Formation, Evolution, and Antioxidant Activity of Melanoidins in Black Garlic under Different Storage Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Preparation
2.3. Absorbance Measurement
2.4. Reaction Kinetics
2.5. Chemical Compositions
2.5.1. Reducing Sugar
2.5.2. Protein
2.5.3. Total Phenolic (TPC)
2.6. Elemental Analysis
2.7. Py-GC-MS
2.8. Molecular Weight Measurement
2.9. Fourier Transform Infrared Spectroscopy (FT-IR) Analysis
2.10. Morphology Analysis
2.11. Antioxidant Activity
2.12. Statistical Analysis
3. Results and Discussions
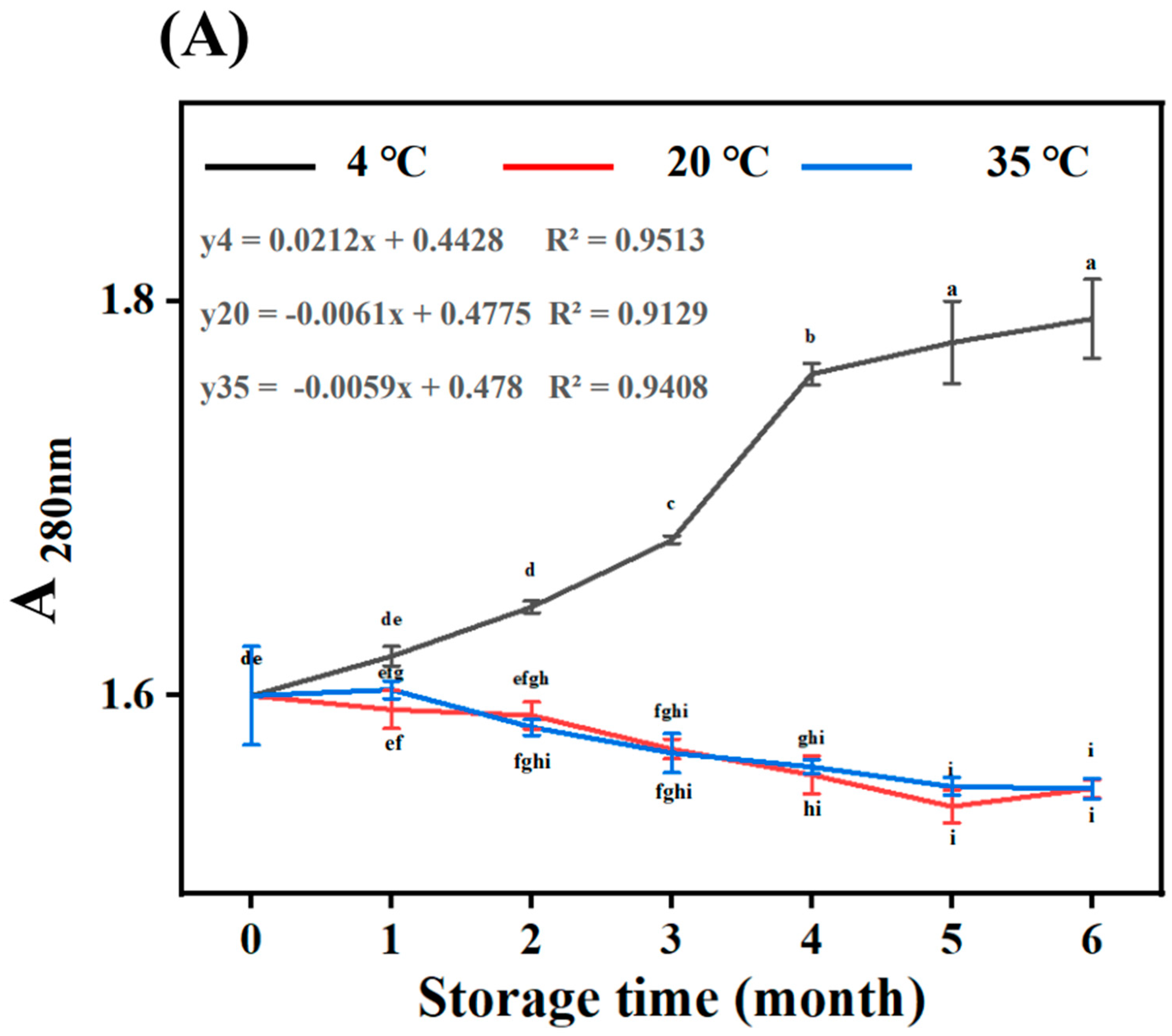
3.1. Absorbance Measurement
3.2. Chemical Compositions of MLDs under Storage at Different Temperatures
3.2.1. Reducing Sugar
3.2.2. Protein and Amino Acids
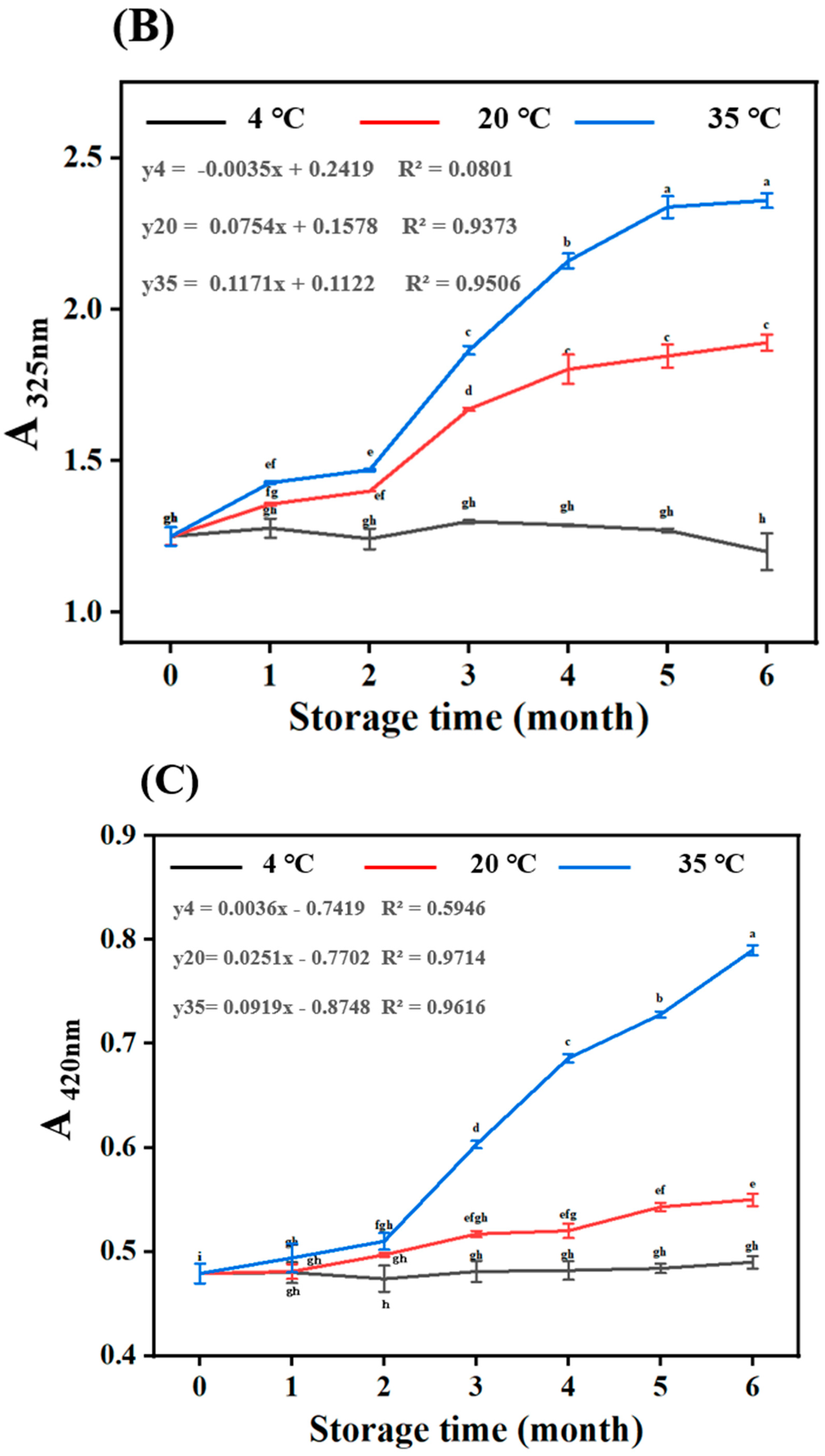
3.2.3. Total Phenolic
3.3. Elemental Analysis
3.4. Py-GC-MS Analysis
3.4.1. Characteristics of Volatile Compounds Formed through MLD Pyrolysis
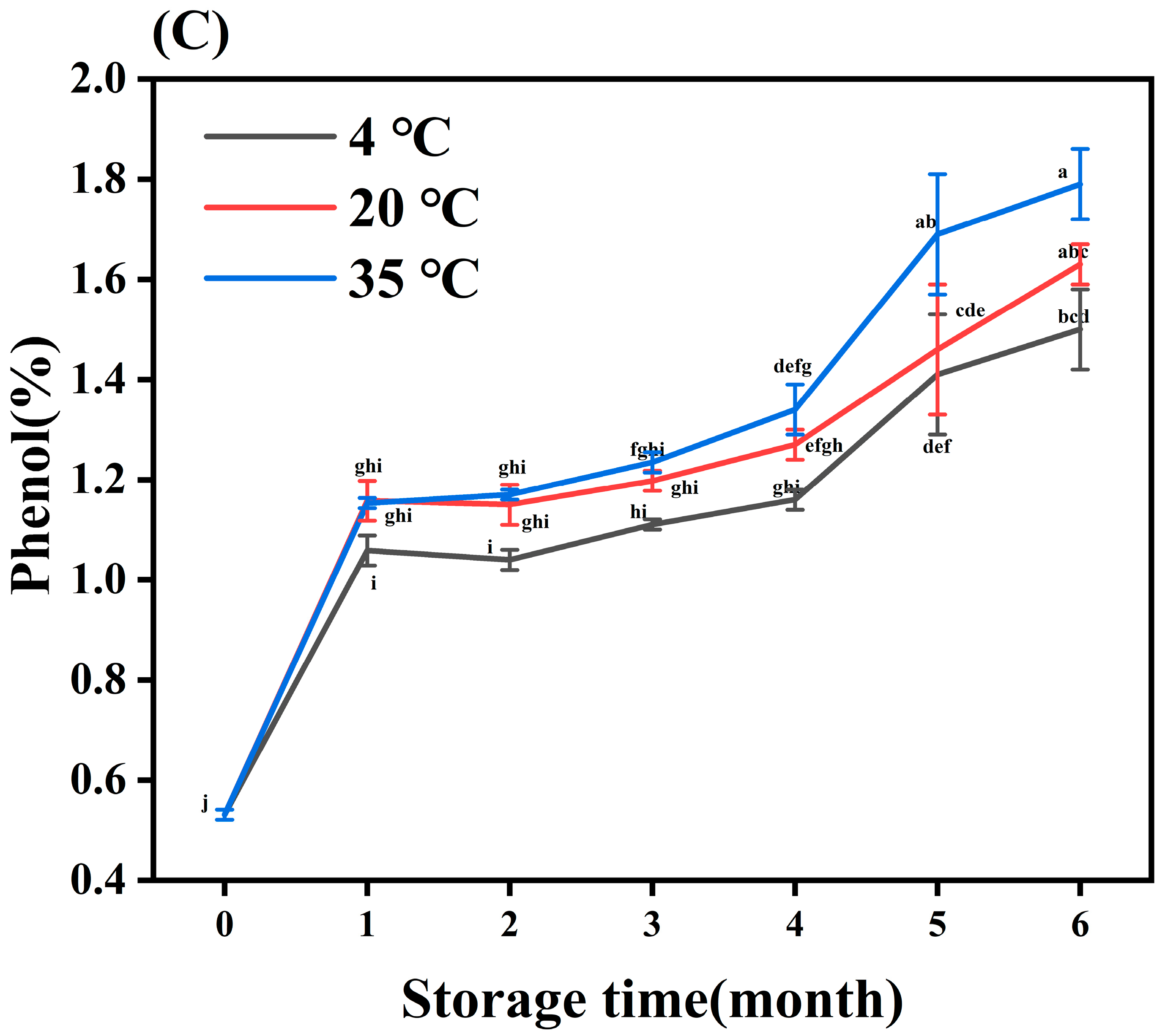
3.4.2. Principal Component Analysis (PCA), Partial Least Squares–Discriminant Analysis (PLS-DA) and Heat Map Analysis
3.5. The Structural Characteristics of MLDs
3.5.1. Molecular Weight (MW) Measurement
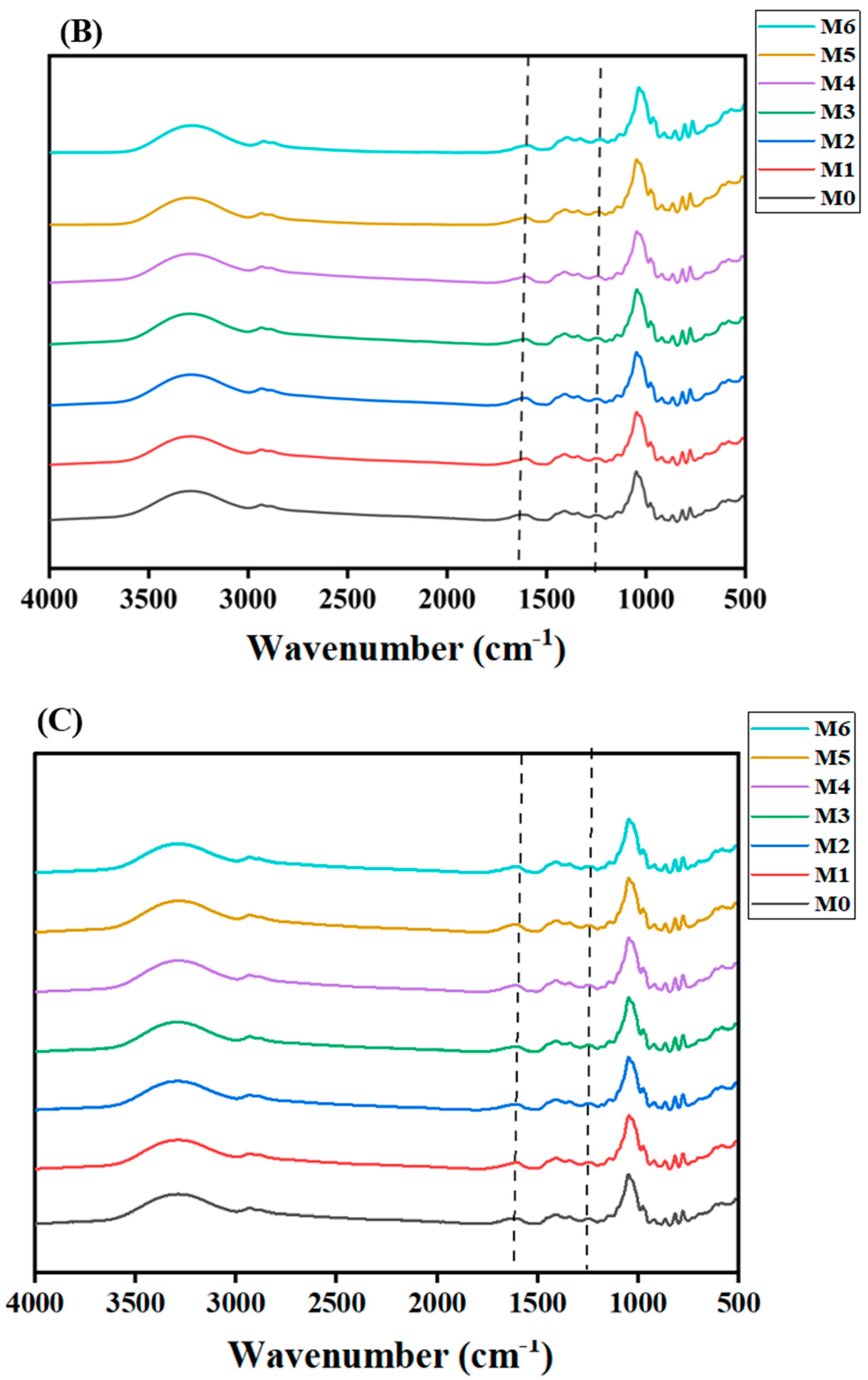
3.5.2. FT-IR Analysis
3.5.3. Morphology Analysis
3.6. Antioxidant Activity
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Temperature | Storage Time (Month) | Arg (mg·100g−1) | P-Ser (mg·100g−1) | Glu (mg·100g−1) | Lys (mg·100g−1) | Asp (mg·100g−1) | Ser (mg·100g−1) | Thr (mg·100g−1) | Met (mg·100g−1) | His (mg·100g−1) |
|---|---|---|---|---|---|---|---|---|---|---|
| 4 °C | 0 | 844.26 | 119.12 | 65.98 | 51.35 | 44.79 | 31.50 | 27.95 | 23.90 | 4.53 |
| 3 | 850.63 | 67.91 | 72.42 | 55.40 | 41.78 | 31.11 | 27.49 | 26.81 | 4.29 | |
| 6 | 673.82 | 39.34 | 52.45 | 39.76 | 22.75 | 22.13 | 17.11 | 15.69 | 2.04 | |
| 20 °C | 0 | 844.26 | 119.12 | 65.98 | 51.35 | 44.79 | 31.50 | 27.95 | 23.90 | 4.53 |
| 3 | 1016.12 | 75.99 | 89.09 | 65.23 | 53.46 | 39.60 | 32.96 | 29.00 | 6.12 | |
| 6 | 737.95 | 33.19 | 53.90 | 47.19 | 19.11 | 23.25 | 15.67 | 16.53 | 3.58 | |
| 35 °C | 0 | 844.26 | 119.12 | 65.98 | 51.35 | 44.79 | 31.50 | 27.95 | 23.90 | 4.53 |
| 3 | 758.45 | 68.66 | 67.88 | 38.15 | 42.95 | 32.35 | 31.34 | 26.50 | 3.94 | |
| 6 | 798.00 | 30.26 | 55.61 | 48.44 | 17.98 | 20.68 | 15.50 | 16.14 | 3.51 |
Appendix B
| Temperature | Storage Time (Month) | Heterocyclic Compounds | Acids | Aliphatic Hydrocarbons | Ketones | Alcohols | Aromatic Hydrocarbons | Other Compounds | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n | P (%) | n | P (%) | n | P (%) | n | P (%) | n | P (%) | n | P (%) | n | P (%) | ||
| 4 °C | 0 | 22 | 56.6 | 5 | 19.68 | 21 | 9.28 | 22 | 5.93 | 5 | 1.79 | 8 | 4.39 | 18 | 2.33 |
| 1 | 24 | 57.11 | 4 | 16.11 | 17 | 9.01 | 23 | 6.37 | 5 | 2.27 | 7 | 4.86 | 23 | 4.27 | |
| 2 | 29 | 55.74 | 5 | 11.09 | 26 | 5.62 | 24 | 6.19 | 4 | 2.7 | 7 | 4.5 | 15 | 14.16 | |
| 3 | 19 | 61.29 | 9 | 4.53 | 15 | 19.38 | 25 | 5.55 | 7 | 2.14 | 5 | 3.36 | 13 | 3.75 | |
| 4 | 19 | 59.01 | 12 | 16.44 | 17 | 5.15 | 17 | 5.89 | 6 | 2.33 | 4 | 2.31 | 14 | 8.86 | |
| 5 | 23 | 60.79 | 16 | 19.16 | 19 | 3.09 | 25 | 5.97 | 7 | 2.69 | 3 | 2.61 | 12 | 5.69 | |
| 6 | 23 | 59.2 | 16 | 20.12 | 19 | 1.83 | 15 | 6 | 4 | 2.78 | 3 | 2.63 | 15 | 7.44 | |
| Total number | 101 | 103 | |||||||||||||
| 20 °C | 0 | 22 | 56.6 | 5 | 19.68 | 21 | 9.28 | 22 | 5.93 | 5 | 1.79 | 8 | 4.39 | 18 | 2.33 |
| 1 | 29 | 61.9 | 6 | 17.54 | 19 | 5.76 | 18 | 5.96 | 7 | 2.31 | 3 | 5.02 | 24 | 1.51 | |
| 2 | 33 | 66.97 | 7 | 12.91 | 23 | 5.71 | 16 | 5.47 | 8 | 2.94 | 6 | 5.36 | 14 | 0.64 | |
| 3 | 23 | 69.46 | 11 | 13.66 | 19 | 1.12 | 30 | 7.43 | 6 | 2.21 | 6 | 4.56 | 17 | 1.56 | |
| 4 | 27 | 69.52 | 15 | 12.43 | 16 | 4.24 | 24 | 7.02 | 6 | 2.24 | 5 | 3.09 | 13 | 1.47 | |
| 5 | 28 | 73.22 | 10 | 11.63 | 16 | 2.72 | 29 | 8.59 | 5 | 2.41 | 3 | 1.41 | 11 | 0.01 | |
| 6 | 28 | 72.5 | 9 | 11.78 | 22 | 1.76 | 18 | 8.9 | 7 | 2.65 | 3 | 2.24 | 13 | 0.17 | |
| Total number | 190 | 63 | 136 | 157 | 44 | 34 | 110 | ||||||||
| 35 °C | 0 | 22 | 56.6 | 5 | 19.68 | 21 | 9.28 | 22 | 5.93 | 5 | 1.79 | 8 | 4.39 | 18 | 2.33 |
| 1 | 31 | 62.1 | 7 | 18.91 | 21 | 5.73 | 27 | 6.05 | 6 | 2.31 | 5 | 3.4 | 19 | 1.5 | |
| 2 | 24 | 69.14 | 8 | 11.89 | 25 | 4.78 | 26 | 5.95 | 9 | 2.26 | 9 | 5.23 | 17 | 0.75 | |
| 3 | 29 | 73.57 | 10 | 2.77 | 24 | 6.82 | 29 | 7.8 | 9 | 2.39 | 7 | 4.29 | 20 | 2.36 | |
| 4 | 29 | 70.75 | 12 | 13.12 | 18 | 2.5 | 28 | 8.65 | 5 | 1.89 | 4 | 2.87 | 17 | 0.22 | |
| 5 | 25 | 79.84 | 5 | 1.94 | 21 | 3.91 | 17 | 8.67 | 5 | 2.53 | 4 | 2.21 | 15 | 0.9 | |
| 6 | 32 | 78.89 | 6 | 4.94 | 20 | 1.69 | 20 | 9.2 | 5 | 2.69 | 3 | 2.3 | 15 | 0.29 | |
| Total number | 192 | 53 | 150 | 169 | 44 | 40 | 121 | ||||||||
Appendix C
| Temperature | Storage Time (Month) | Mean Roughness (Ra, nm) | Skewness | Kurtosis |
|---|---|---|---|---|
| 4 °C | 0 | 0.147 | 2.29 | 56 |
| 1 | 0.248 | 2.44 | 14.1 | |
| 3 | 0.332 | 1.9 | 8.18 | |
| 6 | 0.259 | 1.85 | 9.44 | |
| 20 °C | 0 | 0.147 | 2.29 | 56 |
| 1 | 0.282 | 4.14 | 50.8 | |
| 3 | 0.269 | 1.41 | 8.64 | |
| 6 | 2.830 | 2.8 | 10.9 | |
| 35 °C | 0 | 0.147 | 2.29 | 56 |
| 1 | 0.198 | 12.2 | 424 | |
| 3 | 0.588 | 13.6 | 276 | |
| 6 | 1.070 | 10.3 | 124 |
References
- Chae, J.; Lee, E.; Oh, S.M.; Ryu, H.W.; Kim, S.; Nam, J.O. Aged black garlic (Allium sativum L.) and aged black elephant garlic (Allium ampeloprasum L.) alleviate obesity and attenuate obesity-induced muscle atrophy in diet-induced obese C57BL/6 mice. Biomed. Pharmacother. 2023, 163, 114810. [Google Scholar] [CrossRef]
- Ried, K.; Travica, N.; Sali, A. The effect of aged garlic extract on blood pressure and other cardiovascular risk factors in uncontrolled hypertensives: The AGE at Heart trial. Integr. Blood Control 2016, 9, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Park, C.; Park, S.; Chung, Y.; Kim, G.; Choi, Y.; Kim, B.; Choi, Y. Induction of apoptosis by a hexane extract of aged black garlic in the human leukemic U937 cells. Nutr. Res. Pract. 2014, 8, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Recinella, L.; Libero, M.; Citi, V.; Chiavaroli, A.; Martelli, A.; Foligni, R.; Mannozzi, C.; Acquaviva, A.; Simone, S.; Calderone, V.; et al. Anti-Inflammatory and Vasorelaxant Effects Induced by an Aqueous Aged Black Garlic Extract Supplemented with Vitamins D, C, and B12 on Cardiovascular System. Foods 2023, 12, 1558. [Google Scholar] [CrossRef]
- Molina-Calle, M.; Medinaa, V.S.; Priego-Capote, F.; Castro, M.D. Establishing compositional differences between fresh and black garlic by a metabolomics approach based on LC–QTOF MS/MS analysis. J. Food Compos. Anal. 2017, 62, 155–163. [Google Scholar] [CrossRef]
- Pérez-Burilloa, S.; Rajakarunab, S.; Pastoriza, S.; Paliy, O.; Rufián-Henares, J.A. Bioactivity of food melanoidins is mediated by gut microbiota. Food Chem. 2020, 316, 126309. [Google Scholar] [CrossRef] [PubMed]
- Peña-Correa, R.F.; Mogol, B.A.; Fryganas, C.; Fogliano, V. Fluidized-bed-roasted cocoa has different chemical characteristics than conventionally roasted cocoa. J. Agric. Food Chem. 2023, 71, 10203–10211. [Google Scholar] [CrossRef]
- Bruhns, P.; Kanzler, C.; Degenhardt, A.G.; Kroh, T.J.; Lothar, W.K. Basic structure of melanoidins formed in the maillard reaction of 3-deoxyglucosone and gamma-aminobutyric acid. J. Agric. Food Chem. 2019, 67, 5197–5203. [Google Scholar] [CrossRef]
- Morales, F.J.; Somoza, V.; Fogliano, V. Physiological relevance of dietary melanoidins. Amino Acids 2012, 42, 1097–1109. [Google Scholar] [CrossRef]
- Zhao, Y.-M.; Jiang, Y.-L.; Ding, Y.-F.; Wang, D.-F.; Deng, Y. High hydrostatic pressure-assisted extraction of high-molecular-weight melanoidins from black garlic: Composition, structure, and bioactive properties. J. Food Qual. 2019, 2019, 1682749. [Google Scholar] [CrossRef]
- Qi, Z.; Chen, L.-Q.; Pu, X.-X.; Du, G.-C.; Yang, F.; Lu, J.-J.; Peng, Z.; Zhang, J.; Tu, H.-B. The Differences in the Composition of Maillard Components between Three Kinds of Sauce-Flavor Daqu. Fermentation 2023, 9, 860. [Google Scholar] [CrossRef]
- Zhao, Y.-M.; Ding, Y.-F.; Wang, D.-F.; Deng, Y.; Zhao, Y.-M. Effect of high hydrostatic pressure conditions on the composition, morphology, rheology, thermal behavior, color, and stability of black garlic melanoidins. Food Chem. 2021, 337, 127790. [Google Scholar] [CrossRef] [PubMed]
- Lan, X.-H.; Liu, P.; Xia, S.-Q.; Jia, C.-S.; Mukunzi, D.; Zhang, X.-M.; Xia, W.-S.; Tian, H.-X.; Xiao, Z.-B. Temperature effect on the non-volatile compounds of Maillard reaction products derived from xylose-soybean peptide system: Further insights into thermal degradation and cross-linking. Food Chem. 2010, 120, 967–972. [Google Scholar] [CrossRef]
- Rochat, S.; Laumer, J.Y.S.; Chaintreau, A. Analysis of sulfur compounds from the in-oven roast beef aroma by comprehensive two-dimensional gas chromatography. J. Chromatogr. A 2007, 1147, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-Q.; Fan, W.-L.; Nie, Y.; Xu, Y. The formation and structural characteristics of melanoidins from fermenting and distilled grains of Chinese liquor (baijiu). Food Chem. 2023, 410, 135372. [Google Scholar] [CrossRef] [PubMed]
- Kchaou, H.; Benbettaieb, N.; Jridi, M.; Nasri, M.; Debeaufort, F. Influence of Maillard reaction and temperature on functional, structure and bioactive properties of fish gelatin films. Food Hydrocoll. 2019, 97, 105196. [Google Scholar] [CrossRef]
- Brudzynski, K.; Miotto, D. Honey melanoidins: Analysis of the compositions of the high molecular weight melanoidins exhibiting radical-scavenging activity. Food Chem. 2011, 127, 1023–1030. [Google Scholar] [CrossRef]
- Wang, H.-Y.; Qian, H.; Yao, W.-R. Melanoidins produced by the Maillard reaction: Structure and biological activity. Food Chem. 2011, 128, 573–584. [Google Scholar] [CrossRef]
- Mohsin, G.F.; Schmitt, F.J.; Kanzler, C.; Alzubaidi, A.K.; Hornemann, A. How alanine catalyzes melanoidin formation and dehydration during synthesis from glucose. Eur. Food Res. Technol. 2022, 248, 1615–1624. [Google Scholar] [CrossRef]
- Babbar, N.; Oberoi, H.S.; Uppal, D.S.; Patil, R.T. Total phenolic content and antioxidant capacity of extracts obtained from six important fruit residues. Food Res. Int. 2011, 44, 391–396. [Google Scholar] [CrossRef]
- Grigorious, A.; Pinakoulaki, E. Linking the dynamic changes in the in vitro antioxidant activity of carob kibbles upon roasting to the chemical and structural changes revealed by FTIR spectroscopy. Antioxidants 2021, 10, 2025. [Google Scholar] [CrossRef] [PubMed]
- Mohsin, G.F.; Schmitt, F.J.; Kanzler, C.; Hoehl, A.; Hornemann, A. PCA-based identification and differentiation of FTIR data from model melanoidins with specific molecular compositions. Food Chem. 2019, 281, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Bekedam, E.K.; Loots, M.J.; Schols, H.A.; Boekel, M.A.J.S.V.; Smit, G. Roasting effects on formation mechanisms of coffee brew melanoidins. J. Agric. Food Chem. 2008, 56, 7138–7145. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-L.; Gao, H.; Wang, C.-Y.; Qayum, A.; Mu, Z.-S.; Gao, Z.-L.; Jiang, Z.-M. Characterization and comparison of the structure and antioxidant activity of glycosylated whey peptides from two pathways. Food Chem. 2018, 257, 279–288. [Google Scholar] [CrossRef]
- Xia, T.; Zhang, B.; Duan, W.-H.; Zhang, J.; Wang, M. Nutrients and bioactive components from vinegar: A fermented and functional food. J. Funct. Foods 2020, 64, 103681. [Google Scholar] [CrossRef]
- Quiroz-Reyes, C.N.; Fogliano, V. Design cocoa processing towards healthy cocoa products: The role of phenolics and melanoidins. J. Funct. Foods 2018, 45, 480–490. [Google Scholar] [CrossRef]
- Kang, O. Evaluation of melanoidins formed from black garlic after different thermal processing steps. Prev. Nutr. Food Sci. 2016, 21, 398–405. [Google Scholar] [CrossRef]
- Carvalho, W.S.; Junior, J.A.S.; Pires de Oliveira, T.J.; Ataide, C.H. Fast pyrolysis of sweet sorghum bagasse in a fluidized bed reactor: Product characterization and comparison with vapors generated in analytical pyrolysis. Energy 2017, 131, 186–197. [Google Scholar] [CrossRef]
- Davídek, T.; Devaud, S.; Robert, F.; Blank, I. Sugar fragmentation in the Maillard reaction cascade: Isotope labeling studies on the formation of acetic acid by a hydrolytic-dicarbonyl cleavage mechanism. J. Agric. Food Chem. 2006, 54, 6667–6676. [Google Scholar] [CrossRef]
- Yao, Y.-G.; Wang, W.-Y.; Chen, L.-Y.; Liu, H.-M.; Yan, R.-Z.; Li, S.; Wang, X.-D. Structural changes of cellulosic polysaccharides in sesame hull during roasting. Qual. Assur. Saf. Crop 2021, 13, 13–24. [Google Scholar] [CrossRef]
- Mohsin, G.F.; Schmitt, F.J.; Kanzler, C.; Dirk Epping, J.; Flemig, S.; Hornemann, A. Structural characterization of melanoidin formed from d-glucose and l-alanine at different temperatures applying FTIR, NMR, EPR, and MALDI-ToF-MS. Food Chem. 2018, 45, 761–767. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.-Q.; Bao, Y.-H.; Chen, Y. Characteristics and antioxidant activity of water-soluble Maillard reaction products from interactions in a whey protein isolate and sugars system. Food Chem. 2013, 139, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Patrignani, M.; González-Forte, L.S. Characterisation of melanoidins derived from brewers’ spent grain: New insights into their structure and antioxidant activity. Int. J. Food Sci. Technol. 2021, 56, 384–391. [Google Scholar] [CrossRef]
- Bekedam, E.K.; Schols, H.A.; Boekel, M.A.J.S.; Smit, G. High molecular weight melanoidins from coffee brew. J. Agric. Food Chem. 2006, 54, 7658–7666. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.-F.; Jin, Y.; Zhang, M. Evaluation on the physicochemical and digestive properties of melanoidin from black garlic and their antioxidant activities in vitro. Food Chem. 2020, 340, 127934. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-Q.; Fan, W.-L.; Xu, Y. Melanoidins from Chinese distilled spent grain: Content, preliminary structure, antioxidant, and ACE-inhibitory activities in vitro. Foods 2019, 8, 516. [Google Scholar] [CrossRef] [PubMed]
- Kuisma, R.; Pesonen-Leinonen, E.; Redsven, I.; Kymalainen, H.-R.; Saarikoski, I.; Sjoberg, A.-M.; Hautala, M. Utilization of profilometry, SEM, AFM and contact angle measurements in describing surfaces of plastic floor coverings and explaining their cleanability. Surf. Sci. 2005, 584, 119–125. [Google Scholar] [CrossRef]
- Liu, J.-Y.; Gan, J.; Yu, Y.-J.; Zhu, S.-H.; Yin, L.-J.; Cheng, Y.-Q. Effect of laboratory-scale decoction on the antioxidative activity of zhenjiang aromatic vinegar: The contribution of melanoidins. J. Funct. Foods 2016, 21, 75–86. [Google Scholar] [CrossRef]
- Molina-Calle, M.; Priego-capote, F.; Luque de Castro, M.D. Headspace–GC–MS volatile profile of black garlic vs fresh garlic: Evolution along fermentation and behavior under heating. LWT-Food Sci. Techhnol. 2017, 80, 98–105. [Google Scholar] [CrossRef]
- Zeng, Y.; Zhang, X.-X.; Guan, Y.-P.; Sun, Y.-X. Characteristics and antioxidant activity of Maillard reaction products from psicose-lysine and fructose-lysine model systems. J. Food Sci. 2011, 76, C398–C403. [Google Scholar] [CrossRef]
- Oracz, J.; Zyzelewicz, D. In vitro antioxidant activity and FTIR characterization of high-molecular weight melanoidin fractions from different types of cocoa beans. Antioxidants 2019, 8, 560. [Google Scholar] [CrossRef] [PubMed]
- Verzelloni, E.; Tagliazucchi, D.; Conte, A. Changes in major antioxidant compounds during aging of traditional balsamic vinegar. J. Food Biochem. 2010, 34, 152–171. [Google Scholar] [CrossRef]









| Temperature | Storage Time (Month) | O (%) | C (%) | H (%) | N (%) |
|---|---|---|---|---|---|
| 4 °C | 0 | 55.81 ± 0.02 a | 36.07 ± 0.49 l | 6.8 ± 0.01 f | 1.03 ± 0.01 g |
| 1 | 54.07 ± 0.01 c | 38.01 ± 0.06 j | 6.64 ± 0.01 i | 1.09 ± 0.01 f | |
| 2 | 52.76 ± 0.02 ef | 38.76 ± 0.07 f | 6.86 ± 0.02 e | 1.12 ± 0.01 e | |
| 3 | 53.41 ± 0.06 d | 38.45 ± 0.06 g | 6.74 ± 0.01 h | 1.11 ± 0.01 e | |
| 4 | 52.41 ± 0.02 fg | 38.99 ± 0.01 e | 6.99 ± 0.05 c | 1.11 ± 0.02 e | |
| 5 | 52.58 ± 0.03 fg | 39.07 ± 0.01 cd | 7.00 ± 0.01 c | 1.12 ± 0.01 e | |
| 6 | 52.59 ± 0.04 fg | 39.03 ± 0.01 d | 7.01 ± 0.01 c | 1.12 ± 0.01 e | |
| 20 °C | 0 | 55.81 ± 0.02 a | 36.07 ± 0.01 l | 6.80 ± 0.02 f | 1.03 ± 0.01 g |
| 1 | 54.74 ± 0.04 b | 36.35 ± 0.16 k | 6.77 ± 0.01 g | 1.02 ± 0.01 g | |
| 2 | 53.17 ± 0.07 d | 38.17 ± 0.07 i | 6.92 ± 0.02 d | 1.02 ± 0.01 g | |
| 3 | 52.61 ± 0.02 fg | 38.38 ± 0.53 gh | 6.86 ± 0.01 e | 1.11 ± 0.02 e | |
| 4 | 52.00 ± 0.03 h | 38.37 ± 0.03 h | 6.99 ± 0.05 c | 1.15 ± 0.02 cd | |
| 5 | 52.75 ± 0.01 d | 39.05 ± 0.03 cd | 6.95 ± 0.01 d | 1.13 ± 0.01 e | |
| 6 | 52.59 ± 0.02 fg | 39.07 ± 0.02 cd | 7.00 ± 0.02 c | 1.13 ± 0.01 e | |
| 35 °C | 0 | 55.81 ± 0.02 a | 36.07 ± 0.09 l | 6.80 ± 0.01 f | 1.03 ± 0.01 g |
| 1 | 53.58 ± 0.04 d | 38.21 ± 0.03 i | 6.77 ± 0.04 fg | 1.13 ± 0.01 e | |
| 2 | 53.16 ± 0.05 de | 38.40 ± 0.01 gh | 7.06 ± 0.01 b | 1.15 ± 0.01 cd | |
| 3 | 52.50 ± 0.05 fg | 39.11 ± 0.11 bc | 6.99 ± 0.03 c | 1.15 ± 0.01 cd | |
| 4 | 52.19 ± 0.06 gh | 39.19 ± 0.05 a | 7.04 ± 0.01 b | 1.19 ± 0.01 a | |
| 5 | 52.51 ± 0.03 fg | 39.03 ± 0.02 cd | 7.11 ± 0.01 a | 1.17 ± 0.01 b | |
| 6 | 52.32 ± 0.05 fgh | 39.17 ± 0.01 ab | 7.12 ± 0.01 a | 1.18 ± 0.01 ab |
| Storage Time (Month) | Temperature | ||
|---|---|---|---|
| 4 °C | 20 °C | 35 °C | |
| 0 | 14,300 ± 203 ab | 14,300 ± 203 ab | 14,300 ± 203 ab |
| 1 | 14,295 ± 124 ab | 14,310 ± 148 ab | 14,326 ± 117 ab |
| 2 | 14,196 ± 147 b | 14,309 ± 104 ab | 14,320 ± 139 ab |
| 3 | 14,203 ± 118 b | 14,305 ± 150 ab | 14,342 ± 112 ab |
| 4 | 14,181 ± 132 b | 14,326 ± 123 ab | 14,341 ± 171 ab |
| 5 | 14,171 ± 115 b | 14,320 ± 112 ab | 14,456 ± 121 a |
| 6 | 14,175 ± 112 b | 14,325 ± 102 ab | 14,449 ± 121 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, B.; Zhong, Y.; Wang, D.; Meng, F.; Li, Y.; Deng, Y. Formation, Evolution, and Antioxidant Activity of Melanoidins in Black Garlic under Different Storage Conditions. Foods 2023, 12, 3727. https://doi.org/10.3390/foods12203727
Wang B, Zhong Y, Wang D, Meng F, Li Y, Deng Y. Formation, Evolution, and Antioxidant Activity of Melanoidins in Black Garlic under Different Storage Conditions. Foods. 2023; 12(20):3727. https://doi.org/10.3390/foods12203727
Chicago/Turabian StyleWang, Bobo, Yu Zhong, Danfeng Wang, Fanbing Meng, Yuncheng Li, and Yun Deng. 2023. "Formation, Evolution, and Antioxidant Activity of Melanoidins in Black Garlic under Different Storage Conditions" Foods 12, no. 20: 3727. https://doi.org/10.3390/foods12203727
APA StyleWang, B., Zhong, Y., Wang, D., Meng, F., Li, Y., & Deng, Y. (2023). Formation, Evolution, and Antioxidant Activity of Melanoidins in Black Garlic under Different Storage Conditions. Foods, 12(20), 3727. https://doi.org/10.3390/foods12203727





