Valorization of Second-Grade Date Fruit Byproducts and Nonstandard Sweet Potato Tubers to Produce Novel Biofortified Functional Jam
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Plant Material
2.1.2. Study Materials
2.2. Methods
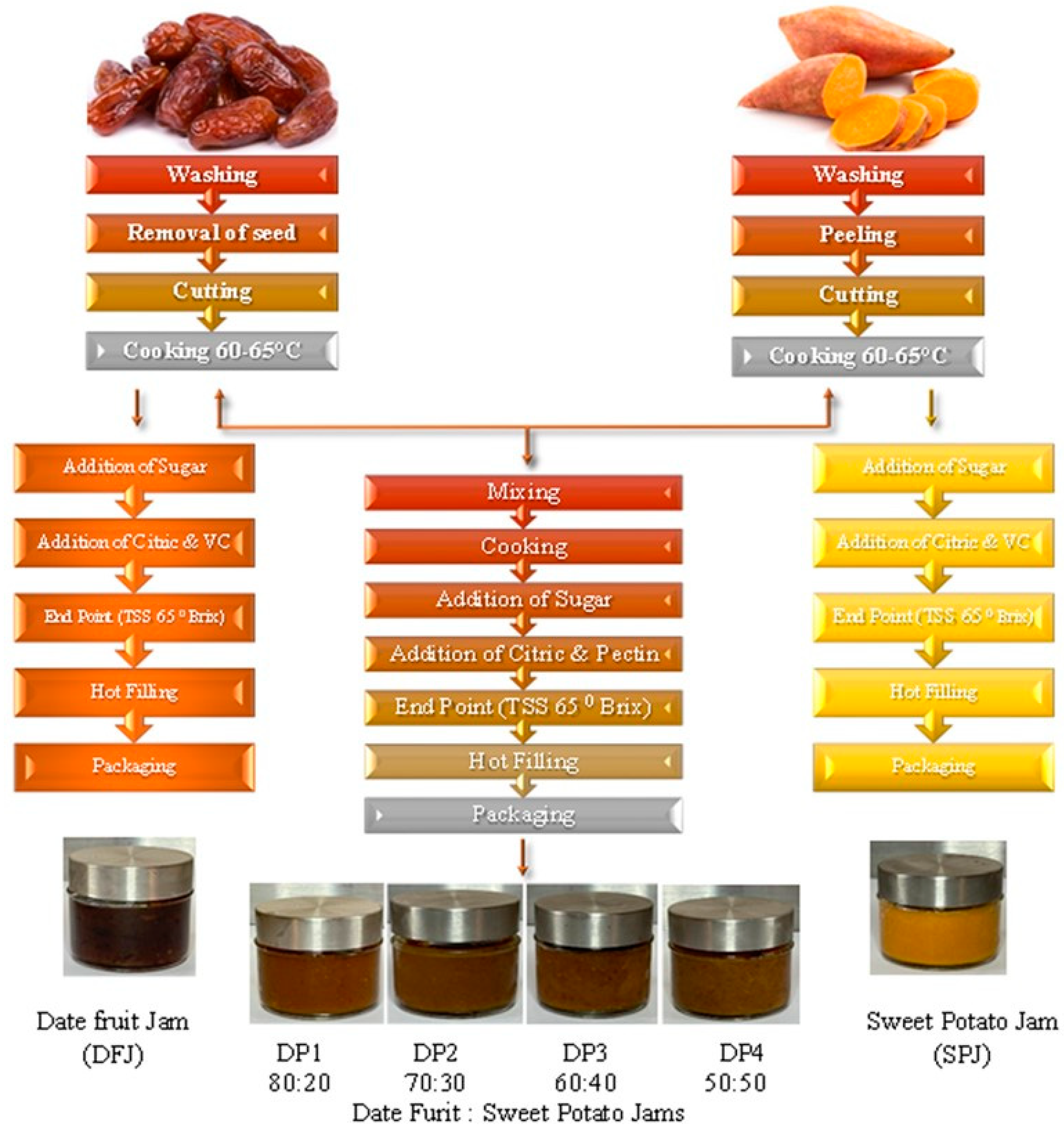
2.2.1. Preparation of Paste of Date Fruits and Sweet Potato Tubers
2.2.2. Preparation of Date Fruit Paste
2.2.3. Preparation of Sweet Potato Tuber Paste
2.2.4. Combining the Sweet Potato Tuber and Date Fruit Pastes
2.3. Physiochemical Properties of Date Fruit and Sweet Potato Tubers
2.3.1. pH
2.3.2. Total Soluble Solids (TSSs)
2.3.3. Color Measurement
2.3.4. Moisture Content
2.3.5. Total Sugars
2.3.6. Total Protein
2.3.7. Ash
2.3.8. Total Fat
2.3.9. Total Fiber
2.3.10. Minerals
2.4. Bioactive Compounds and Antioxidants
2.4.1. Extraction of Carotenoids
2.4.2. Extraction of Phenols
2.4.3. Total Phenolics
2.4.4. Total Flavonoids
2.4.5. DPPH• Free Radical-Scavenging Assay
2.4.6. ABTS• Free Radical-Scavenging Assay
2.4.7. Ferric-Reducing/Antioxidant Power (FRAP) Assay
2.4.8. Superoxide Anion Radical-Quenching Assay
2.5. Sensory Evaluation
2.6. Statistical Analysis
3. Results and Discussion
3.1. Physicochemical Properties of Raw Materials
3.2. Raw Material Bioactive Compounds and Antioxidant Activity
3.3. Physicochemical Properties of Jams
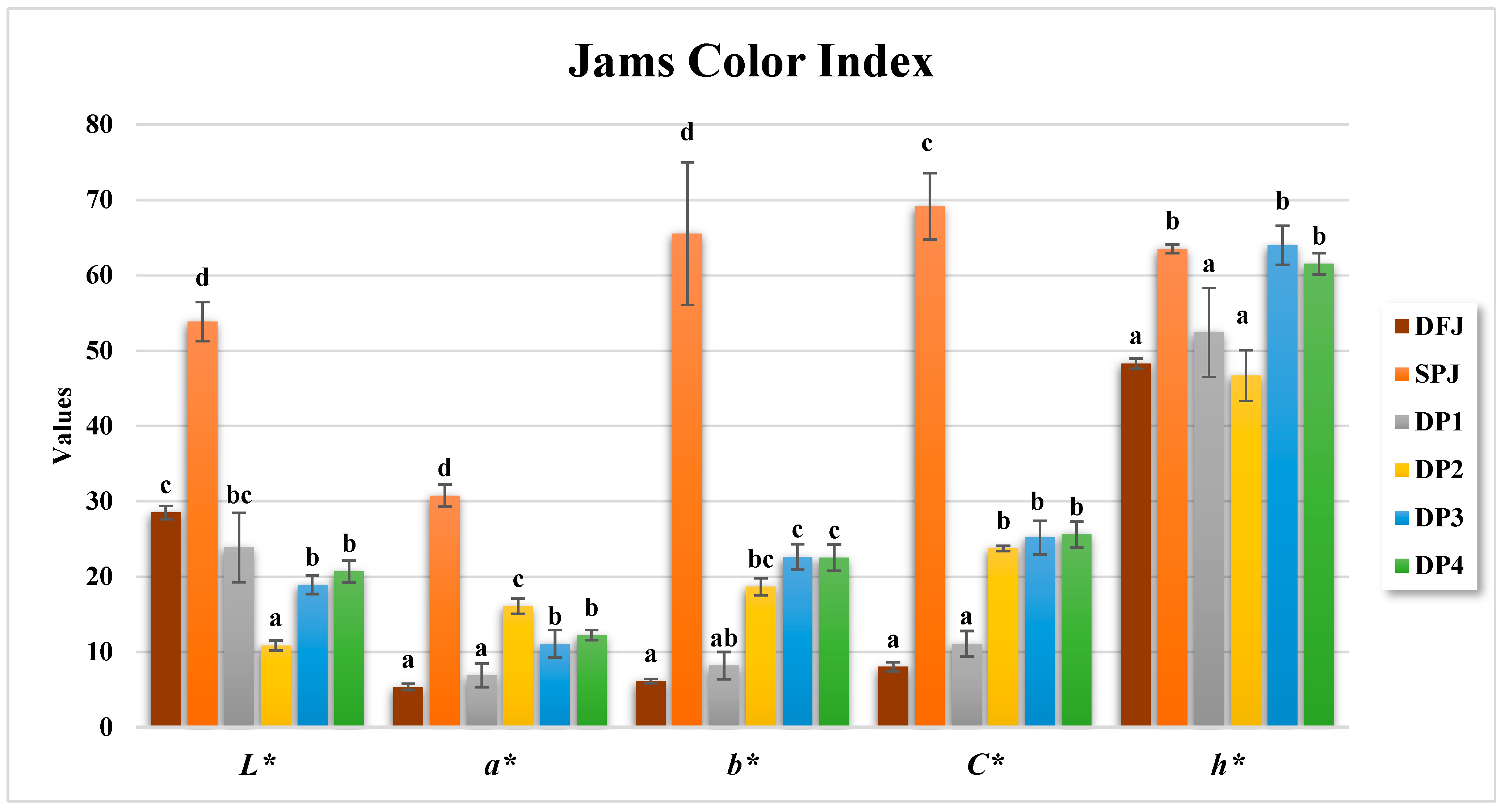
3.4. Color Parameters of Formulated Jams
3.5. Mineral Content of Jams
3.6. Bioactive Compounds in Formulated Jams
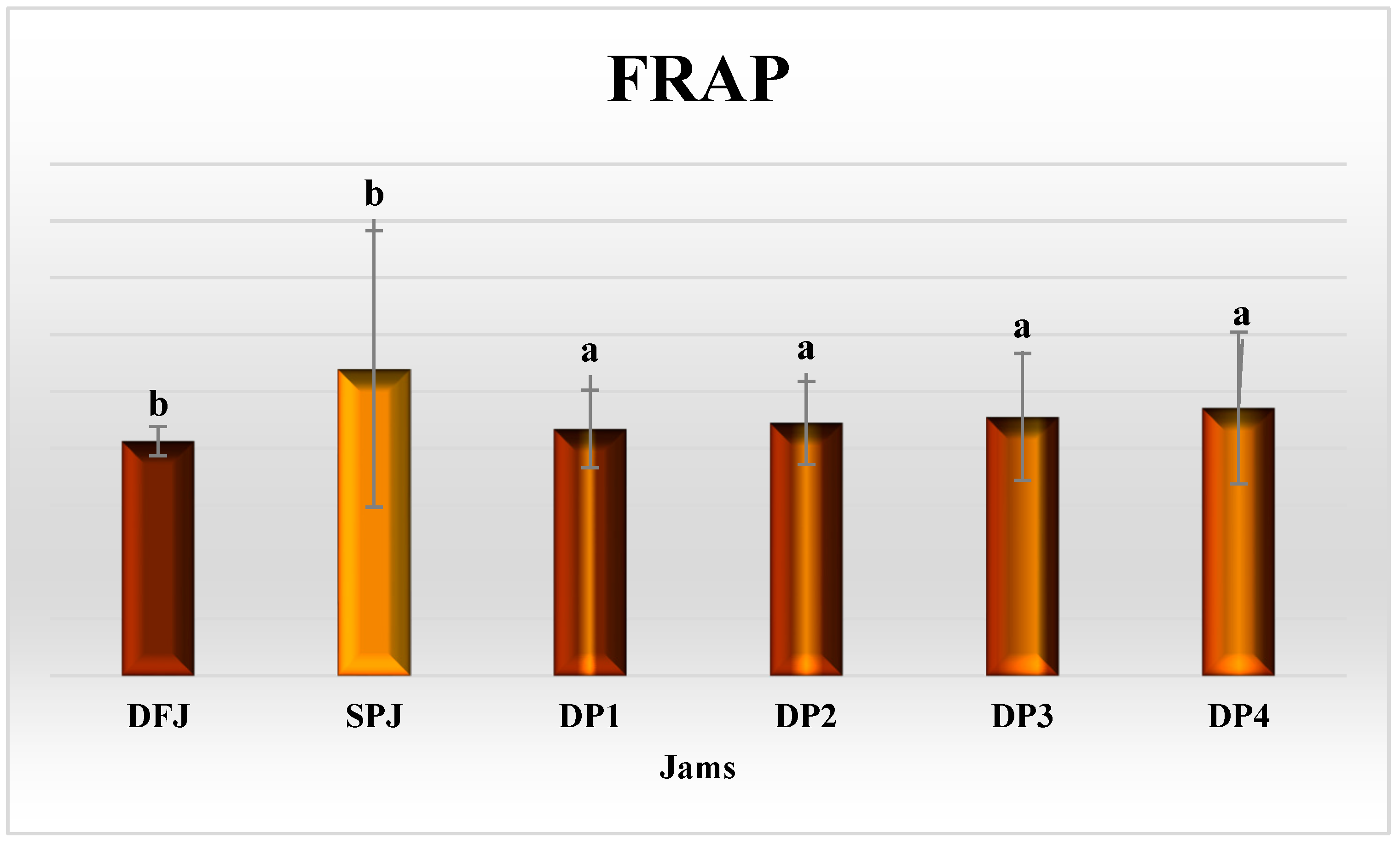
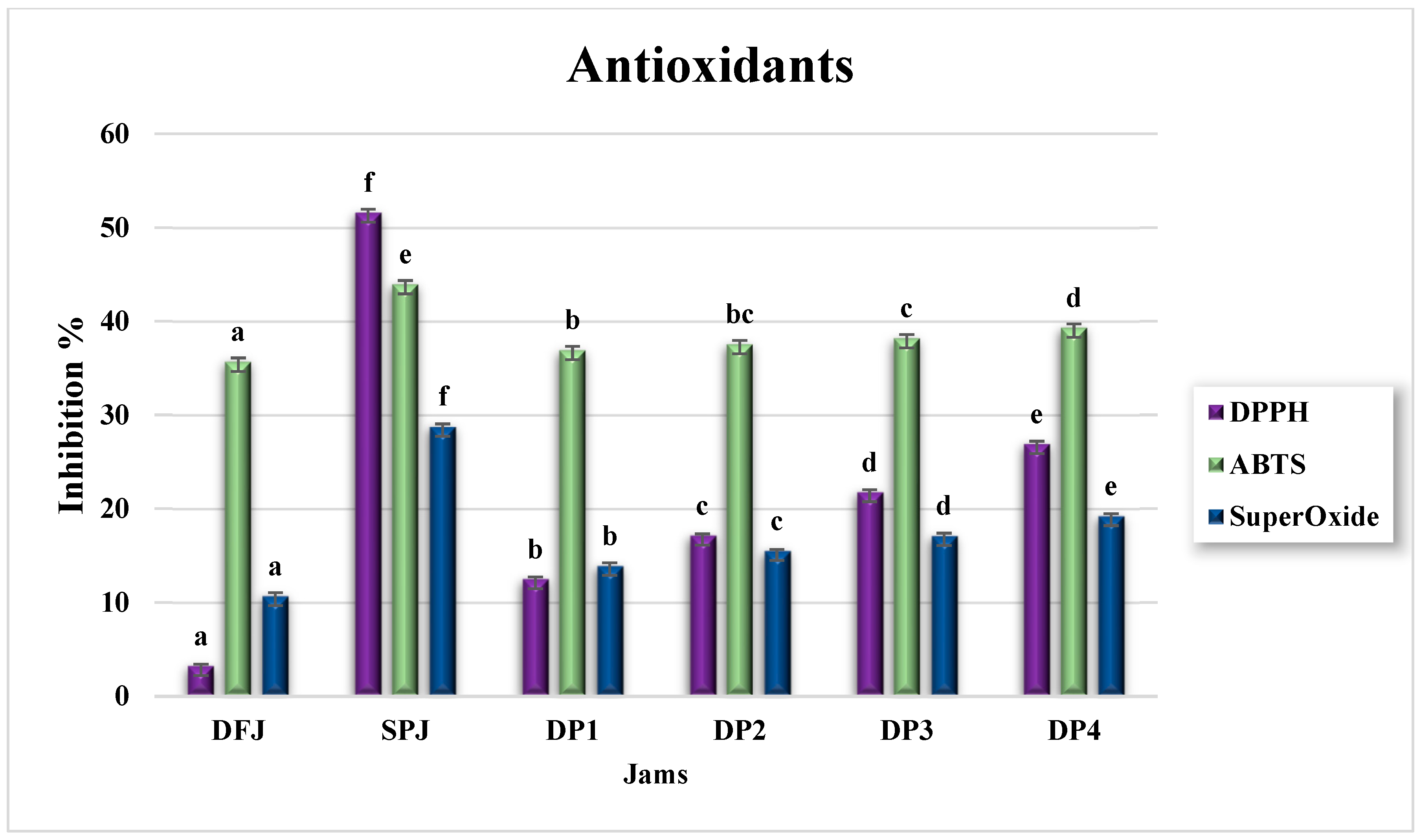
3.7. Antioxidant Activity of Jams
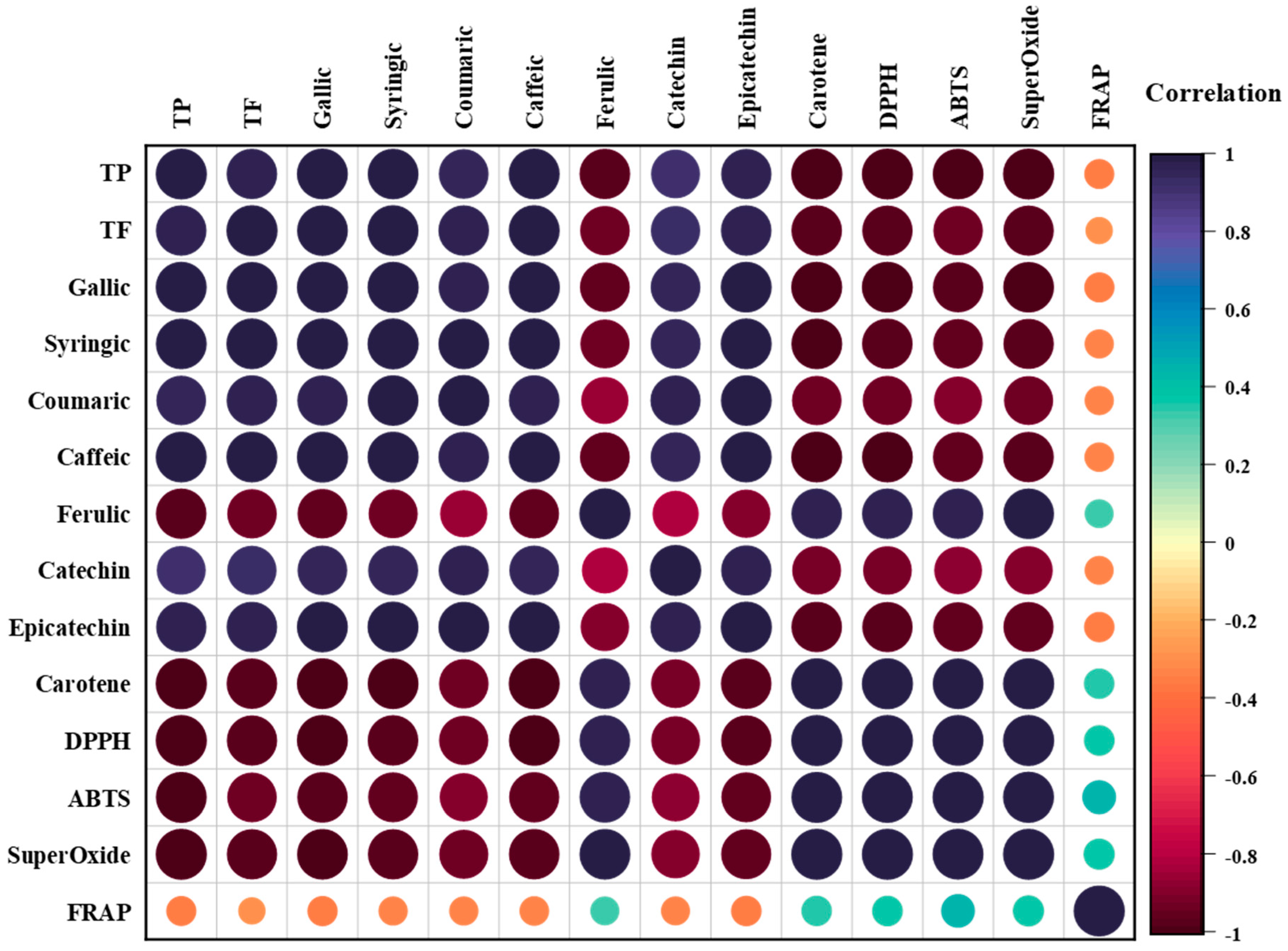
3.8. Correlation Analyses
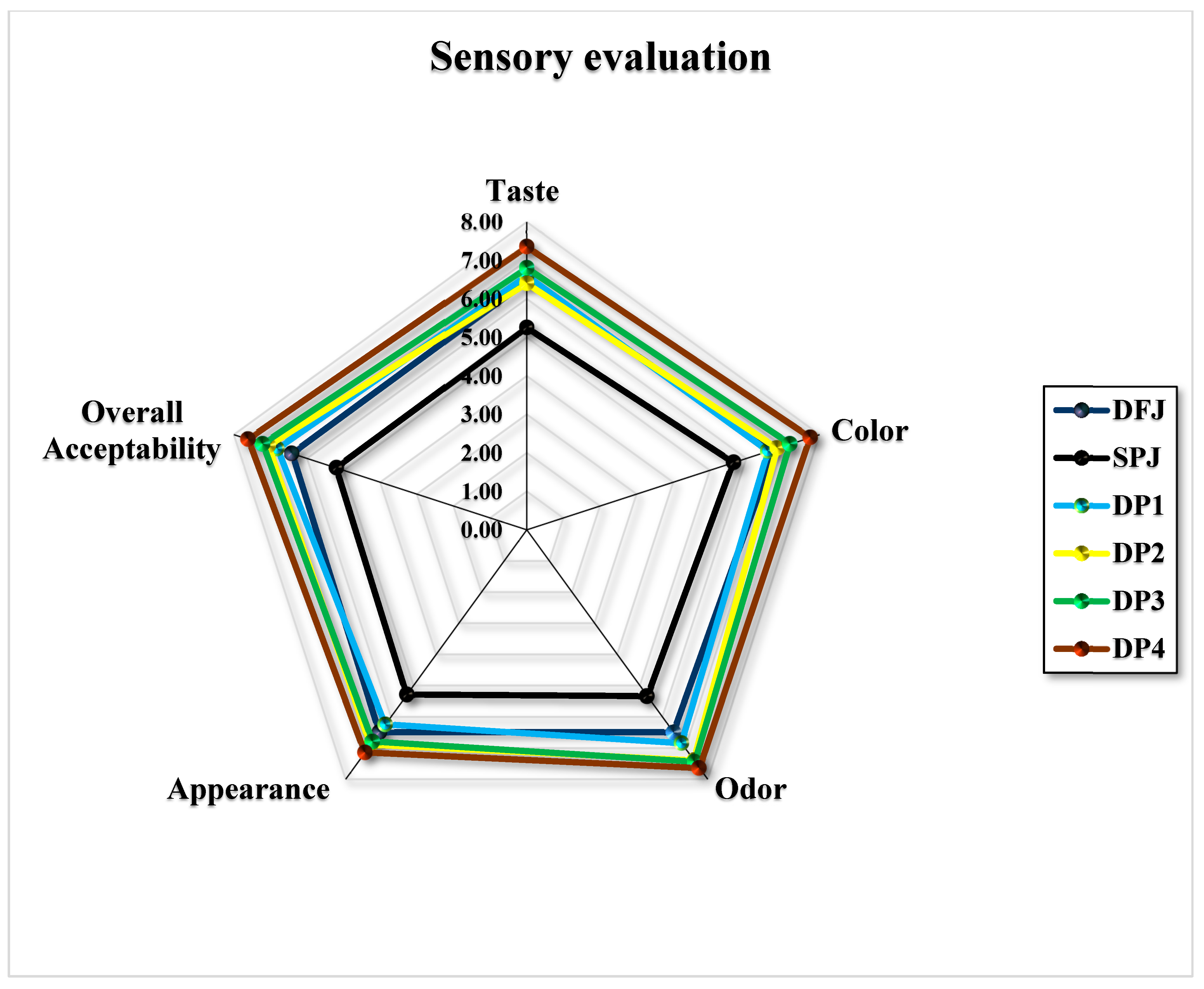
3.9. Sensory Evaluation
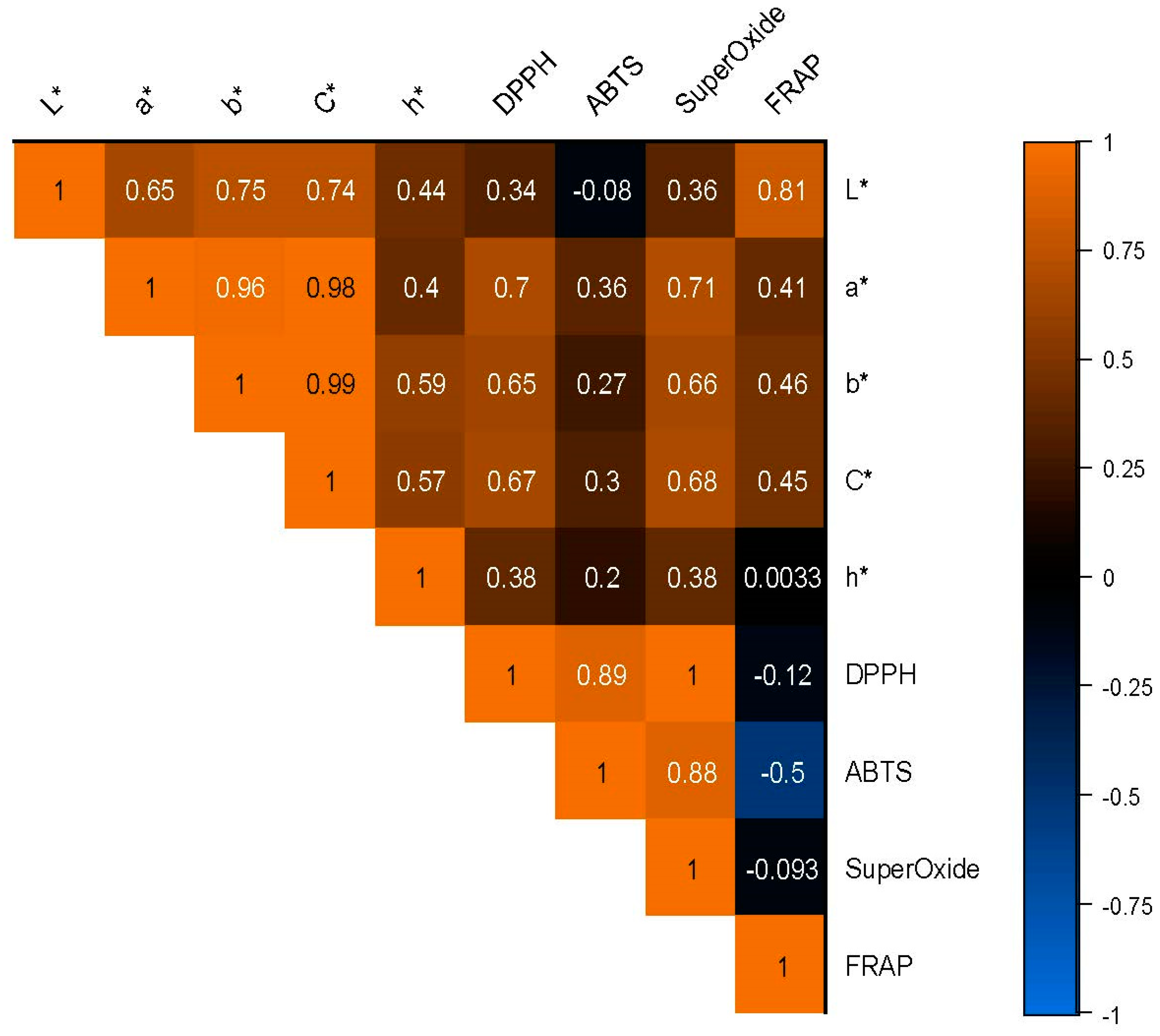
3.10. Correlation Analysis of Color Results Determined Instrumentally and Evaluated Sensorily
3.11. Correlation Analysis of Color Parameter and Antioxidant Activity
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kandemir, K.; Piskin, E.; Xiao, J.; Tomas, M.; Capanoglu, E. Fruit Juice Industry Wastes as a Source of Bioactives. J. Agric. Food Chem. 2022, 70, 6805–6832. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.; Bast, A.; de Boer, A. Valorized Food Processing By-Products in the EU: Finding the Balance between Safety, Nutrition, and Sustainability. Sustainability 2021, 13, 4428. [Google Scholar] [CrossRef]
- Caponio, F.; Piga, A.; Poiana, M. Valorization of Food Processing By-Products. Foods 2022, 11, 3246. [Google Scholar] [CrossRef]
- Iriondo-DeHond, M.; Miguel, E.; del Castillo, M. Food Byproducts as Sustainable Ingredients for Innovative and Healthy Dairy Foods. Nutrients 2018, 10, 1358. [Google Scholar] [CrossRef] [PubMed]
- Alqahtani, N.K.; Alnemr, T.M.; Alqattan, A.M.; Aleid, S.M.; Habib, H.M. Physicochemical and Sensory Properties and Shelf Life of Block-Type Processed Cheeses Fortified with Date Seeds (Phoenix dactylifera L.) as a Functional Food. Foods 2023, 12, 679. [Google Scholar] [CrossRef]
- Picciotti, U.; Massaro, A.; Galiano, A.; Garganese, F. Cheese Fortification: Review and Possible Improvements. Food Rev. Int. 2022, 38, 474–500. [Google Scholar] [CrossRef]
- Olson, R.; Gavin-Smith, B.; Ferraboschi, C.; Kraemer, K. Food Fortification: The Advantages, Disadvantages and Lessons from Sight and Life Programs. Nutrients 2021, 13, 1118. [Google Scholar] [CrossRef]
- Kumar, D.S. Herbal Bioactives and Food Fortification: Extraction and Formulation; CRC Press: Boca Raton, FL, USA, 2015. [Google Scholar]
- Chadare, F.J.; Idohou, R.; Nago, E.; Affonfere, M.; Agossadou, J.; Fassinou, T.K.; Kénou, C.; Honfo, S.; Azokpota, P.; Linnemann, A.R.; et al. Conventional and Food-to-food Fortification: An Appraisal of Past Practices and Lessons Learned. Food Sci. Nutr. 2019, 7, 2781–2795. [Google Scholar] [CrossRef]
- FAO. 2022. Available online: https://www.fao.org/3/i3396e/i3396e.pdf (accessed on 3 January 2022).
- Kamal, H.; Habib, H.M.; Ali, A.; Show, P.L.; Koyande, A.K.; Kheadr, E.; Ibrahim, W.H. Food Waste Valorization Potential: Fiber, Sugar, and Color Profiles of 18 Date Seed Varieties (Phoenix dactylifera, L.). J. Saudi Soc. Agric. Sci. 2022, 22, 133–138. [Google Scholar] [CrossRef]
- Oladzad, S.; Fallah, N.; Mahboubi, A.; Afsham, N.; Taherzadeh, M.J. Date Fruit Processing Waste and Approaches to Its Valorization: A Review. Bioresour. Technol. 2021, 340, 125625. [Google Scholar] [CrossRef]
- Taghian Dinani, S.; van der Goot, A.J. Challenges and Solutions of Extracting Value-Added Ingredients from Fruit and Vegetable by-Products: A Review. Crit. Rev. Food Sci. Nutr. 2022, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Mandal, D.D.; Singh, G.; Majumdar, S.; Chanda, P. Challenges in Developing Strategies for the Valorization of Lignin—A Major Pollutant of the Paper Mill Industry. Environ. Sci. Pollut. Res. 2022, 30, 11119–11140. [Google Scholar] [CrossRef] [PubMed]
- Devaux, A.; Goffart, J.-P.; Kromann, P.; Andrade-Piedra, J.; Polar, V.; Hareau, G. The Potato of the Future: Opportunities and Challenges in Sustainable Agri-Food Systems. Potato Res. 2021, 64, 681–720. [Google Scholar] [CrossRef] [PubMed]
- Silva Pereira Basílio, L.; Vanz Borges, C.; Forlan Vargas, P. Potential of Colored Sweet Potato Genotypes as Source of Bioactive Compounds; Asociación Iberoamericana de Tecnología: Postchosecha, Mexico, 2020; Volume 21. [Google Scholar]
- Tunio, M.H.; Gao, J.; Shaikh, S.A.; Lakhiar, I.A.; Qureshi, W.A.; Solangi, K.A.; Chandio, F.A. Potato Production in Aeroponics: An Emerging Food Growing System in Sustainable Agriculture Forfood Security. Chil. J. Agric. Res. 2020, 80, 118–132. [Google Scholar] [CrossRef]
- Akinlolu-Ojo, T.; Nwanna, E.E.; Badejo, A.A. Physicochemical Constituents and Anti-Oxidative Properties of Ripening Hog Plum (Spondias Mombin) Fruits and the Quality Attributes of Jam Produced from the Fruits. Meas. Food 2022, 7, 100037. [Google Scholar] [CrossRef]
- Pérez-Herrera, A.; Martínez-Gutiérrez, G.A.; León-Martínez, F.M.; Sánchez-Medina, M.A. The Effect of the Presence of Seeds on the Nutraceutical, Sensory and Rheological Properties of Physalis Spp. Fruits Jam: A Comparative Analysis. Food Chem. 2020, 302, 125141. [Google Scholar] [CrossRef]
- Alqahtani, N.K.; Alnemr, T.M.; Ahmed, A.R.; Ali, S. Effect of Inclusion of Date Press Cake on Texture, Color, Sensory, Microstructure, and Functional Properties of Date Jam. Processes 2022, 10, 2442. [Google Scholar] [CrossRef]
- Jati, I.R.A.P.; Darmoatmodjo, L.M.Y.D.; Suseno, T.I.P.; Ristiarini, S.; Wibowo, C. Effect of Processing on Bioactive Compounds, Antioxidant Activity, Physicochemical, and Sensory Properties of Orange Sweet Potato, Red Rice, and Their Application for Flake Products. Plants 2022, 11, 440. [Google Scholar] [CrossRef]
- AOAC Association of Official Analytical Chemists. Official Methods of Analysis, 19th ed.; AOAC: Arlington, VA, USA, 2012; Volume 2. [Google Scholar]
- Habib, H.M.; Ibrahim, W.H. Nutritional Quality of 18 Date Fruit Varieties. Int. J. Food Sci. Nutr. 2011, 62, 544–551. [Google Scholar] [CrossRef]
- Habib, H.M.; Al Meqbali, F.T.; Kamal, H.; Souka, U.D.; Ibrahim, W.H. Physicochemical and Biochemical Properties of Honeys from Arid Regions. Food Chem. 2014, 153, 35–43. [Google Scholar] [CrossRef]
- Habib, H.M.; El-Fakharany, E.M.; Kheadr, E.; Ibrahim, W.H. Grape Seed Proanthocyanidin Extract Inhibits DNA and Protein Damage and Labile Iron, Enzyme, and Cancer Cell Activities. Sci. Rep. 2022, 12, 12393. [Google Scholar] [CrossRef] [PubMed]
- Habib, H.M.; Theuri, S.W.; Kheadr, E.E.; Mohamed, F.E. Functional, Bioactive, Biochemical, and Physicochemical Properties of the Dolichos Lablab Bean. Food Funct. 2017, 8, 872–880. [Google Scholar] [CrossRef] [PubMed]
- Habib, H.M.; El-Fakharany, E.M.; Souka, U.D.; Elsebaee, F.M.; El-Ziney, M.G.; Ibrahim, W.H. Polyphenol-Rich Date Palm Fruit Seed (Phoenix dactylifera L.) Extract Inhibits Labile Iron, Enzyme, and Cancer Cell Activities, and DNA and Protein Damage. Nutrients 2022, 2022, 3536. [Google Scholar] [CrossRef] [PubMed]
- Habib, H.M.; Theuri, S.W.; Kheadr, E.E.; Mohamed, F.E. DNA and BSA Damage Inhibitory Activities, and Anti-Acetylcholinesterase, Anti-Porcine α-Amylase and Antioxidant Properties of Dolichos Lablab Beans. Food Funct. 2017, 8, 881–887. [Google Scholar] [CrossRef] [PubMed]
- Pinto, D.; Moreira, M.M.; Vieira, E.F.; Švarc-Gajić, J.; Vallverdú-Queralt, A.; Brezo-Borjan, T.; Delerue-Matos, C.; Rodrigues, F. Development and Characterization of Functional Cookies Enriched with Chestnut Shells Extract as Source of Bioactive Phenolic Compounds. Foods 2023, 12, 640. [Google Scholar] [CrossRef] [PubMed]
- Kavaya, R.I.; Omwamba, M.N.; Chikamai, B.N.; Mahungu, S.M. Sensory Evaluation of Syneresis Reduced Jam and Marmalade Containing Gum Arabic from Acacia senegal var. kerensis. Food Nutr. Sci. 2019, 10, 1334–1343. [Google Scholar] [CrossRef]
- Amagloh, F.C.; Kaaya, A.N.; Yada, B.; Chelangat, D.M.; Katungisa, A.; Amagloh, F.K.; Tumuhimbise, G.A. Bioactive Compounds and Antioxidant Activities in Peeled and Unpeeled Sweetpotato Roots of Different Varieties and Clones in Uganda. Future Foods 2022, 6, 100183. [Google Scholar] [CrossRef]
- Pazos, J.; Zema, P.; Corbino, G.B.; Gabilondo, J.; Borioni, R.; Malec, L.S. Growing Location and Root Maturity Impact on the Phenolic Compounds, Antioxidant Activity and Nutritional Profile of Different Sweet Potato Genotypes. Food Chem. Mol. Sci. 2022, 5, 100125. [Google Scholar] [CrossRef]
- Baah, R.O.; Duodu, K.G.; Emmambux, M.N. Cooking Quality, Nutritional and Antioxidant Properties of Gluten-Free Maize—Orange-Fleshed Sweet Potato Pasta Produced by Extrusion. LWT 2022, 162, 113415. [Google Scholar] [CrossRef]
- Gabilondo, J.; Corbino, G.; Chludil, H.; Malec, L. Bioactive Compounds of Two Orange-Fleshed Sweet Potato Cultivars (Ipomoea Batatas (L.) Lam.) in Fresh, Stored and Processed Roots. Appl. Food Res. 2022, 2, 100061. [Google Scholar] [CrossRef]
- Rambabu, K.; Bharath, G.; Hai, A.; Banat, F.; Hasan, S.W.; Taher, H.; Mohd Zaid, H.F. Nutritional Quality and Physico-Chemical Characteristics of Selected Date Fruit Varieties of the United Arab Emirates. Processes 2020, 8, 256. [Google Scholar] [CrossRef]
- Aleid, S.M.; Hassan, B.H.; Almaiman, S.A.; Al-Kahtani, S.H.; Ismail, S.M. Microbial Loads and Physicochemical Characteristics of Fruits from Four Saudi Date Palm Tree Cultivars: Conformity with Applicable Date Standards. Food Nutr. Sci. 2014, 5, 316–327. [Google Scholar] [CrossRef]
- Derouich, M.; Meziani, R.; Bourkhis, B.; Filali-Zegzouti, Y.; Alem, C. Nutritional, Mineral and Organic Acid Composition of Syrups Produced from Six Moroccan Date Fruit (Phoenix dactylifera L.) Varieties. J. Food Compos. Anal. 2020, 93, 103591. [Google Scholar] [CrossRef]
- Alem, C.; Ennassir, J.; Benlyas, M.; Mbark, A.N.; Zegzouti, Y.F. Phytochemical Compositions and Antioxidant Capacity of Three Date (Phoenix dactylifera L.) Seeds Varieties Grown in the South East Morocco. J. Saudi Soc. Agric. Sci. 2017, 16, 350–357. [Google Scholar] [CrossRef]
- Hmidani, A.; Bourkhis, B.; Khouya, T.; Ramchoun, M.; Filali-Zegzouti, Y.; Alem, C. Phenolic Profile and Anti-Inflammatory Activity of Four Moroccan Date (Phoenix dactylifera L.) Seed Varieties. Heliyon 2020, 6, e03436. [Google Scholar] [CrossRef]
- Sanoussi, A.F.; Adjatin, A.; Dansi, A.; Adebowale, A.; Sanni, L.O.; Sanni, A. Mineral Composition of Ten Elites Sweet Potato (Ipomoea Batatas [L] Lam) Landraces of Benin. Int. J. Curr. Microbiol. Appl. Sci. 2016, 5, 103–115. [Google Scholar] [CrossRef]
- Franková, H.; Musilová, J.; Árvay, J.; Šnirc, M.; Jančo, I.; Lidiková, J.; Vollmannová, A. Changes in Antioxidant Properties and Phenolics in Sweet Potatoes (Ipomoea batatas L.) Due to Heat Treatments. Molecules 2022, 27, 1884. [Google Scholar] [CrossRef]
- Al-Mssallem, M.Q.; Alqurashi, R.M.; Al-Khayri, J.M. Bioactive Compounds of Date Palm (Phoenix dactylifera L.). In Bioactive Compounds in Underutilized Fruits and Nuts; Springer: Berlin/Heidelberg, Germany, 2019; pp. 1–15. [Google Scholar]
- Al-Shwyeh, H. Date Palm (Phoenix dactylifera L.) Fruit as Potential Antioxidant and Antimicrobial Agents. J. Pharm. Bioallied Sci. 2019, 11, 1. [Google Scholar] [CrossRef]
- Chaari, A.; Abdellatif, B.; Nabi, F.; Khan, R.H. Date Palm (Phoenix dactylifera L.) Fruit’s Polyphenols as Potential Inhibitors for Human Amylin Fibril Formation and Toxicity in Type 2 Diabetes. Int. J. Biol. Macromol. 2020, 164, 1794–1808. [Google Scholar] [CrossRef]
- Kim, M.Y.; Lee, B.W.; Lee, H.; Lee, Y.Y.; Kim, M.H.; Lee, J.Y.; Lee, B.K.; Woo, K.S.; Kim, H. Phenolic Compounds and Antioxidant Activity in Sweet Potato after Heat Treatment. J. Sci. Food Agric. 2019, 99, 6833–6840. [Google Scholar] [CrossRef]
- Kwatra, B. A review on potential properties and therapeutic applications of grape seed extract. Artic. World J. Pharm. Res. 2020, 9, 2519. [Google Scholar] [CrossRef]
- Manca, M.L.; Casula, E.; Marongiu, F.; Bacchetta, G.; Sarais, G.; Zaru, M.; Escribano-Ferrer, E.; Peris, J.E.; Usach, I.; Fais, S.; et al. From Waste to Health: Sustainable Exploitation of Grape Pomace Seed Extract to Manufacture Antioxidant, Regenerative and Prebiotic Nanovesicles within Circular Economy. Sci. Rep. 2020, 10, 14184. [Google Scholar] [CrossRef] [PubMed]
- Gülcü, M.; Uslu, N.; Özcan, M.M.; Gökmen, F.; Özcan, M.M.; Banjanin, T.; Gezgin, S.; Dursun, N.; Geçgel, Ü.; Ceylan, D.A.; et al. The Investigation of Bioactive Compounds of Wine, Grape Juice and Boiled Grape Juice Wastes. J. Food Process Preserv. 2019, 43, e13850. [Google Scholar] [CrossRef]
- Sridhar, K.; Charles, A.L. In Vitro Antioxidant Activity of Kyoho Grape Extracts in DPPH and ABTS Assays: Estimation Methods for EC50 Using Advanced Statistical Programs. Food Chem. 2019, 275, 41–49. [Google Scholar] [CrossRef]
- Rousserie, P.; Lacampagne, S.; Vanbrabant, S.; Rabot, A.; Geny-Denis, L. Influence of Berry Ripeness on Seed Tannins Extraction in Wine. Food Chem. 2020, 315, 126307. [Google Scholar] [CrossRef] [PubMed]
- Mohd Naeem, M.N.; Mohd Fairulnizal, M.N.; Norhayati, M.K.; Zaiton, A.; Norliza, A.H.; Wan Syuriahti, W.Z.; Mohd Azerulazree, J.; Aswir, A.R.; Rusidah, S. The Nutritional Composition of Fruit Jams in the Malaysian Market. J. Saudi Soc. Agric. Sci. 2017, 16, 89–96. [Google Scholar] [CrossRef]
- Chalchisa, T.; Zegeye, A.; Dereje, B.; Tolesa, Y. Effect of Sugar, Pectin, and Processing Temperature on the Qualities of Pineapple Jam. Int. J. Fruit Sci. 2022, 22, 711–724. [Google Scholar] [CrossRef]
- Bikila, A.M.; Tola, Y.B.; Esho, T.B.; Forsido, S.F. Anchote (Coccinia abyssinica [Lam.] Cogn.) Powder, an Underutilized Indigenous Crop, as a Substitute to Commercial Pectin in the Production of Strawberry Jam. Heliyon 2022, 8, e10700. [Google Scholar] [CrossRef]
- Teixeira, F.; dos Santos, B.A.; Nunes, G.; Soares, J.M.; do Amaral, L.A.; de Souza, G.H.O.; de Resende, J.T.V.; Menegassi, B.; Rafacho, B.P.M.; Schwarz, K.; et al. Addition of Orange Peel in Orange Jam: Evaluation of Sensory, Physicochemical, and Nutritional Characteristics. Molecules 2020, 25, 1670. [Google Scholar] [CrossRef]
- Aghajanzadeh, S.; Ziaiifar, A.M.; Verkerk, R. Effect of Thermal and Non-Thermal Treatments on the Color of Citrus Juice: A Review. Food Rev. Int. 2021, 1–23. [Google Scholar] [CrossRef]



| Ingredients/100 g Jam | Khalas Date Fruit | Control Treatments | ||||
|---|---|---|---|---|---|---|
| DP1 | DP2 | DP3 | DP4 | DFJ | SPJ | |
| Date fruit | 40% | 35% | 30% | 25% | 50% | 0.0% |
| Sweet potato | 10% | 15% | 20% | 25% | 0.0% | 50% |
| White sugar, sucrose | 11% | 13.5% | 16.5% | 17.5% | 20% | 65% |
| Citric acid | 0.030% | 0.030% | 0.030% | 0.030% | 0.030% | 0.030% |
| Ascorbic acid | 0.035% | 0.035% | 0.035% | 0.035% | 0.035% | 0.035% |
| Sterile distilled water | 50% | 50% | 50% | 50% | 50% | 50% |
| Total water loss | 11% | 13.5% | 16.5% | 17.5% | 20% | 65% |
| Total | 100.065 | 100.065 | 100.065 | 100.065 | 100.065 | 100.065 |
| Contents | Date Fruit | Sweet Potato |
|---|---|---|
| (g/100 g wt.) | ||
| Moisture % | 22.07 ± 0.18 | 71.17 ± 0.22 |
| TS % | 77.93 ± 1.01 | 28.83 ± 0.37 |
| Carbohydrate % | 66.51 ± 0.28 | 20.57 ± 0.16 |
| Protein % | 2.35 ± 0.28 | 3.64 ± 0.24 |
| Fat % | 0.16 ± 0.02 | 0.36 ± 0.01 |
| Total fiber % | 7.35 ± 0.40 | 2.81 ± 0.73 |
| Ash % | 1.56 ± 0.11 | 1.44 ± 0.41 |
| pH | 5.83 ± 0.07 | 7.17 ± 0.05 |
| a* | 13.75 ± 0.13 | −1.08 ± 0.81 |
| b* | 20.23 ± 0.11 | 18.54 ± 0.99 |
| L* | 26.57 ± 0.08 | 79.17 ± 1.35 |
| °h | 55.70 ± 027 | 93.30 ± 2.04 |
| C* | 24.50 ± 0.78 | 18.50 ± 0.69 |
| Minerals | Date Fruit | Sweet Potato |
|---|---|---|
| (mg/100 g) | ||
| Na | 8.71 ± 0.31 | 5.33 ± 0.41 |
| K | 402.32 ± 3.22 | 76.34 ± 1.02 |
| Ca | 39.82 ± 0.68 | 84.23 ± 0.83 |
| P | 57.37 ± 0.74 | 27.18 ± 0.48 |
| Mg | 53.23 ± 0.48 | 0.18 ± 0.03 |
| Zn | 1.43 ± 0.08 | 0.07 ± 0.01 |
| Fe | 1.84 ± 0.12 | 0.95 ± 0.02 |
| Contents | Date Fruit | Sweet Potato |
|---|---|---|
| (mg/100 g) | ||
| Total phenolics (mg of gallic acid equivalent (GAE)/100 g) | 387.54 ± 2.23 | 187.67 ± 0.13 |
| Total flavonoids (mg of rutin equivalent/100 g) | 74.46 ± 0.71 | 68.57 ± 0.24 |
| Gallic acid | 16.22 ± 0.26 | 2.08 ± 0.14 |
| Syringic acid | 9.39 ± 0.17 | ND |
| Coumaric acid | 6.74 ± 0.21 | 1.39 ± 0.41 |
| Caffeic acid | 13.96 ± 0.52 | 1.48 ±0.36 |
| Ferulic acid | 18.02 ± 0.37 | 28.06 ±0.72 |
| Catechin | 4.89 ± 0.34 | ND |
| Epicatechin | 6.56 ± 0.19 | 0.34 ± 0.01 |
| β-Carotene | 0.39 ± 0.02 | 518.94 ± 0.64 |
| FRAP (mmol ferrous equivalents/100 g) | 907.12 ± 2.74 | 1024.64 ± 6.24 |
| DPPH (inhibition %) | 5.27 ± 0.42 | 78.31 ± 0.75 |
| ABTS (inhibition %) | 67.51 ± 1.08 | 82.47 ± 2.36 |
| Superoxide (inhibition %) | 19.19 ± 0.74 | 42.14 ± 0.86 |
| Physicochemical Properties of Jams | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Jams | Moisture % | TS % | TSS (°Brix) % | Carbohydrate % | Protein % | Fat % | Total Fiber % | Ash % | pH |
| DFJ 100% | 25.89 ± 3.52 a | 74.11 ± 3.52 b | 64.00 ± 0.70 b | 67.00 ± 0.26 c | 1.30 ± 0.03 d | 0.12 ± 0.02 a | 4.62 ± 0.20 e | 1.78 ± 0.04 d | 3.19 ± 0.04 a |
| SPJ 100% | 28.87 ± 0.18 ab | 71.13 ± 0.18 ab | 66.00 ± 0.27 c | 69.00 ± 0.17 d | 1.59 ± 0.40 e | 0.23 ± 0.02 c | 1.62 ± 0.05 a | 1.91 ± 0.03 e | 3.18 ± 0.08 a |
| DP1 80:20 | 33.16 ± 1.58 b | 65.84 ± 1.58 a | 64.00 ± 0.22 b | 71.83 ± 0.40 e | 0.96 ± 0.02 c | 0.15 ± 0.01 ab | 3.62 ± 0.17 d | 1.41 ± 0.03 c | 3.61 ± 0.05 b |
| DP2 70:30 | 31.46 ± 0.88 b | 68.54 ± 0.88 a | 62.00 ± 0.41 a | 68.53 ± 0.19 d | 0.79 ± 0.03 b | 0.16 ± 0.02 ab | 3.12 ± 0.15 c | 1.22 ± 0.02 b | 3.77 ± 0.06 c |
| DP3 60:40 | 32.77 ± 2.84 b | 67.23 ± 2.84 a | 63.00 ± 0.19 ab | 65.93 ± 0.24 b | 0.62 ± 0.01 a | 0.17 ± 0.01 b | 2.62 ± 0.13 b | 1.03 ± 0.03 a | 3.96 ± 0.04 d |
| DP4 50:50 | 34.34 ± 3.52 b | 65.66 ± 0.78 a | 63.00 ± 0.50 ab | 60.63 ± 0.71 a | 0.95 ± 0.01 c | 0.18 ± 0.01 b | 2.62 ± 0.12 b | 1.35 ± 0.03 c | 3.98 ± 0.03 d |
| Bioactive Compounds mg/100 g of Jams | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Jams | TPC | TFC | Gallic Acid | Syringic Acid | Coumaric Acid | Caffeic Acid | Ferulic Acid | Catechin | Epicatechin | β-Carotene |
| DFJ 100% | 232.74 ± 1.25 f | 44.68 ± 0.48 e | 8.90 ± 0.16 f | 5.07 ± 0.13 e | 3.64 ± 0.12 e | 7.81 ± 0.18 f | 10.61 ± 0.37 a | 2.40 ± 0.39 d | 4.54 ± 0.21 e | 0.17 ± 0.00 a |
| SPJ 100% | 114.05 ± 1.87 a | 38.80 ± 0.45 a | 1.15 ± 0.07 a | ND a | 0.90 ± 0.03 a | 0.83 ± 0.05 a | 15.33 ± 0.36 d | ND a | 0.18 ± 0.00 a | 310.77 ± 1.68 f |
| DP1 80:20 | 208.60 ± 1.37 e | 43.11 ± 0.32 d | 6.95 ± 0.11 e | 3.66 ± 0.10 d | 2.69 ± 0.09 d | 6.01 ± 0.14 e | 11.15 ± 0.23 ab | 1.77 ± 0.19 c | 3.27 ± 0.17 d | 61.89 ± 0.34 b |
| DP2 70:30 | 196.53 ± 1.43 d | 42.32 ± 0.25 cd | 5.98 ± 0.09 d | 2.95 ± 0.09 c | 2.22 ± 0.08 c | 5.11 ± 0.11 d | 11.42 ± 0.16 b | 1.08 ± 0.27 b | 2.63 ± 0.15 c | 92.75 ± 0.50 c |
| DP3 60:40 | 184.46 ± 1.49 c | 41.53 ± 0.19 bc | 5.00 ± 0.07 c | 2.24 ± 0.08 b | 1.74 ± 0.07 b | 4.22 ± 0.09 c | 11.69 ± 0.09 b | 0.77 ± 0.10 b | 2.00 ± 0.12 b | 123.61 ± 0.67 d |
| DP4 50:50 | 172.89 ± 1.56 b | 41.24 ± 0.16 b | 4.53 ± 0.04 b | 2.04 ± 0.07 b | 1.77 ± 0.05 b | 3.82 ± 0.07 b | 12.47 ± 0.05 c | 0.80 ± 0.08 b | 1.86 ± 0.10 b | 154.97 ± 0.84 e |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Almaghlouth, B.J.; Alqahtani, N.K.; Alnabbat, K.I.; Mohamed, H.A.; Alnemr, T.M.; Habib, H.M. Valorization of Second-Grade Date Fruit Byproducts and Nonstandard Sweet Potato Tubers to Produce Novel Biofortified Functional Jam. Foods 2023, 12, 1906. https://doi.org/10.3390/foods12091906
Almaghlouth BJ, Alqahtani NK, Alnabbat KI, Mohamed HA, Alnemr TM, Habib HM. Valorization of Second-Grade Date Fruit Byproducts and Nonstandard Sweet Potato Tubers to Produce Novel Biofortified Functional Jam. Foods. 2023; 12(9):1906. https://doi.org/10.3390/foods12091906
Chicago/Turabian StyleAlmaghlouth, Bayan J., Nashi K. Alqahtani, Khadijah I. Alnabbat, Hisham A. Mohamed, Tareq M. Alnemr, and Hosam M. Habib. 2023. "Valorization of Second-Grade Date Fruit Byproducts and Nonstandard Sweet Potato Tubers to Produce Novel Biofortified Functional Jam" Foods 12, no. 9: 1906. https://doi.org/10.3390/foods12091906
APA StyleAlmaghlouth, B. J., Alqahtani, N. K., Alnabbat, K. I., Mohamed, H. A., Alnemr, T. M., & Habib, H. M. (2023). Valorization of Second-Grade Date Fruit Byproducts and Nonstandard Sweet Potato Tubers to Produce Novel Biofortified Functional Jam. Foods, 12(9), 1906. https://doi.org/10.3390/foods12091906









