Safety and Quality of Canned Sardines after Opening: A Shelf-Stability Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Standards and Reagents
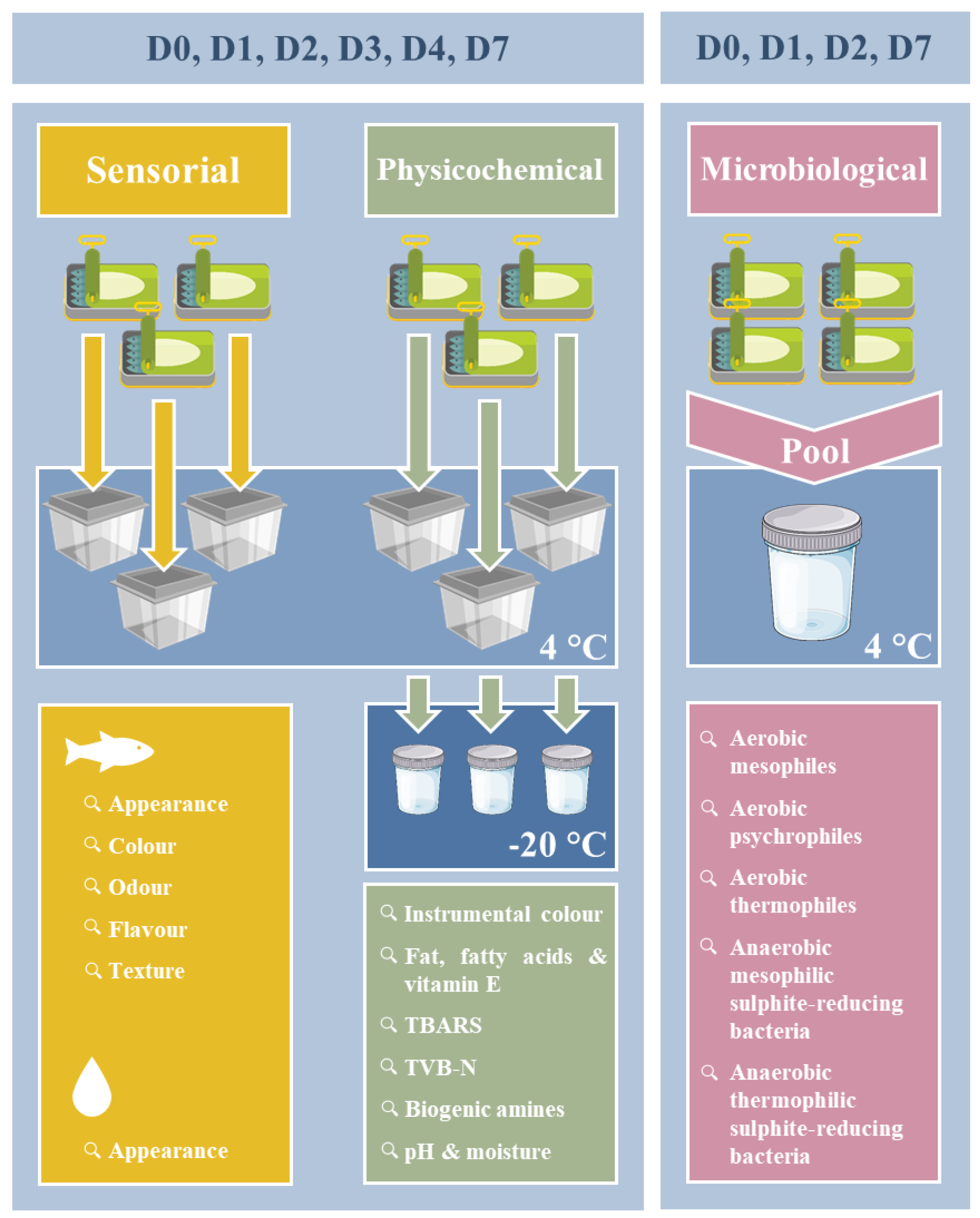
2.2. Sampling and Sample Preparation
2.2.1. Physicochemical and Sensorial Analysis
2.2.2. Microbiological Analysis
2.3. Physicochemical Analysis
2.3.1. Colour Measurement
2.3.2. Estimation of Fat, Fatty Acids, and Vitamin E
2.3.3. Thiobarbituric Acid Reactive Species
2.3.4. Total Volatile Basic Nitrogen
2.3.5. Biogenic Amines
2.3.6. Other Measurements
2.4. Microbiological Analysis
2.5. Sensorial Analysis
2.6. Statistical Analysis
3. Results and Discussion
3.1. Physicochemical Quality and Safety
3.1.1. Nutritional Composition
3.1.2. Lipid Oxidation and Proteolysis
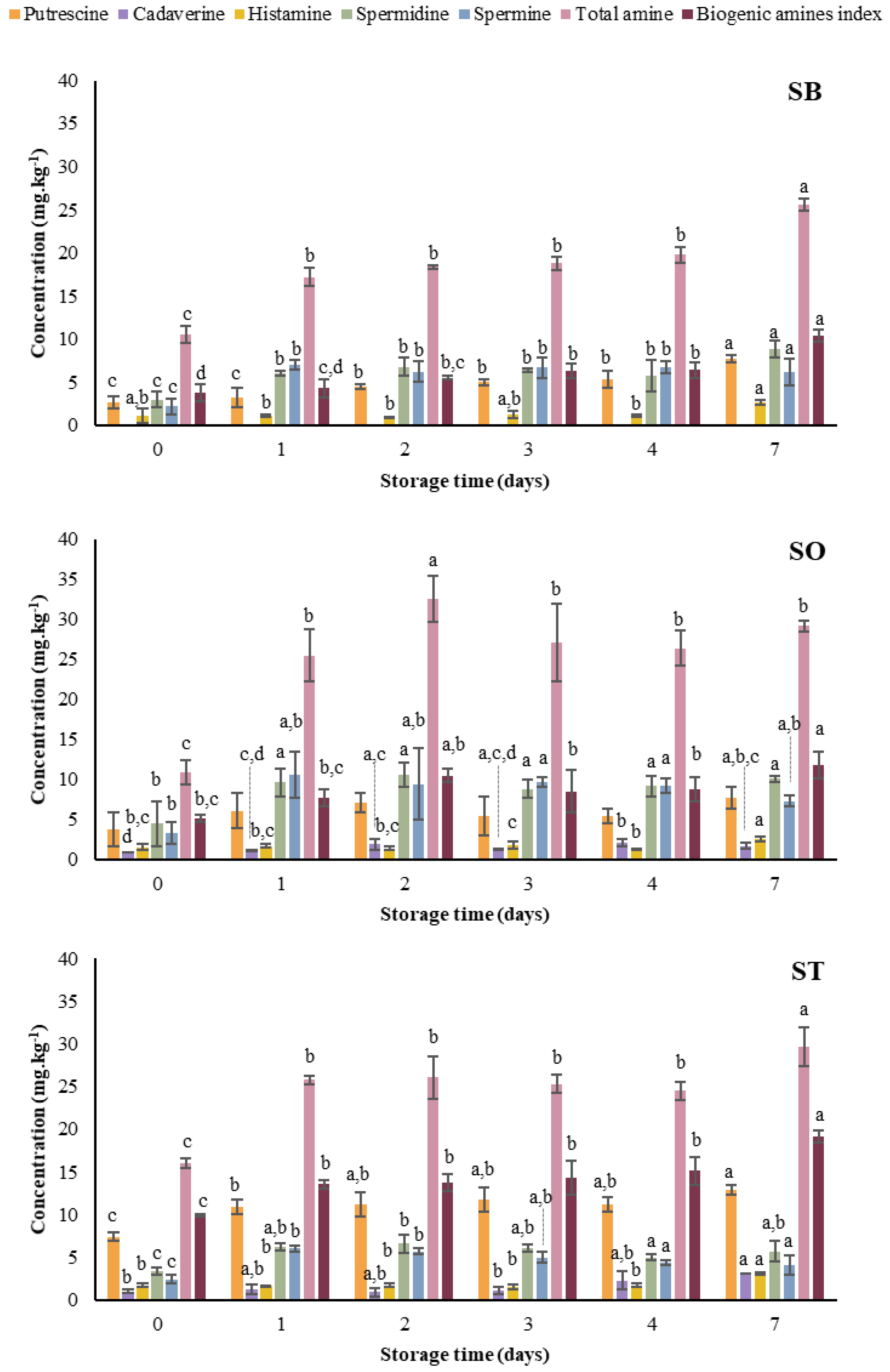
3.1.3. Biogenic Amines
3.2. Instrumental Colour
3.3. Microbiological Quality
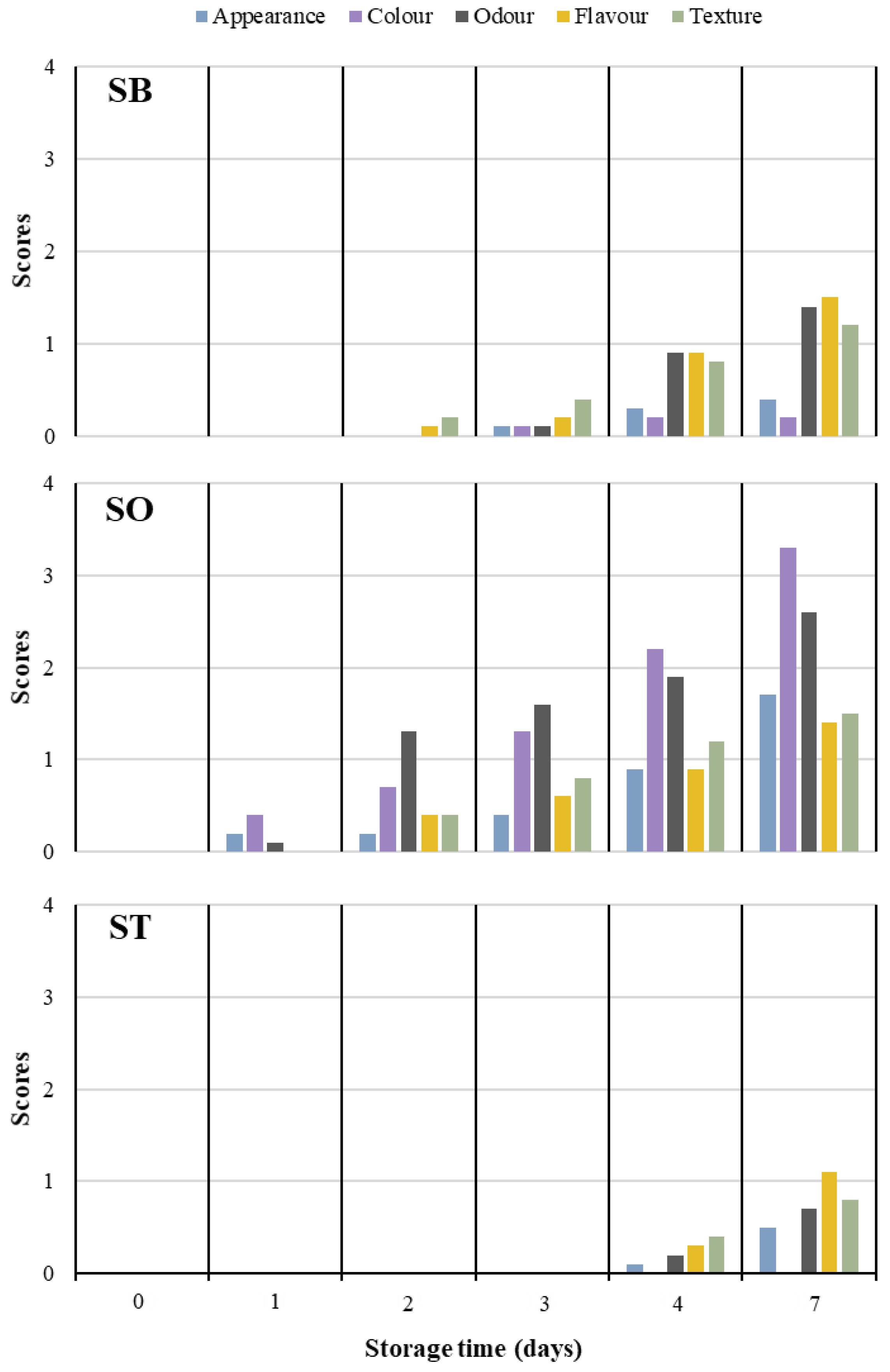
3.4. Sensorial Performance
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. The State of World Fisheries and Aquaculture 2018—Meeting the Sustainable Development Goals; Food and Agriculture Organization of the United Nations: Rome, Italy, 2018. [Google Scholar]
- WHO. Population Nutrient Intake Goals for Preventing Diet-Related Chronic Diseases. Available online: https://www.who.int/nutrition/topics/5_population_nutrient/en/index13.html (accessed on 30 March 2020).
- Khalili Tilami, S.; Sampels, S. Nutritional value of fish: Lipids, proteins, vitamins, and minerals. Rev. Fish. Sci. Aquac. 2018, 26, 243–253. [Google Scholar] [CrossRef]
- Aubourg, S.P. Loss of quality during the manufacture of canned fish products. Food Sci. Technol. Int. 2001, 7, 199–215. [Google Scholar] [CrossRef]
- Gökoğlu, N.; Cengız, E.; Yerlıkaya, P. Determination of the shelf life of marinated sardine (Sardina pilchardus) stored at 4 °C. Food Control 2004, 15, 1–4. [Google Scholar] [CrossRef]
- Kilinc, B.; Cakli, S.; Tolasa, S. Quality changes of sardine (Sardina pilchardus) patties during refrigerated storage. J. Food Qual. 2008, 31, 366–381. [Google Scholar] [CrossRef]
- Losada, V.; Rodríguez, A.; Ortiz, J.; Aubourg, S.P. Quality enhancement of canned sardine (Sardina pilchardus) by a preliminary slurry ice chilling treatment. Eur. J. Lipid Sci. Technol. 2006, 108, 598–605. [Google Scholar] [CrossRef][Green Version]
- Oyelese, O.; Opatokun, M. Exposure time on bacteria flora/count and shelf life of canned sardine (Sardinella pilchardus) under ambient and cold storage conditions. J. Food Process. Preserv. 2007, 31, 517–530. [Google Scholar] [CrossRef]
- Landry, W.L.; Schwab, A.H.; Lancette, G.A. Examination of canned foods. In Bacteriological Analytical Manual; FDA: Silver Spring, MD, USA, 2001. [Google Scholar]
- Pathare, P.B.; Opara, U.L.; Al-Said, F.A.-J. Colour measurement and analysis in fresh and processed foods: A review. Food Bioprocess Technol. 2013, 6, 36–60. [Google Scholar] [CrossRef]
- Cruz, R.; Casal, S.; Mendes, E.; Costa, A.; Santos, C.; Morais, S. Validation of a single-extraction procedure for sequential analysis of vitamin E, cholesterol, fatty acids, and total fat in seafood. Food Anal. Methods 2013, 6, 1196–1204. [Google Scholar] [CrossRef]
- Papastergiadis, A.; Mubiru, E.; Van Langenhove, H.; De Meulenaer, B. Malondialdehyde measurement in oxidized foods: Evaluation of the spectrophotometric thiobarbituric acid reactive substances (TBARS) test in various foods. J. Agric. Food Chem. 2012, 60, 9589–9594. [Google Scholar] [CrossRef]
- Casal, S.; Oliveira, M.; Ferreira, M. Determination of biogenic amines in coffee by an optimized liquid chromatographic method. J. Liq. Chromatogr. Relat. Technol. 2002, 25, 2535–2549. [Google Scholar] [CrossRef]
- Prester, L. Biogenic amines in fish, fish products and shellfish: A review. Food Addit. Contam. Part A 2011, 28, 1547–1560. [Google Scholar] [CrossRef] [PubMed]
- ISO 6887-1:2017. Microbiology of the Food Chain—Preparation of Test Samples, Initial Suspension and Decimal Dilutions for Microbiological Examination—Part 1: General Rules for the Preparation of the Initial Suspension and Decimal Dilutions; ISO: Geneva, Switzerland, 2017. [Google Scholar]
- Codex Alimentarius Commission. Code of Hygienic Practice for Low and Acidified Low Acid Canned Foods; CAC/RCP 23; FAO: Rome, Italy, 2011. [Google Scholar]
- ISO 15213:2003. Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Enumeration of Sulfite-Reducing Bacteria Growing under Anaerobic Conditions; ISO: Geneva, Switzerland, 2003. [Google Scholar]
- ISO 4833-2:2013. Microbiology of the Food Chain—Horizontal Method for the Enumeration of Microorganisms—Part 2: Colony Count at 30 Degrees C by the Surface Plating Technique; ISO: Geneva, Switzerland, 2013. [Google Scholar]
- ISO 4833-1:2013. Microbiology of the Food Chain—Horizontal Method for the Enumeration of Microorganisms—Part 1: Colony Count at 30 Degrees C by the Pour Plate Technique; ISO: Geneva, Switzerland, 2013. [Google Scholar]
- ISO 7218:2007. Microbiology of Food and Animal Feeding Stuffs—General Requirements and Guidance for Microbiological Examinations; ISO: Geneva, Switzerland, 2007. [Google Scholar]
- Health Protection Agency. Guidelines for Assessing the Microbiological Safety of Ready-to-Eat Foods Placed on the Market; Health Protection Agency: London, UK, 2009.
- Santos, M.; Correia, C.; Cunha, M.; Saraiva, M.; Novais, M. Valores Guia para Avaliação da Qualidade Microbiológica de Alimentos Prontos a Comer Preparados em Estabelecimentos de Restauração; INSA: Lisbon, Portugal, 2005; pp. 66–68. [Google Scholar]
- ISO 8586:2012. Sensory Analysis–General Guidelines for the Selection, Training and Monitoring of Selected Assessors and Expert Sensory Assessors; ISO: Geneva, Switzerland, 2012. [Google Scholar]
- Mesías, M.; Holgado, F.; Sevenich, R.; Briand, J.C.; Márquez Ruiz, G.; Morales, F.J. Fatty acids profile in canned tuna and sardine after retort sterilization and high pressure thermal sterilization treatment. J. Food Nutr. Res. 2015, 54, 171–178. [Google Scholar]
- Munshi, A.; Ali, S.; Shakir, S. Seasonal variations in biochemical composition of sardines and mullet from Pakistani Waters. J. Chem. Soc. Pak. 2005, 27, 190. [Google Scholar]
- Howgate, P. Traditional methods. In Fishery Products: Quality, Safety and Authenticity; Rehbein, H., Oehlenschlager, J., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Selmi, S.; Monser, L.; Sadok, S. The influence of local canning process and storage on pelagic fish from Tunisia: Fatty acid profiles and quality indicators. J. Food Process. Preserv. 2008, 32, 443–457. [Google Scholar] [CrossRef]
- Afonso, C.; Bandarra, N.M.; Nunes, L.; Cardoso, C. Tocopherols in seafood and aquaculture products. Crit. Rev. Food Sci. Nutr. 2016, 56, 128–140. [Google Scholar] [CrossRef]
- Polat, A.; Özogul, Y.; Kuley, E.; Özogul, F.; Özyurt, G.; Şimşek, A. Tocopherol content of commercial fish species as affected by microwave cooking. J. Food Biochem. 2013, 37, 381–387. [Google Scholar] [CrossRef]
- Hong, H.; Luo, Y.; Zhou, Z.; Bao, Y.; Lu, H.; Shen, H. Effects of different freezing treatments on the biogenic amine and quality changes of bighead carp (Aristichthys nobilis) heads during ice storage. Food Chem. 2013, 138, 1476–1482. [Google Scholar] [CrossRef]
- Siriamornpun, S.; Yang, L.; Kubola, J.; Li, D. changes of omega-3 fatty acid content and lipid composition in canned tuna during 12-month storage. J. Food Lipids 2008, 15, 164–175. [Google Scholar] [CrossRef]
- European Commission. Commission Regulation (EC) No 1022/2008 of 17 October 2008 amending Regulation (EC) No 2074/2005 as regards the total volatile basic nitrogen (TVB-N) limits. Off. J. Eur. Union 2008, 52, 86–88. [Google Scholar]
- Shakila, R.J.; Jeyasekaran, G.; Vyla, S.A.P.; Kumar, R.S. Effect of delayed processing on changes in histamine and other quality characteristics of 3 commercially canned fishes. J. Food Sci. 2005, 70, M24–M29. [Google Scholar] [CrossRef]
- FDA. Fish and Fishery Products Hazards and Controls Guidance; US Department of Health and Human Services Food and Drug Administration: Tampa, FL, USA, 2019.
- Atiya Ali, M.; Poortvliet, E.; Strömberg, R.; Yngve, A. Polyamines in foods: Development of a food database. Food Nutr. Res. 2011, 55, 5572. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Commission Regulation (EC) No of 5 December 2007 amending Regulation (EC) No 2073/2005 on microbiological criteria for foodstuffs. Off. J. Eur. Union 2007, 1441, 12–29. [Google Scholar]
- Carpenter, R.P. What is sensory analysis used for? In Guidelines for Sensory Analysis in Food Product Development and Quality Control, 2nd ed.; Carpenter, R.P., Lyon, D.H., Hasdell, T.A., Eds.; Springer Science & Business Media: Gaithersburg, MD, USA, 2012. [Google Scholar]
- Ashie, I.; Smith, J.; Simpson, B.; Haard, N.F. Spoilage and shelf-life extension of fresh fish and shellfish. Crit. Rev. Food Sci. Nutr. 1996, 36, 87–121. [Google Scholar] [CrossRef] [PubMed]
- Qiao, M.; Fletcher, D.; Smith, D.; Northcutt, J. Effects of raw broiler breast meat color variation on marination and cooked meat quality. Poult. Sci. 2002, 81, 276–280. [Google Scholar] [CrossRef]
- Oranusi, S.; Braide, W.; Osigwe, G. Investigation on the microbial profile of canned foods. J. Biol. Food Sci. Res. 2012, 1, 15–18. [Google Scholar]
- Triqui, R.; Bouchriti, N. Freshness Assessments of Moroccan Sardine (Sardina pilchardus): Comparison of Overall Sensory Changes to Instrumentally Determined Volatiles. J. Agric. Food Chem. 2003, 51, 7540–7546. [Google Scholar] [CrossRef]
- Ali, A.; Sudhir, B.; Srinivisa Gopal, T.K. Effect of Heat Processing on the Texture Profile of Canned and Retort Pouch Packed Oil Sardine (Sardinella longiceps) in Oil Medium. J. Food Sci. 2005, 70, S350–S354. [Google Scholar] [CrossRef]
| Parameters | Sample | Storage Time (Days) | p | r | |||||
|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 7 | ||||
| Water (%) | SB | 68.6 ± 1.0 | 69.9 ± 1.1 | 69.7 ± 1.5 | 67.6 ± 0.5 | 68.7 ± 1.2 | 70.1 ± 1.0 | 0.104 | n.s. |
| SO | 58.8 ± 0.7 | 57.7 ± 2.5 | 61.7 ± 1.1 | 58.2 ± 2.2 | 58.2 ± 2.1 | 58.6 ± 0.2 | 0.128 | n.s. | |
| ST | 61.7 ± 1.1 | 62.9 ± 0.6 | 61.8 ± 0.4 | 62.5 ± 0.2 | 60.0 ± 0.7 | 62.0 ± 0.8 | 0.336 | n.s. | |
| pH | SB | 6.65 ± 0.04 c | 6.93 ± 0.04 a | 6.97 ± 0.05 a | 6.81 ± 0.05 b | 6.76 ± 0.06 b | 6.62 ± 0.06 c | ˂0.001 | −0.390 * |
| SO | 6.64 ± 0.04 a | 6.56 ± 0.04 b | 6.51 ± 0.04 b | 6.45 ± 0.04 c | 6.55 ± 0.06 b | 6.52 ± 0.06 b | ˂0.001 | n.s. | |
| ST | 5.95 ± 0.05 | 5.93 ± 0.07 | 5.89 ± 0.03 | 5.87± 0.11 | 5.94 ± 0.11 | 5.90 ± 0.08 | 0.484 | n.s. | |
| Fat (g.100 g−1) | SB | 5.77 ± 2.90 | 4.08 ± 1.95 | 6.13 ± 0.65 | 5.52 ± 0.67 | 5.92 ± 1.21 | 4.30 ± 0.96 | 0.517 | n.s. |
| SO | 13.68 ± 0.84 | 13.40 ± 1.35 | 12.63 ± 2.35 | 13.75 ± 1.00 | 11.81 ± 0.41 | 12.32 ± 1.38 | 0.448 | n.s. | |
| ST | 10.31 ± 1.28 a | 7.62 ± 0.97 b | 9.69 ± 0.81 a | 9.69 ± 0.16 a | 10.23 ± 0.65 a | 10.36 ± 0.71 a | 0.014 | n.s. | |
| LC-PUFAs (g.100 g−1) | SB | 1.93 ± 1.07 | 1.48 ± 0.61 | 2.19 ± 0.24 | 2.01 ± 0.13 | 2.22 ± 0.40 | 1.71± 0.38 | 0.584 | n.s. |
| SO | 2.67 ± 0.26 | 2.04 ± 0.49 | 2.14 ± 0.74 | 2.08 ± 0.39 | 2.06 ± 0.13 | 2.19 ±0.15 | 0.468 | n.s. | |
| ST | 3.62 ± 0.51 a | 2.53 ± 0.52 b | 3.49 ± 0.44 a | 3.18 ± 0.24 a,b | 3.69 ± 0.29 a | 3.73 ± 0.20 a | 0.020 | n.s. | |
| Vitamin E (mg.100 g−1) | SB | 0.23 ± 0.12 | 0.20 ± 0.11 | 0.45 ± 0.06 | 0.33 ± 0.11 | 0.34 ± 0.13 | 0.22 ± 0.02 | 0.057 | n.s. |
| SO | 4.90 ± 0.29 | 6.79 ± 2.09 | 4.63 ± 1.14 | 6.30 ± 1.27 | 5.36 ± 1.49 | 4.36 ± 0.55 | 0.208 | n.s. | |
| ST | 1.61 ± 0.11 | 1.68 ± 0.21 | 1.62 ± 0.14 | 1.46 ± 0.18 | 1.67 ± 0.23 | 1.25 ± 0.12 | 0.063 | n.s. | |
| TBARS (mg MDA.kg−1) | SB | 3.54 ± 0.18 | 3.38 ± 0.82 | 3.26 ± 0.32 | 3.37 ± 0.14 | 4.24 ± 0.95 | 4.40 ± 0.88 | 0.203 | n.s. |
| SO | 3.75 ± 0.45 | 4.04 ± 0.39 | 3.60 ± 0.32 | 3.91 ± 0.27 | 4.22 ± 0.71 | 3.99 ± 0.47 | 0.635 | n.s. | |
| ST | 1.96 ± 0.40 c | 1.86 ± 0.23 c | 2.50 ± 0.65 b,c | 3.44 ± 1.29 a,b | 3.35 ± 0.29 a,b | 3.80 ± 0.37 a | 0.012 | 0.742 ** | |
| TVB-N (mg N.100 g−1) | SB | 40.2 ± 0.3 d | 41.1 ± 0.3 c | 41.0 ± 0.4 c | 43.1 ± 0.3 b | 38.9 ± 0.3 e | 44.7 ± 0.3 a | ˂0.001 | 0.586 * |
| SO | 43.8 ± 0.6 | 44.0 ± 1.1 | 41.9 ± 0.6 | 43.1 ± 1.2 | 44.3 ± 0.7 | 43.8 ± 0.8 | 0.053 | n.s. | |
| ST | 36.2 ± 0.4 | 36.0 ± 1.2 | 36.3 ± 0.6 | 35.6 ± 1.3 | 35.9 ± 0.4 | 35.3 ± 1.1 | 0.767 | n.s. | |
| Parameters | Sample | Storage Time (Days) | p | r | |||||
|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 7 | ||||
| L* | SB | 37.0 ± 1.6 | 36.0 ± 1.4 | 36.7 ± 2.8 | 36.3 ± 2.6 | 35.4 ± 1.5 | 35.9 ± 1.6 | 0.930 | n.s. |
| SO | 30.1 ± 2.2 | 29.6 ± 1.5 | 31.8 ± 1.3 | 30.1 ± 1.7 | 31.4 ± 1.2 | 31.0 ± 1.1 | 0.475 | n.s. | |
| ST | 35.4 ± 1.3 | 35.0 ± 1.7 | 35.9 ± 1.2 | 36.3 ± 1.0 | 35.6 ± 1.2 | 35.2 ± 1.2 | 0.807 | n.s. | |
| a* | SB | −0.9 ± 0.1 b | −0.8 ± 0.1 a,b | −0.9 ± 0.0 b | −0.9 ± 0.0 b | −0.9 ± 0.0 b | −0.8 ± 0.1 a | 0.021 | 0.470 * |
| SO | −0.5 ± 0.1 b | −0.6 ± 0.1 a,b | −0.6 ± 0.1 a,b | −0.6 ± 0.0 b | −0.7 ± 0.2 a,b | −0.8 ± 0.1 a | 0.029 | −0.700 ** | |
| ST | 17.6 ± 1.1 | 17.7 ± 0.4 | 17.6 ± 0.9 | 17.2 ± 0.8 | 17.9 ± 1.0 | 17.5 ± 0.7 | 0.949 | n.s. | |
| b* | SB | 4.3 ± 0.4 a | 4.4 ± 0.3 a,b | 4.2 ± 0.1 a,b,c | 4.7 ± 0.5 a | 3.7 ± 0.2 c | 4.1 ± 0.0 b,c | 0.029 | −0.310 * |
| SO | 1.9 ± 0.1 | 1.8 ± 0.1 | 1.9 ± 0.2 | 1.8 ± 0.1 | 1.8 ± 0.1 | 1.9 ± 0.1 | 0.266 | n.s. | |
| ST | 19.6 ± 1.2 | 20.3 ± 2.0 | 20.4 ± 1.2 | 21.1 ± 0.3 | 20.9 ± 1.2 | 20.4 ± 0.8 | 0.746 | n.s. | |
| ΔE | SB | 0.0 ± 0.0 c | 1.1 ± 0.2 b | 1.2 ± 0.5 a,b | 1.5 ± 0.4 a,b | 2.8 ± 1.2 a,b | 1.3 ± 1.5 a,b | 0.034 | n.s. |
| SO | 0.0 ± 0.0 | 0.9 ± 0.3 | 1.8 ± 1.3 | 0.6 ± 0.4 | 1.4 ± 1.7 | 1.1 ± 0.8 | 0.339 | n.s. | |
| ST | 0.0 ± 0.0 | 2.3 ± 0.3 | 2.3 ± 1.1 | 3.0 ± 0.7 | 3.0 ± 1.2 | 2.3 ± 0.8 | 0.339 | n.s. | |
| Parameters | Sample | Storage Time (Days) | p | r | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | 7 | |||||
| Exterior | L* | SB | 76.3 ± 5.1 a | 72.4 ± 4.5 a,b | 72.4 ± 4.3 a,b | 73.4 ± 2.0 a | 70.2 ± 4.4 b | 69.9 ± 5.0 b | ˂0.001 | −0.369 ** |
| SO | 73.1 ± 4.5 a | 71.0 ± 8.8 a,b | 76.4 ± 5.2 b,c | 63.6 ± 4.3 c,d | 61.9 ± 6.6 c,d | 61.8 ± 5.7 d | ˂0.001 | −0.524 ** | ||
| ST | 62.2 ± 5.9 a | 56.8 ± 2.2 b | 55.7 ± 6.6 b | 57.9 ± 6.5 a,b | 58.9 ± 5.9 a,b | 58.4 ± 6.3 a,b | 0.001 | n.s. | ||
| a* | SB | −0.8 ± 0.6 | −0.7 ± 0.5 | −0.8 ± 0.4 | −0.8 ± 0.5 | −0.7 ± 0.6 | −0.7 ± 0.6 | 0.946 | n.s. | |
| SO | −0.2 ± 0.7 | −0.6 ± 0.9 | −0.5 ± 0.9 | −0.6 ± 0.7 | −0.6 ± 0.7 | −0.7 ± 0.9 | 0.393 | n.s. | ||
| ST | 11.8 ± 3.7 | 10.9 ± 2.5 | 10.6 ± 3.7 | 10.2 ± 3.3 | 10.9 ± 3.6 | 10.3 ± 2.6 | 0.516 | n.s. | ||
| b* | SB | 12.5 ± 2.9 | 13.9 ± 2.7 | 12.3 ± 2.1 | 12.1 ± 2.9 | 13.3 ± 3.0 | 12.1 ±2.9 | 0.130 | n.s. | |
| SO | 10.3 ± 2.9 a | 9.9 ± 3.7 a | 10.3 ± 2.5 a | 10.1 ± 2.8 a | 8.3 ± 2.4 b | 8.0 ±3.0 b | 0.006 | −0.277 ** | ||
| ST | 36.9 ± 6.4 a | 32.9 ± 5.5 b | 32.6 ± 6.5 b | 32.7 ± 5.2 b | 32.3 ± 5.9 b | 33.0 ± 4.1 b | 0.032 | n.s. | ||
| ΔE | SB | 0.0 ± 0.0 b | 8.7 ± 4.7 a | 7.7 ± 4.3 a | 6.9 ± 3.3 a | 8.1 ± 5.0 a | 8.0 ± 4.6 a | ˂0.001 | 0.335 ** | |
| SO | 0.0 ± 0.0 b | 9.5 ± 5.1 a | 8.8 ± 5.3 a | 10.7 ± 4.7 a | 11.5 ± 5.3 a | 13.0 ± 6.6 a | ˂0.001 | 0.510 ** | ||
| ST | 0.0 ± 0.0 b | 11.4 ± 4.7 a | 13.3 ± 7.6 a | 12.3 ± 5.6 a | 12.8 ± 6.8 a | 12.3 ± 4.4 a | ˂0.001 | 0.375 ** | ||
| Interior | L* | SB | 52.4 ± 3.9 a | 46.7 ± 8.4 b | 45.8 ± 6.0 b | 46.6 ± 2.1 b | 45.5 ± 4.2 b | 46.6 ± 4.5 b | ˂0.001 | −0.325 ** |
| SO | 48.9 ± 5.0 a | 39.0 ± 6.0 b | 43.0 ± 6.4 c | 45.6 ± 5.4 c | 44.5 ± 6.7 c | 42.1 ± 6.4 b,c | ˂0.001 | n.s. | ||
| ST | 53.9 ± 3.6 a | 48.0 ± 3.0 b | 48.4 ± 5.4 b | 48.1 ± 3.4 b | 49.1 ± 1.8 b | 48.9 ± 3.3 b | ˂0.001 | −0.216 ** | ||
| a* | SB | 7.4 ± 2.6 a | 6.4 ± 2.7 a,b,c | 6.6 ± 2.0 a,b | 6.9 ± 2.7 a,b | 5.2 ± 1.8 b,c | 5.0 ± 1.7 c | ˂0.001 | −0.314 ** | |
| SO | 6.9 ± 2.0 b,c | 7.3 ± 2.2 a,b | 8.1 ± 2.2 a | 6.7 ± 2.0 b,c | 5.9 ± 2.0 c | 6.8 ± 1.9 b,c | 0.006 | n.s. | ||
| ST | 11.1 ± 2.2 a | 8.3 ± 2.1 b | 8.6 ± 2.0 b,c | 9.4 ± 2.5 b,c | 9.3 ± 2.0 b,c | 9.8 ± 2.7 c | ˂0.001 | n.s. | ||
| b* | SB | 18.2 ± 2.0 a | 16.0 ± 2.5 b | 17.3 ± 2.8 a,b | 16.1 ± 1.8 b | 16.0 ± 1.4 b | 16.7 ± 1.8 b | ˂0.001 | −0.156 * | |
| SO | 19.3 ± 2.5 a | 13.7 ± 2.5 c | 15.2 ± 2.2 b | 15.8 ± 2.0 b | 14.8 ± 2.9 b,c | 14.7 ± 2.5 b,c | ˂0.001 | −0.280 ** | ||
| ST | 24.0 ± 2.6 a | 19.3 ± 3.1 b | 19.4 ± 2.2 b | 19.5 ± 2.7 b | 19.8 ± 1.7 b | 19.3 ± 2.1 b | ˂0.001 | −0.315 ** | ||
| ΔE | SB | 0.0 ± 0.0 a | 9.3 ± 4.7 b | 8.3 ± 5.0 b | 8.1 ± 2.8 b | 9.6 ± 4.3 b | 9.4 ± 3.9 b | ˂0.001 | 0.409 ** | |
| SO | 0.0 ± 0.0 c | 10.8 ± 4.5 a | 9.7 ± 5.8 b | 7.7 ± 5.3 b | 8.8 ± 4.1 b,c | 10.4 ± 8.5 a,b | ˂0.001 | 0.326 ** | ||
| ST | 0.0 ± 0.0 b | 9.6 ± 3.8 a | 10.1 ± 4.9 a | 9.1 ± 3.1 a | 8.0 ± 3.9 a | 8.6 ± 4.0 a | ˂0.001 | 0.304 ** | ||
| Quality Indicators | Microbiological Analysis (CFU/g) | |||
|---|---|---|---|---|
| Storage Time (Days) | ||||
| 0 | 1 | 2 | 7 | |
| Aerobic mesophiles | ˂10 | ˂10 | ˂10 | ˂10 |
| Aerobic psychrophiles | ˂102 | ˂102 | ˂102 | * present but <4 × 102 |
| Aerobic thermophiles | ˂102 | ˂102 | ˂102 | * present but <4 × 102 |
| Anaerobic mesophilic sulphite-reducing bacteria | ˂10 | ˂10 | ˂10 | ˂10 |
| Anaerobic thermophilic sulphite-reducing bacteria | ˂10 | ˂10 | ˂10 | ˂10 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cruz, R.; Pereira, V.; Pinho, T.; Ferreira, I.M.P.L.V.O.; Novais, C.; Casal, S. Safety and Quality of Canned Sardines after Opening: A Shelf-Stability Study. Foods 2022, 11, 991. https://doi.org/10.3390/foods11070991
Cruz R, Pereira V, Pinho T, Ferreira IMPLVO, Novais C, Casal S. Safety and Quality of Canned Sardines after Opening: A Shelf-Stability Study. Foods. 2022; 11(7):991. https://doi.org/10.3390/foods11070991
Chicago/Turabian StyleCruz, Rebeca, Vânia Pereira, Teresa Pinho, Isabel M. P. L. V. O. Ferreira, Carla Novais, and Susana Casal. 2022. "Safety and Quality of Canned Sardines after Opening: A Shelf-Stability Study" Foods 11, no. 7: 991. https://doi.org/10.3390/foods11070991
APA StyleCruz, R., Pereira, V., Pinho, T., Ferreira, I. M. P. L. V. O., Novais, C., & Casal, S. (2022). Safety and Quality of Canned Sardines after Opening: A Shelf-Stability Study. Foods, 11(7), 991. https://doi.org/10.3390/foods11070991









