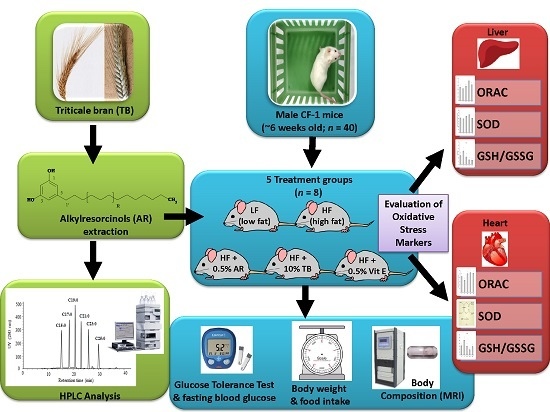
Triticale Bran Alkylresorcinols Enhance Resistance to Oxidative Stress in Mice Fed a High-Fat Diet
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Sample Preparation and Analysis
2.3. Animals and Diets
| Ingredient | Low-Fat Diet | High-Fat Diet |
|---|---|---|
| Carbohydrate (%) | 48.0 | 27.3 |
| Protein (%) | 14.3 | 23.5 |
| Fat (%) | 4.0 | 34.3 |
| Saturated (%) | 0.7 | 12.7 |
| Monounsaturated (%) | 0.8 | 16.1 |
| Polyunsaturated (%) | 2.5 | 5.5 |
| Fibre (%) | 22.1 | 6.5 |
| Soluble (%) | 4.1 | − |
| Insoluble (%) | 18.0 | 6.5 |
| Energy density (kcal/g) | 2.9 | 5.1 |
| Energy from carbohydrate (%) | 67.3 | 21.3 |
| Energy from protein (%) | 20.1 | 18.4 |
| Energy from fat (%) | 12.6 | 60.3 |
2.4. Glucose Tolerance Test (GTT)
2.5. Carcass Analysis
2.6. Tissue Preparation
2.7. Protein Determination
2.8. Antioxidant Activity Assays
2.8.1. Oxygen Radical Absorbance Capacity (ORAC) Assay
2.8.2. Super Oxide Dismutase (SOD) Activity
2.8.3. Gluatathione (GSH) Assay
2.9. Statistical Analysis
3. Results
3.1. Characterization of ARs Extracts from TB
| AR Homologue (mg/100 g) | |
|---|---|
| Saturated | |
| 5-n-heptadecylresorcinol C 15:0 | 1.06 ± 0.2 |
| 5-n-heptadecylresorcinol C 17:0 | 12.44 ± 0.5 |
| 5-n-nonadecanylresorcinol C 19:0 | 28.08 ± 0.3 |
| 5-n-heneicosylresorcinol C 21:0 | 39.76 ± 0.3 |
| 5-n-tricosylresorcinol C 23:0 | 20.33 ± 0.2 |
| 5-n-pentacosylresorcinol C 25:0 | 15.86 ± 0.1 |
| Unsaturated | 12.12 ± 0.2 |
| Unknown | 13.61 ± 0.6 |
| Total | 143.29 ± 0.3 |
3.2. Weight and Intake Parameters
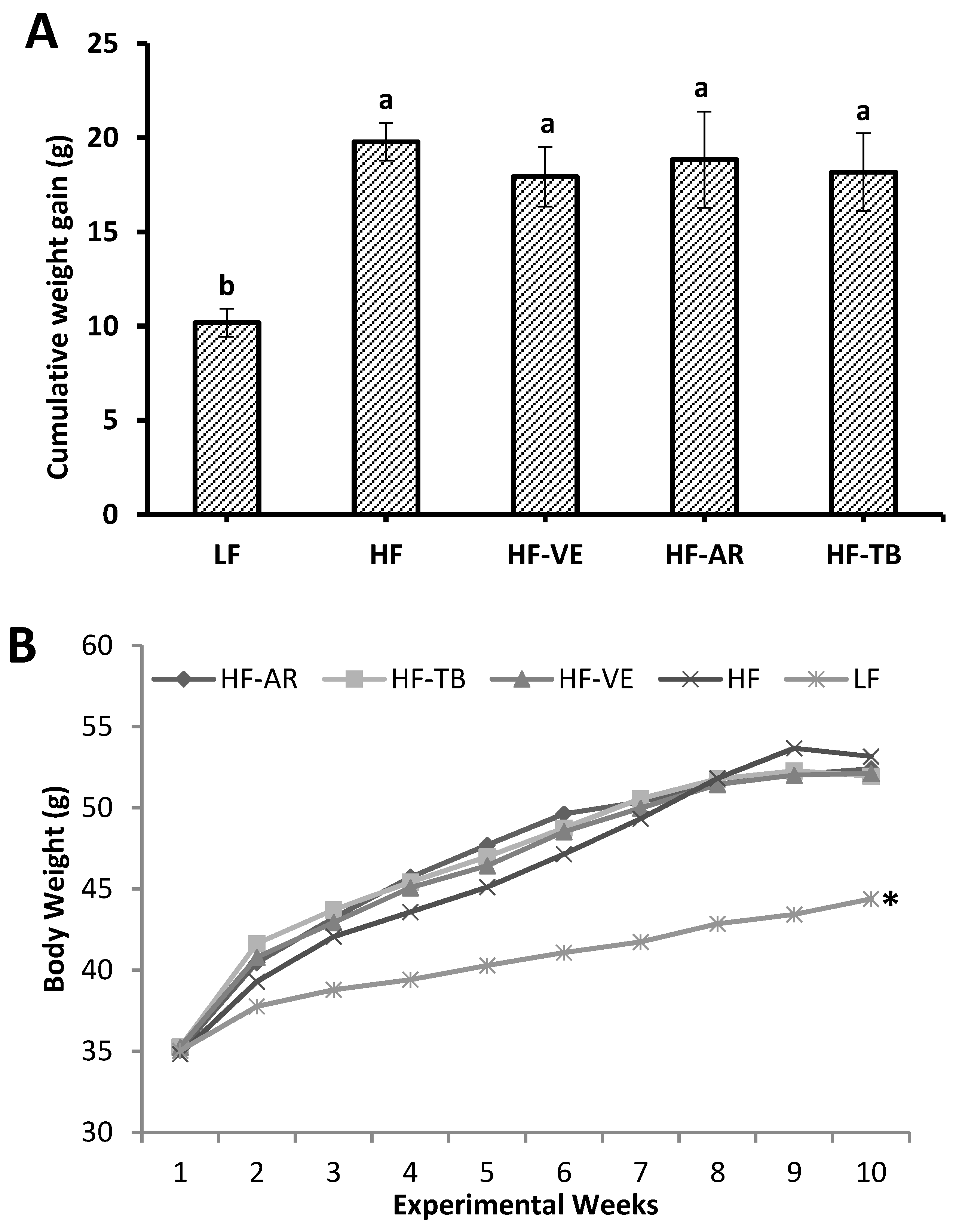

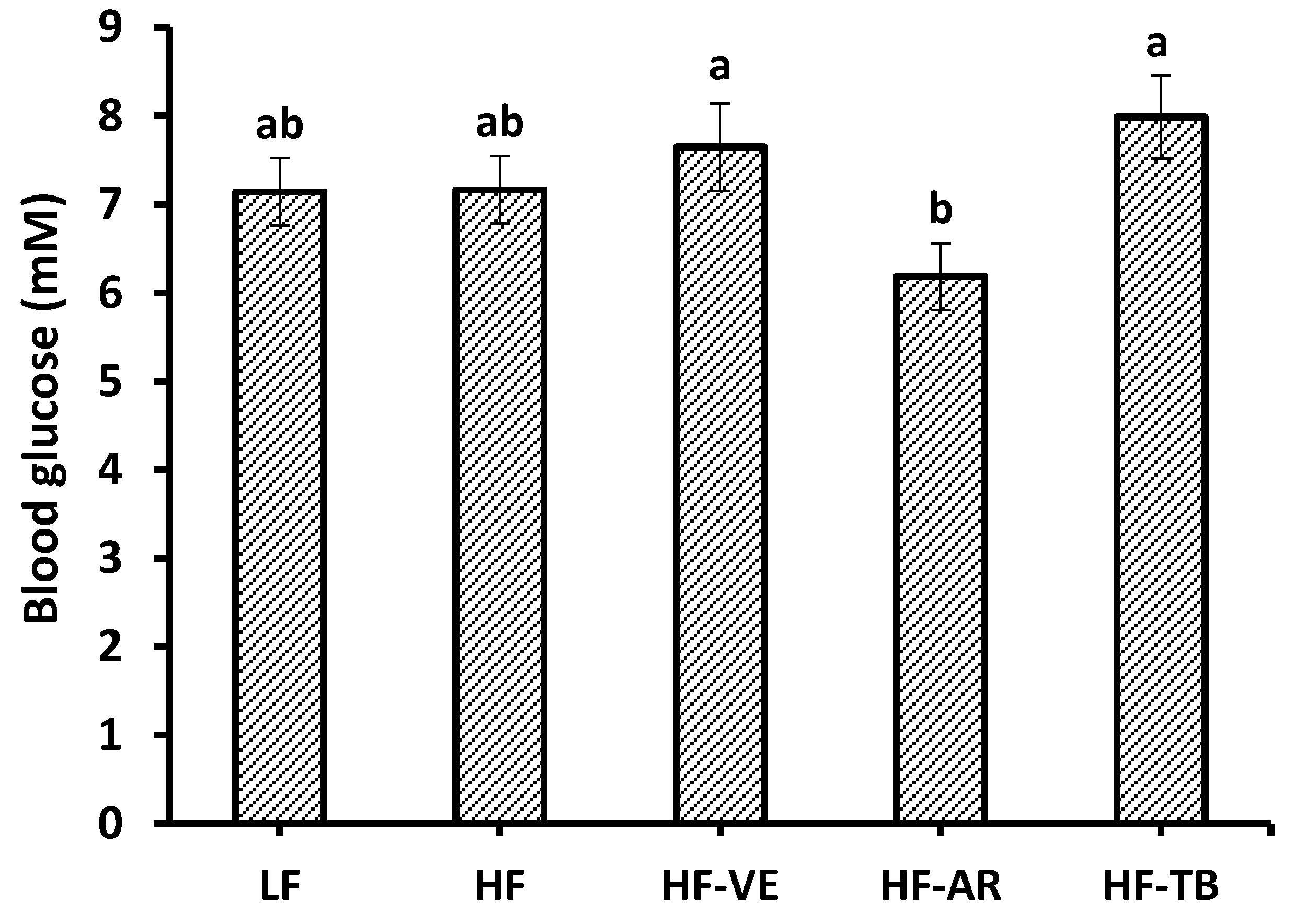
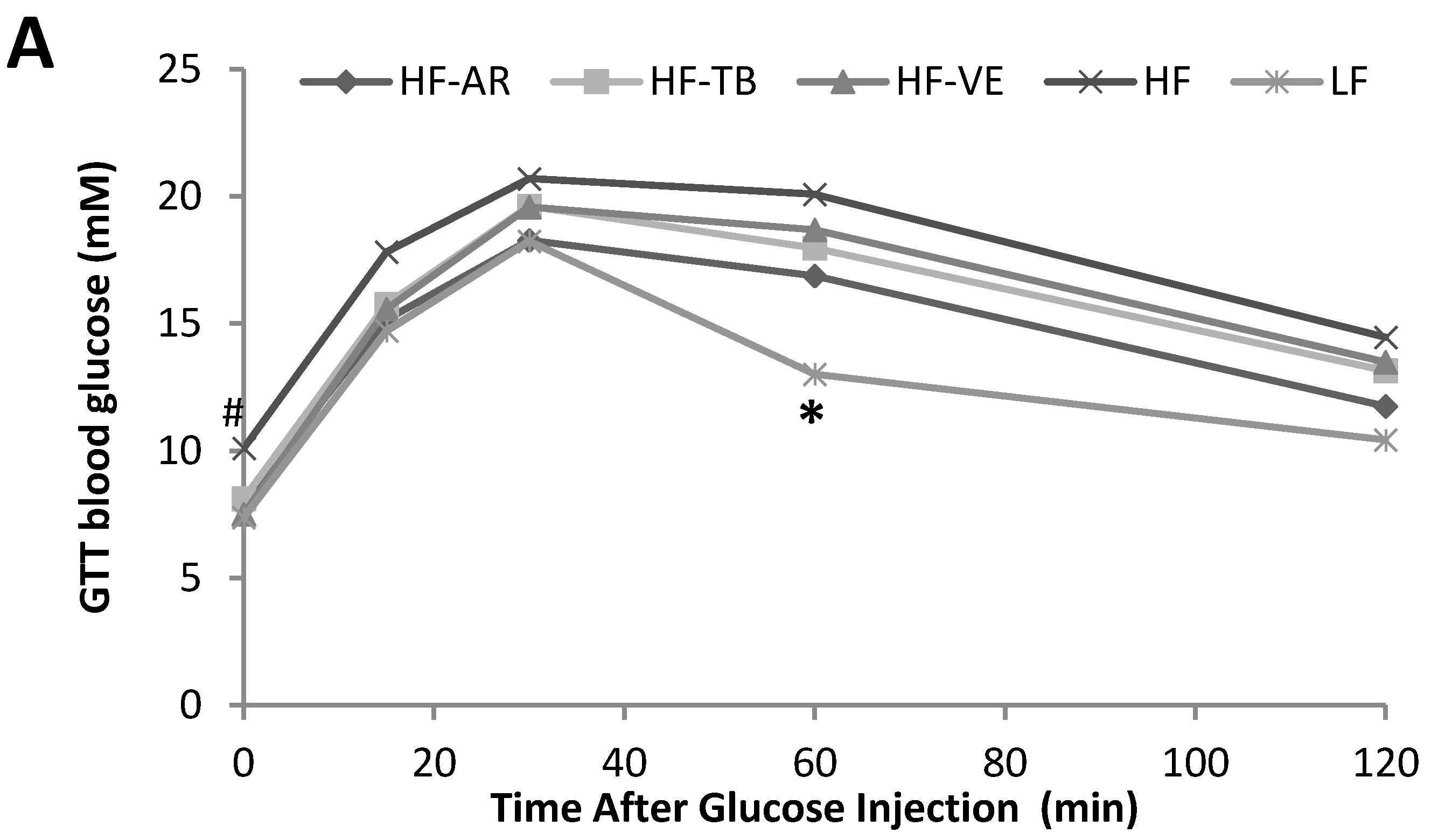
3.3. Glucose Tolerance and Fasting Blood Glucose
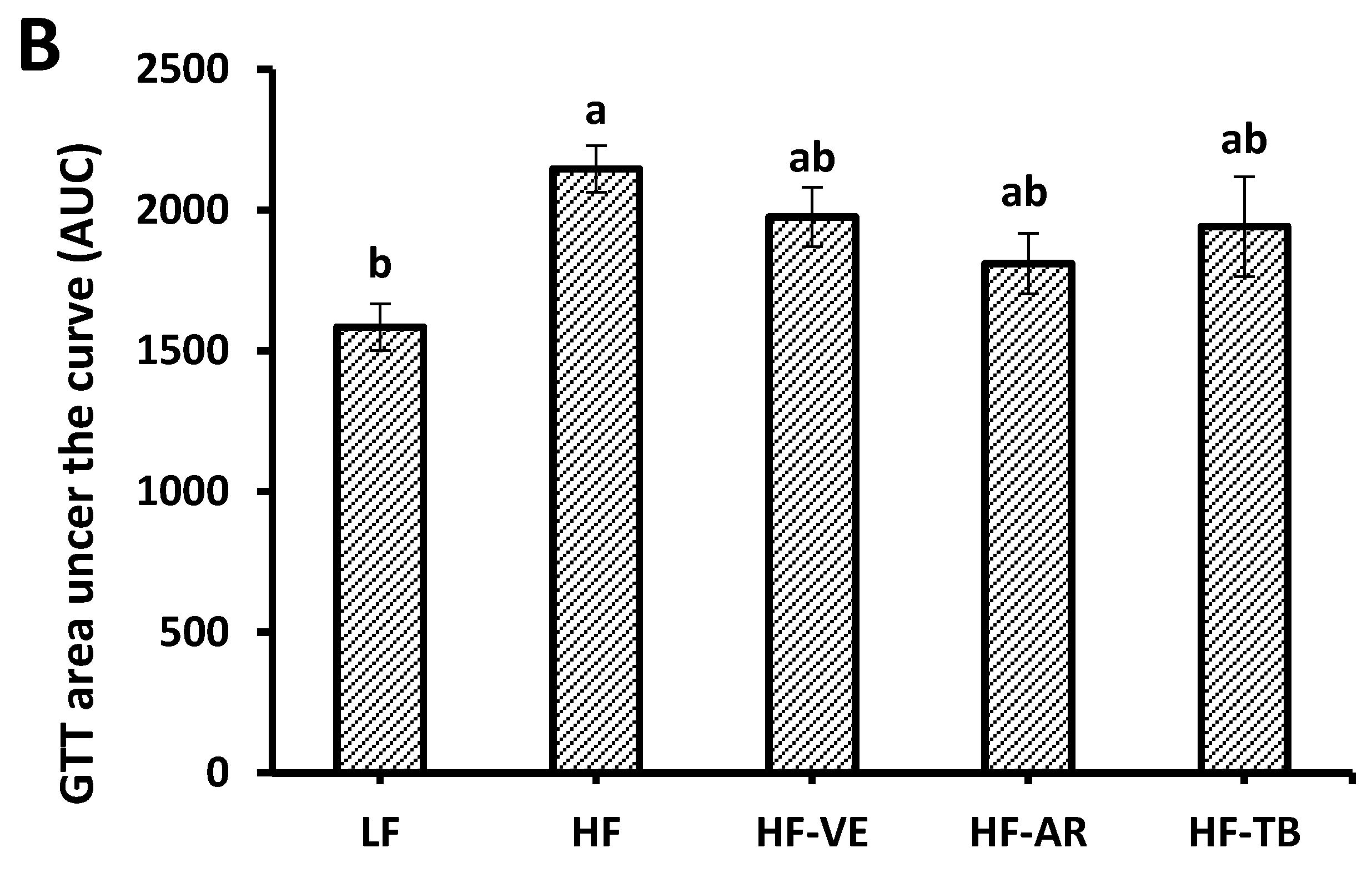
3.4. Body Composition
| Treatment 1 | Weight (g) | Fat (%) | Lean Muscle (%) |
|---|---|---|---|
| LF | 36.60 ± 1.35 b | 18.02 ± 1.00 b | 75.90 ± 0.89 a |
| HF | 46.66 ± 1.06 a | 27.00 ± 1.03 a | 69.89 ± 1.35 c |
| HF-VE | 45.17 ± 2.18 a | 26.09 ± 0.63 a | 70.87 ± 0.62 bc |
| HF-AR | 45.83 ± 3.15 a | 28.10 ± 1.92 a | 67.79 ± 1.89 c |
| HF-TB | 45.06 ± 2.32 a | 24.14 ± 1.35 a | 72.87 ± 1.43 ab |
3.5. Oxygen Radical Absorbance Capacity (ORAC)
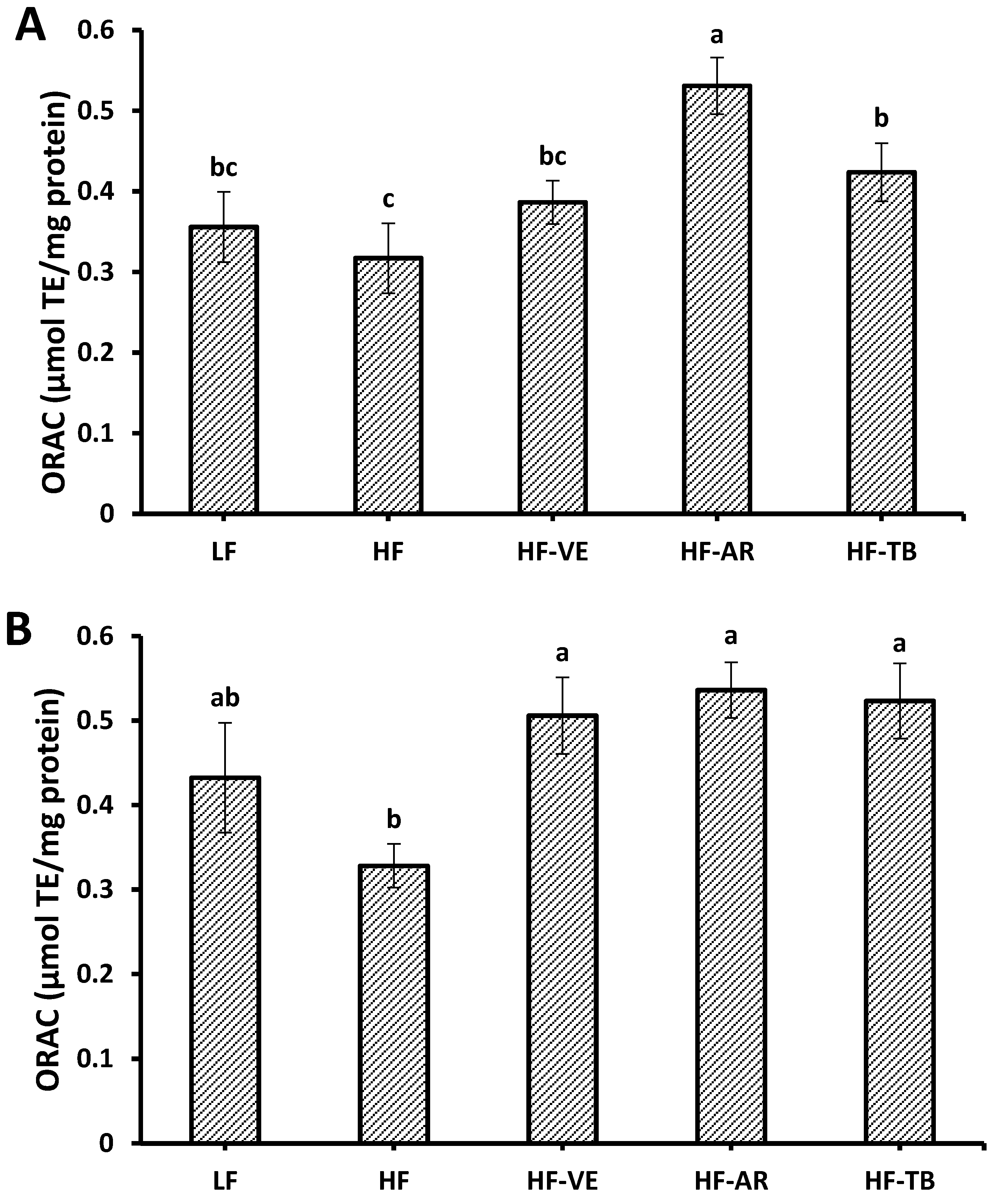
3.6. Superoxide Dismutase (SOD) Activity
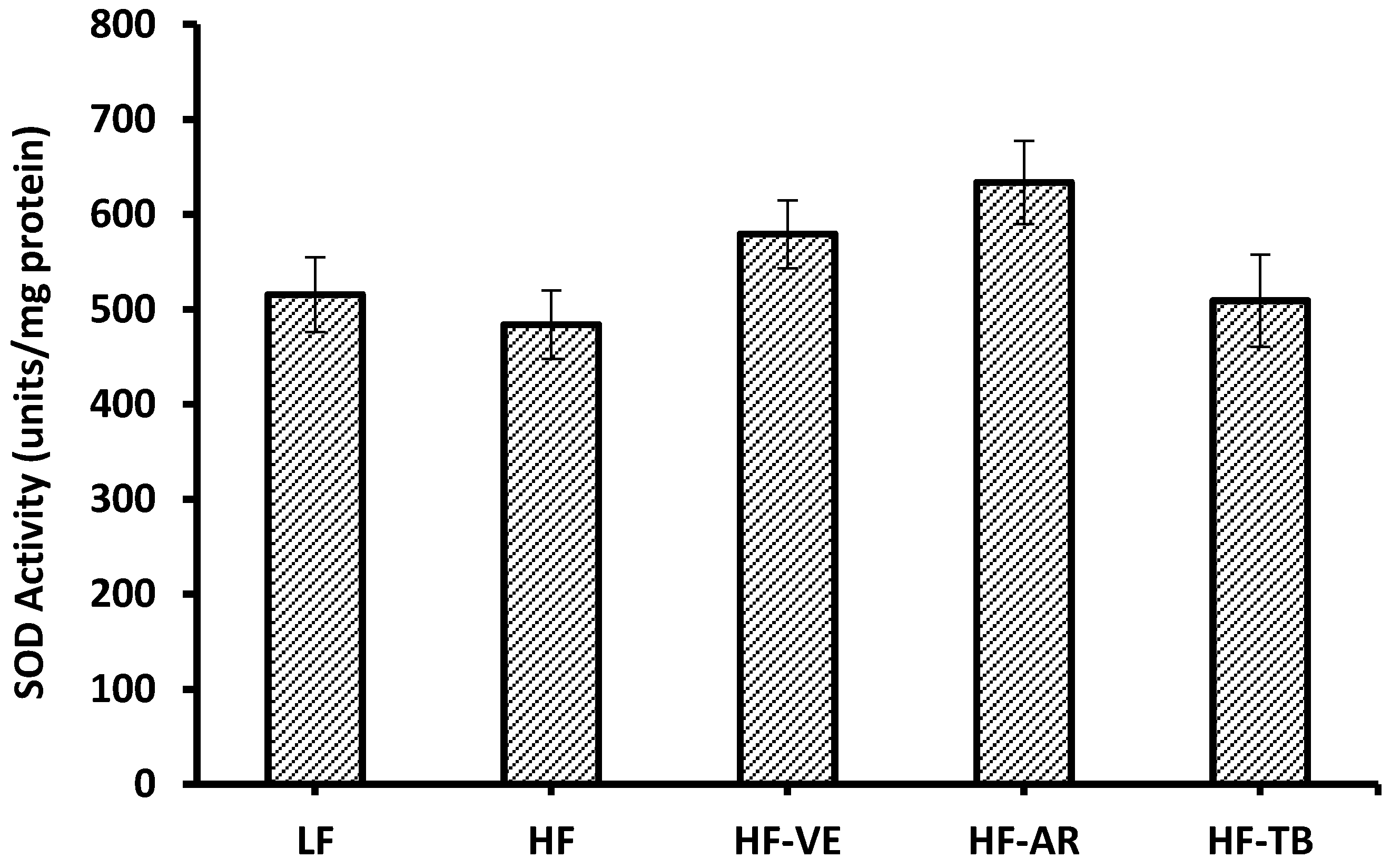
3.7. Reduced Glutathione (GSH) Content
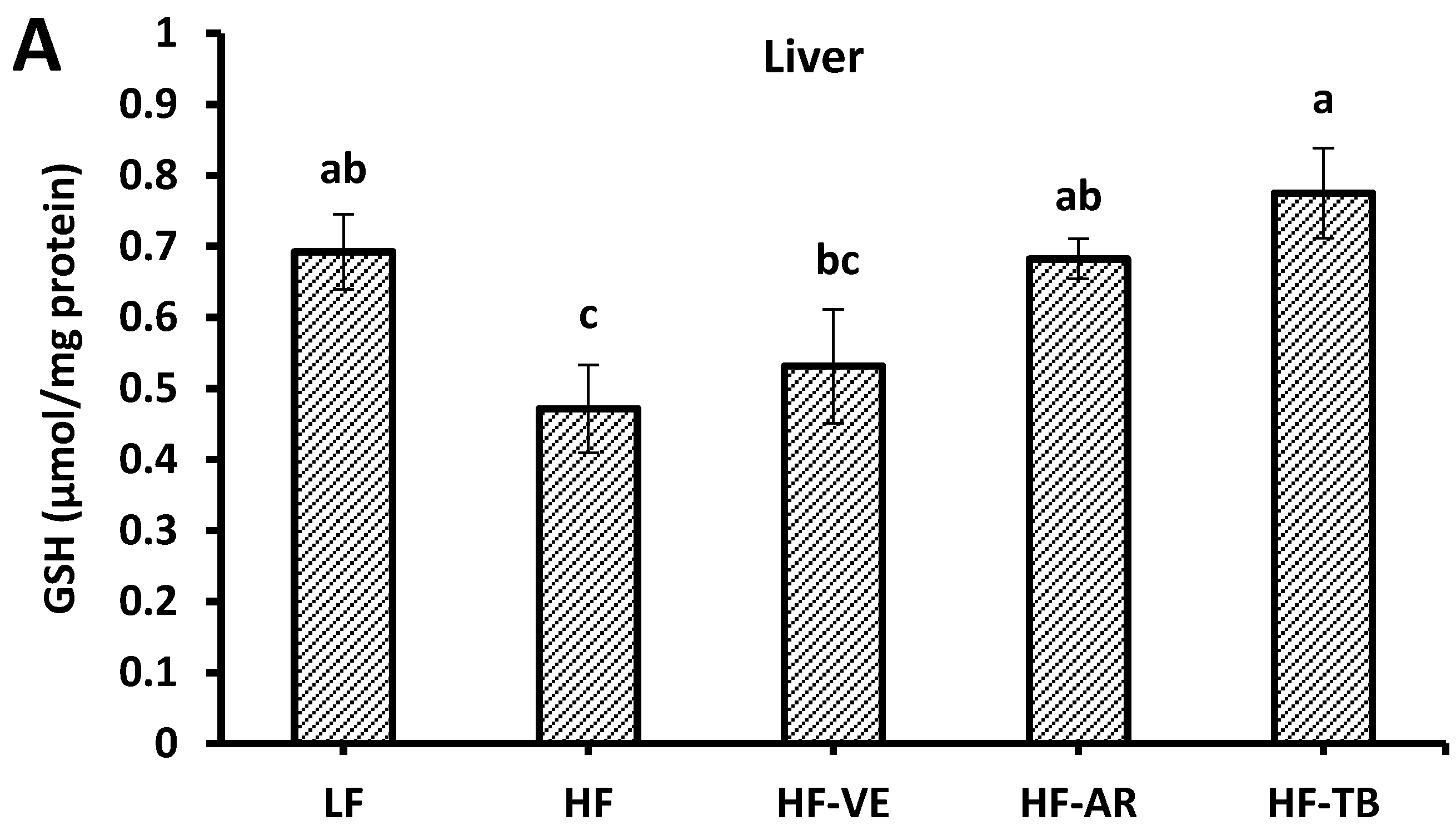

4. Discussion
4.1. Weight and Intake Parameters
4.2. Glucose Tolerance and Fasting Blood Glucose
4.3. Oxygen Radical Absorbance Capacity (ORAC)
4.4. Superoxide Dismutase (SOD) Activity
4.5. Reduced Glutathione (GSH) Contents
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lordan, S.; Ross, R.P.; Stanton, C. Marine bioactives as functional food ingredients: Potential to reduce the incidence of chronic diseases. Marine Drugs 2011, 9, 1056–1100. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, S.; Hashim, S.N.; Rahman, H.A. Seaweeds: A sustainable functional food for complementary and alternative therapy. Trends Food Sci. Technol. 2012, 23, 83–96. [Google Scholar] [CrossRef]
- Vincent, H.K.; Innes, K.E.; Vincent, K.R. Oxidative stress and potential interventions to reduce oxidative stress in overweight and obesity. Diabetes Obes. Metab. 2007, 9, 813–839. [Google Scholar] [CrossRef] [PubMed]
- Seifried, H.E.; Anderson, D.E.; Fisher, E.I.; Milner, J.A. A review of the interaction among dietary antioxidants and reactive oxygen species. J. Nutr. Biochem. 2007, 18, 567–579. [Google Scholar] [CrossRef] [PubMed]
- Limón-Pacheco, J.; Gonsebatt, M.E. The role of antioxidants and antioxidant-related enzymes in protective responses to environmentally induced oxidative stress. Mutat. Res./Genet. Toxicol. Environ. Mutagen. 2009, 674, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Sánchez, A.; Madrigal-Santillán, E.; Bautista, M.; Esquivel-Soto, J.; Morales-González, Á.; Esquivel-Chirino, C.; Durante-Montiel, I.; Sánchez-Rivera, G.; Valadez-Vega, C.; Morales-González, J.A. Inflammation, oxidative stress, and obesity. Int. J. Mol. Sci. 2011, 12, 3117–3132. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J. Why whole grains are protective: Biological mechanisms. Proc. Nutr. Soc. 2003, 62, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J.L. Dietary fiber and body weight. Nutrition 2005, 21, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Ross, A.B.; Shea, M.K.; Bruce, S.J.; Jacques, P.F.; Saltzman, E.; Lichtenstein, A.H.; Booth, S.L.; McKeown, N.M. Plasma alkylresorcinols, biomarkers of whole-grain intake, are related to lower BMI in older adults. J. Nutr. 2012, 142, 1859–1864. [Google Scholar] [CrossRef] [PubMed]
- Agil, R.; Hosseinian, F. Bioactivity of alkylresorcinols. In Bioactive Molecules in Plant Foods; Uruakpa, F.O., Ed.; Nova Science Publishers: New York, NY, USA, 2012; pp. 131–162. [Google Scholar]
- Korycinska, M.; Czelna, K.; Jaromin, A.; Kozubek, A. Antioxidant activity of rye bran alkylresorcinols and extracts from whole-grain cereal products. Food Chem. 2009, 116, 1013–1018. [Google Scholar] [CrossRef]
- Soderholm, P.P.; Lundin, J.E.; Koskela, A.H.; Tikkanen, M.J.; Adlercreutz, H.C. Pharmacokinetics of alkylresorcinol metabolites in human urine. Br. J. Nutr. 2011, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.B.; Kamal-Eldin, A.; Aman, P. Dietary alkylresorcinols: Absorption, bioactivities, and possible use as biomarkers of whole-grain wheat- and rye-rich foods. Nutr. Rev. 2004, 62, 81–95. [Google Scholar] [CrossRef] [PubMed]
- Andersson, U.; Dey, E.S.; Holm, C.; Degerman, E. Rye bran alkylresorcinols suppress adipocyte lipolysis and hormone-sensitive lipase activity. Mol. Nutr. Food Res. 2011, 55, 290–293. [Google Scholar] [CrossRef] [PubMed]
- Gliwa, J. The antioxidant and anti-inflammatory activities of alkylresorcinols from rye bran; Carleton University: Ottawa, ON, Canada, 2013. [Google Scholar]
- Gliwa, J.; Gunenc, A.; Ames, N.; Willmore, W.G.; Hosseinian, F.S. Antioxidant activity of alkylresorcinols from rye bran and their protective effects on cell viability of PC-12 AC cells. J. Agric. Food Chem. 2011, 59, 11473–11482. [Google Scholar] [CrossRef] [PubMed]
- Oishi, K.; Yamamoto, S.; Itoh, N.; Nakao, R.; Yasumoto, Y.; Tanaka, K.; Kikuchi, Y.; Fukudome, S.; Okita, K.; Takano-Ishikawa, Y. Wheat alkylresorcinols suppress high-fat, high-sucrose diet-induced obesity and glucose intolerance by increasing insulin sensitivity and cholesterol excretion in male mice. J. Nutr. 2015, 145, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Agil, R.; Hosseinian, F. Dual functionality of triticale as a novel dietary source of prebiotics with antioxidant activity in fermented dairy products. Plant Foods Hum. Nutr. 2012, 67, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Gunenc, A.; HadiNezhad, M.; Farah, I.; Hashem, A.; Hosseinian, F. Impact of supercritical CO2 and traditional solvent extraction systems on the extractability of alkylresorcinols, phenolic profile and their antioxidant activity in wheat bran. J. Funct. Foods. 2015, 12, 109–119. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Paoletti, F.; Aldinucci, D.; Mocali, A.; Caparrini, A. A sensitive spectrophotometric method for the determination of superoxide dismutase activity in tissue extracts. Anal. Biochem. 1986, 154, 536–541. [Google Scholar] [CrossRef]
- Griffith, O.W. Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinylpyridine. Anal. Biochem. 1980, 106, 207–212. [Google Scholar] [CrossRef]
- Winzell, M.S.; Ahrén, B. The high-fat diet–fed mouse: A model for studying mechanisms and treatment of impaired glucose tolerance and type 2 diabetes. Diabetes 2004, 53, S215–S219. [Google Scholar] [CrossRef] [PubMed]
- Hakkak, R.; Korourian, S.; Pavliv, O.; Melnyk, S. Serum metabolic profile of the 7,12-dimethylbenz(a)anthracene (DMBA)-induced mammary tumor in obese Zucker rat model. FASEB J. 2014, 28, 824–831. [Google Scholar]
- Hsu, C.L.; Yen, G.C. Phenolic compounds: Evidence for inhibitory effects against obesity and their underlying molecular signaling mechanisms. Mol. Nutr. Food Res. 2008, 52, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Sergent, T.; Vanderstraeten, J.; Winand, J.; Beguin, P.; Schneider, Y.J. Phenolic compounds and plant extracts as potential natural anti-obesity substances. Food Chem. 2012, 135, 68–73. [Google Scholar] [CrossRef]
- Ross, A.B.; Chen, Y.; Frank, J.; Swanson, J.E.; Parker, R.S.; Kozubek, A.; Lundh, T.; Vessby, B.; Aman, P.; Kamal-Eldin, A. Cereal alkylresorcinols elevate gamma-tocopherol levels in rats and inhibit gamma-tocopherol metabolism in vitro. J. Nutr. 2004, 134, 506–510. [Google Scholar] [PubMed]
- Jonnalagadda, S.S.; Harnack, L.; Hai Liu, R.; McKeown, N.; Seal, C.; Liu, S.; Fahey, G.C. Putting the whole grain puzzle together: Health benefits associated with whole Grains. J. Nutr. 2011, 141, 1011–1022. [Google Scholar] [CrossRef] [PubMed]
- Ye, E.Q.; Chacko, S.A.; Chou, E.L.; Kugizaki, M.; Liu, S. Greater whole-grain intake is associated with lower risk of type 2 diabetes, cardiovascular disease, and weight gain. J. Nutr. 2012, 142, 1304–1313. [Google Scholar] [CrossRef] [PubMed]
- Kahn, S.E.; Hull, R.L.; Utzschneider, K.M. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 2006, 444, 840–846. [Google Scholar] [CrossRef] [PubMed]
- Magnusdottir, O.K.; Landberg, R.; Gunnarsdottir, I.; Cloetens, L.; Akesson, B.; Landin-Olsson, M.; Rosqvist, F.; Iggman, D.; Schwab, U.; Herzig, K.H.; et al. Plasma alkylresorcinols C17:0/C21:0 ratio, a biomarker of relative whole-grain rye intake, is associated to insulin sensitivity: A randomized study. Eur. J. Clin. Nutr. 2014, 68, 453–458. [Google Scholar] [CrossRef] [PubMed]
- Sealock, R.R.; Murlin, J.R.; Driver, R.L. A comparative study of the effect of alkylresorcinols and related compounds upon the absorption of insulin from isolated intestinal loops of anesthetized dogs. Am. J. Physiol.--Legacy Content 1939, 128, 92–96. [Google Scholar]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef] [PubMed]
- López, P.; Ferraro, G.; Anesini, C. Comparative antioxidant activity of an extract of Lithraea molleoides and an isolated 5-alkyl resorcinol derivative. Effects on the proliferation of normal and tumoral lymphocytes. Phytother. Res. 2011, 25, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Parikka, K.; Rowland, I.R.; Welch, R.W.; Wahala, K. In vitro antioxidant activity and antigenotoxicity of 5-n-alkylresorcinols. J. Agric. Food Chem. 2006, 54, 1646–1650. [Google Scholar] [CrossRef] [PubMed]
- Gunenc, A.; HadiNezhad, M.; Tamburic-Ilincic, L.; Mayer, P.M.; Hosseinian, F. Effects of region and cultivar on alkylresorcinols content and composition in wheat bran and their antioxidant activity. J. Cereal Sci. 2013, 57, 405–410. [Google Scholar] [CrossRef]
- Gunenc, A.; Tavakoli, H.; Seetharaman, K.; Mayer, P.M.; Fairbanks, D.; Hosseinian, F. Stability and antioxidant activity of alkyresorcinols in breads enriched with hard and soft wheat brans. Food Res. Int. 2013, 51, 571–578. [Google Scholar] [CrossRef]
- McCord, J.M.; Edeas, M.A. SOD, oxidative stress and human pathologies: a brief history and a future vision. Biomed. Pharmacother. 2005, 59, 139–142. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, P.; Soulsby, M. Oxidant/anti-oxidant status in rats exposed to simulated weightlessness by hind-limb unloading and reloading. Open Clin. Chem. J. 2008, 1, 47–56. [Google Scholar] [CrossRef]
- Lushchak, V.I. Adaptive response to oxidative stress: Bacteria, fungi, plants and animals. Comp. Biochem. Physiol. Part C: Toxicol. Pharmacol. 2011, 153, 175–190. [Google Scholar] [CrossRef] [PubMed]
- Bartłomiej, S.; Justyna, R.K.; Ewa, N. Bioactive compounds in cereal grains—Occurrence, structure, technological significance and nutritional benefits—A review. Food Sci. Technol. Int. 2012, 18, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Fardet, A. New hypotheses for the health-protective mechanisms of whole-grain cereals: What is beyond fibre? Nutr. Res. Rev. 2010, 23, 65–134. [Google Scholar] [CrossRef] [PubMed]
- Marklund, M.; Strömberg, E.A.; Hooker, A.C.; Hammarlund-Udenaes, M.; Åman, P.; Landberg, R.; Kamal-Eldin, A. Chain length of dietary alkylresorcinols affects their in vivo elimination kinetics in rats. J. Nutr. 2013, 143, 1573–1578. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agil, R.; Patterson, Z.R.; Mackay, H.; Abizaid, A.; Hosseinian, F. Triticale Bran Alkylresorcinols Enhance Resistance to Oxidative Stress in Mice Fed a High-Fat Diet. Foods 2016, 5, 5. https://doi.org/10.3390/foods5010005
Agil R, Patterson ZR, Mackay H, Abizaid A, Hosseinian F. Triticale Bran Alkylresorcinols Enhance Resistance to Oxidative Stress in Mice Fed a High-Fat Diet. Foods. 2016; 5(1):5. https://doi.org/10.3390/foods5010005
Chicago/Turabian StyleAgil, Rania, Zachary R. Patterson, Harry Mackay, Alfonso Abizaid, and Farah Hosseinian. 2016. "Triticale Bran Alkylresorcinols Enhance Resistance to Oxidative Stress in Mice Fed a High-Fat Diet" Foods 5, no. 1: 5. https://doi.org/10.3390/foods5010005