The Role of Aquaculture and Capture Fisheries in Meeting Food and Nutrition Security: Testing a Nutrition-Sensitive Pond Polyculture Intervention in Rural Zambia
Abstract
1. Introduction
2. Methods and Materials
2.1. Sampling and Site Selection
2.2. Intervention: Polyculture Pond Farming and Nutrition Training
- Take fish from their ponds whenever they wanted to, rather than at the end of the growth cycle (promoting intermittent harvesting).
- Cultivate a diversity of species and not eliminate SIS (promoting polyculture).
- Use natural rather than formulated feeds since the aim did not require maximizing the growth of a single species in a pond (promoting natural feeding regimes).
2.3. Data Collection
2.3.1. Primary Data: Demographic Information and Fish Food Diaries
2.3.2. Secondary Data: Nutrient Composition of Fish Species and Recommended Nutrient Intake
2.4. Analysis of Longitudinal Fish Consumption and Individual Nutrient Intake
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fluet-Chouinard, E.; Funge-Smith, S.; McIntyre, P.B. Global hidden harvest of freshwater fish revealed by household surveys. Proc. Natl. Acad. Sci. USA 2018, 115, 7623–7628. [Google Scholar] [CrossRef] [PubMed]
- Funge-Smith, S.; Bennett, A. A fresh look at inland fisheries and their role in food security and livelihoods. Fish Fish. 2019, 20, 1176–1195. [Google Scholar] [CrossRef]
- O’Meara, L.; Cohen, P.J.; Simmance, F.; Marinda, P.; Nagoli, J.; Teoh, S.J.; Funge-Smith, S.; Mills, D.J.; Thilsted, S.H.; Byrd, K.A. Inland fisheries critical for the diet quality of young children in sub-Saharan Africa. Glob. Food Secur. 2021, 28, 100483. [Google Scholar] [CrossRef]
- Kawarazuka, N.; Béné, C. The potential role of small fish species in improving micronutrient deficiencies in developing countries: Building evidence. Public Health Nutr. 2011, 14, 1927–1938. [Google Scholar] [CrossRef]
- Thilsted, S.H.; Thorne-Lyman, A.; Subasinghe, R.; Webb, P.; Bogard, J.R.; Phillips, M.J.; Allison, E.H. Sustaining healthy diets: The role of capture fisheries and aquaculture for improving nutrition in the post-2015 era. Food Policy 2016, 61, 126–131. [Google Scholar] [CrossRef]
- de Bruyn, J.; Wesana, J.; Bunting, S.W.; Thilsted, S.H.; Cohen, P.J. Fish Acquisition and Consumption in the African Great Lakes Region through a Food Environment Lens: A Scoping Review. Nutrients 2021, 13, 2408. [Google Scholar] [CrossRef]
- Hicks, C.C.; Cohen, P.J.; Graham, N.A.J.; Nash, K.L.; Allison, E.H.; D’Lima, C.; Mills, D.J.; Roscher, M.; Thilsted, S.H.; Thorne-Lyman, A.L.; et al. Harnessing global fisheries to tackle micronutrient deficiencies. Nature 2019, 574, 95–98. [Google Scholar] [CrossRef]
- Lynch, A.J.; Cowx, I.G.; Fluet-Chouinard, E.; Glaser, S.M.; Phang, S.C.; Beard, T.D.; Bower, S.D.; Brooks, J.L.; Bunnell, D.B.; Claussen, J.E.; et al. Inland fisheries—Invisible but integral to the UN Sustainable Development Agenda for ending poverty by 2030. Glob. Environ. Chang. 2017, 47, 167–173. [Google Scholar] [CrossRef]
- Chan, C.Y.; Tran, N.; Pethiyagoda, S.; Crissman, C.C.; Sulser, T.B.; Phillips, M.J. Prospects and challenges of fish for food security in Africa. Glob. Food Secur. 2019, 20, 17–25. [Google Scholar] [CrossRef]
- Chikowi, C.T.M.; Ochieng, D.O.; Jumbe, C.B.L. Consumer choices and demand for Tilapia in urban Malawi: What are the complementarities and trade-offs? Aquaculture 2021, 530, 735755. [Google Scholar] [CrossRef]
- Genschick, S.; Marinda, P.; Tembo, G.; Kaminski, A.M.; Thilsted, S.H. Fish consumption in urban Lusaka: The need for aquaculture to improve targeting of the poor. Aquaculture 2018, 492, 280–289. [Google Scholar] [CrossRef]
- Aiga, H.; Matsuoka, S.; Kuroiwa, C.; Yamamoto, S. Malnutrition among children in rural Malawian fish-farming households. Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 827–833. [Google Scholar] [CrossRef] [PubMed]
- Obiero, K.; Meulenbroek, P.; Drexler, S.; Dagne, A.; Akoll, P.; Odong, R.; Kaunda-Arara, B.; Waidbacher, H. The Contribution of Fish to Food and Nutrition Security in Eastern Africa: Emerging Trends and Future Outlooks. Sustainability 2019, 11, 1636. [Google Scholar] [CrossRef]
- Kaminski, A.M.; Genschick, S.; Kefi, A.S.; Kruijssen, F. Commercialization and upgrading in the aquaculture value chain in Zambia. Aquaculture 2018, 493, 355–364. [Google Scholar] [CrossRef]
- Avadí, A.; Cole, S.M.; Kruijssen, F.; Dabat, M.H.; Mungule, C.M. How to enhance the sustainability and inclusiveness of smallholder aquaculture production systems in Zambia? Aquaculture 2022, 547, 737494. [Google Scholar] [CrossRef]
- Harris, J.; Chisanga, B.; Drimie, S.; Kennedy, G. Nutrition transition in Zambia: Changing food supply, food prices, household consumption, diet and nutrition outcomes. Food Secur. 2019, 11, 371–387. [Google Scholar] [CrossRef]
- Longley, C.; Thilsted, S.H.; Beveridge, M.; Cole, S.; Nyirenda, D.B.; Heck, S.; Hother, A.-L. The Role of Fish in the First 1000 Days in Zambia; IDS Special Collection; Institute of Development Studies: Brighton, UK, 2014; Available online: https://opendocs.ids.ac.uk/opendocs/handle/123456789/4384 (accessed on 1 November 2021).
- Marinda, P.A.; Genschick, S.; Khayeka-Wandabwa, C.; Kiwanuka-Lubinda, R.; Thilsted, S.H. Dietary diversity determinants and contribution of fish to maternal and under-five nutritional status in Zambia. PLoS ONE 2018, 13, e0204009. [Google Scholar] [CrossRef]
- Hasselberg, A.E.; Wessels, L.; Aakre, I.; Reich, F.; Atter, A.; Steiner-Asiedu, M.; Amponsah, S.; Pucher, J.; Kjellevold, M. Composition of nutrients, heavy metals, polycyclic aromatic hydrocarbons and microbiological quality in processed small indigenous fish species from Ghana: Implications for food security. PLoS ONE 2020, 15, e0242086. [Google Scholar] [CrossRef]
- Roos, N.; Leth, T.; Jakobsen, J.; Thilsted, S.H. High vitamin A content in some small indigenous fish species in Bangladesh: Perspectives for food-based strategies to reduce vitamin A deficiency. Int. J. Food Sci. Nutr. 2002, 53, 425–437. [Google Scholar] [CrossRef]
- Bogard, J.R.; Thilsted, S.H.; Marks, G.C.; Wahab, M.A.; Hossain, M.A.R.; Jakobsen, J.; Stangoulis, J. Nutrient composition of important fish species in Bangladesh and potential contribution to recommended nutrient intakes. J. Food Compos. Anal. 2015, 42, 120–133. [Google Scholar] [CrossRef]
- Roos, N.; Wahab, M.A.; Hossain, M.A.R.; Thilsted, S.H. Linking human nutrition and fisheries: Incorporating micronutrient-dense, small indigenous fish species in carp polyculture production in Bangladesh. Food Nutr. Bull. 2007, 28, S280–S293. [Google Scholar] [CrossRef]
- Castine, S.A.; Bogard, J.R.; Barman, B.K.; Karim, M.; Mokarrom Hossain, M.; Kunda, M.; Mahfuzul Haque, A.B.M.; Phillips, M.J.; Thilsted, S.H. Homestead pond polyculture can improve access to nutritious small fish. Food Secur. 2017, 9, 785–801. [Google Scholar] [CrossRef]
- Wahab, M.A.; Kadir, A.; Milstein, A.; Kunda, M. Manipulation of species combination for enhancing fish production in polyculture systems involving major carps and small indigenous fish species. Aquaculture 2011, 321, 289–297. [Google Scholar] [CrossRef]
- Milstein, A. Ecological aspects of fish species interactions in polyculture ponds. Hydrobiologia 1992, 231, 177–186. [Google Scholar] [CrossRef]
- Robinson, E.H.; Li, M.H. Channel catfish Ictalurus punctatus size and feed conversion ratio. J. World Aquac. Soc. 2010, 41, 829–833. [Google Scholar] [CrossRef]
- Ahmad, S.A.S.; Bart, A.N.; Yi, Y.; Rakocy, J.E.; Diana, J.S. The effect of the introduction of Nile tilapia (Oreochromis niloticus L.) on small indigenous fish species (mola, Amblypharyngodon mola, Hamilton; chela, Chela cachius, Hamilton; punti, Puntius sophore, Hamilton). Aquac. Res. 2010, 41, 904–912. [Google Scholar] [CrossRef]
- Karim, M.; Little, D.C.; Kabir, M.S.; Verdegem, M.J.C.; Telfer, T.; Wahab, M.A. Enhancing benefits from polycultures including tilapia (Oreochromis niloticus) within integrated pond-dike systems: A participatory trial with households of varying socio-economic level in rural and peri-urban areas of Bangladesh. Aquaculture 2011, 314, 225–235. [Google Scholar] [CrossRef]
- Brummett, R.E.; Katambalika, K. Protocols for the development of indigenous species: Polyculture of indigenous species under Malawian smallholder conditions. Aquac. Res. 1996, 27, 225–233. [Google Scholar] [CrossRef]
- Kakwasha, K.; Mudege, N.N.; Sichilima, T.; Sebele, M.; Nabiwa, L.; Lundeba, M. Smallholder Fish Farmers Population Census Report 2020: Northern and Luapula Provinces, Zambia. Penang, Malaysia: WorldFish. Program Report: 2020-40. 2020. Available online: https://digitalarchive.worldfishcenter.org/handle/20.500.12348/4500?show=full (accessed on 1 November 2021).
- Lundeba, M.; Cole, S.M.; Mekkawy, W.; Yossa, R.; Basiita, R.K.; Nyirenda, M.; Muyuni, N.; Benzie, J.A.H. On-farm participatory evaluation of feeding approaches used by farmers for tilapia (Oreochromis macrochir) production in northern Zambia. Aquaculture 2022, 549, 737–747. [Google Scholar] [CrossRef]
- Kaminski, A.; Gellner, M.; Giese, D.; Jabborov, S.; Lootz, M.; Lundeba, M.; Nyika, B.; Patt, N.; Sadeghi, A.; Siachinga, M. Opportunities and Challenges for Small-Scale Aquaculture in Zambia; Humboldt-Universität zu Berlin, SLE Postgraduate Studies on International Cooperation for Sustainable Development (SLE): Berlin, Germany, 2019. [Google Scholar]
- Nkonde, C.; Audain, K.; Kiwanuka-Lubinda, R.N.; Marinda, P. Effect of agricultural diversification on dietary diversity in rural households with children under 5 years of age in Zambia. Food Sci. Nutr. 2021, 9, 6274–6285. [Google Scholar] [CrossRef]
- Brummett, R.E.; Lazard, J.; Moehl, J. African aquaculture: Realizing the potential. Food Policy 2008, 33, 371–385. [Google Scholar] [CrossRef]
- Genschick, S.; Kaminski, A.; Cole, S.; Tran, N.; Chimatiro, S.; Lundeba, M. Toward More Inclusive and Sustainable Development of Zambian Aquaculture. Penang, Malaysia: CGIAR Research Program on Fish Agri-Food Systems Program Brief: FISH-2017-07 12. 2017. Available online: http://pubs.iclarm.net/resource_centre/FISH-2017-07.pdf (accessed on 1 November 2021).
- Gellner, M. Polyculture in Semi-Concrete Ponds of Oreochromis macrochir, Barbus radiatus and Marcusenius macrolepidotus Cultured under Different Combinations in Northern Zambia. Master’s Thesis, Albrecht Daniel Thaer-Institute of Agricultural and Horticultural Sciences, Humboldt-University, Berlin, Germany, 2017. [Google Scholar]
- Ruel, M.T.; Alderman, H. Nutrition-sensitive interventions and programmes: How can they help to accelerate progress in improving maternal and child nutrition? Lancet 2013, 382, 536–551. [Google Scholar] [CrossRef]
- Ruel, M.T.; Quisumbing, A.R.; Balagamwala, M. Nutrition-sensitive agriculture: What have we learned so far? Glob. Food Secur. 2018, 17, 128–153. [Google Scholar] [CrossRef]
- Simmance, F.A.; Cohen, P.J.; Huchery, C.; Sutcliffe, S.; Suri, S.K.; Tezzo, X.; Thilsted, S.H.; Oosterveer, P.; McDougall, C.; Ahern, M.; et al. Nudging fisheries and aquaculture research towards food systems. Fish Fish. 2022, 23, 34–53. [Google Scholar] [CrossRef]
- Tezzo, X.; Bush, S.R.; Oosterveer, P.; Belton, B. Food system perspective on fisheries and aquaculture development in Asia. Agric. Hum. Values 2021, 38, 73–90. [Google Scholar] [CrossRef]
- Dinerstein, E.; Olson, D.; Joshi, A.; Vynne, C.; Burgess, N.D.; Wikramanayake, E.; Hahn, N.; Palminteri, S.; Hedao, P.; Noss, R.; et al. An Ecoregion-Based Approach to Protecting Half the Terrestrial Realm. Bioscience 2017, 67, 534–545. [Google Scholar] [CrossRef] [PubMed]
- Linke, S.; Lehner, B.; Ouellet Dallaire, C.; Ariwi, J.; Grill, G.; Anand, M.; Beames, P.; Burchard-Levine, V.; Maxwell, S.; Moidu, H.; et al. Global hydro-environmental sub-basin and river reach characteristics at high spatial resolution. Sci. Data 2019, 6, 283. [Google Scholar] [CrossRef]
- Lehner, B.; WWF-Zambia. HydroATLAS-Zambia. Technical Documentation Version 1.0. Lusaka: WWF. 2020. Available online: https://www.hydrosheds.org/hydroatlas-zambia (accessed on 1 November 2021).
- Huchzermeyer, C.F. Fishes and Fisheries of the Bangweulu Wetlands and Lavushi Manda National Park. Master’s Thesis, Department of Ichthyology and Fisheries Science, Rhodes University, Makhanda, South Africa, 2013. [Google Scholar]
- Birbeck, G.; Chomba, E.; Atadzhanov, M.; Mbewe, E.; Haworth, A. The social and economic impact of epilepsy in Zambia: A cross-sectional study. Lancet Neurol. 2007, 6, 39–44. [Google Scholar] [CrossRef]
- Nölle, N.; Genschick, S.; Schwadorf, K.; Hrenn, H.; Brandner, S.; Biesalski, H.K. Fish as a source of (micro)nutrients to combat hidden hunger in Zambia. Food Secur. 2020, 12, 1385–1406. [Google Scholar] [CrossRef]
- World Health Organization/Food and Agriculture Organization (WHO/FAO). Vitamin and Mineral Requirements in Human Nutrition, 2nd ed.; FAO: Rome, Italy, 2004; Available online: https://www.who.int/publications/i/item/9241546123 (accessed on 1 November 2021).
- National Academies of Sciences, Engineering, and Medicine (NASEM). Dietary Reference Intakes for Sodium and Potassium; The National Academies Press: Washington, DC, USA, 2019. [Google Scholar] [CrossRef]
- World Health Organization/Food and Agriculture Organization (WHO/FAO). Fats and Fatty Acids in Human Nutrition: Report of an Expert Consultation; FAO: Rome, Italy, 2010; Food and Nutrition Paper 91. Available online: https://pubmed.ncbi.nlm.nih.gov/21812367/ (accessed on 1 November 2021).
- Zhang, Z.; Fulgoni, V.L.; Kris-Etherton, P.M.; Mitmesser, S.H. Dietary Intakes of EPA and DHA Omega-3 Fatty Acids among US Childbearing-Age and Pregnant Women: An Analysis of NHANES 2001–2014. Nutrients 2018, 10, 416. [Google Scholar] [CrossRef]
- National Academies of Sciences, Engineering, and Medicine (NASEM). Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids; The National Academies Press: Washington, DC, USA, 2005. [Google Scholar] [CrossRef]
- Hallström, E.; Bergman, K.; Mifflin, K.; Parker, R.; Tyedmers, P.; Troell, M.; Ziegler, F. Combined climate and nutritional performance of seafoods. J. Clean. Prod. 2019, 230, 402–411. [Google Scholar] [CrossRef]
- Tweddle, D.; Cowx, I.G.; Peel, R.A.; Weyl, O.L.F. Challenges in fisheries management in the Zambezi, one of the great rivers of Africa. Fish. Manag. Ecol. 2015, 22, 99–111. [Google Scholar] [CrossRef]
- Mussa, H.; Kaunda, E.; Chimatiro, S.; Kakwasha, K.; Banda, L.; Nankwenya, B.; Nyengere, J. Assessment of Informal Cross-Border Fish Trade in the Southern Africa Region: A Case of Malawi and Zambia. J. Agric. Sci. Technol. 2017, 7, 358–366. [Google Scholar] [CrossRef]
- Kaminski, A.M.; Kruijssen, F.; Cole, S.M.; Beveridge, M.C.M.; Dawson, C.; Mohan, C.V.; Suri, S.; Karim, M.; Chen, O.L.; Phillips, M.J.; et al. A review of inclusive business models and their application in aquaculture development. Rev. Aquac. 2020, 12, 1881–1902. [Google Scholar] [CrossRef]
- Thilsted, S.H. Diversifying aquaculture systems to nourish nations. Nat. Food 2021, 2, 450–451. [Google Scholar] [CrossRef]
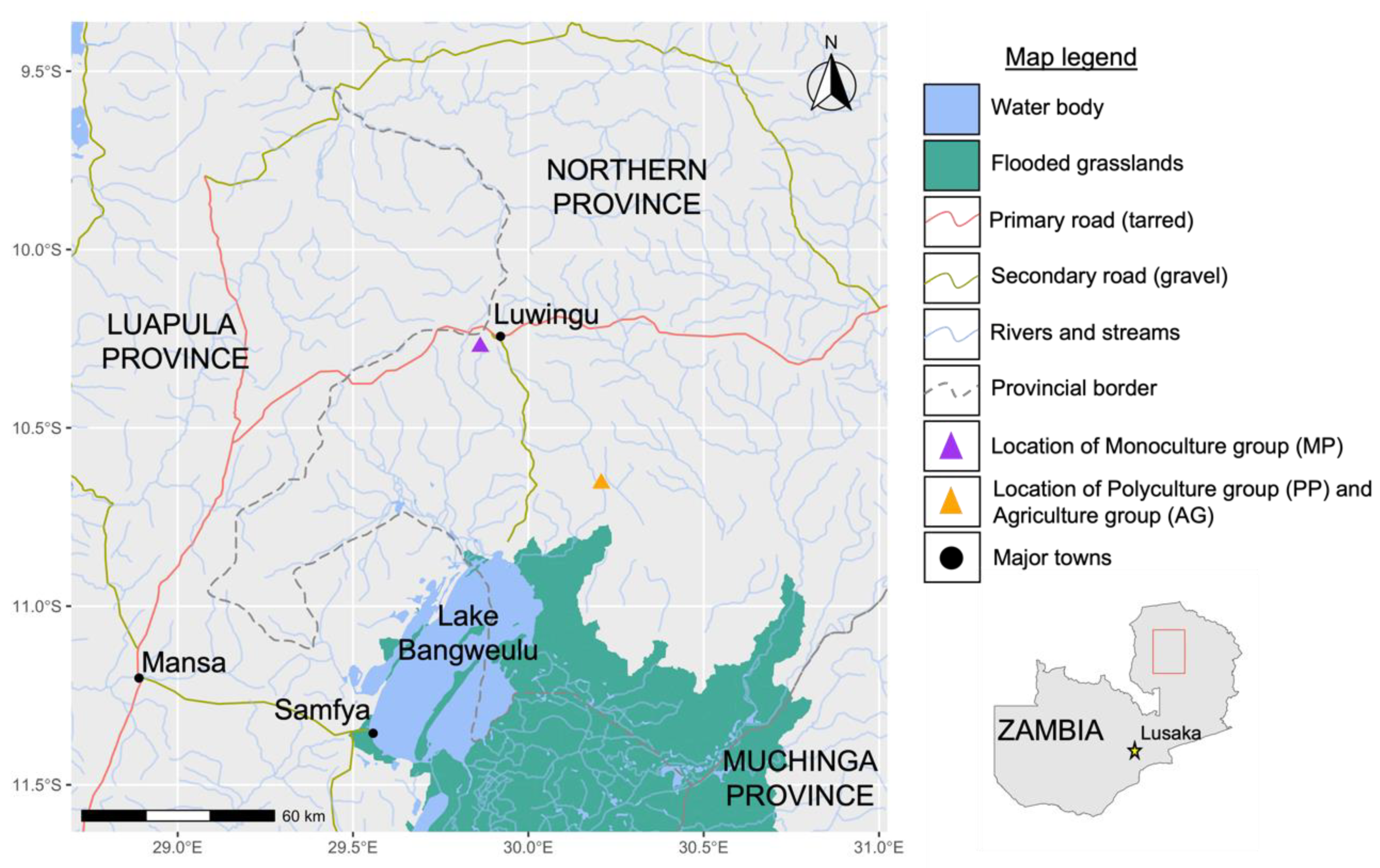
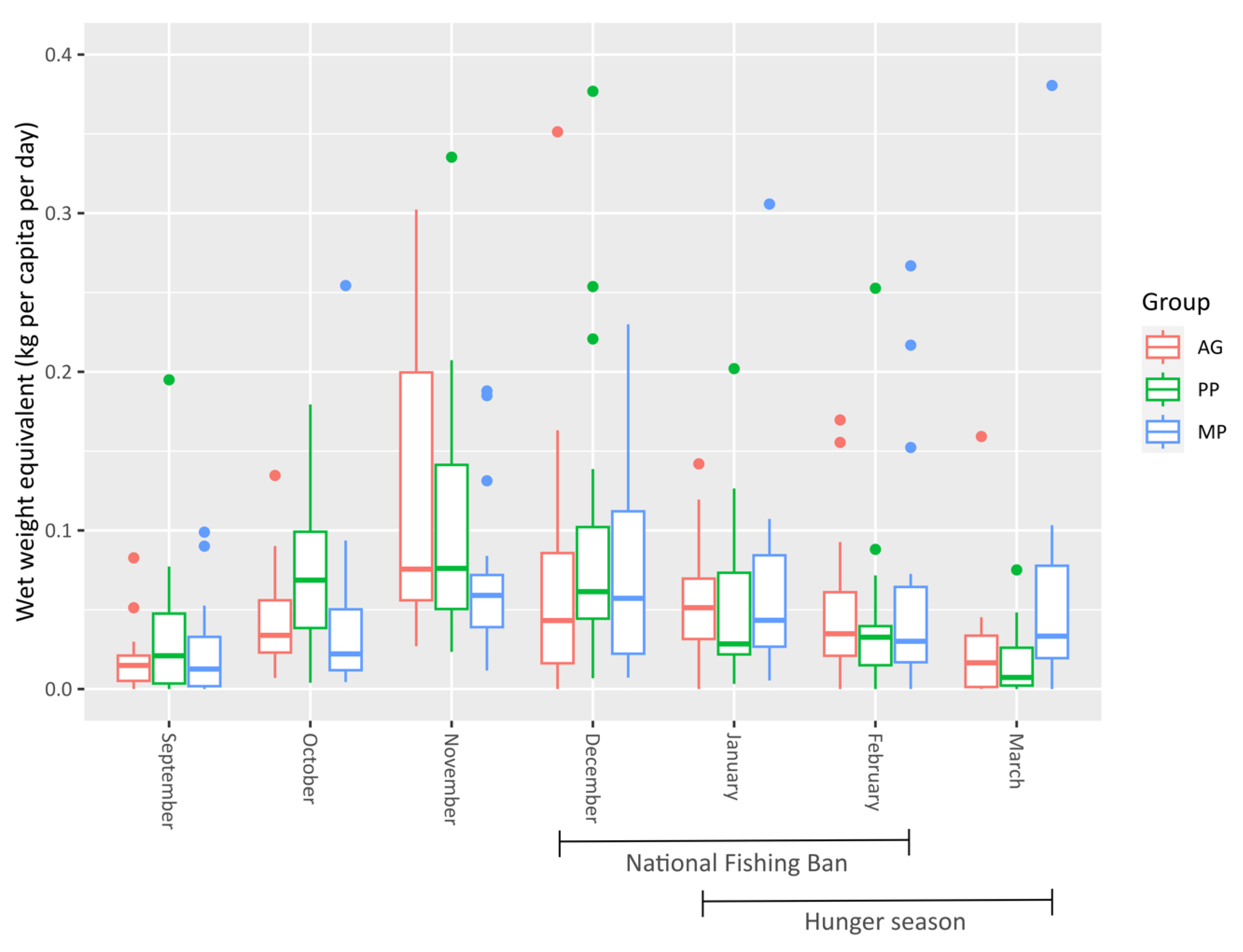
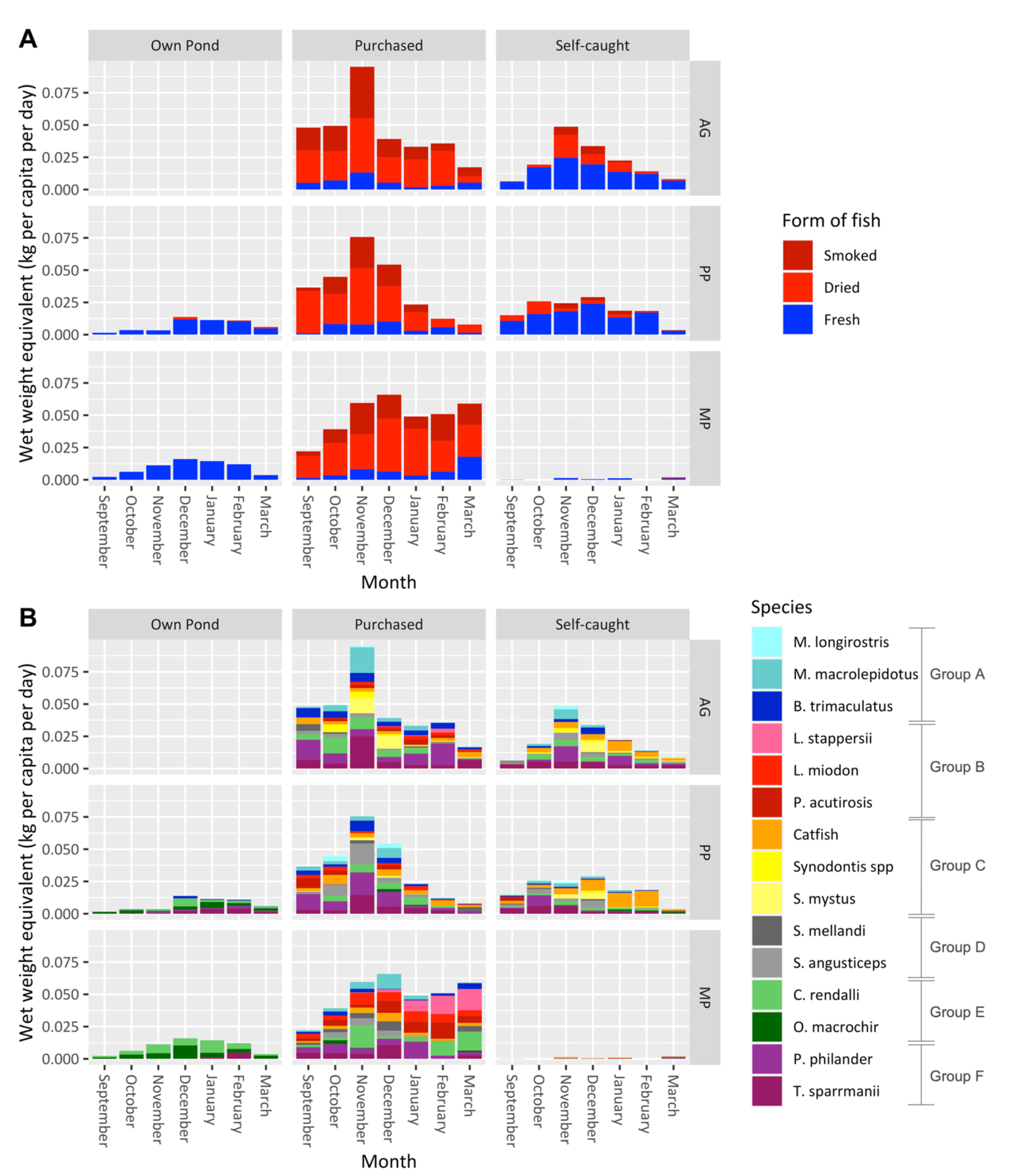
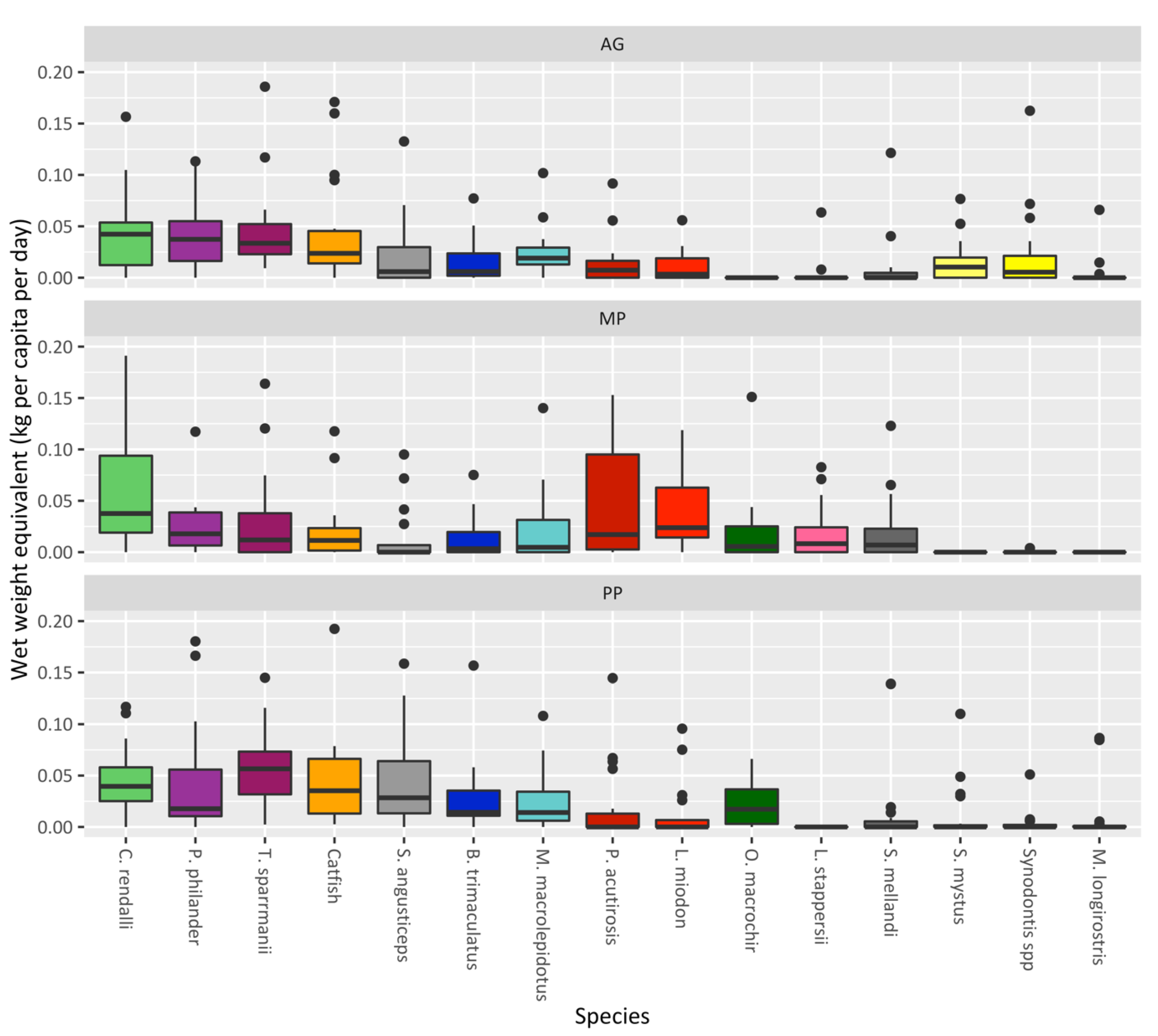
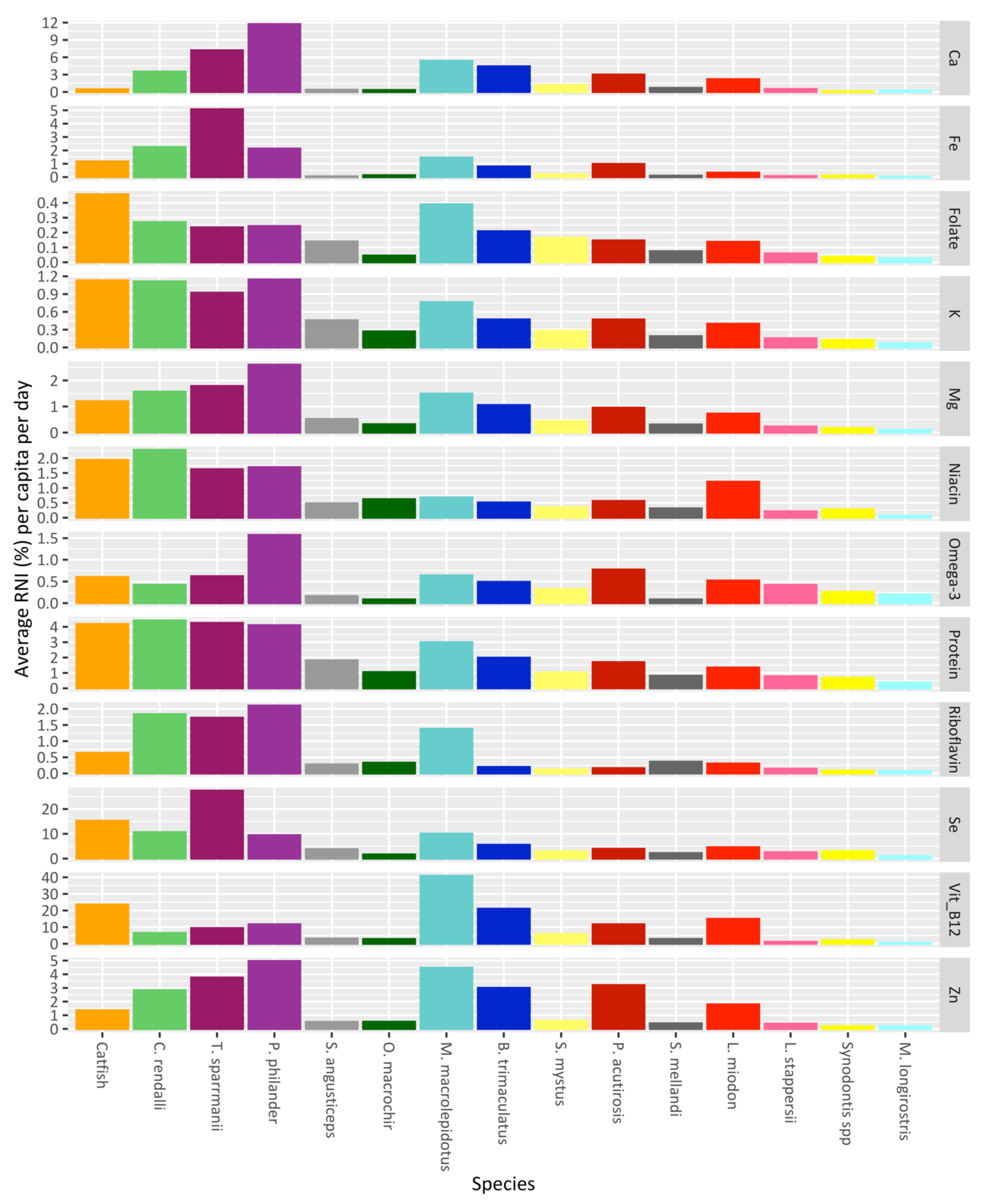
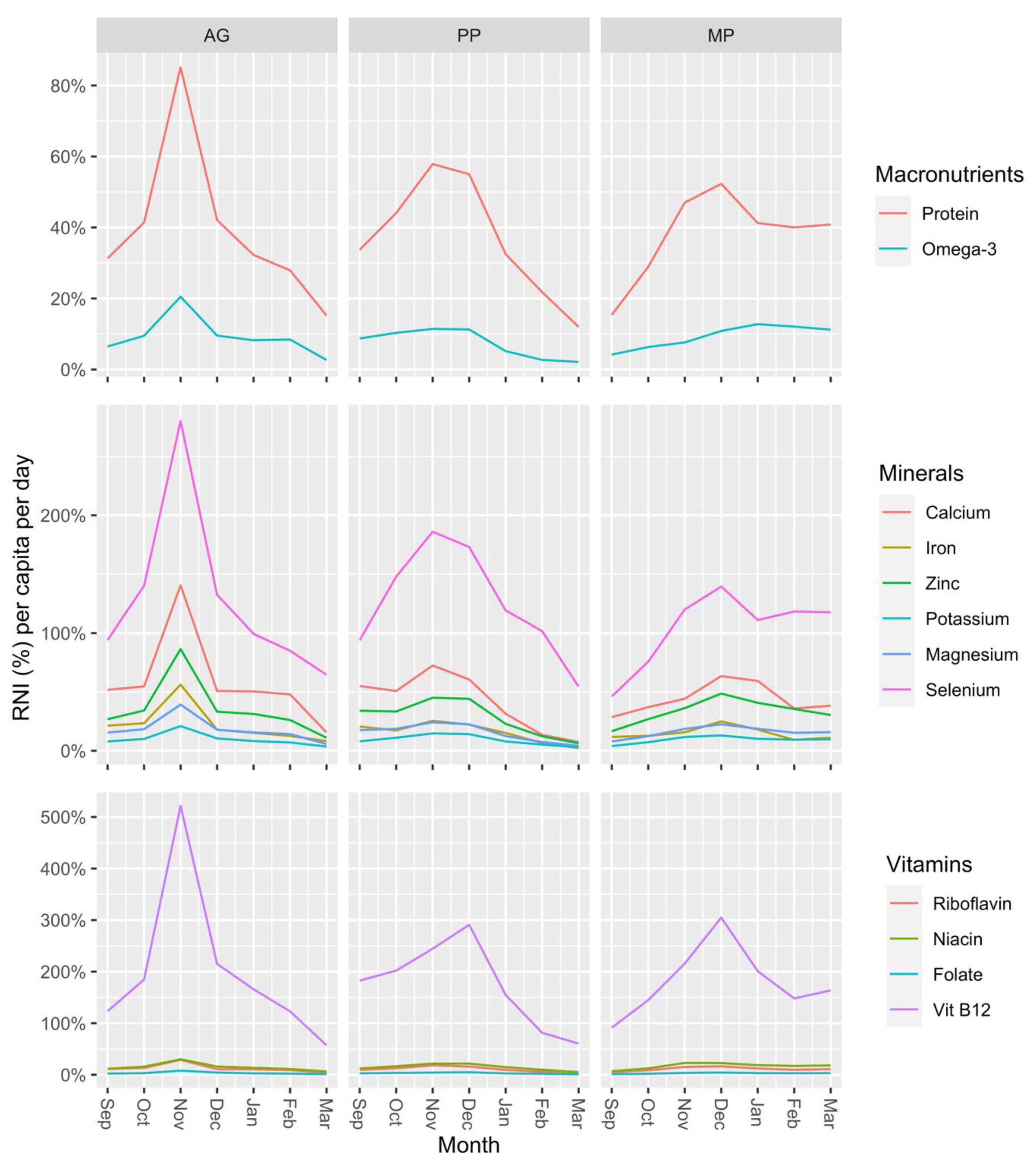
| Species | Total Fish | Number of Fish Stocked in Ponds (n = 20) | StockingDensity (Fish/m2) | Weight of Fish (g) | Length of Fish (cm) | ||||
|---|---|---|---|---|---|---|---|---|---|
| M | SD | M | SD | M | SD | M | SD | ||
| O. macrochir | 8554 | 427.26 | 11.63 | 1.98 | 1.01 | 13.37 | 4.63 | 8.56 | 1.00 |
| Small Indigenous Species | |||||||||
| T. sparrmanii | 2000 | 100 | 0.00 | 0.46 | 0.24 | 5.04 | 1.01 | 5.92 | 0.60 |
| P. philander | 2000 | 100 | 0.00 | 0.46 | 0.24 | 1.80 † | 0.84 † | 4.10† | 0.75 † |
| B. trimaculatus | 1000 | 50 | 0.00 | 0.23 | 0.12 | ||||
| Total (N = 53) a | Polyculture (PP) (n = 19) | Monoculture (MP) (n = 16) | Agriculture (AG) (n = 18) | |
|---|---|---|---|---|
| Age (Mean Years ± SD) | 40.6 ± 11.4 | 39.9 ± 10.1 | 44.9 ± 12.2 | 37.4 ± 11.5 |
| Education (Mean Years ± SD) | 7.6 ± 2.0 | 6.7 ± 2.3 | 7.7 ± 1.9 | 8.5 ± 1.3 |
| Household size (Mean No. of People ± SD) | 6.3 ± 2.5 | 6.2 ± 2.6 | 7.2 ± 2.4 | 5.6 ± 2.5 |
| Number of Children (Mean No. ± SD) | 4.3 ± 2.4 | 4.3 ± 2.4 | 4.7 ± 2.6 | 3.9 ± 2.4 |
| Marital Status (Freq. and % Single) | 14 (26%) | 6 (32%) | 1 (6%) | 6 (33%) |
| Head of Household (Freq. and % Female-headed) | 13 (25%) | 6 (32%) b | 1 (6%) | 6 (33%) |
| Average Disposable Income (Mean ZMW c ± SD) | 5265 ± 7982 | 5237 ± 10,943 | 6215 ± 6200 | 4449 ± 5709 |
| Category * | Scientific Name | Local Name | Frequency | Measured Weight (kg) | Wet Weight Equivalent (kg) |
|---|---|---|---|---|---|
| A: Mormyrids and barbs (wetland species) | Mormyrus longirstris | Mbubu | 38 | 13.7 | 33.9 |
| Marcusenius macrolepidotus | Mintesa | 278 | 119.6 | 234.1 | |
| Barbus trimaculatus † | Mushipa | 243 | 122 | 242.5 | |
| B: Pelagic small/medium fish | Luciolates stappersii | Buka-Buka | 59 | 63.3 | 141.8 |
| Limnothrissa miodon and Stolothrissa tanganicae | Kapenta | 138 | 71.9 | 197.6 | |
| Potamothrissa acutirostris and Poecilothrissa moeruensis | Chisense | 133 | 66.3 | 214.1 | |
| C: Catfishes (large and small) | Clarias spp. | Milonge | 465 | 333.4 | 350.7 |
| Syndontis spp. | Cingongo | 79 | 44.9 | 70.3 | |
| Schilbe mystus | Lupata | 41 | 70.7 | 120 | |
| D: Large cichlids | Sargochromis mellandi | Imbelya | 89 | 75.1 | 139.8 |
| Serranochromis angusticeps | Polwe | 133 | 157.7 | 274.4 | |
| E: Tilapias (often cultivated) | Coptodon rendalli | Mpende | 326 | 388 | 508.4 |
| Oreochromis machrochir | Nkamba | 121 | 178 | 193.3 | |
| F: Small cichlids from local capture fisheries | Pseudocrenilabrus philander | Cikundu | 384 | 165.2 | 480.2 |
| Tilapia sparrmanii | Matuku | 553 | 282.3 | 479.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaminski, A.M.; Little, D.C.; Middleton, L.; Syapwaya, M.; Lundeba, M.; Johnson, J.; Huchzermeyer, C.; Thilsted, S.H. The Role of Aquaculture and Capture Fisheries in Meeting Food and Nutrition Security: Testing a Nutrition-Sensitive Pond Polyculture Intervention in Rural Zambia. Foods 2022, 11, 1334. https://doi.org/10.3390/foods11091334
Kaminski AM, Little DC, Middleton L, Syapwaya M, Lundeba M, Johnson J, Huchzermeyer C, Thilsted SH. The Role of Aquaculture and Capture Fisheries in Meeting Food and Nutrition Security: Testing a Nutrition-Sensitive Pond Polyculture Intervention in Rural Zambia. Foods. 2022; 11(9):1334. https://doi.org/10.3390/foods11091334
Chicago/Turabian StyleKaminski, Alexander M., David C. Little, Lucinda Middleton, Muleya Syapwaya, Mary Lundeba, Jacob Johnson, Carl Huchzermeyer, and Shakuntala H. Thilsted. 2022. "The Role of Aquaculture and Capture Fisheries in Meeting Food and Nutrition Security: Testing a Nutrition-Sensitive Pond Polyculture Intervention in Rural Zambia" Foods 11, no. 9: 1334. https://doi.org/10.3390/foods11091334
APA StyleKaminski, A. M., Little, D. C., Middleton, L., Syapwaya, M., Lundeba, M., Johnson, J., Huchzermeyer, C., & Thilsted, S. H. (2022). The Role of Aquaculture and Capture Fisheries in Meeting Food and Nutrition Security: Testing a Nutrition-Sensitive Pond Polyculture Intervention in Rural Zambia. Foods, 11(9), 1334. https://doi.org/10.3390/foods11091334






