Use of Cocktail of Bacteriophage for Salmonella Typhimurium Control in Chicken Meat
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Culture Medium
2.2. Bacteriophage Isolation
2.3. Host Range
2.4. Lysis Kinetics
2.5. One Step Growth
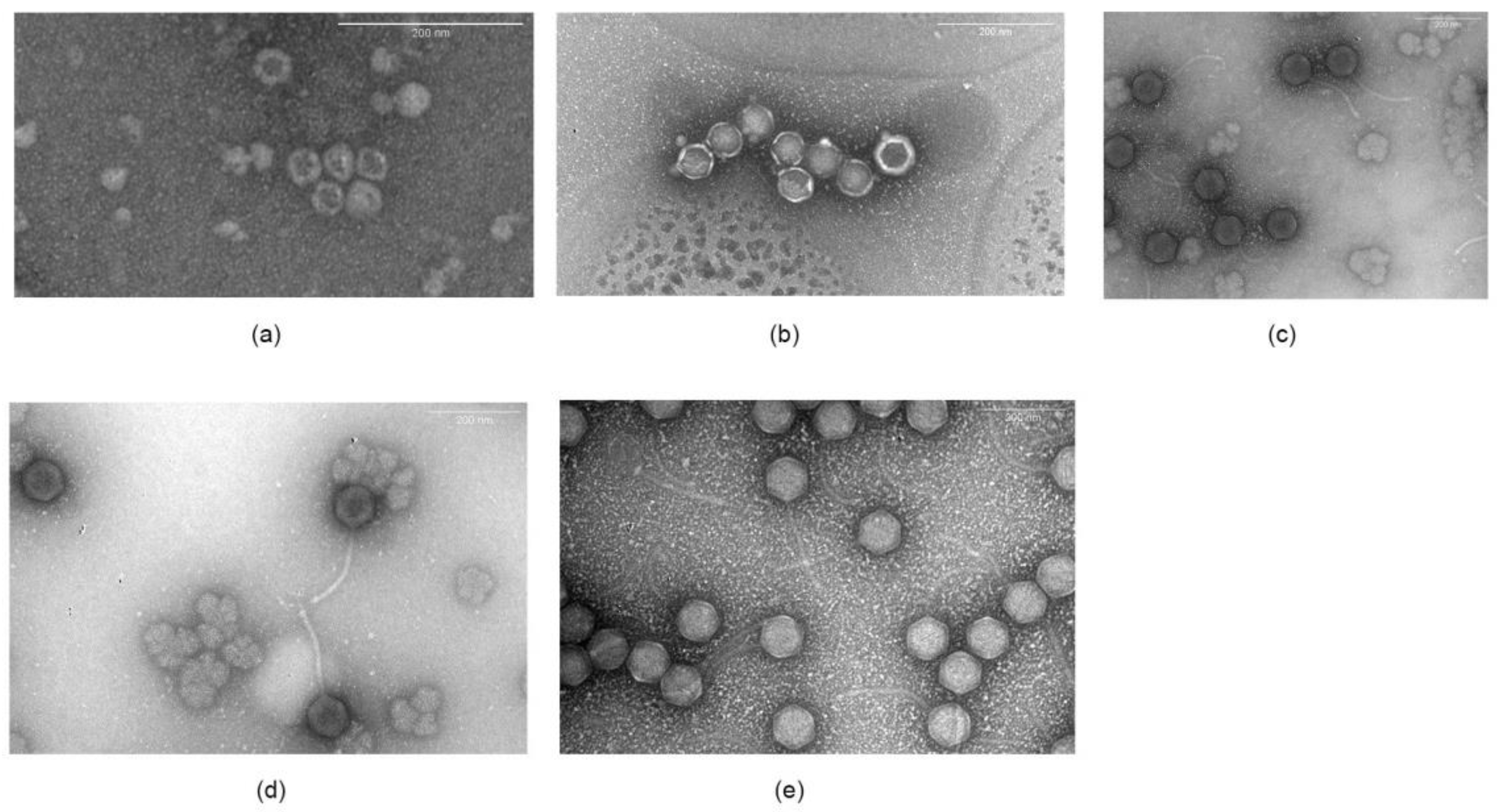
2.6. Electron Microscopy
2.7. DNA Extraction and Restriction of Enzyme Digestion
2.8. Assay in Chicken Meat
2.9. Statistical Analysis
3. Results
3.1. Bacteriophage Isolation
3.2. Characterization of Phages
3.2.1. Host Range and Selection of Phage
3.2.2. Microscopy
3.3. One-Step Growth and Lysis Kinetics
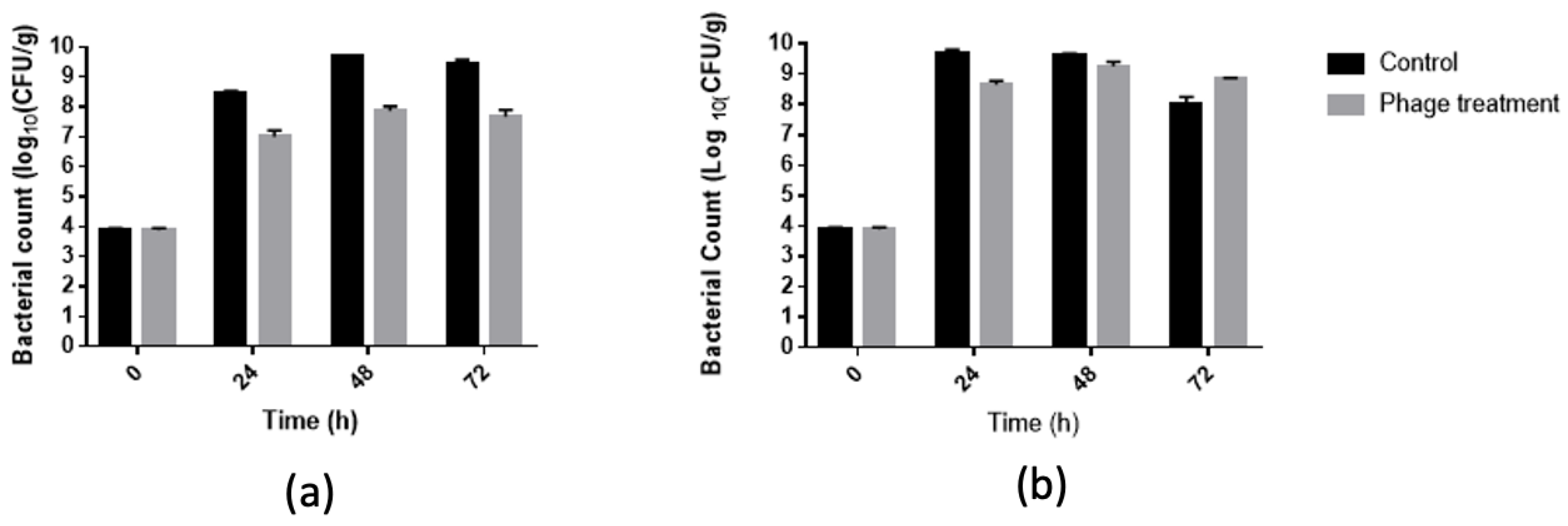
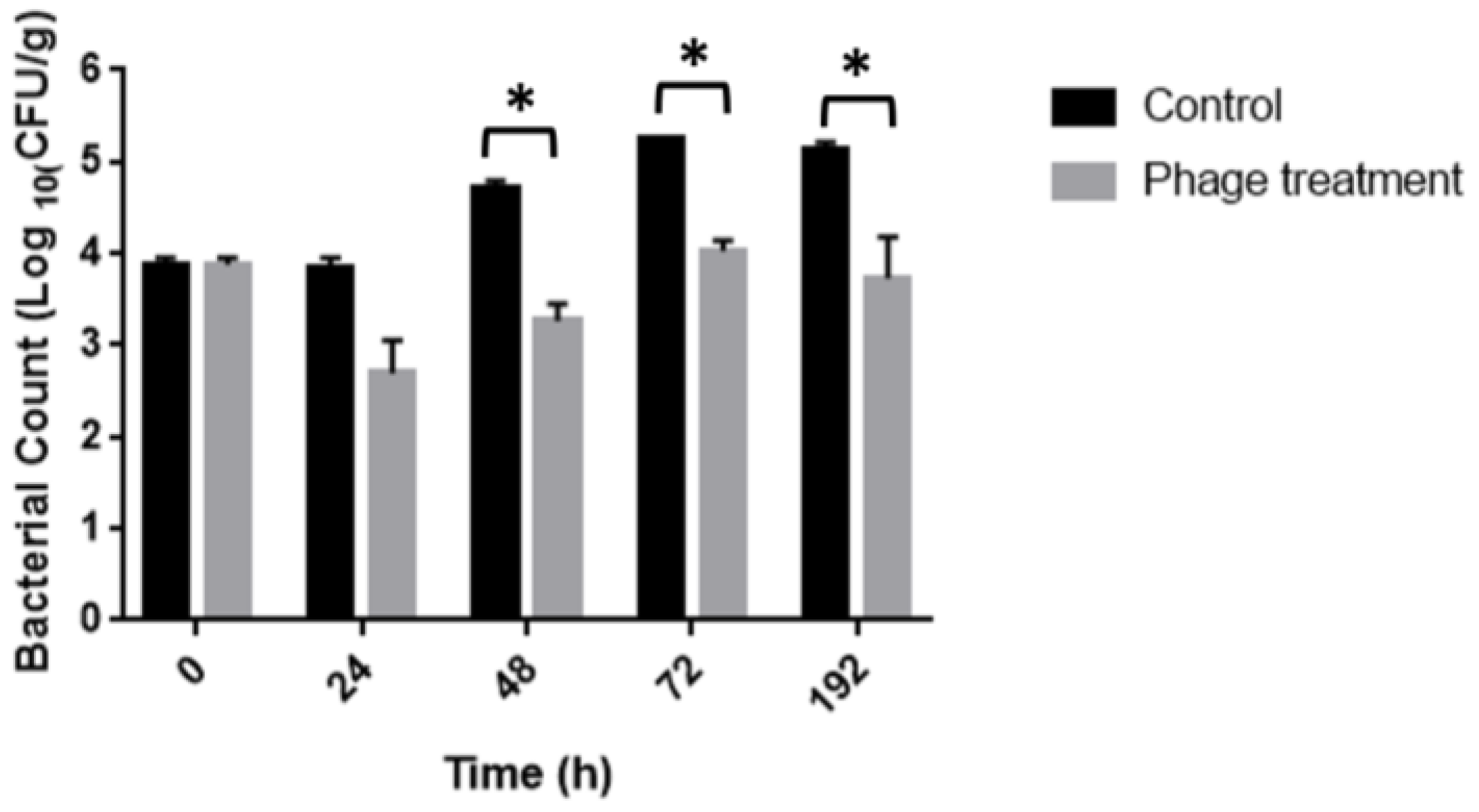
3.4. Salmonella Reduction in Chicken Meat
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shi, X.; Namvar, A.; Kostrzynska, M.; Hora, R.; Warriner, K. Persistence and Growth of Different Salmonella Serovars on Pre- and Postharvest Tomatoes. J. Food Prot. 2007, 70, 2725–2731. [Google Scholar] [CrossRef] [PubMed]
- Nesse, L.L.; Nordby, K.; Heir, E.; Bergsjoe, B.; Vardund, T.; Nygaard, H.; Holstad, G. Molecular Analyses of Salmonella Enterica Isolates from Fish Feed Factories and Fish Feed Ingredients. Appl. Environ. Microbiol. 2003, 69, 1075–1081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Condell, O.; Iversen, C.; Cooney, S.; Power, K.A.; Walsh, C.; Burgess, C.; Fanning, S. Efficacy of Biocides Used in the Modern Food Industry to Control Salmonella Enterica, and Links between Biocide Tolerance and Resistance to Clinically Relevant Antimicrobial Compounds. Appl. Environ. Microbiol. 2012, 78, 3087–3097. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Threlfall, E.J. Antimicrobial Drug Resistance in Salmonella: Problems and Perspectives in Food- and Water-Borne Infections. FEMS Microbiol. Rev. 2002, 26, 141–148. [Google Scholar] [CrossRef]
- CDC. CDC Yellow Book 2020: Health Information for International Travel; Brunette, G.W., Nemhauser, J.B., Eds.; Oxford University Press: Oxford, UK, 2020; ISBN 978-0190065973. [Google Scholar]
- Cosby, D.E.; Cox, N.A.; Harrison, M.A.; Wilson, J.L.; Jeff Buhr, R.; Fedorka-Cray, P.J. Salmonella and Antimicrobial Resistance in Broilers: A Review. J. Appl. Poult. Res. 2015, 24, 408–426. [Google Scholar] [CrossRef]
- Hugas, M.; Beloeil, P.A. Controlling Salmonella along the Food Chain in the European Union-Progress over the Last Ten Years. Eurosurveillance 2014, 19, 208204. [Google Scholar] [CrossRef] [Green Version]
- Foley, S.L.; Nayak, R.; Hanning, I.B.; Johnson, T.J.; Han, J.; Ricke, S.C. Population Dynamics of Salmonella Enterica Serotypes in Commercial Egg and Poultry Production. Appl. Environ. Microbiol. 2011, 77, 4273–4279. [Google Scholar] [CrossRef] [Green Version]
- Jones, F.T.; Ricke, S.C. Observations on the History of the Development of Antimicrobials and Their Use in Poultry Feeds. Poult. Sci. 2003, 82, 613–617. [Google Scholar] [CrossRef]
- Castanon, J.I.R. History of the Use of Antibiotic as Growth Promoters in European Poultry Feeds. Poult. Sci. 2007, 86, 2466–2471. [Google Scholar] [CrossRef]
- García, P.; Martínez, B.; Obeso, J.M.; Rodríguez, A. Bacteriophages and Their Application in Food Safety. Lett. Appl. Microbiol. 2008, 47, 479–485. [Google Scholar] [CrossRef]
- Hagens, S.; Loessner, M.J. Bacteriophage for Biocontrol of Foodborne Pathogens: Calculations and Considerations. Curr. Pharm. Biotechnol. 2010, 11, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Goodridge, L.D.; Bisha, B. Phage-Based Biocontrol Strategies to Reduce Foodborne Pathogens in Foods. Bacteriophage 2011, 1, 130–137. [Google Scholar] [CrossRef] [Green Version]
- Wernicki, A.; Nowaczek, A.; Urban-Chmiel, R. Bacteriophage Therapy to Combat Bacterial Infections in Poultry. Virol. J. 2017, 14, 179. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Shi, J.; Ma, W.; Li, Z.; Wang, J.; Li, J.; Wang, X. Isolation, Characterization, and Application of a Novel Specific Salmonella Bacteriophage in Different Food Matrices. Food Res. Int. 2018, 111, 631–641. [Google Scholar] [CrossRef] [PubMed]
- Endersen, L.; O’Mahony, J.; Hill, C.; Ross, R.P.; McAuliffe, O.; Coffey, A. Phage Therapy in the Food Industry. Annu. Rev. Food Sci. Technol. 2014, 5, 327–349. [Google Scholar] [CrossRef] [PubMed]
- Blair, J.E. Bacteriophages. Mark H. Adams, with Chapters by E. S. Anderson, J. S. Gots, F. Jacob and E. L. Wollman. Interscience Publishers, Inc., New York, 1959. Illustrated, pp. Xviii + 592, $15.00. Clin. Chem. 1959, 5, 634. [Google Scholar] [CrossRef]
- Janež, N.; Loc-Carrillo, C. Use of Phages to Control Campylobacter spp. J. Microbiol. Methods 2013, 95, 68–75. [Google Scholar] [CrossRef] [Green Version]
- Clokie, M.R.J.; Kropinski, A.M. Methods and Protocols. In Bacteriophages; Humana Press: Totowa, NJ, USA, 2018. [Google Scholar]
- Deveau, H.; Labrie, S.J.; Chopin, M.C.; Moineau, S. Biodiversity and Classification of Lactococcal Phages. Appl. Environ. Microbiol. 2006, 72, 4338–4346. [Google Scholar] [CrossRef] [Green Version]
- Pickard, D.J.J. Preparation of Bacteriophage Lysates and Pure DNA. Methods Mol. Biol. 2009, 502, 3–9. [Google Scholar] [CrossRef]
- Moye, Z.D.; Woolston, J.; Sulakvelidze, A. Bacteriophage Applications for Food Production and Processing. Viruses 2018, 10, 205. [Google Scholar] [CrossRef] [Green Version]
- Xiang, Y.; Li, F.; Dong, N.; Tian, S.; Zhang, H.; Du, X.; Zhou, X.; Xu, X.; Yang, H.; Xie, J.; et al. Investigation of a Salmonellosis Outbreak Caused by Multidrug Resistant Salmonella Typhimurium in China. Front. Microbiol. 2020, 11, 801. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, M.; Viazis, S.; Diez-Gonzalez, F. Isolation, Identification and Characterization of Lytic, Wide Host Range Bacteriophages from Waste Effluents against Salmonella Enterica Serovars. Food Control 2014, 38, 67–74. [Google Scholar] [CrossRef]
- Li, M.; Lin, H.; Wang, L.; Wang, J. Complete Genome Sequence of the Extreme-PH-Resistant Salmonella Bacteriophage Aα of the Family Microviridae. Arch. Virol. 2021, 166, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Székely, A.J.; Breitbart, M. Single-Stranded DNA Phages: From Early Molecular Biology Tools to Recent Revolutions in Environmental Microbiology. FEMS Microbiol. Lett. 2016, 363. [Google Scholar] [CrossRef] [PubMed]
- Fiorentin, L.; Vieria, N.D.; Barioni Júnior, W. Use of Lytic Bacteriophages to Reduce Salmonella Enteritidis in Experimentally Contaminated Chicken Cuts. Braz. J. Poult. Sci. 2005, 7, 255–260. [Google Scholar] [CrossRef] [Green Version]
- Hungaro, H.M.; Mendonça, R.C.S.; Gouvêa, D.M.; Vanetti, M.C.D.; de Oliveira Pinto, C.L. Use of Bacteriophages to Reduce Salmonella in Chicken Skin in Comparison with Chemical Agents. Food Res. Int. 2013, 52, 75–81. [Google Scholar] [CrossRef] [Green Version]
- Sukumaran, A.T.; Nannapaneni, R.; Kiess, A.; Sharma, C.S. Reduction of Salmonella on Chicken Breast Fillets Stored under Aerobic or Modified Atmosphere Packaging by the Application of Lytic Bacteriophage Preparation SalmoFreshTM. Poult. Sci. 2016, 95, 668–675. [Google Scholar] [CrossRef]
- Higgins, J.P.; Higgins, S.E.; Guenther, K.L.; Huff, W.; Donoghue, A.M.; Donoghue, D.J.; Hargis, B.M. Use of a Specific Bacteriophage Treatment to Reduce Salmonella in Poultry Products. Poult. Sci. 2005, 84, 1141–1145. [Google Scholar] [CrossRef]
- Juneja, V.K.; Fan, X.; Peña-Ramos, A.; Diaz-Cinco, M.; Pacheco-Aguilar, R. The Effect of Grapefruit Extract and Temperature Abuse on Growth of Clostridium Perfringens from Spore Inocula in Marinated, Sous-Vide Chicken Products. Innov. Food Sci. Emerg. Technol. 2006, 7, 100–106. [Google Scholar] [CrossRef]
- Alonso-Hernando, A.; Alonso-Calleja, C.; Capita, R. Effectiveness of Several Chemical Decontamination Treatments against Gram-Negative Bacteria on Poultry during Storage under Different Simulated Cold Chain Disruptions. Food Control 2013, 34, 574–580. [Google Scholar] [CrossRef]
- Han, H.; Wei, X.; Wei, Y.; Zhang, X.; Li, X.; Jiang, J.; Wang, R. Isolation, Characterization, and Bioinformatic Analyses of Lytic Salmonella Enteritidis Phages and Tests of Their Antibacterial Activity in Food. Curr. Microbiol. 2017, 74, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Sritha, K.S.; Bhat, S.G. In Vitro Efficiency Evaluation of Phage Cocktail for Biocontrol of Salmonella Spp. in Food Products. Arch. Microbiol. 2021, 203, 5445–5452. [Google Scholar] [CrossRef]
- Grygorcewicz, B.; Chajęcka-Wierzchowska, W.; Augustyniak, A.; Wasak, A.; Stachurska, X.; Nawrotek, P.; Dołęgowska, B. In-Milk Inactivation of Escherichia Coli O157:H7 by the Environmental Lytic Bacteriophage ECPS-6. J. Food Saf. 2020, 40, e12747. [Google Scholar] [CrossRef]
- Hammerl, J.A.; Barac, A.; Erben, P.; Fuhrmann, J.; Gadicherla, A.; Kumsteller, F.; Lauckner, A.; Müller, F.; Hertwig, S. Properties of Two Broad Host Range Phages of Yersinia Enterocolitica Isolated from Wild Animals. Int. J. Mol. Sci. 2021, 22, 11381. [Google Scholar] [CrossRef]
- Tokman, J.I.; Kent, D.J.; Wiedmann, M.; Denes, T. Temperature Significantly Affects the Plaquing and Adsorption Efficiencies of Listeria Phages. Front. Microbiol. 2016, 7, 631. [Google Scholar] [CrossRef]
- D’Aoust, J.Y. Salmonella and the International Food Trade. Int. J. Food Microbiol. 1994, 24, 11–31. [Google Scholar] [CrossRef]
- Hong, Y.; Schmidt, K.; Marks, D.; Hatter, S.; Marshall, A.; Albino, L.; Ebner, P. Treatment of Salmonella-Contaminated Eggs and Pork with a Broad-Spectrum, Single Bacteriophage: Assessment of Efficacy and Resistance Development. Foodborne Pathog. Dis. 2016, 13, 679–688. [Google Scholar] [CrossRef]
- Møretrø, T.; Heir, E.; Nesse, L.L.; Vestby, L.K.; Langsrud, S. Control of Salmonella in Food Related Environments by Chemical Disinfection. Food Res. Int. 2012, 45, 532–544. [Google Scholar] [CrossRef]
- Nabil, N.M.; Tawakol, M.M.; Hassan, H.M. Assessing the Impact of Bacteriophages in the Treatment of Salmonella in Broiler Chickens. Infect. Ecol. Epidemiol. 2018, 8, 1539056. [Google Scholar] [CrossRef]


| Salmonella enterica Serovar | Phage | Total Phage | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A1 | A2 | A3 | A4 * | A5 * | A5.2 * | A6 | A7 * | A8 * | A9 * | A10 | B1 | B2 | B3 * | B4 | B5 | B6 * | B7 | B8 * | B9 | ||
| Choleraesiu FSL R9-1343 a | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 10 | ||||||||||
| Javiana FSL S5-406 a | 3 | 3 | 2 | ||||||||||||||||||
| Infantis FSL S5-506 a | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 10 | ||||||||||
| Typhimuriu FSL A4-737 a | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 9 | |||||||||||
| Agora FSL S5-667 a | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 7 | ||||||||||||
| Cerro FSL R8-370 a | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 9 | |||||||||||
| Newport FSL S5-515 a | 3 | 3 | 2 | ||||||||||||||||||
| 4,5,12, i:- FSL S5-390 a | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 8 | |||||||||||
| Enteritidis SARB16 b | 3 | 3 | 2 | ||||||||||||||||||
| Enteritidis SARB17 b | 3 | 3 | 3 | 3 | 4 | ||||||||||||||||
| Enteritidis SARB19 b | 3 | 3 | 3 | 4 | |||||||||||||||||
| Typhimuriu ATCC 14028 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 20 |
| Total serovars | 6 | 8 | 6 | 1 | 1 | 1 | 5 | 3 | 2 | 3 | 10 | 8 | 5 | 2 | 6 | 4 | 2 | 6 | 3 | 5 | |
| Phage | Burst Size (log 10 Unit) | Lag Time (min) |
|---|---|---|
| A4 | 4.9 | 20 |
| A5.1 | 2.5 | 20 |
| A7 | 1.8 | >60 |
| A8 | 1.7 | >100 |
| B3 | 0.8 | 4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aguilera, M.; Martínez, S.; Tello, M.; Gallardo, M.J.; García, V. Use of Cocktail of Bacteriophage for Salmonella Typhimurium Control in Chicken Meat. Foods 2022, 11, 1164. https://doi.org/10.3390/foods11081164
Aguilera M, Martínez S, Tello M, Gallardo MJ, García V. Use of Cocktail of Bacteriophage for Salmonella Typhimurium Control in Chicken Meat. Foods. 2022; 11(8):1164. https://doi.org/10.3390/foods11081164
Chicago/Turabian StyleAguilera, Matías, Sofía Martínez, Mario Tello, María José Gallardo, and Verónica García. 2022. "Use of Cocktail of Bacteriophage for Salmonella Typhimurium Control in Chicken Meat" Foods 11, no. 8: 1164. https://doi.org/10.3390/foods11081164
APA StyleAguilera, M., Martínez, S., Tello, M., Gallardo, M. J., & García, V. (2022). Use of Cocktail of Bacteriophage for Salmonella Typhimurium Control in Chicken Meat. Foods, 11(8), 1164. https://doi.org/10.3390/foods11081164







