Microencapsulation of Carotenoid-Rich Extract from Guaraná Peels and Study of Microparticle Functionality through Incorporation into an Oatmeal Paste
Abstract
1. Introduction
2. Material and Methods
2.1. Materials
2.2. Carotenoid-Rich Extract Preparation
2.3. Microencapsulation by Spray Drying
2.4. Total Carotenoid Content
2.5. Stability Study of Microparticles
2.5.1. Carotenoid Degradation Kinetics
2.5.2. Color
2.5.3. Mean Diameter
2.5.4. Moisture Content and Water Activity
2.6. Dynamic Vapor Sorption (DVS) of Microparticles and Oat Flakes
2.7. Thermal Properties of Oat Flakes and Oat Flakes Containing Microparticles
2.8. Pasting Properties
2.9. Retention of β-Carotene, Lutein, and Total Carotenoid Content in the Oatmeal Paste
2.10. Statistics
3. Results and Discussion
3.1. Total Carotenoid Content of Microparticles
3.2. Stability Study of Microparticles during Storage
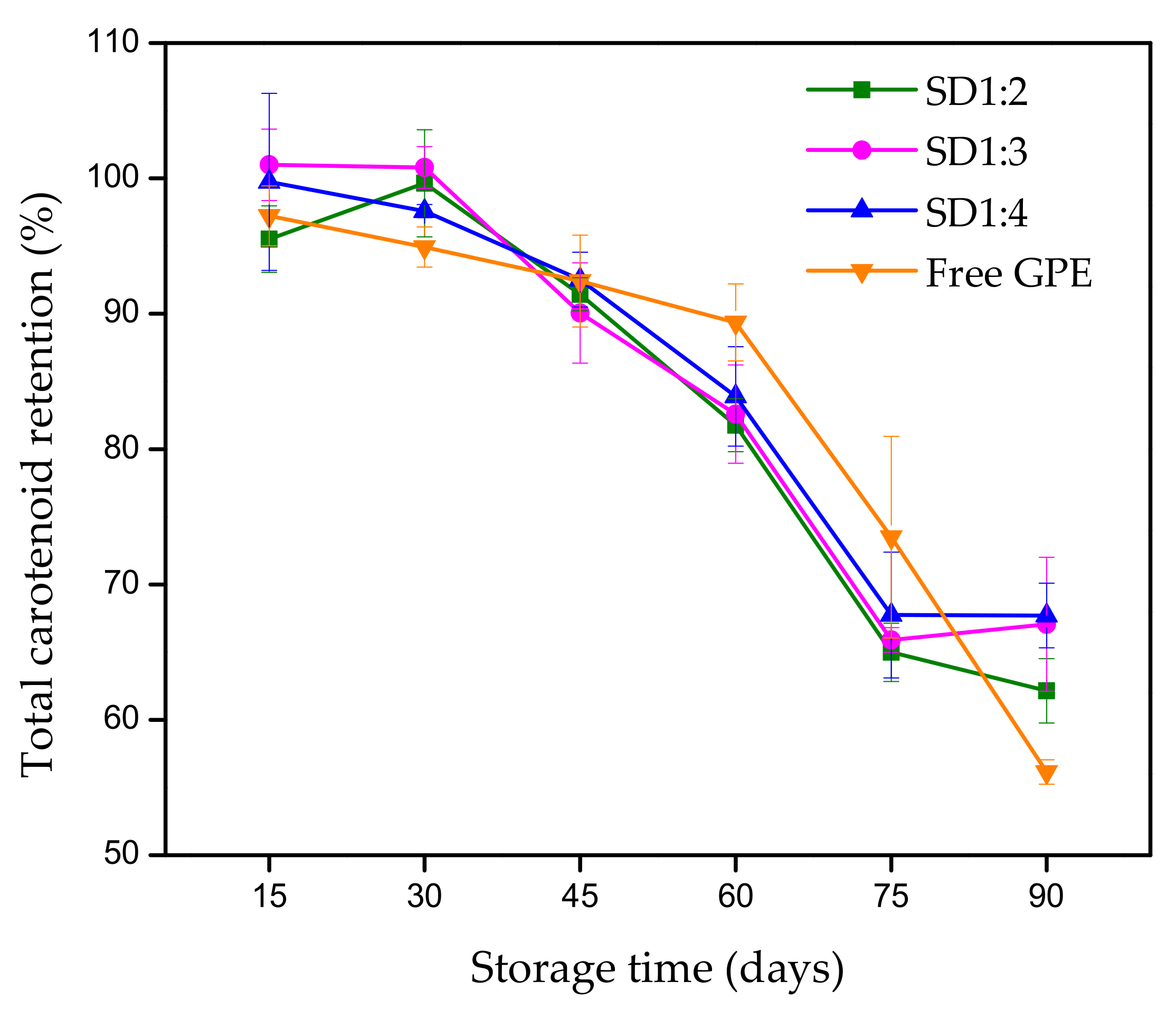
3.2.1. Carotenoid Retention
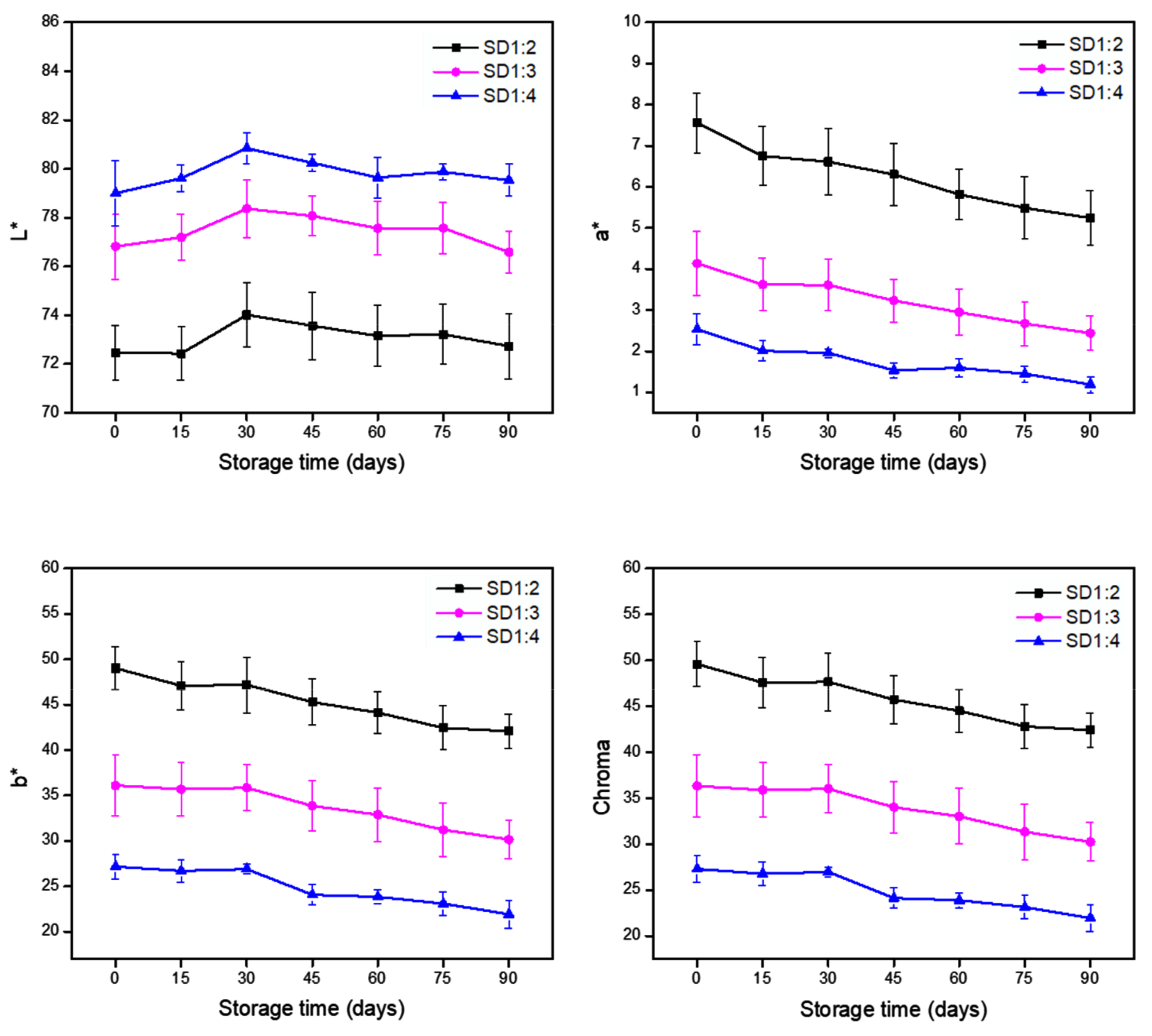
3.2.2. Color Parameters
3.2.3. Mean Particle Diameter
3.2.4. Moisture Content and aw
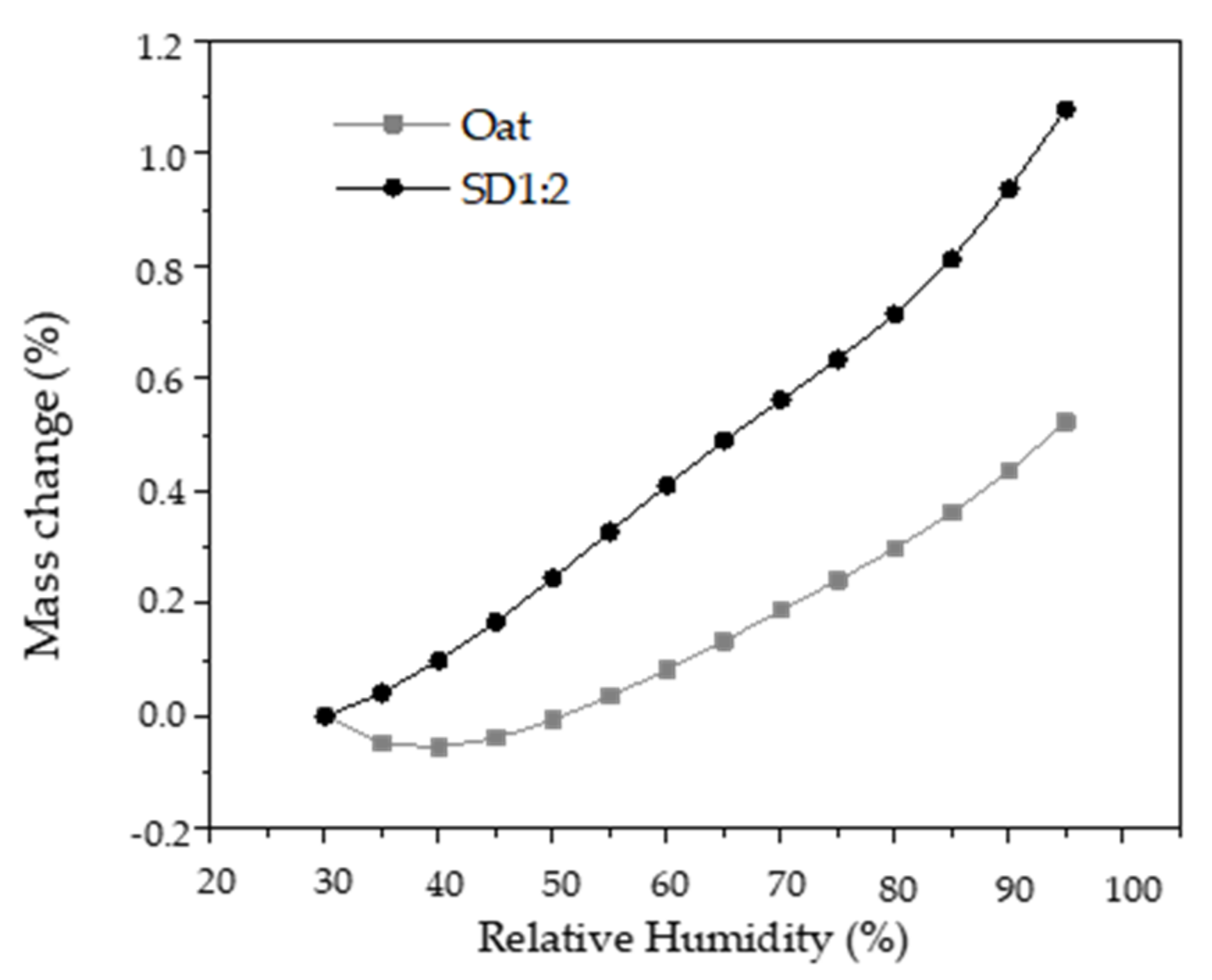
3.3. Dynamic Vapor Sorption (DVS) of Oat Flakes and Microparticles
3.4. Thermal Properties
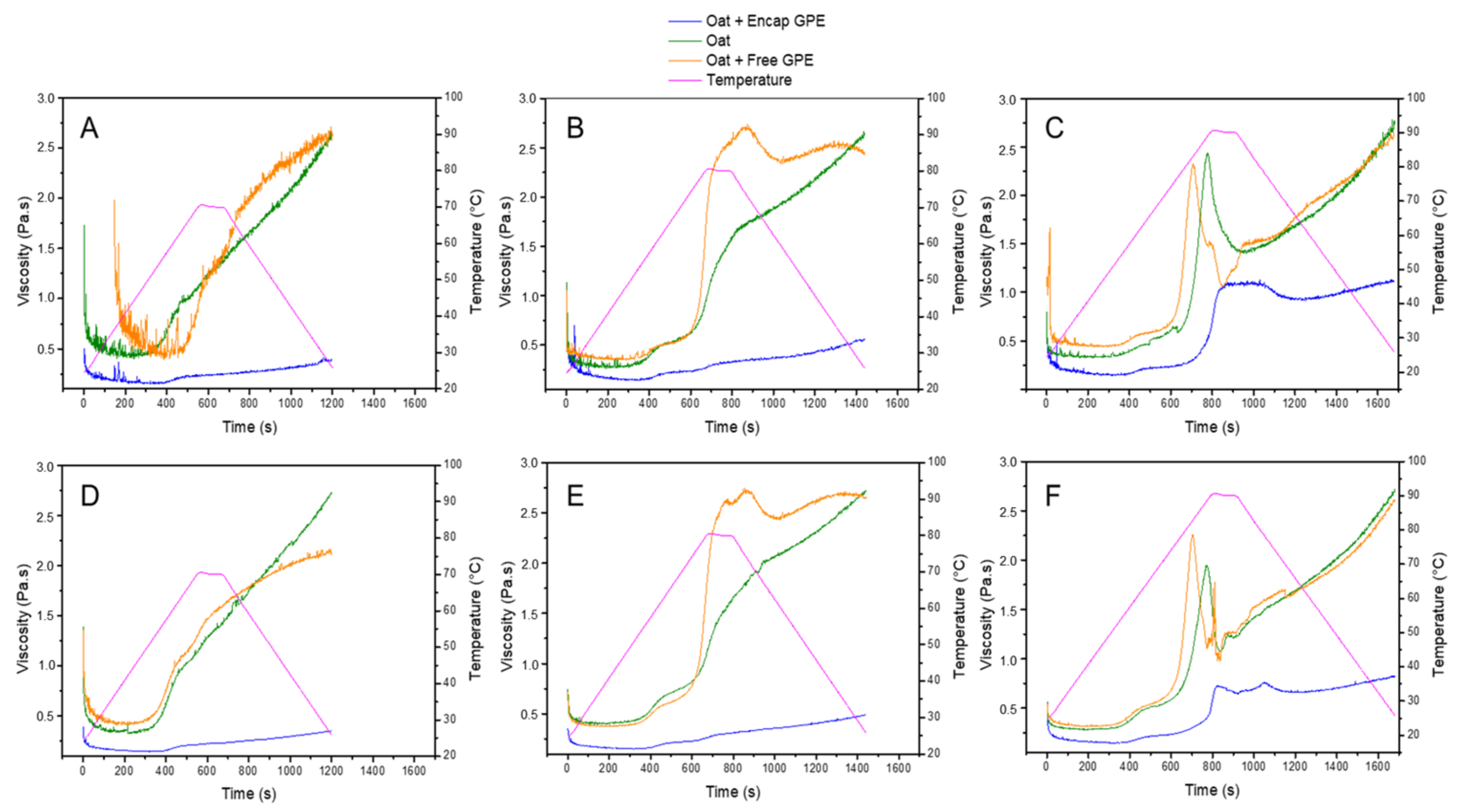
3.5. Pasting Properties
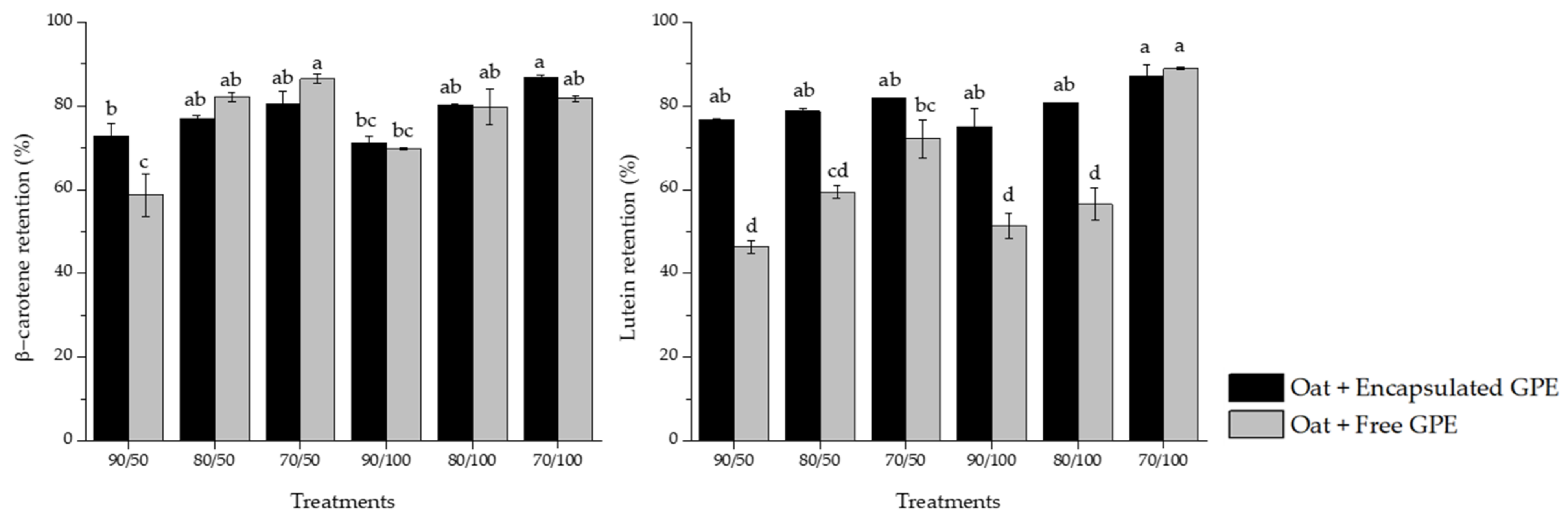
3.6. Carotenoid Retention
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Micozzi, M.S.; Beecher, G.R.; Taylor, P.R.; Khachik, F. Carotenoid analyses of selected raw and cooked foods associated with a lower risk for cancer. JNCI J. Natl. Cancer Inst. 1990, 82, 282–285. [Google Scholar] [CrossRef]
- Toma, S.; Losardo, P.L.; Vincent, M.; Palumbo, R. Effectiveness of beta-carotene in cancer chemoprevention. Eur. J. Cancer Prev. 1995, 4, 213–224. [Google Scholar] [CrossRef]
- Kumar, S.R.; Hosokawa, M.; Miyashita, K. Fucoxanthin: A marine carotenoid exerting anti-cancer effects by affecting multiple mechanisms. Mar. Drugs 2013, 11, 5130–5147. [Google Scholar] [CrossRef]
- Riccioni, G. Carotenoids and cardiovascular disease. Curr. Atheroscler. Rep. 2009, 11, 434–439. [Google Scholar] [CrossRef]
- Monroy-Ruiz, J.; Sevilla, M.Á.; Carrón, R.; Montero, M.J. Astaxanthin-enriched-diet reduces blood pressure and improves cardiovascular parameters in spontaneously hypertensive rats. Pharmacol. Res. 2011, 63, 44–50. [Google Scholar] [CrossRef]
- Gale, C.R.; Hall, N.F.; Phillips, D.I.; Martyn, C.N. Lutein and zeaxanthin status and risk of age-related macular degeneration. Investig. Ophthalmol. Vis. Sci. 2003, 44, 2461–2465. [Google Scholar] [CrossRef]
- Stahl, W.; Sies, H. Antioxidant activity of carotenoids. Mol. Asp. Med. 2003, 24, 345–351. [Google Scholar] [CrossRef]
- Olson, J.A. Provitamin A function of carotenoids: The conversion of β-carotene into vitamin A. J. Nutr. 1989, 119, 105–108. [Google Scholar] [CrossRef]
- Chen, B.H.; Tang, Y.C. Processing and stability of carotenoid powder from carrot pulp waste. J. Agric. Food Chem. 1998, 46, 2312–2318. [Google Scholar] [CrossRef]
- Ajila, C.M.; Bhat, S.G.; Rao, U.P. Valuable components of raw and ripe peels from two Indian mango varieties. Food Chem. 2007, 102, 1006–1011. [Google Scholar] [CrossRef]
- De Andrade Lima, M.; Kestekoglou, I.; Charalampopoulos, D.; Chatzifragkou, A. Supercritical fluid extraction of carotenoids from vegetable waste matrices. Molecules 2019, 24, 466. [Google Scholar] [CrossRef]
- De Souza Mesquita, L.M.; Martins, M.; Maricato, É.; Nunes, C.; Quinteiro, P.S.; Dias, A.C.; Coutinho, J.A.; Pisani, L.P.; de Rosso, V.V.; Ventura, S.P. Ionic liquid-mediated recovery of carotenoids from the Bactris gasipaes fruit waste and their application in food-packaging chitosan films. ACS Sustain. Chem. Eng. 2020, 8, 4085–4095. [Google Scholar] [CrossRef]
- Caris-Veyrat, C.; Schmid, A.; Carail, M.; Böhm, V. Cleavage products of lycopene produced by in vitro oxidations: Characterization and mechanisms of formation. J. Agric. Food Chem. 2003, 51, 7318–7325. [Google Scholar] [CrossRef]
- Boon, C.S.; McClements, D.J.; Weiss, J.; Decker, E.A. Factors influencing the chemical stability of carotenoids in foods. Crit. Rev. Food Sci. Nutr. 2010, 50, 515–532. [Google Scholar] [CrossRef]
- Xiao, Y.D.; Huang, W.Y.; Li, D.J.; Song, J.F.; Liu, C.Q.; Wei, Q.Y.; Zhang, M.; Yang, Q.M. Thermal degradation kinetics of all-trans and cis-carotenoids in a light-induced model system. Food Chem. 2018, 239, 360–368. [Google Scholar] [CrossRef]
- Sagar, V.R.; Kumar, P.S. Recent advances in drying and dehydration of fruits and vegetables: A review. J. Food Sci. Technol. 2010, 47, 15–26. [Google Scholar] [CrossRef]
- Subtil, S.F.; Rocha-Selmi, G.A.; Thomazini, M.; Trindade, M.A.; Netto, F.M.; Favaro-Trindade, C.S. Effect of spray drying on the sensory and physical properties of hydrolysed casein using gum arabic as the carrier. J. Food Sci. Technol. 2014, 51, 2014–2021. [Google Scholar] [CrossRef]
- Tuyen, C.K.; Nguyen, M.H.; Roach, P.D. Effects of spray drying conditions on the physicochemical and antioxidant properties of the Gac (Momordica cochinchinensis) fruit aril powder. J. Food Eng. 2010, 98, 385–392. [Google Scholar] [CrossRef]
- Hojjati, M.; Razavi, S.H.; Rezaei, K.; Gilani, K. Spray drying microencapsulation of natural canthaxantin using soluble soybean polysaccharide as a carrier. Food Sci. Biotechnol. 2011, 20, 63–69. [Google Scholar] [CrossRef]
- Etzbach, L.; Meinert, M.; Faber, T.; Klein, C.; Schieber, A.; Weber, F. Effects of carrier agents on powder properties, stability of carotenoids, and encapsulation efficiency of goldenberry (Physalis peruviana L.) powder produced by co-current spray drying. Curr. Res. Food Sci. 2020, 3, 73–81. [Google Scholar] [CrossRef]
- Ai, Y.; Jane, J.L. Gelatinization and rheological properties of starch. Starch-Stärke 2015, 67, 213–224. [Google Scholar] [CrossRef]
- Wang, J.; Xia, S.; Wang, B.; Ali, F.; Li, X. Effect of twin-screw extrusion on gelatinization characteristics of oat powder. J. Food Process Eng. 2019, 42, e13014. [Google Scholar] [CrossRef]
- Silva, M.P.; Thomazini, M.; Holkem, A.T.; Pinho, L.S.; Genovese, M.I.; Fávaro-Trindade, C.S. Production and characterization of solid lipid microparticles loaded with guaraná (Paullinia cupana) seed extract. Food Res. Int. 2019, 123, 144–152. [Google Scholar] [CrossRef]
- Pinho, L.P.; Silva, M.P.; Thomazini, M.; Cooperstone, J.L.; Campanella, O.; Rodrigues, C.E.C.; Fávaro-Trindade, C.S. Guaraná (Paullinia cupana) by-product as a source of bioactive compounds and as a natural antioxidant for food applications. Food Process. Preserv. 2021, 45, e15854. [Google Scholar] [CrossRef]
- Cuevas, M.S.; Rodrigues, C.E.; Gomes, G.B.; Meirelles, A.J. Vegetable Oils Deacidification by Solvent Extraction: Liquid-Liquid Equilibrium Data for Systems Containing Sunflower Seed Oil at 298.2 K. J. Chem. Eng. Data 2010, 55, 3859–3862. [Google Scholar] [CrossRef]
- Rocha, G.A.; Fávaro-Trindade, C.S.; Grosso, C.R.F. Microencapsulation of lycopene by spray drying: Characterization, stability and application of microcapsules. Food Bioprod. Process. 2012, 90, 37–42. [Google Scholar] [CrossRef]
- Rodriguez-Amaya, D.B. A Guide to Carotenoid Analysis in Foods; ILSI Press: Washington, DC, USA, 2001. [Google Scholar]
- Tonon, R.V.; Brabet, C.; Hubinger, M.D. Anthocyanin stability and antioxidant activity of spray-dried açai (Euterpe oleracea Mart.) juice produced with different carrier agents. Food Res. Int. 2010, 43, 907–914. [Google Scholar] [CrossRef]
- Hidalgo, A.; Brandolini, A. Kinetics of carotenoids degradation during the storage of einkorn (Triticum monococcum L. ssp. monococcum) and bread wheat (Triticum aestivum L. ssp. aestivum) flours. J. Agric. Food Chem. 2008, 56, 11300–11305. [Google Scholar] [CrossRef]
- Song, J.; Wang, X.; Li, D.; Liu, C. Degradation kinetics of carotenoids and visual colour in pumpkin (Cucurbita maxima L.) slices during microwave-vacuum drying. Int. J. Food Prop. 2017, 20 (Suppl. S1), S632–S643. [Google Scholar] [CrossRef]
- Minolta Corporation. Precise Color Communication: Color Control from Feeling to Instrumentation; Minolta Corporation: Ramsey, NJ, USA, 2008; pp. 1–49. [Google Scholar]
- Kopec, R.E.; Cooperstone, J.L.; Schweiggert, R.M.; Young, G.S.; Harrison, E.H.; Francis, D.M.; Clinton, S.K.; Schwartz, S.J. Avocado consumption enhances human postprandial provitamin a absorption and conversion from a novel high-β-carotene tomato sauce and from carrots. J. Nutr. 2014, 144, 1158–1166. [Google Scholar] [CrossRef]
- Cooperstone, J.L.; Ralston, R.A.; Riedl, K.M.; Haufe, T.C.; Schweiggert, R.M.; King, S.A.; Timmers, C.D.; Francis, D.M.; Lesinski, G.B.; Clinton, S.K.; et al. Enhanced bioavailability of lycopene when consumed as cis-isomers from tangerine compared to red tomato juice, a randomized, cross-over clinical trial. Mol. Nutr. Food Res. 2015, 59, 658–669. [Google Scholar] [CrossRef]
- Rascón, M.P.; Beristain, C.I.; García, H.S.; Salgado, M.A. Carotenoid retention and storage stability of spray-dried encapsulated paprika oleoresin using gum Arabic and soy protein isolate as wall materials. LWT-Food Sci. Technol. 2011, 44, 549–557. [Google Scholar] [CrossRef]
- Favaro-Trindade, C.S.; Okuro, P.K.; de Matos, F.E., Jr. Encapsulation via spray. In Handbook of Encapsulation and Controlled Release; CRC Press: Boca Raton, FL, USA, 2015; pp. 71–88. [Google Scholar]
- Walton, D.E.; Mumford, C.J. Spray dried products—Characterization of particle morphology. Chem. Eng. Res. Des. 1999, 77, 21–38. [Google Scholar] [CrossRef]
- Ghosal, S.; Indira, T.N.; Bhattacharya, S. Agglomeration of a model food powder: Effect of maltodextrin and gum Arabic dispersions on flow behavior and compacted mass. J. Food Eng. 2010, 96, 222–228. [Google Scholar] [CrossRef]
- Ezhilarasi, P.N.; Karthik, P.; Chhanwal, N.; Anandharamakrishnan, C. Nanoencapsulation techniques for food bioactive components: A review. Food Bioprocess Technol. 2013, 6, 628–647. [Google Scholar] [CrossRef]
- Chiou, D.; Langrish, T.A.G. Development and characterisation of novel nutraceuticals with spray drying technology. J. Food Eng. 2007, 82, 84–91. [Google Scholar] [CrossRef]
- Labuza, T.P.; Dugan, L.R., Jr. Kinetics of lipid oxidation in foods. Crit. Rev. Food Sci. Nutr. 1971, 2, 355–405. [Google Scholar] [CrossRef]
- Álvarez-Henao, M.V.; Saavedra, N.; Medina, S.; Cartagena, C.J.; Alzate, L.M.; Londoño-Londoño, J. Microencapsulation of lutein by spray-drying: Characterization and stability analyses to promote its use as a functional ingredient. Food Chem. 2018, 256, 181–187. [Google Scholar] [CrossRef]
- Hettrich, K.; Fanter, C. Novel xylan gels prepared from oat spelts. In Macromolecular Symposia; WILEY-VCH Verlag: Weinheim, Germany, 2010; Volume 294, pp. 141–150. [Google Scholar] [CrossRef]
- Al-Muhtaseb, A.H.; McMinn, W.A.M.; Magee, T.R.A. Moisture sorption isotherm characteristics of food products: A review. Food Bioprod. Process. 2002, 80, 118–128. [Google Scholar] [CrossRef]
- Steenberg, E.; Harris, P.J. Adsorption of carboxymethylcellulose, guar gum and starch onto talc, sulphides, oxides and salt type minerals. South Afr. J. Chem. 1984, 37, 85–90. [Google Scholar] [CrossRef]
- Gabas, A.L.; Telis, V.R.N.; Sobral, P.J.A.; Telis-Romero, J. Effect of maltodextrin and arabic gum in water vapor sorption thermodynamic properties of vacuum dried pineapple pulp powder. J. Food Eng. 2007, 82, 246–252. [Google Scholar] [CrossRef]
- Damodaran, S.; Parkin, K.L.; Fennema, O.R. (Eds.) Fennema’s Food Chemistry; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Brett, B.; Figueroa, M.; Sandoval, A.J.; Barreiro, J.A.; Müller, A.J. Moisture sorption characteristics of starchy products: Oat flour and rice flour. Food Biophys. 2009, 4, 151–157. [Google Scholar] [CrossRef]
- Díaz-Calderón, P.; MacNaughtan, B.; Hill, S.; Foster, T.; Enrione, J.; Mitchell, J. Changes in gelatinisation and pasting properties of various starches (wheat, maize and waxy maize) by the addition of bacterial cellulose fibrils. Food Hydrocoll. 2018, 80, 274–280. [Google Scholar] [CrossRef]
- Eliasson, A.C. Differential scanning calorimetry studies on wheat starch—Gluten mixtures: I. Effect of gluten on the gelatinization of wheat starch. J. Cereal Sci. 1983, 1, 199–205. [Google Scholar] [CrossRef]
- Punia, S.; Sandhu, K.S.; Dhull, S.B.; Siroha, A.K.; Purewal, S.S.; Kaur, M.; Kidwai, M.K. Oat starch: Physico-chemical, morphological, rheological characteristics and its applications-A review. Int. J. Biol. Macromol. 2020, 154, 493–498. [Google Scholar] [CrossRef]
- Taghizadeh, A.; Favis, B.D. Effect of high molecular weight plasticizers on the gelatinization of starch under static and shear conditions. Carbohydr. Polym. 2013, 92, 1799–1808. [Google Scholar] [CrossRef]
- Singh, A.; Geveke, D.J.; Yadav, M.P. Improvement of rheological, thermal and functional properties of tapioca starch by using gum arabic. LWT 2017, 80, 155–162. [Google Scholar] [CrossRef]
- Shahzad, S.A.; Hussain, S.; Mohamed, A.A.; Alamri, M.S.; Ibraheem, M.A.; Qasem, A.A.A. Effect of hydrocolloid gums on the pasting, thermal, rheological and textural properties of chickpea starch. Foods 2019, 8, 687. [Google Scholar] [CrossRef]
- Farrag, Y.; Sabando, C.; Rodríguez-Llamazares, S.; Bouza, R.; Rojas, C.; Barral, L. Preparation of donut-shaped starch microparticles by aqueous-alcoholic treatment. Food Chem. 2018, 246, 1–5. [Google Scholar] [CrossRef]
- Sun, Y.; Li, F.; Luan, Y.; Li, P.; Dong, X.; Chen, M.; Dai, L.; Sun, Q. Gelatinization, pasting, and rheological properties of pea starch in alcohol solution. Food Hydrocoll. 2021, 112, 106331. [Google Scholar] [CrossRef]
- Dhuique-Mayer, C.; Tbatou, M.; Carail, M.; Caris-Veyrat, C.; Dornier, M.; Amiot, M.J. Thermal degradation of antioxidant micronutrients in citrus juice: Kinetics and newly formed compounds. J. Agric. Food Chem. 2007, 55, 4209–4216. [Google Scholar] [CrossRef]
- Achir, N.; Randrianatoandro, V.A.; Bohuon, P.; Laffargue, A.; Avallone, S. Kinetic study of β-carotene and lutein degradation in oils during heat treatment. Eur. J. Lipid Sci. Technol. 2010, 112, 349–361. [Google Scholar] [CrossRef]
- Aman, R.; Schieber, A.; Carle, R. Effects of heating and illumination on trans− cis isomerization and degradation of β-carotene and lutein in isolated spinach chloroplasts. J. Agric. Food Chem. 2005, 53, 9512–9518. [Google Scholar] [CrossRef]
- Waramboi, J.G.; Gidley, M.J.; Sopade, P.A. Carotenoid contents of extruded and non-extruded sweetpotato flours from Papua New Guinea and Australia. Food Chem. 2013, 141, 1740–1746. [Google Scholar] [CrossRef]
- Vásquez-Caicedo, A.L.; Schilling, S.; Carle, R.; Neidhart, S. Impact of packaging and storage conditions on colour and β-carotene retention of pasteurised mango purée. Eur. Food Res. Technol. 2007, 224, 581–590. [Google Scholar] [CrossRef]
- Gama, J.J.T.; de Sylos, C.M. Effect of thermal pasteurization and concentration on carotenoid composition of Brazilian Valencia orange juice. Food Chem. 2007, 100, 1686–1690. [Google Scholar] [CrossRef]
- Provesi, J.G.; Dias, C.O.; Amante, E.R. Changes in carotenoids during processing and storage of pumpkin puree. Food Chem. 2011, 128, 195–202. [Google Scholar] [CrossRef]
- Pinho, L.S.; de Lima, P.M.; de Sá, S.H.G.; Chen, D.; Campanella, O.H.; da Costa Rodrigues, C.E. Favaro-Trindade, C.S. Encapsulation of Rich-Carotenoids Extract from Guaraná (Paullinia cupana) Byproduct by a Combination of Spray Drying and Spray Chilling. Foods 2022, 11, 2557. [Google Scholar] [CrossRef]
| Treatments | Parameters | |
|---|---|---|
| Temperature (°C) | Shear (1/s) | |
| 70/50 | 70 | 50 |
| 80/50 | 80 | 50 |
| 90/50 | 90 | 50 |
| 70/100 | 70 | 100 |
| 80/100 | 80 | 100 |
| 90/100 | 90 | 100 |
| Formulations | Carotenoid Content (µg/g) | Carotenoid Retention (%) |
|---|---|---|
| SD1:2 | 96.0 ± 1.0 a | 100.0 ± 1.0 a |
| SD1:3 | 57.6 ± 0.7 b | 99.4 ± 0.2 a |
| SD1:4 | 40.7 ± 0.9 c | 96.7 ± 0.3 b |
| Sample | k (s−1) | t 1/2 (Days) | R2 |
|---|---|---|---|
| Free extract | 0.006 | 108.082 | 0.811 |
| SD1:2 | 0.005 | 141.924 | 0.888 |
| SD1:3 | 0.005 | 152.286 | 0.897 |
| SD1:4 | 0.005 | 144.676 | 0.874 |
| Time (days) | Formulations | Median Diameter (µm) |
|---|---|---|
| 0 | SD1:2 | 11.9 ± 1.5 a |
| 0 | SD1:3 | 9.1 ± 1.8 ab |
| 0 | SD1:4 | 8.9 ± 0.9 b |
| 90 | SD1:2 | 15.1 ± 2.8 a |
| 90 | SD1:3 | 13.4 ± 2.4 a |
| 90 | SD1:4 | 18.6 ± 5.5 a |
| Time (Days) | Formulations | Parameters | |
|---|---|---|---|
| Moisture (%) | aw | ||
| 0 | SD1:2 | 3.9 ± 0.4 d | 0.21 ± 0.02 c |
| 0 | SD1:3 | 4.8 ± 0.4 c | 0.24 ± 0.03 c |
| 0 | SD1:4 | 4.1 ± 0.7 cd | 0.16 ± 0.02 d |
| 90 | SD1:2 | 6.9 ± 0.3 b | 0.46 ± 0.01 b |
| 90 | SD1:3 | 8.1 ± 0.3 a | 0.48 ± 0.01 a |
| 90 | SD1:4 | 8.2 ± 0.1 a | 0.50 ± 0.02 a |
| Onset Temp. (°C) | Peak Temp. (°C) | Enthalpy (J/g) | |
|---|---|---|---|
| Oat | 53.05 ± 0.01 b | 60.1 ± 0.3 b | 3.1 ± 0.1 a |
| Oat + Encapsulated GPE | 54.3 ± 0.6 a | 61.0 ± 0.3 a | 2.6 ± 0.1 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pinho, L.S.; Patel, B.K.; Campanella, O.H.; Rodrigues, C.E.d.C.; Favaro-Trindade, C.S. Microencapsulation of Carotenoid-Rich Extract from Guaraná Peels and Study of Microparticle Functionality through Incorporation into an Oatmeal Paste. Foods 2023, 12, 1170. https://doi.org/10.3390/foods12061170
Pinho LS, Patel BK, Campanella OH, Rodrigues CEdC, Favaro-Trindade CS. Microencapsulation of Carotenoid-Rich Extract from Guaraná Peels and Study of Microparticle Functionality through Incorporation into an Oatmeal Paste. Foods. 2023; 12(6):1170. https://doi.org/10.3390/foods12061170
Chicago/Turabian StylePinho, Lorena Silva, Bhavesh K. Patel, Osvaldo H. Campanella, Christianne Elisabete da Costa Rodrigues, and Carmen Sílvia Favaro-Trindade. 2023. "Microencapsulation of Carotenoid-Rich Extract from Guaraná Peels and Study of Microparticle Functionality through Incorporation into an Oatmeal Paste" Foods 12, no. 6: 1170. https://doi.org/10.3390/foods12061170
APA StylePinho, L. S., Patel, B. K., Campanella, O. H., Rodrigues, C. E. d. C., & Favaro-Trindade, C. S. (2023). Microencapsulation of Carotenoid-Rich Extract from Guaraná Peels and Study of Microparticle Functionality through Incorporation into an Oatmeal Paste. Foods, 12(6), 1170. https://doi.org/10.3390/foods12061170









