Metabolomics Combined with Correlation Analysis Revealed the Differences in Antioxidant Activities of Lotus Seeds with Varied Cultivars
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals Reagents
2.2. Plant Materials
2.3. Main Chemical Composition Analysis
2.4. Metabolomics Analysis by UPLC-Q/TOF-MS
2.4.1. Extraction of Metabolites from LS
2.4.2. The Conditions of UPLC-Q/TOF-MS Analysis
2.4.3. Data Preprocessing
2.5. UPLC-QqQ-MS Analysis
2.6. Antioxidant Activities Evaluation
2.6.1. DPPH•
2.6.2. Ferric Reducing Antioxidant Power (FRAP)
2.6.3. ABTS•+
2.7. Statistical Analysis
3. Results and Discussion
3.1. Analysis of the Composition of Different LS Cultivars
3.1.1. Main Chemical Composition Analysis
3.1.2. Fatty Acid Composition
3.2. Metabolomics Analysis of Different LS Cultivars Based on UPLC-Q/TOF-MS
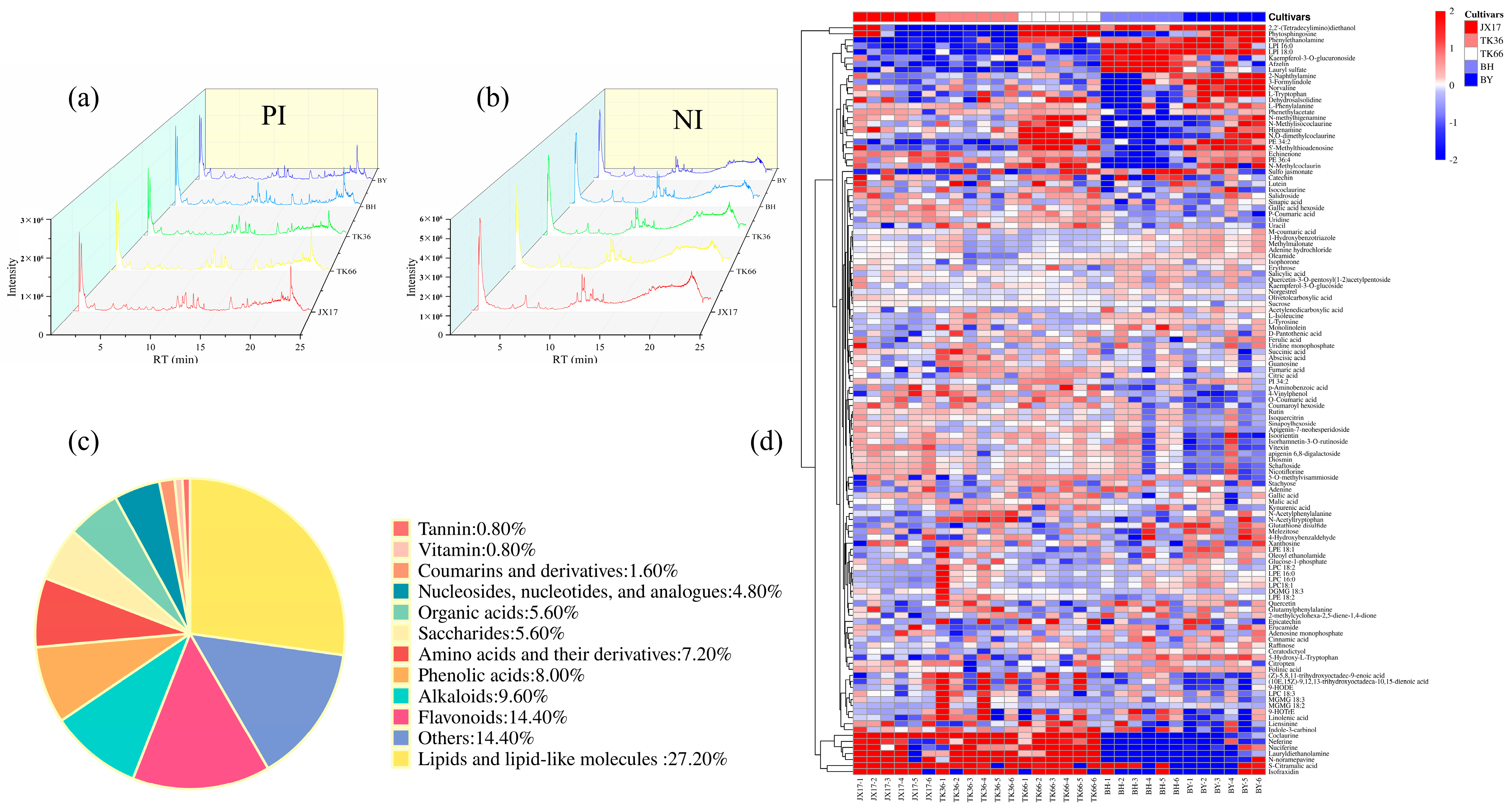
3.2.1. Metabolites Analysis of LS
3.2.2. Principal Component Analysis (PCA)
3.2.3. Partial Least Squares-Discriminant Analysis (PLS-DA)
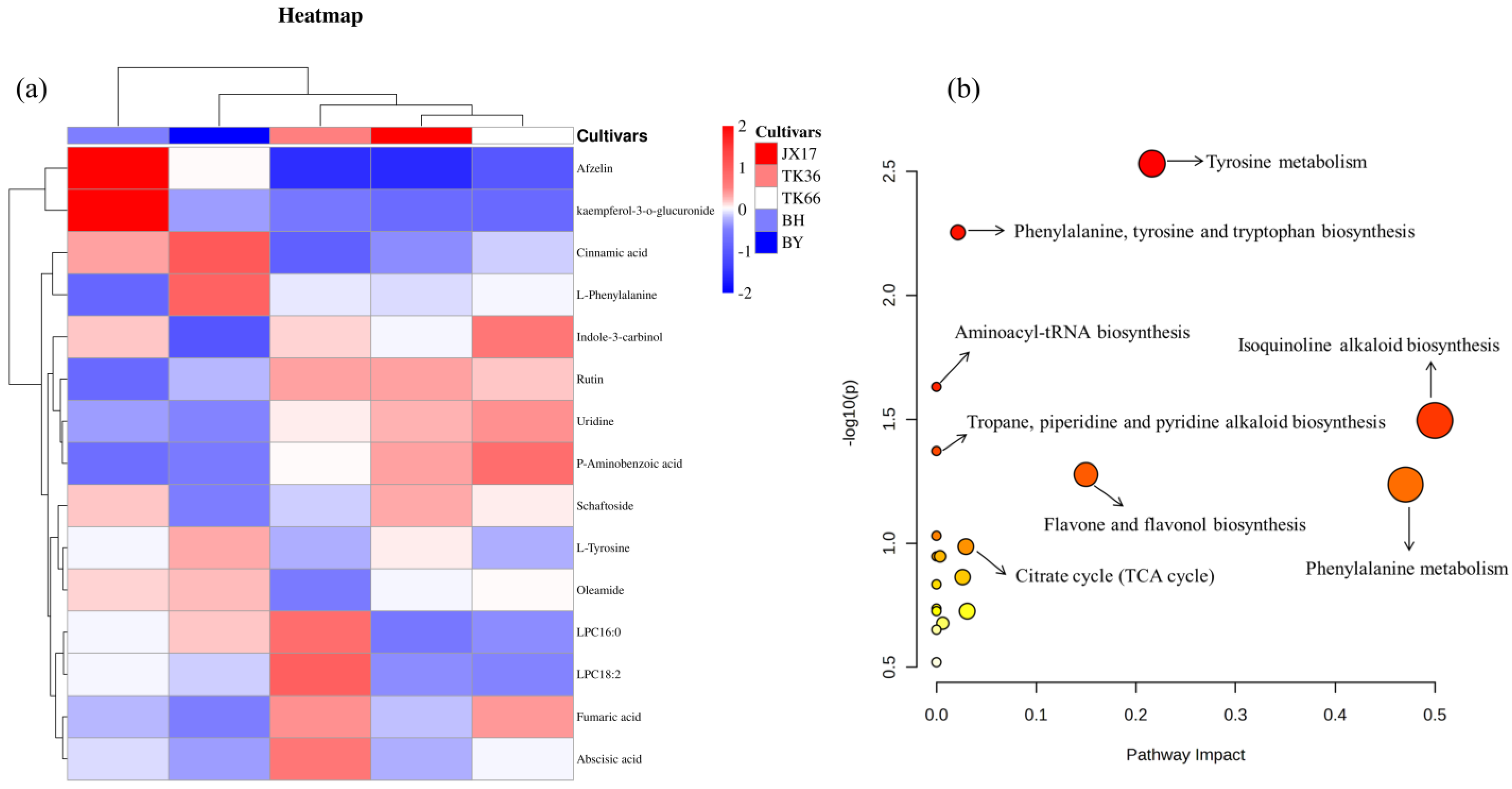
3.2.4. Metabolic Pathway Analysis
3.3. UPLC-QqQ-MS Analysis
3.4. Antioxidant Activities of Different LS Cultivars
3.5. Correlation Analysis of Chemical Composition and Antioxidant Activities
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chen, G.; Zhu, M.; Guo, M. Research advances in traditional and modern use of Nelumbo nucifera: Phytochemicals, health promoting activities and beyond. Crit. Rev. Food Sci. Nutr. 2019, 59, S189–S209. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Cheng, Y.; Zeng, M.; Wang, Z.; Qin, F.; Wang, Y.; Chen, J.; He, Z. Lotus (Nelumbo nucifera Gaertn.) leaf: A narrative review of its Phytoconstituents, health benefits and food industry applications. Trends Food Sci. Technol. 2021, 112, 631–650. [Google Scholar] [CrossRef]
- Arooj, M.; Imran, S.; Inam-Ur-Raheem, M.; Rajoka, M.S.R.; Sameen, A.; Siddique, R.; Sahar, A.; Tariq, S.; Riaz, A.; Hussain, A.; et al. Lotus seeds (Nelumbinis semen) as an emerging therapeutic seed: A comprehensive review. Food Sci. Nutr. 2021, 9, 3971–3987. [Google Scholar] [CrossRef]
- Punia Bangar, S.; Dunno, K.; Kumar, M.; Mostafa, H.; Maqsood, S. A comprehensive review on lotus seeds (Nelumbo nucifera Gaertn.): Nutritional composition, health-related bioactive properties, and industrial applications. J. Funct. Foods 2022, 89, 104937. [Google Scholar] [CrossRef]
- Wang, M.; Hu, W.-J.; Wang, Q.-H.; Yang, B.-Y.; Kuang, H.-X. Extraction, purification, structural characteristics, biological activities, and application of the polysaccharides from Nelumbo nucifera Gaertn. (lotus): A review. Int. J. Biol. Macromol. 2023, 226, 562–579. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Gao, H.; Liu, F.; Gong, L. Red-Skin Extracts of Lotus Seeds Alleviate High-Fat-Diet Induced Obesity via Regulating Lipoprotein Lipase Activity. Foods 2022, 11, 2085. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Sha, T.; Tian, W.; Wu, L.; Chen, J.; Huang, J.; Xia, Z.; Liu, K.; Sun, P.; Fan, H.; et al. Anti-inflammatory Activity of Total Alkaloids in Nelumbo nucifera and Simultaneous Determination of Major Bisbenzylisoquinolines. Rev. Bras. Farmacogn. 2023, 33, 353–363. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhu, H.; Xu, J.; Zhuang, W.; Zheng, B.; Lo, Y.M.; Huang, Z.; Tian, Y. Microwave vacuum drying of lotus (Nelumbo nucifera Gaertn.) seeds: Effects of ultrasonic pretreatment on color, antioxidant activity, and rehydration capacity. LWT—Food Sci. Technol. 2021, 149, 111603. [Google Scholar] [CrossRef]
- Zhang, Y.; Zheng, B.; Tian, Y.; Huang, S. Microwave-assisted extraction and anti-oxidation activity of polyphenols from lotus (Nelumbo nucifera Gaertn.) seeds. Food Sci. Biotechnol. 2012, 21, 1577–1584. [Google Scholar] [CrossRef]
- Zhu, Z.; Zhong, B.; Yang, Z.; Zhao, W.; Shi, L.; Aziz, A.; Rauf, A.; Aljohani, A.S.M.; Alhumaydhi, F.A.; Suleria, H.A.R. LC-ESI-QTOF-MS/MS Characterization and Estimation of the Antioxidant Potential of Phenolic Compounds from Different Parts of the Lotus (Nelumbo nucifera) Seed and Rhizome. ACS Omega 2022, 7, 14630–14642. [Google Scholar] [CrossRef]
- Cao, J.; Yu, X.; Deng, Z.; Pan, Y.; Zhang, B.; Tsao, R.; Li, H. Chemical Compositions, Antiobesity, and Antioxidant Effects of Proanthocyanidins from Lotus Seed Epicarp and Lotus Seed Pot. J. Agric. Food Chem. 2018, 66, 13492–13502. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zheng, Y.; Chen, T.; Yi, J.; Qin, L.; Rahman, K.; Lin, W. Evaluation of the quality of lotus seed of Nelumbo nucifera Gaertn from outer space mutation. Food Chem. 2007, 105, 540–547. [Google Scholar] [CrossRef]
- Sun, H.; Li, J.; Song, H.; Yang, D.; Deng, X.; Liu, J.; Wang, Y.; Ma, J.; Xiong, Y.; Liu, Y.; et al. Comprehensive analysis of AGPase genes uncovers their potential roles in starch biosynthesis in lotus seed. BMC Plant Biol. 2020, 20, 457. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.-Q.; Zhang, J.; Zhang, Y.-T.; Sun, J.-Y.; Prieto, M.A.; Simal-Gandara, J.; Putnik, P.; Li, N.-Y.; Liu, C. The link between the phenolic composition and the antioxidant activity in different small berries: A metabolomic approach. LWT 2023, 182, 114853. [Google Scholar] [CrossRef]
- Fraga-Corral, M.; Carpena, M.; Garcia-Oliveira, P.; Pereira, A.G.; Prieto, M.A.; Simal-Gandara, J. Analytical Metabolomics and Applications in Health, Environmental and Food Science. Crit. Rev. Anal. Chem. 2020, 52, 712–734. [Google Scholar] [CrossRef] [PubMed]
- Qiao, R.; Zhou, L.; Zhong, M.; Zhang, M.; Yang, L.; Yang, Y.; Chen, H.; Yang, W.; Yuan, J. Spectrum-effect relationship between UHPLC-Q-TOF/MS fingerprint and promoting gastrointestinal motility activity of Fructus aurantii based on multivariate statistical analysis. J. Ethnopharmacol. 2021, 279, 114366. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Fu, J.; Li, M.; Fragner, L.; Weckwerth, W.; Yang, P. Metabolomic and Proteomic Profiles Reveal the Dynamics of Primary Metabolism during Seed Development of Lotus (Nelumbo nucifera). Front. Plant Sci. 2016, 7, 750. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Wei, X.; Liu, Y.; Dong, G.; Hao, C.; Zhang, J.; Jiang, J.; Cheng, J.; Liu, A.; Chen, S. Identification and quantification of oligomeric proanthocyanidins, alkaloids, and flavonoids in lotus seeds: A potentially rich source of bioactive compounds. Food Chem. 2022, 379, 132124. [Google Scholar] [CrossRef]
- GB5009.3-2016; Determination of Moisture in Foods. Standards Press of China: Beijing, China, 2016.
- GB5009.5-2016; Determination of Protein in Foods. Standards Press of China: Beijing, China, 2016.
- Luo, H.-L.; Liu, X.-X.; Huang, X.-Y.; Dai, X.-M.; Zhang, M.; Fang, F.-F.; Luo, L.-P. Chemical Deterioration of Lotus Seeds During Storage. J. Food Qual. 2016, 39, 496–503. [Google Scholar] [CrossRef]
- GB5009.6-2016; Determination of Fat in Foods. Standards Press of China: Beijing, China, 2016.
- Chen, Y.; Yang, Y.; Nie, S.; Yang, X.; Wang, Y.; Yang, M.; Li, C.; Xie, M. The analysis of trans fatty acid profiles in deep frying palm oil and chicken fillets with an improved gas chromatography method. Food Control 2014, 44, 191–197. [Google Scholar] [CrossRef]
- Zhong, J.; Wang, Y.; Li, C.; Yu, Q.; Xie, J.; Dong, R.; Xie, Y.; Li, B.; Tian, J.; Chen, Y. Natural variation on free, esterified, glycosylated and insoluble-bound phenolics of Rubus chingii Hu: Correlation between phenolic constituents and antioxidant activities. Food Res. Int. 2022, 162, 112043. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-N.; Xie, R.-P.; Li, J.; Fan, Y.-W.; Liu, X.-R.; Zhang, B.; Deng, Z.-Y. Alteration on phenolic acids and the appearance of lotus (Nelumbo nucifera Gaertn) seeds dealt with antistaling agents during storage. Int. J. Food Prop. 2018, 21, 1481–1494. [Google Scholar] [CrossRef]
- Hu, L.; Zhang, H.; Hu, Z.; Chin, Y.; Li, G.; Huang, J.; Zhang, X.; Jiang, B.; Hu, Y. Differentiation of three commercial tuna species through Q-Exactive Orbitrap mass spectrometry based lipidomics and chemometrics. Food Res. Int. 2022, 158, 111509. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Sun, Z.; Zhao, Y.; Li, L.; Yang, X.; Cen, J.; Chen, S.; Li, C.; Wang, Y. Application of UHPLC-Q-TOF-MS/MS metabolomics approach to investigate the taste and nutrition changes in tilapia fillets treated with different thermal processing methods. Food Chem. 2021, 356, 129737. [Google Scholar] [CrossRef] [PubMed]
- Kadum, H.; Hamid, A.A.; Abas, F.; Ramli, N.S.; Mohammed, A.K.S.; Muhialdin, B.J.; Jaafar, A.H. Bioactive Compounds Responsible for Antioxidant Activity of Different Varieties of Date (Phoenix dactylifera L.) Elucidated by 1H-NMR Based Metabolomics. Int. J. Food Prop. 2019, 22, 462–476. [Google Scholar] [CrossRef]
- Tu, Y.; Yan, S.; Li, J. Impact of harvesting time on the chemical composition and quality of fresh lotus seeds. Hortic. Environ. Biotechnol. 2020, 61, 735–744. [Google Scholar] [CrossRef]
- Zeng, H.Y.; Cai, L.H.; Cai, X.L.; Wang, Y.J.; Li, Y.Q. Amino acid profiles and quality from lotus seed proteins. J. Sci. Food Agric. 2013, 93, 1070–1075. [Google Scholar] [CrossRef]
- Fu, Z.-F.; Tu, Z.-C.; Zhang, L.; Wang, H.; Wen, Q.-H.; Huang, T. Antioxidant activities and polyphenols of sweet potato (Ipomoea batatas L.) leaves extracted with solvents of various polarities. Food Biosci. 2016, 15, 11–18. [Google Scholar] [CrossRef]
- Chen, S.K.; Lin, H.F.; Wang, X.; Yuan, Y.; Yin, J.Y.; Song, X.X. Comprehensive analysis in the nutritional composition, phenolic species and in vitro antioxidant activities of different pea cultivars. Food Chem. X 2023, 17, 100599. [Google Scholar] [CrossRef]
- Hamany Djande, C.Y.; Piater, L.A.; Steenkamp, P.A.; Tugizimana, F.; Dubery, I.A. A Metabolomics Approach and Chemometric Tools for Differentiation of Barley Cultivars and Biomarker Discovery. Metabolites 2021, 11, 578. [Google Scholar] [CrossRef]
- Ghorbani, A. Mechanisms of antidiabetic effects of flavonoid rutin. Biomed. Pharmacother. 2017, 96, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Wang, T.; Liu, M.; Liu, J.; Zhang, Z. Transcriptome analysis of filling stage seeds among three buckwheat species with emphasis on rutin accumulation. PLoS ONE 2017, 12, e0189672. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.; Zhang, M.; Xue, H.; Yu, R.; Bao, Y.O.; Kuang, Y.; Chai, Y.; Ma, W.; Wang, J.; Shi, X.; et al. Schaftoside inhibits 3CL(pro) and PL(pro) of SARS-CoV-2 virus and regulates immune response and inflammation of host cells for the treatment of COVID-19. Acta Pharm. Sin. B 2022, 12, 4154–4164. [Google Scholar] [CrossRef]
- Hu, X.L.; Niu, Y.J.; Chen, M.; Feng, J.H.; Shen, W.; Jiang, Z.Z.; Zhang, X.Q.; Ye, W.C.; Xiong, F.; Wang, H. Preventive Effects of Total Flavonoid C-Glycosides from Abrus mollis on Nonalcoholic Fatty Liver Disease through Activating the PPARalpha Signaling Pathway. Planta Medica 2019, 85, 678–688. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kim, M.; Kim, J.M.; Lee, M.K.; Seo, S.J.; Park, K.Y. Afzelin suppresses proinflammatory responses in particulate matter-exposed human keratinocytes. Int. J. Mol. Med. 2019, 43, 2516–2522. [Google Scholar] [CrossRef] [PubMed]
- Parvez, M.K.; Ahmed, S.; Al-Dosari, M.S.; Abdelwahid, M.A.S.; Arbab, A.H.; Al-Rehaily, A.J.; Al-Oqail, M.M. Novel Anti-Hepatitis B Virus Activity of Euphorbia schimperi and Its Quercetin and Kaempferol Derivatives. ACS Omega 2021, 6, 29100–29110. [Google Scholar] [CrossRef] [PubMed]
- Righetti, L.; Rubert, J.; Galaverna, G.; Folloni, S.; Ranieri, R.; Stranska-Zachariasova, M.; Hajslova, J.; Dall’Asta, C. Characterization and Discrimination of Ancient Grains: A Metabolomics Approach. Int. J. Mol. Sci. 2016, 17, 1217. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.; Xu, Q.; Guo, H.Y.; Huang, X.; Chen, F.; Jin, L.; Quan, Z.S.; Shen, Q.K. Application of cinnamic acid in the structural modification of natural products: A review. Phytochemistry 2023, 206, 113532. [Google Scholar] [CrossRef]
- Ilica, R.A.; Kloetzer, L.; Galaction, A.I.; Cascaval, D. Fumaric acid: Production and separation. Biotechnol. Lett. 2019, 41, 47–57. [Google Scholar] [CrossRef]
- Zaib Un, N.; Akhtar, T. para-Aminobenzoic Acid-A Substrate of Immense Significance. Mini-Rev. Org. Chem. 2020, 17, 686–700. [Google Scholar] [CrossRef]
- Rozova, E.V.; Mankovskaya, I.N.; Belosludtseva, N.V.; Khmil, N.V.; Mironova, G.D. Uridine as a protector against hypoxia-induced lung injury. Sci. Rep. 2019, 9, 9418. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.A.; Patil, M.P.; Kang, M.-J.; Niyonizigiye, I.; Kim, G.-D. Biomedical application of Indole-3-carbinol: A mini-review. Phytochem. Lett. 2021, 41, 49–54. [Google Scholar] [CrossRef]
- Du, J.; Lu, X.; Geng, Z.; Yuan, Y.; Liu, Y.; Li, J.; Wang, M.; Wang, J. Metabolites changes of Eucommia ulmoides Olive samaras from different regions and cultivars. Ind. Crops Prod. 2022, 189, 115824. [Google Scholar] [CrossRef]
- Mo, Y.-N.; Cheng, F.; Yang, Z.; Shang, X.-F.; Liang, J.-P.; Shang, R.-F.; Hao, B.-C.; Wang, X.-H.; Zhang, H.-J.; Wali, A.; et al. Antioxidant Activity and the Potential Mechanism of the Fruit From Ailanthus altissima Swingle. Front. Vet. Sci. 2021, 8, 784898. [Google Scholar] [CrossRef]
- Ning, K.; Hou, C.; Wei, X.; Zhou, Y.; Zhang, S.; Chen, Y.; Yu, H.; Dong, L.; Chen, S. Metabolomics Analysis Revealed the Characteristic Metabolites of Hemp Seeds Varieties and Metabolites Responsible for Antioxidant Properties. Front. Plant Sci. 2022, 13, 904163. [Google Scholar] [CrossRef] [PubMed]
- Duan, W.; Guan, Q.; Zhang, H.L.; Wang, F.Z.; Lu, R.; Li, D.M.; Geng, Y.; Xu, Z.H. Improving flavor, bioactivity, and changing metabolic profiles of goji juice by selected lactic acid bacteria fermentation. Food Chem. 2023, 408, 135155. [Google Scholar] [CrossRef]
- Šťastná, K.; Mrázková, M.; Sumczynski, D.; Cındık, B.; Yalçın, E. The Nutritional Value of Non-Traditional Gluten-Free Flakes and Their Antioxidant Activity. Antioxidants 2019, 8, 565. [Google Scholar] [CrossRef] [PubMed]
- Eldahshan, O.A.; Azab, S.S. Anti-inflammatory Effect of Apigenin-7-neohesperidoside (Rhoifolin) in Carrageenin-Induced Rat Oedema Model. J. Appl. Pharm. Sci. 2012, 2, 74–79. [Google Scholar] [CrossRef]
- Zhu, X.; Si, F.; Hao, R.; Zheng, J.; Zhang, C. Nuciferine Protects against Obesity-Induced Nephrotoxicity through Its Hypolipidemic, Anti-Inflammatory, and Antioxidant Effects. J. Agric. Food Chem. 2023, 71, 18769–18779. [Google Scholar] [CrossRef] [PubMed]
- Sirota, T.V.; Lyamina, N.E.; Weisfeld, L.I. The Antioxidant properties of para-Aminobenzoic acid and its sodium salt. Biophysics 2017, 62, 691–695. [Google Scholar] [CrossRef]
- Lu, X.; Li, N.; Qiao, X.; Qiu, Z.; Liu, P. Composition analysis and antioxidant properties of black garlic extract. J. Food Drug Anal. 2017, 25, 340–349. [Google Scholar] [CrossRef] [PubMed]



| Components | JX17 | TK66 | TK36 | BH | BY |
|---|---|---|---|---|---|
| Moisture g/100 g FW | 58.41 ± 1.41 bc | 61.49 ± 1.65 a | 60.64 ± 1.22 a | 59.71 ± 1.86 ab | 57.00 ± 1.98 c |
| Total starch g/100 g DW | 72.79 ± 3.77 a | 72.16 ± 1.63 a | 61.34 ± 3.98 b | 62.89 ± 3.88 b | 64.08 ± 4.44 b |
| Protein g/100 g DW | 21.35 ± 0.94 b | 21.71 ± 0.72 b | 23.64 ± 0.66 a | 22.76 ± 1.03 a | 20.19 ± 0.45 c |
| Fat g/100 g DW | 1.56 ± 0.06 c | 1.76 ± 0.09 b | 2.04 ± 0.11 a | 1.74 ± 0.06 b | 1.79 ± 0.10 b |
| TPC mg GAE/100 g DW | 73.19 ± 6.51 ab | 80.60 ± 4.9 a | 73.58 ± 5.8 ab | 70.75 ± 5.59 b | 71.24 ± 5.66 b |
| Fatty Acid | JX17 | TK66 | TK36 | BH | BY |
|---|---|---|---|---|---|
| C11:0 | 0.08 ± 0.00 a | 0.06 ± 0.01 a | ND | ND | 0.08 ± 0.02 a |
| C14:0 | 0.30 ± 0.01 a | 0.29 ± 0.01 a | 0.30 ± 0.01 a | 0.29 ± 0.02 a | 0.30 ± 0.01 a |
| C16:0 | 15.66 ± 0.28 b | 15.42 ± 0.42 b | 16.74 ± 1.29 a | 15.98 ± 0.49 ab | 16.57 ± 0.69 a |
| C16:1 | 0.15 ± 0.00 ab | 0.15 ± 0.01 a | 0.15 ± 0.01b | 0.16 ± 0.01 a | 0.15 ± 0.01 b |
| C18:0 | 2.18 ± 0.11 b | 2.13 ± 0.13 bc | 2.36 ± 0.10 a | 2.00 ± 0.09 c | 2.43 ± 0.11 a |
| C18:1 | 11.33 ± 0.79 bc | 9.90 ± 0.26 c | 11.01 ± 1.27 bc | 13.00 ± 2.60 b | 16.50 ± 3.93 a |
| C18:2 | 53.79 ± 1.10 ab | 55.03 ± 0.56 a | 52.99 ± 1.19 b | 52.89 ± 1.78 b | 48.52 ± 2.41 c |
| C20:0 | 2.10 ± 0.04 bc | 2.45 ± 0.11 a | 2.23 ± 0.28 b | 1.94 ± 0.19 c | 2.62 ± 0.20 a |
| C18:3 | 5.50 ± 0.35 ab | 5.33 ± 0.25 b | 5.50 ± 0.33 ab | 5.80 ± 0.37 a | 3.60 ± 0.24 c |
| C20:3 | 6.93 ± 0.28 a | 7.25 ± 0.38 a | 6.86 ± 0.70 a | 6.11 ± 0.66 b | 7.38 ± 0.63 a |
| C20:4 | 0.20 ± 0.02 ab | 0.20 ± 0.01 ab | 0.22 ± 0.02 a | 0.18 ± 0.03 b | 0.20 ± 0.01 ab |
| C22:2 | 1.83 ± 0.13 a | 1.80 ± 0.08 a | 1.68 ± 0.20 a | 1.64 ± 0.15 a | 1.65 ± 0.17 a |
| SFA | 20.30 ± 0.31 b | 20.34 ± 0.43 b | 21.64 ± 0.96 a | 20.22 ± 0.58 b | 22.00 ± 0.77 a |
| UFA | 79.70 ± 0.31 a | 79.66 ± 0.43 a | 78.36 ± 0.96 b | 79.78 ± 0.58 a | 78.00 ± 0.77b |
| PUFA | 68.26 ± 1.09 ab | 69.61 ± 0.41 a | 67.25 ± 0.69 ab | 66.62 ± 2.40 b | 61.35 ± 3.23 c |
| UFA/SFA | 3.93 ± 0.07 a | 3.92 ± 0.11 a | 3.63 ± 0.20b | 3.95 ± 0.14 a | 3.55 ± 0.17 b |
| Compounds | TK36 | TK66 | JX17 | BY | BH |
|---|---|---|---|---|---|
| Malic acid | 402.37 ± 25.81 b | 418.14 ± 41.29 a | 357.25 ± 39.63 bc | 359.76 ± 40.18 bc | 323.47 ± 7.19 c |
| Fumaric acid | 16.76 ± 0.79 a | 15.26 ± 0.66 ab | 14.25 ± 0.43 b | 15.17 ± 1.24 b | 14.77 ± 1.16 b |
| Gallic acid | 0.28 ± 0.03 b | 0.27 ± 0.02 ba | 0.33 ± 0.03 b | 0.29 ± 0.03 b | 0.43 ± 0.09 a |
| Catechin | 1.99 ± 0.21 b | 2.89 ± 0.39 a | 2.07 ± 0.29 b | 2.53 ± 0.20 ab | 2.40 ± 0.31 ab |
| Epicatechin | 0.26 ± 0.03 ab | 0.29 ± 0.04 a | 0.21 ± 0.03 c | 0.29 ± 0.03 ab | 0.22 ± 0.03 bc |
| Schaftoside | 7.13 ± 1.95 b | 6.80 ± 1.05 bc | 11.33 ± 1.61 a | 4.09 ± 0.66 c | 7.65 ± 1.43 b |
| Isoorientin | 3.03 ± 0.63 bc | 4.44 ± 1.13 b | 8.24 ± 1.21 a | 2.02 ± 0.66 c | 4.97 ± 1.01 b |
| p-Coumaric acid | 0.88 ± 0.16 bc | 1.36 ± 0.15 a | 0.91 ± 0.15 b | 0.60 ± 0.10 d | 0.64 ± 0.10 cd |
| Sinapic acid | 0.28 ± 0.02 ab | 0.26 ± 0.07 b | 0.39 ± 0.08 a | 0.21 ± 0.06 b | 0.27 ± 0.03 b |
| Ferulic acid | 0.45 ± 0.07 b | 0.45 ± 0.08 b | 0.61 ± 0.09 ab | 0.75 ± 0.12 a | 0.44 ± 0.09 b |
| Rutin | 7.80 ± 1.57 b | 10.61 ± 2.01 a | 10.04 ± 0.82 a | 7.50 ± 1.03 b | 7.86 ± 0.83 b |
| Isoquercitrin | 1.86 ± 0.44 ab | 1.78 ± 0.30 bc | 2.35 ± 0.33 ab | 1.53 ± 0.25 c | 2.32 ± 0.12 a |
| Cinnamic acid | 0.42 ± 0.07 bc | 0.32 ± 0.09 c | 0.57 ± 0.58 b | 0.58 ± 0.10 b | 0.91 ± 0.18 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, X.; Wang, Y.; Yan, X.; Leng, T.; Xie, J.; Yu, Q.; Chen, Y. Metabolomics Combined with Correlation Analysis Revealed the Differences in Antioxidant Activities of Lotus Seeds with Varied Cultivars. Foods 2024, 13, 1084. https://doi.org/10.3390/foods13071084
Yu X, Wang Y, Yan X, Leng T, Xie J, Yu Q, Chen Y. Metabolomics Combined with Correlation Analysis Revealed the Differences in Antioxidant Activities of Lotus Seeds with Varied Cultivars. Foods. 2024; 13(7):1084. https://doi.org/10.3390/foods13071084
Chicago/Turabian StyleYu, Xinjin, Yuting Wang, Xiaoli Yan, Tuo Leng, Jianhua Xie, Qiang Yu, and Yi Chen. 2024. "Metabolomics Combined with Correlation Analysis Revealed the Differences in Antioxidant Activities of Lotus Seeds with Varied Cultivars" Foods 13, no. 7: 1084. https://doi.org/10.3390/foods13071084
APA StyleYu, X., Wang, Y., Yan, X., Leng, T., Xie, J., Yu, Q., & Chen, Y. (2024). Metabolomics Combined with Correlation Analysis Revealed the Differences in Antioxidant Activities of Lotus Seeds with Varied Cultivars. Foods, 13(7), 1084. https://doi.org/10.3390/foods13071084







