Validation of a Real-Time PCR Assay for Identification of Fresh and Processed Carica papaya Botanical Material: Using Synthetic DNA to Supplement Specificity Evaluation
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Primer and Probe Design
2.3. DNA Extraction and Quantification
2.4. Real-Time PCR
2.5. Assay Specificity
gBlocks® for Unprocured Species and ddPCR Copy Number Determination
2.6. Assay Efficiency and Sensitivity
2.7. Assay Repeatability
2.8. Assay Reproducability
2.9. ddPCR Comparison
3. Results
3.1. Authenticity Testing of Papaya Samples
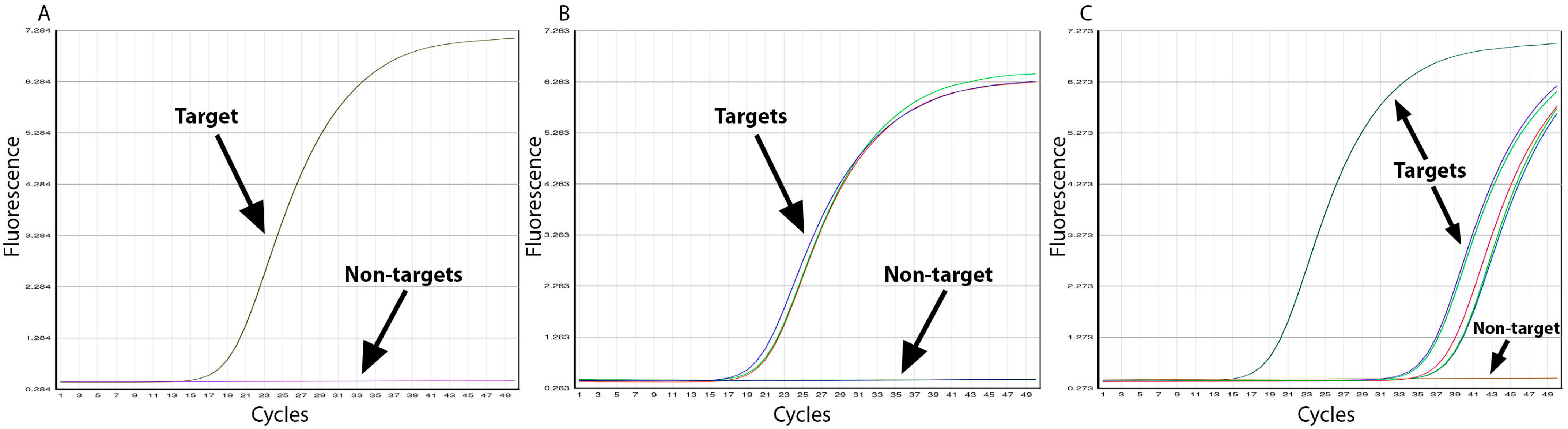
3.2. Assay Specificity
3.3. Assay Efficiency and Sensitivity
3.4. Assay Repeatability and Reproducability
3.5. gBlocks® Used for Further Specificity Evaluation
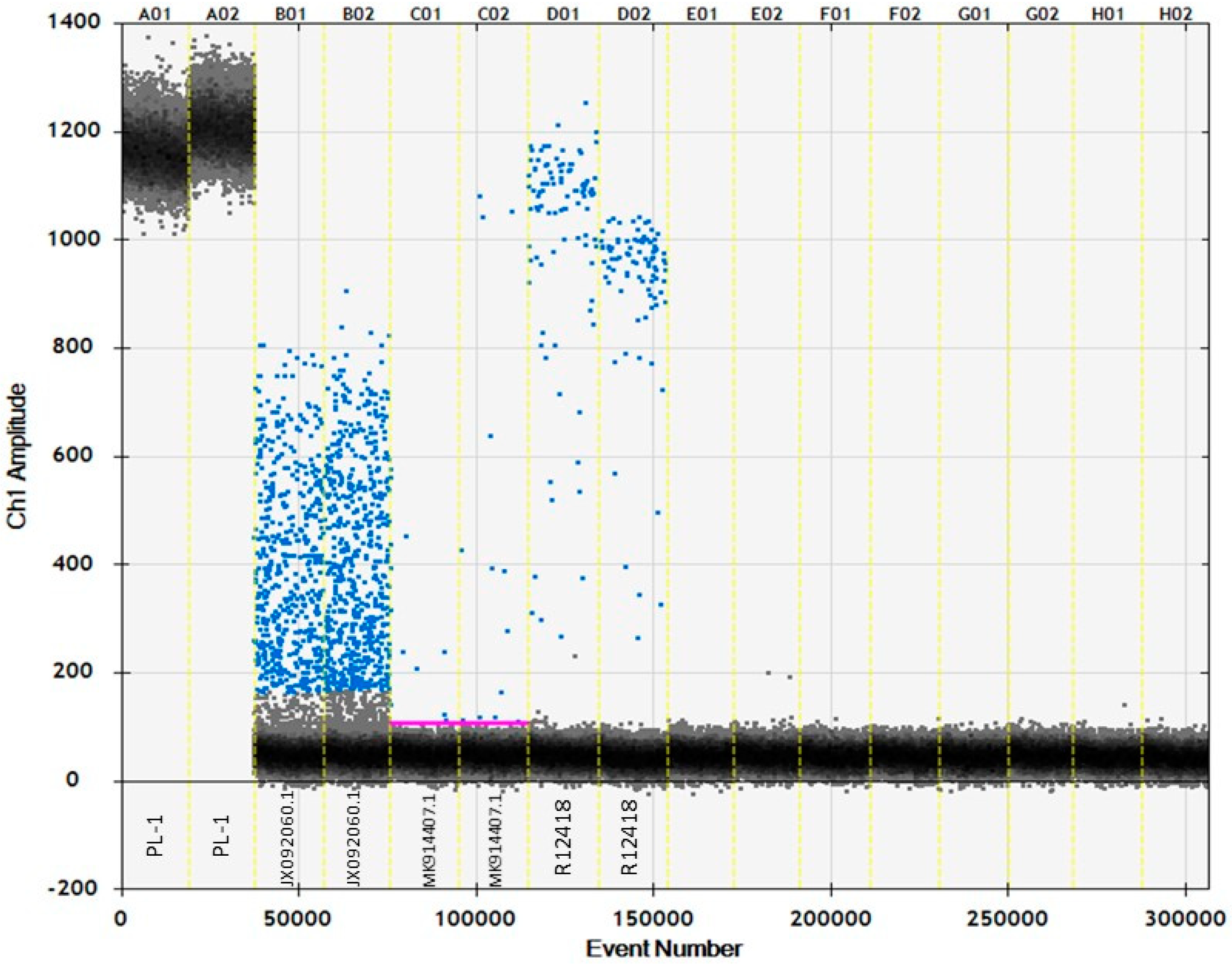
3.6. ddPCR
4. Discussion
4.1. Assay Design and Performance
4.2. Investigating Accuracy Using ddPCR
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Krishna, K.L.; Paridhavi, M.; Patel, J.A. Review on nutritional, medicinal and pharmacological properties of Papaya (Carica papaya Linn.). Nat. Prod. Radiance 2008, 7, 364–373. [Google Scholar]
- Vij, T.; Prashar, Y. A review on medicinal properties of Carica papaya Linn. Asian Pac. J. Trop. Dis. 2015, 5, 1–6. [Google Scholar] [CrossRef]
- Ismawanti, Z.; Suparyatmo, J.B.; Wiboworini, B. The effects of papaya fruit as anti diabetes: A review. J. Nutr. Sci. 2019, 4, 65–70. [Google Scholar]
- Zunjar, V.; Mammen, D.; Trivedi, B.M.; Daniel, M. Pharmacognostic, physicochemical and phytochemical studies on Carica papaya Linn. leaves. Pharmacogn. J. 2011, 3, 5–8. [Google Scholar] [CrossRef]
- TechNavio: Global Papaya Market 2022–2026. 2021. Available online: https://www.researchandmarkets.com/reports/5504962/global-papaya-market-2022-2026 (accessed on 1 November 2022).
- Evans, E.A.; Ballen, F.H.; Crane, J. An overview of global papaya production, trade, and consumption. EDIS 2012, FE913, 1–4. [Google Scholar] [CrossRef]
- Food and Agricultural Organization of the United Nations Statistics Division (FAOSTAT): Crops. 2020. Available online: https://www.fao.org/faostat/en/#data/QCL (accessed on 30 November 2022).
- Farina, V.; Passafiume, R.; Tinebra, I.; Scuderi, D.; Saletta, F.; Gugliuzza, G.; Gallotta, A.; Sortino, G. Postharvest application of aloe vera gel-based edible coating to improve the quality and storage stability of fresh-cut papaya. J. Food Qual. 2020, 2020, 8303140. [Google Scholar] [CrossRef]
- Farina, V.; Tinebra, I.; Perrone, A.; Sortino, G.; Palazzolo, E.; Mannino, G.; Gentile, C. Physicochemical, nutraceutical and sensory traits of six papaya (Carica papaya L.) cultivars grown in greenhouse conditions in the Mediterranean climate. Agronomy 2020, 10, 501. [Google Scholar] [CrossRef]
- Everstine, K.; Spink, J.; Kennedy, S. Economically motivated adulteration (EMA) of food: Common characteristics of EMA incidents. J. Food Prot. 2013, 76, 723–735. [Google Scholar] [CrossRef]
- Food and Drug Administration. 21 CFR 111.75: Current Good Manufacturing Practice in Manufacturing, Packaging, Labeling, or Holding Operations for Dietary Supplements. 2007. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=111.75 (accessed on 18 November 2022).
- de Boer, H.J.; Ichim, M.C.; Newmaster, S.G. DNA barcoding and pharmacovigilance of herbal medicines. Drug Saf. 2015, 38, 611–620. [Google Scholar] [CrossRef]
- Cankar, K.; Štebih, D.; Dreo, T.; Žel, J.; Gruden, K. Critical points of DNA quantification by real-time PCR—Effects of DNA extraction method and sample matrix on quantification of genetically modified organisms. BMC Biotechnol. 2006, 6, 37. [Google Scholar] [CrossRef]
- da Silva, F.F.; Pereira, M.G.; Campos, W.F.; Junior, P.C.D.; Pereira, T.N.S.; de Souza Filho, G.A.; Ramos, H.C.C.; Viana, A.P.; Ferreguetti, G.A. DNA marker-assisted sex conversion in elite papaya genotype (Carica papaya L.). Crop Breed. Appl. Biotechnol. 2007, 7, 52–58. [Google Scholar] [CrossRef]
- Gul, I.; Nasrullah, N.; Nissar, U.; Saifi, M.; Abdin, M.Z. Development of DNA and GC-MS fingerprints for authentication and quality control of Piper nigrum L. and its adulterant Carica papaya L. Food Anal. Methods 2018, 11, 1209–1222. [Google Scholar] [CrossRef]
- Lemos, E.G.M.; Silva, C.L.S.P.; Zaidan, H.A. Identification of sex in Carica papaya L. using RAPD markers. Euphytica 2002, 127, 179–184. [Google Scholar] [CrossRef]
- Urasaki, N.; Tokumoto, M.; Tarora, K.; Ban, Y.; Kayano, T.; Tanaka, H.; Oku, I.; Chinen, R.; Terauchi, R. A male and hermaphrodite specific RAPD marker for papaya (Carica papaya L.). Theor. Appl. Genet. 2002, 104, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Dhanya, K.; Syamkumar, S.; Sasikumar, B. Development and application of SCAR marker for the detection of papaya seed adulteration in traded black pepper powder. Food Biotechnol. 2009, 23, 97–106. [Google Scholar] [CrossRef]
- Wolfender, J.-L. HPLC in natural product analysis: The detection issue. Planta Med. 2009, 75, 719–734. [Google Scholar] [CrossRef]
- Kuddus, M.; Arif, J.M.; Ali, A.; Kausar, M.A.; Shahid, S.M.A.; Parveen, K.; Siddiqui, W.A. Chemical fingerprinting of bioactive compounds of Carica papaya Linn (Indian variety) seed oil. Biochem. Cell. Arch. 2016, 16, 9–20. [Google Scholar]
- Vadivel, V.; Ravichandran, N.; Rajalakshmi, P.; Brindha, P.; Gopal, A.; Kumaravelu, C. Microscopic, phytochemical, HPTLC, GC–MS and NIRS methods to differentiate herbal adulterants: Pepper and papaya seeds. J. Herb. Med. 2018, 11, 36–45. [Google Scholar] [CrossRef]
- Singh, V.; Rizvi, A.; Sara, U.V. Standardization and Phytochemical screening of Carica papaya seeds. Res. J. Pharm. Technol. 2021, 14, 4540–4546. [Google Scholar] [CrossRef]
- Böhme, K.; Calo-Mata, P.; Barros-Velázquez, J.; Ortea, I. Review of recent DNA-based methods for main food-authentication topics. J. Agric. Food Chem. 2019, 67, 3854–3864. [Google Scholar] [CrossRef] [PubMed]
- Parveen, I.; Gafner, S.; Techen, N.; Murch, S.J.; Khan, I.A. DNA Barcoding for the Identification of Botanicals in Herbal Medicine and Dietary Supplements: Strengths and Limitations. Planta Med. 2016, 82, 1225–1235. [Google Scholar] [CrossRef]
- Han, J.; Zhu, Y.; Chen, X.; Liao, B.; Yao, H.; Song, J.; Chen, S.; Meng, F. The short ITS2 sequence serves as an efficient taxonomic sequence tag in comparison with the full-length ITS. BioMed Res. Int. 2013, 2013, 741476. [Google Scholar] [CrossRef]
- Burns, M.; Valdivia, H. Modelling the limit of detection in real-time quantitative PCR. Eur. Food Res. Technol. 2008, 226, 1513–1524. [Google Scholar] [CrossRef]
- R Foundation for Statistical Computing. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: https://www.R-project.org/ (accessed on 8 October 2021).
- Coyne, V.E.; James, M.D.; Reid, S.J.; Rybicki, E.P. Molecular biology techniques manual. Dep. Mol. Cell Biol. Univ. Cape Town S. Afr. 2001, 1, 3–25. [Google Scholar]
- Wang, X.; Seed, B. Chapter 5: High-Throughput Primer and Probe Design. In Real-Time PCR; Dorak, T., Ed.; International University Line: New York, NY, USA, 2006; pp. 93–106. [Google Scholar]
- Prediger, E. How to Design Primers and Probes for PCR and qPCR. 2013. Available online: https://www.idtdna.com/pages/education/decoded/article/designingpcr-primers-and-probes (accessed on 30 October 2022).
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE Guidelines: Minimum Information for Publication of Quantitative Real-Time PCR Experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef] [PubMed]
- Broeders, S.; Huber, I.; Grohmann, L.; Berben, G.; Taverniers, I.; Mazzara, M.; Roosens, N.; Morisset, D. Guidelines for validation of qualitative real-time PCR methods. Trends Food Sci. Technol. 2014, 37, 115–126. [Google Scholar] [CrossRef]
- Hübner, P.; Waiblinger, H.U.; Pietsch, K.; Brodmann, P. Validation of PCR methods for quantitation of genetically modified plants in food. J. AOAC Int. 2001, 84, 1855–1864. [Google Scholar] [CrossRef]
- Maisarah, A.M.; Asmah, R.; Fauziah, O. Proximate analysis, antioxidant and anti-proliferative activities of different parts of Carica papaya. J. Tissue Eng. 2014, 5, 1. [Google Scholar]
- Faller, A.C.; Ragupathy, S.; Shanmughanandhan, D.; Zhang, Y.; Lu, Z.; Chang, P.; Swanson, G.; Newmaster, S.G. DNA quality and quantity analysis of Camellia sinensis through processing from fresh leaves to a green tea extract. J. AOAC Int. 2019, 102, 1798–1807. [Google Scholar] [CrossRef]
- Chen, L.L.; Verpoorte, R.; Yen, H.R.; Peng, W.H.; Cheng, Y.C.; Chao, J.; Pao, L.H. Effects of processing adjuvants on traditional Chinese herbs. J. Food Drug Anal. 2018, 26, S96–S114. [Google Scholar] [CrossRef] [PubMed]
- Azmin, S.N.H.M.; Manan, Z.A.; Alwi, S.R.W.; Chua, L.S.; Mustaffa, A.A.; Yunus, N.A. Herbal processing and extraction technologies. Sep. Purif. Rev. 2016, 45, 305–320. [Google Scholar] [CrossRef]
- Karni, M.; Zidon, D.; Polak, P.; Zalevsky, Z.; Shefi, O. Thermal degradation of DNA. DNA Cell Biol. 2013, 32, 298–301. [Google Scholar] [CrossRef] [PubMed]
- AOAC International Guideline Working Group. AOAC International guidelines for validation of botanical identification methods. J. AOAC Int. 2012, 95, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, F.A.; Renner, S.S. IV: A Dated Phylogeny of the Papaya Family (Caricaceae) Reveals the Crop’s Closest Relatives and the Family’s Biogeographic History. In Molecular Phylogeny, Biogeography and an E-Monograph of the Papaya Family (Caricaceae) as an Example of Taxonomy in the Electronic Age; Springer: Wiesbaden, Germany, 2015; pp. 49–81. [Google Scholar]


| Botanical Name | Family | Code | Source | Common Name | Type of Sample | Type of Material |
|---|---|---|---|---|---|---|
| Carica papaya | Caricaceae | PB–1 (Brazil) | Market | Papaya | Target | Fruit |
| Carica papaya | Caricaceae | PG–1 (Guatemala) | Market | Papaya | Target | Fruit |
| Carica papaya | Caricaceae | PL–1 (USA) | Market | Papaya | Target | Fruit |
| Carica papaya | Caricaceae | R12418 * | Martin Bauer | Papaya | Target | 8–10% fruit extract powder |
| Carica papaya | Caricaceae | R00003 ** | Martin Bauer | Papaya | Target | 8–10% fruit extract powder |
| Carica papaya | Caricaceae | 3787 | AHP | Papaya | Target | Dried fruit |
| Carica papaya | Caricaceae | BRM697 | In-house voucher | Papaya | Target | Dried leaf |
| Angelica sinensis | Apiaceae | SA09609CR38 | Alkemist | Angelica | Non-Target | Dried root |
| Panax ginseng | Araliaceae | 19091QSS | Alkemist | Asian ginseng | Non-Target | Dried root |
| Beta vulgaris | Amaranthaceae | 4413 | AHP | Beet | Non-Target | Dried root |
| Vaccinium corymbosum | Ericaceae | 5314 | Chromadex | Blueberry | Non-Target | Dried fruit |
| Matricaria chamomilla L | Asteraceae | 00030692–495 | Chromadex | Chamomile | Non-Target | Dried flower |
| Mentha canadensis syn. haplocalyx | Lamiaceae | 30984–241 | Chromadex | Chinese mint | Non-Target | Dried leaf |
| Zea mays | Poaceae | 00031127–356 | Chromadex | Corn silk | Non-Target | Dried stigma |
| Taraxacum officinale | Asteraceae | 00030662–697 | Chromadex | Dandelion | Non-Target | Dried root |
| Rosa canina | Rosaceae | 00030792–473 | Chromadex | Dog rose | Non-Target | Dried fruit |
| Zingiber officinale | Zingiberaceae | 5374 | AHP | Ginger | Non-Target | Dried root |
| Camellia sinensis | Theaceae | 00030330–054 | Chromadex | Green tea | Non-Target | Dried leaf |
| Paullinia cupana | Sapindaceae | 00030335–064 | Chromadex | Guarana | Non-Target | Dried seed |
| Melissa officinalis | Lamiaceae | 3335.6 | AHP | Lemon balm | Non-Target | Dried herb |
| Ganoderma lucidum | Ganodermataceae | H20109CRB10 | Alkemist | Lingzhi Mushroom | Non-Target | Dried, whole mushroom |
| Allium cepa | Amaryllidaceae | 4533 | AHP | Onion | Non-Target | Dried bulb |
| Rosmarinus officinalis | Lamiaceae | 5063 | AHP | Rosemary | Non-Target | Dried leaf |
| Schisandra chinensis | Schisandraceae | 3241.4 | AHP | Schisandra | Non-Target | Dried fruit |
| Spinacia oleracea | Amaranthaceae | 4647 | Chromadex | Spinach | Non-Target | Dried leaf |
| Curcuma longa | Zingiberaceae | 00031107–328 | Chromadex | Turmeric | Non-Target | Dried root |
| Daucus carota | Apiaceae | 00031080–288 | Alkemist | Wild carrot | Non-Target | Dried root |
| Petroselinum crispum | Apiaceae | UR29409CRB15 | AHP | Parsley | Non-Target | Dried leaf |
| Jacaratia dolichaula | Caricaceae | JX092060.1 | IDT | Barrilillo | Non-Target | gBlock® |
| Jacaratia digitata | Caricaceae | MK914407.1 | IDT | Papaya Caspi | Non-Target | gBlock® |
| Oligo | Sequence |
|---|---|
| Forward Primer | 5′-TCG AGT CTT TGA ACG CAA GTT-3′ |
| Reverse Primer | 5′-GGG GAA GGA GGT TTT TGT G-3′ |
| Probe | 5′-/56-FAM/ACT GTG CGT/ZEN/GAC ACC CAG GCA GA/3IABkFQ/-3′ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patel, R.; Faller, A.C.; Nguyen, T.; Quan, Z.; Eminger, C.; Kaul, S.; Collins, T.; Zhang, Y.; Chang, P.; Swanson, G.; et al. Validation of a Real-Time PCR Assay for Identification of Fresh and Processed Carica papaya Botanical Material: Using Synthetic DNA to Supplement Specificity Evaluation. Foods 2023, 12, 530. https://doi.org/10.3390/foods12030530
Patel R, Faller AC, Nguyen T, Quan Z, Eminger C, Kaul S, Collins T, Zhang Y, Chang P, Swanson G, et al. Validation of a Real-Time PCR Assay for Identification of Fresh and Processed Carica papaya Botanical Material: Using Synthetic DNA to Supplement Specificity Evaluation. Foods. 2023; 12(3):530. https://doi.org/10.3390/foods12030530
Chicago/Turabian StylePatel, Rajesh, Adam C. Faller, Tiffany Nguyen, Zheng Quan, Corey Eminger, Swetha Kaul, Ted Collins, Yanjun Zhang, Peter Chang, Gary Swanson, and et al. 2023. "Validation of a Real-Time PCR Assay for Identification of Fresh and Processed Carica papaya Botanical Material: Using Synthetic DNA to Supplement Specificity Evaluation" Foods 12, no. 3: 530. https://doi.org/10.3390/foods12030530
APA StylePatel, R., Faller, A. C., Nguyen, T., Quan, Z., Eminger, C., Kaul, S., Collins, T., Zhang, Y., Chang, P., Swanson, G., & Lu, Z. (2023). Validation of a Real-Time PCR Assay for Identification of Fresh and Processed Carica papaya Botanical Material: Using Synthetic DNA to Supplement Specificity Evaluation. Foods, 12(3), 530. https://doi.org/10.3390/foods12030530






