In Vitro Anti-Inflammatory Activity of Peptides Obtained by Tryptic Shaving of Surface Proteins of Streptococcus thermophilus LMD-9
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Growth Conditions
2.2. Enzymatic Shaving of Surface Proteins
2.3. LC-MS/MS Analysis
2.4. Anti-Inflammatory Activity of Peptide Hydrolysate on HT-29 Cells
2.5. Anti-Inflammatory Activity of Peptide Hydrolysate on THP-1 Macrophages
2.6. Measurement of Inflammatory Cytokines by ELISA
2.7. Cell Cytotoxicity
2.8. Western-Blotting
2.9. Statistical Analysis
3. Results
3.1. Analysis of Surface Proteins of S. thermophilus LMD-9 after Tryptic Shaving
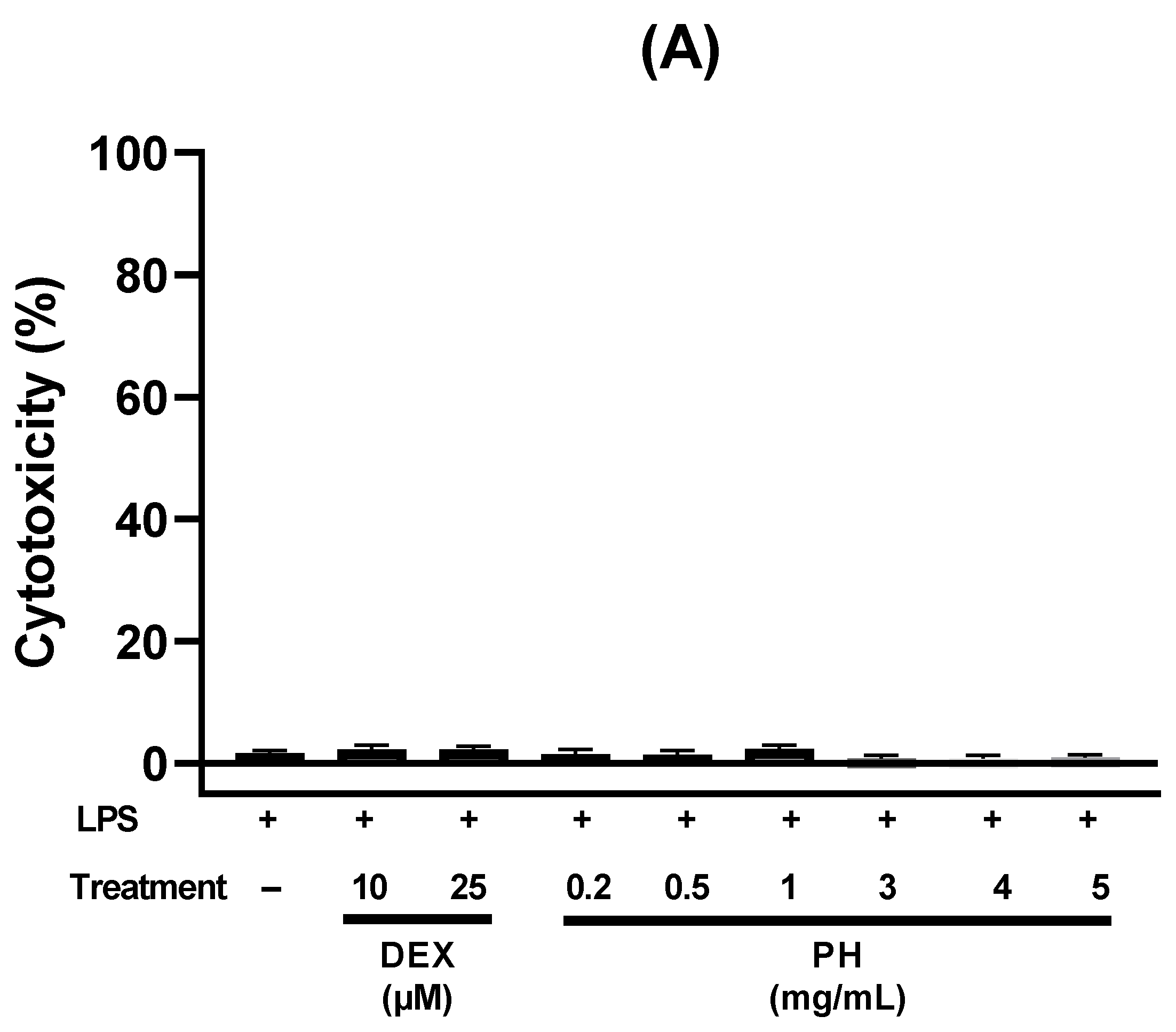
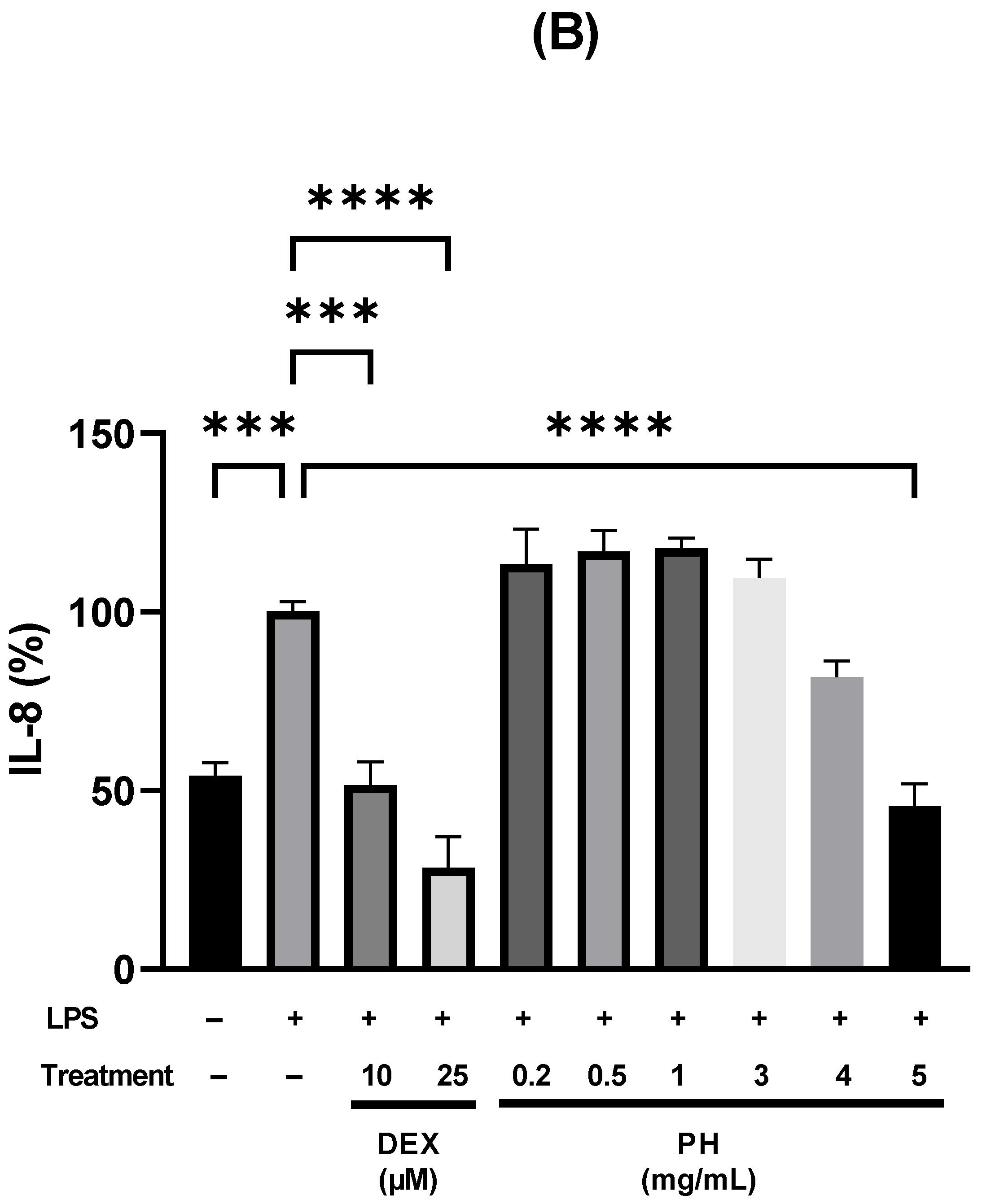
3.2. Anti-Inflammatory Effect of Peptide Hydrolysate on HT-29 Cells
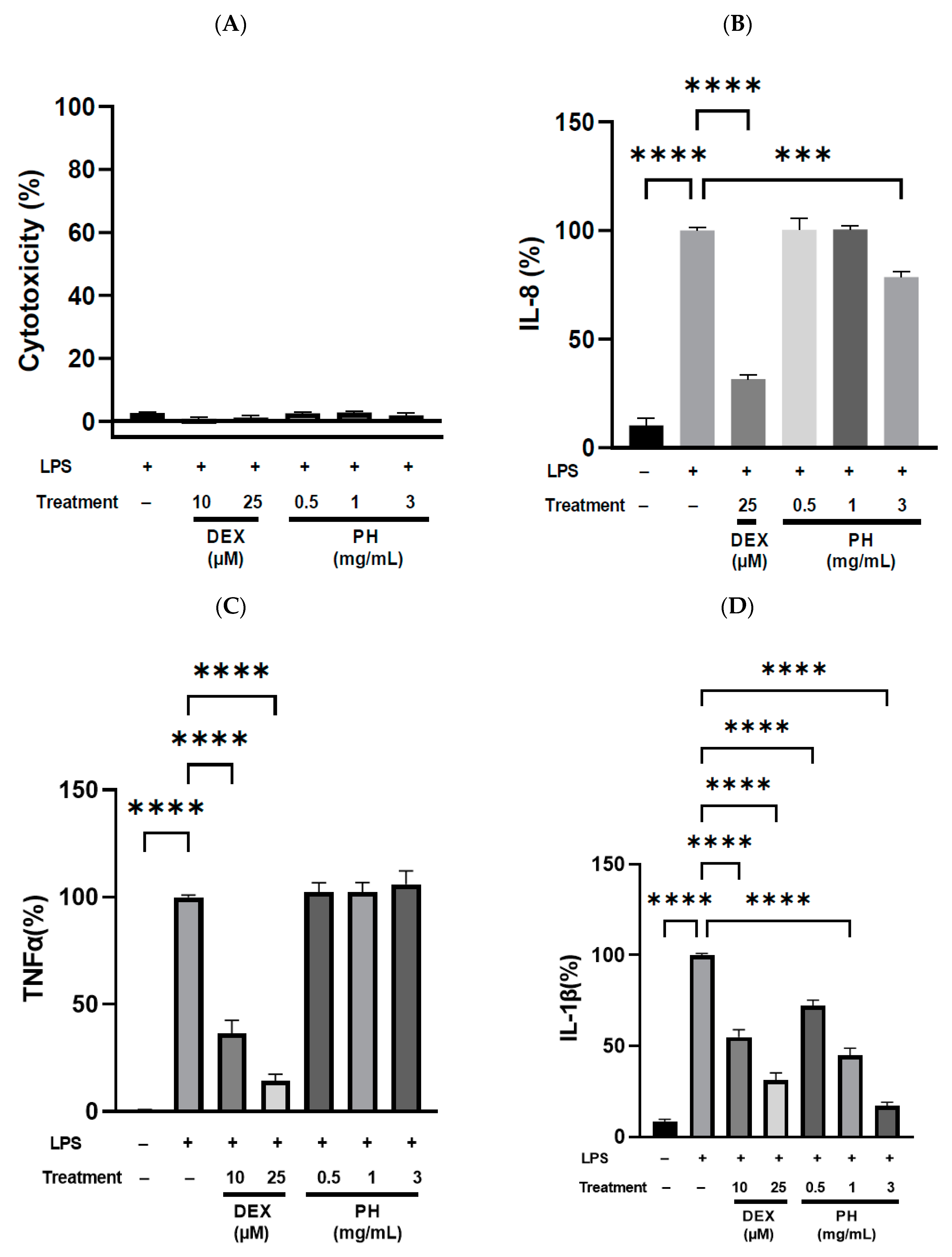
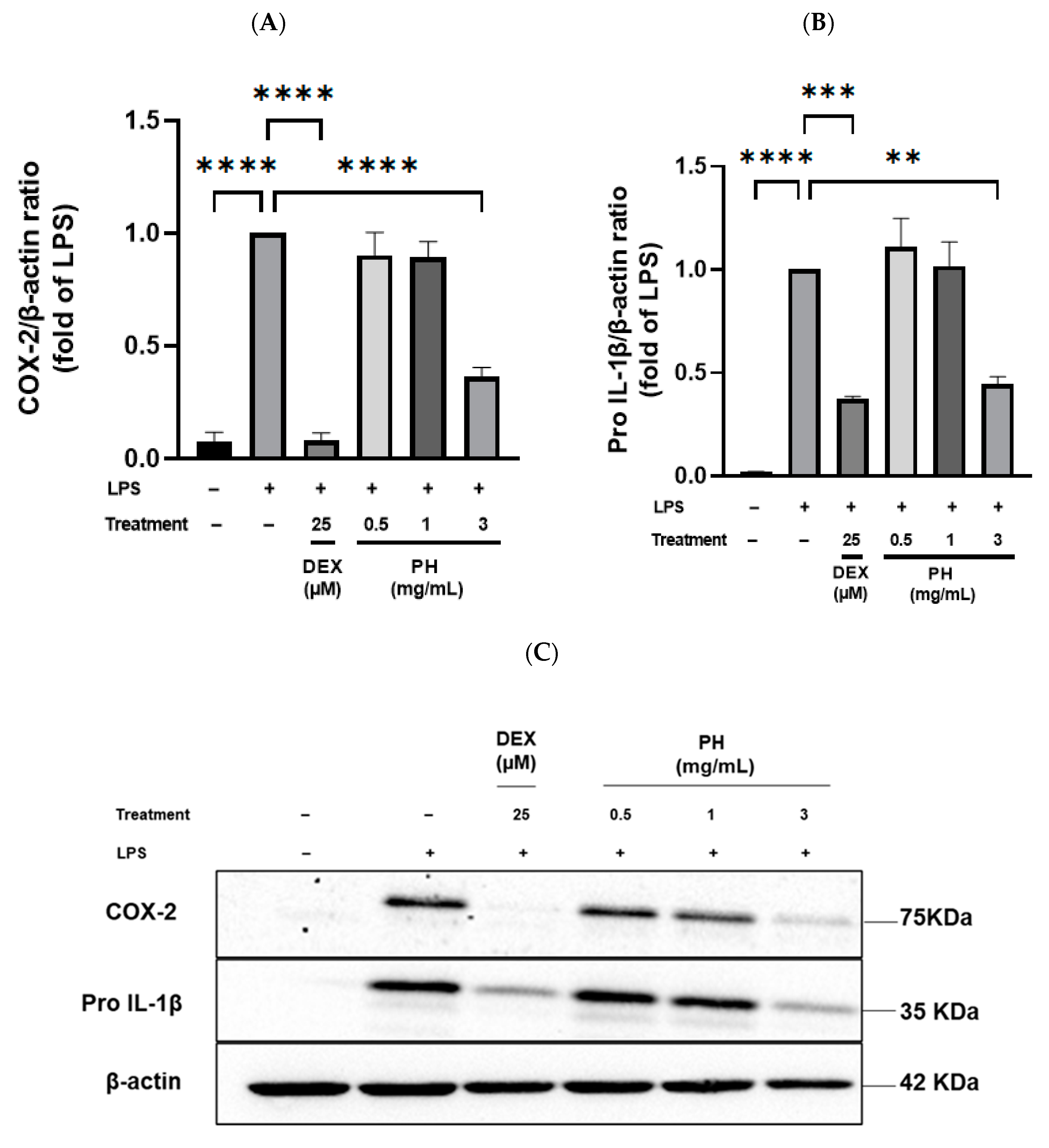
3.3. Anti-Inflammatory Effect of Peptide Hydrolysate on THP-1 Macrophages
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Serhan, C.N.; Savill, J. Resolution of Inflammation: The Beginning Programs the End. Nat. Immunol. 2005, 6, 1191–1197. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Gong, X.; Wan, J.; Zhang, L.; Zhang, Z.; Li, H.; Min, S. Resolvin D1 Protects Mice from LPS-Induced Acute Lung Injury. Pulm. Pharmacol. Ther. 2011, 24, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Ciornei, R.T. Prevention of Severe Coronavirus Disease 2019 Outcomes by Reducing Low-Grade Inflammation in High-Risk Categories. Front. Immunol. 2020, 11, 1762. [Google Scholar] [CrossRef] [PubMed]
- Suárez-Reyes, A.; Villegas-Valverde, C.A. Implications of Low-Grade Inflammation in SARS-CoV-2 Immunopathology. MEDICC Rev. 2021, 23, 42. [Google Scholar] [CrossRef] [PubMed]
- Carter, G.T.; Duong, V.; Ho, S.; Ngo, K.C.; Greer, C.L.; Weeks, D.L. Side Effects of Commonly Prescribed Analgesic Medications. Phys. Med. Rehabil. Clin. N. Am. 2014, 25, 457–470. [Google Scholar] [CrossRef] [PubMed]
- Guha, S.; Majumder, K. Structural-Features of Food-Derived Bioactive Peptides with Anti-Inflammatory Activity: A Brief Review. J. Food Biochem. 2019, 43, e12531. [Google Scholar] [CrossRef] [PubMed]
- Ilich, J.Z. Nutritional and Behavioral Approaches to Body Composition and Low-Grade Chronic Inflammation Management for Older Adults in the Ordinary and COVID-19 Times. Nutrients 2020, 12, 3898. [Google Scholar] [CrossRef]
- Kitts, D.D.; Weiler, K. Bioactive Proteins and Peptides from Food Sources. Applications of Bioprocesses Used in Isolation and Recovery. Curr. Pharm. Des. 2003, 9, 1309–1323. [Google Scholar] [CrossRef]
- Sánchez, A.; Vázquez, A. Bioactive Peptides: A Review. Food Qual. Saf. 2017, 1, 29–46. [Google Scholar] [CrossRef]
- Chakrabarti, S.; Guha, S.; Majumder, K. Food-Derived Bioactive Peptides in Human Health: Challenges and Opportunities. Nutrients 2018, 10, 1738. [Google Scholar] [CrossRef] [Green Version]
- Kanmani, P.; Kim, H. Protective Effects of Lactic Acid Bacteria Against TLR4 Induced Inflammatory Response in Hepatoma HepG2 Cells Through Modulation of Toll-Like Receptor Negative Regulators of Mitogen-Activated Protein Kinase and NF-ΚB Signaling. Front. Immunol. 2018, 9, 1537. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.Y.; Sekhon, S.S.; Kim, H.C.; Won, K.; Ahn, J.-Y.; Lee, K.; Kim, Y.-H. Anti-Inflammatory Effect of Lactic Acid Bacteria Isolated from Kimchi on Acid-Induced Acute Colitis in Model Mice. Toxicol. Environ. Health Sci. 2017, 9, 279–283. [Google Scholar] [CrossRef]
- Suzuki, H.; Yamazaki, T.; Ohshio, K.; Sugamata, M.; Yoshikawa, M.; Kanauchi, O.; Morita, Y. A Specific Strain of Lactic Acid Bacteria, Lactobacillus paracasei, Inhibits Inflammasome Activation In Vitro and Prevents Inflammation-Related Disorders. J. Immunol. 2020, 205, 811–821. [Google Scholar] [CrossRef] [PubMed]
- Delorme, C. Safety Assessment of Dairy Microorganisms: Streptococcus thermophilus. Int. J. Food Microbiol. 2008, 126, 274–277. [Google Scholar] [CrossRef] [PubMed]
- Dandoy, D.; Fremaux, C.; Henry de Frahan, M.; Horvath, P.; Boyaval, P.; Hols, P.; Fontaine, L. The Fast Milk Acidifying Phenotype of Streptococcus thermophilus Can Be Acquired by Natural Transformation of the Genomic Island Encoding the Cell-Envelope Proteinase PrtS. Microb. Cell Factories 2011, 10, S21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galia, W.; Jameh, N.; Perrin, C.; Genay, M.; Dary-Mourot, A. Acquisition of PrtS in Streptococcus thermophilus Is Not Enough in Certain Strains to Achieve Rapid Milk Acidification. Dairy Sci. Technol. 2016, 96, 623–636. [Google Scholar] [CrossRef] [Green Version]
- Hafeez, Z.; Cakir-Kiefer, C.; Roux, E.; Perrin, C.; Miclo, L.; Dary-Mourot, A. Strategies of Producing Bioactive Peptides from Milk Proteins to Functionalize Fermented Milk Products. Food Res. Int. 2014, 63, 71–80. [Google Scholar] [CrossRef]
- Miclo, L.; Roux, É.; Genay, M.; Brusseaux, É.; Poirson, C.; Jameh, N.; Perrin, C.; Dary, A. Variability of Hydrolysis of β-, αs1-, and αs2-Caseins by 10 Strains of Streptococcus thermophilus and Resulting Bioactive Peptides. J. Agric. Food Chem. 2012, 60, 554–565. [Google Scholar] [CrossRef]
- Uriot, O.; Denis, S.; Junjua, M.; Roussel, Y.; Dary-Mourot, A.; Blanquet-Diot, S. Streptococcus thermophilus: From Yogurt Starter to a New Promising Probiotic Candidate? J. Funct. Foods 2017, 37, 74–89. [Google Scholar] [CrossRef]
- Burton, J.P.; Chanyi, R.M.; Schultz, M. Common Organisms and Probiotics: Streptococcus thermophilus (Streptococcus salivarius subsp. thermophilus). In The Microbiota in Gastrointestinal Pathophysiology; Elsevier: Amsterdam, The Netherlands, 2017; pp. 165–169. ISBN 978-0-12-804024-9. [Google Scholar]
- Del Carmen, S.; de Moreno de LeBlanc, A.; Martin, R.; Chain, F.; Langella, P.; Bermúdez-Humarán, L.G.; LeBlanc, J.G. Genetically Engineered Immunomodulatory Streptococcus thermophilus Strains Producing Antioxidant Enzymes Exhibit Enhanced Anti-Inflammatory Activities. Appl. Environ. Microbiol. 2014, 80, 869–877. [Google Scholar] [CrossRef] [Green Version]
- Cheng, F.-S.; Pan, D.; Chang, B.; Jiang, M.; Sang, L.-X. Probiotic Mixture VSL#3: An Overview of Basic and Clinical Studies in Chronic Diseases. World J. Clin. Cases 2020, 8, 1361–1384. [Google Scholar] [CrossRef] [PubMed]
- Dargahi, N.; Johnson, J.; Donkor, O.; Vasiljevic, T.; Apostolopoulos, V. Immunomodulatory Effects of Streptococcus thermophilus on U937 Monocyte Cell Cultures. J. Funct. Foods 2018, 49, 241–249. [Google Scholar] [CrossRef]
- Dargahi, N.; Matsoukas, J.; Apostolopoulos, V. Streptococcus thermophilus ST285 Alters Pro-Inflammatory to Anti-Inflammatory Cytokine Secretion against Multiple Sclerosis Peptide in Mice. Brain Sci. 2020, 10, 126. [Google Scholar] [CrossRef] [Green Version]
- Dargahi, N.; Johnson, J.C.; Apostolopoulos, V. Immune Modulatory Effects of Probiotic Streptococcus thermophilus on Human Monocytes. Biologics 2021, 1, 396–415. [Google Scholar] [CrossRef]
- Dargahi, N.; Johnson, J.; Apostolopoulos, V. Streptococcus thermophilus Alters the Expression of Genes Associated with Innate and Adaptive Immunity in Human Peripheral Blood Mononuclear Cells. PLoS ONE 2020, 15, e0228531. [Google Scholar] [CrossRef] [Green Version]
- Han, F.; Wu, G.; Zhang, Y.; Zheng, H.; Han, S.; Li, X.; Cai, W.; Liu, J.; Zhang, W.; Zhang, X.; et al. Streptococcus thermophilus Attenuates Inflammation in Septic Mice Mediated by Gut Microbiota. Front. Microbiol. 2020, 11, 598010. [Google Scholar] [CrossRef]
- Junjua, M.; Kechaou, N.; Chain, F.; Awussi, A.A.; Roussel, Y.; Perrin, C.; Roux, E.; Langella, P.; Bermúdez-Humarán, L.G.; Le Roux, Y.; et al. A Large Scale in Vitro Screening of Streptococcus thermophilus Strains Revealed Strains with a High Anti-Inflammatory Potential. LWT Food Sci. Technol. 2016, 70, 78–87. [Google Scholar] [CrossRef]
- Wang, Y.; Li, H.; Li, T.; Du, X.; Zhang, X.; Guo, T.; Kong, J. Glutathione Biosynthesis Is Essential for Antioxidant and Anti-Inflammatory Effects of Streptococcus thermophilus. Int. Dairy J. 2019, 89, 31–36. [Google Scholar] [CrossRef]
- Dave, L.A.; Hayes, M.; Montoya, C.A.; Rutherfurd, S.M.; Moughan, P.J. Human Gut Endogenous Proteins as a Potential Source of Angiotensin-I-Converting Enzyme (ACE-I)-, Renin Inhibitory and Antioxidant Peptides. Peptides 2016, 76, 30–44. [Google Scholar] [CrossRef]
- Terzaghi, B.E.; Sandine, W.E. Improved Medium for Lactic Streptococci and Their Bacteriophages. Appl. Microbiol. 1975, 29, 807–813. [Google Scholar] [CrossRef]
- Lecomte, X.; Gagnaire, V.; Briard-Bion, V.; Jardin, J.; Lortal, S.; Dary, A.; Genay, M. The Naturally Competent Strain Streptococcus thermophilus LMD-9 as a New Tool to Anchor Heterologous Proteins on the Cell Surface. Microb. Cell Factories 2014, 13, 82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deglaire, A.; Oliveira, S.D.; Jardin, J.; Briard-Bion, V.; Kroell, F.; Emily, M.; Ménard, O.; Bourlieu, C.; Dupont, D. Impact of Human Milk Pasteurization on the Kinetics of Peptide Release during in Vitro Dynamic Digestion at the Preterm Newborn Stage. Food Chem. 2019, 281, 294–303. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Wang, F.; Zhao, P.; Zhang, R.; Zeng, Q. Effect of Heat-Killed Streptococcus thermophilus on Type 2 Diabetes Rats. PeerJ 2019, 7, e7117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Piqué, N.; Berlanga, M.; Miñana-Galbis, D. Health Benefits of Heat-Killed (Tyndallized) Probiotics: An Overview. Int. J. Mol. Sci. 2019, 20, 2534. [Google Scholar] [CrossRef] [Green Version]
- Lee, I.-C.; Tomita, S.; Kleerebezem, M.; Bron, P.A. The Quest for Probiotic Effector Molecules—Unraveling Strain Specificity at the Molecular Level. Pharmacol. Res. 2013, 69, 61–74. [Google Scholar] [CrossRef]
- Noh, S.Y.; Kang, S.-S.; Yun, C.-H.; Han, S.H. Lipoteichoic Acid from Lactobacillus plantarum Inhibits Pam2CSK4-Induced IL-8 Production in Human Intestinal Epithelial Cells. Mol. Immunol. 2015, 64, 183–189. [Google Scholar] [CrossRef]
- Fernandez, E.M.; Valenti, V.; Rockel, C.; Hermann, C.; Pot, B.; Boneca, I.G.; Grangette, C. Anti-Inflammatory Capacity of Selected Lactobacilli in Experimental Colitis Is Driven by NOD2-Mediated Recognition of a Specific Peptidoglycan-Derived Muropeptide. Gut 2011, 60, 1050–1059. [Google Scholar] [CrossRef]
- Olsen, J.V.; Ong, S.-E.; Mann, M. Trypsin Cleaves Exclusively C-Terminal to Arginine and Lysine Residues. Mol. Cell. Proteomics 2004, 3, 608–614. [Google Scholar] [CrossRef] [Green Version]
- Laskay, Ü.A.; Lobas, A.A.; Srzentić, K.; Gorshkov, M.V.; Tsybin, Y.O. Proteome Digestion Specificity Analysis for Rational Design of Extended Bottom-up and Middle-down Proteomics Experiments. J. Proteome Res. 2013, 12, 5558–5569. [Google Scholar] [CrossRef]
- Siciliano, R.A.; Lippolis, R.; Mazzeo, M.F. Proteomics for the Investigation of Surface-Exposed Proteins in Probiotics. Front. Nutr. 2019, 6, 52. [Google Scholar] [CrossRef]
- Tjalsma, H.; Lambooy, L.; Hermans, P.W.; Swinkels, D.W. Shedding & Shaving: Disclosure of Proteomic Expressions on a Bacterial Face. PROTEOMICS 2008, 8, 1415–1428. [Google Scholar] [CrossRef] [PubMed]
- Mu, Y.; Xin, Y.; Guo, T.; Kong, J. Identification and Characterization of a Moonlighting Protein-Enolase for Surface Display in Streptococcus thermophilus. Microb. Cell Factories 2020, 19, 132. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Jeffery, C.J. An Analysis of Surface Proteomics Results Reveals Novel Candidates for Intracellular/Surface Moonlighting Proteins in Bacteria. Mol. Biosyst. 2016, 12, 1420–1431. [Google Scholar] [CrossRef] [PubMed]
- Byun, E.-B.; Kim, W.S.; Sung, N.-Y.; Byun, E.-H. Epigallocatechin-3-Gallate Regulates Anti-Inflammatory Action Through 67-KDa Laminin Receptor-Mediated Tollip Signaling Induction in Lipopolysaccharide-Stimulated Human Intestinal Epithelial Cells. Cell. Physiol. Biochem. 2018, 46, 2072–2081. [Google Scholar] [CrossRef]
- Chanput, W.; Mes, J.J.; Wichers, H.J. THP-1 Cell Line: An in Vitro Cell Model for Immune Modulation Approach. Int. Immunopharmacol. 2014, 23, 37–45. [Google Scholar] [CrossRef]
- Jones, L.D.; Pangloli, P.; Krishnan, H.B.; Dia, V.P. BG-4, a Novel Bioactive Peptide from Momordica charantia, Inhibits Lipopolysaccharide-Induced Inflammation in THP-1 Human Macrophages. Phytomedicine 2018, 42, 226–232. [Google Scholar] [CrossRef]
- Bisht, A.; Dickens, M.; Rutherfurd-Markwick, K.; Thota, R.; Mutukumira, A.N.; Singh, H. Chlorogenic Acid Potentiates the Anti-Inflammatory Activity of Curcumin in LPS-Stimulated THP-1 Cells. Nutrients 2020, 12, 2706. [Google Scholar] [CrossRef]
- Shi, J.; Shan, S.; Li, H.; Song, G.; Li, Z. Anti-Inflammatory Effects of Millet Bran Derived-Bound Polyphenols in LPS-Induced HT-29 Cell via ROS/MiR-149/Akt/NF-ΚB Signaling Pathway. Oncotarget 2017, 8, 74582–74594. [Google Scholar] [CrossRef] [Green Version]
- Kany, S.; Vollrath, J.T.; Relja, B. Cytokines in Inflammatory Disease. Int. J. Mol. Sci. 2019, 20, 6008. [Google Scholar] [CrossRef] [Green Version]
- Do Carmo, F.L.R.; Rabah, H.; De Oliveira Carvalho, R.D.; Gaucher, F.; Cordeiro, B.F.; da Silva, S.H.; Le Loir, Y.; Azevedo, V.; Jan, G. Extractable Bacterial Surface Proteins in Probiotic–Host Interaction. Front. Microbiol. 2018, 9, 645. [Google Scholar] [CrossRef]
- Boonma, P.; Spinler, J.K.; Venable, S.F.; Versalovic, J.; Tumwasorn, S. Lactobacillus rhamnosus L34 and Lactobacillus casei L39 Suppress Clostridium difficile-Induced IL-8 Production by Colonic Epithelial Cells. BMC Microbiol. 2014, 14, 177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eftang, L.L.; Esbensen, Y.; Tannæs, T.M.; Bukholm, I.R.; Bukholm, G. Interleukin-8 Is the Single Most up-Regulated Gene in Whole Genome Profiling of H. pylori Exposed Gastric Epithelial Cells. BMC Microbiol. 2012, 12, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Do Carmo, F.L.R.; Rabah, H.; Huang, S.; Gaucher, F.; Deplanche, M.; Dutertre, S.; Jardin, J.; Le Loir, Y.; Azevedo, V.; Jan, G. Propionibacterium freudenreichii Surface Protein SlpB Is Involved in Adhesion to Intestinal HT-29 Cells. Front. Microbiol. 2017, 8, 1033. [Google Scholar] [CrossRef] [PubMed]
- Do Carmo, F.L.R.; Rabah, H.; Cordeiro, B.F.; da Silva, S.H.; Pessoa, R.M.; Fernandes, S.O.A.; Cardoso, V.N.; Gagnaire, V.; Deplanche, M.; Savassi, B.; et al. Probiotic Propionibacterium freudenreichii Requires SlpB Protein to Mitigate Mucositis Induced by Chemotherapy. Oncotarget 2019, 10, 7198–7219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, P.; Yu, Q.; Ye, X.; Wang, Z.; Yang, Q. Lactobacillus S-Layer Protein Inhibition of Salmonella-Induced Reorganization of the Cytoskeleton and Activation of MAPK Signalling Pathways in Caco-2 Cells. Microbiology 2011, 157, 2639–2646. [Google Scholar] [CrossRef] [PubMed]
- Hou, Z.; Falcone, D.J.; Subbaramaiah, K.; Dannenberg, A.J. Macrophages Induce COX-2 Expression in Breast Cancer Cells: Role of IL-1β Autoamplification. Carcinogenesis 2011, 32, 695–702. [Google Scholar] [CrossRef] [Green Version]
- García-Lafuente, A.; Moro, C.; Manchón, N.; Gonzalo-Ruiz, A.; Villares, A.; Guillamón, E.; Rostagno, M.; Mateo-Vivaracho, L. In Vitro Anti-Inflammatory Activity of Phenolic Rich Extracts from White and Red Common Beans. Food Chem. 2014, 161, 216–223. [Google Scholar] [CrossRef]
- Mater, D.D.G.; Bretigny, L.; Firmesse, O.; Flores, M.-J.; Mogenet, A.; Bresson, J.-L.; Corthier, G. Streptococcus thermophilus and Lactobacillus delbrueckii subsp. bulgaricus Survive Gastrointestinal Transit of Healthy Volunteers Consuming Yogurt. FEMS Microbiol. Lett. 2005, 250, 185–187. [Google Scholar] [CrossRef] [Green Version]
- Feijó Delgado, F.; Cermak, N.; Hecht, V.C.; Son, S.; Li, Y.; Knudsen, S.M.; Olcum, S.; Higgins, J.M.; Chen, J.; Grover, W.H.; et al. Intracellular Water Exchange for Measuring the Dry Mass, Water Mass and Changes in Chemical Composition of Living Cells. PLoS ONE 2013, 8, e67590. [Google Scholar] [CrossRef] [Green Version]
- Kayser, H.; Meisel, H. Stimulation of Human Peripheral Blood Lymphocytes by Bioactive Peptides Derived from Bovine Milk Proteins. FEBS Lett. 1996, 383, 18–20. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Zhao, Y.; Yao, Y.; Xu, M.; Du, H.; Zhang, M.; Tu, Y. Anti-Inflammatory Activity of Di-Peptides Derived from Ovotransferrin by Simulated Peptide-Cut in TNF-α-Induced Caco-2 Cells. J. Funct. Foods 2017, 37, 424–432. [Google Scholar] [CrossRef]
- Zhao, L.; Wang, X.; Zhang, X.-L.; Xie, Q.-F. Purification and Identification of Anti-Inflammatory Peptides Derived from Simulated Gastrointestinal Digests of Velvet Antler Protein (Cervus elaphus Linnaeus). J. Food Drug Anal. 2016, 24, 376–384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, Y.; Liu, J.; Shi, H.; Yu, L. (Lucy) Isolation and Characterization of Anti-Inflammatory Peptides Derived from Whey Protein. J. Dairy Sci. 2016, 99, 6902–6912. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kozlowski, L.P. Proteome-PI: Proteome Isoelectric Point Database. Nucleic Acids Res. 2017, 45, D1112–D1116. [Google Scholar] [CrossRef] [PubMed]
| Protein ID | New Locus Number | Description | MW (kDa) | Location | Coverage (%) | Nb Specific Sequences Identified |
|---|---|---|---|---|---|---|
| Nucleotide metabolism and transport | ||||||
| STER_1992|ID:1900614|guaB| | STER_RS09740 | IMP dehydrogenase | 52.88 | Cyto | 56.7 | 17 |
| STER_1845|ID:1900521| | STER_RS09015 | DNA-directed RNA polymerase subunit beta | 133.17 | Cyto | 32.3 | 29 |
| STER_0198|ID:1898877| | STER_RS00970 | 2′:3′-cyclic-nucleotide 2′-phosphodiesterase (modular protein) | 91.21 | CS | 64.9 | 156 |
| Post-translational modification, protein turnover, chaperone function | ||||||
| STER_0846|ID:1899371| | STER_RS04165 | exported protein of unknown function (subtilisin-like serine protease PrtS) | 173.05 | CS | 51.3 | 103 |
| STER_1578|ID:1900309|clpB| | STER_RS07755 | protein disaggregation chaperone | 77.15 | Cyto | 69.0 | 70 |
| STER_0253|ID:1898921|groL| | STER_RS01230 | Cpn60 chaperonin GroEL, large subunit of GroESL | 56.89 | Cyto | 69.8 | 32 |
| STER_0163|ID:1898844|dnaK| | STER_RS00790 | chaperone Hsp70, co-chaperone with DnaJ | 64.76 | Cyto | 50.8 | 27 |
| STER_0191|ID:1898870|tig| | STER_RS00935 | peptidyl-prolyl cis/trans isomerase (trigger factor) | 46.65 | Cyto | 61.9 | 26 |
| STER_2002|ID:1899687|degP| | STER_RS09790 | serine endoprotease (protease Do), membrane-associated | 42.77 | CM/M | 72.3 | 37 |
| STER_0014|ID:1898716|ftsH| | STER_RS00070 | protease, ATP-dependent zinc-metallo | 71.95 | M | 34.8 | 16 |
| STER_0648|ID:1899776|clpA| | STER_RS03195 | ATPase and specificity subunit of ClpA-ClpP ATP-dependent serine protease, chaperone activity | 83.69 | CM | 24.4 | 13 |
| Translation | ||||||
| STER_0524|ID:1899138|tufB| | STER_RS02570 | protein chain elongation factor EF-Tu (duplicate of tufA) | 43.84 | Cyto | 68.9 | 35 |
| STER_1762|ID:1900456|fusA| | STER_RS08620 | protein chain elongation factor EF-G, GTP-binding | 76.56 | Cyto | 59.4 | 33 |
| STER_0639|ID:1899231| | STER_RS03135 | 40S ribosomal protein S1 | 43.88 | Cyto | 50.4 | 22 |
| STER_1844|ID:1900520|rpoC| | STER_RS09010 | RNA polymerase, beta prime subunit | 135.16 | Cyto | 21.4 | 21 |
| STER_1526|ID:1900271|deaD| | STER_RS07510 | ATP-dependent RNA helicase | 58.96 | CM | 44.5 | 21 |
| STER_1904|ID:1900567|rplB| | STER_RS09330 | 50S ribosomal subunit protein L2 | 29.91 | Cyto | 49.6 | 19 |
| STER_0383|ID:1899016|infB| | STER_RS01860 | conserved protein of unknown function (translation inibition factor IF-2) | 103.73 | Cyto | 20.3 | 17 |
| STER_0105|ID:1898797|rpsB| | STER_RS00525 | 30S ribosomal subunit protein S2 | 28.40 | Cyto | 69.1 | 17 |
| STER_0247|ID:1898915|proS| | STER_RS01210 | prolyl-tRNA synthetase | 68.48 | Cyto | 35.2 | 16 |
| STER_1893|ID:1900556|rpsH| | STER_RS09275 | 30S ribosomal subunit protein S8 | 14.78 | Cyto | 58.7 | 15 |
| Carbohydrate metabolism and transport | ||||||
| STER_1761|ID:1900455|gapA| | STER_RS08615 | glyceraldehyde-3-phosphate dehydrogenase A | 36.00 | Cyto | 83.4 | 28 |
| STER_0684|ID:1899266|eno| | STER_RS03365 | enolase | 46.95 | CS/M/Cyto | 63.2 | 28 |
| STER_1163|ID:1899979|pykF| | STER_RS05740 | pyruvate kinase I | 54.49 | Cyto | 55.1 | 22 |
| STER_1172|ID:1899988|gpmA| | STER_RS05785 | phosphoglyceromutase 1 | 26.17 | Cyto | 53.7 | 21 |
| STER_1876|ID:1900541|kbaY| | STER_RS09185 | tagatose 6-phosphate aldolase 1, kbaY subunit | 31.51 | Cyto | 47.3 | 14 |
| STER_0241|ID:1898909| | STER_RS01180 | glucose-6-phosphate isomerase A (GPI A) | 49.76 | Cyto | 34.7 | 14 |
| STER_1241|ID:1900050|gabD| | STER_RS06125 | succinate-semialdehyde dehydrogenase I, NADP-dependent | 50.79 | Cyto | 45.0 | 18 |
| STER_1755|ID:1900453|pgk| | STER_RS08580 | phosphoglycerate kinase | 42.21 | Cyto | 58.8 | 20 |
| STER_0895|ID:1899407| | STER_RS04435 | putative ribulose-phosphate 3-epimerase | 58.13 | Cyto | 34.8 | 15 |
| Cell wall/membrane/envelop biogenesis | ||||||
| STER_0042|ID:1898743| | STER_RS00210 | secreted 45 kDa protein precursor | 46.45 | CS | 45.6 | 17 |
| Amino acid transport and metabolism | ||||||
| STER_1411|ID:1900180| | STER_RS06940 | putative transporter subunit: periplasmic-binding component of ABC superfamily | 72.18 | CS | 47.3 | 22 |
| Unknown function | ||||||
| STER_0576|ID:1899177| | STER_RS02840 | mucus-binding protein precursor (fragment) | 108.40 | CS | 29.1 | 31 |
| STER_0856|ID:1899377| | STER_RS04220 | CD4+ T-cell-stimulating antigen precursor | 37.62 | CS | 53.2 | 14 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Allouche, R.; Hafeez, Z.; Papier, F.; Dary-Mourot, A.; Genay, M.; Miclo, L. In Vitro Anti-Inflammatory Activity of Peptides Obtained by Tryptic Shaving of Surface Proteins of Streptococcus thermophilus LMD-9. Foods 2022, 11, 1157. https://doi.org/10.3390/foods11081157
Allouche R, Hafeez Z, Papier F, Dary-Mourot A, Genay M, Miclo L. In Vitro Anti-Inflammatory Activity of Peptides Obtained by Tryptic Shaving of Surface Proteins of Streptococcus thermophilus LMD-9. Foods. 2022; 11(8):1157. https://doi.org/10.3390/foods11081157
Chicago/Turabian StyleAllouche, Rania, Zeeshan Hafeez, Florent Papier, Annie Dary-Mourot, Magali Genay, and Laurent Miclo. 2022. "In Vitro Anti-Inflammatory Activity of Peptides Obtained by Tryptic Shaving of Surface Proteins of Streptococcus thermophilus LMD-9" Foods 11, no. 8: 1157. https://doi.org/10.3390/foods11081157
APA StyleAllouche, R., Hafeez, Z., Papier, F., Dary-Mourot, A., Genay, M., & Miclo, L. (2022). In Vitro Anti-Inflammatory Activity of Peptides Obtained by Tryptic Shaving of Surface Proteins of Streptococcus thermophilus LMD-9. Foods, 11(8), 1157. https://doi.org/10.3390/foods11081157







