Plant-Based Meat Analogues in the Human Diet: What Are the Hazards?
Abstract
1. Introduction
2. Plant-Based Protein Sources
3. Extrusion Processing of Plant-Based Meat Analogues
4. Potential Hazards in Plant-Based Meat Analogues
5. Hazard Assessment
5.1. Nutritional Aspects
5.1.1. Protein Quality
5.1.2. Nutritional Value (H15) and Protein Metabolites (H16)
5.1.3. Anti-Nutritional Factors (H17)
| Parameter | Soy | Pea | SPC or SPI | PPC or PPI | Extruded Soy | LME Pea | HME Pea |
|---|---|---|---|---|---|---|---|
| Phytic acid/% | 1.32 | 0.54–1.2 | 1.1–2 | 0.46–1.6 | – | – | – |
| Trypsin inhibitor/TIU/g | 49,450 | 2100 | 110–2110 | 1300–2300 | 190–1480 | 378–1410 | 168 |
| Lectin/g/kg | 5.88 | 5.77 | <0.1 | 5.98–23.2 | ~0 | – | – |
| Tannin/% | 0.46 | 1.12 | – | 0.47–1.79 | – | – | – |
5.2. Allergens (H11)
5.3. Microbiological Aspects
Mycotoxins (H14)
5.4. Chemical Aspects
Process-Related by Products (H4–H7)
6. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- De Smet, S.; Vossen, E. Meat: The Balance between Nutrition and Health. A Review. Meat Sci. 2016, 120, 145–156. [Google Scholar] [CrossRef]
- European Commission. EU Agricultural Outlook for Markets, Income and Environment 2021–2031; Publications Office of the European Union: Brussels, Belgium, 2021; pp. 1–83. [Google Scholar] [CrossRef]
- Bohrer, B.M. An Investigation of the Formulation and Nutritional Composition of Modern Meat Analogue Products. Food Sci. Hum. Wellness 2019, 8, 320–329. [Google Scholar] [CrossRef]
- Caporgno, M.P.; Böcker, L.; Müssner, C.; Stirnemann, E.; Haberkorn, I.; Adelmann, H.; Handschin, S.; Windhab, E.J.; Mathys, A. Extruded Meat Analogues Based on Yellow, Heterotrophically Cultivated Auxenochlorella Protothecoides Microalgae. Innov. Food Sci. Emerg. Technol. 2020, 59, 102275. [Google Scholar] [CrossRef]
- Palanisamy, M.; Töpfl, S.; Berger, R.G.; Hertel, C. Physico-Chemical and Nutritional Properties of Meat Analogues Based on Spirulina/Lupin Protein Mixtures. Eur. Food Res. Technol. 2019, 245, 1889–1898. [Google Scholar] [CrossRef]
- Smetana, S.; Pernutz, C.; Toepfl, S.; Heinz, V.; van Campenhout, L. High-Moisture Extrusion with Insect and Soy Protein Concentrates: Cutting Properties of Meat Analogues under Insect Content and Barrel Temperature Variations. J. Insects Food Feed. 2019, 5, 29–34. [Google Scholar] [CrossRef]
- Stephan, A.; Ahlborn, J.; Zajul, M.; Zorn, H. Edible Mushroom Mycelia of Pleurotus Sapidus as Novel Protein Sources in a Vegan Boiled Sausage Analog System: Functionality and Sensory Tests in Comparison to Commercial Proteins and Meat Sausages. Eur. Food Res. Technol. 2018, 244, 913–924. [Google Scholar] [CrossRef]
- Bonny, S.P.F.; Gardner, G.E.; Pethick, D.W.; Hocquette, J.-F. What Is Artificial Meat and What Does It Mean for the Future of the Meat Industry? J. Integr. Agric. 2015, 14, 255–263. [Google Scholar] [CrossRef]
- Lee, H.J.; Yong, H.I.; Kim, M.; Choi, Y.-S.; Jo, C. Status of Meat Alternatives and Their Potential Role in the Future Meat Market—A Review. Asian-Australas. J. Anim. Sci. 2020, 33, 1533–1543. [Google Scholar] [CrossRef]
- Banach, J.L.; van der Berg, J.P.; Kleter, G.; van Bokhorst-van de Veen, H.; Bastiaan-Net, S.; Pouvreau, L.; van Asselt, E.D. Alternative Proteins for Meat and Dairy Replacers: Food Safety and Future Trends. Crit. Rev. Food Sci. Nutr. 2023, 63, 11063–11080. [Google Scholar] [CrossRef]
- Curtain, F.; Grafenauer, S. Plant-Based Meat Substitutes in the Flexitarian Age: An Audit of Products on Supermarket Shelves. Nutrients 2019, 11, 2603. [Google Scholar] [CrossRef]
- Smetana, S.; Profeta, A.; Voigt, R.; Kircher, C.; Heinz, V. Meat Substitution in Burgers: Nutritional Scoring, Sensorial Testing, and Life Cycle Assessment. Future Foods 2021, 4, 100042. [Google Scholar] [CrossRef]
- Boye, J.; Zare, F.; Pletch, A. Pulse Proteins: Processing, Characterization, Functional Properties and Applications in Food and Feed. Food Res. Int. 2010, 43, 414–431. [Google Scholar] [CrossRef]
- Nadathur, S.R.; Wanasundara, J.P.D.; Scanlin, L. (Eds.) Sustainable Protein Sources; Elsevier: Amsterdam, The Netherlands, 2017; ISBN 978-0-12-802776-9. [Google Scholar]
- Kumar, M.; Tomar, M.; Potkule, J.; Verma, R.; Punia, S.; Mahapatra, A.; Belwal, T.; Dahuja, A.; Joshi, S.; Berwal, M.K.; et al. Advances in the Plant Protein Extraction: Mechanism and Recommendations. Food Hydrocoll. 2021, 115, 106595. [Google Scholar] [CrossRef]
- Arif, M.; Pauls, K.P. Properties of Plant Proteins. In Plant Bioproducts; Springer: New York, NY, USA, 2018; pp. 121–142. [Google Scholar]
- Bouvier, J.; Campanella, O.H. Extrusion Processing Technology; Wiley: Hoboken, NJ, USA, 2014; ISBN 9781444338119. [Google Scholar]
- Arêas, J.A.G. Extrusion of Food Proteins. Crit. Rev. Food Sci. Nutr. 1992, 32, 365–392. [Google Scholar] [CrossRef]
- Kinsella, J.E.; Franzen, K.L. Texturized Proteins: Fabrication, Flavoring, and Nutrition. CRC Crit. Rev. Food Sci. Nutr. 1978, 10, 147–207. [Google Scholar] [CrossRef]
- Pommet, M.; Morel, M.-H.; Redl, A.; Guilbert, S. Aggregation and Degradation of Plasticized Wheat Gluten during Thermo-Mechanical Treatments, as Monitored by Rheological and Biochemical Changes. Polymer 2004, 45, 6853–6860. [Google Scholar] [CrossRef]
- Redl, A.; Guilbert, S.; Morel, M.-H. Heat and Shear Mediated Polymerisation of Plasticized Wheat Gluten Protein upon Mixing. J. Cereal Sci. 2003, 38, 105–114. [Google Scholar] [CrossRef]
- Redl, A.; Morel, M.H.; Bonicel, J.; Vergnes, B.; Guilbert, S. Extrusion of Wheat Gluten Plasticized with Glycerol: Influence of Process Conditions on Flow Behavior, Rheological Properties, and Molecular Size Distribution. Cereal Chem. J. 1999, 76, 361–370. [Google Scholar] [CrossRef]
- Harper, J.M. Extrusion Texturization of Foods. Food Technol. 1986, 40, 70–76. [Google Scholar]
- Kinsella, J.E. Functional Properties of Soy Proteins. J. Am. Oil Chem. Soc. 1979, 56, 242–258. [Google Scholar] [CrossRef]
- Akdogan, H. High Moisture Food Extrusion. Int. J. Food Sci. Technol. 1999, 34, 195–207. [Google Scholar] [CrossRef]
- Camire, M.E. Protein Functionality Modification by Extrusion Cooking. J. Am. Oil Chem. Soc. 1991, 68, 200–205. [Google Scholar] [CrossRef]
- Cheftel, J.C.; Kitagawa, M.; Quéguiner, C. New Protein Texturization Processes by Extrusion Cooking at High Moisture Levels. Food Rev. Int. 1992, 8, 235–275. [Google Scholar] [CrossRef]
- Chen, F.L.; Wei, Y.M.; Zhang, B. Chemical Cross-Linking and Molecular Aggregation of Soybean Protein during Extrusion Cooking at Low and High Moisture Content. LWT—Food Sci. Technol. 2011, 44, 957–962. [Google Scholar] [CrossRef]
- Dekkers, B.L.; Boom, R.M.; van der Goot, A.J. Structuring Processes for Meat Analogues. Trends Food Sci. Technol. 2018, 81, 25–36. [Google Scholar] [CrossRef]
- van der Sman, R.G.M.; van der Goot, A.J. Hypotheses Concerning Structuring of Extruded Meat Analogs. Curr. Res. Food Sci. 2023, 6, 100510. [Google Scholar] [CrossRef]
- Kyriakopoulou, K.; Keppler, J.K.; van der Goot, A.J. Functionality of Ingredients and Additives in Plant-Based Meat Analogues. Foods 2021, 10, 600. [Google Scholar] [CrossRef]
- Cantwell, M.; Elliott, C. Nitrates, Nitrites and Nitrosamines from Processed Meat Intake and Colorectal Cancer Risk. J. Clin. Nutr. Diet. 2017, 3, 4–27. [Google Scholar] [CrossRef]
- Herrmann, S.S.; Duedahl-Olesen, L.; Granby, K. Occurrence of Volatile and Non-Volatile N-Nitrosamines in Processed Meat Products and the Role of Heat Treatment. Food Control 2015, 48, 163–169. [Google Scholar] [CrossRef]
- Engel, E.; Ratel, J.; Bouhlel, J.; Planche, C.; Meurillon, M. Novel Approaches to Improving the Chemical Safety of the Meat Chain towards Toxicants. Meat Sci. 2015, 109, 75–85. [Google Scholar] [CrossRef]
- Andrée, S.; Jira, W.; Schwind, K.-H.; Wagner, H.; Schwägele, F. Chemical Safety of Meat and Meat Products. Meat Sci. 2010, 86, 38–48. [Google Scholar] [CrossRef]
- Felton, J.S.; Fultz, E.; Dolbeare, F.A.; Knize, M.G. Effect of Microwave Pretreatment on Heterocyclic Aromatic Amine Mutagens/Carcinogens in Fried Beef Patties. Food Chem. Toxicol. 1994, 32, 897–903. [Google Scholar] [CrossRef]
- Ni, W.; McNaughton, L.; LeMaster, D.M.; Sinha, R.; Turesky, R.J. Quantitation of 13 Heterocyclic Aromatic Amines in Cooked Beef, Pork, and Chicken by Liquid Chromatography−Electrospray Ionization/Tandem Mass Spectrometry. J. Agric. Food Chem. 2008, 56, 68–78. [Google Scholar] [CrossRef]
- He, J.; Evans, N.M.; Liu, H.; Shao, S. A Review of Research on Plant-Based Meat Alternatives: Driving Forces, History, Manufacturing, and Consumer Attitudes. Compr. Rev. Food Sci. Food Saf. 2020, 19, 2639–2656. [Google Scholar] [CrossRef]
- Xie, Y.; Cai, L.; Zhao, D.; Liu, H.; Xu, X.; Zhou, G.; Li, C. Real Meat and Plant-Based Meat Analogues Have Different in Vitro Protein Digestibility Properties. Food Chem. 2022, 387, 132917. [Google Scholar] [CrossRef]
- Xie, Y.; Wang, C.; Zhao, D.; Zhou, G.; Li, C. Processing Method Altered Mouse Intestinal Morphology and Microbial Composition by Affecting Digestion of Meat Proteins. Front. Microbiol. 2020, 11, 511. [Google Scholar] [CrossRef]
- Joye, I. Protein Digestibility of Cereal Products. Foods 2019, 8, 199. [Google Scholar] [CrossRef]
- Dupont, D. Peptidomic as a Tool for Assessing Protein Digestion. Curr. Opin. Food Sci. 2017, 16, 53–58. [Google Scholar] [CrossRef]
- Shi, L.; Arntfield, S.D.; Nickerson, M. Changes in Levels of Phytic Acid, Lectins and Oxalates during Soaking and Cooking of Canadian Pulses. Food Res. Int. 2018, 107, 660–668. [Google Scholar] [CrossRef]
- Ma, Z.; Boye, J.I.; Hu, X. In Vitro Digestibility, Protein Composition and Techno-Functional Properties of Saskatchewan Grown Yellow Field Peas (Pisum sativum L.) as Affected by Processing. Food Res. Int. 2017, 92, 64–78. [Google Scholar] [CrossRef]
- Herreman, L.; Nommensen, P.; Pennings, B.; Laus, M.C. Comprehensive Overview of the Quality of Plant- and Animal-sourced Proteins Based on the Digestible Indispensable Amino Acid Score. Food Sci. Nutr. 2020, 8, 5379–5391. [Google Scholar] [CrossRef] [PubMed]
- Schlemmer, U.; Frølich, W.; Prieto, R.M.; Grases, F. Phytate in Foods and Significance for Humans: Food Sources, Intake, Processing, Bioavailability, Protective Role and Analysis. Mol. Nutr. Food Res. 2009, 53, S330–S375. [Google Scholar] [CrossRef] [PubMed]
- Savelkoul, F.H.M.G.; van der Poel, A.F.B.; Tamminga, S. The Presence and Inactivation of Trypsin Inhibitors, Tannins, Lectins and Amylase Inhibitors in Legume Seeds during Germination. A Review. Plant Foods Hum. Nutr. 1992, 42, 71–85. [Google Scholar] [CrossRef] [PubMed]
- Champ, M.M.-J. Non-Nutrient Bioactive Substances of Pulses. Br. J. Nutr. 2002, 88, 307–319. [Google Scholar] [CrossRef] [PubMed]
- Nadathur, S.R.; Wanasundara, J.P.D.; Scanlin, L. Proteins in the Diet: Challenges in Feeding the Global Population. In Sustainable Protein Sources; Elsevier: Amsterdam, The Netherlands, 2017; pp. 1–19. [Google Scholar]
- USDA. Isoflavone Content of Selected Foods; USDA: Washington, DC, USA, 2008. [Google Scholar]
- DiSilvestro, R.A.; Mattern, C.; Wood, N.; Devor, S.T. Soy Protein Intake by Active Young Adult Men Raises Plasma Antioxidant Capacity without Altering Plasma Testosterone. Nutr. Res. 2006, 26, 92–95. [Google Scholar] [CrossRef]
- Bredsdorff, L.; Fagt, S.; Boberg, J.; Pilegaard, K.; Widenfalk, A.; Steffensen, I.-L. Soy Intake and Possible Adverse Health Effects in Nordic Children and Pregnant Women (Unborn Children); Nordic Council of Ministers: Copenhagen, Denmark, 2020; ISBN 9789289367059. [Google Scholar]
- Thrane, M.; Paulsen, P.V.; Orcutt, M.W.; Krieger, T.M. Soy Protein. In Sustainable Protein Sources; Elsevier: Amsterdam, The Netherlands, 2017; pp. 23–45. [Google Scholar]
- Messina, M. A Brief Historical Overview of the Past Two Decades of Soy and Isoflavone Research. J. Nutr. 2010, 140, 1350S–1354S. [Google Scholar] [CrossRef] [PubMed]
- Reinkensmeier, A.; Bußler, S.; Schlüter, O.; Rohn, S.; Rawel, H.M. Characterization of Individual Proteins in Pea Protein Isolates and Air Classified Samples. Food Res. Int. 2015, 76, 160–167. [Google Scholar] [CrossRef]
- Rivera del Rio, A.; Möller, A.C.; Boom, R.M.; Janssen, A.E.M. In Vitro Gastro-Small Intestinal Digestion of Conventional and Mildly Processed Pea Protein Ingredients. Food Chem. 2022, 387, 132894. [Google Scholar] [CrossRef] [PubMed]
- Avilés-Gaxiola, S.; Chuck-Hernández, C.; Serna Saldívar, S.O. Inactivation Methods of Trypsin Inhibitor in Legumes: A Review. J. Food Sci. 2018, 83, 17–29. [Google Scholar] [CrossRef]
- Wang, N.; Maximiuk, L. Effect of Air Classification Processing Variables on Yield, Composition, and Certain Antinutrients of Air-classified Fractions from Field Peas by Response Surface Methodology. J. Food Process Preserv. 2019, 43, e13999. [Google Scholar] [CrossRef]
- Morel, P.C.H.; Melai, J.; Eady, S.L.; Coles, G.D. Effect of Non-Starch Polysaccharides and Resistant Starch on Mucin Secretion and Endogenous Amino Acid Losses in Pigs. Asian-Australas. J. Anim. Sci. 2005, 18, 1634–1641. [Google Scholar] [CrossRef]
- Opazo-Navarrete, M.; Tagle Freire, D.; Boom, R.M.; Janssen, A.E.M. The Influence of Starch and Fibre on In Vitro Protein Digestibility of Dry Fractionated Quinoa Seed (Riobamba Variety). Food Biophys. 2019, 14, 49–59. [Google Scholar] [CrossRef]
- FAO/WHO/UNU. Energy and Protein Requirements. Report of a Joint FAO/WHO/UNU Expert Consultation. World Health Organ. Tech. Rep. Ser. 1985, 724, 1–206. [Google Scholar]
- FAO/WHO. Protein Quality Evaluation. Report of the Joint FAO/WHO Expert Consultation. FAO Food Nutr. Pap. 1991, 51, 1–66. [Google Scholar]
- FAO. Dietary Protein Quality Evaluation in Human Nutrition; FAO: Rome, Italy, 2011. [Google Scholar]
- Hughes, G.J.; Ryan, D.J.; Mukherjea, R.; Schasteen, C.S. Protein Digestibility-Corrected Amino Acid Scores (PDCAAS) for Soy Protein Isolates and Concentrate: Criteria for Evaluation. J. Agric. Food Chem. 2011, 59, 12707–12712. [Google Scholar] [CrossRef] [PubMed]
- Santos-Hernández, M.; Alfieri, F.; Gallo, V.; Miralles, B.; Masi, P.; Romano, A.; Ferranti, P.; Recio, I. Compared Digestibility of Plant Protein Isolates by Using the INFOGEST Digestion Protocol. Food Res. Int. 2020, 137, 109708. [Google Scholar] [CrossRef]
- Faris, R.J.; Wang, H.; Wang, T. Improving Digestibility of Soy Flour by Reducing Disulfide Bonds with Thioredoxin. J. Agric. Food Chem. 2008, 56, 7146–7150. [Google Scholar] [CrossRef]
- Bühler, J.M.; Dekkers, B.L.; Bruins, M.E.; van der Goot, A.J. Modifying Faba Bean Protein Concentrate Using Dry Heat to Increase Water Holding Capacity. Foods 2020, 9, 1077. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Martin, B.R.; Weaver, C.M. Calcium Bioavailability of Calcium Carbonate Fortified Soymilk Is Equivalent to Cow’s Milk in Young Women. J. Nutr. 2005, 135, 2379–2382. [Google Scholar] [CrossRef]
- Weaver, C.M.; Heaney, R.P.; Connor, L.; Martin, B.R.; Smith, D.L.; Nielsen, S. Bioavailability of Calcium from Tofu as Compared with Milk in Premenopausal Women. J. Food Sci. 2002, 67, 3144–3147. [Google Scholar] [CrossRef]
- van den Berg, L.A.; Mes, J.J.; Mensink, M.; Wanders, A.J. Protein Quality of Soy and the Effect of Processing: A Quantitative Review. Front. Nutr. 2022, 9, 1004754. [Google Scholar] [CrossRef]
- Nosworthy, M.G.; Franczyk, A.J.; Medina, G.; Neufeld, J.; Appah, P.; Utioh, A.; Frohlich, P.; House, J.D. Effect of Processing on the In-Vitro and in-Vivo Protein Quality of Yellow and Green Split Peas (Pisum sativum). J. Agric. Food Chem. 2017, 65, 7790–7796. [Google Scholar] [CrossRef]
- Mathai, J.K.; Liu, Y.; Stein, H.H. Values for Digestible Indispensable Amino Acid Scores (DIAAS) for Some Dairy and Plant Proteins May Better Describe Protein Quality than Values Calculated Using the Concept for Protein Digestibility-Corrected Amino Acid Scores (PDCAAS). Br. J. Nutr. 2017, 117, 490–499. [Google Scholar] [CrossRef]
- Zhou, H.; Hu, Y.; Tan, Y.; Zhang, Z.; McClements, D.J. Digestibility and Gastrointestinal Fate of Meat versus Plant-Based Meat Analogs: An in Vitro Comparison. Food Chem. 2021, 364, 130439. [Google Scholar] [CrossRef] [PubMed]
- Ketnawa, S.; Ogawa, Y. In Vitro Protein Digestibility and Biochemical Characteristics of Soaked, Boiled and Fermented Soybeans. Sci. Rep. 2021, 11, 14257. [Google Scholar] [CrossRef]
- Fanelli, N.S.; Bailey, H.M.; Thompson, T.W.; Delmore, R.; Nair, M.N.; Stein, H.H. Digestible Indispensable Amino Acid Score (DIAAS) Is Greater in Animal-Based Burgers than in Plant-Based Burgers If Determined in Pigs. Eur. J. Nutr. 2022, 61, 461–475. [Google Scholar] [CrossRef]
- Nosworthy, M.G.; Hernandez-Alvarez, A.J.; Franczyk, A.J.; Medina, G.; Neufeld, J.; Arcand, Y.; Ribéreau, S.; Sánchez-Velázquez, O.A.; House, J.D. Effect of Cooking on the in Vitro and in Vivo Protein Quality of Soy, Oat and Wheat Varieties. Cereal Chem. 2022, 100, 460–472. [Google Scholar] [CrossRef]
- Tulbek, M.C.; Lam, R.S.H.; Wang, Y.C.; Asavajaru, P.; Lam, A. Pea. In Sustainable Protein Sources; Elsevier: Amsterdam, The Netherlands, 2017; pp. 145–164. [Google Scholar]
- Hodgkinson, S.M.; Montoya, C.A.; Scholten, P.T.; Rutherfurd, S.M.; Moughan, P.J. Cooking Conditions Affect the True Ileal Digestible Amino Acid Content and Digestible Indispensable Amino Acid Score (DIAAS) of Bovine Meat as Determined in Pigs. J. Nutr. 2018, 148, 1564–1569. [Google Scholar] [CrossRef]
- de Marchi, M.; Costa, A.; Pozza, M.; Goi, A.; Manuelian, C.L. Detailed Characterization of Plant-Based Burgers. Sci. Rep. 2021, 11, 2049. [Google Scholar] [CrossRef]
- van Vliet, S.; Bain, J.R.; Muehlbauer, M.J.; Provenza, F.D.; Kronberg, S.L.; Pieper, C.F.; Huffman, K.M. A Metabolomics Comparison of Plant-Based Meat and Grass-Fed Meat Indicates Large Nutritional Differences despite Comparable Nutrition Facts Panels. Sci. Rep. 2021, 11, 13828. [Google Scholar] [CrossRef]
- Tallima, H.; el Ridi, R. Arachidonic Acid: Physiological Roles and Potential Health Benefits—A Review. J. Adv. Res. 2018, 11, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Wu, G. Important Roles of Dietary Taurine, Creatine, Carnosine, Anserine and 4-Hydroxyproline in Human Nutrition and Health. Amino Acids 2020, 52, 329–360. [Google Scholar] [CrossRef] [PubMed]
- Madeo, F.; Eisenberg, T.; Pietrocola, F.; Kroemer, G. Spermidine in Health and Disease. Science 2018, 359, eaan2788. [Google Scholar] [CrossRef] [PubMed]
- Márquez Campos, E.; Stehle, P.; Simon, M.C. Microbial Metabolites of Flavan-3-Ols and Their Biological Activity. Nutrients 2019, 11, 2260. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.-Z.; Yang, S.; Wu, G. Free Radicals, Antioxidants, and Nutrition. Nutrition 2002, 18, 872–879. [Google Scholar] [CrossRef] [PubMed]
- Othman, R.A.; Moghadasian, M.H. Beyond Cholesterol-Lowering Effects of Plant Sterols: Clinical and Experimental Evidence of Anti-Inflammatory Properties. Nutr. Rev. 2011, 69, 371–382. [Google Scholar] [CrossRef] [PubMed]
- Toribio-Mateas, M.A.; Bester, A.; Klimenko, N. Impact of Plant-Based Meat Alternatives on the Gut Microbiota of Consumers: A Real-World Study. Foods 2021, 10, 2040. [Google Scholar] [CrossRef]
- Shevlyakov, A.; Nikogosov, D.; Stewart, L.-A.; Toribio-Mateas, M. Reference Values for Intake of Six Types of Soluble and Insoluble Fibre in Healthy UK Inhabitants Based on the UK Biobank Data. Public. Health Nutr. 2022, 25, 1321–1335. [Google Scholar] [CrossRef] [PubMed]
- Westhoek, H.; Lesschen, J.P.; Leip, A.; Rood, G.; Wagner, S.; Murphy-Bokern, D.; Pallière, C.; Howard, C.; Oenema, O.; Sutton, O. Nitrogen on the Table: The Influence of Food Choices on Nitrogen Emissions and the Environment; NERC/Centre for Ecology & Hydrology: Wallingford, UK, 2016. [Google Scholar]
- FAO. Daily Protein Supply from Plant-Based Foods, World, 1962 to 2021; FAO: Rome, Italy, 2024. [Google Scholar]
- Arntfield, S.D.; Maskus, H.D. Peas and Other Legume Proteins. In Handbook of Food Proteins; Elsevier: Amsterdam, The Netherlands, 2011; pp. 233–266. [Google Scholar]
- Kumar, V.; Sinha, A.K.; Makkar, H.P.S.; Becker, K. Dietary Roles of Phytate and Phytase in Human Nutrition: A Review. Food Chem. 2010, 120, 945–959. [Google Scholar] [CrossRef]
- Mohan, V.R.; Tresina, P.S.; Daffodil, E.D. Antinutritional Factors in Legume Seeds: Characteristics and Determination. In Encyclopedia of Food and Health; Elsevier: Amsterdam, The Netherlands, 2016; pp. 211–220. [Google Scholar]
- Hall, A.E.; Moraru, C.I. Comparative Effects of High Pressure Processing and Heat Treatment on in Vitro Digestibility of Pea Protein and Starch. NPJ Sci. Food 2022, 6, 2. [Google Scholar] [CrossRef]
- Guy, R. Extrusion Cooking: Technologies and Applications; Woodhead publishing: Sawston, UK, 2001. [Google Scholar]
- Alonso, R.; Aguirre, A.; Marzo, F. Effects of Extrusion and Traditional Processing Methods on Antinutrients and in Vitro Digestibility of Protein and Starch in Faba and Kidney Beans. Food Chem. 2000, 68, 159–165. [Google Scholar] [CrossRef]
- Kelkar, S.; Siddiq, M.; Harte, J.B.; Dolan, K.D.; Nyombaire, G.; Suniaga, H. Use of Low-Temperature Extrusion for Reducing Phytohemagglutinin Activity (PHA) and Oligosaccharides in Beans (Phaseolus vulgaris L.) Cv. Navy and Pinto. Food Chem. 2012, 133, 1636–1639. [Google Scholar] [CrossRef]
- Rathod, R.P.; Annapure, U.S. Effect of Extrusion Process on Antinutritional Factors and Protein and Starch Digestibility of Lentil Splits. LWT—Food Sci. Technol. 2016, 66, 114–123. [Google Scholar] [CrossRef]
- Honig, D.H.; Wolf, W.J.; Rackis, J.J. Phytic Acid and Phosphorus Content of Various Soybean Protein Fractions. Cereal Chem. 1984, 61, 523–526. [Google Scholar]
- Khalil, A.A.; Mohamed, S.S.; Taha, F.S.; Karlsson, E.N. Production of Functional Protein Hydrolysates from Egyptian Breeds of Soybean and Lupin Seeds. Afr. J. Biotechnol. 2006, 5, 907–916. [Google Scholar]
- Clarke, E.; Wiseman, J. Effects of Extrusion Conditions on Trypsin Inhibitor Activity of Full Fat Soybeans and Subsequent Effects on Their Nutritional Value for Young Broilers. Br. Poult. Sci. 2007, 48, 703–712. [Google Scholar] [CrossRef]
- Adamidou, S.; Nengas, I.; Grigorakis, K.; Nikolopoulou, D.; Jauncey, K. Chemical Composition and Antinutritional Factors of Field Peas (Pisum Sativum), Chickpeas (Cicer Arietinum), and Faba Beans (Vicia Faba) as Affected by Extrusion Preconditioning and Drying Temperatures. Cereal Chem. J. 2011, 88, 80–86. [Google Scholar] [CrossRef]
- Broucke, K.; Van Poucke, C.; Duquenne, B.; De Witte, B.; Baune, M.-C.; Lammers, V.; Terjung, N.; Ebert, S.; Gibis, M.; Weiss, J.; et al. Ability of (Extruded) Pea Protein Products to Partially Replace Pork Meat in Emulsified Cooked Sausages. Innov. Food Sci. Emerg. Technol. 2022, 78, 102992. [Google Scholar] [CrossRef]
- Björck, I.; Asp, N.-G. The Effects of Extrusion Cooking on Nutritional Value—A Literature Review. J. Food Eng. 1983, 2, 281–308. [Google Scholar] [CrossRef]
- Furuichi, Y.; Kubota, Y.; Sugiura, Y.; Umekawa, H.; Takahashi, T.; Kouno, S. Effects of Low-Moisture Extrusion Cooking on the Chemical Composition and Nutritional Value of Whole Soybeans. Nippon. Eiyo Shokuryo Gakkaishi 1989, 42, 165–172. [Google Scholar] [CrossRef]
- Abd El-Hady, E.A.; Habiba, R.A. Effect of Soaking and Extrusion Conditions on Antinutrients and Protein Digestibility of Legume Seeds. LWT—Food Sci. Technol. 2003, 36, 285–293. [Google Scholar] [CrossRef]
- Alonso, R.; Orúe, E.; Zabalza, M.J.; Grant, G.; Marzo, F. Effect of Extrusion Cooking on Structure and Functional Properties of Pea and Kidney Bean Proteins. J. Sci. Food Agric. 2000, 80, 397–403. [Google Scholar] [CrossRef]
- Qi, M.; Zhang, G.; Ren, Z.; He, Z.; Peng, H.; Zhang, D.; Ma, C. Impact of Extrusion Temperature on In Vitro Digestibility and Pasting Properties of Pea Flour. Plant Foods Hum. Nutr. 2021, 76, 26–30. [Google Scholar] [CrossRef]
- Devi, S.; Varkey, A.; Dharmar, M.; Holt, R.R.; Allen, L.H.; Sheshshayee, M.S.; Preston, T.; Keen, C.L.; Kurpad, A. v Amino Acid Digestibility of Extruded Chickpea and Yellow Pea Protein Is High and Comparable in Moderately Stunted South Indian Children with Use of a Dual Stable Isotope Tracer Method. J. Nutr. 2020, 150, 1178–1185. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Bhirud, P.R.; Tyler, R.T. Extrusion Texturization of Air-Classified Pea Protein. J. Food Sci. 1999, 64, 509–513. [Google Scholar] [CrossRef]
- Omosebi, M.O.; Osundahunsi, O.F.; Fagbemi, T.N. Effect of Extrusion on Protein Quality, Antinutritional Factors, and Digestibility of Complementary Diet from Quality Protein Maize and Soybean Protein Concentrate. J. Food Biochem. 2018, 42, e12508. [Google Scholar] [CrossRef]
- Baune, M.-C.; Jeske, A.-L.; Profeta, A.; Smetana, S.; Broucke, K.; van Royen, G.; Gibis, M.; Weiss, J.; Terjung, N. Effect of Plant Protein Extrudates on Hybrid Meatballs—Changes in Nutritional Composition and Sustainability. Future Foods 2021, 4, 100081. [Google Scholar] [CrossRef]
- Osen, R.; Toelstede, S.; Eisner, P.; Schweiggert-Weisz, U. Effect of High Moisture Extrusion Cooking on Protein-Protein Interactions of Pea (Pisum Sativum L.) Protein Isolates. Int. J. Food Sci. Technol. 2015, 50, 1390–1396. [Google Scholar] [CrossRef]
- Palanisamy, M.; Franke, K.; Berger, R.G.; Heinz, V.; Töpfl, S. High Moisture Extrusion of Lupin Protein: Influence of Extrusion Parameters on Extruder Responses and Product Properties. J. Sci. Food Agric. 2019, 99, 2175–2185. [Google Scholar] [CrossRef] [PubMed]
- Pasqualone, A.; Costantini, M.; Coldea, T.E.; Summo, C. Use of Legumes in Extrusion Cooking: A Review. Foods 2020, 9, 958. [Google Scholar] [CrossRef]
- Alonso, R.; Rubio, L.A.; Muzquiz, M.; Marzo, F. The Effect of Extrusion Cooking on Mineral Bioavailability in Pea and Kidney Bean Seed Meals. Anim. Feed. Sci. Technol. 2001, 94, 1–13. [Google Scholar] [CrossRef]
- Jeunink, J.; Cheftel, J.C. Chemical and Physicochemical Changes in Field Bean and Soybean Proteins Texturized by Extrusion. J. Food Sci. 1979, 44, 1322–1325. [Google Scholar] [CrossRef]
- Singh, S.; Gamlath, S.; Wakeling, L. Nutritional Aspects of Food Extrusion: A Review. Int. J. Food Sci. Technol. 2007, 42, 916–929. [Google Scholar] [CrossRef]
- Tso, R.; Forde, C.G. Unintended Consequences: Nutritional Impact and Potential Pitfalls of Switching from Animal- to Plant-Based Foods. Nutrients 2021, 13, 2527. [Google Scholar] [CrossRef]
- Gendel, S.M. Sequence Analysis for Assessing Potential Allergenicity. Ann. N. Y. Acad. Sci. 2006, 964, 87–98. [Google Scholar] [CrossRef] [PubMed]
- Shewry, P.R.; Beaudoin, F.; Jenkins, J.; Griffiths-Jones, S.; Mills, E.N.C. Plant Protein Families and Their Relationships to Food Allergy. Biochem. Soc. Trans. 2002, 30, 906–910. [Google Scholar] [CrossRef] [PubMed]
- Pi, X.; Sun, Y.; Fu, G.; Wu, Z.; Cheng, J. Effect of Processing on Soybean Allergens and Their Allergenicity. Trends Food Sci. Technol. 2021, 118, 316–327. [Google Scholar] [CrossRef]
- Helm, R.M.; Cockrell, G.; Connaughton, C.; Sampson, H.A.; Bannon, G.A.; Beilinson, V.; Livingstone, D.; Nielsen, N.C.; Burks, A.W. A Soybean G2 Glycinin Allergen. Int. Arch. Allergy Immunol. 2000, 123, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Verhoeckx, K.C.M.; Vissers, Y.M.; Baumert, J.L.; Faludi, R.; Feys, M.; Flanagan, S.; Herouet-Guicheney, C.; Holzhauser, T.; Shimojo, R.; van der Bolt, N.; et al. Food Processing and Allergenicity. Food Chem. Toxicol. 2015, 80, 223–240. [Google Scholar] [CrossRef] [PubMed]
- Sicherer, S.H.; Morrow, E.H.; Sampson, H.A. Dose-Response in Double-Blind, Placebo-Controlled Oral Food Challenges in Children with Atopic Dermatitis. J. Allergy Clin. Immunol. 2000, 105, 582–586. [Google Scholar] [CrossRef]
- Sanchez-Monge, R.; Lopez-Torrejon, G.; Pascual, C.Y.; Varela, J.; Martin-Esteban, M.; Salcedo, G. Vicilin and Convicilin Are Potential Major Allergens from Pea. Clin. Exp. Allergy 2004, 34, 1747–1753. [Google Scholar] [CrossRef]
- Taylor, S.L.; Marsh, J.T.; Koppelman, S.J.; Kabourek, J.L.; Johnson, P.E.; Baumert, J.L. A Perspective on Pea Allergy and Pea Allergens. Trends Food Sci. Technol. 2021, 116, 186–198. [Google Scholar] [CrossRef]
- Troszynska, A.; Szymkiewicz, A.; Wolejszo, A. The effects of germination on the sensory quality and immunoreactive properties of pea (Pisum sativum L.) And soybean (Glycine max). J. Food Qual. 2007, 30, 1083–1100. [Google Scholar] [CrossRef]
- Ding, J.; Ju, H.; Zhong, L.; Qi, L.; Sun, N.; Lin, S. Reducing the Allergenicity of Pea Protein Based on the Enzyme Action of Alcalase. Food Funct. 2021, 12, 5940–5948. [Google Scholar] [CrossRef]
- Ogawa, T.; Samoto, M.; Takahashi, K. Soybean Allergens and Hypoallergenic Soybean Products. J. Nutr. Sci. Vitaminol. 2000, 46, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Gu, X.; Shang, F.; Qiu, J.; Li, J.; Li, J. Investigation and Analysis of Main Antinutritional Factors in Soybean and Extruded Soybean. Sci. Agric. Sin. 2016, 49, 2174–2182. [Google Scholar] [CrossRef]
- Yamanishi, R.; Huang, T.; Tsuji, H.; Bando, N.; Ogawa, T. Reduction of the Soybean Allergenicity by the Fermentation with Bacillus Natto. Food Sci. Technol. Int. Tokyo 1995, 1, 14–17. [Google Scholar] [CrossRef]
- Pi, X.; Wan, Y.; Yang, Y.; Li, R.; Wu, X.; Xie, M.; Li, X.; Fu, G. Research Progress in Peanut Allergens and Their Allergenicity Reduction. Trends Food Sci. Technol. 2019, 93, 212–220. [Google Scholar] [CrossRef]
- Sathe, S.K.; Teuber, S.S.; Roux, K.H. Effects of Food Processing on the Stability of Food Allergens. Biotechnol. Adv. 2005, 23, 423–429. [Google Scholar] [CrossRef]
- Jiménez-Saiz, R.; Benedé, S.; Molina, E.; López-Expósito, I. Effect of Processing Technologies on the Allergenicity of Food Products. Crit. Rev. Food Sci. Nutr. 2015, 55, 1902–1917. [Google Scholar] [CrossRef]
- Chizoba Ekezie, F.-G.; Cheng, J.-H.; Sun, D.-W. Effects of Nonthermal Food Processing Technologies on Food Allergens: A Review of Recent Research Advances. Trends Food Sci. Technol. 2018, 74, 12–25. [Google Scholar] [CrossRef]
- Abbas, A.K.; Lichtman, A.; Pillai, S. Antibodies and Antigens. In Cellular and Molecular Immunology; Elsevier: Amsterdam, The Netherlands, 2021. [Google Scholar]
- Yin, H.; Jia, F.; Huang, J.; Zhang, Y.; Zheng, X.; Zhang, X. Effect of Extrusion on the Structure and Antigenicity of Soybean β-Conglycinin. Grain Oil Sci. Technol. 2019, 2, 67–72. [Google Scholar] [CrossRef]
- Zheng, H.; Yan, G.; Lee, Y.; Alcaraz, C.; Marquez, S.; de Mejia, E.G. Effect of the Extrusion Process on Allergen Reduction and the Texture Change of Soybean Protein Isolate-Corn and Soybean Flour-Corn Mixtures. Innov. Food Sci. Emerg. Technol. 2020, 64, 102421. [Google Scholar] [CrossRef]
- Davis, P.J.; Smales, C.M.; James, D.C. How Can Thermal Processing Modify the Antigenicity of Proteins? Allergy 2001, 56, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Ishaq, A.; Irfan, S.; Sameen, A.; Khalid, N. Plant-Based Meat Analogs: A Review with Reference to Formulation and Gastrointestinal Fate. Curr. Res. Food Sci. 2022, 5, 973–983. [Google Scholar] [CrossRef] [PubMed]
- Buyukunal, S.K.; Issa, G.; Aksu, F.; Vural, A. Microbiological Quality of Fresh Vegetables and Fruits Collected from Supermarkets in Istanbul, Turkey. J. Food Nutr. Sci. 2015, 3, 152. [Google Scholar] [CrossRef]
- Alp, D.; Bulantekin, Ö. The Microbiological Quality of Various Foods Dried by Applying Different Drying Methods: A Review. Eur. Food Res. Technol. 2021, 247, 1333–1343. [Google Scholar] [CrossRef] [PubMed]
- Kleter, G.A.; Banach, J.L.; Dijkxhoorn, Y.; Fischer, A.R.H.; Kool, J.; Giller, K.E.; Oostewechel, R.J.A.; Schlangen, M.; Timmer, R.; van der Goot, A.J.; et al. Pulse of Africa: Exploring West-African Soybean, Cowpea, and Bambara Groundnut; Report; Wageningen University & Research: Wageningen, The Netherlands, 2020. [Google Scholar]
- FAO/WHO Shiga Toxin-Producing Escherichia Coli (STEC) and Food: Attribution, Characterization, and Monitoring; FAO/WHO: Bethesda, MD, USA, 2018.
- EFSA/BIOHAZ. Scientific Opinion on the Risk Posed by Pathogens in Food of Non-Animal Origin. Part 1 (Outbreak Data Analysis and Risk Ranking of Food/Pathogen Combinations). EFSA J. 2013, 11, 3025. [Google Scholar] [CrossRef]
- NCEZID. Reports of Foodborne and Waterborne Disease Outbreaks and Enteric (Intestinal) Disease Outbreaks Spread by Contact with Environmental Sources, Infected People or Animals, and Other Means. National Outbreak Reporting System (NORS) Dashboard. Available online: https://wwwn.cdc.gov/norsdashboard/ (accessed on 9 May 2022).
- EFSA/ECDC. The European Union One Health 2019 Zoonoses Report. EFSA J. 2021, 19, e06406. [Google Scholar] [CrossRef]
- Munimbazi, C.; Bullerman, L.B. Molds and Mycotoxins in Foods from Burundi†. J. Food Prot. 1996, 59, 869–875. [Google Scholar] [CrossRef]
- Hitokoto, H.; Morozumi, S.; Wauke, T.; Sakai, S.; Kurata, H. Fungal Contamination and Mycotoxin-Producing Potential of Dried Beans. Mycopathologia 1981, 73, 33–38. [Google Scholar] [CrossRef] [PubMed]
- FDA. Hazard Analysis and Risk-Based Preventive Controls for Human Food: Draft Guidance for Industry; FDA: Silver Spring, MD, USA, 2018. [Google Scholar]
- Filho, G.C.S.; Vessoni Penna, T.C.; Schaffner, D.W. Microbiological Quality of Vegetable Proteins during the Preparation of a Meat Analog. Ital. J. Food Sci. 2005, 17, 269–283. [Google Scholar]
- NIZO. Unlock the Potential of Alternative Proteins; NIZO: Ede, The Netherlands, 2022. [Google Scholar]
- CFIA. Bacterial Pathogens in Seed Powder and Plant-Based Protein Powder; CFIA: Ottawa, ON, Canada, 2018. [Google Scholar]
- Tóth, A.J.; Dunay, A.; Battay, M.; Illés, C.B.; Bittsánszky, A.; Süth, M. Microbial Spoilage of Plant-Based Meat Analogues. Appl. Sci. 2021, 11, 8309. [Google Scholar] [CrossRef]
- Leutgeb, K. Microbial Examination of Raw and Extruded Products for the Production of a Vegetarian Meat Analogue. Master’s Thesis, University of Natural Resources and Life Sciences, Vienna, Austria, 2015. [Google Scholar]
- Wild, F.; Czerny, M.; Janssen, A.M.; Kole, A.P.W.; Zunabovic, M.; Domig, K.J. The Evolution of a Plant-Based Alternative to Meat: From Niche Markets to Widely Accepted Meat Alternatives. Agro Food Ind. Hi Tech. 2014, 25, 45–49. [Google Scholar]
- Mustakas, G.C.; Griffin, E.L.; Allen, L.E.; Smith, O.B. Production and Nutritional Evaluation of Extrusion-Cooked Full-Fat Soybean Flour. J. Am. Oil Chem. Soc. 1964, 41, 607–614. [Google Scholar] [CrossRef]
- DeMeulaere, H.J.H.; Buzzard, J.L. Cooker Extruder in Service of World Feeding. Food Technol. 1969, 23, 345–351. [Google Scholar]
- Kauffman, K.B.; Hatch, R.T. Shear Effects on Cell Viability during Extrusion of Semi-Moist Foods. J. Food Sci. 1977, 42, 906–910. [Google Scholar] [CrossRef]
- Quéguiner, C.; Dumay, E.; Cavalier, C.; Cheftel, J.C. Reduction of Streptococcus Thermophilus in a Whey Protein Isolate by Low Moisture Extrusion Cooking without Loss of Functional Properties. Int. J. Food Sci. Technol. 2007, 24, 601–612. [Google Scholar] [CrossRef]
- Geeraerts, W.; de Vuyst, L.; Leroy, F. Ready-to-Eat Meat Alternatives, a Study of Their Associated Bacterial Communities. Food Biosci. 2020, 37, 100681. [Google Scholar] [CrossRef]
- Geeraerts, W.; Pothakos, V.; de Vuyst, L.; Leroy, F. Variability within the Dominant Microbiota of Sliced Cooked Poultry Products at Expiration Date in the Belgian Retail. Food Microbiol. 2018, 73, 209–215. [Google Scholar] [CrossRef]
- Hai, D.; Guo, B.; Qiao, M.; Jiang, H.; Song, L.; Meng, Z.; Huang, X. Evaluating the Potential Safety Risk of Plant-Based Meat Analogues by Analyzing Microbial Community Composition. Foods 2023, 13, 117. [Google Scholar] [CrossRef]
- Janik, E.; Niemcewicz, M.; Ceremuga, M.; Stela, M.; Saluk-Bijak, J.; Siadkowski, A.; Bijak, M. Molecular Aspects of Mycotoxins—A Serious Problem for Human Health. Int. J. Mol. Sci. 2020, 21, 8187. [Google Scholar] [CrossRef]
- Domijan, A.-M.; Peraica, M.; Žlender, V.; Cvjetković, B.; Jurjević, Ž.; Topolovec-Pintarić, S.; Ivić, D. Seed-Borne Fungi and Ochratoxin A Contamination of Dry Beans (Phaseolus vulgaris L.) in the Republic of Croatia. Food Chem. Toxicol. 2005, 43, 427–432. [Google Scholar] [CrossRef]
- Kolakowski, B.; O’Rourke, S.M.; Bietlot, H.P.; Kurz, K.; Aweryn, B. Ochratoxin A Concentrations in a Variety of Grain-Based and Non–Grain-Based Foods on the Canadian Retail Market from 2009 to 2014. J. Food Prot. 2016, 79, 2143–2159. [Google Scholar] [CrossRef]
- Ahn, S.; Lee, S.; Lee, J.; Kim, B. Accurate Determination of Ochratoxin A in Korean Fermented Soybean Paste by Isotope Dilution-Liquid Chromatography Tandem Mass Spectrometry. Food Chem. 2016, 190, 368–373. [Google Scholar] [CrossRef]
- Kunz, B.M.; Wanko, F.; Kemmlein, S.; Bahlmann, A.; Rohn, S.; Maul, R. Development of a Rapid Multi-Mycotoxin LC-MS/MS Stable Isotope Dilution Analysis for Grain Legumes and Its Application on 66 Market Samples. Food Control 2020, 109, 106949. [Google Scholar] [CrossRef]
- Valenta, H.; Dänicke, S.; Blüthgen, A. Mycotoxins in Soybean Feedstuffs Used in Germany. Mycotoxin Res. 2002, 18, 208–211. [Google Scholar] [CrossRef]
- Owolabi, I.O.; Kolawole, O.; Jantarabut, P.; Elliott, C.T.; Petchkongkaew, A. The Importance and Mitigation of Mycotoxins and Plant Toxins in Southeast Asian Fermented Foods. NPJ Sci. Food 2022, 6, 39. [Google Scholar] [CrossRef]
- Rafai, P.; Bata, R.; Jakab, L.; Vanyi, A. Evaluation of Mycotoxin-Contaminated Cereals for Their Use in Animal Feeds in Hungary. Food Addit. Contam. 2000, 17, 799–808. [Google Scholar] [CrossRef]
- Iha, M.H.; Trucksess, M.W.; Tournas, V.H. Effect of Processing on Ochratoxin A Content in Dried Beans. Food Addit. Contam. Part A 2009, 26, 1389–1395. [Google Scholar] [CrossRef]
- The Clean Label Project Protein Powder: Our Point of View. 2018. Available online: https://cleanlabelproject.org/protein-powder-infographic/ (accessed on 1 December 2022).
- Castells, M.; Marín, S.; Sanchis, V.; Ramos, A.J. Fate of Mycotoxins in Cereals during Extrusion Cooking: A Review. Food Addit. Contam. 2005, 22, 150–157. [Google Scholar] [CrossRef]
- Zheng, H.; Wei, S.; Xu, Y.; Fan, M. Reduction of Aflatoxin B1 in Peanut Meal by Extrusion Cooking. LWT—Food Sci. Technol. 2015, 64, 515–519. [Google Scholar] [CrossRef]
- Saalia, F.K.; Phillips, R.D. Reduction of Aflatoxins in Peanut Meal by Extrusion Cooking in the Presence of Nucleophiles. LWT—Food Sci. Technol. 2011, 44, 1511–1516. [Google Scholar] [CrossRef]
- Kaushik, G.; Satya, S.; Naik, S.N. Pesticide Residue Dissipation Upon Storage and Processing in Chickpea Legume for Food Safety. Adv. Food Technol. Nutr. Sci.—Open J. 2016, 2, 64–72. [Google Scholar] [CrossRef]
- Han, C.-T.; Kim, M.-J.; Moon, S.-H.; Jeon, Y.-R.; Hwang, J.-S.; Nam, C.; Park, C.-W.; Lee, S.-H.; Na, J.-B.; Park, C.-S.; et al. Acute and 28-Day Subacute Toxicity Studies of Hexane Extracts of the Roots of Lithospermum Erythrorhizon in Sprague-Dawley Rats. Toxicol. Res. 2015, 31, 403–414. [Google Scholar] [CrossRef][Green Version]
- González-Pérez, S.; Arellano, J.B. Vegetable Protein Isolates. In Handbook of Hydrocolloids; Elsevier: Amsterdam, The Netherlands, 2009; pp. 383–419. [Google Scholar]
- Schuh, V.; Allard, K.; Herrmann, K.; Gibis, M.; Kohlus, R.; Weiss, J. Impact of Carboxymethyl Cellulose (CMC) and Microcrystalline Cellulose (MCC) on Functional Characteristics of Emulsified Sausages. Meat Sci. 2013, 93, 240–247. [Google Scholar] [CrossRef]
- Jenkins, D.J.; Wolever, T.M.; Leeds, A.R.; Gassull, M.A.; Haisman, P.; Dilawari, J.; Goff, D.V.; Metz, G.L.; Alberti, K.G. Dietary Fibres, Fibre Analogues, and Glucose Tolerance: Importance of Viscosity. BMJ 1978, 1, 1392–1394. [Google Scholar] [CrossRef]
- Younes, M.; Aggett, P.; Aguilar, F.; Crebelli, R.; di Domenico, A.; Dusemund, B.; Filipič, M.; Jose Frutos, M.; Galtier, P.; Gott, D.; et al. Re-evaluation of Celluloses E 460(i), E 460(Ii), E 461, E 462, E 463, E 464, E 465, E 466, E 468 and E 469 as Food Additives. EFSA J. 2018, 16, e05047. [Google Scholar] [CrossRef]
- Benard, C.; Cultrone, A.; Michel, C.; Rosales, C.; Segain, J.-P.; Lahaye, M.; Galmiche, J.-P.; Cherbut, C.; Blottière, H.M. Degraded Carrageenan Causing Colitis in Rats Induces TNF Secretion and ICAM-1 Upregulation in Monocytes through NF-ΚB Activation. PLoS ONE 2010, 5, e8666. [Google Scholar] [CrossRef] [PubMed]
- Engster, M.; Abraham, R. Cecal Response to Different Molecular Weights and Types of Carrageenan in the Guinea Pig. Toxicol. Appl. Pharmacol. 1976, 38, 265–282. [Google Scholar] [CrossRef] [PubMed]
- Capron, I.; Yvon, M.; Muller, G. In-Vitro Gastric Stability of Carrageenan. Food Hydrocoll. 1996, 10, 239–244. [Google Scholar] [CrossRef]
- David, S.; Shani Levi, C.; Fahoum, L.; Ungar, Y.; Meyron-Holtz, E.G.; Shpigelman, A.; Lesmes, U. Revisiting the Carrageenan Controversy: Do We Really Understand the Digestive Fate and Safety of Carrageenan in Our Foods? Food Funct. 2018, 9, 1344–1352. [Google Scholar] [CrossRef]
- Bao, W.; Rong, Y.; Rong, S.; Liu, L. Dietary Iron Intake, Body Iron Stores, and the Risk of Type 2 Diabetes: A Systematic Review and Meta-Analysis. BMC Med. 2012, 10, 119. [Google Scholar] [CrossRef]
- Fraser, R.Z.; Shitut, M.; Agrawal, P.; Mendes, O.; Klapholz, S. Safety Evaluation of Soy Leghemoglobin Protein Preparation Derived From Pichia Pastoris, Intended for Use as a Flavor Catalyst in Plant-Based Meat. Int. J. Toxicol. 2018, 37, 241–262. [Google Scholar] [CrossRef]
- Zhu, Z.; Xu, Y.; Huang, T.; Yu, Y.; Bassey, A.P.; Huang, M. The Contamination, Formation, Determination and Control of Polycyclic Aromatic Hydrocarbons in Meat Products. Food Control 2022, 141, 109194. [Google Scholar] [CrossRef]
- Visvanathan, R.; Krishnakumar, T. Acrylamide in Food Products: A Review. J. Food Process Technol. 2014, 5, 344. [Google Scholar] [CrossRef]
- Oz, F.; Kaban, G.; Kaya, M. Effects of Cooking Methods and Levels on Formation of Heterocyclic Aromatic Amines in Chicken and Fish with Oasis Extraction Method. LWT—Food Sci. Technol. 2010, 43, 1345–1350. [Google Scholar] [CrossRef]
- Oz, F.; Kaban, G.; Kaya, M. Effects of Cooking Methods on the Formation of Heterocyclic Aromatic Amines of Two Different Species Trout. Food Chem. 2007, 104, 67–72. [Google Scholar] [CrossRef]
- Toribio, F.; Busquets, R.; Puignou, L.; Galceran, M.T. Heterocyclic Amines in Griddled Beef Steak Analysed Using a Single Extract Clean-up Procedure. Food Chem. Toxicol. 2007, 45, 667–675. [Google Scholar] [CrossRef]
- Barzegar, F.; Kamankesh, M.; Mohammadi, A. Heterocyclic Aromatic Amines in Cooked Food: A Review on Formation, Health Risk-Toxicology and Their Analytical Techniques. Food Chem. 2019, 280, 240–254. [Google Scholar] [CrossRef]
- Murkovic, M. Formation of Heterocyclic Aromatic Amines in Model Systems. J. Chromatogr. B 2004, 802, 3–10. [Google Scholar] [CrossRef]
- Pfau, W.; Skog, K. Exposure to β-Carbolines Norharman and Harman. J. Chromatogr. B 2004, 802, 115–126. [Google Scholar] [CrossRef]
- Dong, H.; Xian, Y.; Li, H.; Bai, W.; Zeng, X. Potential Carcinogenic Heterocyclic Aromatic Amines (HAAs) in Foodstuffs: Formation, Extraction, Analytical Methods, and Mitigation Strategies. Compr. Rev. Food Sci. Food Saf. 2020, 19, 365–404. [Google Scholar] [CrossRef]
- Tareke, E.; Rydberg, P.; Karlsson, P.; Eriksson, S.; Törnqvist, M. Analysis of Acrylamide, a Carcinogen Formed in Heated Foodstuffs. J. Agric. Food Chem. 2002, 50, 4998–5006. [Google Scholar] [CrossRef]
- Stadler, R.H.; Blank, I.; Varga, N.; Robert, F.; Hau, J.; Guy, P.A.; Robert, M.-C.; Riediker, S. Acrylamide from Maillard Reaction Products. Nature 2002, 419, 449–450. [Google Scholar] [CrossRef]
- Gertz, C.; Klostermann, S. Analysis of Acrylamide and Mechanisms of Its Formation in Deep-Fried Products. Eur. J. Lipid Sci. Technol. 2002, 104, 762–771. [Google Scholar] [CrossRef]
- Umano, K.; Shibamoto, T. Analysis of Acrolein from Heated Cooking Oils and Beef Fat. J. Agric. Food Chem. 1987, 35, 909–912. [Google Scholar] [CrossRef]
- Yaylayan, V.A.; Locas, C.P.; Wnorowski, A.; O’Brien, J. The Role of Creatine in the Generation of N-Methylacrylamide: A New Toxicant in Cooked Meat. J. Agric. Food Chem. 2004, 52, 5559–5565. [Google Scholar] [CrossRef]
- Stadler, R.H.; Verzegnassi, L.; Varga, N.; Grigorov, M.; Studer, A.; Riediker, S.; Schilter, B. Formation of Vinylogous Compounds in Model Maillard Reaction Systems. Chem. Res. Toxicol. 2003, 16, 1242–1250. [Google Scholar] [CrossRef]
- Claeys, W.L.; de Vleeschouwer, K.; Hendrickx, M.E. Effect of Amino Acids on Acrylamide Formation and Elimination Kinetics. Biotechnol. Prog. 2005, 21, 1525–1530. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation (EU) 2017/2158 of 20 November 2017 Establishing Mitigation Measures and Benchmark Levels for the Reduction of the Presence of Acrylamide in Food 2017. Off. J. 2017, L204, 24–44. [Google Scholar]
- Squeo, G.; De Angelis, D.; Caputi, A.F.; Pasqualone, A.; Summo, C.; Caponio, F. Screening of Acrylamide Content in Commercial Plant-Based Protein Ingredients from Different Technologies. Foods 2023, 12, 1331. [Google Scholar] [CrossRef]
- Pospiech, J.; Hoelzle, E.; Schoepf, A.; Melzer, T.; Granvogl, M.; Frank, J. Acrylamide Increases and Furanoic Compounds Decrease in Plant-Based Meat Alternatives during Pan-Frying. Food Chem. 2024, 439, 138063. [Google Scholar] [CrossRef]
- Li, C.; Li, C.; Yu, H.; Cheng, Y.; Xie, Y.; Yao, W.; Guo, Y.; Qian, H. Chemical Food Contaminants during Food Processing: Sources and Control. Crit. Rev. Food Sci. Nutr. 2021, 61, 1545–1555. [Google Scholar] [CrossRef]
- Singh, L.; Agarwal, T.; Simal-Gandara, J. PAHs, Diet and Cancer Prevention: Cooking Process Driven-Strategies. Trends Food Sci. Technol. 2020, 99, 487–506. [Google Scholar] [CrossRef]
- Badger, G.M.; Buttery, R.G. The Formation of Aromatic Hydrocarbons at High Temperatures. Part IV. The Pyrolysis of Styrene. J. Chem. Soc. (Resumed) 1958, 2458–2463. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation (EU) 2020/1255 of 7 September 2020 Amending Regulation (EC) No 1881/2006 as Regards Maximum Levels of Polycyclic Aromatic Hydrocarbons (PAHs) in Traditionally Smoked Meat and Smoked Meat Products and Traditionally Smoked Fish and Smoked Fishery Products and Establishing a Maximum Level of PAHs in Powders of Food of Plant Origin Used for the Preparation of Beverages. Off. J. 2020, L293, 1–4. [Google Scholar]
- da Paz, A.P.S.; Nascimento, E.C.P.; Marcondes, H.C.; da Silva, M.C.F.; Hamoy, M.; Mello, V.J. de Presença de Hidrocarbonetos Policíclicos Aromáticos Em Produtos Alimentícios e a Sua Relação Com o Método de Cocção e a Natureza Do Alimento. Braz. J. Food Technol. 2017, 20. [Google Scholar] [CrossRef]
- Friedman, M. Chemistry, Biochemistry, Nutrition, and Microbiology of Lysinoalanine, Lanthionine, and Histidinoalanine in Food and Other Proteins. J. Agric. Food Chem. 1999, 47, 1295–1319. [Google Scholar] [CrossRef]
- Nielsen, S.D.; Knudsen, L.J.; Bækgaard, L.T.; Rauh, V.; Larsen, L.B. Influence of Lactose on the Maillard Reaction and Dehydroalanine-Mediated Protein Cross-Linking in Casein and Whey. Foods 2022, 11, 897. [Google Scholar] [CrossRef]
- Sternberg, M.; Kim, C.Y. Lysinoalanine Formation in Protein Food Ingredients. In Protein Crosslinking; Advances in Experimental Medicine and Biology, Springer: Boston, MA, USA, 1977; Volume 86, pp. 73–84. [Google Scholar]
- Maga, J.A. Lysinoalanine in Foods. J. Agric. Food Chem. 1984, 32, 955–964. [Google Scholar] [CrossRef]
- De Groot, A.P.; Slump, P. Effects of Severe Alkali Treatment of Proteins on Amino Acid Composition and Nutritive Value. J. Nutr. 1969, 98, 45–56. [Google Scholar] [CrossRef]
- Friedman, M. Chapter 10: Lysinoalanine Formation in Soybean Proteins: Kinetics and Mechanisms. In Food Protein Deterioration; American Chemical Society: Washington, DC, USA, 1982; pp. 231–273. [Google Scholar]
- Li, T.; Guo, X.-N.; Zhu, K.-X.; Zhou, H.-M. Effects of Alkali on Protein Polymerization and Textural Characteristics of Textured Wheat Protein. Food Chem. 2018, 239, 579–587. [Google Scholar] [CrossRef]
- Friedman, M.; Gumbmann, M.R.; Masters, P.M. Protein-Alkali Reactions: Chemistry, Toxicology, and Nutritional Consequences. In Nutritional and Toxicological Aspects of Food Safety; Springer: Berlin/Heidelberg, Germany, 1984; pp. 367–412. [Google Scholar]



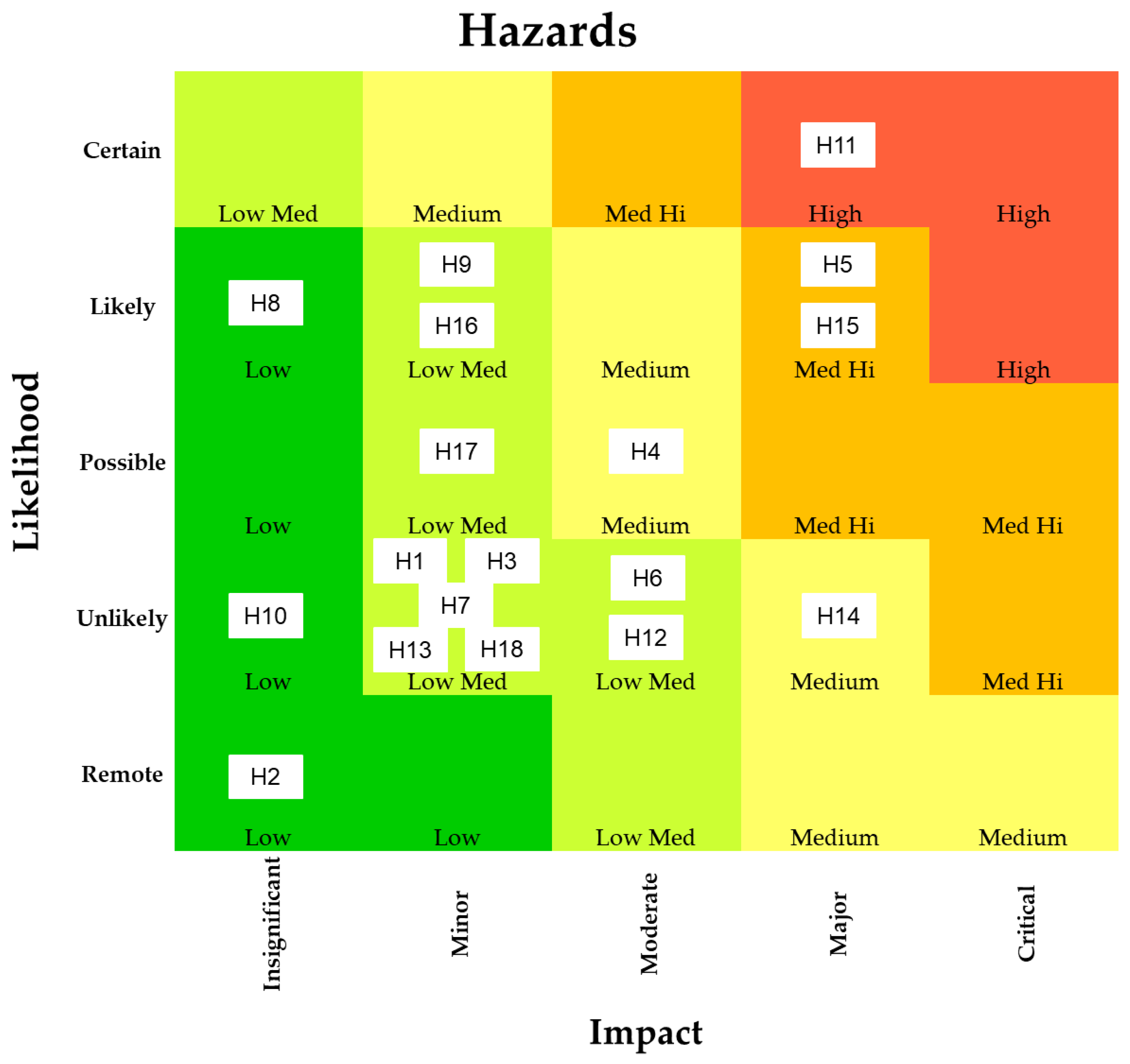
| Probability | Score | Description |
|---|---|---|
| Remote | 1 | Probability of less than 10% |
| Unlikely | 2 | Probability between 10% and 35% |
| Possible | 3 | Probability between 36% to 64% |
| Likely | 4 | Probability between 65% to 90% |
| Certain | 5 | Probability above 90% |
| Impact | Score | Description |
|---|---|---|
| Insignificant | 1 | Compounds are categorized as not hazardous or even though hazardous compounds might be present in consumed products, adverse reactions are very unlikely |
| Substances that have a positive effect on health are present in the consumed products | ||
| Minor | 2 | Substances are contaminants, but their concentration is regulated. Therefore, no adverse reaction is expected if the regulations are followed. However, they present a hazard and should be monitored |
| Substances might have adverse effects when ingested by humans, but there is no regulation or definitive information in this regard | ||
| Substances that have a positive effect on health are present but information about solely their consumption in regards to related animal-based products is lacking | ||
| Moderate | 3 | Substances are contaminants, regulations exist, and they might be present in consumed products |
| Substances are contaminants and might be present in consumed products, but no regulations for plant-based meat analogues exist. Until their concentration is also regulated in the investigated products, they possess a moderate threat | ||
| Major | 4 | Significant possibility of adverse reactions, no regulations exist, and substances might be present in the consumed products. |
| Susbtance considered a health concern but no epidemiological evidence exists. No regulations exist and substance was found in the consumed products | ||
| Substances that have a positive effect on health are only present in certain products at lower concentrations than animal-based products or other products with excellent nutritional properties | ||
| Critical | 5 | Significant possibility of adverse reactions, no regulations exist, and the substance was found in several related products |
| Substances that are known to have a positive effect on health or have excellent nutritional properties are missing |
| Code | Identified Hazard |
|---|---|
| Chemical hazards | |
| H1 | Heavy metals |
| H2 | Compounds related to protein |
| H3 | Compounds related to protein extraction—Chloropropanol |
| H4 | Compounds related to processing—NNAs and HAAs |
| H5 | Compounds related to processing—AAs |
| H6 | Compounds related to processing—PAHs |
| H7 | Compounds related to processing—Lysinolanine and lanthionine |
| H8 | Binders and gums—Methylcellulose |
| H9 | Binders and gums—Carrageenan |
| H10 | Flavorings and colorants—Leghemoglobin |
| Allergens | |
| H11 | Allergens |
| Microbial hazards | |
| H12 | Spore-and non-spore-forming bacteria |
| H13 | Spoilage organisms |
| H14 | Mycotoxins |
| Nutritional hazards | |
| H15 | Nutritional values |
| H16 | Protein metabolites |
| H17 | Anti-nutritional factors |
| H18 | Isoflavones |
| Parameter | Cooked Soy | Cooked Pea | SPC or SPI | PPC or PPI | Extruded Soy | Plant-Based Beef/Pork | Plant-Based Burger |
|---|---|---|---|---|---|---|---|
| PDCAAS | 73 | 50–89 | 86–100 | 71–93 | >54 | – | – |
| DIAAS | 65 | 46–89 | 84–95 | 62–70 | – | – | 71–107 |
| IVPD | 49 | – | 96 | 54–97 | 73–96 | 50–70 | – |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gräfenhahn, M.; Beyrer, M. Plant-Based Meat Analogues in the Human Diet: What Are the Hazards? Foods 2024, 13, 1541. https://doi.org/10.3390/foods13101541
Gräfenhahn M, Beyrer M. Plant-Based Meat Analogues in the Human Diet: What Are the Hazards? Foods. 2024; 13(10):1541. https://doi.org/10.3390/foods13101541
Chicago/Turabian StyleGräfenhahn, Maria, and Michael Beyrer. 2024. "Plant-Based Meat Analogues in the Human Diet: What Are the Hazards?" Foods 13, no. 10: 1541. https://doi.org/10.3390/foods13101541
APA StyleGräfenhahn, M., & Beyrer, M. (2024). Plant-Based Meat Analogues in the Human Diet: What Are the Hazards? Foods, 13(10), 1541. https://doi.org/10.3390/foods13101541








