From Nucleation to Fat Crystal Network: Effects of Stearic–Palmitic Sucrose Ester on Static Crystallization of Palm Oil
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Protocols for Crystallization
2.3. Differential Scanning Calorimetry
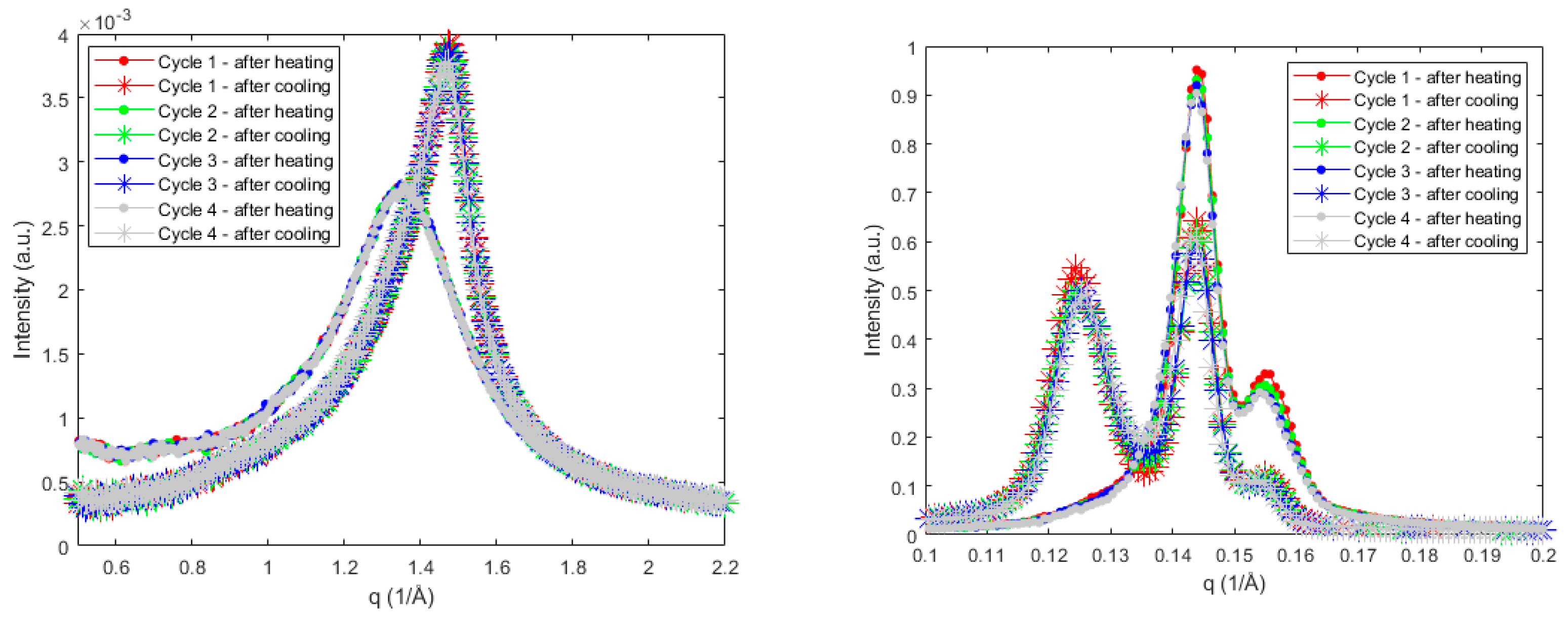
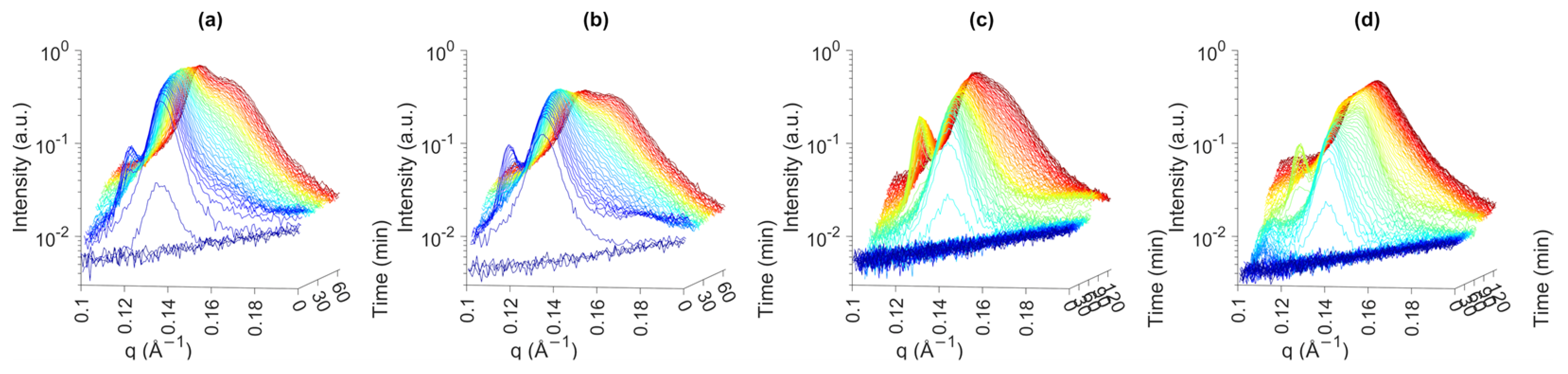
2.4. Small-Angle X-ray Scattering
- The lamellar spacing (d001) was derived from the peak position (q001): d001 = (2 × π)/q001.
- The thickness τ (domain) of the nanoplatelets was derived from the Scherrer equation: τ = (K × λ)/(FWHM001 × cos θ), with K being a numerical constant of 0.9, λ the wavelength, FWHM001 the full width at half maximum from the first-order crystallization peak (radians) and θ the Bragg angle (radians).
- A strain analysis was applied in order to assess the effect of the SE addition on the fat crystal network: FWHM001 × cos θ = (K × λ)/L + 4 × ε × sin θ. FWHM001, θ, K and λ are as described above. L provides a domain size corrected for strain, and ε is the strain. A plot of FWHM001 × cos θ as a function of 4 × sin θ was made based on the first (001)- and third (003)-order SAXS peaks. The slope provided a value for the strain ε. The goal was not to obtain an absolute strain value, but to investigate the strain differences with and without the SE.
2.5. Wide- and Ultra-Small-Angle X-ray Scattering
2.6. Polarized Light Microscopy
2.7. Cryo-Scanning Electron Microscopy
2.8. Graphs and Statistics
3. Results and Discussion
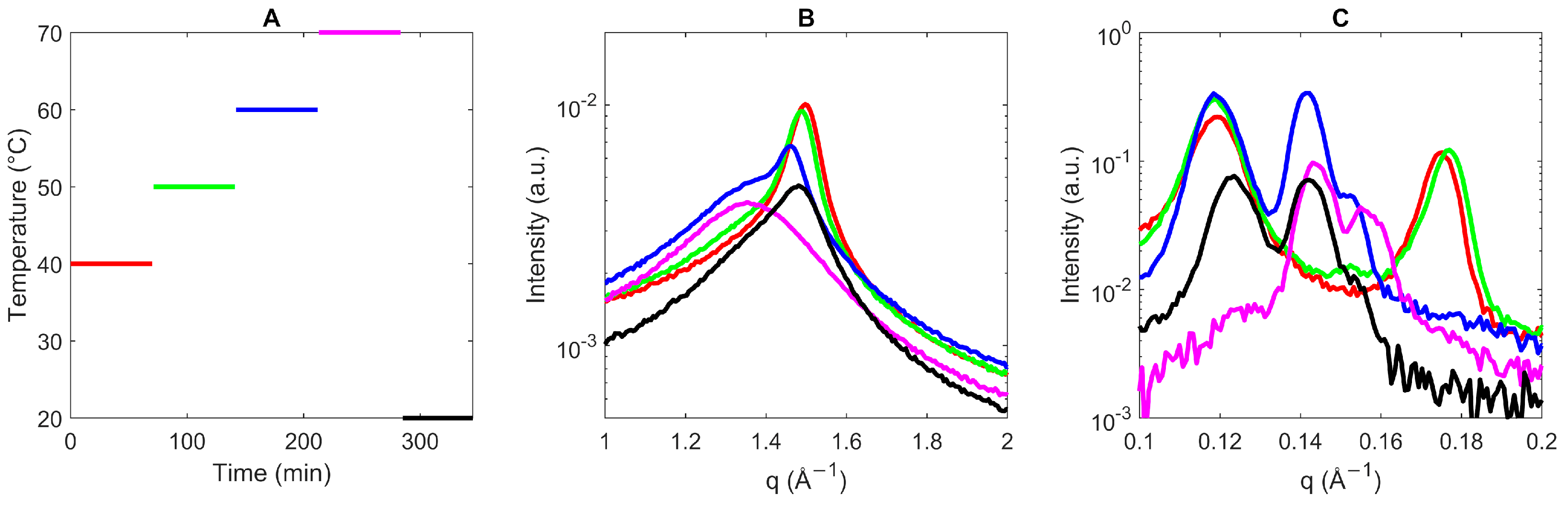
3.1. Crystallization Mechanism of Sucrose Ester
3.1.1. Crystallization and Melting Behaviors of the Sucrose Ester
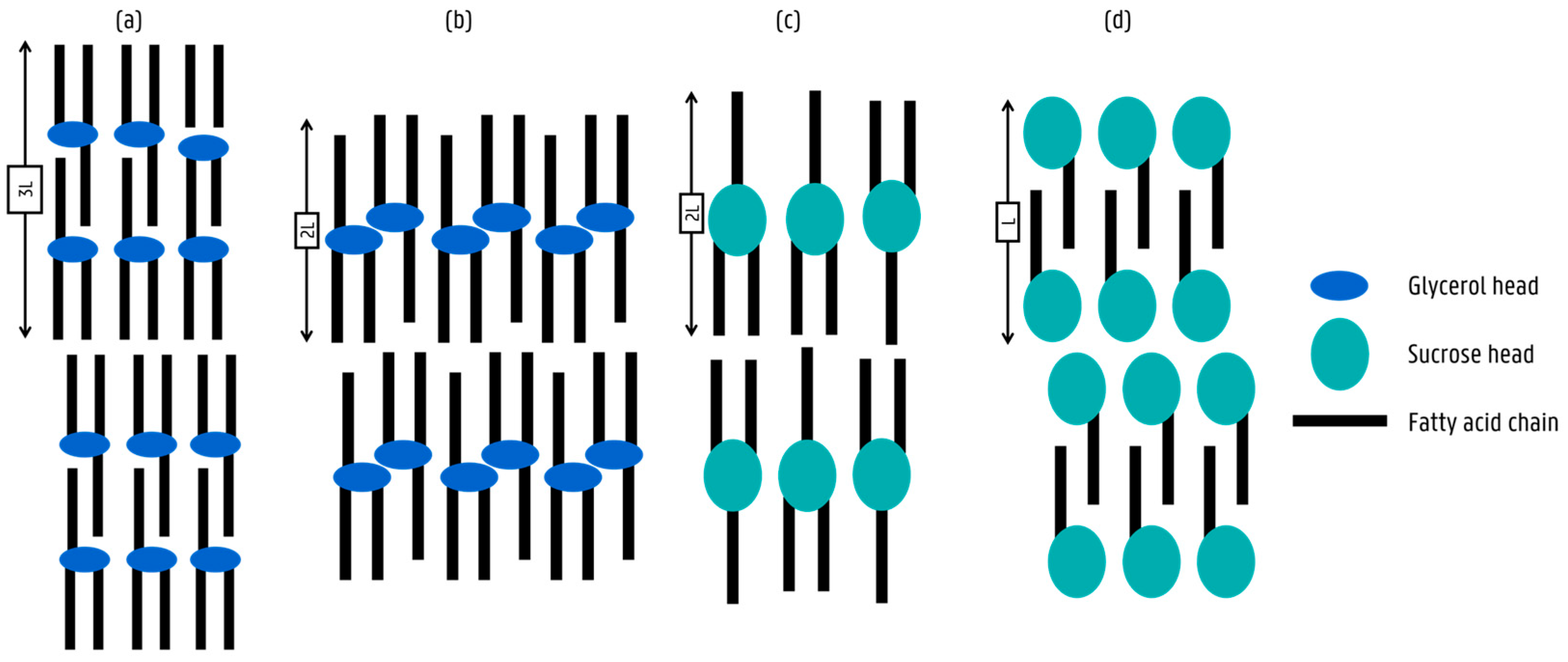
3.1.2. Subcell Packing and Chain-Length Structure of Sucrose Ester
3.2. Effect of Sucrose Ester Addition on Crystallization of Palm Oil
3.2.1. Onset of Crystallization
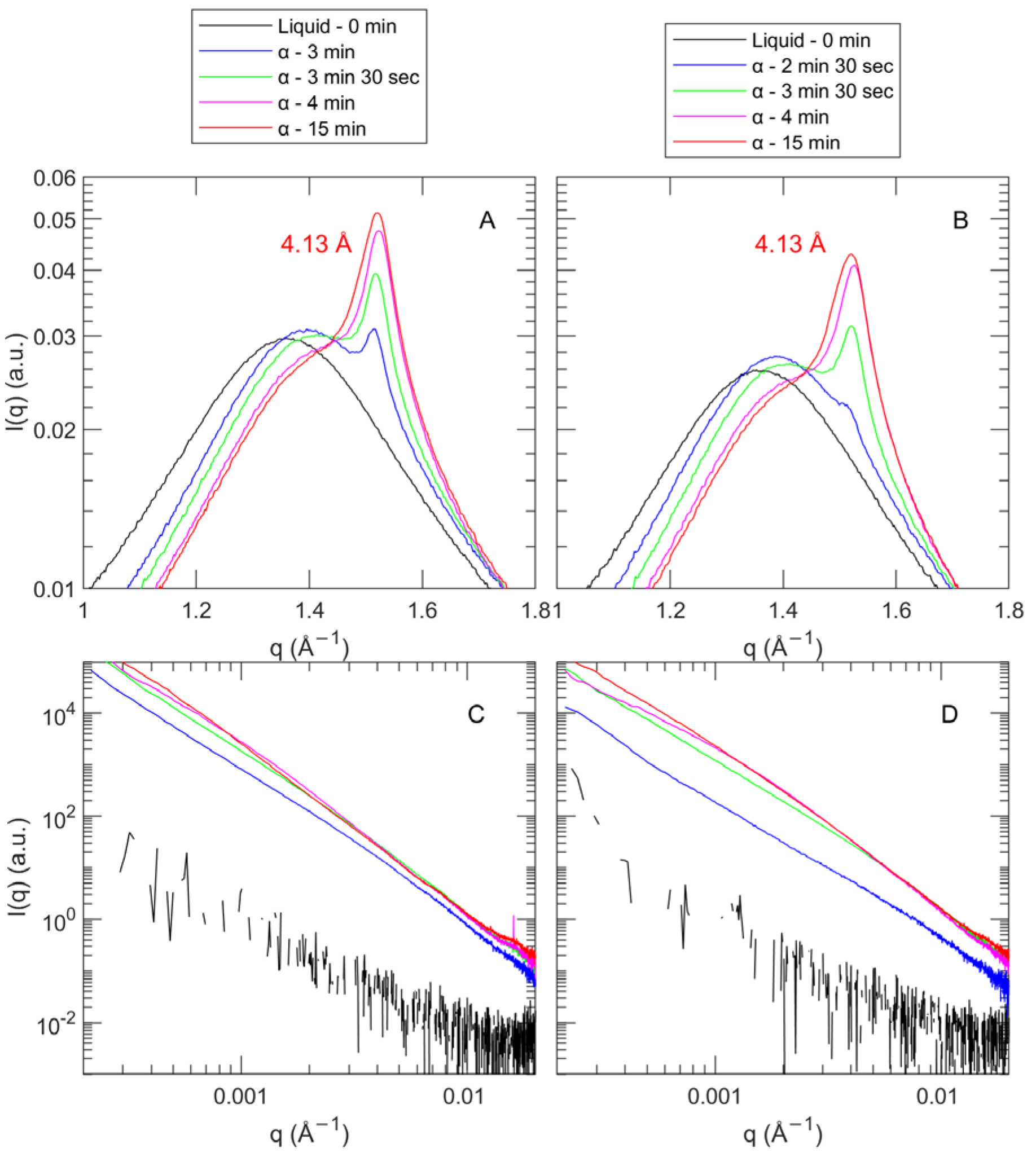
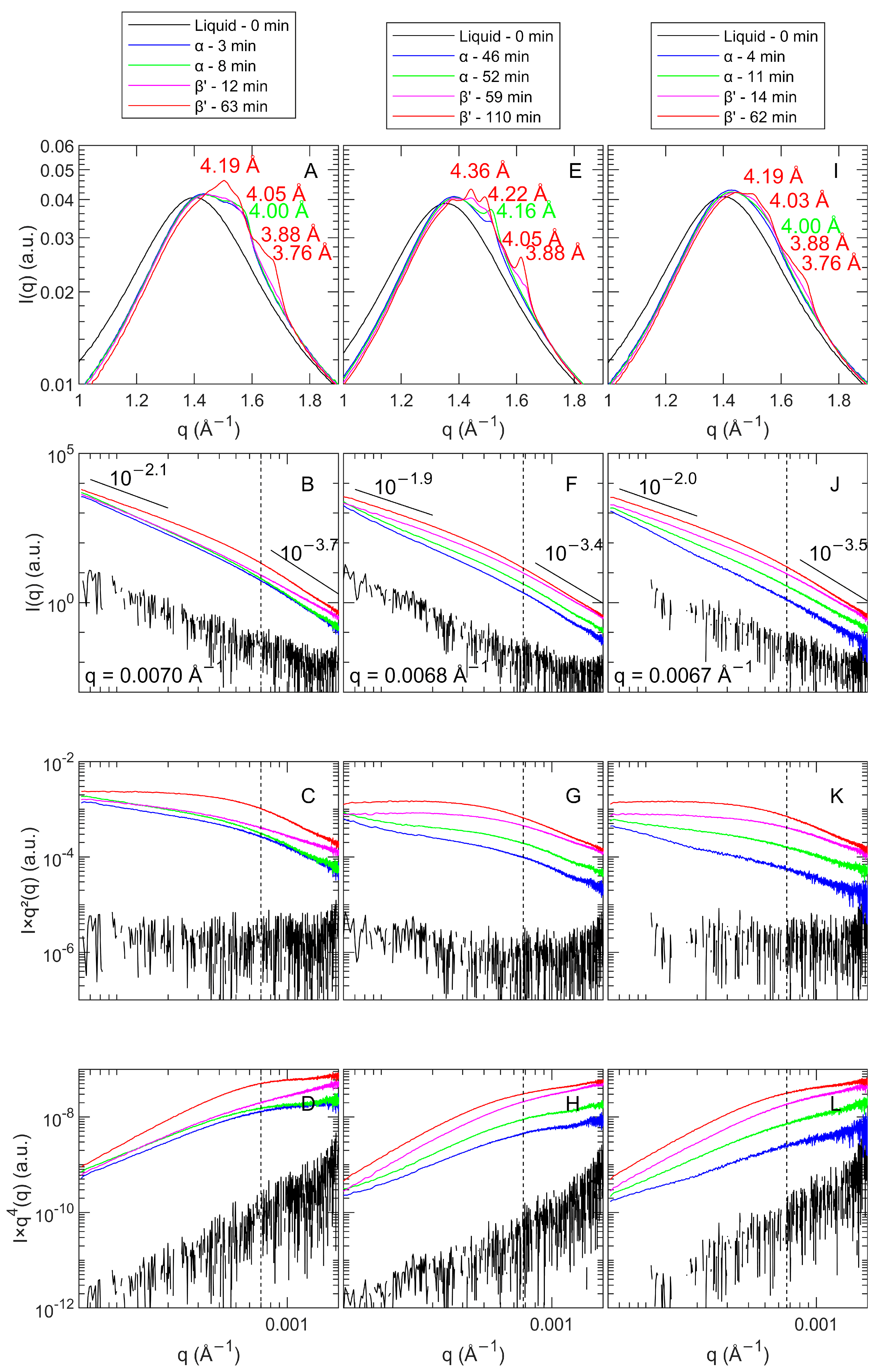
3.2.2. Polymorphic Transition and Lamellar Thickness
Polymorphism
Lamellar Spacing
3.2.3. CNP Structure and Aggregation
Crystal Nanoplatelet Thickness and Thickness Distribution
Crystal Strain
Crystal Nanoplatelet Size
Microstructure of Fat Crystal Network
3.2.4. Melting Properties
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Crystallization and Melting of SP-30
| Crystallization Protocol | Onset (°C) | Peak 1 (°C) | Peak 2 (°C) | Peak 3 (°C) | Peak 4 (°C) |
|---|---|---|---|---|---|
| FC0 | 58.9 ± 0.2 c | 56.7 ± 0.6 b | 39.5 ± 0.2 b | 31.4 ± 0.8 b | 26.7 ± 1.4 ab |
| FC20 | 59.1 ± 0.2 bc | 56.9 ± 0.3 b | 40.0 ± 0.2 ab | 31.7 ± 0.6 ab | 27.4 ± 0.6 ab |
| FC25 | 59.0 ± 0.1 c | 57.1 ± 0.0 ab | 39.2 ± 0.5 b | 31.2 ± 0.5 b | 29.0 ± 0.3 a |
| SC0 | 60.9 ± 0.1 a | 59.7 ± 0.3 a | 42.9 ± 0.3 a | 34.2 ± 0.6 a | 28.7 ± 0.6 a |
| SC20 | 60.8 ± 0.1 abc | 59.9 ± 0.1 a | 43.3 ± 0.9 a | 34.4 ± 1.0 a | 25.9 ± 1.0 b |
| SC25 | 60.8 ± 0.1 ab | 59.9 ± 0.2 a | 43.0 ± 0.3 a | 34.3 ± 0.4 a | 28.8 ± 1.3 a |
| Crystallization Protocol | Peak 1 (°C) | Peak 2 (°C) | Peak 3 (°C) |
|---|---|---|---|
| FC0 | 44.6 ± 0.1 ab | 49.3 ± 0.2 ab | 64.8 ± 0.2 a |
| FC20 | 44.4 ± 0.1 b | 49.2 ± 0.2 b | 64.7 ± 0.1 a |
| FC25 | 44.5 ± 0.1 b | 49.2 ± 0.1 b | 64.9 ± 0.2 a |
| SC0 | 44.9 ± 0.1 a | 49.7 ± 0.1 a | 65.6 ± 0.2 a |
| SC20 | 44.9 ± 0.2 a | 49.7 ± 0.1 a | 65.2 ± 0.5 a |
| SC25 | 45.0 ± 0.2 a | 49.8 ± 0.2 a | 65.4 ± 0.4 a |

| Temperature (°C) | WAXS | SAXS | ||||||
|---|---|---|---|---|---|---|---|---|
| Peak 1 | Peak 2 | Peak 3 | ||||||
| q (Å−1) | d (Å) | q (Å−1) | d (Å) | q (Å−1) | d (Å) | q (Å−1) | d (Å) | |
| 40 | 1.50 | 4.19 | 0.119 | 52.8 | 0.175 | 35.9 | 0.352 | 17.8 |
| 50 | 1.49 | 4.22 | 0.118 | 53.2 | 0.177 | 35.5 | 0.355 | 17.7 |
| 60 | Amorphous + 1.46 | Amorphous + 4.30 | 0.118 | 53.2 | 0.142 | 44.2 | 0.152 | 41.3 |
| 70 | Amorphous | Amorphous | 0.143 | 43.9 | 0.160 | 39.3 | - | - |
| 20 | 1.48 | 4.24 | 0.124 | 50.7 | 0.142 | 44.2 | - | - |
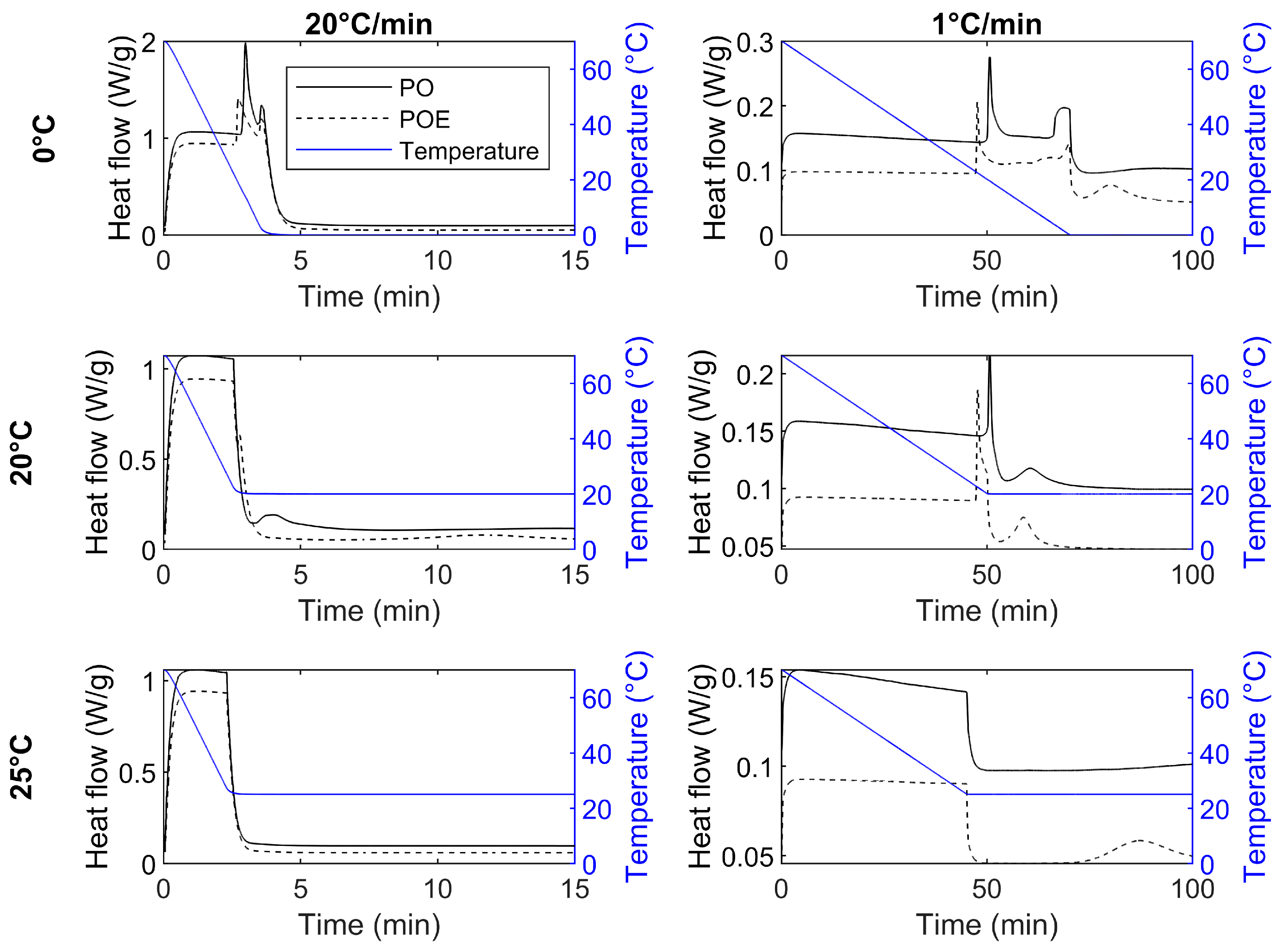
Appendix B. Crystallization and Melting of PO and POE
| Sample | Protocol | Onset | Peak 1 | Peak 2 | Peak 3 | ||||
|---|---|---|---|---|---|---|---|---|---|
| Temperature (°C) | Time (min) | Temperature (°C) | Time (min) | Temperature (°C) | Time (min) | Temperature (°C) | Time (min) | ||
| PO | FC0 | 17.5 ± 0.2 b,A | 2.8 ± 0.1 c,A | 13.8 ± 0.1 | 3.0 ± 0.1 | 2.3 ± 0.1 | 3.6 ± 0.1 | 0.0 ± 0.1 | 29.4 ± 0.6 |
| SC0 | 21.9 ± 0.5 ab,A | 48.2 ± 0.5 ab,A | 19.5 ± 0.1 | 50.8 ± 0.1 | 1.7 ± 0.1 | 68.6 ± 0.1 | 0.0 ± 0.1 | 93.5 ± 2.0 | |
| FC20 SC20 | 20.1 ± 0.1 b,B | 3.4 ± 0.1 bc,A | 20.1 ± 0.1 | 3.9 ± 0.1 | 20.0 ± 0.1 | 14.1 ± 0.5 | - | - | |
| 22.3 ± 1.1 ab,A | 47.8 ± 1.1 ab,A | 20.1 ± 0.1 | 50.1 ± 0.1 | 20.0 ± 0.1 | 60.6 ± 0.2 | - | - | ||
| FC25 SC25 | 25.0 ± 0.1 a,A | 30.1 ± 4.6 bc,A | ND | ND | ND | ND | ND | ND | |
| 25.0 ± 0.1 a,A | 77.9 ± 6.2 a,A | ND | ND | ND | ND | ND | ND | ||
| POE | FC0 SC0 | 20.6 ± 0.4 b,A | 2.6 ± 0.1 c,B | 18.6 ± 0.4 | 2.8 ± 0.1 | 2.4 ± 0.1 | 3.6 ± 0.1 | 0.0 ± 0.1 | 12.0 ± 2.0 |
| 22.9 ± 0.2 ab,A | 47.2 ± 0.2 ab,A | 22.5 ± 0.1 | 47.8 ± 0.1 | 4.7 ± 0.3 0.0 ± 0.1 | 65.5 ± 0.3 70.0 ± 0.1 | 0.0 ± 0.1 | 80.5 ± 0.7 | ||
| FC20 SC20 | 21.0 ± 0.1 b,A | 2.7 ± 0 bc,B | 20.8 ± 0.1 | 2.8 ± 0.1 | 20.0 ± 0.1 | 11.8 ± 0.2 | - | - | |
| 23.0 ± 0.3 ab,A | 47.2 ± 0.3 ab,A | 22.4 ± 0.1 | 47.8 ± 0.1 | 20.0 ± 0.1 | 59.3 ± 0.6 | - | - | ||
| FC25 SC25 | 25.0 ± 0.1 a,A | 28.8 ± 6.4 bc,A | 25.0 ± 0.1 | 49.6 ± 9.1 | - | - | - | - | |
| 25.0 ± 0.1 a,A | 70.9 ± 5.3 a,A | 25.0 ± 0.1 | 92.7 ± 8.1 | - | - | - | - | ||
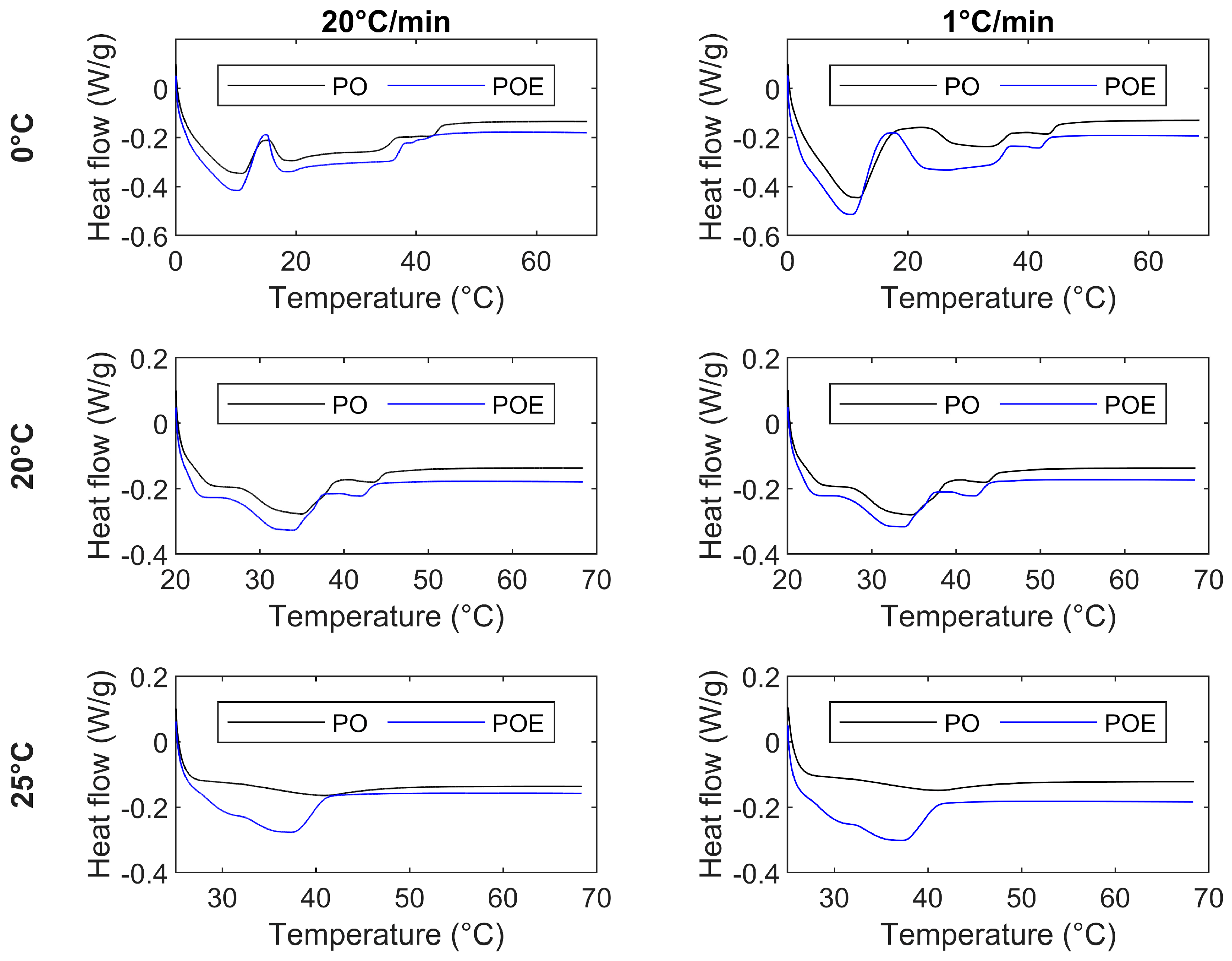
| Sample | Protocol | Peak 1 (°C) | Peak 2 (°C) | Peak 3 (°C) | Peak 4 (°C) |
|---|---|---|---|---|---|
| PO | FC0 | 10.7 ± 0.1 | 19.2 ± 0.1 | 33.5 ± 0.6 | 42.6 ± 0.2 |
| SC0 | 11.6 ± 0.1 | 27.6 ± 0.4 | 34.1 ± 0.3 | 43.1 ± 0.1 | |
| FC20 SC20 | 24.3 ± 0.2 | 34.7 ± 0.2 | 43.4 ± 0.1 | - | |
| 24.4 ± 0.2 | 34.6 ± 0.1 | 43.4 ± 0.1 | - | ||
| FC25 SC25 | 41.0 ± 0.1 | - | - | - | |
| 41.2 ± 0.1 | - | - | - | ||
| POE | FC0 SC0 | 10.3 ± 0.1 | 18.5 ± 0.8 | 36.2 ± 0.2 | 39.3 ± 0.1 |
| 10.7 ± 0.2 | 26.8 ± 0.4 | 41.7 ± 0.1 | 41.7 ± 0.2 | ||
| FC20 SC20 | 23.2 ± 0.2 | 34.0 ± 0.3 | 41.7 ± 0.3 | - | |
| 23.5 ± 0.2 | 33.7 ± 0.1 | 42.1 ± 0.1 | - | ||
| FC25 SC25 | 29.5 ± 2.2 | 37.6 ± 0.6 | - | - | |
| 31.4 ± 0.9 | 37.2 ± 0.4 | - | - |
Appendix C. Crystallization of PO and POE at 0 °C (FC0 and SC0)

Appendix D. Crystal Strain Assessments of PO and POE
| Sample | Cooling Protocol | Strain ε |
|---|---|---|
| PO | FC20 | 7.2 × 10−5 ± 9.8 × 10−6 a,A |
| SC20 | 6.1 × 10−5 ± 5.3 × 10−6 a,A | |
| POE | FC20 | 6.1 × 10−5 ± 7.4 × 10−6 a,A |
| SC20 | 4.7 × 10−5 ± 8.1 × 10−6 a,A |
References
- Zaliha, O.; Hishamuddin, E.; Mat Sahri, M.; Mohamad Fauzi, S.; Mat Dian, N.L.; Ramli, M.; Norizzah, A.R. Palm Oil Crystallisation: A Review. J. Oil Palm Res. 2015, 27, 97–106. [Google Scholar]
- Sainlaud, C.; Taché, O.; Testard, F.; Saiter, J.; Bohin, M.C.; Coquerel, G. Impact of Cooling Profile on Refined Palm Oil Crystallization: Microscopic and Small and Wide-Angle X-ray Scattering Investigations. Eur. J. Lipid Sci. Technol. 2021, 124, 2100045. [Google Scholar] [CrossRef]
- Nguyen, V.; Rimaux, T.; Truong, V.; Dewettinck, K.; Van Bockstaele, F. Granular Crystals in Palm Oil Based Shortening/Margarine: A Review. Cryst. Growth Des. 2019, 20, 1363–1372. [Google Scholar] [CrossRef]
- Sato, K.; Bayés-García, L.; Calvet, T.; Cuevas-Diarte, M.; Ueno, S. External factors affecting polymorphic crystallization of lipids. Eur. J. Lipid Sci. Technol. 2013, 115, 1224–1238. [Google Scholar] [CrossRef]
- Marangoni, A.G.; Wesdorp, L.H. Structure and Properties of Fat Crystal Networks; Taylor & Francis Ltd.: London, UK, 2012. [Google Scholar]
- Acevedo, N.C.; Marangoni, A.G. Nanostructured Fat Crystal Systems. Annu. Rev. Food Sci. Technol. 2015, 6, 71–96. [Google Scholar] [CrossRef] [PubMed]
- Nikolaeva, T. Multiscale Assessment of Food Lipid Structures; Wageningen University: Wageningen, The Netherlands, 2019. [Google Scholar]
- Garbolino, C.; Bartoccini, M.; Flöter, E. The influence of emulsifiers on the crystallisation behaviour of a palm oil-based blend. Eur. J. Lipid Sci. Technol. 2005, 107, 616–626. [Google Scholar] [CrossRef]
- Nelen, B.A.P.; Cooper, J.M. Sucrose Esters. In Emulsifiers in Food Technology; John Wiley & Sons: Hoboken, NJ, USA, 2004; pp. 131–161. [Google Scholar]
- Molinier, V.; Kouwer, P.J.J.; Fitremann, J.; Bouchu, A.; Mackenzie, G.; Queneau, Y.; Goodby, J.W. Self-organizing properties of monosubstituted sucrose fatty acid esters: The effects of chain length and un-saturation. Chemistry 2006, 12, 3547–3557. [Google Scholar] [CrossRef] [PubMed]
- Muller, A.-S.; Gagnaire, J.; Queneau, Y.; Karaoglanian, M.; Maitre, J.-P.; Bouchu, A. Winsor behaviour of sucrose fatty acid esters: Choice of the cosurfactant and effect of the surfactant com-position. Colloids Surf. A Physicochem. Eng. Asp. 2002, 203, 55–66. [Google Scholar] [CrossRef]
- Teng, Y.; Stewart, S.G.; Hai, Y.-W.; Li, X.; Banwell, M.G.; Lan, P. Sucrose fatty acid esters: Synthesis, emulsifying capacities, biological activities and structure-property profiles. Crit. Rev. Food Sci. Nutr. 2020, 61, 3297–3317. [Google Scholar] [CrossRef]
- Aulton, M.E. Pharmaceutics: The Science of Dosage form Design, 2nd ed.; Churchill Livingstone Edinburgh: Edinburgh, UK, 2002. [Google Scholar]
- da Silva, T.L.T.; Baeten, V.; Danthine, S. Modifying sucrose esters oleogels properties using different structuration routes. Food Chem. 2023, 405, 134927. [Google Scholar] [CrossRef]
- The European Parliament; The Council of the European Union. Regulation (EC) No 1333/2008 of the European Parliament and of the council of 16 December 2008 on food additives. Off. J. Eur. Union 2008, 34, 67–84. [Google Scholar]
- The European Commission. Commission Regulation (EU) No 1129/2011 of 11 November 2011 amending Annex II to Regulation (EC) No 1333/2008 of the European Parliament and of the Council by establishing a Union list of food additives. Off. J. Eur. Union 2011, 45, 131–307. [Google Scholar]
- The European Commission. Commission Regulation (EU) No 231/2012 of 9 March 2012 laying down specifications for food additives listed in Annexes II and III to Regulation (EC) No 1333/2008 of the European Parliament and of the Council. Off. J. Eur. Union 2012, 37, 3–297. [Google Scholar]
- Chen, C.; Zhang, H.; Bi, Y.; Cheong, L. Effects of Sucrose Esters on Isothermal Crystallization of Palm Oil-based Blend. J. Am. Oil Chem. Soc. 2014, 92, 277–286. [Google Scholar] [CrossRef]
- Tangsanthatkun, J.; Sonwai, S. Crystallisation of palm olein under the influence of sucrose esters. Int. J. Food Sci. Technol. 2019, 54, 3032–3041. [Google Scholar] [CrossRef]
- Martini, S.; Puppo, M.; Hartel, R.; Herrera, M. Effect of Sucrose Esters and Sunflower Oil Addition on Crystalline Microstructure of a High-melting Milk Fat Fraction. J. Food Sci. 2002, 67, 3412–3418. [Google Scholar] [CrossRef]
- Puppo, M.; Martini, S.; Hartel, R.; Herrera, M. Effects of Sucrose Esters on Isothermal Crystallization and Rheological Behavior of Blends of Milk-fat Fraction Sunflower Oil. J. Food Sci. 2002, 67, 3419–3426. [Google Scholar] [CrossRef]
- Elisabettini, P.; Desmedt, A.; Durant, F. Polymorphism of stabilized and nonstabilized tristearin, pure and in the presence of food emulsifiers. J. Am. Oil Chem. Soc. 1996, 73, 187–192. [Google Scholar] [CrossRef]
- Domingues, M.A.F.; Paulo, B.D.; da Silva, T.L.T.; Ribeiro, A.P.B. Physical properties of cocoa butter: Effect of sucrose esters on oil migration. Int. J. Food Sci. Technol. 2023, 58, 6059–6067. [Google Scholar] [CrossRef]
- Rincón-Cardona, J.A.; Agudelo-Laverde, L.M.; Martini, S.; Candal, R.J.; Herrera, M.L. In situ synchrotron radiation X-ray scattering study on the effect of a stearic sucrose ester on polymorphic behavior of a new sunflower oil variety. Food Res. Int. 2014, 64, 9–17. [Google Scholar] [CrossRef]
- Rodríguez-Negrette, A.C.; Rodríguez-Batiller, M.J.; García-Londoño, V.A.; Borroni, V.; Candal, R.J.; Herrera, M.L. Effect of sucrose esters on polymorphic behavior and crystallization kinetics of cupuassu fat and its fractions. J. Am. Oil Chem. Soc. 2021, 99, 27–41. [Google Scholar] [CrossRef]
- Li, Y.; Liao, T.; Liu, T.; Wang, J.; Sun, Z.; Zhao, M.; Deng, X.; Zhao, Q. Effect of stearic and oleic acid-based lipophilic emulsifiers on the crystallization of the fat blend and the stability of whipped cream. Food Chem. 2023, 428, 136762. [Google Scholar] [CrossRef]
- Zeng, D.; Cai, Y.; Liu, T.; Huang, L.; Wang, J.; Zhao, M.; Zhu, S.; Zhao, Q. Effect of hydrophobic sucrose esters with different fat acid composition and esterification degree on whipped cream properties. Food Hydrocoll. 2024, 146, 109183. [Google Scholar] [CrossRef]
- Zeng, D.; Cai, Y.; Liu, T.; Huang, L.; Liu, P.; Zhao, M.; Zhao, Q. Effect of sucrose ester S370 on interfacial layers and fat crystals network of whipped cream. Food Hydrocoll. 2021, 113, 106541. [Google Scholar] [CrossRef]
- Zeng, D.; Cai, Y.; Liu, T.; Huang, L.; Zeng, Y.; Zhao, Q.; Zhao, M. The effect of sucrose esters S1570 on partial coalescence and whipping properties. Food Hydrocoll. 2021, 125, 107429. [Google Scholar] [CrossRef]
- Liu, Y.; Binks, B.P. A novel strategy to fabricate stable oil foams with sucrose ester surfactant. J. Colloid Interface Sci. 2021, 594, 204–216. [Google Scholar] [CrossRef]
- Wakui, R.; Kamigaki, T.; Nishino, Y.; Ito, Y.; Miyazawa, A.; Shiota, M. Effect of Sucrose Esterified Fatty Acid Moieties on the Crystal Nanostructure and Physical Properties of Water-in-oil Palm-based Fat Blends. J. Oleo Sci. 2021, 70, 479–490. [Google Scholar] [CrossRef]
- De Witte, F.; Penagos, I.A.; Rondou, K.; Moens, K.; Lewille, B.; Tzompa-Sosa, D.A.; Van de Walle, D.; Van Bockstaele, F.; Skirtach, A.G.; Dewettinck, K. Insights in the Structural Hierarchy of Statically Crystallized Palm Oil. Crystals 2024, 14, 142. [Google Scholar] [CrossRef]
- Adel, R.D.; van Malssen, K.; van Duynhoven, J.; Mykhaylyk, O.O.; Voda, A. Fat Crystallite Thickness Distribution Based on SAXD Peak Shape Analysis. Eur. J. Lipid Sci. Technol. 2018, 120, 1800222. [Google Scholar] [CrossRef]
- Rondou, K.; De Witte, F.; Van Bockstaele, F.; Dewettinck, K. Microstructure development in semi-liquid shortenings upon storage. J. Am. Oil Chem. Soc. 2022, 99, 57. [Google Scholar]
- Narayanan, T.; Sztucki, M.; Zinn, T.; Kieffer, J.; Homs-Puron, A.; Gorini, J.; Van Vaerenbergh, P.; Boesecke, P. Performance of the time-resolved ultra-small-angle X-ray scattering beamline with the Extremely Brilliant Source. J. Appl. Crystallogr. 2022, 55, 98–111. [Google Scholar] [CrossRef] [PubMed]
- De Witte, F.; Van Bockstaele, F.; Dewettinck, K. Comparative study of six sucrose esters. 2024; submitted. [Google Scholar]
- Szűts, A.; Pallagi, E.; Regdon, G.; Aigner, Z.; Szabó-Révész, P. Study of thermal behaviour of sugar esters. Int. J. Pharm. 2007, 336, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Domingues, M.A.F.; da Silva, T.L.T.; Chiu, M.C.; Ribeiro, A.P.B.; Gonçalves, L.A.G. Tailoring crystallization and physical properties of palm mid-fraction with sorbitan tristearate and sucrose stearate. Food Chem. 2021, 369, 130943. [Google Scholar] [CrossRef] [PubMed]
- Molinier, V.; Kouwer, P.J.J.; Fitremann, J.; Bouchu, A.; Mackenzie, G.; Queneau, Y.; Goodby, J.W. Shape Dependence in the Formation of Condensed Phases Exhibited by Disubstituted Sucrose Esters. Chem.–A Eur. J. 2007, 13, 1763–1775. [Google Scholar] [CrossRef] [PubMed]
- Krog, N.; Sparsø, F.V. Food Emulsifiers: Their Chemical and Physical Properties; Marcel Dekker Inc.: New York, NY, USA, 2004. [Google Scholar]
- Zhou, J.; Meng, X. Effect of Glycerol Incorporation on the Liquid Crystal Structure of Sucrose Fatty Acid Ester in Aqueous Solution. Colloids Surf. A Physicochem. Eng. Asp. 2024, 684, 133213. [Google Scholar] [CrossRef]
- Bayés-García, L.; Patel, A.R.; Dewettinck, K.; Rousseau, D.; Sato, K.; Ueno, S. Lipid crystallization kinetics—Roles of external factors influencing functionality of end products. Curr. Opin. Food Sci. 2015, 4, 32–38. [Google Scholar] [CrossRef]
- Mazzanti, G.; Marangoni, A.G.; Idziak, S.H.J. Modeling phase transitions during the crystallization of a multicomponent fat under shear. Phys. Rev. E 2005, 71, 041607. [Google Scholar] [CrossRef] [PubMed]
- Cholakova, D.; Denkov, N. Polymorphic phase transitions in triglycerides and their mixtures studied by SAXS/WAXS techniques: In bulk and in emulsions. Adv. Colloid Interface Sci. 2024, 323, 103071. [Google Scholar] [CrossRef] [PubMed]
- Koizumi, H.; Kimura, K.; Takagi, M.; Michikawa, S.; Hirai, Y.; Sato, K.; Ueno, S. Effect of accumulated strain on fat bloom in CBS-based compound chocolates. CrystEngComm 2023, 25, 4562–4567. [Google Scholar] [CrossRef]
- Glatter, O. Chapter 2—General Theorems and Special Cases. In Scattering Methods and Their Application in Colloid and Interface Science; Glatter, O., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 19–32. [Google Scholar]
- Peyronel, F.; Ilavsky, J. Edible oil structures at low and intermediate concentrations. II. Ultra-small angle X-ray scattering of in situ tristearin solids in triolein. J. Appl. Phys. 2013, 114, 234902. [Google Scholar] [CrossRef]
- Peyronel, F.; Quinn, B.; Marangoni, A.G.; Pink, D.A. Edible fat structures at high solid fat concentrations: Evidence for the existence of oil-filled nanospaces. Appl. Phys. Lett. 2015, 106, 023109. [Google Scholar] [CrossRef]
- Yuki, A.; Matsuda, K.; Nishimura, A. Effect of Sucrose Polyesters on Crystallization Behavior of Vegetable Shortening and Margarine Fat. J. Jpn. Oil Chem. Soc. 1990, 39, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Bin Sintang, M.D.; Danthine, S.; Patel, A.R.; Rimaux, T.; Van De Walle, D.; Dewettinck, K. Mixed surfactant systems of sucrose esters and lecithin as a synergistic approach for oil structuring. J. Colloid Interface Sci. 2017, 504, 387–396. [Google Scholar] [CrossRef] [PubMed]

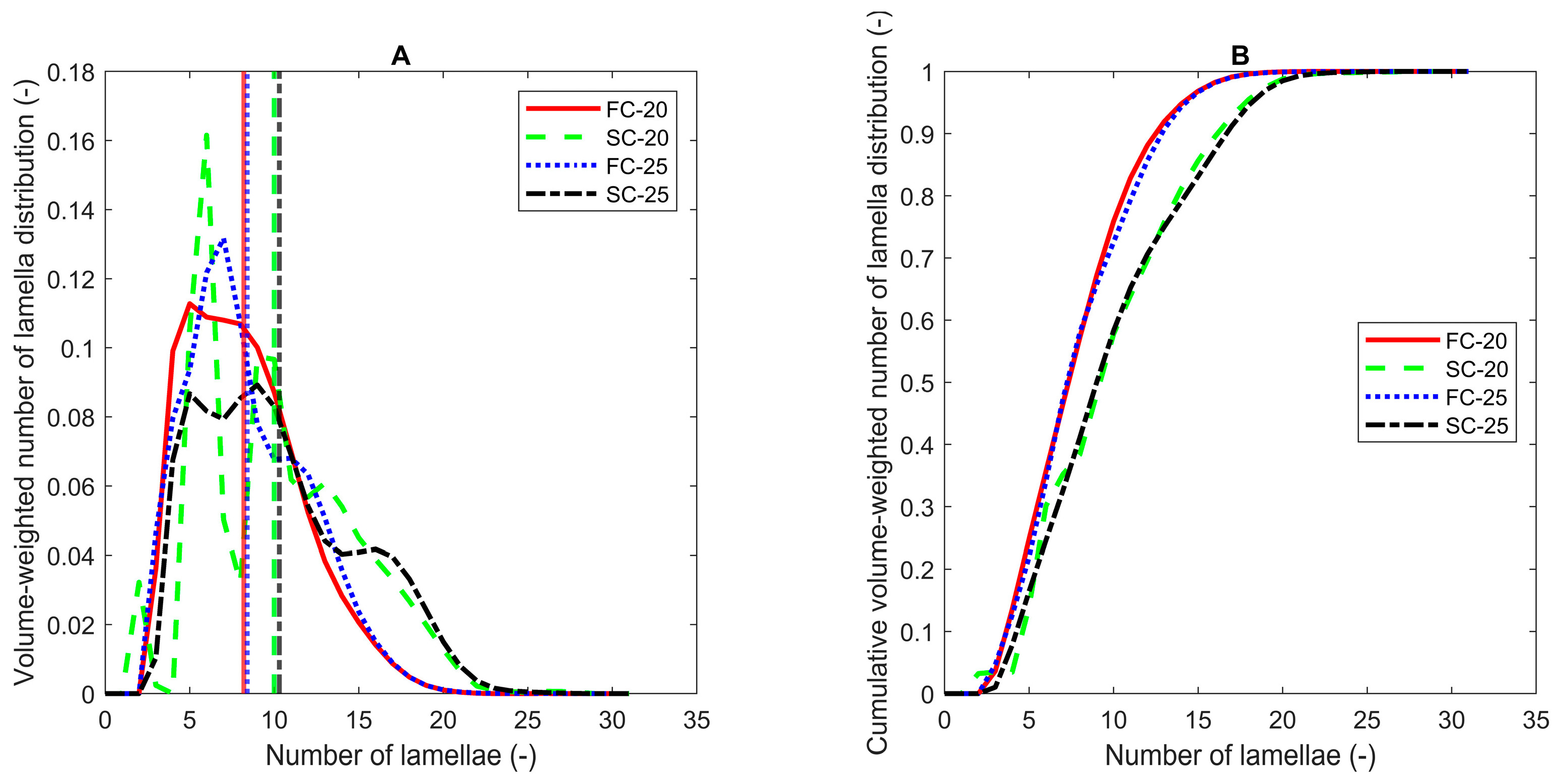


| Sample | Cooling Protocol | SAXS | |||
|---|---|---|---|---|---|
| Polymorph and Chain-Length Structure | d001 (Å) | Scherrer Average Number of Lamellae (-) | BWA Average Number of Lamellae (-) | ||
| PO | FC20 | β′-2L | 42.1 ± 0.1 a,B | 12.0 ± 0.1 b,A | 10.1 ± 0.1 a,A |
| SC20 | β′-2L | 42.1 ± 0.1 a,A | 12.3 ± 0.1 b,A | 10.2 ± 0.1 a,A | |
| FC25 | β′-2L | 42.1 ± 0.1 a,A | 12.7 ± 0.2 a,A | 10.6 ± 0.3 a,A | |
| SC25 | β′-2L | 42.1 ± 0.1 a,A | 11.5 ± 0.5 b,A | 9.6 ± 0.5 a,A | |
| POE | FC20 | β′-2L | 42.2 ± 0.1 a,A | 9.6 ± 0.2 b,A | 8.2 ± 0.1 bc,A |
| SC20 | β′-2L | 41.9 ± 0.1 b,B | 12.2 ± 0.2 a,A | 10.0 ± 0.1 ab,A | |
| FC25 | β′-2L | 41.9 ± 0.1 b,B | 10.0 ± 0.1 ab,A | 8.4 ± 0.1 b,A | |
| SC25 | β′-2L | 41.9 ± 0.1 b,B | 12.3 ± 0.2 a,A | 10.3 ± 0.1 a,A | |
| Sample | Crystallization Protocol | USAXS | ||
|---|---|---|---|---|
| Slope at Low q (-) | Cut-Off Point (Å−1) | Slope at High q (-) | ||
| PO | FC0 * | 2.7 | 0.0054 | 3.4 |
| FC20 | 1.9 | 0.0051 | 3.5 | |
| FC25 | 2.2 | 0.0025 | 3.4 | |
| SC20 | 2.0 | 0.0035 | 3.3 | |
| SC25 | 2.2 | 0.0025 | 3.4 | |
| POE | FC0 * | 2.6 | 0.0066 | 3.1 |
| FC20 | 2.1 | 0.0070 | 3.7 | |
| FC25 | 1.9 | 0.0068 | 3.4 | |
| SC20 | 2.0 | 0.0067 | 3.5 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Witte, F.; Penagos, I.A.; Van de Walle, D.; Skirtach, A.G.; Dewettinck, K.; Van Bockstaele, F. From Nucleation to Fat Crystal Network: Effects of Stearic–Palmitic Sucrose Ester on Static Crystallization of Palm Oil. Foods 2024, 13, 1372. https://doi.org/10.3390/foods13091372
De Witte F, Penagos IA, Van de Walle D, Skirtach AG, Dewettinck K, Van Bockstaele F. From Nucleation to Fat Crystal Network: Effects of Stearic–Palmitic Sucrose Ester on Static Crystallization of Palm Oil. Foods. 2024; 13(9):1372. https://doi.org/10.3390/foods13091372
Chicago/Turabian StyleDe Witte, Fien, Ivana A. Penagos, Davy Van de Walle, Andre G. Skirtach, Koen Dewettinck, and Filip Van Bockstaele. 2024. "From Nucleation to Fat Crystal Network: Effects of Stearic–Palmitic Sucrose Ester on Static Crystallization of Palm Oil" Foods 13, no. 9: 1372. https://doi.org/10.3390/foods13091372
APA StyleDe Witte, F., Penagos, I. A., Van de Walle, D., Skirtach, A. G., Dewettinck, K., & Van Bockstaele, F. (2024). From Nucleation to Fat Crystal Network: Effects of Stearic–Palmitic Sucrose Ester on Static Crystallization of Palm Oil. Foods, 13(9), 1372. https://doi.org/10.3390/foods13091372







