Recent Advances Regarding Polyphenol Oxidase in Camellia sinensis: Extraction, Purification, Characterization, and Application
Abstract
1. Introduction
2. Extraction of PPO
2.1. Acetone Extraction
2.2. Buffer Extraction
2.3. Surfactant Extraction
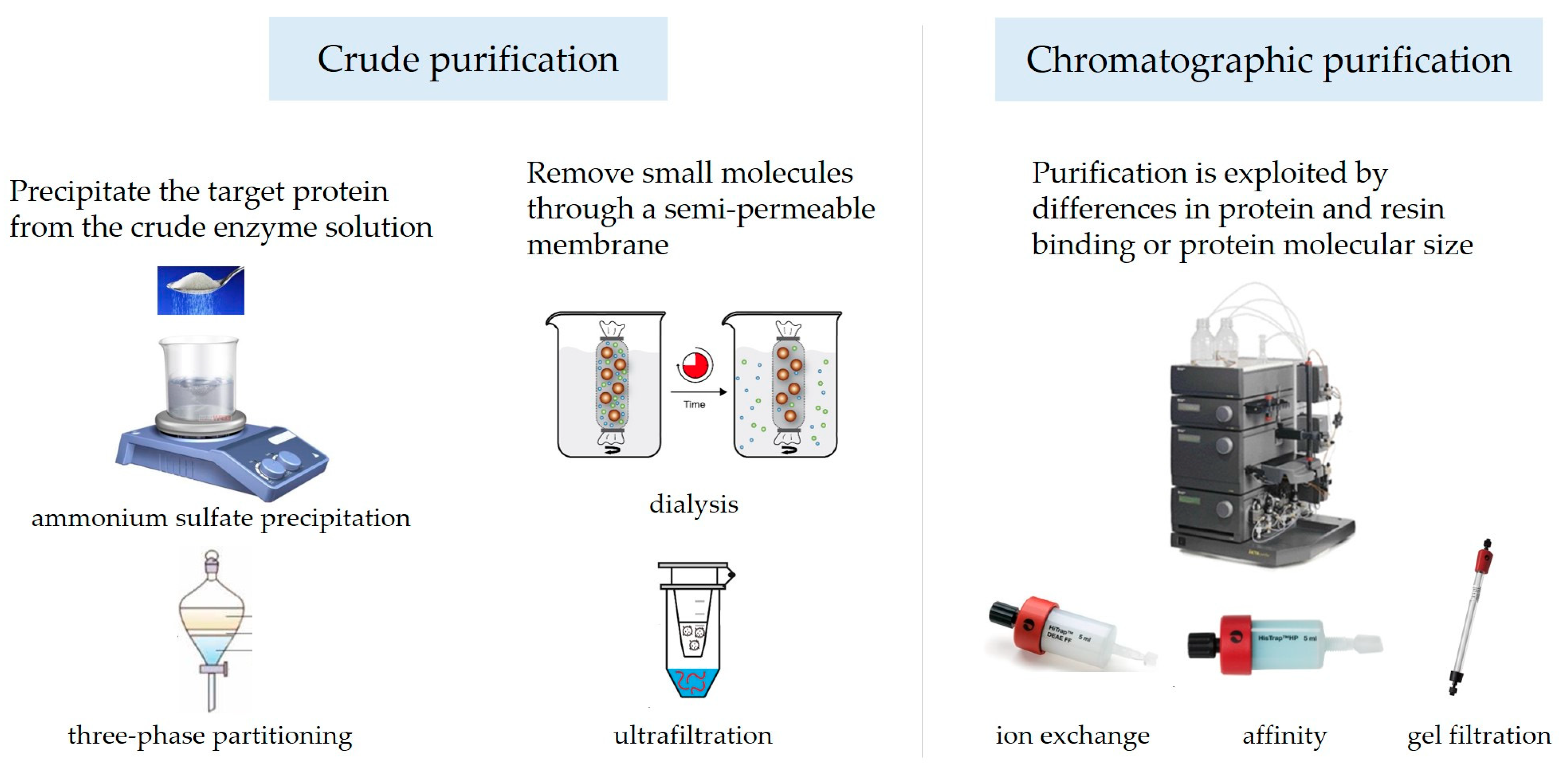
3. Purification of PPO
3.1. Crude Purification
3.1.1. Ammonium Sulfate Precipitation
3.1.2. Three-Phase Partitioning
3.1.3. Dialysis
3.1.4. Ultrafiltration
3.2. Chromatographic Purification
3.2.1. Ion Exchange Chromatography
3.2.2. Gel Filtration Chromatography
3.2.3. Affinity Chromatography
4. Characterizations of PPO
4.1. Optimal pH of PPO
4.2. Optimal Temperature of PPO
4.3. Molecular Weight of PPO
4.4. Substrate Specificity of PPO
4.5. Activators and Inhibitors of PPO
5. Application of PPO
5.1. The Role of PPO in Tea Processing
5.1.1. Black Tea
5.1.2. Dark Tea
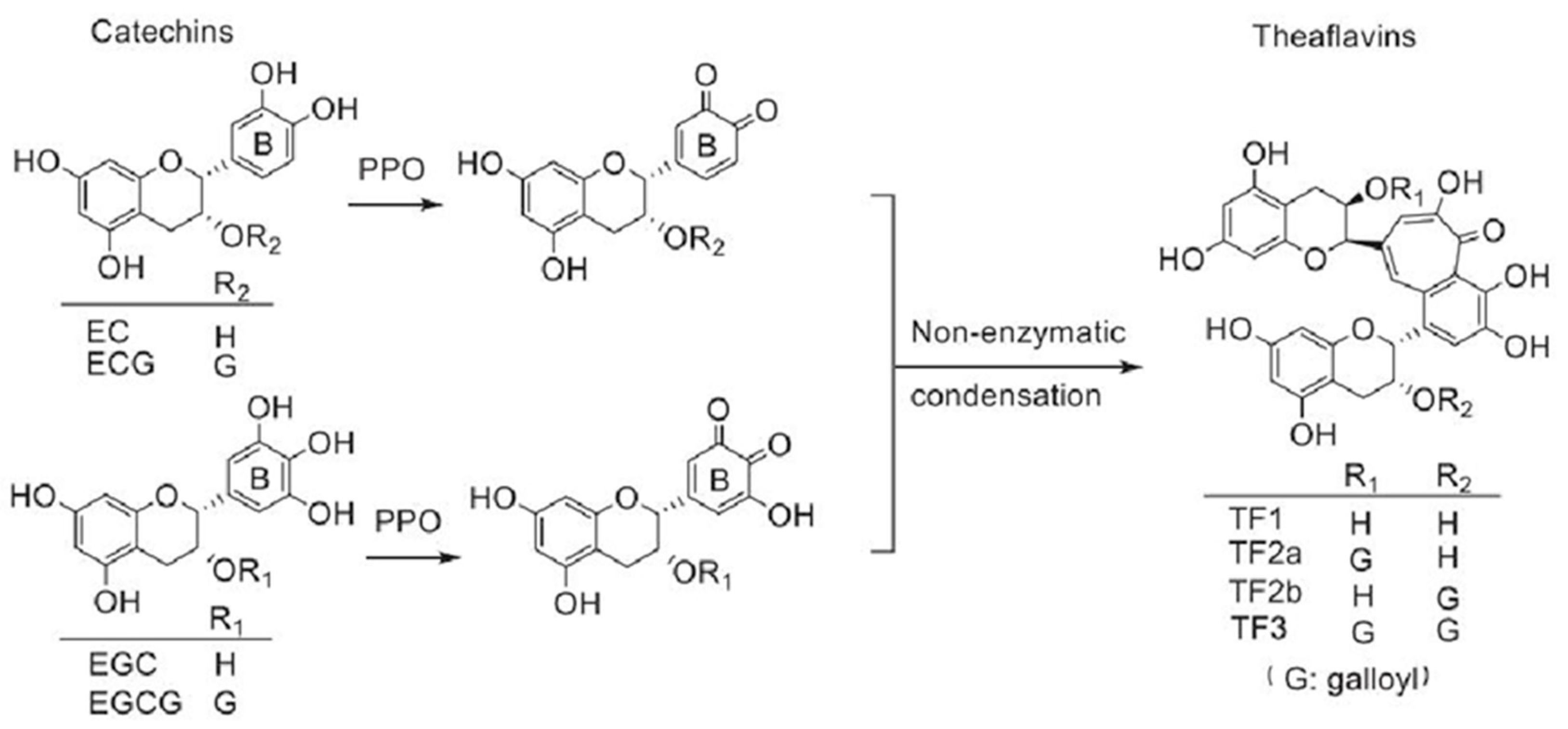
5.2. Synthesis of Theaflavins by PPO
6. Conclusions and Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Wei, X.M.; Shu, J.; Fahad, S.; Tao, K.L.; Zhang, J.W.; Chen, G.L.; Liang, Y.C.; Wang, M.Q.; Chen, S.Y.; Liao, J.G. Polyphenol oxidases regulate pollen development through modulating flavonoids homeostasis in tobacco. Plant Physiol. Bioch. 2023, 198, 107702. [Google Scholar] [CrossRef]
- Hong, Q.; Chen, Y.L.; Lin, D.Q.; Yang, R.Q.; Cao, K.Y.; Zhang, L.J.; Liu, Y.M.; Sun, L.C.; Cao, M.J. Expression of polyphenol oxidase of Litopenaeus vannamei and its characterization. Food Chem. 2024, 432, 137258. [Google Scholar] [CrossRef] [PubMed]
- Sarsenova, A.; Demir, D.; Caglayan, K.; Abiyev, S.; Darbayeva, T.; Eken, C. Purification and Properties of Polyphenol Oxidase of Dried Volvariella bombycina. Biology 2023, 12, 53. [Google Scholar] [CrossRef] [PubMed]
- Tinello, F.; Lante, A. Recent advances in controlling polyphenol oxidase activity of fruit and vegetable products. Innov. Food Sci. Emerg. 2018, 50, 73–83. [Google Scholar] [CrossRef]
- Mayer, A.M. Polyphenol oxidases in plants and fungi: Going places? A review. Phytochemistry 2006, 67, 2318–2331. [Google Scholar] [CrossRef] [PubMed]
- McLarin, M.A.; Leung, I.K.H. Substrate specificity of polyphenol oxidase. Crit. Rev. Biochem. Mol. 2020, 55, 274–308. [Google Scholar] [CrossRef]
- Li, J.F.; Deng, Z.Y.; Dong, H.H.; Tsao, R.; Liu, X.R. Substrate specificity of polyphenol oxidase and its selectivity towards polyphenols: Unlocking the browning mechanism of fresh lotus root (Nelumbo nucifera Gaertn.). Food Chem. 2023, 424, 136392. [Google Scholar] [CrossRef]
- Hua, J.J.; Xu, Q.; Yuan, H.B.; Wang, J.J.; Wu, Z.Q.; Li, X.T.; Jiang, Y.W. Effects of novel fermentation method on the biochemical components change and quality formation of Congou black tea. J. Food Compos. Anal. 2021, 96, 103751. [Google Scholar] [CrossRef]
- Zhang, G.Y.; Yang, J.H.; Cui, D.D.; Zhao, D.D.; Li, Y.Y.; Wan, X.C.; Zhao, J. Transcriptome and Metabolic Profiling Unveiled Roles of Peroxidases in Theaflavin Production in Black Tea Processing and Determination of Tea Processing Suitability. J. Agric. Food Chem. 2020, 68, 3528–3538. [Google Scholar] [CrossRef]
- Song, F.H.; Zheng, Y.; Li, R.Y.; Li, Z.F.; Liu, B.Y.; Wu, X. Intelligent control of green tea fixation with Microwave Processing. J. Food Eng. 2023, 349, 111481. [Google Scholar] [CrossRef]
- Zuo, H.; Si, X.Y.; Li, P.; Li, J.; Chen, Z.H.; Li, P.H.; Chen, C.S.; Liu, Z.H.; Zhao, J. Dynamic change of tea (Camellia sinensis) leaf cuticular wax in white tea processing for contribution to tea flavor formation. Food Res. Int. 2023, 163, 112182. [Google Scholar] [CrossRef]
- Feng, X.Y.; Yang, S.Y.; Pan, Y.N.; Zhou, S.; Ma, S.C.; Ou, C.S.; Fan, F.Y.; Gong, S.Y.; Chen, P.; Chu, Q. Yellow tea: More than turning green leaves to yellow. Crit. Rev. Food Sci. 2023, 3, 1–18. [Google Scholar] [CrossRef]
- He, C.; Zhou, J.T.; Li, Y.C.; Zhang, D.; Ntezimana, B.; Zhu, J.Y.; Wang, X.Y.; Xu, W.L.; Wen, X.J.; Chen, Y.Q.; et al. The aroma characteristics of oolong tea are jointly determined by processing mode and tea cultivars. Food Chem. X 2023, 18, 100730. [Google Scholar] [CrossRef]
- Kaur, A.; Farooq, S.; Sehgal, A. A Comparative Study of Antioxidant Potential and Phenolic Content in White (Silver Needle), Green and Black Tea. Curr. Nutr. Food Sci. 2019, 15, 415–420. [Google Scholar] [CrossRef]
- Liu, K.H.; Liu, W.T.; Ding, X.W.; Gao, X.; Lv, J.L.; Li, J.M. The key role of LeuRS in the development of the cleistothecium and the metabolization of the flavor during the fermentation of dark tea with Aspergillus montevidensis. LWT Food Sci. Technol. 2023, 185, 115188. [Google Scholar] [CrossRef]
- Long, P.P.; Rakariyatham, K.; Ho, C.T.; Zhang, L. Thearubigins: Formation, structure, health benefit and sensory property. Trends Food Sci. Technol. 2023, 133, 37–48. [Google Scholar] [CrossRef]
- Ramkumar, S.; Perumal, P.C.; Gandhi, P.S.; Mandal, A.K.A.; Padmanabhan, M.; Kumar, P.S.; Gopalakrishnan, V.K. Computational structure analysis of PPO enzyme from UPASI selected clone of Camellia sinensis (L.) O. Kuntze. J. Young Pharm. 2015, 7, 118–121. [Google Scholar] [CrossRef]
- Huang, X.X.; Ou, S.Q.; Li, Q.; Luo, Y.; Lin, H.Y.; Li, J.; Zhu, M.Z.; Wang, K.B. The R2R3 Transcription Factor CsMYB59 Regulates Polyphenol Oxidase Gene in Tea Plants (Camellia sinensis). Front. Plant Sci. 2021, 12, 739951. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Luo, Y.; Zheng, C.; Li, J.; Li, Q.; Lin, H.; Wang, K. The identification and analysis of polyphenol oxidase gene family in tea plant (Camellia sinensis). J. Tea Sci. 2018, 38, 385–395. [Google Scholar]
- Gregory, R.P.F.; Bendall, D.S.J.B.J. The purification and some properties of the polyphenol oxidase from tea (Camellia sinensis). Biochem. J. 1967, 101, 569–581. [Google Scholar] [CrossRef] [PubMed]
- Sui, X.; Meng, Z.; Dong, T.T.; Fan, X.T.; Wang, Q.G. Enzymatic browning and polyphenol oxidase control strategies. Curr. Opin. Biotech. 2023, 81, 102921. [Google Scholar] [CrossRef]
- Ravichandran, R.; Parthiban, R. Changes in enzyme activities (polyphenol oxidase and phenylalanine ammonia lyase) with type of tea leaf and during black tea manufacture and the effect of enzyme supplementation of dhool on black tea quality. Food Chem. 1998, 62, 277–281. [Google Scholar] [CrossRef]
- Ünal, M.Ü.; Yabacı, S.N.; Şener, A. Extraction, partial purification and characterisation of polyphenol oxidase from tea leaf (Camellia sinensis). Gida 2011, 36, 137–144. [Google Scholar]
- Takeo, T. Tea Leaf Polyphenol Oxidase: Part IV. The Localization of Polyphenol Oxidase in Tea Leaf Cell. Agric. Biol. Chem. 1966, 30, 931–934. [Google Scholar] [CrossRef]
- Xu, L.; Zhang, S.Q.; Xu, X.Y.; Chen, S.H.; Yao, Y.N.; Huang, Y.Y. Optimization of the extraction method of polyphenol oxidase from Camellia sinensis var. Longjing. J. Food Saf. Qual. 2015, 6, 1237–1242. [Google Scholar]
- Zhan, K.; Yang, Z.; Xu, Z.; Lai, Z.; Zhou, S.; Gan, Y.; Li, M.; Li, J.; Chen, L. Comparison of soluble and membrane-bound polyphenol oxidase from cultivars suitable to Ninghong tea production. J. Tea Sci. 2023, 43, 356–366. [Google Scholar]
- Liu, Y.; Chen, Q.; Liu, D.; Yang, L.; Hu, W.; Kuang, L.; Teng, J.; Liu, Y. Comparison of the biochemical properties and enzymatic synthesis of theaflavins by soluble and membrane-bound polyphenol oxidases from tea (Camellia sinensis) leaves. Food Sci. Technol. 2022, 42, 117321. [Google Scholar] [CrossRef]
- Öztürk, C.; Aksoy, M.; Küfrevioğlu, Ö.İ. Purification of tea leaf (Camellia sinensis) polyphenol oxidase by using affinity chromatography and investigation of its kinetic properties. J. Food Meas. Charact. 2020, 14, 31–38. [Google Scholar] [CrossRef]
- Villamil-Galindo, E.; Van de Velde, F.; Piagentini, A.M. Extracts from strawberry by-products rich in phenolic compounds reduce the activity of apple polyphenol oxidase. LWT Food Sci. Technol. 2020, 133, 110097. [Google Scholar] [CrossRef]
- Bravo, K.; Osorio, E. Characterization of polyphenol oxidase from Cape gooseberry (Physalis peruviana L.) fruit. Food Chem. 2016, 197, 185–190. [Google Scholar] [CrossRef]
- Huang, J.; Gao, X.; Su, L.; Liu, X.; Guo, L.; Zhang, Z.; Zhao, D.; Hao, J. Purification, characterization and inactivation kinetics of polyphenol oxidase extracted from Cistanche deserticola. Planta 2023, 257, 85. [Google Scholar] [CrossRef]
- Agunbiade, O.J.; Adewale, I.O. Studies on latent and soluble polyphenol oxidase from Moringa oleifera Lam. leaves. Biocatal. Agric. Biotechnol. 2022, 45, 102515. [Google Scholar] [CrossRef]
- Jia, S.; Jiang, S.; Chen, Y.; Wei, Y.; Shao, X. Comparison of Inhibitory Effects of Cinnamic Acid, β-Cyclodextrin, L-Cysteine, and Ascorbic Acid on Soluble and Membrane-Bound Polyphenol Oxidase in Peach Fruit. Foods 2022, 12, 167. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Pan, M.; Lu, Y.; Wang, M.; Huang, S.; Li, J.; Luo, K.; Luo, L.; Yao, M.; Hua, D. Purification and comparison of soluble and membrane-bound polyphenol oxidase from potato (Solanum tuberosum) tubers. Protein Expr. Purif. 2023, 202, 106195. [Google Scholar] [CrossRef]
- Zhou, H.; Bie, S.; Li, Z.; Zhou, L. Comparing the Effect of HPP on the Structure and Stability of Soluble and Membrane-Bound Polyphenol Oxidase from ‘Lijiang Snow’ Peach: Multispectroscopic and Molecular Dynamics Simulation. Foods 2023, 12, 1820. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.-Y.; Liu, F.; Li, M.; Wang, K.-L.; Ni, Y.-Y. Comparison of biochemical properties of membrane-bound and soluble polyphenol oxidase from Granny Smith apple (Malus × domestica Borkh.). Food Chem. 2019, 289, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Ioniţă, E.; Gurgu, L.; Aprodu, I.; Stănciuc, N.; Dalmadi, I.; Bahrim, G.; Râpeanu, G. Characterization, purification, and temperature/pressure stability of polyphenol oxidase extracted from plums (Prunus domestica). Process Biochem. 2017, 56, 177–185. [Google Scholar] [CrossRef]
- Alici, E.H.; Arabaci, G. Purification of polyphenol oxidase from borage (Trachystemon orientalis L.) by using three-phase partitioning and investigation of kinetic properties. Int. J. Biol. Macromol. 2016, 93, 1051–1056. [Google Scholar] [CrossRef] [PubMed]
- Adeseko, C.J.; Fatoki, T.H. Isolation and partial purification of polyphenol oxidase from seed of melon (Cucumeropsis edulis). Biointerface Res. Appl. Chem. 2021, 11, 9085–9096. [Google Scholar]
- Schmidt, J.M.; Greve-Poulsen, M.; Damgaard, H.; Hammershøj, M.; Larsen, L.B. Effect of membrane material on the separation of proteins and polyphenol oxidase in ultrafiltration of potato fruit juice. Food Bioprocess. Technol. 2016, 9, 822–829. [Google Scholar] [CrossRef]
- Çınar, F.; Aksay, S. Purification and characterization of polyphenol oxidase from myrtle berries (Myrtus communis L.). J. Food Meas. Charact. 2022, 16, 2282–2291. [Google Scholar] [CrossRef]
- Li, F. Purification, kinetic parameters, and isoforms of polyphenol oxidase from “Xushu 22” sweet potato skin. J. Food Biochem. 2020, 44, e13452. [Google Scholar] [CrossRef]
- Alishah, M.M.; Yıldız, S.; Bilen, Ç.; Karakuş, E. Purification and characterization of avocado (Persea americana) polyphenol oxidase by affinity chromatography. Prep. Biochem. Biotechnol. 2023, 53, 40–53. [Google Scholar] [CrossRef]
- Burgess, R.R. Protein precipitation techniques. Methods Enzymol. 2009, 463, 331–342. [Google Scholar] [PubMed]
- Zhang, S.Q.; Xu, L.; Chen, S.H.; Xu, X.Y.; Huang, Y.J.; Yao, Y.N.; Huang, Y.Y. Study on the Characterization of Polyphenol Oxidase Isoenzyme Isolated from Camellia sinensis cv. Longjing 43. Hubei Agric. Sci. 2016, 55, 1487–1491. [Google Scholar] [CrossRef]
- Xu, L.; Zhang, S.Q.; Chen, S.H.; Xu, X.Y.; Yao, Y.N.; Huang, Y.Y. Isolation and purification of polyphenol oxidase isoenzyme from Camellia sinensis var. J. Huazhong Agric. Univ. 2015, 34, 114–118. [Google Scholar]
- Yuzugullu Karakus, Y.; Kahveci, B.; Acemi, A.; Kocak, G. Application of three-phase partitioning to the purification and characterization of polyphenol oxidase from antioxidant rosemary (Rosmarinus officinalis L.). Int. J. Food Eng. 2020, 16, 20200118. [Google Scholar] [CrossRef]
- Saki, N.; Akin, M.; Alici, E.H.; Arabaci, G. Partial purification and characterization of polyphenol oxidase from the wild edible mushroom Lepiota procera using three-phase partitioning. Int. J. Food Eng. 2018, 14, 20170208. [Google Scholar] [CrossRef]
- Zawada, A.M.; Lang, T.; Ottillinger, B.; Kircelli, F.; Stauss-Grabo, M.; Kennedy, J.P. Impact of Hydrophilic Modification of Synthetic Dialysis Membranes on Hemocompatibility and Performance. Membranes 2022, 12, 932. [Google Scholar] [CrossRef]
- Krediet, R.T.; Barreto, D.L.; van Diepen, A.T.N. Assessment of the size selectivity of peritoneal permeability by the restriction coefficient to protein transport. Periton Dial. Int. 2022, 42, 335–343. [Google Scholar] [CrossRef]
- Teng, J.; Gong, Z.H.; Deng, Y.L.; Chen, L.; Li, Q.; Shao, Y.Y.; Lin, L.; Xiao, W.J. Purification, characterization and enzymatic synthesis of theaflavins of polyphenol oxidase isozymes from tea leaf (Camellia sinensis). LWT Food Sci. Technol. 2017, 84, 263–270. [Google Scholar] [CrossRef]
- Zhao, Y.; Tian, R.; Xu, Z.J.; Jiang, L.Z.; Sui, X.A. Recent advances in soy protein extraction technology. J. Am. Oil Chem. Soc. 2023, 100, 187–195. [Google Scholar] [CrossRef]
- Schuster, B.; Sleytr, U.B. S-Layer Ultrafiltration Membranes. Membranes 2021, 11, 275. [Google Scholar] [CrossRef] [PubMed]
- Haas, A.; Vaz, C.; Kempka, A.P. Extraction and Purification of Vegetable Peroxidase: A Review. Period. Tche Quim. 2019, 16, 692–703. [Google Scholar] [CrossRef]
- Sánchez-Trasviña, C.; Flores-Gatica, M.; Enriquez-Ochoa, D.; Rito-Palomares, M.; Mayolo-Deloisa, K. Purification of Modified Therapeutic Proteins Available on the Market: An Analysis of Chromatography-Based Strategies. Front. Bioeng. Biotech. 2021, 9, 717326. [Google Scholar] [CrossRef] [PubMed]
- Ke, L.J.; Xu, W.; Gao, J.N.; Gao, G.Z.; Wang, H.Q.; Zhou, J.W.; Liu, J.; Rao, P.F.; Xu, Y.Q. Isolation and characterization of thermo-tolerant polyphenol oxidases in a black tea infusion. Food Control 2021, 119, 107465. [Google Scholar] [CrossRef]
- Cai, H.L.; Zhong, Z.H.; Chen, Y.R.; Zhang, S.Y.; Ling, H.; Fu, H.W.; Zhang, L. Genes cloning, sequencing and function identification of recombinant polyphenol oxidase isozymes for production of monomeric theaflavins from Camellia sinensis. Int. J. Biol. Macromol. 2023, 240, 124353. [Google Scholar] [CrossRef]
- Cummins, P.M.; Rochfort, K.D.; O’Connor, B.F. Ion-Exchange Chromatography: Basic Principles and Application. Methods Mol. Biol. 2017, 1485, 209–223. [Google Scholar] [CrossRef]
- Agustoni, E.; Teixeira, R.D.; Huber, M.; Flister, S.; Hiller, S.; Schirmer, T. Acquisition of enzymatic progress curves in real time by quenching-free ion exchange chromatography. Anal. Biochem. 2022, 639, 114523. [Google Scholar] [CrossRef]
- Taddia, A.; Rito-Palomares, M.; Mayolo-Deloisa, K.; Tubio, G. Purification of xylanase from NRRL3 extract by an integrated strategy based on aqueous two-phase systems followed by ion exchange chromatography. Sep. Purif. Technol. 2021, 255, 117699. [Google Scholar] [CrossRef]
- Ma, Y.; Luo, M.; Xu, Y.P.; Liu, Y.J.; Liu, X.C.; Bi, X.F.; Yuan, Y.P.; Su, F.; Yin, X.C. Purification and characterization of a thaumatin-like protein-1 with polyphenol oxidase activity found in Prunus mume. RSC Adv. 2020, 10, 28746–28754. [Google Scholar] [CrossRef] [PubMed]
- Vishwasrao, C.; Chakraborty, S.; Ananthanarayan, L. Partial purification, characterisation and thermal inactivation kinetics of peroxidase and polyphenol oxidase isolated from Kalipatti sapota (Manilkara zapota). J. Sci. Food Agric. 2017, 97, 3568–3575. [Google Scholar] [CrossRef]
- Mogilnaya, O.; Ronzhin, N.; Posokhina, E.; Bondar, V. Extracellular Oxidase from the Fungus as a Promising Enzyme for Analytical Applications. Protein J. 2021, 40, 731–740. [Google Scholar] [CrossRef]
- Benaceur, F.; Gouzi, H.; Meddah, B.; Neifar, A.; Guergouri, A. Purification and characterization of catechol oxidase from Tadela (Phoenix dactylifera L.) date fruit. Int. J. Biol. Macromol. 2019, 125, 1248–1256. [Google Scholar] [CrossRef]
- Lin, H.B.; Ng, A.W.R.; Wong, C.W. Partial purification and characterization of polyphenol oxidase from Chinese parsley (Coriandrum sativum). Food Sci. Biotechnol. 2016, 25, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Sajjad, N.; Naqvi, S.M.S.; Asad, M.J.; Raja, N.I.; Pusztai-Carey, M.; Ahmad, M.S. Biochemical, Purification, Sequencing and Alignment Studies of the Novel Polyphenol Oxidase Isoforms from Musa acuminata Fruit Pulp. J. Anim. Plant Sci. 2021, 31, 542–555. [Google Scholar] [CrossRef]
- Popovic, G.; Kirby, N.C.; Dement, T.C.; Peterson, K.M.; Daub, C.E.; Belcher, H.A.; Guthold, M.; Offenbacher, A.R.; Hudson, N.E. Development of Transient Recombinant Expression and Affinity Chromatography Systems for Human Fibrinogen. Int. J. Mol. Sci. 2022, 23, 1054. [Google Scholar] [CrossRef] [PubMed]
- Altunkaya, A. Partial Purification and Characterization of Polyphenoloxidase from Turkish Tea Leaf (Camellia sinensis L.). Int. J. Food Prop. 2014, 17, 1490–1497. [Google Scholar] [CrossRef]
- Halder, J.; Tamuli, P.; Bhaduri, A.N. Isolation and characterization of polyphenol oxidase from Indian tea leaf (Camellia sinensis). J. Nutr. Biochem. 1998, 9, 75–80. [Google Scholar] [CrossRef]
- Zhang, S. Recent Advances of Polyphenol Oxidases in Plants. Molecules 2023, 28, 2158. [Google Scholar] [CrossRef]
- Siddiq, M.; Dolan, K.D. Characterization of polyphenol oxidase from blueberry (Vaccinium corymbosum L.). Food Chem. 2017, 218, 216–220. [Google Scholar] [CrossRef] [PubMed]
- Cheema, S.; Sommerhalter, M. Characterization of polyphenol oxidase activity in Ataulfo mango. Food Chem. 2015, 171, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Batista, K.A.; Batista, G.L.A.; Alves, G.L.; Fernandes, K.F. Extraction, partial purification and characterization of polyphenol oxidase from Solanum lycocarpum fruits. J. Mol. Catal. B Enzym. 2014, 102, 211–217. [Google Scholar] [CrossRef]
- Laad, S.; Premakshi, H.G.; Mirjankar, M.; Mulla, S.; Pujari, N.; Kamanavalli, C. Partial Purification, Characterization and Investigation of Inhibitory Effects of Organic Compounds on Cinnamomum verum Polyphenoloxidase Enzymes. Appl. Food Biotechnol. 2020, 7, 183–193. [Google Scholar] [CrossRef]
- Peng, X.Y.; Du, C.; Yu, H.Y.; Zhao, X.Y.; Zhang, X.Y.; Wang, X.Y. Purification and characterization of polyphenol oxidase (PPO) from water yam (Dioscorea alata). CyTA J. Food 2019, 17, 676–684. [Google Scholar] [CrossRef]
- Benaceur, F.; Chaibi, R.; Berrabah, F.; Neifar, A.; Leboukh, M.; Benaceur, K.; Nouioua, W.; Rezzoug, A.; Bouazzara, H.; Gouzi, H.; et al. Purification and characterization of latent polyphenol oxidase from truffles (Terfezia arenaria). Int. J. Biol. Macromol. 2020, 145, 885–893. [Google Scholar] [CrossRef]
- Zaini, N.A.M.; Osman, A.; Hamid, A.A.; Ebrahimpour, A.; Saari, N. Purification and characterization of membrane-bound polyphenoloxidase (mPPO) from Snake fruit [Salacca zalacca (Gaertn.) Voss]. Food Chem. 2013, 136, 407–414. [Google Scholar] [CrossRef]
- Liu, J.W.; Huang, Y.Y.; Ding, J.A.; Liu, C.; Xiao, X.D.; Ni, D.J. Prokaryotic expression and purification of Camellia sinensis polyphenol oxidase. J. Sci. Food Agric. 2010, 90, 2490–2494. [Google Scholar] [CrossRef]
- Wu, Y.L.; Pan, L.P.; Yu, S.L.; Li, H.H. Cloning, microbial expression and structure-activity relationship of polyphenol oxidases from Camellia sinensis. J. Biotechnol. 2010, 145, 66–72. [Google Scholar] [CrossRef]
- Liu, J.P.; Zhang, J.Y.; Liao, T.; Zhou, L.; Zou, L.Q.; Liu, Y.F.; Zhang, L.; Liu, W. Thermal Inactivation Kinetics of Kudzu (Pueraria lobata) Polyphenol Oxidase and the Influence of Food Constituents. Foods 2021, 10, 1320. [Google Scholar] [CrossRef]
- Liu, B.; Zhou, X.F.; Guan, H.Y.; Pang, X.Q.; Zhang, Z.Q. Purification and Characterization of a Dark Red Skin Related Dimeric Polyphenol Oxidase from Huaniu Apples. Foods 2022, 11, 1790. [Google Scholar] [CrossRef] [PubMed]
- Adeseko, C.J.; Sanni, D.M.; Salawu, S.O.; Kade, I.J.; Bamidele, S.O.; Lawal, O.T. Purification and biochemical characterization of polyphenol oxidase of African bush mango (Irvingia gabonensis) fruit peel. Biocatal. Agric. Biotech. 2021, 36, 102119. [Google Scholar] [CrossRef]
- Gorshkov, V.; Tarasova, N.; Gogoleva, N.; Osipova, E.; Petrova, O.; Kovtunov, E.; Gogolev, Y. Polyphenol oxidase from Pectobacterium atrosepticum: Identification and cloning of gene and characteristics of the enzyme. J. Basic. Microb. 2017, 57, 998–1009. [Google Scholar] [CrossRef] [PubMed]
- Kampatsikas, I.; Bijelic, A.; Rompel, A. Biochemical and structural characterization of tomato polyphenol oxidases provide novel insights into their substrate specificity. Sci. Rep. 2019, 9, 4022. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.R.; Jiang, X.F.; Li, C.; Qiu, L.; Chen, Y.Q.; Yu, Z.; Ni, D.J. Effect of Fermentation Humidity on Quality of Congou Black Tea. Foods 2023, 12, 1726. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.Y.; Liu, K.Y.; Yu, Q.; Shuo, X.; Li, D.Y.; Hua, G.Z.; Jun, X.W. Activity Changes of Polyphenol Oxidase, Peroxidase and β-Glycosidase in Black Tea Processing. J. Agric. 2014, 4, 96–99+113. [Google Scholar]
- Mahanta, P.K.; Boruah, S.K.; Boruah, H.K.; Kalita, J.N. Changes of polyphenol oxidase and peroxidase activities and pigment composition of some manufactured black teas (Camellia sinensis L.). J. Agric. Food Chem. 1993, 41, 272–276. [Google Scholar] [CrossRef]
- Chen, L.; Wang, H.J.; Ye, Y.; Wang, Y.F.; Xu, P. Structural insight into polyphenol oxidation during black tea fermentation. Food Chem. X 2023, 17, 100615. [Google Scholar] [CrossRef]
- Zhu, J.Y.; Wang, J.J.; Yuan, H.B.; Ouyang, W.; Li, J.; Hua, J.J.; Jiang, Y.W. Effects of Fermentation Temperature and Time on the Color Attributes and Tea Pigments of Yunnan Congou Black Tea. Foods 2022, 11, 1845. [Google Scholar] [CrossRef]
- Wang, H.J.; Shen, S.; Wang, J.J.; Jiang, Y.W.; Li, J.; Yang, Y.Q.; Hua, J.J.; Yuan, H.B. Novel insight into the effect of fermentation time on quality of Yunnan Congou black tea. LWT Food Sci. Technol. 2022, 155, 112939. [Google Scholar] [CrossRef]
- Samanta, T.; Cheeni, V.; Das, S.; Roy, A.B.; Ghosh, B.C.; Mitra, A. Assessing biochemical changes during standardization of fermentation time and temperature for manufacturing quality black tea. J. Food Sci. Technol. 2015, 52, 2387–2393. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.L.; Xie, J.L.; Deng, Y.L.; Jiang, Y.W.; Tong, H.R.; Yuan, H.B.; Yang, Y.Q. Volatile profile characterization during the drying process of black tea by integrated volatolomics analysis. LWT Food Sci. Technol. 2023, 184, 115039. [Google Scholar] [CrossRef]
- Jiang, Y.W.; Hua, J.J.; Wang, B.; Yuan, H.B.; Ma, H.L. Effects of Variety, Season, and Region on Theaflavins Content of Fermented Chinese Congou Black Tea. J. Food Qual. 2018, 2018, 5427302. [Google Scholar] [CrossRef]
- Das, S.; Datta, A.K. Mass transfer coefficient and mass diffusivity of O2 and CO2 during oxidation of macerated CTC and rolled orthodox leaves in black tea manufacturing. J. Food Process Eng. 2018, 41, 12875. [Google Scholar] [CrossRef]
- Hua, J.J.; Wang, H.J.; Yuan, H.B.; Yin, P.; Wang, J.J.; Guo, G.Y.; Jiang, Y.W. New insights into the effect of fermentation temperature and duration on catechins conversion and formation of tea pigments and theasinensins in black tea. J. Sci. Food Agric. 2022, 102, 2750–2760. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Liu, F.; Yang, Y.F.; Tu, Z.; Lin, J.Z.; Ye, Y.; Xu, P. Oxygen-enriched fermentation improves the taste of black tea by reducing the bitter and astringent metabolites. Food Res. Int. 2021, 148, 110613. [Google Scholar] [CrossRef]
- Ye, F.; Gao, S.W.; Gong, Z.M. Effects of Pyrus pyrifolia Nakai Polyphenol Oxidase Treatment on the Quality of Black Tea in Summer and Autumn. Food Sci. 2013, 34, 92–95. [Google Scholar]
- Chiang, S.H.; Yang, K.M.; Wang, S.Y.; Chen, C.W. Enzymatic treatment in black tea manufacturing processing: Impact on bioactive compounds, quality, and bioactivities of black tea. LWT Food Sci. Technol. 2022, 163, 113560. [Google Scholar] [CrossRef]
- Zhu, M.Z.; Li, N.; Zhou, F.; Ouyang, J.; Lu, D.M.; Xu, W.; Li, J.; Lin, H.Y.; Zhang, Z.; Xiao, J.B.; et al. Microbial bioconversion of the chemical components in dark tea. Food Chem. 2020, 312, 126043. [Google Scholar] [CrossRef]
- Hu, Z.Y.; Liu, S.C.; Xu, Z.G.; Liu, S.Q.; Li, T.T.; Yu, S.L.; Zhao, W.P. Comparison of and related species in dark tea at different aspects: Morphology, enzyme activity and mitochondrial genome. J. Food Process Pres. 2021, 45, 15903. [Google Scholar] [CrossRef]
- Liu, L.; Shi, J.J.; Yuan, Y.H.; Yue, T.L. Changes in the metabolite composition and enzyme activity of fermented tea during processing. Food Res. Int. 2022, 158, 111428. [Google Scholar] [CrossRef]
- Fu, Y.X.; Xun, L.T. The Effect of Polyphenol Oxidase on the Pile-fermentation Process and Quality of Pu-erh Tea. Mod. Food Sci. Technol. 2015, 31, 197–201. [Google Scholar] [CrossRef]
- He, H.F. Research progress on theaflavins: Efficacy, formation, and preparation. Food Nutr. Res. 2017, 61, 1344521. [Google Scholar] [CrossRef]
- Takemoto, M.; Takemoto, H. Synthesis of Theaflavins and Their Functions. Molecules 2018, 23, 918. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.Y.; Chen, Q.Y.; Luo, H.; Li, R.Y.; Chen, L.J.; Jiang, B.; Liang, Z.W.; Wang, T.; Ma, Y.; Zhao, M. An In Vitro Catalysis of Tea Polyphenols by Polyphenol Oxidase. Molecules 2023, 28, 1722. [Google Scholar] [CrossRef] [PubMed]
- Teng, J.; Liu, Y.; Zeng, W.; Zhou, M.Z.; Liu, Y.F.; Huang, Y.H.; Chen, Q.C. In vitro enzymatic synthesis of a monomeric theaflavin using a polyphenol oxidase isozyme from tea (Camellia sinensis) leaf. Int. J. Food Sci. Technol. 2022, 57, 5621–5631. [Google Scholar] [CrossRef]
- Singh, S.; Singh, D.; Kumar, S. Expression and biochemical analysis of codon-optimized polyphenol oxidase from Camellia sinensis (L.) O. Kuntze in E. coli. Process Biochem. 2017, 59, 180–186. [Google Scholar] [CrossRef]
- Liu, C.W.; Zhou, J.H.; Huang, J.A.; Xu, W.; Liu, Z.H. Study on the Synthesis of Theaflavin-3,3’-Digallate Catalyzed by Expressing Tea Tree Polyphenol Oxidase Isozymes and Its Enzymatic Solution. Fermentation 2023, 9, 770. [Google Scholar] [CrossRef]

| Method | Extract Solvent | Specific Enzyme Activity (U/mg) | Advantage | Disadvantage | References |
|---|---|---|---|---|---|
| Acetone extraction | Acetone | 24,789 | High enzyme activity, stable and easy to store | Low extraction rate of enzyme | [22,23] |
| Buffer extraction | Phosphate/citrate buffer | 192 | Easy operation, less impurity | Low enzyme activity and extraction rate | [24,25] |
| Surfactant extraction | Triton X-100 | 20,544 | High extraction rate of enzyme | Surfactant needs to be removed | [26,27,28] |
| Type of Chromatography | Chromatographic Matrix | Elution Buffer | Purification Fold | References |
|---|---|---|---|---|
| Anion exchange | DEAE-cellulose | A linear gradient of phosphate buffer (pH 6.8) concentration from 10 to 200 mM | 3.32 | [23] |
| Anion exchange | UNOsphere™ Q | A linear concentration gradient (0–1.0 M) of NaCl in 20 mM Tris-HCl (pH 9.0) | 11.8 | [56] |
| Gel filtration | Sephadex G-75 | 0.02 M Tris–HCl buffer (pH 7.5) containing 100 mL/L glycerol and 0.1 M NaCl | 48.94 | [51] |
| Affinity | Ni-NTA | Imidazole solution of 25–500 mM | Unknown | [57] |
| Affinity | Sepharose 4B-L-tyrosine-p-aminobenzoic acid | 0.1 M Tris–HCl buffer (pH 8.5) containing 1 M NaCl | 19.77 | [28] |
| Source | pH | Temperature (°C) | Molecular Weight (kDa) | References |
|---|---|---|---|---|
| Two PPO isozymes from Camellia sinensis var. Zhenghedabai | 5.5 and 6.0 | 33 and 38 | 85 and 42 | [51] |
| PPO from Camellia sinensis var. Lapsang souchong | 6.2 | 35 | 66 | [56] |
| PPO from Turkish tea leaves | 6.0 | 30 | 72 | [23] |
| PPO from Turkish tea leaves | 6.0 | 30 | Unknown | [68] |
| PPO from Indian tea leaves | 5.0 | Unknown | 72 | [69] |
| Two recombinant PPO isozymes from Huangjinya tea | 6.0 and 5.5 | 35 and 30 | 61.15 and 61.21 | [57] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zou, C.; Zhang, X.; Xu, Y.; Yin, J. Recent Advances Regarding Polyphenol Oxidase in Camellia sinensis: Extraction, Purification, Characterization, and Application. Foods 2024, 13, 545. https://doi.org/10.3390/foods13040545
Zou C, Zhang X, Xu Y, Yin J. Recent Advances Regarding Polyphenol Oxidase in Camellia sinensis: Extraction, Purification, Characterization, and Application. Foods. 2024; 13(4):545. https://doi.org/10.3390/foods13040545
Chicago/Turabian StyleZou, Chun, Xin Zhang, Yongquan Xu, and Junfeng Yin. 2024. "Recent Advances Regarding Polyphenol Oxidase in Camellia sinensis: Extraction, Purification, Characterization, and Application" Foods 13, no. 4: 545. https://doi.org/10.3390/foods13040545
APA StyleZou, C., Zhang, X., Xu, Y., & Yin, J. (2024). Recent Advances Regarding Polyphenol Oxidase in Camellia sinensis: Extraction, Purification, Characterization, and Application. Foods, 13(4), 545. https://doi.org/10.3390/foods13040545








