Variations in Key Aroma Compounds and Aroma Profiles in Yellow and White Cultivars of Flammulina filiformis Based on Gas Chromatography–Mass Spectrometry–Olfactometry, Aroma Recombination, and Omission Experiments Coupled with Odor Threshold Concentrations
Abstract
1. Introduction
2. Materials and Methods
2.1. Mushroom Samples and Chemicals
2.2. Headspace Solid-Phase Microextraction (HS–SPME) Analysis
2.3. Solvent-Assisted Flavor Evaporation (SAFE)
2.4. Gas Chromatography–Mass Spectrometry–Olfactometry (GC-MS-O) Analysis
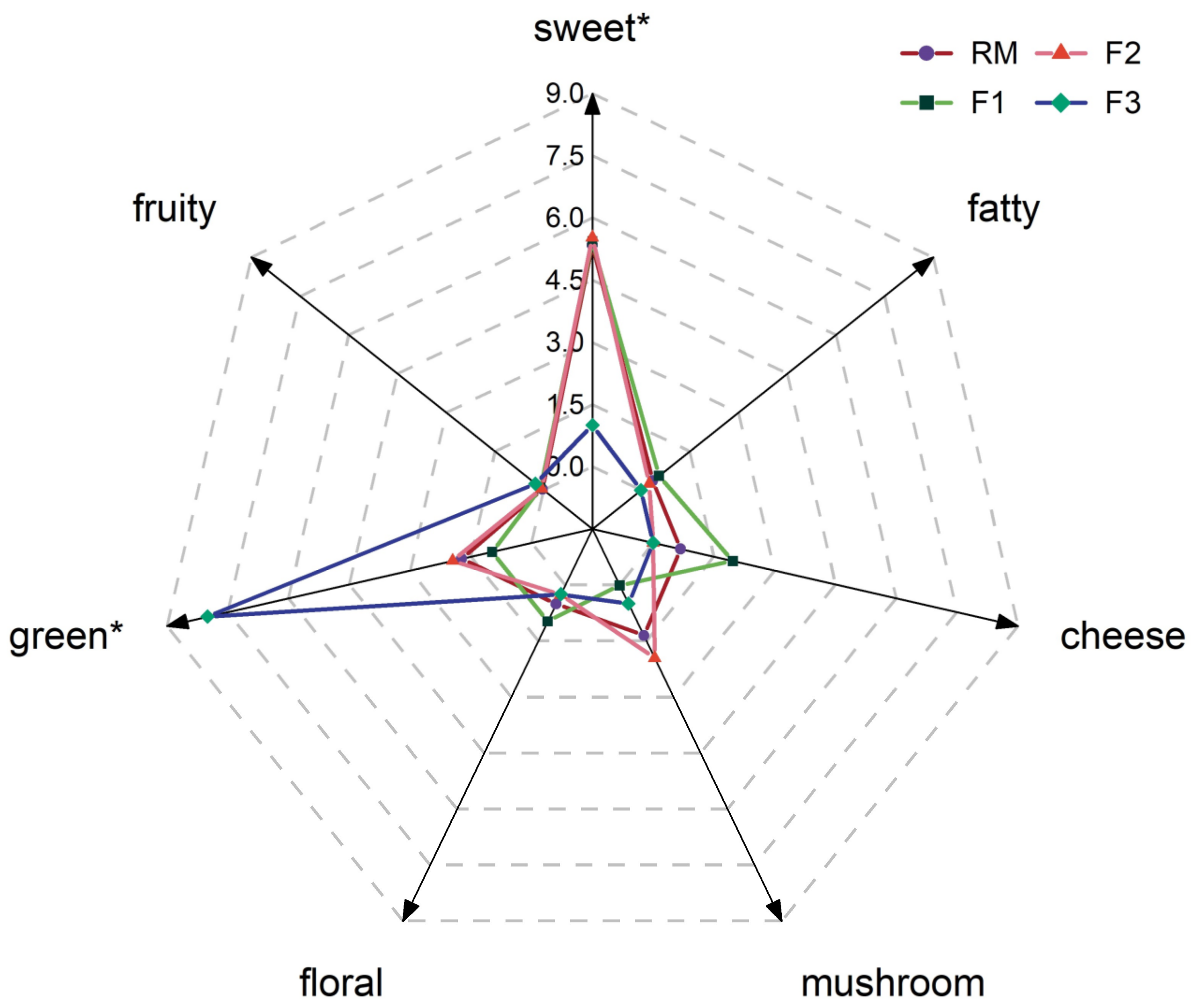
2.5. Aroma Profile Analysis
2.6. Aroma Extract Dilution Analysis (AEDA)
2.7. Identification and Quantification of the Key Aroma Compounds
2.8. Odor Threshold Concentrations
2.9. Odor Activity Values (OAVs)
2.10. Aroma Omission/Recombination Experiments
2.11. Statistical Analysis
3. Results and Discussion
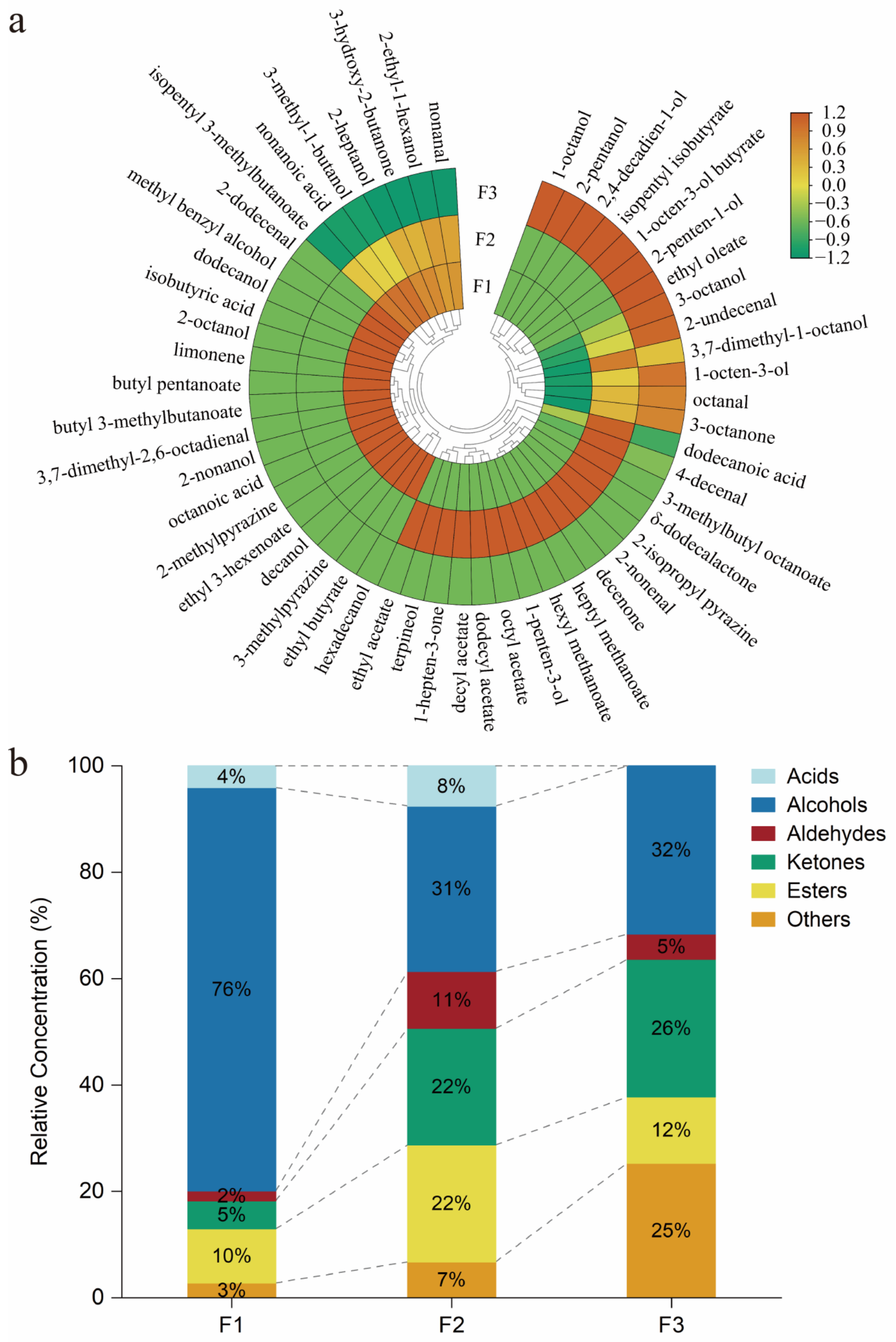
3.1. Identification of the Aroma-Active Compounds in F. filiformis
3.2. Further Confirmation for Aroma Attributes by GC-O and Contributions by AEDA
3.3. Quantitative Analysis and OAV Referring to Volatile Compounds
3.4. Aroma Recombination and Omission Experiments
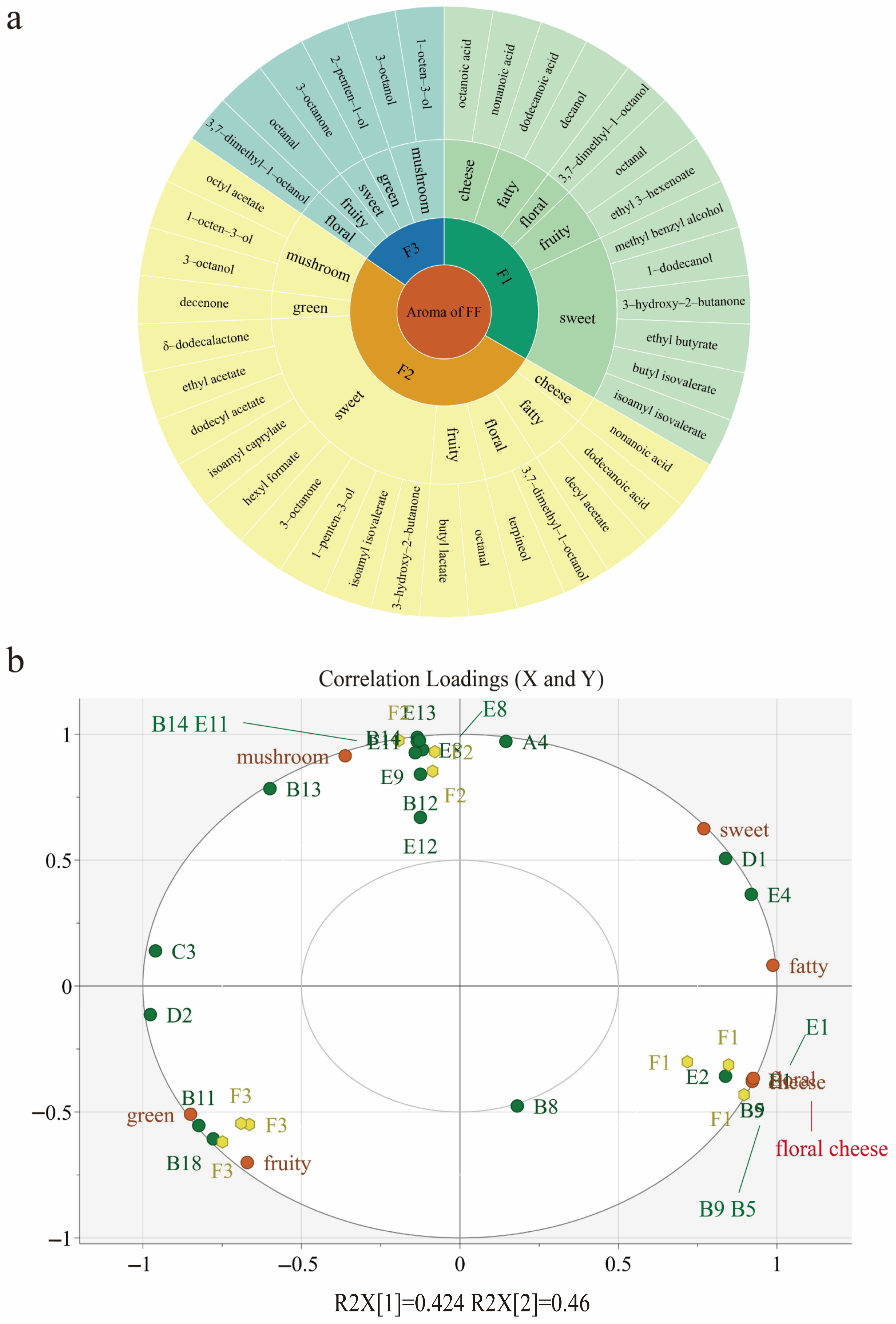
3.5. Aroma Attributes’ Correlation with Aroma-Active Compounds Using Partial Least-Squares Regression (PLSR)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Fu, Y.; Yu, Y.; Tan, H.; Wang, B.; Peng, W.; Sun, Q. Metabolomics reveals dopa melanin involved in the enzymatic browning of the yellow cultivars of East Asian golden needle mushroom (Flammulina filiformis). Food Chem. 2022, 370, 131295. [Google Scholar] [CrossRef]
- Li, H.; Shi, L.; Tang, W.; Xia, W.; Zhong, Y.; Xu, X.; Xie, B.; Tao, Y. Comprehensive Genetic Analysis of Monokaryon and Dikaryon Populations Provides Insight into Cross-Breeding of Flammulina filiformis. Front. Microbiol. 2022, 13, 887259. [Google Scholar] [CrossRef]
- Liu, X.B.; Feng, B.; Li, J.; Yan, C.; Yang, Z.L. Genetic diversity and breeding history of Winter Mushroom (Flammulina velutipes) in China uncovered by genomic SSR markers. Gene 2016, 591, 227–235. [Google Scholar] [CrossRef]
- Fang, D.; Yang, W.; Kimatu, B.M.; Zhao, L.; An, X.; Hu, Q. Comparison of flavour qualities of mushrooms (Flammulina velutipes) packed with different packaging materials. Food Chem. 2017, 232, 1–9. [Google Scholar] [CrossRef]
- Hou, Z.; Xia, R.; Li, Y.; Xu, H.; Wang, Y.; Feng, Y.; Pan, S.; Wang, Z.; Ren, H.; Qian, G.; et al. Key components, formation pathways, affecting factors, and emerging analytical strategies for edible mushrooms aroma: A review. Food Chem. 2023, 438, 137993. [Google Scholar] [CrossRef]
- Sun, M.; Ni, L.; Huang, Y.; Yang, M.; Cheng, G.; Zhang, M.; Wu, M.; Ma, C. Effects of different drying treatments on the microstructure, free amino acids, volatile compounds and antioxidant activity of Flammulina velutipes root. Food Chem. X 2023, 18, 100656. [Google Scholar] [CrossRef]
- Xia, R.; Wang, L.; Xin, G.; Bao, X.; Sun, L.; Xu, H.; Hou, Z. Preharvest and postharvest applications of 1-MCP affect umami taste and aroma profiles of mushrooms (Flammulina velutipes). LWT 2021, 144, 111176. [Google Scholar] [CrossRef]
- Yang, W.; Yu, J.; Pei, F.; Mariga, A.M.; Ma, N.; Fang, Y.; Hu, Q. Effect of hot air drying on volatile compounds of Flammulina velutipes detected by HS-SPME–GC–MS and electronic nose. Food Chem. 2016, 196, 860–866. [Google Scholar] [CrossRef]
- Zhu, R.; Wen, Y.; Wu, W.; Zhang, L.; Salman Farid, M.; Shan, S.; Wen, J.; Farag, M.A.; Zhang, Y.; Zhao, C. The flavours of edible mushrooms: A comprehensive review of volatile organic compounds and their analytical methods. Crit. Rev. Food Sci. Nutr. 2022, 1–15. [Google Scholar] [CrossRef]
- Pennerman, K.K.; Yin, G.; Bennett, J.W. Eight-carbon volatiles: Prominent fungal and plant interaction compounds. J. Exp. Bot. 2021, 73, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Wan, J.; Wang, Z.; Sun, M.; Feng, T.; Ho, C.T.; Song, S. Variation of Volatile Compounds and Corresponding Aroma Profiles in Chinese Steamed Bread by Various Yeast Species Fermented at Different Times. J. Agric. Food Chem. 2022, 70, 3795–3806. [Google Scholar] [CrossRef]
- Xu, X.; Xu, R.; Jia, Q.; Feng, T.; Huang, Q.; Ho, C.-T.; Song, S. Identification of dihydro-β-ionone as a key aroma compound in addition to C8 ketones and alcohols in Volvariella volvacea mushroom. Food Chem. 2019, 293, 333–339. [Google Scholar] [CrossRef]
- Yao, L.; Mo, Y.; Chen, D.; Feng, T.; Song, S.; Wang, H.; Sun, M. Characterization of key aroma compounds in Xinjiang dried figs (Ficus carica L.) by GC–MS, GC–olfactometry, odor activity values, and sensory analyses. LWT 2021, 150, 111982. [Google Scholar] [CrossRef]
- Wan, J.; Liu, Q.; Ma, C.; Muhoza, B.; Huang, Y.; Sun, M.; Song, S.; Ho, C.T. Characteristic flavour fingerprint disclosure of dzo beef in Tibet by applying SAFE-GC-O-MS and HS-GC-IMS technology. Food Res. Int. 2023, 166, 112581. [Google Scholar] [CrossRef]
- Liu, F.; Wang, S.H.; Jia, D.H.; Tan, H.; Wang, B.; Zhao, R.L. Development of Multiple Nucleotide Polymorphism Molecular Markers for Enoki Mushroom (Flammulina filiformis) Cultivars Identification. J. Fungi 2023, 9, 330. [Google Scholar] [CrossRef]
- Li, W.; Li, R.; Chen, W.; Feng, J.; Wu, D.; Zhang, Z.; Zhang, J.; Yang, Y. The anabolism of sulphur aroma volatiles responds to enzymatic and non-enzymatic reactions during the drying process of shiitake mushrooms. Food Chem. 2022, 371, 131123. [Google Scholar] [CrossRef]
- ISO 8589; Sensory Analysis—General Guidance for the Design of Test Rooms. ISO: Geneva, Switzerland, 2007.
- ISO 8586; Sensory Analysis—Selection and Training of Sensory Assessors. ISO: Geneva, Switzerland, 2023.
- Feng, T.; Sun, J.; Wang, K.; Song, S.; Chen, D.; Zhuang, H.; Lu, J.; Li, D.; Meng, X.; Shi, M.; et al. Variation in Volatile Compounds of Raw Pu-Erh Tea upon Steeping Process by Gas Chromatography–Ion Mobility Spectrometry and Characterization of the Aroma-Active Compounds in Tea Infusion Using Gas Chromatography–Olfactometry–Mass Spectrometry. J. Agric. Food Chem. 2022, 70, 13741–13753. [Google Scholar] [CrossRef]
- Zhao, Y.; Wei, W.; Tang, L.; Wang, D.; Wang, Y.; Wu, Z.; Zhang, W. Characterization of aroma and bacteria profiles of Sichuan industrial paocai by HS-SPME-GC-O-MS and 16S rRNA amplicon sequencing. Food Res. Int. 2021, 149, 110667. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zeng, X.; Song, H.; Xi, Y.; Li, Y.; Hui, B.; Li, H.; Li, J. Characterization of the aroma profiles of cold and hot break tomato pastes by GC-O-MS, GC × GC-O-TOF-MS, and GC-IMS. Food Chem. 2023, 405, 134823. [Google Scholar] [CrossRef] [PubMed]
- ASTM. E679-19; Standard Practice for Determination of Odor and Taste Threshold by a Forced-Choice Ascending Concentration Series Method of Limits. ASTM International: West Conshohocken, PA, USA, 2019.
- Gottmann, J.; Vestner, J.; Fischer, U. Sensory relevance of seven aroma compounds involved in unintended but potentially fraudulent aromatization of wine due to aroma carryover. Food Chem. 2023, 402, 134160. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Zhang, G.; Xu, L.; Duan, J.; Li, H.; Sun, J.; Sun, B. Investigation on the interaction between 1,3-dimethyltrisulfide and aroma-active compounds in sesame-flavour baijiu by Feller Additive Model, Odor Activity Value and Partition Coefficient. Food Chem. 2023, 410, 135451. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yu, P.; Sun, J.; Jia, Y.; Wan, C.; Zhou, Q.; Huang, F. Investigation of volatile thiol contributions to rapeseed oil by odor active value measurement and perceptual interactions. Food Chem. 2022, 373, 131607. [Google Scholar] [CrossRef] [PubMed]
- ISO 4120; Sensory Analysis—Methodology—Triangle Test. ISO: Geneva, Switzerland, 2021.
- Wang, L.; Wu, L.; Xiang, D.; Huang, H.; Han, Y.; Zhen, P.; Shi, B.; Chen, S.; Xu, Y. Characterization of key aroma compounds in aged Qingxiangxing baijiu by comparative aroma extract dilution analysis, quantitative measurements, aroma recombination, and omission studies. Food Chem. 2023, 419, 136027. [Google Scholar] [CrossRef]
- Wang, P.; Kan, Q.; Yang, L.; Huang, W.; Wen, L.; Fu, J.; Liu, Z.; Lan, Y.; Huang, Q.; Ho, C.T.; et al. Characterization of the key aroma compounds in soy sauce by gas chromatography-mass spectrometry-olfactometry, headspace-gas chromatography-ion mobility spectrometry, odor activity value, and aroma recombination and omission analysis. Food Chem. 2023, 419, 135995. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Chen, H.; Zhang, Y.; Thomas, H.R.; Frank, M.H.; He, Y.; Xia, R. TBtools: An Integrative Toolkit Developed for Interactive Analyses of Big Biological Data. Mol. Plant 2020, 13, 1194–1202. [Google Scholar] [CrossRef] [PubMed]
- Vlot, A.C.; Rosenkranz, M. Volatile compounds—The language of all kingdoms? J. Exp. Bot. 2022, 73, 445–448. [Google Scholar] [CrossRef] [PubMed]
- Okur, S.; Qin, P.; Chandresh, A.; Li, C.; Zhang, Z.; Lemmer, U.; Heinke, L. An Enantioselective e-Nose: An array of Nanoporous Homochiral MOF Films for Stereospecific Sensing of Chiral Odors. Angew. Chem. Int. Ed. 2020, 60, 3566–33571. [Google Scholar] [CrossRef]
- Smit, M.S.; Maseme, M.J.; van Marwijk, J.; Aschenbrenner, J.C.; Opperman, D.J. Delineation of the CYP505E subfamily of fungal self-sufficient in-chain hydroxylating cytochrome P450 monooxygenases. Appl. Microbiol. Biotechnol. 2023, 107, 735–747. [Google Scholar] [CrossRef]
- Xia, R.; Wang, Z.; Xu, H.; Hou, Z.; Li, Y.; Wang, Y.; Feng, Y.; Zhang, X.; Xin, G. Cutting root treatment combined with low-temperature storage regimes on non-volatile and volatile compounds of Oudemansiella raphanipes. LWT 2022, 166, 113754. [Google Scholar] [CrossRef]
- Cho, I.H.; Kim, S.Y.; Choi, H.K.; Kim, Y.S. Characterization of Aroma-Active Compounds in Raw and Cooked Pine-Mushrooms (Tricholoma matsutake Sing.). J. Agric. Food Chem. 2006, 54, 6332–6335. [Google Scholar] [CrossRef]
- Sun, L.; Xin, G.; Hou, Z.; Zhao, X.; Xu, H.; Bao, X.; Xia, R.; Li, Y.; Li, L. Biosynthetic Mechanism of Key Volatile Biomarkers of Harvested Lentinula edodes Triggered by Spore Release. J. Agric. Food Chem. 2021, 69, 9350–9361. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Wan, J.; Sun, M.; Feng, T.; Liu, Q.; Song, S.; Zhang, X.; Ho, C.T. Flavour profile disclosure of Chinese steamed breads (CSBs) by sensomics approach. Food Biosci. 2023, 51, 102198. [Google Scholar] [CrossRef]
- Selli, S.; Guclu, G.; Sevindik, O.; Kelebek, H. Variations in the key aroma and phenolic compounds of champignon (Agaricus bisporus) and oyster (Pleurotus ostreatus) mushrooms after two cooking treatments as elucidated by GC–MS-O and LC-DAD-ESI-MS/MS. Food Chem. 2021, 354, 129576. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Li, Z.; Liu, H.; Zhang, H.; Wang, L.; Chen, H. Characterization of the key aroma compounds in Semnostachya menglaensis Tsui by gas chromatography-olfactometry, odor activity values, aroma recombination, and omission analysis. Food Res. Int. 2020, 131, 108948. [Google Scholar] [CrossRef]
- Yin, C.; Fan, X.; Fan, Z.; Shi, D.; Yao, F.; Gao, H. Comparison of non-volatile and volatile flavour compounds in six Pleurotus mushrooms. J. Sci. Food Agric. 2018, 99, 1691–1699. [Google Scholar] [CrossRef]
- Lu, H.; Song, W.; Shang, X.D.; Liu, J.Y.; Zhang, D.; Li, L.; Wang, R.J.; Zhai, X.T.; Feng, T. Expression of terpene synthase-related genes in parents and offspring of Flammulina filiformis based on differences in volatile aroma components. Food Chem. Mol. Sci. 2023, 6, 100156. [Google Scholar] [CrossRef]
| FD c | ||||||||
|---|---|---|---|---|---|---|---|---|
| No. | Name | RI/KI a | OD b | F1 | F2 | F3 | IM d | |
| acids | A1 | isobutyric acid | 1318 | sour, cheese | 1 | MS, O, RI | ||
| A2 | dodecanoic acid | 1947 | fatty, coconut | 128 | 4 | MS, O, RI | ||
| A3 | octanoic acid | 2053 | cheese, fat, grass | 4 | MS, O, RI | |||
| A4 | nonanoic acid | 2157 | cheese | 512 | 1 | MS, O, RI | ||
| alcohols | B1 | 3-methyl-1-butanol | 1218 | burnt, cocoa, floral, malt | 1 | 512 | MS, O, RI | |
| B2 | dodecanol | 1264 | earthy, sweet, honey, coconut | 2 | MS, O, RI | |||
| B3 | 2-ethyl-1-hexanol | 1483 | green, rose | 2 | 2 | 1 | MS, O, RI | |
| B4 | 2-heptanol | 1575 | citrus, earth, fried, mushroom | 1 | 8 | MS, O, RI | ||
| B5 | 2-octanol | 1599 | fat, mushroom | 1 | MS, O, RI | |||
| B6 | 2-nonanol | 1618 | cucumber, green | 1 | MS, O, RI | |||
| B7 | hexadecanol | 1638 | waxy, clean, floral, oily | 1 | MS, O, RI | |||
| B8 | 3,7-dimethyl-1-octanol | 1641 | floral | 256 | 8 | 1 | MS, O, RI | |
| B9 | methyl benzyl alcohol | 2180 | sweet, gardenia, floral | 2 | MS, O, RI | |||
| B10 | decanol | 1385 | fat, oil | 16 | MS, O, RI | |||
| B11 | 3-octanol | 1261 | mushroom | 512 | 1 | MS, O, RI | ||
| B12 | 1-penten-3-ol | 1333 | butter, sweet | 64 | MS, O, RI | |||
| B13 | 1-octen-3-ol | 1410 | earth, fat, mushroom | 4 | 1 | MS, O, RI | ||
| B14 | terpineol | 1867 | floral | 128 | MS, O, RI | |||
| B15 | 2-pentanol | 1095 | oil, green | 1 | MS, O, RI | |||
| B16 | 2,4-decadien-1-ol | 1225 | fatty, citrus | 2 | MS, O, RI | |||
| B17 | 1-octanol | 1403 | bitter almond, fat, floral | 1 | MS, O, RI | |||
| B18 | 2-penten-1-ol | 1333 | green | 32 | MS, O, RI | |||
| aldehydes | C1 | 2-dodecenal | 1293 | fruit, citrus | 1 | MS, O, RI | ||
| C2 | 3,7-dimethyl-2,6-octadienal | 1339 | lemon | 8 | MS, O, RI | |||
| C3 | octanal | 1347 | citrus, fat, green, oil | 8 | 16 | 1 | MS, O, RI | |
| C4 | nonanal | 1399 | fat, floral, green, lemon | 2 | 1 | MS, O, RI | ||
| C5 | 2-undecenal | 1245 | citrus, orange peel | 8 | 1 | MS, O, RI | ||
| C6 | 2-nonenal | 1293 | green, cucumber | 1024 | MS, O, RI | |||
| C7 | 4-decenal | 1738 | citrus | 1 | 1 | MS, O, RI | ||
| ketones | D1 | 3-hydroxy-2-butanone | 1583 | sweet, buttery, creamy | 2 | 1 | MS, O, RI | |
| D2 | 3-octanone | 1269 | butter, herb, sweet, mushroom | 512 | 4 | MS, O, RI | ||
| D3 | 1-hepten-3-one | 1641 | metallic | 16 | MS, O, RI | |||
| D4 | decenone | 1692 | fatty, green, fruity | 1024 | MS, O, RI | |||
| D5 | δ-dodecalactone | 2146 | fruit, sweet, peach, coconut | 1 | MS, O, RI | |||
| esters | E1 | ethyl butyrate | 1188 | apple, sweet, cheese, pineapple | 1 | MS, O, RI | ||
| E2 | butyl 3-methylbutanoate | 1685 | fruity, apple, sweet | 64 | MS, O, RI | |||
| E3 | ethyl 3-hexenoate | 1841 | fruity, pineapple, green | 8 | MS, O, RI | |||
| E4 | isopentyl 3-methylbutanoate | 1855 | sweet, fruity, apple | 64 | 256 | MS, O, RI | ||
| E5 | butyl pentanoate | 1888 | sweet, fruity, pineapple | 256 | MS, O, RI | |||
| E6 | heptyl methanoate | 1222 | green, floral, apple | 1 | MS, O, RI | |||
| E7 | hexyl methanoate | 1403 | apple, banana, sweet | 1 | MS, O, RI | |||
| E8 | octyl acetate | 1458 | green, earthy, mushroom | 512 | MS, O, RI | |||
| E9 | 3-methylbutyl octanoate | 1829 | sweet, fruity, pineapple | 1024 | MS, O, RI | |||
| E10 | dodecyl acetate | 1907 | sweet, waxy | 4 | MS, O, RI | |||
| E11 | decyl acetate | 1929 | oil, orange | 128 | MS, O, RI | |||
| E12 | ethyl acetate | 2114 | sweet, pineapple | 8 | MS, O, RI | |||
| E13 | isopentyl isobutyrate | 1655 | fruity, green, grape | 2 | MS, O, RI | |||
| E14 | ethyl oleate | 2225 | fatty, milky | 8 | MS, O, RI | |||
| E15 | 1-octen-3-ol butyrate | 1874 | fruity, floral | 8 | MS, O, RI | |||
| others | F1 | limonene | 1222 | citrus, mint | 1 | MS, O, RI | ||
| F2 | 2-methylpyrazine | 1277 | cocoa, green | 1 | MS, O, RI | |||
| F3 | 3-methylpyrazine | 2013 | nut | 1 | MS, O, RI | |||
| F4 | 2-isopropyl pyrazine | 2181 | minty, green, nutty, honey | 4 | MS, O, RI | |||
| Concentration (mg/kg) c | OAV f | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| No. | Name | F1 | F2 | F3 | QI d | OT e | F1 | F2 | F3 |
| A2 | dodecanoic acid | 0.0447 ± 0.0136 b | 0.1446 ± 0.0428 a | 43, 60, 73 | 11.2681 | 4 | 13 | ||
| A3 | octanoic acid | 0.0207 ± 0.0095 | 60, 73 | 0.19 | 109 | ||||
| A4 | nonanoic acid | 0.0832 ± 0.0070 b | 0.2966 ± 0.0435 a | 57, 60, 73 | 5.8471 | 14 | 51 | ||
| B9 | methyl benzyl alcohol | 0.5072 ± 0.0756 | 77, 79, 107 | 0.4074 | >1000 | ||||
| B10 | decanol | 0.0675 ± 0.0008 | 55, 70 | 2.5918 | 26 | ||||
| B11 | 3-octanol | 0.0604 ± 0.0266 b | 0.7607 ± 0.1035 a | 55, 59, 83 | 0.1709 | 354 | >1000 | ||
| B12 | 1-penten-3-ol | 0.0189 ± 0.0129 | 57 | 0.1578 | 120 | ||||
| B13 | 1-octen-3-ol | 0.2851 ± 0.0189 a | 0.1421 ± 0.0357 b | 43, 57 | 0.0625 | >1000 | >1000 | ||
| B14 | terpineol | 0.1339 ± 0.0048 | 59, 93, 121 | 0.7509 | 178 | ||||
| B2 | 1-dodecanol | 0.0052 ± 0.0069 | 43, 55, 69 | 3.4348 | 2 | ||||
| B18 | 2-penten-1-ol | 3.4681 ± 0.6225 | 57 | 0.72 | >1000 | ||||
| B8 | 3,7-dimethyl-1-octanol | 0.5577 ± 0.0211 a | 0.5355 ± 0.0262 a | 0.5531 ± 0.0039 a | 41, 55, 56 | 0.0009 | >1000 | >1000 | >1000 |
| C3 | octanal | 0.0259 ± 0.0006 b | 0.0317 ± 0.0007 a | 0.0336 ± 0.0014 a | 43, 44 | 0.0034 | >1000 | >1000 | >1000 |
| D1 | 3-hydroxy-2-butanone | 27.9547 ± 4.9313 a | 24.4867 ± 3.6591 a | 43, 45 | 0.59 | >1000 | >1000 | ||
| D2 | 3-octanone | 0.8568 ± 0.2728 b | 1.6570 ± 0.1777 a | 43, 57, 72 | 0.0330 | >1000 | >1000 | ||
| D4 | decenone | 0.1801 ± 0.0148 | 43, 55 | 10.2799 | 18 | ||||
| D5 | δ-dodecalactone | 0.6141 ± 0.0720 | 99 | 0.098 | >1000 | ||||
| E1 | ethyl butyrate | 0.0972 ± 0.0109 | 43, 71 | 0.0104 | >1000 | ||||
| E7 | hexyl methanoate | 0.0852 ± 0.0372 | 56 | 8.8135 | 10 | ||||
| E8 | octyl acetate | 0.2401 ± 0.0665 | 43 | 0.1105 | >1000 | ||||
| E9 | 3-methylbutyl octanoate | 2.0699 ± 0.8334 | 70, 127 | 0.07 | >1000 | ||||
| E10 | dodecyl acetate | 0.1956 ± 0.0039 | 43, 55 | 49.9471 | 4 | ||||
| E11 | decyl acetate | 0.1208 ± 0.0254 | 43, 70 | 0.2903 | 416 | ||||
| E12 | ethyl acetate | 0.0277 ± 0.0322 | 43 | 0.0194 | >1000 | ||||
| E2 | butyl 3-methylbutanoate | 0.0519 ± 0.0261 | 56, 57, 85 | 0.1786 | 290 | ||||
| E3 | ethyl 3-hexenoate | 0.3146 ± 0.0596 | 29, 41, 69 | 103.7098 | 3 | ||||
| E4 | isopentyl 3-methylbutanoate | 0.3177 ± 0.0357 a | 0.2264 ± 0.0270 b | 43, 70, 85 | 0.02 | >1000 | >1000 | ||
| E5 | butyl pentanoate | 0.0064 ± 0.0016 | 56, 57, 85 | 25.4167 | <1 | ||||
| Test No. | Omitted Odorants | Difference in Odor | Number of Correct Answers a |
|---|---|---|---|
| 1 | sweet note compounds | less sweet | 9 ** |
| 1-1 | 1-penten-3-ol | less butter, less sweet | 5 * |
| 1-2 | 3-hydroxy-2-butanone | less creamy, less sweet | 9 ** |
| 1-3 | isopentyl 3-methylbutanoate | less sweet, less fruity | 8 ** |
| 1-4 | δ-dodecalactone | less fruity, less sweet | 7 ** |
| 1-5 | ethyl butyrate | less cheese, less sweet | 8 ** |
| 1-6 | butyl 3-methylbutanoate | less sweet, less apple-like | 5 * |
| 1-7 | hexyl methanoate | nd b | 2 |
| 1-8 | 3-methylbutyl octanoate | less sweet, less fruity | 9 ** |
| 1-9 | ethyl acetate | less sweet | 6 * |
| 1-10 | methyl benzyl alcohol | less floral, less sweet | 6 * |
| 1-11 | 1-dodecanol | nd b | 1 |
| 1-12 | dodecyl acetate | nd b | 1 |
| 1-13 | 3-octanone | less sweet, less herb | 9 ** |
| 2 | fatty note compounds | mildly less fatty | 6 * |
| 2-1 | dodecanoic acid | nd b | 2 |
| 2-2 | decanol | nd b | 3 |
| 2-3 | decyl acetate | less fatty | 5 * |
| 3 | cheese note compounds | decreased acidic | 4 * |
| 3-1 | octanoic acid | nd b | 1 |
| 3-2 | nonanoic acid | mildly less acidic | 4 * |
| 4 | mushroom note compounds | less mushroom-like | 9 ** |
| 4-1 | 3-octanol | less earthy | 5 * |
| 4-2 | 1-octen-3-ol | less mushroom | 7 ** |
| 4-3 | 2-octanol | less mushroom | 9 ** |
| 4-4 | octyl acetate | less mushroom | 6 * |
| 5 | floral note compounds | slightly less floral | 9 ** |
| 5-1 | terpineol | decreased floral | 5 * |
| 5-2 | 3,7-dimethyl-1-octanol | decreased floral | 9 ** |
| 6 | green note compounds | less green | 8 ** |
| 6-1 | decenone | nd b | 2 |
| 6-2 | 2-penten-1-ol | less green | 7 ** |
| 7 | fruity note compounds | less fruity, less sweet | 8 ** |
| 7-1 | ethyl 3-hexenoate | nd b | 1 |
| 7-2 | octanal | less citrus | 8 ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, W.; Sun, M.; Lu, H.; Wang, S.; Wang, R.; Shang, X.; Feng, T. Variations in Key Aroma Compounds and Aroma Profiles in Yellow and White Cultivars of Flammulina filiformis Based on Gas Chromatography–Mass Spectrometry–Olfactometry, Aroma Recombination, and Omission Experiments Coupled with Odor Threshold Concentrations. Foods 2024, 13, 684. https://doi.org/10.3390/foods13050684
Song W, Sun M, Lu H, Wang S, Wang R, Shang X, Feng T. Variations in Key Aroma Compounds and Aroma Profiles in Yellow and White Cultivars of Flammulina filiformis Based on Gas Chromatography–Mass Spectrometry–Olfactometry, Aroma Recombination, and Omission Experiments Coupled with Odor Threshold Concentrations. Foods. 2024; 13(5):684. https://doi.org/10.3390/foods13050684
Chicago/Turabian StyleSong, Wei, Min Sun, Huan Lu, Shengyou Wang, Ruijuan Wang, Xiaodong Shang, and Tao Feng. 2024. "Variations in Key Aroma Compounds and Aroma Profiles in Yellow and White Cultivars of Flammulina filiformis Based on Gas Chromatography–Mass Spectrometry–Olfactometry, Aroma Recombination, and Omission Experiments Coupled with Odor Threshold Concentrations" Foods 13, no. 5: 684. https://doi.org/10.3390/foods13050684
APA StyleSong, W., Sun, M., Lu, H., Wang, S., Wang, R., Shang, X., & Feng, T. (2024). Variations in Key Aroma Compounds and Aroma Profiles in Yellow and White Cultivars of Flammulina filiformis Based on Gas Chromatography–Mass Spectrometry–Olfactometry, Aroma Recombination, and Omission Experiments Coupled with Odor Threshold Concentrations. Foods, 13(5), 684. https://doi.org/10.3390/foods13050684







