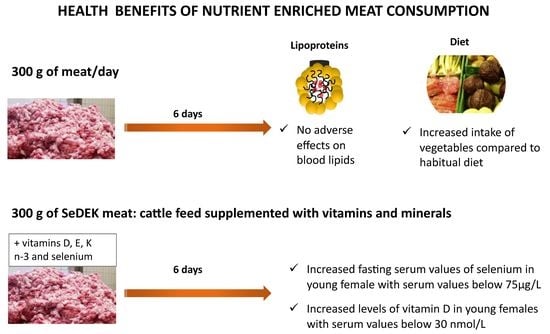
Nutrient-Optimized Beef Enhances Blood Levels of Vitamin D and Selenium among Young Women
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Study Design
2.3. Anthropometric Measurements
2.4. Blood Analyses
2.4.1. Serum Lipids and Other Blood Markers
2.4.2. Vitamin D3 and 25(OH)D3 Analyzed in Whole Blood
2.4.3. 25(OH)D3 in Whole Blood Compared to Serum, a Small Pilot Study
2.4.4. Determination of Blood Glucose and Hemoglobin
2.5. Diet Registration
2.6. Beef Production
2.7. Nutrient Intake Calculations from Food Composition Databases and Our Own Analyses
2.8. Estimation of Satiety in Habitual vs. Diets Holding 300 g Beef per Day
2.9. Statistical Analyses
3. Results
3.1. Baseline Characteristics
- Iodine; all the 34 persons had a lower intake than the recommended 150 µg per day, the average intake was 69 µg/day; only 46% of the recommended intake. The lower intake level is 70 µg per day [17], and 23 participants were below this level;
- Vitamin D; 29 of the 34. The lower intake level is 2.5 µg per day [17], and 9 of the participants were below this level;
- Iron; 28 of the 34 participants had a lower intake than the recommended 15 mg per day [17], and the average intake was 10.7 mg per day. The lower intake level was 9 mg per day, and 13 of the participants were below this level;
- Selenium: 17 of the 34 participants had a lower intake than the recommended 50 µg per day, and the average intake was 52 µg per day. The lower intake level is 9 mg per day [17], and 13 of the participants were below this level.
3.2. Nutrient Intakes Were Calculated from Databases and Own Analyses
3.3. Comparison of the ‘300 g Beef per Day’ Diet to the Habitual Diet
3.3.1. Calculated Nutrient Intake of the Two Diets
3.3.2. Measured Changes in Physical and Blood Variables from Habitual Diets to a Diet including 300 g Beef per Day
3.4. Consuming SeDEK Beef Compared to REGULAR Beef, Calculated and Measured Data
3.4.1. Calculated Nutrient Intake during the Intervention Consuming SeDEK Beef Compared to REGULAR Beef
3.4.2. Measured Study Variables following Consuming SeDEK Beef per Day Compared to REGULAR Beef
- Body weight, BMI, Blood Pressure, and Pulse
- Serum lipids and Other Blood Markers
- Inflammatory Markers
4. Discussion
4.1. The Nature of the Intervention (High Meat and Short Time)
4.1.1. Compliance
4.1.2. The Length of the Intervention Study
4.2. Health Outcomes
4.2.1. Serum Lipids
4.2.2. Blood Glucose
4.2.3. Micronutrients
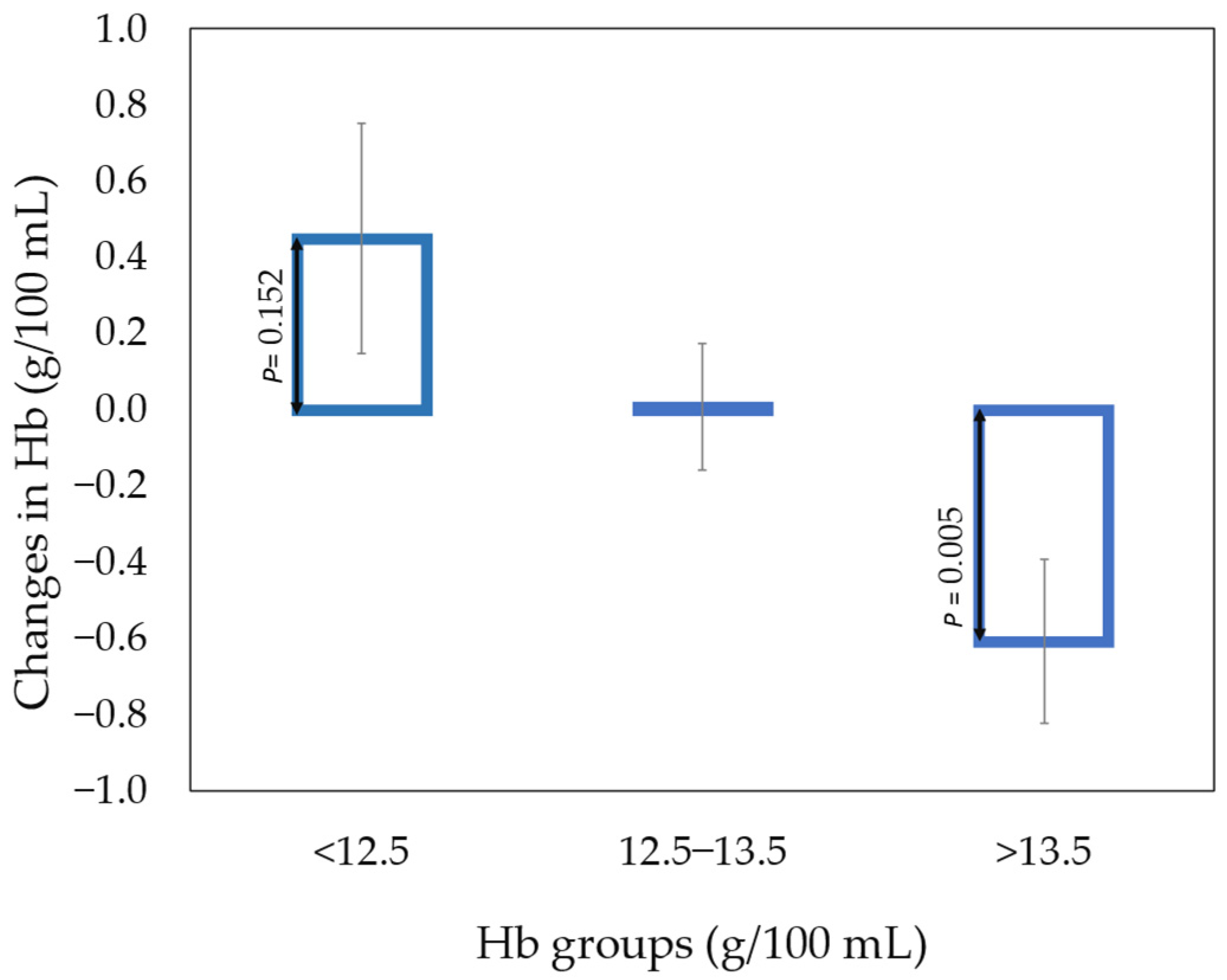
- Blood Hb changes with increased meat intake
- Iodine challenges with increased meat intake
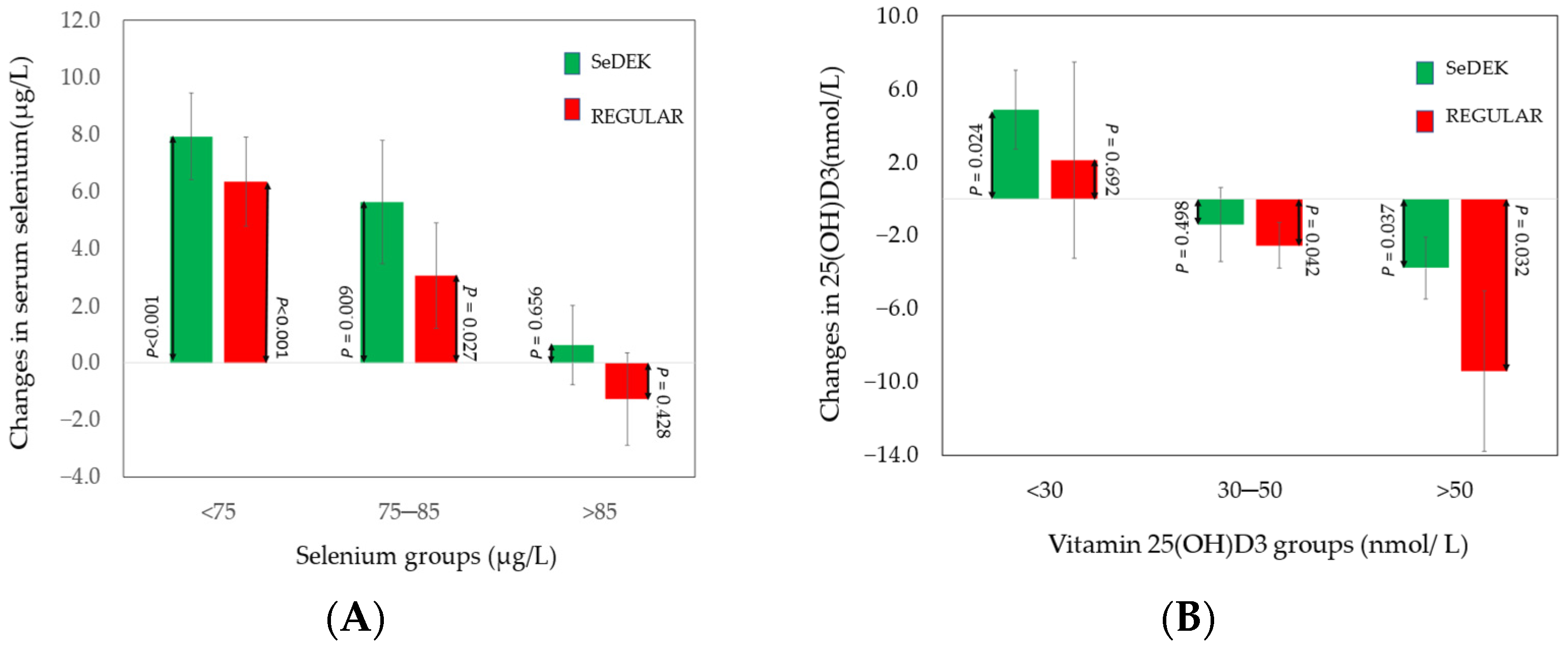
- Selenium
- Vitamin D
- Vitamin K
4.2.4. Inflammation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Biesalski, H.K. Meat as a component of a healthy diet-are there any risks or benefits if meat is avoided in the diet? Meat Sci. 2005, 70, 509–524. [Google Scholar] [CrossRef] [PubMed]
- Bellavia, A.; Stilling, F.; Wolk, A. High red meat intake and all-cause cardiovascular and cancer mortality: Is the risk modified by fruit and vegetable intake? Am. J. Clin. Nutr. 2016, 104, 1137–1143. [Google Scholar] [CrossRef] [PubMed]
- Wurtz, A.M.; Hansen, M.D.; Tjonneland, A.; Rimm, E.B.; Schmidt, E.B.; Overvad, K.; Jakobsen, M.U. Substitution of meat and fish with vegetables or potatoes and risk of myocardial infarction. Br. J. Nutr. 2016, 116, 1602–1610. [Google Scholar] [CrossRef] [PubMed]
- InterAct, C. Adherence to predefined dietary patterns and incident type 2 diabetes in European populations: EPIC-InterAct Study. Diabetologia 2014, 57, 321–333. [Google Scholar]
- Pan, A.; Sun, Q.; Bernstein, A.M.; Manson, J.E.; Willett, W.C.; Hu, F.B. Changes in red meat consumption and subsequent risk of type 2 diabetes mellitus: Three cohorts of US men and women. JAMA Intern. Med. 2013, 173, 1328–1335. [Google Scholar] [CrossRef]
- Zelber-Sagi, S.; Ivancovsky-Wajcman, D.; Fliss Isakov, N.; Webb, M.; Orenstein, D.; Shibolet, O.; Kariv, R. High red and processed meat consumption is associated with non-alcoholic fatty liver disease and insulin resistance. J. Hepatol. 2018, 68, 1239–1246. [Google Scholar] [CrossRef]
- Al-Shaar, L.; Satija, A.; Wang, D.D.; Rimm, E.B.; Smith-Warner, S.A.; Stampfer, M.J.; Hu, F.B.; Willett, W.C. Red meat intake and risk of coronary heart disease among US men: Prospective cohort study. BMJ 2020, 371, m4141. [Google Scholar] [CrossRef]
- Rayman, M.P.; Winther, K.H.; Pastor-Barriuso, R.; Cold, F.; Thvilum, M.; Stranges, S.; Guallar, E.; Cold, S. Effect of long-term selenium supplementation on mortality: Results from a multiple-dose, randomised controlled trial. Free Radic Biol. Med. 2018, 127, 46–54. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed. Pharmacother. 2002, 56, 365–379. [Google Scholar] [CrossRef]
- Cashman, K.D.; Dowling, K.G.; Skrabakova, Z.; Gonzalez-Gross, M.; Valtuena, J.; De Henauw, S.; Moreno, L.; Damsgaard, C.T.; Michaelsen, K.F.; Mølgaard, C.; et al. Vitamin D deficiency in Europe: Pandemic? Am. J. Clin. Nutr. 2016, 103, 1033–1044. [Google Scholar] [CrossRef]
- Haug, A.; Nyquist, N.F.; Mosti, T.J.; Andersen, M.; Hostmark, A.T. Increased EPA levels in serum phospholipids of humans after four weeks daily ingestion of one portion chicken fed linseed and rapeseed oil. Lipids Health Dis. 2012, 11, 104. [Google Scholar] [CrossRef]
- Christophersen, O.A.; Haug, A. Animal products, diseases and drugs: A plea for better integration between agricultural sciences, human nutrition and human pharmacology. Lipids Health Dis. 2011, 10, 16. [Google Scholar] [CrossRef] [PubMed]
- Comitato, R.; Ambra, R.; Virgili, F. Tocotrienols: A Family of Molecules with Specific Biological Activities. Antioxidants 2017, 6, 93. [Google Scholar] [CrossRef] [PubMed]
- Hosnedlova, B.; Kepinska, M.; Skalickova, S.; Fernandez, C.; Ruttkay-Nedecky, B.; Malevu, T.D.; Sochor, J.; Baron, M.; Melcova, M.; Zidkova, J.; et al. A Summary of New Findings on the Biological Effects of Selenium in Selected Animal Species-A Critical Review. Int. J. Mol. Sci. 2017, 18, 2209. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Scientific Opinion on the substantiation of health claims related to selenium and protection of DNA, proteins and lipids from oxidative damage (ID 277, 283, 286, 1289, 1290, 1291, 1293, 1751), function of the immune system (ID 278), thyroid function (ID 2). EFSA J. 2009, 7, 1220. [Google Scholar] [CrossRef]
- EFSA. Scientific Opinion on the substantiation of health claims related to vitamin K and maintenance of bone (ID 123, 127, 128, and 2879), blood coagulation (ID 124 and 126), and function of the heart and blood vessels (ID 124, 125 and 2880) pursuant to Article. EFSA J. 2009, 7, 2714. [Google Scholar]
- Nordic Council of Ministers. Nordic Nutrition Recommendations 2012: Integrating Nutrition and Physical Activity, 5th ed.; Nordic Council of Ministers: Copenhagen, Denmark, 2014. [Google Scholar] [CrossRef]
- Mortensen, C.; Damsgaard, C.T.; Hauger, H.; Ritz, C.; Lanham-New, S.A.; Smith, T.J.; Hennessy, A.; Dowling, K.; Cashman, K.D.; Kiely, M.; et al. Estimation of the dietary requirement for vitamin D in white children aged 4–8 y: A randomized, controlled, dose-response trial. Am. J. Clin. Nutr. 2016, 104, 1310–1317. [Google Scholar] [CrossRef]
- Aspell, N.; Lawlor, B.; O’Sullivan, M. Is there a role for vitamin D in supporting cognitive function as we age? Proc. Nutr. Soc. 2018, 77, 124–134. [Google Scholar] [CrossRef]
- Laird, E.; O’Halloran, A.M.; Carey, D.; Healy, M.; O’Connor, D.; Moore, P.; Shannon, T.; Molloy, A.M.; Kenny, R.A. The Prevalence of Vitamin D Deficiency and the Determinants of 25(OH)D Concentration in Older Irish Adults: Data From The Irish Longitudinal Study on Ageing (TILDA). J. Gerontol. A Biol. Sci. Med. Sci. 2018, 73, 519–525. [Google Scholar] [CrossRef]
- Nasjonalt Råd for Ernæring. Vitamin D i Norge: Behov for Tiltak for å Sikre Vitamin D Status? Report Series 11/18. 2018, pp. 1–44. Available online: https://www.helsedirektoratet.no/rapporter/vitamin-d-i-norge-behov-for-tiltak-for-a-sikre-god-vitamin-d-status (accessed on 15 December 2021).
- Berridge, M.J. Vitamin D deficiency accelerates ageing and age-related diseases: A novel hypothesis. J. Physiol. 2017, 595, 6825–6836. [Google Scholar] [CrossRef]
- Wei, R.; Christakos, S. Mechanisms Underlying the Regulation of Innate and Adaptive Immunity by Vitamin, D. Nutrients 2015, 7, 8251–8260. [Google Scholar] [CrossRef] [PubMed]
- Fayet, F.; Flood, V.; Petocz, P.; Samman, S. Avoidance of meat and poultry decreases intakes of omega-3 fatty acids, vitamin B12, selenium and zinc in young women. J. Hum. Nutr. Diet. 2014, 27 (Suppl. 2), 135–142. [Google Scholar] [CrossRef] [PubMed]
- Rimstad, S.H. Interesse for et Sunnere Storfekjøtt Blant Forbrukere i Norge. Master’s Thesis, Norwegian University of Life Sciences, Ås, Norway, 15 May 2016. Available online: https://brage.bibsys.no/xmlui/handle/11250/92927/browse?value=Rimstad%2C+Silje+Helene&type=author (accessed on 15 April 2021).
- Segovia-Siapco, G.; Burkholder-Cooley, N.; Haddad Tabrizi, S.; Sabate, J. Beyond Meat: A Comparison of the Dietary Intakes of Vegetarian and Non-vegetarian Adolescents. Front. Nutr. 2019, 6, 86. [Google Scholar] [CrossRef] [PubMed]
- Fallon, N.; Dillon, S.A. Low Intakes of Iodine and Selenium Amongst Vegan and Vegetarian Women Highlight a Potential Nutritional Vulnerability. Front. Nutr. 2020, 7, 72. [Google Scholar] [CrossRef]
- Reddy, M.B.; Hurrell, R.F.; Cook, J.D. Meat consumption in a varied diet marginally influences nonheme iron absorption in normal individuals. J. Nutr. 2006, 136, 576–581. [Google Scholar] [CrossRef]
- Samuelson, G.; Lonnerdal, B.; Kempe, B.; Elverby, J.E.; Bratteby, L.E. A follow-up study of serum ferritin and transferrin receptor concentrations in Swedish adolescents at age 17 age 15. Acta Paediatr. 2000, 89, 1162–1168. [Google Scholar] [CrossRef]
- Finley, J.W.; Grusak, M.A.; Keck, A.-S.; Gregoire, B.R. Bioavailability of selenium from meat and broccoli as determined by retention and distribution of 75Se. Biol. Trace Elem. Res. 2004, 99, 191–209. [Google Scholar] [CrossRef]
- Tetens, I.; Bendtsen, K.M.; Henriksen, M.; Ersboll, A.K.; Milman, N. The impact of a meat- versus a vegetable-based diet on iron status in women of childbearing age with small iron stores. Eur. J. Nutr. 2007, 46, 439–445. [Google Scholar] [CrossRef]
- Krebs, N.F.; Westcott, J.E.; Culbertson, D.L.; Sian, L.; Miller, L.V.; Hambidge, K.M. Comparison of complementary feeding strategies to meet zinc requirements of older breastfed infants. Am. J. Clin. Nutr. 2012, 96, 30–35. [Google Scholar] [CrossRef]
- Mrazova, J.; Gazarova, M.; Kopcekova, J.; Kolesarova, A.; Bucko, O.; Bobcek, B. The effect of consumption of pork enriched by organic selenium on selenium status and lipid profile in blood serum of consumers. J. Environ. Sci. Health Part B 2020, 55, 69–74. [Google Scholar] [CrossRef]
- Bjorn-Rasmussen, E.; Hallberg, L.; Isaksson, B.; Arvidsson, B. Food iron absorption in man. Applications of the two-pool extrinsic tag method to measure heme and nonheme iron absorption from the whole diet. J. Clin. Investg. 1974, 53, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Egelandsdal, B.; Oostindjer, M.; Hovland, E.M.; Okholm, B.; Saarem, K.; Bjerke, F.; Ruud, L.; Grabež, V.; Haug, A. Identifying labelling and marketing advantages of nutrients in minced beef meat: A case study. Meat Sci. 2020, 159, 107920. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D. The role of vitamers and dietary-based metabolites of vitamin D in prevention of vitamin D deficiency. Food Nutr. Res. 2012, 56, 5383. [Google Scholar] [CrossRef] [PubMed]
- Jakobsen, J. Bioavailability and bioactivity of vitamin D3 active compounds–Which potency should be used for 25-hydroxyvitamin D3? Int. Congr. Ser. 2007, 1297, 133–142. [Google Scholar] [CrossRef]
- National Food Institute TUoD. FRIDA, DTU Foods Public Food Database; DTU Fødevareinstitutet: Kongens Lyngby, Denmark, 2019; Available online: https://frida.fooddata.dk/pdf/frida-dokumentation-da_DK-august-2019.pdf (accessed on 15 April 2018).
- Haug, A.; Vhile, S.; Berg, J.; Hove, K.; Egelandsdal, B. Feeding potentially health promoting nutrients to finishing bulls changes meat composition and allow for product health claims. Meat Sci. 2018, 145, 461–468. [Google Scholar] [CrossRef]
- Norwegian Directorate of Health; Norwegian Food Safety Authority. A Diet Tool from the Norwegian Directorate of Health and the Norwegian Food Safety Authority; Norwegian Directorate of Health and the Norwegian Food Safety Authority: Oslo, Norway, 2021. Available online: https://www.kostholdsplanleggeren.no/ (accessed on 1 September 2019).
- Council of the European Communities. Council Directive 90/496/EEC of 24 September 1990 on Nutrition Labelling for Foodstuffs; EUR-Lex (European Union Law): Brussels, Belgium, 1990. Available online: https://eur-lex.europa.eu/legal-content/en/ALL/?uri=CELEX:31990L0496 (accessed on 2 February 2016).
- Brattbakk, H.R.; Arbo, I.; Aagaard, S.; Lindseth, I.; de Soysa, A.K.; Langaas, M.; Kulseng, B.; Lindberg, F.; Johansen, B. Balanced caloric macronutrient composition downregulates immunological gene expression in human blood cells-adipose tissue diverges. Omics A J. Integr. Biol. 2013, 17, 41–52. [Google Scholar] [CrossRef]
- Fürst Medisinsk Laboratorium. 2021. Available online: https://www.furst.no/analyse-og-klinikk/ (accessed on 1 November 2021).
- Schurgers, L.J.; Vermeer, C. Determination of phylloquinone and menaquinones in food. Effect of food matrix on circulating vitamin K concentrations. Haemostasis 2000, 30, 298–307. [Google Scholar]
- Authority NFS. The Norwegian Food Composition Database. 2021. Available online: https://matvaretabellen.no/?language=en (accessed on 3 January 2017).
- Agricultural Research Service US Department of Agriculture. Food Data Central (2017). 2019. Available online: https://fdc.nal.usda.gov/ (accessed on 17 April 2018).
- EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP); Bampidis, V.; Azimonti, G.; Bastos, M.D.L.; Christensen, H.; Dusemund, B.; Kouba, M.; Kos Durjava, M.; López-Alonso, M.; López Puente, S.; et al. Safety and efficacy of sodium selenate as feed additive for ruminants. EFSA J. 2019, 17, e05788. [Google Scholar]
- Wellek, S.; Blettner, M. On the proper use of the crossover design in clinical trials: Part 18 of a series on evaluation of scientific publications. Dtsch. Arztebl. Int. 2012, 109, 276–281. [Google Scholar]
- Liu, Z.; Li, J.; Chen, D.; Gao, R.; Zeng, W.; Chen, S.; Huang, Y.; Huang, J.; Long, W.; Li, M.; et al. Dynamic Interleukin-6 Level Changes as a Prognostic Indicator in Patients With COVID-19. Front. Pharmacol. 2020, 11, 1093. [Google Scholar] [CrossRef]
- Biovendor Research and Diagnostics. Quantitative Determination Ofhuman Interleukin-8. 2016. Available online: https://www.biovendor.com (accessed on 26 August 2021).
- Toh, J.; Gavrilova, T.; Rubinstein, A. Three Cases of Elevated IL-1beta in Common Variable Immunodeficiency (CVID) with Autoimmune Complications. J. Allergy Clin. Immunol. 2016, 137, AB19. [Google Scholar] [CrossRef]
- Zou, Y.; Chen, X.; Liu, J.; Zhou, D.B.; Kuang, X.; Xiao, J.; Yu, Q.; Lu, X.; Li, W.; Xie, B.; et al. Serum IL-1beta and IL-17 levels in patients with COPD: Associations with clinical parameters. Int. J. Chron. Obstruct. Pulmon. Dis. 2017, 12, 1247–1254. [Google Scholar] [CrossRef] [PubMed]
- Thane, C.W.; Bates, C.J.; Shearer, M.J.; Unadkat, N.; Harrington, D.J.; Paul, A.A.; Prentice, A.; Bolton-Smith, C. Plasma phylloquinone (vitamin K1) concentration and its relationship to intake in a national sample of British elderly people. Br. J. Nutr. 2002, 87, 615–622. [Google Scholar] [CrossRef]
- Thane, C.W.; Wang, L.Y.; Coward, W.A. Plasma phylloquinone (vitamin K1) concentration and its relationship to intake in British adults aged 19–64 years. Br. J. Nutr. 2006, 96, 1116–1124. [Google Scholar] [CrossRef] [PubMed]
- Schurgers, L.J.; Shearer, M.J.; Hamulyak, K.; Stocklin, E.; Vermeer, C. Effect of vitamin K intake on the stability of oral anticoagulant treatment: Dose-response relationships in healthy subjects. Blood 2004, 104, 2682–2689. [Google Scholar] [CrossRef][Green Version]
- Shearer, M.J.; Rahim, S.; Barkhan, P.; Stimmler, L. Plasma Vitamin-K1 in Mothers and Their Newborn Babies. Lancet 1982, 2, 460–463. [Google Scholar] [CrossRef]
- Thijssen, H.H.; Drittij, M.J.; Vermeer, C.; Schoffelen, E. Menaquinone-4 in breast milk is derived from dietary phylloquinone. Br. J. Nutr. 2002, 87, 219–226. [Google Scholar] [CrossRef]
- Knapen, M.H.; Braam, L.A.; Teunissen, K.J.; Van’t Hoofd, C.M.; Zwijsen, R.M.; van den Heuvel, E.G.H.M.; Vermeer, C. Steady-state vitamin K2 (menaquinone-7) plasma concentrations after intake of dairy products and soft gel capsules. Eur. J. Clin. Nutr. 2016, 70, 831–836. [Google Scholar] [CrossRef]
- Hodges, S.J.; Akesson, K.; Vergnaud, P.; Obrant, K.; Delmas, P.D. Circulating levels of vitamins K1 and K2 decreased in elderly women with hip fracture. J. Bone. Miner. Res. 1993, 8, 1241–1245. [Google Scholar] [CrossRef]
- EFSA. Dietary Reference Values: EFSA Publishes Advice on Vitamin K. Available online: https://www.efsa.europa.eu/en/press/news/170522-12017 (accessed on 22 May 2017).
- Institute of Medicine. Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc. In Nutrients; SoURLo, Intakes; SoIaUoDR, Intakes SCotSEoDR, Medicine; Institute of Medicine (US) Panel on Micronutrients; National Academies Press: Washington, DC, USA, 2001. [Google Scholar]
- Cashman, K.D.; Seamans, K.M.; Lucey, A.J.; Stocklin, E.; Weber, P.; Kiely, M.; Hill, T.R. Relative effectiveness of oral 25-hydroxyvitamin D3 and vitamin D3 in raising wintertime serum 25-hydroxyvitamin D in older adults. Am. J. Clin. Nutr. 2012, 95, 1350–1356. [Google Scholar] [CrossRef]
- Pettersson, J.; Hindorf, U.; Persson, P.; Bengtsson, T.; Malmqvist, U.; Werkstrom, V.; Ekelund, M. Muscular exercise can cause highly pathological liver function tests in healthy men. Br. J. Clin. Pharmacol. 2008, 65, 253–259. [Google Scholar] [CrossRef]
- Bragstad, K.; Paulsen, T.H.; Tønnessen, R.; Klüwer, B.; Rydland, K.M.; Aune, T.; Hungnes, O. Influensasesongen i Norge 2020-21; Norwegian Institute of Public Health: Oslo, Norway, 2021. Available online: https://www.fhi.no/globalassets/dokumenterfiler/influensa/influensaovervaking/influensasesongen-i-norge-2020-21 (accessed on 1 December 2021).
- An, R.; Andrade, F.; Grigsby-Toussaint, D. Sandwich consumption in relation to daily dietary intake and diet quality among US adults, 2003–2012. Public Health 2016, 140, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Mensink, R.P. Effects of Saturated Fatty Acids on Serum Lipids and Lipoproteins: A Systematic Review and Regression Analysis; World Health Organization: Geneva, Switzerland, 2016; Available online: https://apps.who.int/iris/handle/10665/246104 (accessed on 13 December 2021).
- Ke, Q.; Chen, C.; He, F.; Ye, Y.; Bai, X.; Cai, L.; Xia, M. Association between dietary protein intake and type 2 diabetes varies by dietary pattern. Diabetol. Metab. Syndr. 2018, 10, 48. [Google Scholar] [CrossRef] [PubMed]
- Gibson, S.; Ashwell, M. The association between red and processed meat consumption and iron intakes and status among British adults. Public Health Nutr. 2003, 6, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Alleyne, M.; Horne, M.K.; Miller, J.L. Individualized treatment for iron-deficiency anemia in adults. Am. J. Med. 2008, 121, 943–948. [Google Scholar] [CrossRef] [PubMed]
- Houghton, L.A.; Parnell, W.R.; Thomson, C.D.; Green, T.J.; Gibson, R.S. Serum Zinc Is a Major Predictor of Anemia and Mediates the Effect of Selenium on Hemoglobin in School-Aged Children in a Nationally Representative Survey in New Zealand. J. Nutr. 2016, 146, 1670–1676. [Google Scholar] [CrossRef]
- Maehre, H.K.; Malde, M.K.; Eilertsen, K.E.; Elvevoll, E.O. Characterization of protein, lipid and mineral contents in common Norwegian seaweeds and evaluation of their potential as food and feed. J. Sci. Food Agric. 2014, 94, 3281–3290. [Google Scholar] [CrossRef]
- Grabež, V.; Mydland, L.T.; Kidane, A.; Øverland, M.; Berg, P.; Fulladosa, E.; Øverland, M.; Mydland, M.T.; Brasas, E.C. Inclusion of Intact Saccharina Latissima to Finishing Lamb Diet Improved Micronutrient Content in the Meat; International Congress of Meat Science and Technology: Krakow, Poland, 2021; Available online: https://www.researchgate.net/publication/354544292_SubID_62152808826_Preferred_session_type_VOS_-virtual_oral_session_only_online_presentation (accessed on 13 December 2021).
- Rayman, M.P.; Blundell-Pound, G.; Pastor-Barriuso, R.; Guallar, E.; Steinbrenner, H.; Stranges, S. A randomized trial of selenium supplementation and risk of type-2 diabetes, as assessed by plasma adiponectin. PLoS ONE 2012, 7, e45269. [Google Scholar] [CrossRef]
- Aro, A.; Alfthan, G.; Varo, P. Effects of supplementation of fertilizers on human selenium status in Finland. Analyst 1995, 120, 841–843. [Google Scholar] [CrossRef]
- Hartikainen, H. Biogeochemistry of selenium and its impact on food chain quality and human health. J. Trace Elem. Med. Biol. 2005, 18, 309–318. [Google Scholar] [CrossRef]
- Schmid, A.; Walther, B. Natural vitamin D content in animal products. Adv. Nutr. 2013, 4, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Schurgers, L.J.; Vermeer, C. Differential lipoprotein transport pathways of K-vitamins in healthy subjects. Biochim. Biophys. Acta-Gen. Subj. 2002, 1570, 27–32. [Google Scholar] [CrossRef]
- Cui, G.; Yuan, A.; Sun, Z.; Zheng, W.; Pang, Z. IL-1beta/IL-6 network in the tumor microenvironment of human colorectal cancer. Pathol. Res. Pract. 2018, 214, 986–992. [Google Scholar] [CrossRef] [PubMed]

| In 100 g a | REGULAR | SeDEK |
|---|---|---|
| N = 6 Bulls | N = 6 Bulls | |
| Selenium, µg | 10.0 | 12.6 |
| Myoglobin b, g | 0.38 | 0.45 |
| Alpha-tocopherol, mg | 0.16 | 0.65 |
| K1, µg | 2.1 | 2.0 |
| MK4, µg | 9.1 | 20.3 |
| Vit D3, µg | <0.01 | 0.04 |
| 25(OH)D3, µg | 0.10 | 0.29 |
| Cholesterol, mg | 73.0 | 57.5 |
| Fat, g | 13.6 | 14.9 |
| C14:0, g | 0.36 | 0.39 |
| C16:0, g | 3.35 | 3.48 |
| C16:1 n-7, g | 0.37 | 0.42 |
| C18:0, g | 2.59 | 2.82 |
| C18:1 n-9, g | 5.01 | 5.56 |
| C18:1 trans, g | 0.43 | 0.47 |
| C18:2 n-6, g | 0.27 | 0.25 |
| C18:3 n-3, g | 0.059 | 0.070 |
| C20:4 n-6, g | 0.032 | 0.029 |
| C20:5 n-3, g | 0.006 | 0.006 |
| C22:5 n-3, g | 0.017 | 0.019 |
| n-6/n-3 | 3.6 | 2.9 |
| N = 34 | Baseline | Min | Max | Ref Values |
|---|---|---|---|---|
| Mean ± SD | ||||
| Age, years | 21.4 ± 2.0 | 19 | 29 | |
| Height, cm | 170 ± 6 | 157 | 180 | |
| Body weight, kg | 66.1 ± 8.5 | 50 | 88 | |
| BMI, kg/m2 | 22.9 ± 2.7 | 18 | 29 | |
| Systolic BP | 113 ± 8 | 94 | 132 | 90–130 |
| Diastolic BP | 70 ± 7 | 54 | 84 | 60–85 |
| Heart rate | 74 ± 13 | 48 | 102 | 60–100 |
| Blood glucose, mmol/L, blood | 5.0 ± 0.5 | 3.9 | 6.6 | 3.8–6.1 |
| Insulin, pmol/L serum | 72.6 ± 31 | 24 | 150 | 18–173 |
| C-peptide, pmol/L serum | 476 ± 146 | 205 | 850 | 270–1290 |
| Selenium µg/L serum | 84.9 ± 15.1 | 65.4 | 151.8 | 47–142 |
| AST, U/L serum | 18.3 ± 4.5 | 10 | 27 | <35 |
| ALT, U/L serum | 14.7± 5.7 | 8 | 35 | <45 |
| IL-6, ng/L plasma | 3.6 ± 0.8 | 2.2 | 6.8 | <7 a |
| IL-8, ng/L plasma | 21.5 ± 1.2 | 19.4 | 23.6 | <16 b |
| IL-1β, ng/L plasma | 12.3 ± 2.6 | 9.9 | 19.6 | <12 c,d |
| Hb, g/100 mL blood | 13.3 ± 1.0 | 11.4 | 15.6 | 11.5–16 |
| Triglycerides, mmol/L serum | 0.93 ± 0.28 | 0.5 | 1.6 | <2.6 |
| LDL-cholesterol, mmol/L serum | 2.7 ± 0.81 | 0.9 | 4.8 | 1.2–4.3 |
| HDL-cholesterol, mmol/L serum | 1.6 ± 0.41 | 1.2 | 3.0 | 1.0–2.7 |
| Phylloquinone, µg/L plasma | 0.35 ± 0.22 | nd | 0.8 | 0.2–0.8 e,f,g,h |
| MK4, µg/L plasma | n.d | n.d. | n.d. | <0.3 i |
| MK7, µg/L plasma | 0.18 ± 0.14 | nd | 0.82 | <0.8 j,k |
| MK8, µg/L plasma | 0.13± 0.18 | nd | 0.48 | <0.45 k |
| 25(OH)D3, nmol/L serum | 54.6 l ± 19.6 | 11.8 | 91.7 | 50–150 |
| N = 34 | Habitual Diet | ‘300 g Beef’ | p | SeDEK-Beef | REGULAR Beef | p |
|---|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | |||
| Energy, kcal | 1848 ± 336 | 1971 ± 306 | 0.117 | 1894 ± 313 | 2049 ± 383 | 0.071 |
| Higher satiety, % of participants | 3 | 77 | <0.001 | - | - | - |
| Intake of beef, g | 12.7 ± 2.3 | 312.2 ± 8.4 | <0.001 | - | - | - |
| Intake of all meat, g | 84.8 ± 17.8 | 391.0 ± 28.8 | <0.001 | - | - | - |
| Intake of vegetables, g | 221 ± 145 | 314 ± 176 | 0.020 | - | - | - |
| Intake of cereals, g | 222 ± 80 | 186 ± 86 | 0.080 | - | - | - |
| Fat E% | 36.33 ± 6.01 | 42.73 ± 4.55 | <0.001 | 42.26 ± 5.2 | 43.53 ± 6.6 | 0.385 |
| Saturated fat E% | 12.53 ± 3.10 | 17.60 ± 2.69 | <0.001 | 17.65 ± 3.3 | 17.56 ± 3.7 | 0.918 |
| Trans unsaturated E% | 0.08 ± 0.18 | 0.45 ± 0.42 | <0.001 | 0.44 ± 0.56 | 0.46 ± 0.50 | 0.909 |
| Monounsaturated E% | 13.21 ± 3.20 | 15.71 ± 2.53 | <0.001 | 14.97 ± 3.2 | 16.44 ± 3.2 | 0.062 |
| Polyunsaturated fat E% | 6.40 ± 1.89 | 4.56 ± 1.53 | 0.007 | 4.24 ± 1.5 | 4.88 ± 1.7 | 0.107 |
| Carbohydrate (incl. fiber) E% | 45.68 ± 7.11 | 35.54 ± 5.13 | <0.001 | 36.15 ± 5.8 | 34.94 ± 6.7 | 0.429 |
| Added sugar E% | 5.65 ± 4.98 | 3.97 ± 3.34 | 0.105 | 4.03 ± 4.4 | 3.88 ± 3.7 | 0.881 |
| Protein E% | 18.12 ± 3.67 | 21.37 ± 2.30 | <0.001 | 21.53 ± 3.0 | 21.21 ± 2.3 | 0.620 |
| Fat, g | 73.7 ± 21.1 | 91.5 ± 15.8 | <0.001 | 90.0 ± 15.9 | 95.0 ± 21.1 | 0.128 |
| Saturated fat, g | 25.08 ± 8.41 | 37.05 ± 6.05 | <0.001 | 36.1 ± 6.5 | 38.0 ± 9.2 | 0.333 |
| Trans unsaturated fat, g | 0.51 ± 0.33 | 1.07 ± 0.25 | <0.001 | 1.01 ± 0.3 | 1.14 ± 0.4 | 0.095 |
| Monounsaturated fat, g | 26.85 ± 8.73 | 34.62 ± 6.27 | <0.001 | 31.91 ± 6.0 | 35.92 ± 9.2 | 0.036 |
| Polyunsaturated fat, g | 13.39 ± 5.71 | 10.07 ± 3.98 | 0.007 | 9.07 ± 3.8 | 11.10 ± 4.8 | 0.055 |
| n-3, g | 2.43 ± 1.99 | 1.76 ± 1.82 | 0.151 | 1.63 ± 1.9 | 1.94 ± 1.9 | 0.599 |
| n-6, g | 9.51 ± 3.35 | 7.46 ± 2.78 | 0.008 | 6.65 ± 2.4 | 8.28 ± 3.5 | 0.032 |
| n-6/n-3 | 5.86 ± 3.02 | 5.64 ± 1.80 | 0.722 | 5.29 ± 1.9 | 5.99 ± 2.7 | 0.220 |
| Cholesterol, mg | 281 ± 134 | 262 ± 97 | 0.514 | 249 ± 107 | 276 ± 123 | 0.341 |
| Carbohydrate (incl. fiber) g | 219 ± 47.8 | 185.3 ± 46.9 | 0.005 | 178 ± 51 | 193 ± 60 | 0.263 |
| Starch, g | 108.5 ± 29.7 | 95.3 ± 33.4 | 0.089 | 91.6 ± 35 | 99.0 ± 45 | 0.458 |
| Mono- and disaccharides, g | 76.2 ± 35.0 | 62.0 ± 24.3 | 0.057 | 60.8 ± 25 | 63.3 ± 31 | 0.714 |
| Sugar, added, g | 26.6 ± 25.1 | 18.1 ± 15.3 | 0.094 | 16.63 ± 16 | 19.49 ± 17 | 0.498 |
| Fiber, g | 26.7 ± 10.8 | 23.1 ± 9.8 | 0.149 | 22.33±10 | 23.83 ± 11 | 0.560 |
| Protein, g | 81.4 ± 21.0 | 104.9 ± 14.9 | <0.001 | 102 ± 18 | 108 ± 16 | 0.214 |
| Salt, g | 6.07 ± 2.11 | 6.18 ± 2.47 | 0.843 | 6.66 ± 3.2 | 5.69 ± 2.7 | 0.175 |
| Vitamin A, (RAE) | 1227 ± 1327 | 634 ± 349 | 0.014 | 593 ± 436 | 675 ± 491 | 0.469 |
| Retinol, µg | 648 ± 1007 | 250 ± 210 | 0.028 | 217 ± 205 | 283 ± 383 | 0.379 |
| Beta-carotene, µg | 4217 ± 3562 | 3885 ± 3589 | 0.704 | 3858 ± 4350 | 3913 ± 4252 | 0.957 |
| Vitamin D a, µg | 4.91 ± 3.26 | 5.20 ± 1.78 | 0.650 | 6.39 ± 1.7 | 4.01 ± 2.9 | <0.001 |
| Vitamin E, αTE | 13.16 ± 4.75 | 11.49±3.70 | 0.112 | 11.38 ± 3.3 | 11.61 ± 5.0 | 0.821 |
| Vitamin K (total) b µg | 100.71 ± 83.5 | 157.7 ± 88.0 | 0.008 | 155.6 ± 94 | 159.9 ± 119 | 0.869 |
| MK4, µg | 10.57 ± 7.05 | 54.15 ± 5.63 | <0.001 | 69.29 ± 2.8 | 39.00 ± 10.2 | <0.001 |
| Thiamin, mg | 1.53 ± 0.46 | 1.26 ± 0.49 | 0.022 | 1.29 ± 0.6 | 1.23 ± 0.5 | 0.648 |
| Riboflavin, mg | 1.50 ± 0.42 | 1.50 ± 0.34 | 0.923 | 1.45 ± 0.4 | 1.55 ± 0.4 | 0.288 |
| Niacin, mg | 18.28 ± 6.05 | 22.15 ± 3.91 | 0.003 | 21.38 ± 3.8 | 22.92 ± 4.7 | 0.141 |
| Vitamin B6, mg | 1.80 ± 0.57 | 1.72 ± 0.49 | 0.527 | 1.64 ± 0.5 | 1.79 ± 0.6 | 0.243 |
| Folate, µg | 309 ± 127 | 282 ± 107 | 0.336 | 261 ± 91 | 302 ± 144 | 0.173 |
| Vitamin B12, µg | 5.16 ± 2.22 | 7.10 ± 1.31 | <0.001 | 6.96 ± 1.5 | 7.24 ± 1.7 | 0.480 |
| Vitamin C, mg | 104.4 ± 67.1 | 119.5 ± 70.8 | 0.370 | 120.5 ± 95 | 118.6 ± 72 | 0.929 |
| Calcium, mg | 835 ± 328 | 737 ± 254 | 0.169 | 726 ± 357 | 748 ± 327 | 0.793 |
| Iron, mg | 10.77 ± 3.20 | 14.08 ± 2.90 | <0.001 | 13.88 ± 3.0 | 14.28 ± 3.5 | 0.616 |
| Sodium, mg | 2405 ± 871 | 2454 ± 1016 | 0.833 | 2644 ± 1297 | 2263 ± 1074 | 0.192 |
| Potassium, mg | 3210 ± 1071 | 3440 ± 979 | 0.358 | 3382 ± 913 | 3499 ± 1242 | 0.662 |
| Magnesium, mg | 337 ± 115 | 326 ± 103 | 0.693 | 313 ± 99 | 340 ± 123 | 0.316 |
| Zinc, mg | 9.76 ± 2.16 | 20.55 ± 2.08 | <0.001 | 20.11 ± 2.5 | 20.98 ± 2.6 | 0.162 |
| Selenium, µg | 49.12 ± 25.69 | 61.61 ± 28.54 | 0.023 | 66.82 ± 37.9 | 56.41 ± 12.8 | 0.134 |
| Copper, mg | 1.31 ± 0.53 | 1.30 ± 0.42 | 0.955 | 1.24 ± 0.4 | 1.36 ± 0.5 | 0.330 |
| Phosphorous, mg | 1509 ± 350 | 1573 ± 307 | 0.424 | 1527 ± 334 | 1620 ± 389 | 0.296 |
| Iodine, µg | 76.38 ± 34.81 | 55.72 ± 27.45 | 0.008 | 51.33 ± 29 | 60.10 ± 37 | 0.283 |
| Part I | Part II | Part III | |||||
|---|---|---|---|---|---|---|---|
| Variable (N = 34) | Habitual Diet | 300 g Beef | p a | SeDEK-Beef | REGULAR-Beef | p | Cross Over |
| Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | p b | |||
| Body weight, kg | 66.35 ± 8.58 | 66.64 ± 8.64 | 0.895 | 66.45 ± 8.71 | 66.47 ± 8.83 | 0.991 | 0.372 |
| 0.001 c ± 0.011 | 0.002 ± 0.010 | 0.681 | |||||
| BMI | 22.99 ± 2.75 | 23.04 ± 2.79 | 0.944 | 23.03 ± 2.80 | 23.04 ± 2.82 | 0.993 | 0.839 |
| 0.001 ± 0.010 | 0.002 ± 0.010 | 0.666 | |||||
| Systolic BP | 113.3 ± 7.16 | 115.8 ± 7.17 | 0.153 | 116.4 ± 9.02 | 115.4 ± 7.32 | 0.617 | 0.204 |
| 0.035 ** ± 0.081 | 0.018 ± 0.076 | 0.372 | |||||
| Diastolic PB | 70.4 ± 5.46 | 71.3 ± 5.88 | 0.544 | 71.3 ± 7.49 | 71.4 ± 5.95 | 0.957 | 0.175 |
| 0.011 ± 0.120 | 0.025 * ± 0.072 | 0.543 | |||||
| Pulse | 74.18 ± 9.87 | 75.29 ± 8.88 | 0.625 | 76.50 ± 11.46 | 74.35 ± 10.26 | 0.419 | 0.378 |
| 0.030 ± 0.162 | 0.025 ± 0.146 | 0.884 | |||||
| Blood glucose, mmol/L blood | 5.19 ± 0.52 | 5.39 ± 0.42 | 0.078 | 5.47 ± 0.58 | 5.31 ± 0.41 | 0.190 | 0.125 |
| 0.058 ** ± 0.127 | 0.039 ± 0.122 | 0.538 | |||||
| Insulin, pmol/L serum | 70.10 ± 25.17 | 74.02 ± 30.24 | 0.564 | 74.75 ± 31.65 | 75.44 ± 30.44 | 0.928 | 0.038 |
| 0.113 ± 0.435 | 0.274 ± 0.610 | 0.221 | |||||
| C-peptide, pmol/L serum | 459 ± 111 | 471 ± 138 | 0.687 | 492 ± 156 | 462 ± 124 | 0.386 | 0.413 |
| 0.078 ± 0.290 | 0.085 ± 0.318 | 0.921 | |||||
| AST, U/L serum | 19.13 ± 5.23 | 17.59 ± 4.86 | 0.212 | 17.62 ± 5.10 | 18.03 ± 5.79 | 0.758 | 0.007 |
| 0.047 ± 0.212 | 0.052 ± 0.207 | 0.914 | |||||
| ALT, U/L serum | 15.41 ± 6.64 | 15.50 ± 6.41 | 0.955 | 15.53 ± 6.45 | 15.91 ± 8.16 | 0.833 | 0.171 |
| 0.023 ± 0.191 | 0.040 ± 0.215 | 0.741 | |||||
| AST/ALT | 1.31 ± 0.26 | 1.22 ± 0.26 | 0.139 | 1.22 ± 0.34 | 1.21 ± 0.25 | 0.905 | 0.030 |
| −0.059 ± 0.195 | 0.076 ± 0.178 | 0.702 | |||||
| IL-1β, ng/L plasma | 11.3 ± 2.7 | 10.5 ± 3.0 | 0.085 | 10.7 ± 3.6 | 10.3 ± 2.2 | 0.511 | 0.514 |
| −0.065 ± 0.245 | 0.089 *** ± 0.141 | 0.638 | |||||
| IL-6, ng/L plasma | 3.6 ± 0.8 | 4.1 ± 1.3 | 0.038 | 4.2 ± 1.4 | 4.0 ± 1.2 | 0.556 | 0.455 |
| 0.119 * ± 0.296 | 0.051 ± 0.220 | 0.294 | |||||
| IL-8, ng/L plasma | 22.3 ± 2.0 | 23.2 ± 2.5 | 0.037 | 23.4 ± 2.8 | 23.0 ± 2.2 | 0.519 | 0.252 |
| 0.049 *** ± 0.074 | 0.027 * ± 0.071 | 0.221 | |||||
| IL-1β/IL-6 | 3.3 ± 1.1 | 2.7 ± 0.7 | 0.001 | 2.8 ± 0.8 | 2.6 ± 0.6 | 0.500 | 0.379 |
| −0.016 ± 0.301 | 0.014 ± 0.220 | 0.972 | |||||
| Hb, g/100 mL blood | 13.17 ± 0.90 | 13.23 ± 0.92 | 0.755 | 13.13 ± 1.14 | 13.32 ± 0.90 | 0.432 | 0.350 |
| −0.016 ± 0.064 | 0.030 * ± 0.078 | 0.010 | |||||
| Triacylglycerol, mmol/L serum | 0.89 ± 0.27 | 0.89 ± 0.29 | 0.945 | 0.89 ± 0.26 | 0.90 ± 0.34 | 0.850 | 0.616 |
| 0.033 ± 0.262 | 0.025 ± 0.220 | 0.892 | |||||
| LDL-cholesterol, mmol/L serum | 2.62 ± 0.78 | 2.74 ± 0.86 | 0.556 | 2.72 ± 0.85 | 2.81 ± 0.85 | 0.638 | 0.021 |
| 0.043 ± 0.169 | 0.069 ** ± 0.135 | 0.492 | |||||
| HDL-cholesterol, mmol/L serum | 1.62 ± 0.37 | 1.63 ± 0.38 | 0.950 | 1.65 ± 0.34 | 1.67 ± 0.41 | 0.864 | 0.227 |
| 0.015 ± 0.098 | 0.043 ** ± 0.072 | 0.192 | |||||
| Selenium, µg/L serum | 83.6 ± 12.8 | 87.0 ± 10.2 | 0.093 | 87.9 ± 9.0 | 86.1 ± 11.4 | 0.472 | 0.410 |
| 0.057 *** ± 0.085 | 0.032 * ± 0.079 | 0.216 | |||||
| Phylloquinone, µg/L plasma | 0.33 ± 0.22 | 0.39 ± 0.32 | 0.410 | 0.40 ± 0.46 | 0.40 ± 0.38 | 0.989 | 0.302 |
| 0.38 ± 1.15 | 0.36 * ± 0.97 | 0.938 | |||||
| MK7, µg/L plasma | 0.19 ± 0.12 | 0.17 ± 0.11 | 0.436 | 0.16 ± 0.11 | 0.18 ± 0.17 | 0.589 | 0.578 |
| −0.125 ± 0.402 | −0.024 ± 0.556 | 0.456 | |||||
| 25(OH)D3, nmol/L serum | 52.0 ± 18.1 | 49.2 ± 16.8 | 0.512 | 50.4 ± 17.2 | 48.4 ± 16.6 | 0.630 | 0.255 |
| −0.012 ± 0.108 | −0.064 ** ± 0.131 | 0.088 d | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haug, A.; Vermeer, C.; Ruud, L.; Monfort-Pires, M.; Grabež, V.; Egelandsdal, B. Nutrient-Optimized Beef Enhances Blood Levels of Vitamin D and Selenium among Young Women. Foods 2022, 11, 631. https://doi.org/10.3390/foods11050631
Haug A, Vermeer C, Ruud L, Monfort-Pires M, Grabež V, Egelandsdal B. Nutrient-Optimized Beef Enhances Blood Levels of Vitamin D and Selenium among Young Women. Foods. 2022; 11(5):631. https://doi.org/10.3390/foods11050631
Chicago/Turabian StyleHaug, Anna, Cees Vermeer, Lene Ruud, Milena Monfort-Pires, Vladana Grabež, and Bjørg Egelandsdal. 2022. "Nutrient-Optimized Beef Enhances Blood Levels of Vitamin D and Selenium among Young Women" Foods 11, no. 5: 631. https://doi.org/10.3390/foods11050631
APA StyleHaug, A., Vermeer, C., Ruud, L., Monfort-Pires, M., Grabež, V., & Egelandsdal, B. (2022). Nutrient-Optimized Beef Enhances Blood Levels of Vitamin D and Selenium among Young Women. Foods, 11(5), 631. https://doi.org/10.3390/foods11050631