α-Carotene and β-Carotene Content in Raw and Cooked Pulp of Three Mature Stage Winter Squash “Type Butternut”
Abstract
:1. Introduction
2. Experimental Section
2.1. Plant Materials and Fruit Sample Preparation

2.2. β-Carotene and α-Carotene Content
2.3. Color
2.4. Percentage of Reduction of Carotenes by Cooking
2.5. Dry Matter Content
2.6. Experimental Design and Statistical Analysis
3. Results and Discussion
3.1. Color and Content of β-Carotene and α-Carotene in Winter Squash
| β-Carotene | α-Carotene | |||
|---|---|---|---|---|
| Raw | Cooked | Raw | Cooked | |
| Immature | 9.54 ± 0.61 bA | 6.35 ± 0.62 bB | 9.28 ± 0.31 bA | 5.55 ± 0.54 bB |
| Mature | 12.34 ± 0.47 abA | 9.37 ± 0.91 abA | 14.31 ± 0.67 abA | 9.37 ± 0.65 bB |
| Very Mature | 15.77 ± 1.61 aA | 11.08 ± 0.83 aA | 16.19 ± 1.79 aA | 11.31 ± 0.89 aA |
| Mains effects | p Value (β) | p Value (α) | ||
| Stage of Maturity (SM) | 0.0016 | 0.0007 | ||
| Preparation (P) | 0.0016 | 0.0004 | ||
| SM x P | 0.6458 | 0.8421 | ||
| Immature | Mature | Very Mature | |
|---|---|---|---|
| Lightness (L*) | |||
| Raw | 69.4 ± 0.3 aA | 68.5 ± 0.4 aA | 65.8 ± 0.5 bA |
| Cooked | 57.5 ± 1.5 aB | 62.7 ± 1.6 aB | 51.6 ± 1.5 bB |
| Hue Angle (hab) | |||
| Raw | 67.1 ± 0.6 aA | 66.1 ± 0.3 aA | 61.3 ± 0.6 bA |
| Cooked | 73.2 ± 1.0 aB | 67.8 ± 0.8 bA | 65.2 ± 1.2 bB |
| Chroma (C*ab) | |||
| Raw | 62.3 ± 0.3 bA | 63.9 ± 1.4 bA | 67.4 ± 0.8 aA |
| Cooked | 50.7 ± 1.9 bB | 59.8 ± 2.0 aA | 49.5 ± 1.8 bB |
| Dry matter content (%) | p Value | |||
|---|---|---|---|---|
| Immature | Mature | Very Mature | ||
| Raw | 8.75 ± 0.75 | 8.28 ± 0.29 | 8.60 ± 0.59 | 0.8403 |
| Cooked | 9.26 ± 0.53 | 8.83 ± 0.17 | 8.04 ± 0.14 | 0.1426 |
| p Value | 0.6779 | 0.2182 | 0.2131 | |
| Main Effects | p Value | |||
| Maturity stage (MS) | 0.3911 | |||
| Preparation (P) | 0.6874 | |||
| MS × P | 0.6458 | |||
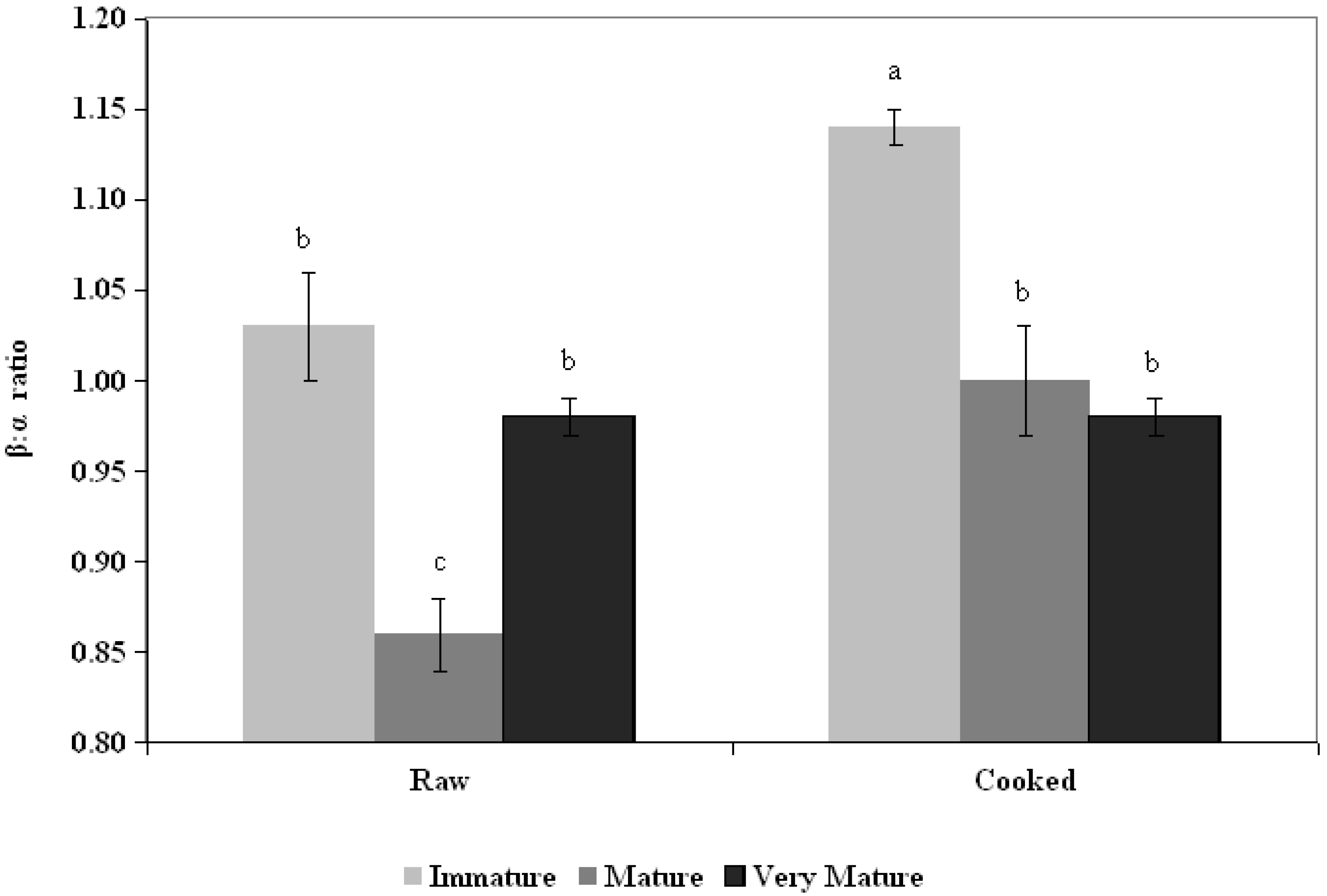
3.2. Ratio β:α
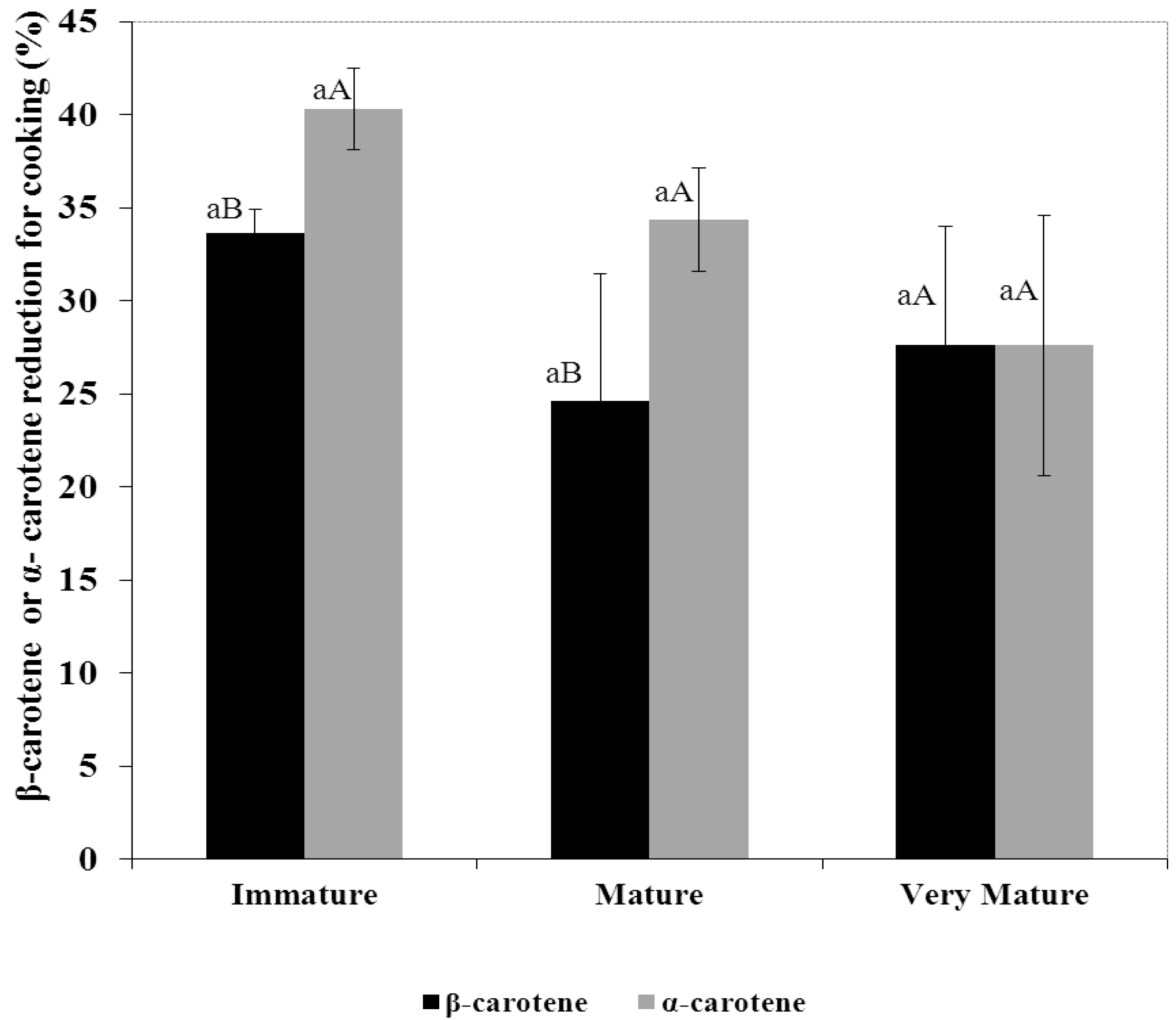
3.3. Percentage Reduction of Carotenes
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zaccari, F.; Sollier, S. Estudio de la conservación de diez cultivares de calabacines. In Resultados de Investigación en Zapallo, Zafra 2003–2004; Facultad de Agronomía, Universidad de la República: Montevideo, Uruguay, 2004; p. 66. Available online: http://biblioteca.fagro.edu.uy/cgi-bin/wxis.exe/iah/ (accessed on 26 March 2015).
- Harvey, W.J.; Grant, D.G. Effect of maturity on the sensory quality of buttercup squash. Proc. Agron. Soc. N. Zeal. 1992, 22, 25–30. [Google Scholar]
- Von Lintig, J.; Sies, H. Carotenoids. Arch. Biochem. Biophys. 2013, 539, 99–101. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.K.; Zhang, M.P.; Marzourek, M.; Tadmor, Y.; Li, L. Regulatory control of carotenoid accumulation in winter squash during storage. Planta 2014, 240, 1063–1074. [Google Scholar] [CrossRef] [PubMed]
- Britton, G.; Khachik, F. Carotenoids. In Food Carotenoids; Britton, G., Liaaen-Jensen, S., Pfand, H., Eds.; Birkhäuser Verlag: Basel, Switzerland, 2009; Chapter 3; Volume 5, pp. 45–67. [Google Scholar]
- Fu, C.; Shi, H.; Li, Q. A review on pharmacological activities and utilization technologies of pumpkin. Plant Foods Hum. Nutr. 2006, 61, 73–80. [Google Scholar]
- Conti, S.; Villari, G.; Amico, E.; Caruso, G. Effects of production system and transplanting time on yield, quality and antioxidant content of organic winter squash (Cucurbita moschata Duch.). Sci. Hortic. 2015, 183, 136–143. [Google Scholar] [CrossRef]
- Harvey, W.J.; Grant, D.G.; Lammerink, J.P. Physical and sensory changes during the development and storage of buttercup squash. N. Zeal. Crop Hortic. Sci. 1997, 25, 341–351. [Google Scholar] [CrossRef]
- Boon, C.S.; McClements, J.D.; Weiss, J.; Decker, E.A. Factors Influencing the Chemical Stability of Carotenoids in Foods. Crit. Rev. Food Sci. Nutr. 2010, 50, 515–532. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Amaya, D.B.; Kimur, M.; Godoy, H.T.; Amaya-Farfan, J. Updated Brazilian database on food carotenoids: Factors affecting carotenoid composition. J. Food Compos. Anal. 2008, 21, 445–463. [Google Scholar] [CrossRef]
- Carvalho, J.L.M.; de Azevedo Sarmet Moreira Smiderle, L.; de Carvalho, J.L.V.; de Souza Neves Cardoso, F.; Bello Koblit, M.G. Assessment of carotenoids in pumpkins after different home cooking conditions. Food Sci. Technol. (Campinas) 2014, 34, 365–370. [Google Scholar] [CrossRef]
- Zaccari, F.; Domingo, C.; Volonterio, E. Efecto del estado de madurez de calabacín (Cosmos F1) y de la conservación refrigerada en la calidad de la pulpa cocida. In Proceedings of the Avances en la Postcosecha de Frutas y Hortalizas, Valencia, España, 21–23 October 2014; Zacarías, L., Ballester, A.R., Marcos, J.F., Pérez-Gago, M.B., Eds.; Fundación Universidad-Empresa ADEIT: Valencia, España, 2014; pp. 553–557. [Google Scholar]
- Zaccari, F.; Cabrera, M.; Ramos, A.; Saadoun, A. In vitro bioaccessibility of β-carotene, Ca, Mg and Zn in landrace carrots (Daucus carota, L.). Food Chem. 2015, 166, 365–371. [Google Scholar] [CrossRef] [PubMed]
- CIE, Colorimetry. Publications CIE. In Commission Internationale de l’Eclairage Central Bureau; Technical Report; CIE: Vienna, Austria, 2004; CIE (15.2). [Google Scholar]
- Provesi, J.G.; OdebrechtDias, C.; Amante, E.R. Changes in carotenoids during processing and storage of pumpkin puree. Food Chem. 2011, 128, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Nakkanong, K.; Yang, J.H.; Zhang, M.F. Carotenoid accumulation and carotenogenic gene expression during fruit development in novel inter-specific inbred squash lines and their parents. J. Agric. Food Chem. 2012, 60, 5936–5944. [Google Scholar] [CrossRef] [PubMed]
- Lv, P.; Li, N.; Liu, H.; Gu, H.; Zhao, W. Changes in carotenoid profiles and in the expression pattern of the genes in carotenoid metabolisms during fruit development and ripening in four watermelon cultivars. Food Chem. 2015, 174, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Yuan, H. Chromoplast biogenesis and carotenoid accumulation. Arch. Biochem. Biophys. 2013, 539, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Murador, D.C.; Cunha, D.T.; de Rossa, V.V. Effects of cooking techniques on vegetable pigments: A meta-analytic approach to carotenoid and anthocyanin levels. Food Res. Int. 2014, 65, 177–183. [Google Scholar] [CrossRef]
- Meléndez-Martínez, A.J.; Britton, G.; Vicario, I.M.; Heredia, F.J. Relationship between the colour and the chemical structure of carotenoid pigments. Food Chem. 2007, 101, 1145–1150. [Google Scholar] [CrossRef]
- Gurak, P.D.; Mercadante, A.Z.; González-Miret, M.L.; Heredia, F.J.; Meléndez-Martínez, A.J. Changes in antioxidant capacity and colour asassociated with the formation of b-carotene epoxides and oxidative ecleavage derivatives. Food Chem. 2014, 147, 160–169. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro-Santana, H.M.; Stringheta, P.C.; Cardoso Brandão, S.; Páez, H.H.; Vitarelli de Querioz, V.M. Evaluation of total carotenoids, α- and β-carotene in carrots (Daucus carota L.) during home processing. Ciência e Tecnología do Alimentos (Campinas) 1998, 18, 39–44. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zaccari, F.; Galietta, G. α-Carotene and β-Carotene Content in Raw and Cooked Pulp of Three Mature Stage Winter Squash “Type Butternut”. Foods 2015, 4, 477-486. https://doi.org/10.3390/foods4030477
Zaccari F, Galietta G. α-Carotene and β-Carotene Content in Raw and Cooked Pulp of Three Mature Stage Winter Squash “Type Butternut”. Foods. 2015; 4(3):477-486. https://doi.org/10.3390/foods4030477
Chicago/Turabian StyleZaccari, Fernanda, and Giovanni Galietta. 2015. "α-Carotene and β-Carotene Content in Raw and Cooked Pulp of Three Mature Stage Winter Squash “Type Butternut”" Foods 4, no. 3: 477-486. https://doi.org/10.3390/foods4030477
APA StyleZaccari, F., & Galietta, G. (2015). α-Carotene and β-Carotene Content in Raw and Cooked Pulp of Three Mature Stage Winter Squash “Type Butternut”. Foods, 4(3), 477-486. https://doi.org/10.3390/foods4030477





