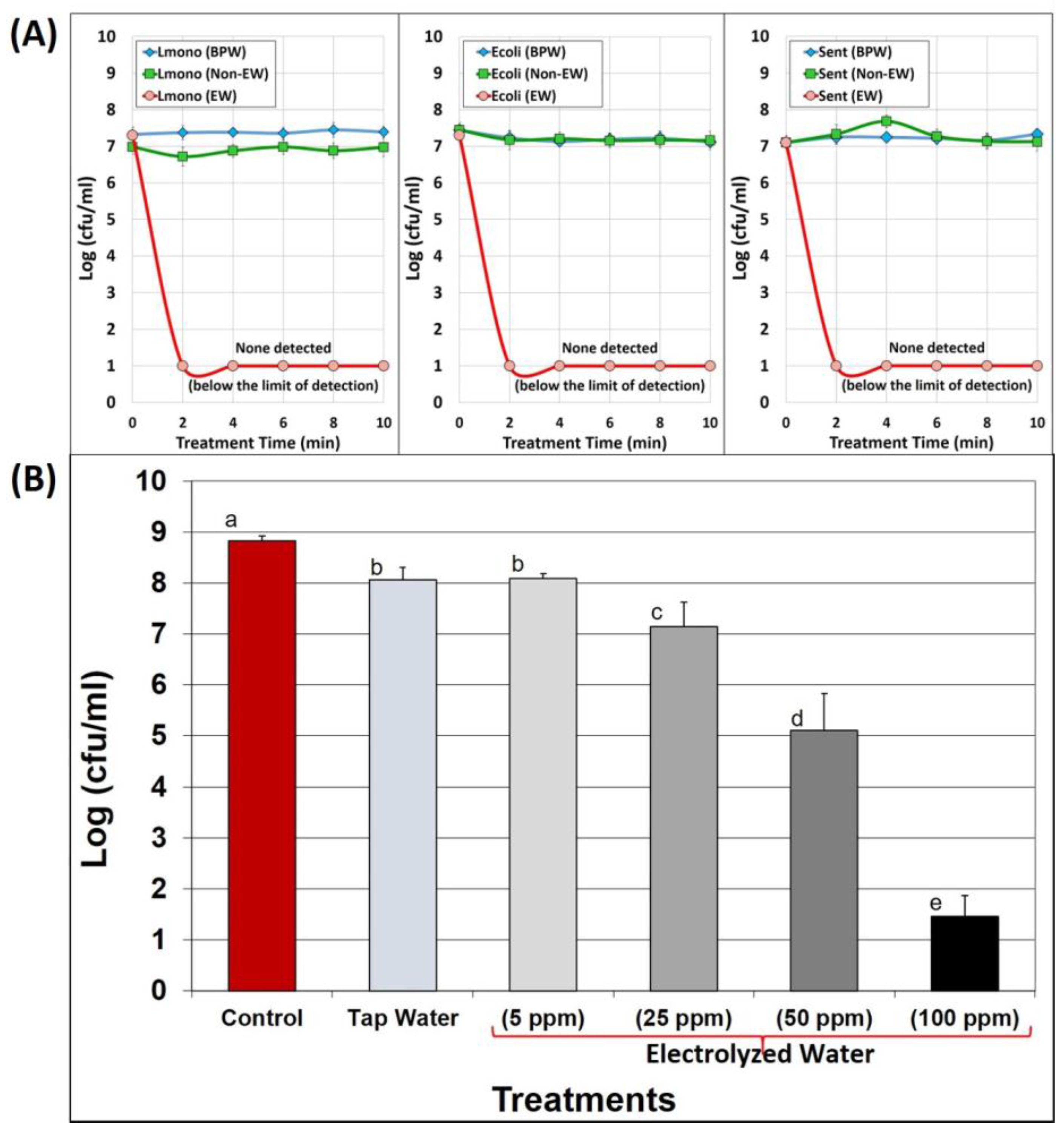
3.1. Antimicrobial Activity of EW Solutions
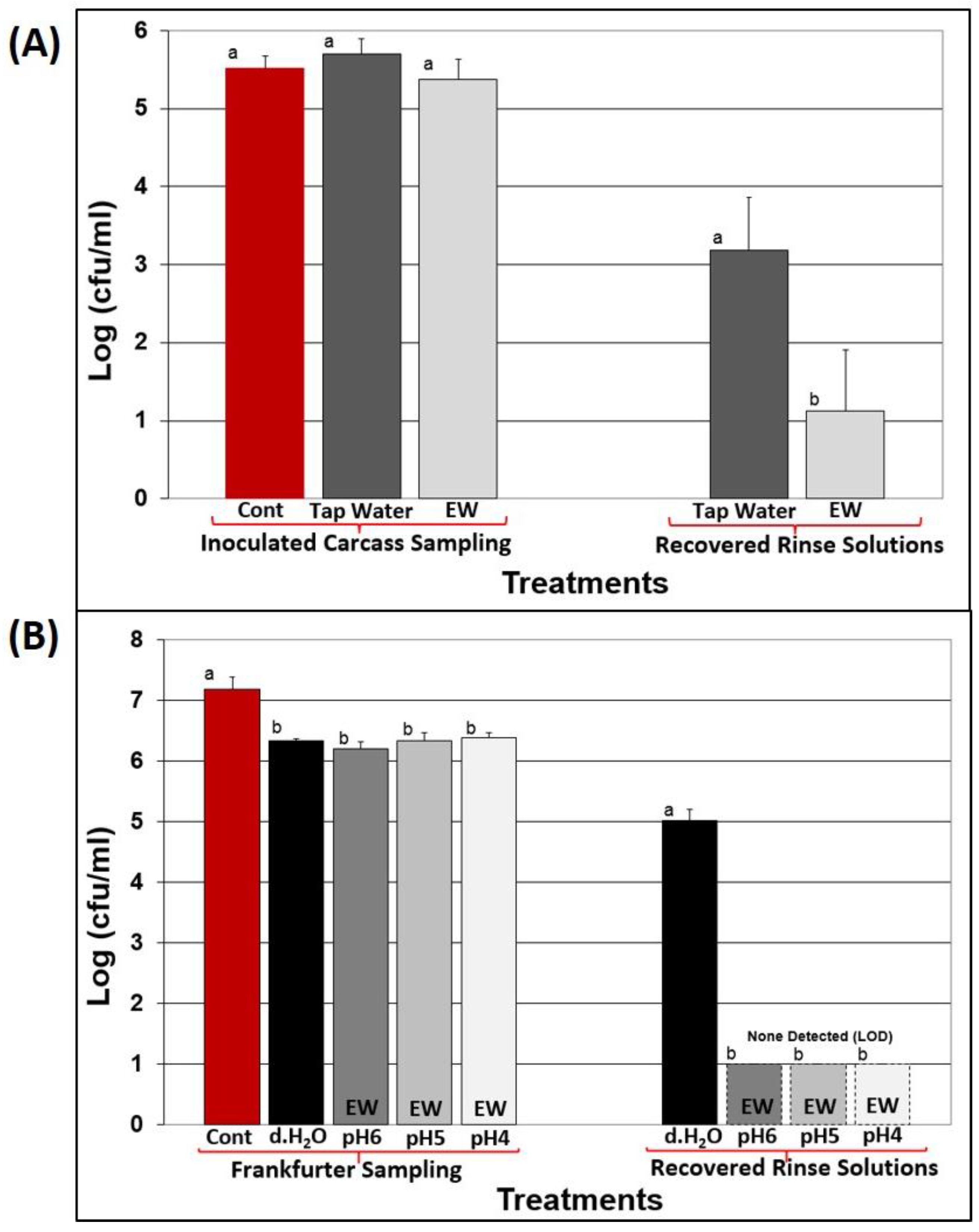
3.2. Application of EW to Raw Beef Carcasses
3.3. Antimicrobial Treatments with RTE Meats
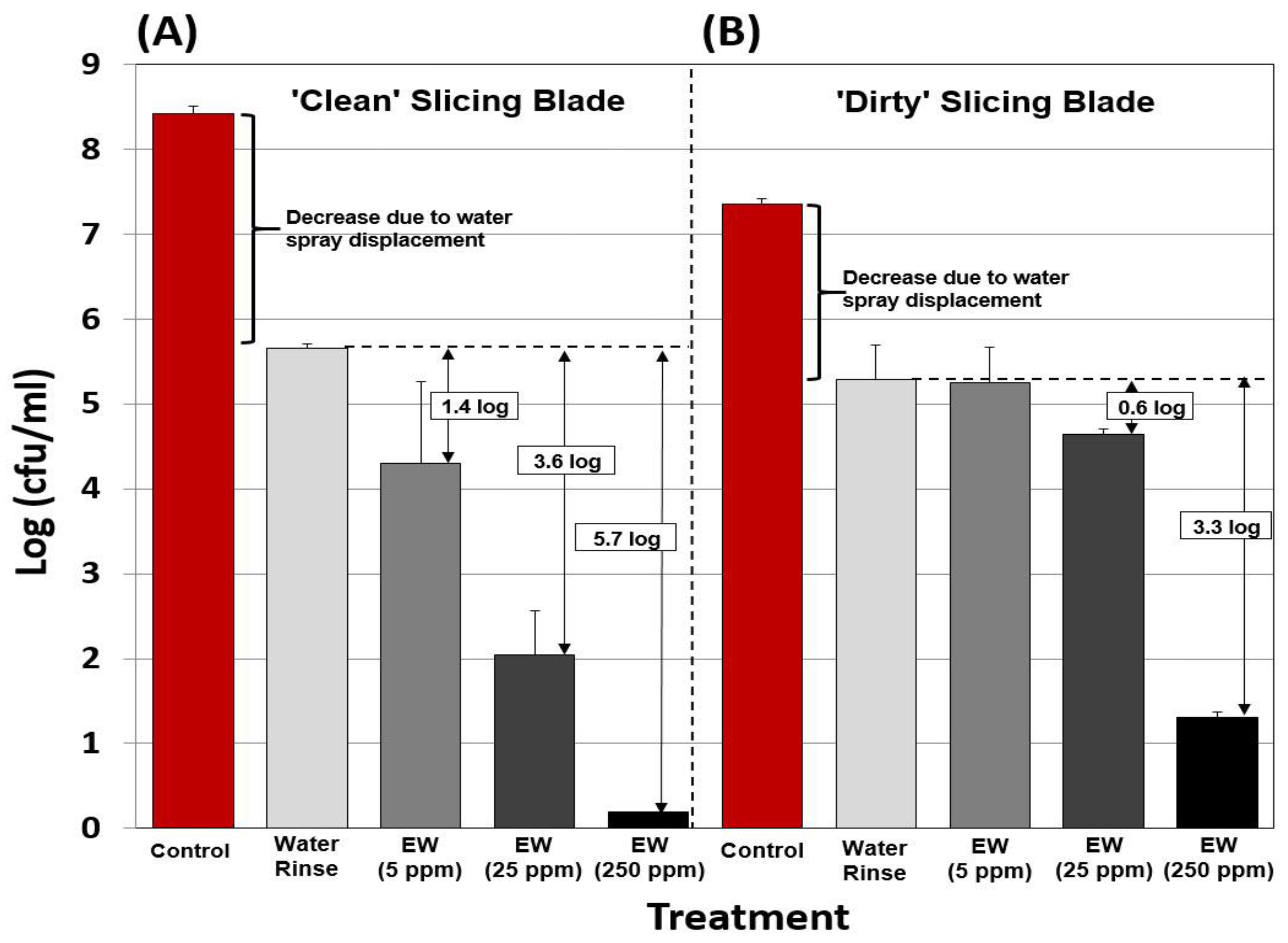
3.4. Antimicrobial Activity of EW on Slicing Blades
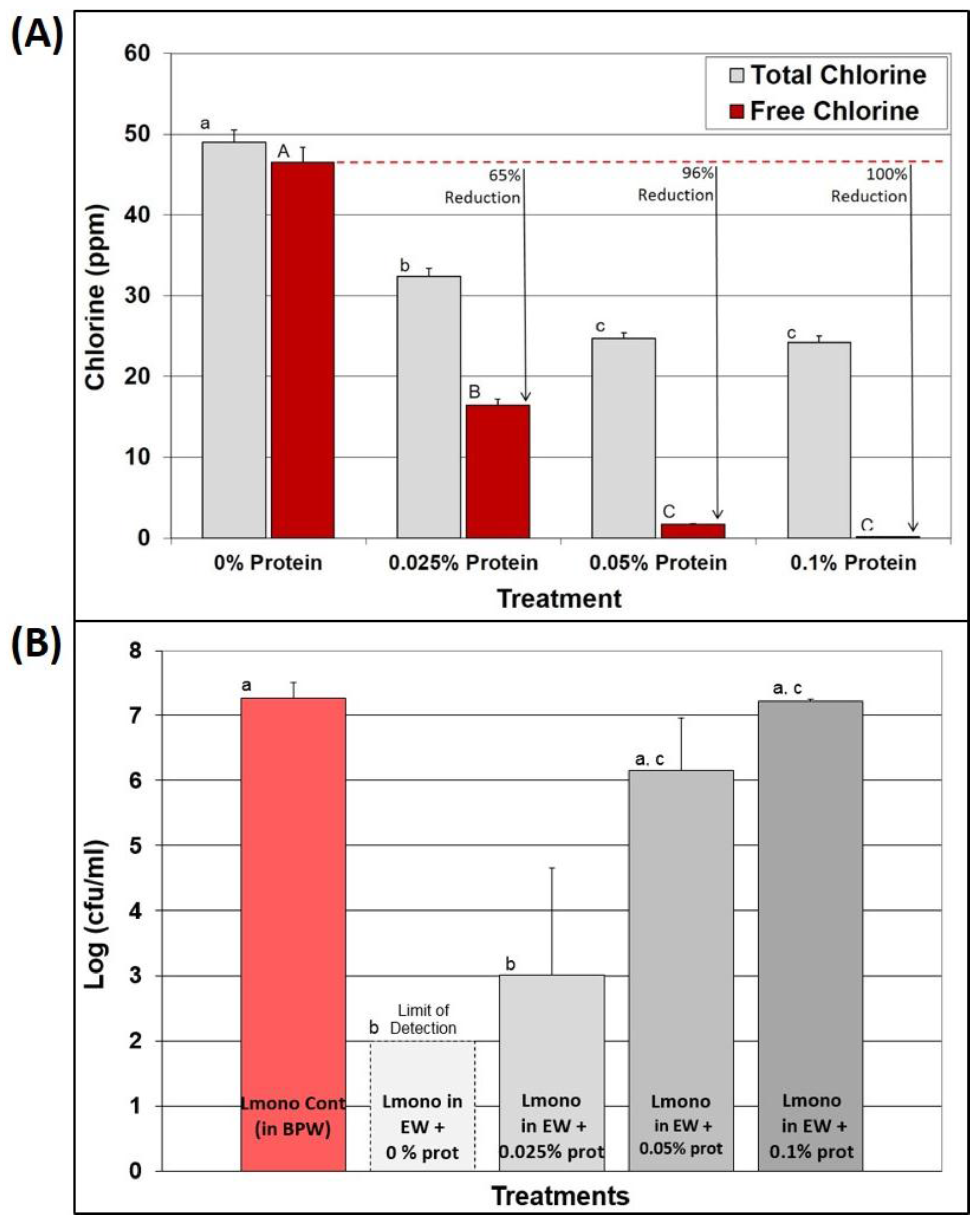
3.5. Effect of EW in the Presence of Known Protein Levels
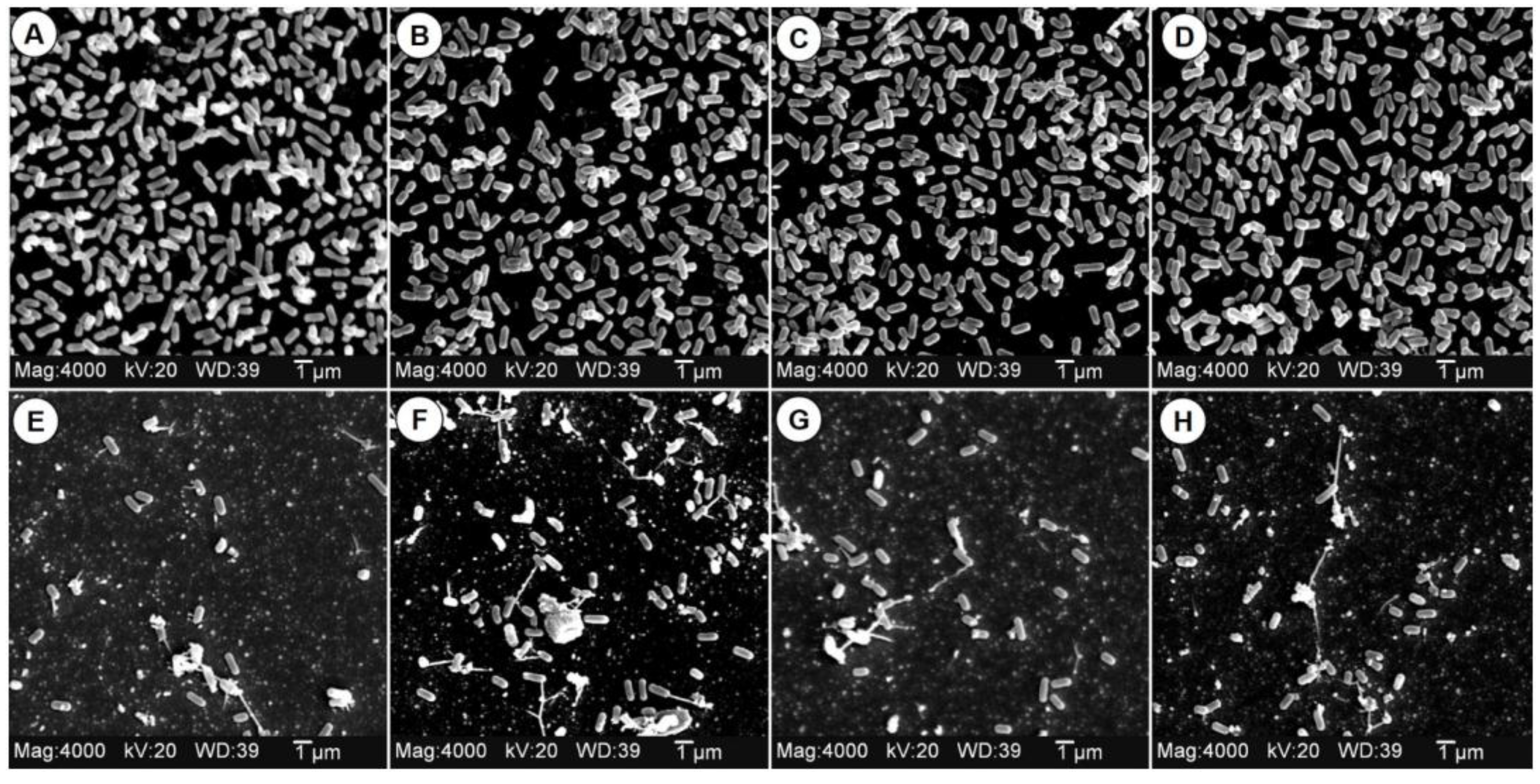
3.6. Effect of EW on L. monocytogenes Biofilms
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| EW | Electrolyzed water |
| FAC | Free available chlorine |
| RTE | Ready-to-eat |
| USDA | United States department of agriculture |
| FSIS | Food safety and inspection service |
| HOCL | Hypochlorous acid |
| PPM | Parts per million |
| OCL | Hypochlorite |
| ECA | Electrochemically activated |
| ATTC | American Type Tissue Collection |
| PSI | pounds per square inch |
| PVC | polyvinyl chloride |
| ORP | Oxidation reduction potential |
| BPW | Buffered peptone water |
| TSA | Tryptic soy agar |
| ANOVA | Analysis of variance |
References
- USDA-FSIS. Compliance Guidelines to Control Listeria monocytogenes in Post-Lethality Exposed Ready-to-Eat Meat and Poultry Products; USDA-FSIS: Washington, DC, USA, 2012. [Google Scholar]
- Chantarapanont, W.; Berrang, M.E.; Frank, J.F. Direct microscopic observation of viability of Campylobacter jejuni on chicken skin treated with selected chemical sanitizing agents. J. Food Prot. 2004, 67, 1146–1152. [Google Scholar] [PubMed]
- Stopforth, J.D.; Yoon, Y.; Belk, K.E.; Scanga, J.A.; Kendall, P.A.; Smith, G.C.; Sofos, J.N. Effect of simulated spray chilling with chemical solutions on acid-habituated and non-acid-habituated Escherichia coli O157:H7 cells attached to beef carcass tissue. J. Food Prot. 2004, 67, 2099–2106. [Google Scholar] [PubMed]
- Van Netten, P.; Mossel, D.A.A.; Huis, J.H.J. Microbial changes on freshly slaughtered pork carcasses due to “hot” lactic acid decontamination. J. Food Saf. 1997, 17, 89–111. [Google Scholar] [CrossRef]
- Juneja, V.K.; Thippareddi, H. Control of Clostridium perfringens in a model roast beef by salts of organic acids during chilling. J. Food Saf. 2004, 24, 95–108. [Google Scholar] [CrossRef]
- Qvist, S.; Sehested, K.; Zeuthen, P. Growth suppression of Listeria monocytogenes in a meat product. Int. J. Food Microbiol. 1994, 24, 283–293. [Google Scholar] [CrossRef]
- Serdengecti, N.; Yildirim, I.; Gokoglu, N. Investigation of inhibitory effects of several combinations of sodium slats on the growth of Listeria monocytogenes and Salmonella enterica serotype Enteritidis in minced beef. J. Food Saf. 2006, 26, 233–243. [Google Scholar] [CrossRef]
- USDA-FSIS. Safe and Suitable Ingredients Used in the Production of Meat and Poultry, and Egg Products; USDA-FSIS: Washington, DC, USA, 2016. [Google Scholar]
- U.S. Environmental Protection Agency. Alternative Disinfectants and Oxidants Guidance Manual; US-EPA: Washington, DC, USA, 1999.
- Albrich, J.M.; Gilbaugh, J.H., 3rd; Callahan, K.B.; Hurst, J.K. Effects of the putative neutrophil-generated toxin, hypochlorous acid, on membrane permeability and transport systems of Escherichia coli. J. Clin. Investig. 1986, 78, 177–184. [Google Scholar] [CrossRef] [PubMed]
- CDC. Ongoing multistate outbreak of Escherichia coli serotype O157:H7 infections associated with consumption of fresh spinach—United States, September 2006. MMWR 2006, 55, 1045–1046. [Google Scholar]
- Barrette, W.C.; Hannum, D.M.; Wheeler, W.D.; Hurst, J.K. General mechanism for the bacterial toxicity of hypochlorous acid: Abolition of ATP production. Biochemistry 1989, 28, 9172–9178. [Google Scholar] [CrossRef] [PubMed]
- Hurst, J.K.; Barrette, W.C.; Michel, B.R.; Rosen, H. Hypochlorous acid and myeloperoxidase-catalyzed oxidation of iron-sulfur clusters in bacterial respiratory dehydrogenases. Eur. J. Biochem. 1991, 202, 1275–1282. [Google Scholar] [CrossRef] [PubMed]
- Ayebah, B.; Hung, Y.C.; Frank, J.F. Enhancing the bactericidal effect of electrolyzed water on Listeria monocytogenes biofilms formed on stainless steel. J. Food Prot. 2005, 68, 1375–1380. [Google Scholar] [PubMed]
- Fabrizio, K.A.; Cutter, C.N. Stability of electrolyzed oxidizing water and its efficacy against cell suspensions of Salmonella Typhimurium and Listeria monocytogenes. J. Food Prot. 2003, 66, 1379–1384. [Google Scholar] [PubMed]
- Park, H.; Hung, Y.C.; Chung, D. Effects of chlorine and pH on efficacy of electrolyzed water for inactivating Escherichia coli O157:H7 and Listeria monocytogenes. Int. J. Food Microbiol. 2004, 91, 13–18. [Google Scholar] [CrossRef]
- Venkitanarayanan, K.S.; Ezeike, G.O.; Hung, Y.C.; Doyle, M.P. Efficacy of electrolyzed oxidizing water for inactivating Escherichia coli O157:H7, Salmonella Enteritidis, and Listeria monocytogenes. Appl. Environ. Microbiol. 1999, 65, 4276–4279. [Google Scholar] [PubMed]
- Ayebah, B.; Hung, Y.C.; Kim, C.; Frank, J.F. Efficacy of electrolyzed water in the inactivation of planktonic and biofilm Listeria monocytogenes in the presence of organic matter. J. Food Prot. 2006, 69, 2143–2150. [Google Scholar] [PubMed]
- Deza, M.A.; Araujo, M.; Garrido, M.J. Efficacy of neutral electrolyzed water to inactivate Escherichia coli, Listeria monocytogenes, Pseudomonas aeruginosa, and Staphylococcus aureus on plastic and wooden kitchen cutting boards. J. Food Prot. 2007, 70, 102–108. [Google Scholar] [PubMed]
- Park, H.; Hung, Y.C.; Kim, C. Effectiveness of electrolyzed water as a sanitizer for treating different surfaces. J. Food Prot. 2002, 65, 1276–1280. [Google Scholar] [PubMed]
- Lin, C.S.; Wu, C.; Yeh, J.Y.; Saalia, F.K. The evaluation of electrolysed water as an agent for reducing micro-organisms on vegetables. Int. J. Food Sci. Technol. 2005, 40, 495–500. [Google Scholar] [CrossRef]
- Deza, M.A.; Araujo, M.; Garrido, M.J. Inactivation of Escherichia coli O157:H7, Salmonella Enteritidis and Listeria monocytogenes on the surface of tomatoes by neutral electrolyzed water. Lett. Appl. Microbiol. 2003, 37, 482–487. [Google Scholar] [CrossRef] [PubMed]
- Izumi, H. Electrolyzed water as a disinfectant for fresh-cut vegetables. J. Food Sci. 1999, 64, 536–539. [Google Scholar] [CrossRef]
- Koseki, S.; Isobe, S.; Itoh, K. Efficacy of acidic electrolyzed water ice for pathogen control on lettuce. J. Food Prot. 2004, 67, 2544–2549. [Google Scholar] [PubMed]
- Koseki, S.; Yoshida, K.; Isobe, S.; Itoh, K. Efficacy of acidic electrolyzed water for microbial decontamination of cucumbers and strawberries. J. Food Prot. 2004, 67, 1247–1251. [Google Scholar] [PubMed]
- Wang, H.; Feng, H.; Luo, Y. Microbial reduction and storage quality of fresh-cut cilantro washed with acidic electrolyzed water and aqueous ozone. Food Res. Int. 2004, 37, 949–956. [Google Scholar] [CrossRef]
- Yang, H.; Swem, B.L.; Li, Y. The Effect of pH on inactivation of pathogenic bacteria on fresh-cut lettuce by dipping treatment with electrolyzed water. J. Food Sci. 2003, 68, 1013–1017. [Google Scholar] [CrossRef]
- Kim, C.; Hung, Y.C.; Russell, S.M. Efficacy of electrolyzed water in the prevention and removal of fecal material attachment and its microbicidal effectiveness during simulated industrial poultry processing. Poult. Sci. 2005, 84, 1778–1784. [Google Scholar] [CrossRef] [PubMed]
- Park, C.M.; Hung, Y.C.; Lin, C.S.; Brackett, R.E. Efficacy of electrolyzed water in inactivating Salmonella Enteritidis and Listeria monocytogenes on shell eggs. J. Food Prot. 2005, 68, 986–990. [Google Scholar] [PubMed]
- Russell, S.M. The effect of electrolyzed oxidative water applied using electrostatic spraying on pathogenic and indicator bacteria on the surface of eggs. Poult. Sci. 2003, 82, 158–162. [Google Scholar] [CrossRef] [PubMed]
- Hrudey, S.E. Chlorination disinfection by-products, public health risk tradeoffs and me. Water Res. 2009, 43, 2057–2092. [Google Scholar] [CrossRef] [PubMed]
- Gamble, R.; Muriana, P.M. Microplate fluorescence assay for measurement of the ability of strains of Listeria monocytogenes from meat and meat-processing plants to adhere to abiotic surfaces. Appl. Environ. Microbiol. 2007, 73, 5235–5244. [Google Scholar] [CrossRef] [PubMed]
- Fabrizio, K.A.; Cutter, C.N. Comparison of electrolyzed oxidizing water with other antimicrobial interventions to reduce pathogens on fresh pork. Meat Sci. 2004, 68, 463–468. [Google Scholar] [CrossRef] [PubMed]
- Fabrizio, K.A.; Cutter, C.N. Application of electrolyzed oxidizing water to reduce Listeria monocytogenes on ready-to-eat meats. Meat Sci. 2005, 71, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Kiura, H.; Sano, K.; Morimatsu, S.; Nakano, T.; Morita, C.; Yamaguchi, M.; Maeda, T.; Katsuoka, Y. Bactericidal activity of electrolyzed acid water from solution containing sodium chloride at low concentration, in comparison with that at high concentration. J. Microbiol. Methods. 2002, 49, 285–293. [Google Scholar] [CrossRef]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Veasey, S.; Muriana, P.M. Evaluation of Electrolytically-Generated Hypochlorous Acid (‘Electrolyzed Water’) for Sanitation of Meat and Meat-Contact Surfaces. Foods 2016, 5, 42. https://doi.org/10.3390/foods5020042
Veasey S, Muriana PM. Evaluation of Electrolytically-Generated Hypochlorous Acid (‘Electrolyzed Water’) for Sanitation of Meat and Meat-Contact Surfaces. Foods. 2016; 5(2):42. https://doi.org/10.3390/foods5020042
Chicago/Turabian StyleVeasey, Shawnna, and Peter M. Muriana. 2016. "Evaluation of Electrolytically-Generated Hypochlorous Acid (‘Electrolyzed Water’) for Sanitation of Meat and Meat-Contact Surfaces" Foods 5, no. 2: 42. https://doi.org/10.3390/foods5020042
APA StyleVeasey, S., & Muriana, P. M. (2016). Evaluation of Electrolytically-Generated Hypochlorous Acid (‘Electrolyzed Water’) for Sanitation of Meat and Meat-Contact Surfaces. Foods, 5(2), 42. https://doi.org/10.3390/foods5020042







