Comprehensive Study of Traditional Plant Ground Ivy (Glechoma hederacea L.) Grown in Croatia in Terms of Nutritional and Bioactive Composition
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Chemicals
2.1.1. Materials
2.1.2. Chemicals
2.2. Methods
2.2.1. Preparation of Plant Materials
2.2.2. Determination of Micro- and Macrocomposition
2.2.3. Extraction of Phenolic Compounds
2.2.4. Determination of Total Phenolic Content (TPC) and Antioxidant Capacity
2.2.5. HPLC Determination of Individual Phenolic Compounds
2.2.6. LC-MS/MS Analysis
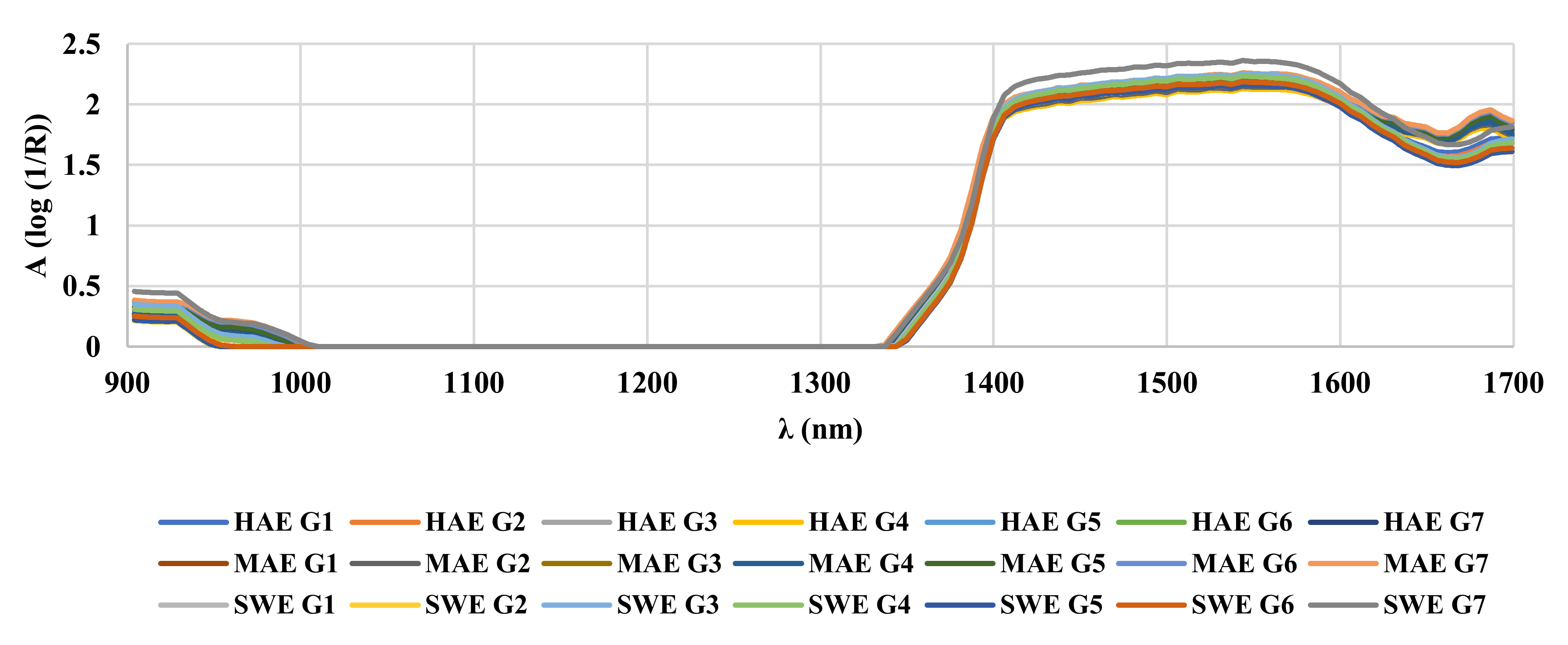
2.2.7. Near-Infrared Spectroscopy (NIR)
2.2.8. Statistical Analysis
3. Results and Discussion
3.1. Macro- and Microcomposition Analysis
3.2. Phenolic Profile Analysis
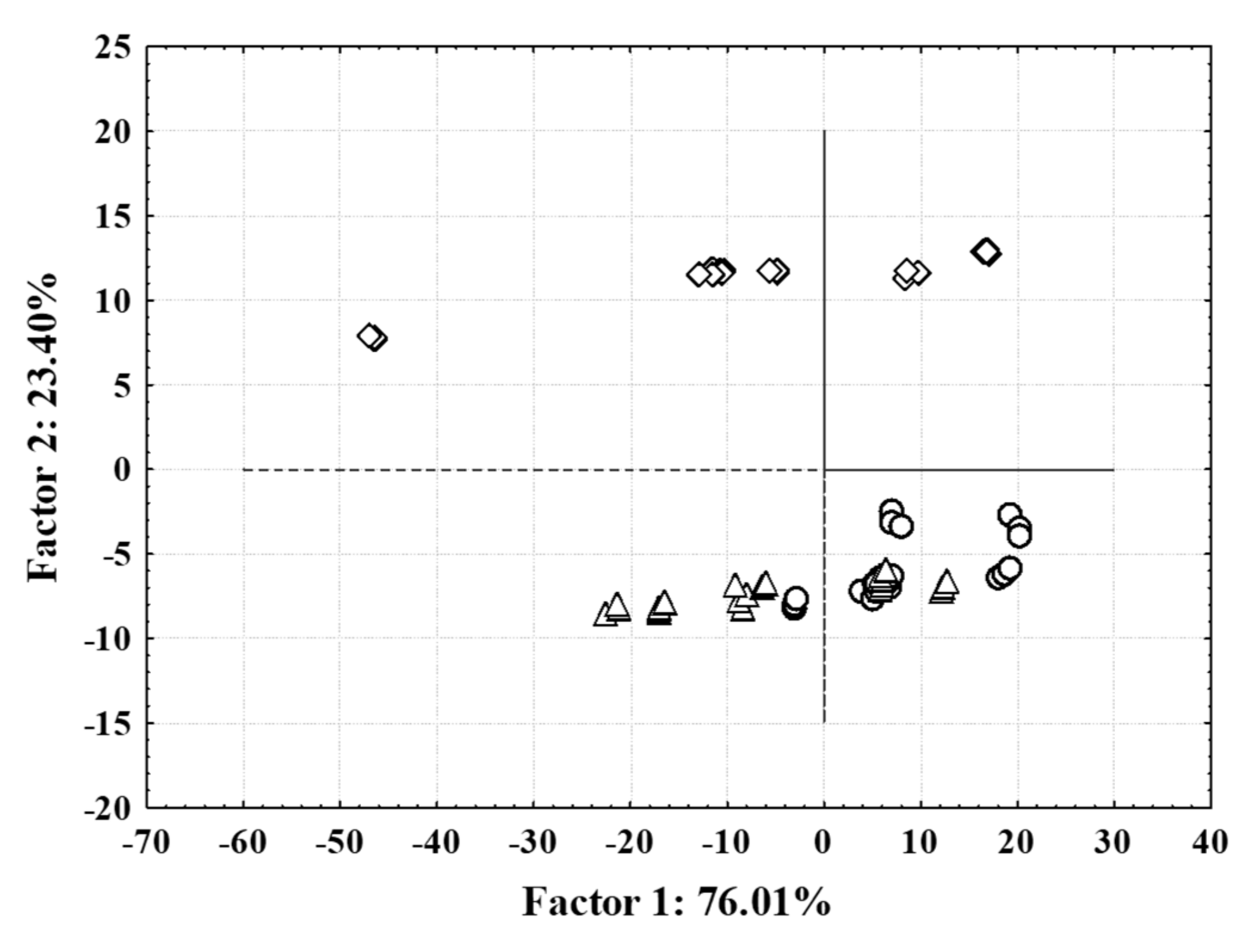
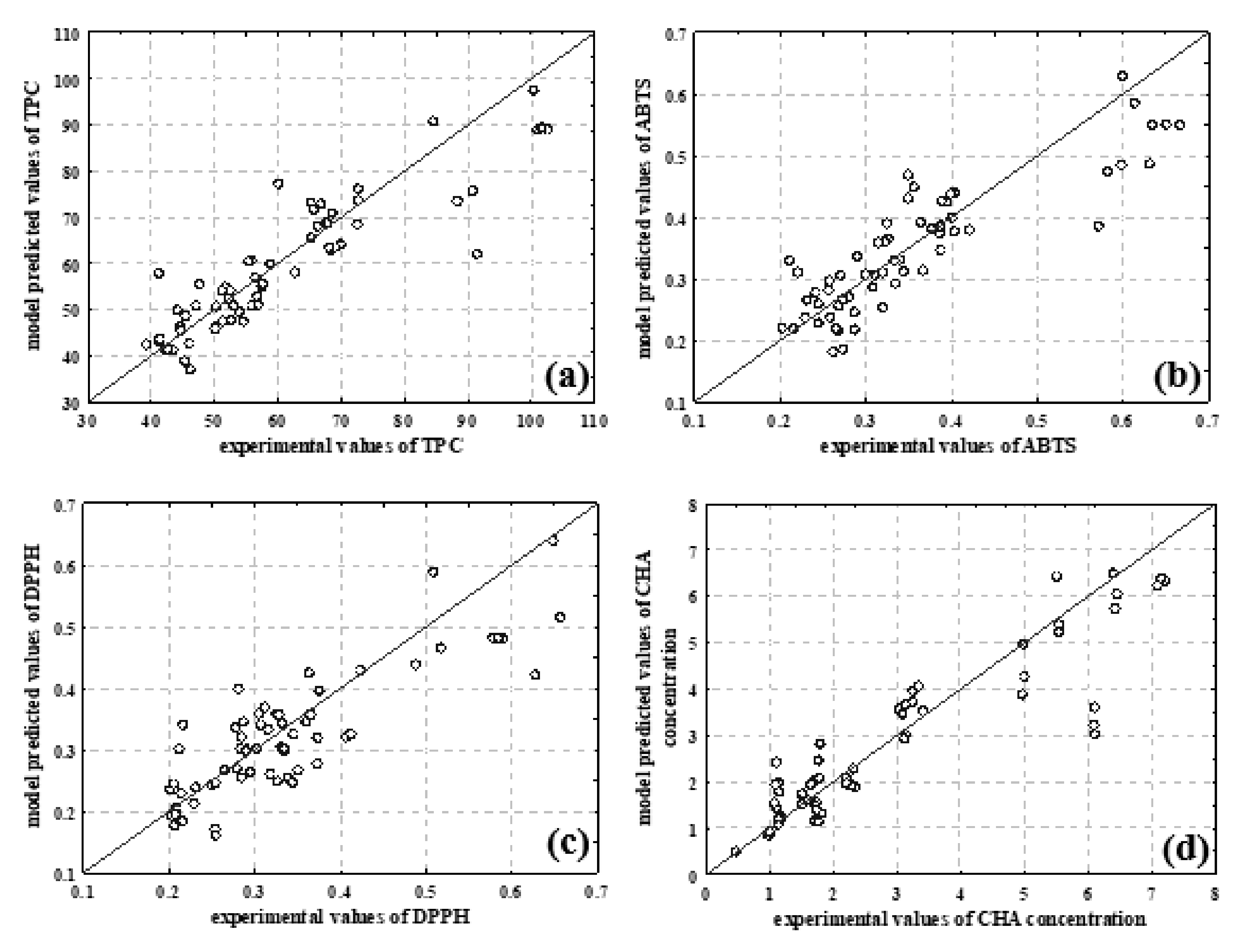
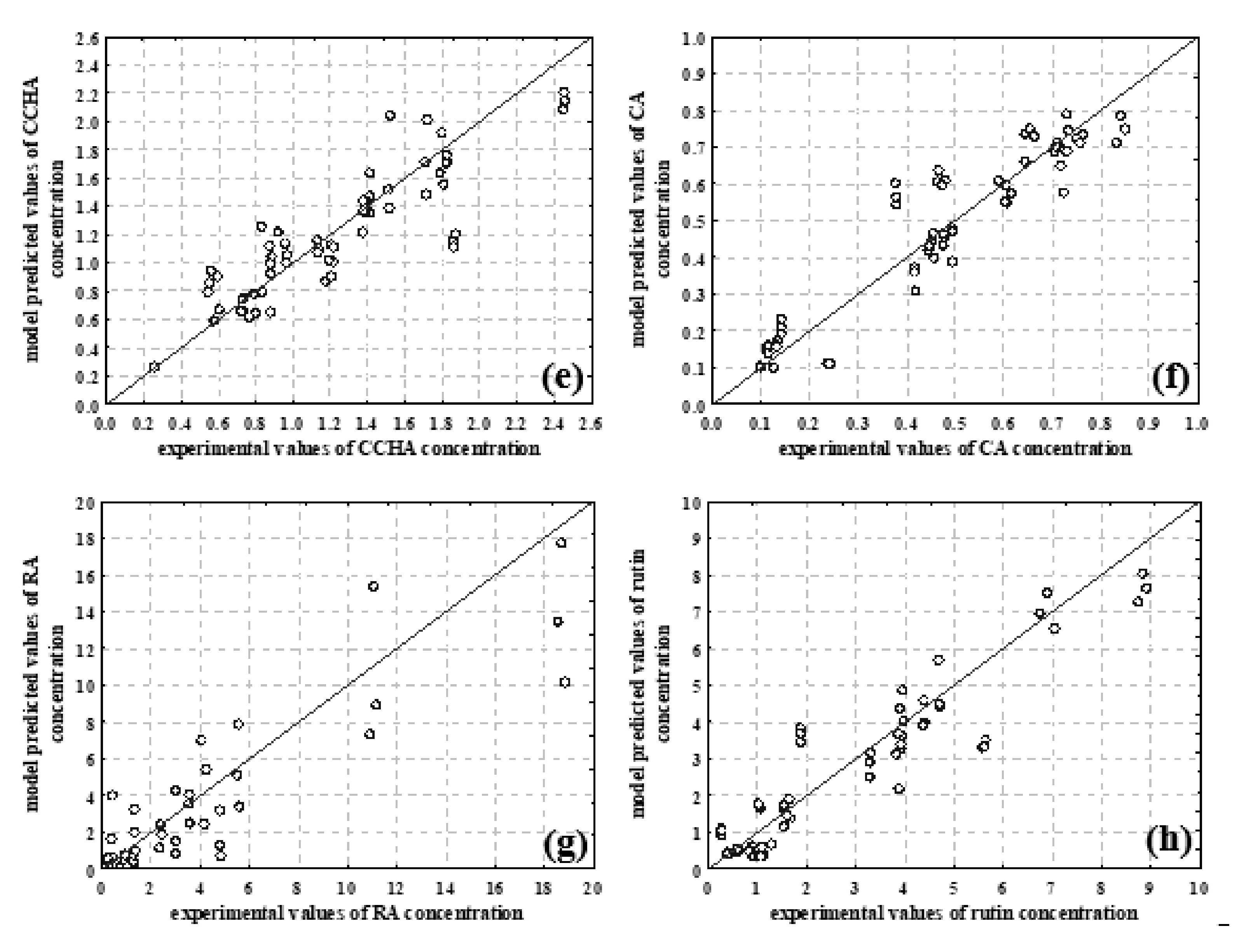
3.3. NIR Spectroscopy, PCA and ANN Modelling
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nikolić, T. The diversity of Croatian vascular flora based on the checklist and CROFlora database. Acta Bot. Croat. 2001, 60, 49–67. [Google Scholar]
- Nikolić, T.; Rešetnik, I. Plant uses in Croatia. Phytol. Balcania 2007, 13, 229–238. [Google Scholar]
- Croatian Flora. Available online: https://botanickivrt.biol.pmf.hr/en/about-our-collections/croatian-flora/ (accessed on 20 December 2020).
- Łuczaj, Ł.; Dolina, K.; Fressel, N.; Perković, S. Ethnobotany and Biocultural Diversities in the Balkans; Pieroni, A., Quave, C.L., Eds.; Springer: New York, NY, USA, 2014. [Google Scholar]
- Sik, B.; Kapcsándi, V.; Székelyhidi, R.; Hanczne, E.L.; Ajtony, Z. Recent advantages in the analysis of rosmarinic acid from herbs in the Lamiaceae family. Nat. Prod. Commun. 2019, 14, 1–10. [Google Scholar] [CrossRef]
- Chou, S.T.; Chan, Y.R.; Chung, Y.C. Studies on the antimutagenicity and antioxidant activity of hot water extract of Glechoma hederacea. J. Food Drug Anal. 2012, 20, 637–645. [Google Scholar] [CrossRef]
- Chou, S.T.; Lin, T.H.; Peng, H.Y.; Chao, W.W. Phytochemical profile of hot water extract of Glechoma hederacea and its antioxidant, and anti-inflammatory activities. Life Sci. 2019, 231, 116519. [Google Scholar] [CrossRef] [PubMed]
- Olszowy, M. What is responsible for antioxidant properties of polyphenolic compounds from plants? Plant Physiol. Biochem. 2019, 144, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Mandal, V.; Mohan, Y.; Hemalatha, S. Microwave assisted extraction—An innovative and promising extraction tool for medicinal plant research. Pharmacogn. Rev. 2007, 1, 7–18. [Google Scholar]
- Zhang, J.; Wen, C.; Zhang, H.; Duan, Y.; Ma, H. Recent advances in the extraction of bioactive compounds with subcritical water: A review. Trends Food Sci. Technol. 2020, 95, 183–195. [Google Scholar] [CrossRef]
- Padmore, J.M. Animal feed—AOAC official method 930.15—Moisture in animal feed. In Official Methods of Analysis, 15th ed.; Helrich, K., Ed.; AOAC International: Arlington, VA, USA, 1990. [Google Scholar]
- Padmore, J.M. Animal feed—AOAC official method 976.05—Protein (crude) in animal feed, automated Kjeldahl method. In Official Methods of Analysis, 15th ed.; Helrich, K., Ed.; AOAC International: Arlington, VA, USA, 1990. [Google Scholar]
- Padmore, J.M. Animal feed—AOAC official method 920.39—Fat (crude) or ether extract in animal feed. In Official Methods of Analysis, 15th ed.; Helrich, K., Ed.; AOAC International: Arlington, VA, USA, 1990. [Google Scholar]
- Padmore, J.M. Animal feed—AOAC official method 942.05—Ash of animal feed. In Official Methods of Analysis, 15th ed.; Helrich, K., Ed.; AOAC International: Arlington, VA, USA, 1990. [Google Scholar]
- EN ISO 5509; Animal and Vegetable Fats and Oils—Preparation of Methyl Esters of Fatty Acids. International Organization for Standardization: Geneva, Switzerland, 2020.
- McCleary, B.V.; DeVries, J.W.; Rader, J.I.; Cohen, G.; Prosky, L.; Mugford, D.C.; Okuma, K. Determination of insoluble, soluble, and total dietary fibre (CODEX definition) by enzymatic-gravimetric method and liquid chromatography: Collaborative study. J. AOAC Int. 2012, 95, 824–844. [Google Scholar] [CrossRef]
- Vihnanek Lazarus, M.; Sekovanić, A.; Kljaković-Gašpić, A.; Orct, T.; Jurasović, J.; Kusak, J.; Reljić, S.; Huber, Đ. Cadmium and lead in grey wolf liver samples: Optimisation of a microwave-assisted digestion method. Arch. Ind. Hyg. Toxicol. 2013, 64, 395–403. [Google Scholar] [CrossRef]
- Šeremet, D.; Jokić, S.; Aladić, K.; Vojvodić Cebin, A.; Mandura, A.; Komes, D. Optimization of heat-, microwave-assisted and subcritical water extraction of phenolic compounds from ground ivy (Glechoma hederacea L.) using response surface methodology. J. Appl. Res. Med. Aromat. Plants 2021, 25, 100346. [Google Scholar] [CrossRef]
- Jokić, S.; Aladić, K.; Šubarić, D. Subcritical water extraction laboratory plant design and application. Annu. Croat. Acad. Eng. 2018, 21, 247–258. [Google Scholar]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphotungstic acid reagents. Am. J. Enol. Viticult. 1965, 16, 144–158. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT—Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorisation assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Fang, N.; Yu, S.; Prior, R.L. LC/MS/MS characterization of phenolic constituents in dried plumps. J. Agric. Food Chem. 2002, 50, 3579–3585. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.B.; Rai, D.K.; Brunton, N.P.; Martin-Diana, A.B.; Barry-Ryan, C. Characterization of phenolic composition in Lamiaceae species by LC-ESI-MS/MS. J. Agric. Food Chem. 2010, 58, 10576–10581. [Google Scholar] [CrossRef] [PubMed]
- Plazonić, A.; Bucar, F.; Maleš, Ž.; Mornar, A.; Nigović, B.; Kujundžić, N. Identification and quantification of flavonoids and phenolic acids in burr parsley (Caucalis platycarpos L.), using high-performance liquid chromatography with diode array detection and electrospray ionization mass spectrometry. Molecules 2009, 14, 2466–2490. [Google Scholar] [CrossRef] [PubMed]
- Arruda, H.S.; Pereira, G.A.; de Morais, D.R.; Eberlin, M.N.; Pastore, G.M. Determination of free, esterified, glycosylated and insoluble-bound phenolics composition in the edible part of araticum fruit (Annona crassiflora Mart.) and its by-products by HPLC-ESI-MS/MS. Food Chem. 2018, 245, 738–749. [Google Scholar] [CrossRef]
- Atoui, A.K.; Mansouri, A.; Boskou, G.; Kefalas, P. Tea and hebal infusions: Their antioxidant activity and phenolic profile. Food Chem. 2005, 89, 27–36. [Google Scholar] [CrossRef]
- Barros, L.; Carvalho, A.M.; Ferreira, C.F.R. From famine plants to tasty and fragrant spices: Three Lamiaceae of general dietary relevance in traditional cuisine of Trás-os-Montes (Portugal). LWT—Food Sci. Technol. 2011, 44, 543–548. [Google Scholar] [CrossRef][Green Version]
- Iqdal, A.; Khalil, I.A.; Ateeq, N.; Khan, M.S. Nutritional quality of important food legumes. Food Chem. 2006, 97, 331–335. [Google Scholar] [CrossRef]
- Jo, D.; Lee, J.; Noh, J.; Kim, O.K.; Kwon, J.H. Chemical Composition and Electron Donating and Nitrite Scavenging Activities of Glechoma hederacea var. longituba NAKAI. Prev. Nutr. Food Sci. 2001, 6, 142–146. [Google Scholar]
- Cacan, E.; Kokten, K.; Kilic, O. Leaf fatty acid composition of some Lamiaceae taxa from Turkey. Prog. Nutr. 2018, 20, 231–236. [Google Scholar] [CrossRef]
- Juranović Cindrić, I.; Zeiner, M.; Glamuzina, E.; Stingeder, G. Elemental characterisation of the medical herbs Salvia officinalis L. and Teucrium montanum L. grown in Croatia. Microchem. J. 2013, 107, 185–189. [Google Scholar] [CrossRef]
- Weaver, C.M. Potassium and health. Adv. Nutr. 2013, 4, 368S–377S. [Google Scholar] [CrossRef]
- Miller, G.D.; Jarvis, J.K.; McBean, L.D. The importance of meeting calcium needs with foods. J. Am. Coll. Nutr. 2001, 20, 168S–185S. [Google Scholar] [CrossRef]
- Rehder, D. The potentiality of vanadium in medicinal applications. Inorg. Chim. Acta 2020, 504, 119445. [Google Scholar] [CrossRef]
- Lovkova, M.Y.; Buzuk, G.N.; Sokolova, S.M.; Kliment’eva, N.I. Chemical features of medicinal plants (Review). Appl. Biochem. Microbiol. 2001, 37, 229–237. [Google Scholar] [CrossRef]
- Zinicovscaia, I.; Gundorina, S.; Vergel, K.; Grozdov, D.; Ciocarlan, A.; Aricu, A.; Dragalin, I.; Ciocarlan, N. Elemental analysis of Lamiaceae medicinal and aromatic plants growing in the Republic of Moldova using neutron activation analysis. Phytochem. Lett. 2020, 35, 119–127. [Google Scholar] [CrossRef]
- Stanojkovic-Sebic, A.; Pivic, R.; Josic, D.; Dinic, Z.; Stanojkovic, A. Heavy metals content in selected medicinal plants commonly used as components for herbal formulations. J. Agric. Sci. 2014, 21, 17–325. [Google Scholar] [CrossRef]
- Gilbert-López, B.; Plaza, M.; Mendiola, J.A.; Ibánez, E.; Herrero, M. Water Extraction of Bioactive Compounds; Dominguez, H., Gonzalez Munoz, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar] [CrossRef]
- Plaza, M.; Amigo-Benavent, M.; del Castillo, M.; Ibánez, E.; Herrero, M. Facts about the formation of new antioxidants in natural samples after subcritical water extraction. Food Res. Int. 2010, 43, 2341–2348. [Google Scholar] [CrossRef]
- Belščak-Cvitanović, A.; Stojanović, R.; Manojlović, V.; Komes, D.; Juranović Cindrić, I.; Nedović, V.; Bugarski, B. Encapsulation of polyphenolic antioxidants from medicinal plant extracts inalginate–chitosan system enhanced with ascorbic acid by electrostatic extrusion. Food Res. Int. 2011, 44, 1094–1101. [Google Scholar] [CrossRef]
- Kikuchi, M.; Goto, J.; Noguchi, S.; Kakuda, R.; Yaoita, Y. Glycosides from whole plants of Glechoma hederacea L. Nat. Med. Note 2008, 62, 479–480. [Google Scholar] [CrossRef]
- Li, X.; Zhang, L.; Zhang, Y.; Wang, D.; Wang, X.; Yu, L.; Zhang, W.; Li, P. Review of NIR spectroscopy methods for nondestructive quality analysis of oilseeds and edible oils. Trends Food Sci. Technol. 2020, 101, 172–181. [Google Scholar] [CrossRef]
- Cozzolino, D. The role of visible and infrared spectroscopy combined with chemometrics to measure phenolic compounds in grape and wine samples. Molecules 2015, 20, 726–737. [Google Scholar] [CrossRef]
- Valinger, D.; Kušen, M.; Jurinjak Tušek, A.; Panić, M.; Jurina, T.; Benković, M.; Radojčić Redovniković, I.; Gajdoš Kljusurić, J. Development of near infrared spectroscopy models for the quantitative prediction of olive leaves bioactive compounds content. Chem. Biochem. Eng. Q. 2018, 32, 535–543. [Google Scholar] [CrossRef]
- Valinger, D.; Jurina, T.; Šain, A.; Matešić, N.; Panić, M.; Benković, M.; Gajdoš Kljusurić, J.; Jurinjak Tušek, A. Development of ANN models based on UV-VIS-NIR spectra for rapid quantification of physical and chemical properties of industrial hemp extracts. Phytochem. Anal. 2020, 32, 326–338. [Google Scholar] [CrossRef]
- Jurinjak Tušek, A.; Marić, L.; Benković, M.; Valinger, D.; Jurina, T.; Gajdoš Kljusurić, J. In-vitro digestion of the bioactives originating from the Lamiaceae family herbal teas: A kinetic and PLS modelling study. J. Food Biochem. 2020, 44, e13233. [Google Scholar] [CrossRef] [PubMed]
- Beć, K.B.; Grabska, J.; Huck, C.W. NIR spectroscopy of natural medicines supported by novel instrumentation and methods for data analysis and interpretation. J. Pharm. Biomed. 2021, 193, 113686. [Google Scholar] [CrossRef] [PubMed]
| Sample | G1 | G2 | G3 | G4 | G5 | G6 | G7 |
|---|---|---|---|---|---|---|---|
| Dry matter (%) | 91.13 ± 0.10 abc | 91.86 ± 0.06 | 90.96 ± 0.03 a | 91.33 ± 0.14 bde | 91.27 ± 0.05 cdf | 91.45 ± 0.01 ef | 93.15 ± 0.12 |
| Crude protein content (% dw *) | 15.63 ± 0.13 abc | 14.75 ± 0.23 d | 15.61 ± 0.20 adef | 17.74 ± 0.17 | 23.13 ± 0.33 | 15.86 ± 0.27 beg | 16.16 ± 0.16 cfg |
| Crude oil content (% dw) | 2.80 ± 0.54 abcde | 2.05 ± 0.07 afghij | 1.10 ± 0.20 fk | 2.63 ± 0.71 bglmn | 1.86 ± 0.15 chklop | 2.39 ± 0.10 dimor | 2.30 ± 0.06 ejnpr |
| 0.18 ± 0.02 a | nd | 4.53 ± 0.48 | 0.18 ± 0.05 a | nd | nd | 1.25 ± 0.03 |
| 7.47 ± 0.09 abc | 7.70 ± 0.23 adef | 13.33 ± 2.28 g | 7.14 ± 0.14 bdh | 10.24 ± 0.82 eij | 11.68 ± 0.86 gi | 7.68 ± 0.42 cfhj |
| 0.93 ± 0.03 | nd | nd | 0.14 ± 0.01 | nd | nd | 1.07 ± 0.03 |
| 0.19 ± 0.01 | nd | nd | 0.07 ± 0.01 | nd | nd | 0.39 ± 0.02 |
| 2.28 ± 0.50 ab | 4.40 ± 1.25 c | 15.46 ± 0.50 d | 1.59 ± 0.00 ae | 16.33 ± 0.30 d | 5.89 ± 0.05 c | 1.88 ± 0.05 be |
| 28.88 ± 0.31 | 33.85 ± 1.76 | 18.19 ± 0.81 | 39.01 ± 0.10 | nd | 21.64 ± 0.03 | 8.70 ± 0.41 |
| 24.51 ± 0.12 a | 22.63 ± 0.22 b | 19.48 ± 0.44 | 21.71 ± 0.50 b | 24.95 ± 0.50 a | 32.35 ± 0.43 | 14.92 ± 0.29 |
| 29.45 ± 0.14 | 24.13 ± 0.81 | 18.25 ± 0.55 | 20.27 ± 0.51 a | 37.92 ± 1.44 | 21.96 ± 0.14 a | 27.15 ± 0.38 |
| 0.34 ± 0.16 a | nd | nd | 0.25 ± 0.01 a | nd | 2.15 ± 0.13 | 0.63 ± 0.04 |
| nd | nd | nd | nd | nd | nd | 6.52 ± 0.19 |
| 2.46 ± 0.13 | nd | nd | 3.22 ± 0.42 | nd | nd | 13.73 ± 0.21 |
| Crude mineral content (% dw) | 9.76 ± 0.09 abcd | 9.59 ± 0.10 aefg | 10.98 ± 0.44 h | 10.95 ± 0.23 h | 10.02 ± 0.13 bei | 9.20 ± 0.02 cf | 9.97 ± 0.40 dgi |
| Total dietary fibre (% dw) | 37.74 ± 2.11 a | 43.14 ± 1.15 bcd | 46.65 ± 1.19 bef | 55.23 ± 3.42 | 41.44 ± 0.65 acg | 44.05 ± 1.08 deg | 49.76 ± 1.10 f |
| 32.26 ± 0.55 a | 34.14 ± 0.65 a | 41.50 ± 0.82 b | 48.03 ± 0.18 | 36.47 ± 0.82 c | 37.03 ± 1.15 c | 40.61 ± 0.85 b |
| 5.48 ± 0.74 abc | 9.00 ± 0.89 d | 5.15 ± 0.43 ae | 7.19 ± 0.72 f | 4.97 ± 0.38 be | 7.02 ± 0.40 cf | 9.14 ± 0.26 d |
| Sample | G1 | G2 | G3 | G4 | G5 | G6 | G7 |
|---|---|---|---|---|---|---|---|
| Macroelements (mg/kg dw *) | |||||||
| Na | 58 ± 2 ab | 98 ± 4 | 72 ± 2 c | 81 ± 1 d | 62 ± 3 ae | 73 ± 7 cd | 62 ± 3 be |
| Mg | 3289 ± 85 a | 2044 ± 19 | 5500 ± 81 | 2835 ± 30 | 6671 ± 139 | 3358 ± 26 a | 3695 ± 32 |
| Al | 1862 ± 94 | 1612 ± 54 | 669 ± 15 ab | 693 ± 56 cd | 219 ± 15 | 667 ± 104 ac | 668 ± 74 bd |
| K | 14,448 ± 355 | 20,289 ± 294 | 28,373 ± 568 ab | 29,794 ± 340 | 27,528 ± 303 ac | 26,020 ± 259 | 27,800 ± 445 bc |
| Ca | 9763 ± 198 | 7783 ± 104 | 14,800 ± 219 | 12,254 ± 125 a | 13,115 ± 277 | 10,803 ± 96 | 12,072 ± 78 a |
| Fe | 1430 ± 62 a | 1322 ± 62 a | 498 ± 16 bcd | 616 ± 69 b | 187 ± 9 | 476 ± 78 ce | 443 ± 44 de |
| P | 1705 ± 31 | 2187 ± 34 | 2409 ± 52 | 2065 ± 22 | 2961 ± 45 | 2877 ± 25 a | 2822 ± 23 a |
| Microelements (µg/kg dw) | |||||||
| V | 3380 ± 96 | 3793 ± 130 | 1103 ± 17 ab | 1474 ± 131 | 383 ± 23 | 1132 ± 185 ac | 1058 ± 112 bc |
| Mn | 48 ± 2 a | 59 ± 1 b | 214 ± 3 | 57 ± 2 b | 185 ± 7 | 43 ± 1 a | 66 ± 1 |
| Cr | 3294 ± 104 | 2873 ± 77 | 2073 ± 16 a | 1980 ± 175 a | 368 ± 16 | 1547 ± 88 | 1118 ± 140 |
| Co | 521 ± 18 | 588 ± 4 | 267 ± 5 | 314 ± 29 | 129 ± 9 | 165 ± 17 a | 177 ± 13 a |
| Ni | 2029 ± 67 a | 1635 ± 17 bc | 2008 ± 9 a | 1668 ± 109 b | 1549 ± 54 c | 3284 ± 162 | 2651 ± 68 |
| Cu | 8 ± 0 | 14 ± 0 | 9 ± 0 a | 8.74 ± 0 a | 9.90 ± 0 b | 11 ± 1 | 9.86 ± 0 b |
| Zn | 26 ± 0 | 29 ± 0 a | 53 ± 1 | 30 ± 1 a | 63 ± 1 | 55 ± 1 | 44 ± 0 |
| As | 369 ± 6 | 460 ± 5 | 209 ± 3 a | 192 ± 55 ab | 62 ± 3 | 147 ± 22 bc | 126 ± 13 c |
| Se | 19 ± 1 | 24 ± 1 abc | 25 ± 3 ade | 14 ± 1 f | 13 ± 1 f | 26 ± 2 bdg | 25 ± 1 ceg |
| Mo | 1228 ± 21 a | 1355 ± 23 | 337 ± 2 | 1231 ± 16 a | 831 ± 10 | 637 ± 50 | 481 ± 0 |
| Cd | 14 ± 0 abc | 19 ± 1 def | 88 ± 5 | 11 ± 1 a | 19 ± 1 bdg | 18 ± 1 ceg | 23 ± 1 f |
| Sn | 106 ± 6 a | 114 ± 3 a | 42 ± 1 bcd | 50 ± 0 bef | 42 ± 1 ceg | 75 ± 16 | 44 ± 5 dfg |
| Sb | 38 ± 1 | 53 ± 1 | 19 ± 1 ab | 23 ± 2 | 19 ± 1 ac | 27 ± 2 | 19 ± 1 bc |
| Hg | 18 ± 2 ab | 30 ± 1 | 18 ± 1 ac | 12 ± 0 de | 13 ± 1 d | 16 ± 1 bcf | 14 ± 0 ef |
| TI | 24 ± 0 | 18 ± 0 | 15 ± 0 | 6 ± 0 ab | 5 ± 0 a | 8 ± 1 c | 7 ± 1 bc |
| Pb | 957 ± 3 | 1323 ± 17 | 418 ± 5 a | 374 ± 52 bc | 261 ± 8 d | 471 ± 71 ab | 327 ± 38 cd |
| Extraction Technique | Sample | Total Phenolic Content (mg GAE/g dw *) | Antioxidant Capacity (mmol Trolox/g dw) | Phenolic Acids (mg/g dw) | Flavonoids (mg/g dw) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| DPPH | ABTS | Chlorogenic Acid | Crypto-Chlorogenic Acid | Caffeic Acid | Rosmarinic Acid | Rutin | Apigenin 7-(6″ Malonyl Glycoside) 1 | Luteolin-7-O-Rutinoside 2 | |||
| HAE | G1 | 50.78 ± 0.67 a | 0.232 ± 0.014 | 0.239 ± 0.013 | 5.53 ± 0.03 | 1.51 ± 0.01 | 0.48 ± 0.02 f | 0.96 ± 0.06 | 4.69 ± 0.02 | 0.31 ± 0.00 | 0.54 ± 0.00 |
| MAE | 51.63 ± 0.66 a | 0.254 ± 0.005 | 0.257 ± 0.027 | 6.10 ± 0.01 | 1.86 ± 0.01 | 0.47 ± 0.01 f | 1.33 ± 0.02 | 5.61 ± 0.03 | 0.26 ± 0.00 | 0.61 ± 0.01 | |
| SWE | 72.62 ± 0.09 | 0.304 ± 0.003 | 0.356 ± 0.007 | 1.73 ± 0.00 | 1.39 ± 0.01 | 0.10 ± 0.00 | 0.17 ± 0.03 | 1.01 ± 0.00 | nd | 0.07 ± 0.00 | |
| HAE | G2 | 56.20 ± 2.75 b | 0.266 ± 0.010 | 0.322 ± 0.003 | 6.42 ± 0.03 | 1.71 ± 0.01 | 0.73 ± 0.00 | 0.75 ± 0.00 | 6.89 ± 0.15 | 0.88 ± 0.00 | 0.42 ± 0.00 |
| MAE | 56.10 ± 0.40 b | 0.324 ± 0.007 d | 0.336 ± 0.008 | 7.15 ± 0.06 | 2.45 ± 0.01 | 0.65 ± 0.01 | 3.00 ± 0.01 | 8.83 ± 0.09 | 0.90 ± 0.00 | 0.51 ± 0.00 | |
| SWE | 66.82 ± 1.66 | 0.329 ± 0.004 d | 0.399 ± 0.004 | 2.20 ± 0.00 | 1.82 ± 0.00 | 0.13 ± 0.00 | 0.17 ± 0.00 | 1.09 ± 0.21 | nd | 0.05 ± 0.00 | |
| HAE | G3 | 40.60 ± 0.93 c | 0.209 ± 0.006 | 0.209 ± 0.005 | 1.13 ± 0.02 e | 0.55 ± 0.01 | 0.38 ± 0.00 | 4.80 ± 0.02 | 1.53 ± 0.01 | nd | nd |
| MAE | 42.88 ± 1.41 c | 0.243 ± 0.007 | 0.263 ± 0.004 | 1.10 ± 0.02 e | 0.59 ± 0.01 | 0.45 ± 0.00 | 5.56 ± 0.04 | 1.64 ± 0.03 | nd | nd | |
| SWE | 60.09 ± 2.55 | 0.280 ± 0.004 | 0.349 ± 0.017 | 0.47 ± 0.00 | 0.26 ± 0.00 | 0.14 ± 0.00 | 0.40 ± 0.00 | 0.40 ± 0.00 | nd | nd | |
| HAE | G4 | 87.87 ± 2.58 | 0.504 ± 0.012 | 0.609 ± 0.020 | 1.65 ± 0.05 | 0.83 ± 0.04 | 0.84 ± 0.01 | 11.03 ± 0.15 | 3.89 ± 0.02 | 0.27 ± 0.00 | 0.35 ± 0.01 |
| MAE | 94.10 ± 4.37 | 0.554 ± 0.012 | 0.589 ± 0.013 | 1.78 ± 0.01 | 0.96 ± 0.01 | 0.60 ± 0.00 | 18.69 ± 0.14 | 4.38 ± 0.03 | 0.26 ± 0.00 | 0.40 ± 0.00 | |
| SWE | 101.72 ± 0.79 | 0.584 ± 0.006 | 0.650 ± 0.016 | 0.99 ± 0.02 | 0.73 ± 0.01 | 0.24 ± 0.00 | 1.07 ± 0.00 | 1.01 ± 0.12 | nd | 0.03 ± 0.00 | |
| HAE | G5 | 44.79 ± 2.00 | 0.246 ± 0.011 | 0.276 ± 0.007 | 1.76 ± 0.06 | 0.77 ± 0.03 | 0.47 ± 0.02 g | 0.94 ± 0.33 | 0.58 ± 0.01 | 0.79 ± 0.02 | 0.43 ± 0.01 |
| MAE | 54.31 ± 0.95 | 0.284 ± 0.001 | 0.317 ± 0.006 | 2.31 ± 0.03 | 1.19 ± 0.02 | 0.47 ± 0.02 g | 4.14 ± 0.08 | 1.03 ± 0.02 | 0.81 ± 0.00 | 0.68 ± 0.00 | |
| SWE | 69.00 ± 0.79 | 0.334 ± 0.001 | 0.403 ± 0.017 | 1.17 ± 0.00 | 0.88 ± 0.00 | 0.42 ± 0.00 | 0.24 ± 0.00 | 0.26 ± 0.00 | nd | 0.13 ± 0.00 | |
| HAE | G6 | 44.86 ± 0.82 | 0.208 ± 0.006 | 0.248 ± 0.006 | 3.14 ± 0.11 | 0.87 ± 0.05 | 0.62 ± 0.03 | 0.40 ± 0.03 | 1.88 ± 0.00 | 0.35 ± 0.01 | 0.35 ± 0.00 |
| MAE | 55.72 ± 1.35 | 0.273 ± 0.002 | 0.319 ± 0.011 | 4.98 ± 0.01 | 1.80 ± 0.01 | 0.71 ± 0.00 | 3.54 ± 0.01 | 3.95 ± 0.02 | 0.40 ± 0.00 | 0.39 ± 0.00 | |
| SWE | 65.63 ± 0.53 | 0.286 ± 0.001 | 0.388 ± 0.012 | 1.52 ± 0.00 | 1.38 ± 0.01 | 0.11 ± 0.00 | 0.21 ± 0.00 | 0.82 ± 0.00 | nd | 0.08 ± 0.00 | |
| HAE | G7 | 44.97 ± 1.18 | 0.206 ± 0.001 | 0.256 ± 0.011 | 3.12 ± 0.02 | 1.21 ± 0.01 | 0.72 ± 0.01 | 1.27 ± 0.00 | 3.29 ± 0.02 | 0.08 ± 0.00 | 0.34 ± 0.00 |
| MAE | 49.95 ± 1.85 | 0.225 ± 0.003 | 0.292 ± 0.005 | 3.33 ± 0.07 | 1.41 ± 0.00 | 0.76 ± 0.01 | 2.37 ± 0.06 | 3.87 ± 0.05 | 0.11 ± 0.00 | 0.39 ± 0.00 | |
| SWE | 67.87 ± 0.17 | 0.289 ± 0.006 | 0.387 ± 0.000 | 1.14 ± 0.00 | 1.13 ± 0.00 | 0.13 ± 0.00 | 0.12 ± 0.00 | 1.07 ± 0.00 | nd | 0.09 ± 0.00 | |
| Output | Network Structure | Training Perf. Training Error | Test Perf. Test Error | Validation Perf. Validation Error | Hidden Activation Function | Output Activation Function |
|---|---|---|---|---|---|---|
| Total phenolic content and antioxidant capacity | MLP 5-6-3 | 0.9475 | 0.8858 | 0.8780 | Tanh | Exponential |
| 0.0113 | 0.0144 | 0.0357 | ||||
| MLP 5-8-3 | 0.8801 | 0.8787 | 0.8726 | Exponential | Exponential | |
| 0.0341 | 0.0416 | 0.0497 | ||||
| MLP 5-8-3 | 0.8862 | 0.8895 | 0.8883 | Exponential | Tanh | |
| 0.0252 | 0.0216 | 0.0332 | ||||
| MLP 5-11-3 | 0.9567 | 0.9400 | 0.9153 | Logistic | Exponential | |
| 0.0093 | 0.0076 | 0.0256 | ||||
| MLP 5-11-3 | 0.9220 | 0.9231 | 0.7813 | Exponential | Identity | |
| 0.0162 | 0.0199 | 0.0582 | ||||
| Content of individual selected phenolic compounds | MLP 5-8-5 | 0.9433 | 0.8909 | 0.8884 | Logistic | Logistic |
| 0.0189 | 0.0379 | 0.05155 | ||||
| MLP 5-9-5 | 0.9309 | 0.8899 | 0.8604 | Exponential | Identity | |
| 0.0223 | 0.0444 | 0.0562 | ||||
| MLP 5-10-5 | 0.9439 | 0.8851 | 0.8625 | Logistic | Exponential | |
| 0.0178 | 0.0380 | 0.0585 | ||||
| MLP 5-10-5 | 0.9341 | 0.8704 | 0.8502 | Logistic | Exponential | |
| 0.0217 | 0.0411 | 0.0661 | ||||
| MLP 5-11-5 | 0.9497 | 0.8688 | 0.8779 | Logistic | Exponential | |
| 0.0164 | 0.0393 | 0.0537 |
| Prediction | ANN Output | R2 | ||
|---|---|---|---|---|
| Training | Test | Validation | ||
| Total phenolic content and antioxidant capacity | TPC | 0.9207 | 0.9079 | 0.8928 |
| ABTS | 0.8830 | 0.8864 | 0.8745 | |
| DPPH | 0.8974 | 0.8677 | 0.8315 | |
| Content of individual selected phenolic compounds | Chlorogenic acid | 0.9545 | 0.9462 | 0.8826 |
| Cryptochlorogenic acid | 0.9283 | 0.8982 | 0.8006 | |
| Caffeic acid | 0.9685 | 0.9613 | 0.8999 | |
| Rosmarinic acid | 0.9407 | 0.9300 | 0.8139 | |
| Rutin | 0.9442 | 0.9386 | 0.9049 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Šeremet, D.; Jokić, S.; Aladić, K.; Butorac, A.; Lovrić, M.; Tušek, A.J.; Obranović, M.; Mandura Jarić, A.; Vojvodić Cebin, A.; Carović-Stanko, K.; et al. Comprehensive Study of Traditional Plant Ground Ivy (Glechoma hederacea L.) Grown in Croatia in Terms of Nutritional and Bioactive Composition. Foods 2022, 11, 658. https://doi.org/10.3390/foods11050658
Šeremet D, Jokić S, Aladić K, Butorac A, Lovrić M, Tušek AJ, Obranović M, Mandura Jarić A, Vojvodić Cebin A, Carović-Stanko K, et al. Comprehensive Study of Traditional Plant Ground Ivy (Glechoma hederacea L.) Grown in Croatia in Terms of Nutritional and Bioactive Composition. Foods. 2022; 11(5):658. https://doi.org/10.3390/foods11050658
Chicago/Turabian StyleŠeremet, Danijela, Stela Jokić, Krunoslav Aladić, Ana Butorac, Marija Lovrić, Ana Jurinjak Tušek, Marko Obranović, Ana Mandura Jarić, Aleksandra Vojvodić Cebin, Klaudija Carović-Stanko, and et al. 2022. "Comprehensive Study of Traditional Plant Ground Ivy (Glechoma hederacea L.) Grown in Croatia in Terms of Nutritional and Bioactive Composition" Foods 11, no. 5: 658. https://doi.org/10.3390/foods11050658
APA StyleŠeremet, D., Jokić, S., Aladić, K., Butorac, A., Lovrić, M., Tušek, A. J., Obranović, M., Mandura Jarić, A., Vojvodić Cebin, A., Carović-Stanko, K., & Komes, D. (2022). Comprehensive Study of Traditional Plant Ground Ivy (Glechoma hederacea L.) Grown in Croatia in Terms of Nutritional and Bioactive Composition. Foods, 11(5), 658. https://doi.org/10.3390/foods11050658











