Effect of Wheatgrass Juice on Nutritional Quality of Apple, Carrot, Beet, Orange and Lemon Juice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Simulation of In Vitro Digestion
2.3. Determination of Total Concentrations of Macro and Microelements
2.4. Total Phenols and Flavonoids Content
2.5. Free Radical Scavenging by DPPH
2.6. Ascorbic Acid Content
2.7. Statistical Analysis
3. Results and Discussion
3.1. Concentrations of Macro and Microelements in Fresh Fruit, Vegetable, and Wheatgrass Juice
3.1.1. Differences in Total and In Vitro Bioaccessible Concentrations of Macro- and Microelements in Different Fruit and Vegetable Juices
3.1.2. The Effect of WGJ Addition on Total and In Vitro Bioaccessible Concentrations of Macro- and Microelements in Different Fruit and Vegetable Juices
3.1.3. The Effect of Lemon Juice Addition on In Vitro Bioaccessibility of Macro and Microelements in Different Fruit and Vegetable Juices
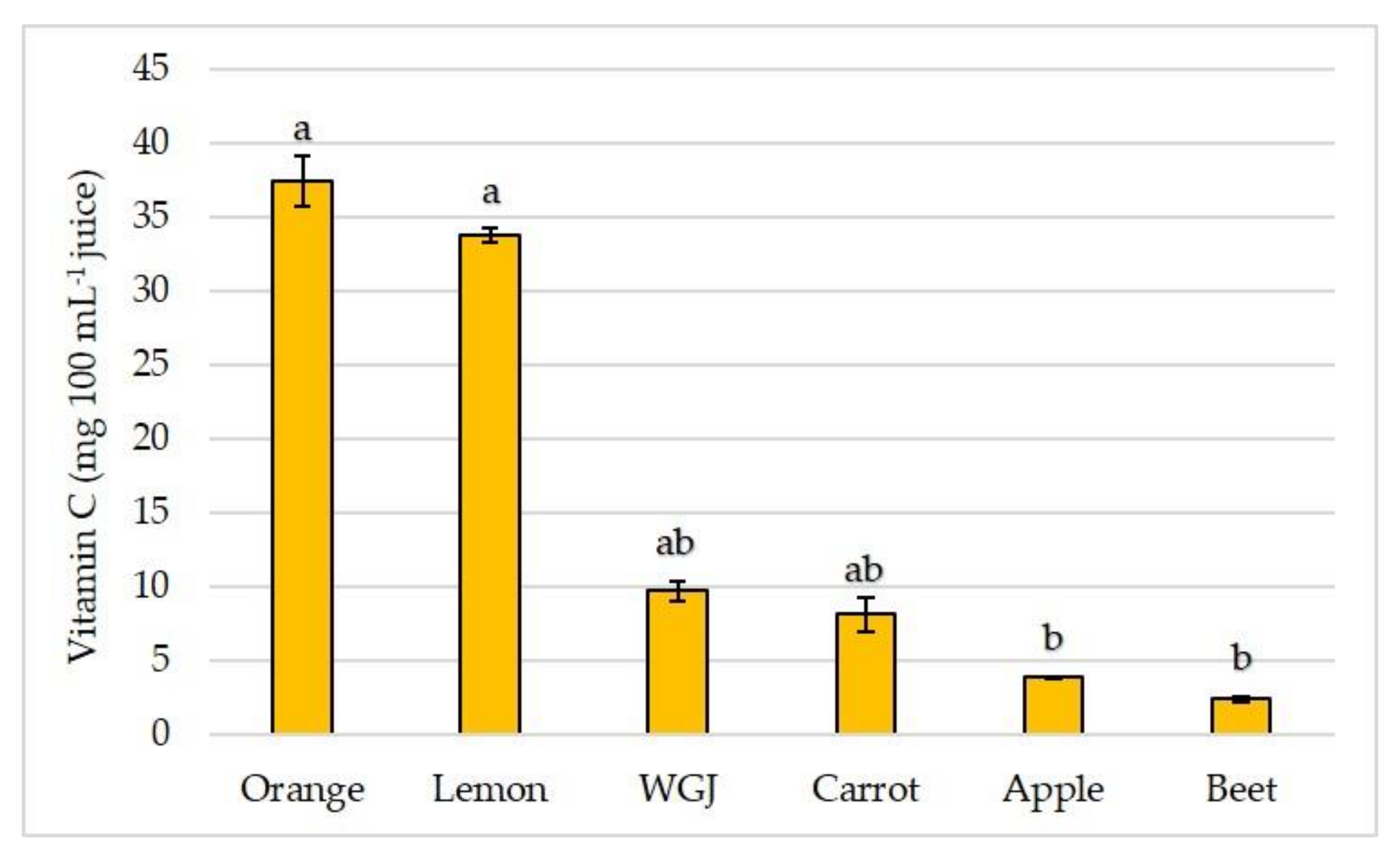
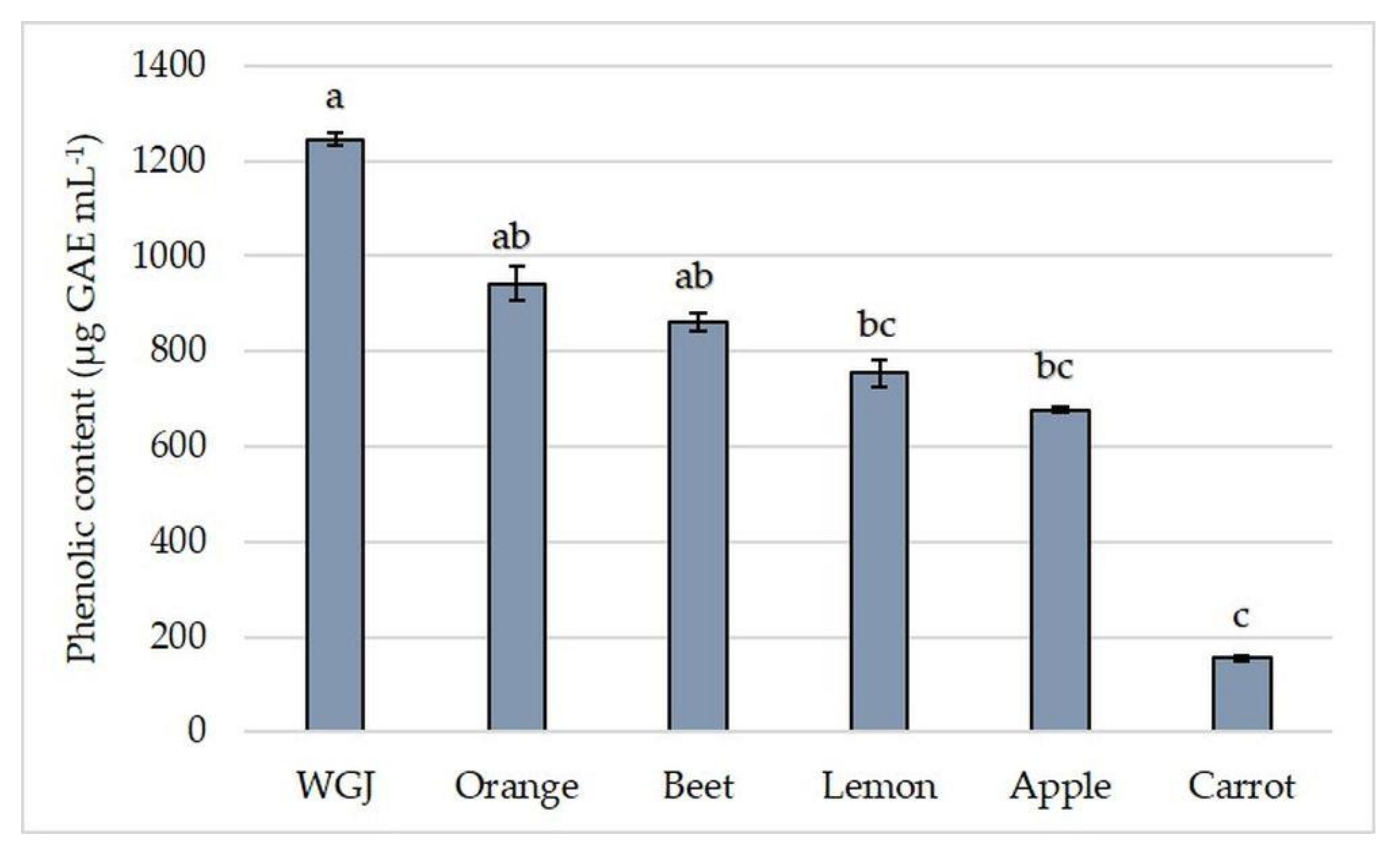
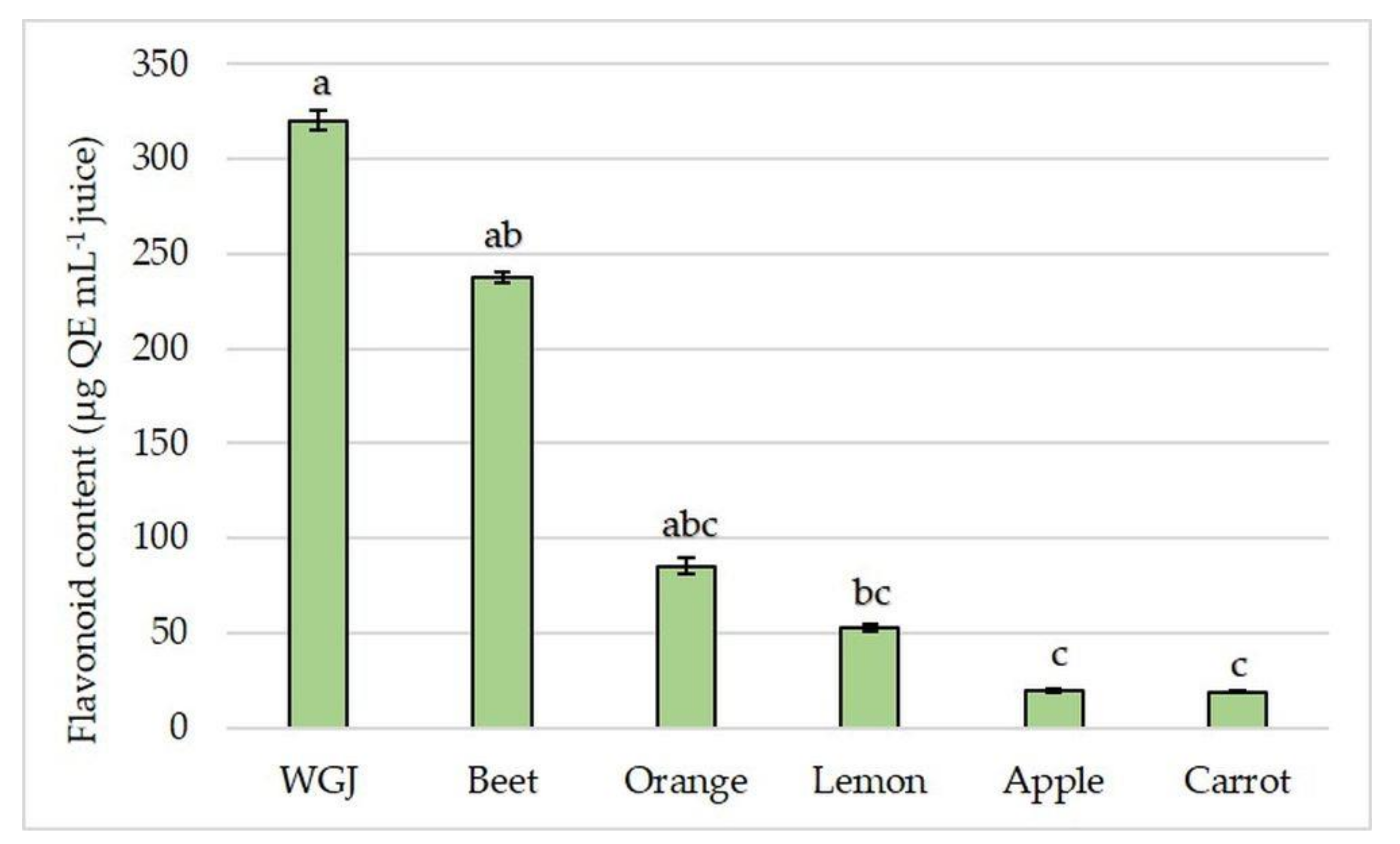
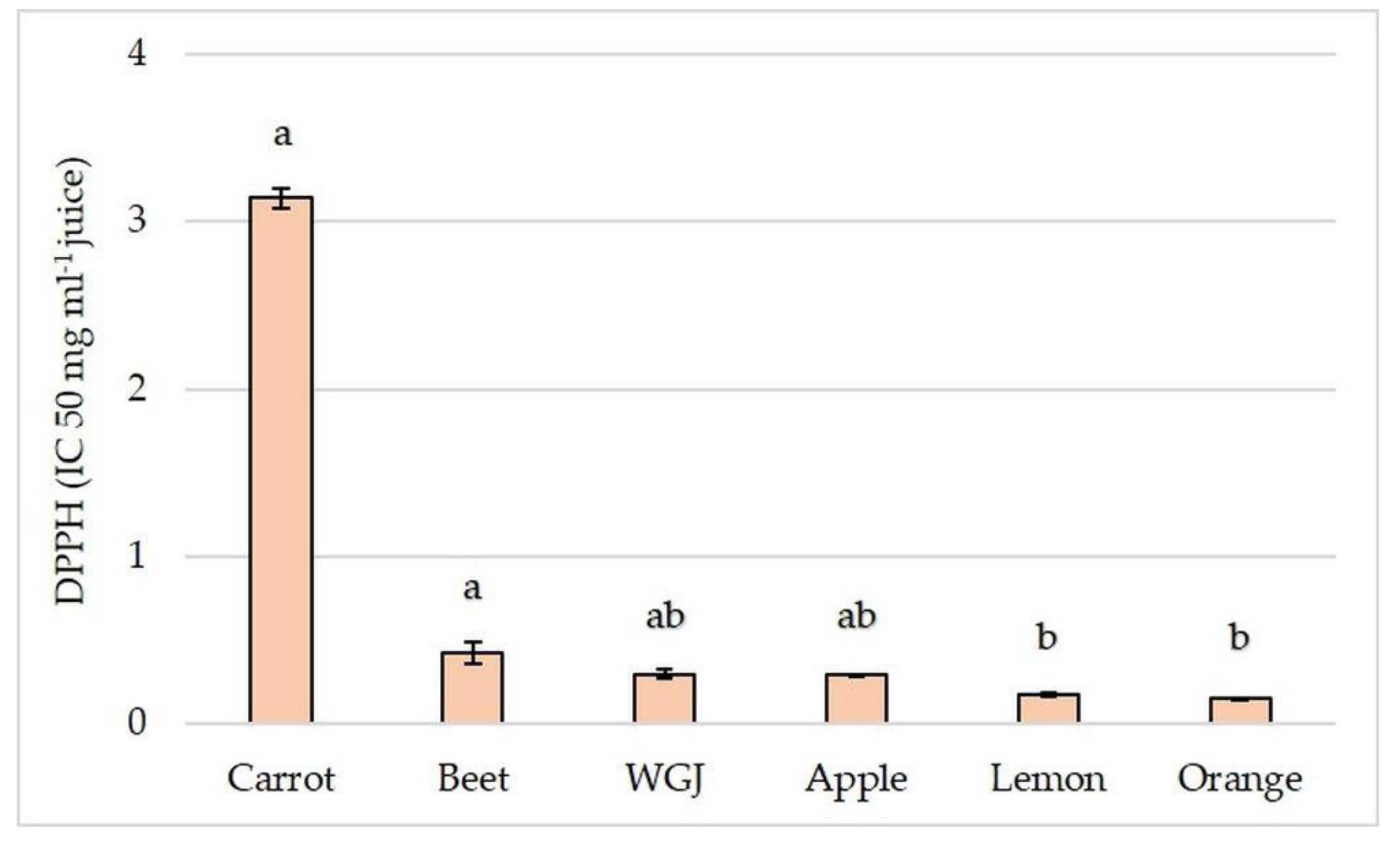
3.2. Total Phenolic and Flavonoid Content and Antioxidant Capacity in Fresh Fruit, Vegetable, and Wheatgrass Juice
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 1 December 2021).
- Padalia, S.; Drabu, S.; Raheja, I.; Gupta, A.; Dhamija, M. Multitude potential of wheatgrass juice (Green Blood): An overview. Chron. Young Sci. 2010, 1, 23–28. [Google Scholar]
- Mathur, S.; Mathur, R.; Kohli, G.K. Therapeutic use of wheat grass juice for the treatment of anemia in young women of Ajmer city (Rajasthan, India). Int. J. Nutr. Sci. 2017, 2, 1014. [Google Scholar]
- Singh, K.; Pannu, M.S.; Singh, P.; Singh, J. Effect of wheat grass tablets on the frequency of blood transfusions in thalassemia major. Indian J. Pediatr. 2010, 77, 90–91. [Google Scholar]
- Grubišić, S.; Orkić, V.; Guberac, S.; Lisjak, M.; Petrović, S.; Rebekić, A. Optimalan način sjetve pšenice (Triticum aestivum L.) za uzgoj pšenične trave. Poljoprivreda 2019, 25, 31–37. [Google Scholar] [CrossRef]
- Awulachew, M.T. The Role of Wheat in Human Nutrition and Its Medicinal Value. Glob. Acad. J. Med. Sci. 2020, 2, 50–54. [Google Scholar]
- Ben-Arye, E.; Goldin, E.; Wengrower, D.; Stamper, A.; Kohn, R.; Berry, E. Wheat grass juice in the treatment of active distal ulcerative colitis: A randomized double-blind placebo-controlled trial. Scand. J. Gastroenter. 2002, 37, 444–449. [Google Scholar] [CrossRef]
- Bar-Sela, G.; Tsalic, M.; Fried, G.; Goldberg, H. Wheat grass juice may improve hematological toxicity related to chemotherapy in breast cancer patients: A pilot study. Nutr. Cancer 2007, 58, 43–48. [Google Scholar] [CrossRef]
- Gore, R.D.; Palaskar, S.J.; Bartake, A.R. Wheatgrass: Green blood can help to fight Cancer. J. Clin. Diagn. 2017, 11, ZC40. [Google Scholar] [CrossRef]
- Hattarki, S.A.; Bogar, C.; Bhat, K. Triticum aestivum (wheat grass) Exhibited Anticancer Activity on Oral Cancer (KB) Cell Line. Int. J. Pharma Res. Health Sci. 2020, 8, 3220–3224. [Google Scholar] [CrossRef]
- Alegría, A.; Garcia-Llatas, G.; Cilla, A. Static digestion models: General introduction. In The Impact of Food Bioactives on Gut Health; Cotter, A., Kleiveland, C., Eds.; Springer: Cham, Switzerland, 2015; pp. 3–12. [Google Scholar]
- Sandberg, A.S. Methods and options for in vitro dialyzability; benefits and limitations. Int. J. Vitam. Nutr. Res. 2005, 75, 395–404. [Google Scholar] [CrossRef]
- Fernández-García, E.; Carvajal-Lérida, I.; Pérez-Gálvez, A. In vitro bioaccessibility assessment as a prediction tool of nutritional efficiency. Nutr. Res. 2009, 29, 751–760. [Google Scholar] [CrossRef]
- Rickards, L.; Lynn, A.; Harrop, D.; Barker, M.E.; Russell, M.; Ranchordas, M.K. Effect of polyphenol-rich foods, juices, and concentrates on recovery from exercise induced muscle damage: A systematic review and meta-analysis. Nutrients 2021, 13, 2988. [Google Scholar] [CrossRef]
- Thakur, N.; Raigond, P.; Singh, Y.; Mishra, T.; Singh, B.; Lal, M.K.; Dutt, S. Recent updates on bioaccessibility of phytonutrients. Trends Food Sci. Technol. 2020, 97, 366–380. [Google Scholar] [CrossRef]
- Minekus, M.; Alminger, M.; Alvito, P.; Ballance, S.; Bohn, T.; Bourlieu, C.; Carriere, F.; Boutron, R.; Corredig, M.; Dupont, D.; et al. A standardised static in vitro digestion method suitable for food—An international consensus. Food Funct. 2014, 5, 1113–1124. [Google Scholar] [CrossRef] [Green Version]
- Kingstone, L.B.; Lassie, H.M.S. Microwave Energy for Acid Decomposition at Elevated Temperatures and Pressures Using 370 Biological and Botanical Samples. Anal. Chem. 1986, 58, 2534–2541. [Google Scholar] [CrossRef] [PubMed]
- Ordonez, A.A.L.; Gomez, J.D.; Vattuone, M.A. Antioxidant activities of Sechium edule (Jacq.) Swartz extracts. Food Chem. 2006, 97, 452–458. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Roe, H.J.; Kuether, A.C. The Determination of ascorbic acid in whole blood and urine through the 2,4-dinitrophenylhydrazine derivative of dehydroascorbic acid. J. Biol. Chem. 1943, 147, 399–407. [Google Scholar] [CrossRef]
- Kaur, N.; Singh, B.; Kaur, A.; Yadav, M.P.; Singh, N.; Ahlawat, A.K.; Singh, A.M. Effect of growing conditions on proximate, mineral, amino acid, phenolic composition and antioxidant properties of wheatgrass from different wheat (Triticum aestivum L.) varieties. Food Chem. 2021, 341, 128201. [Google Scholar] [CrossRef]
- Aghdam, M.S.; Hassanpouraghdam, M.B.; Paliyath, G.; Farmani, B. The language of calcium in postharvest life of fruits, vegetables and flowers. Sci. Hortic. 2012, 144, 102–115. [Google Scholar] [CrossRef]
- Carbonell-Capella, J.M.; Buniowska, M.; Barba, F.J.; Esteve, M.J.; Frígola, A. Analytical methods for determining bioavailability and bioaccessibility of bioactive compounds from fruits and vegetables: A review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 155–171. [Google Scholar] [CrossRef] [PubMed]
- Hambidge, K.M. Micronutrient bioavailability: Dietary Reference Intakes and a future perspective. Am. J. Clin. Nutr. 2010, 91, 1430S–1432S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Popova, A.; Mihaylova, D. Antinutrients in plant-based foods: A review. Open Biotechnol. J. 2019, 13, 68–76. [Google Scholar] [CrossRef] [Green Version]
- Aberoumand, A.; Deokule, S.S. Screening of some nutrients and anti-nutrients components in some plant foods of Iran and India. J. Agric. Technol. 2010, 6, 777–781. [Google Scholar]
- Kamiński, M.; Kręgielska-Narożna, M.; Bogdański, P. Determination of the Popularity of Dietary Supplements Using Google Search Rankings. Nutrients 2020, 12, 908. [Google Scholar] [CrossRef] [Green Version]
- Dickinson, A.; Blatman, J.; El-Dash, N.; Franco, J. Consumer usage and reasons for using dietary supplements: Report of a series of surveys. J. Am. Coll. Nutr. 2014, 33, 176–182. [Google Scholar] [CrossRef]
- Nutrition Research Institut. Available online: https://uncnri.org/2016/06/23/the-sweet-reality-of-eating-nutritious-fruits/ (accessed on 16 December 2021).
- Mujoriya, R.; Bodla, R.B. A study on wheat grass and its nutritional value. Food Sci. Qual. Manag. 2011, 2, 1–8. [Google Scholar]
- Abou-Raya, M.A.; Ghoniem, G.A.; Hameed, A.E. Organoleptic Properties and Acceptability of Fresh Beverages Using Wheat Grass Juice with some Fruit and Vegetable Juices. J. Food Dairy Sci. 2018, 9, 189–192. [Google Scholar] [CrossRef] [Green Version]
- Akbari, A.; Jelodar, G.; Nazifi, S.; Sajedianfard, J. An Overview of the Characteristics and Function of Vitamin C in Various Tissues: Relying on its Antioxidant Function. Zahedan J. Res. Med. Sci. 2016, 18, e4037. [Google Scholar] [CrossRef] [Green Version]
- Padayatty, S.J.; Katz, A.; Wang, Y.; Eck, P.; Kwon, O.; Lee, J.H.; Levine, M. Vitamin C as an antioxidant: Evaluation of its role in disease prevention. J. Am. Coll. Nutr. 2003, 22, 18–35. [Google Scholar] [CrossRef]
- Bendich, A.; Machlin, L.J.; Scandurra, O.; Burton, G.W.; Wayner, D.D.M. The antioxidant role of vitamin C. Free Radic. Biol. Med. 1986, 2, 419–444. [Google Scholar] [CrossRef]
- Salovaara, S.; Sandberg, A.S.; Andlid, T. Organic acids influence iron uptake in the human epithelial cell line Caco-2. J Agric. Food Chem. 2002, 50, 6233–6238. [Google Scholar] [CrossRef]
- Grosicki, A. Influence of vitamin C on cadmium absorption and distribution in rats. J. Trace Elem. Med. Biol. 2004, 18, 183–187. [Google Scholar] [CrossRef]
- Scheers, N. Regulatory Effects of Cu, Zn, and Ca on Fe Absorption: The Intricate Play between Nutrient Transporters. Nutrients 2013, 5, 957–970. [Google Scholar] [CrossRef]
- Zentner, L.E.; Rondó, P.H.; Duran, M.C.; Oliveira, J.M. Relationships of blood lead to calcium, iron, and vitamin C intakes in Brazilian pregnant women. Clin. Nutr. 2007, 27, 100–104. [Google Scholar] [CrossRef]
- Sandström, B.; Cederblad, A. Effect of ascorbic acid on the absorption of zinc and calcium in man. Int. J. Vitam. Nutr. Res. 1987, 57, 87–90. [Google Scholar]
- Rejman, K.; Górska-Warsewicz, H.; Czeczotko, M.; Laskowski, W. Nonalcoholic Beverages as Sources of Nutrients in the Average Polish Diet. Nutrients 2020, 12, 1262. [Google Scholar] [CrossRef]
- Scalbert, A.; Williamson, G. Dietary Intake and Bioaccessibility of Polyphenols. J. Nutr. 2000, 130, 2073S–2085S. [Google Scholar] [CrossRef]
- Perri, M.R.; Romano, C.; Marrelli, M.; Zicarelli, L.; Toma, C.C.; Basta, D.; Conforti, F.; Statti, G. Beneficial Role of Fruits, Their Juices, and Freeze-Dried Powders on Inflammatory Bowel Disease and Related Dysbiosis. Plants 2022, 11, 4. [Google Scholar] [CrossRef]
- Mushtaq, M.; Wani, S.M. Polyphenols and human health-A review. Int. J. Pharma. Bio. Sci. 2013, 4, 338–360. [Google Scholar]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell. Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, J.; Lin, X.; Bordiga, M.; Brennan, C.; Xu, B. Manipulating effects of fruits and vegetables on gut microbiota—A critical review. Int. J. Food Sci. Technol. 2021, 56, 2055–2067. [Google Scholar] [CrossRef]
- Gülçin, I. Antioxidant activity of food constituents: An overview. Arch. Toxicol. 2012, 86, 345–391. [Google Scholar] [CrossRef]
- Vertuani, S.; Angusti, A.; Manfredini, S. The antioxidants and pro-antioxidants network: An overview. Curr. Pharm. Des. 2004, 10, 1677–1694. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, D.; Ou, B.; Prior, R.I. The chemistry behind antioxidant capacity assays. J. Agric. Food. Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
| Juice | Volume Used in Experiment |
|---|---|
| Wheatgrass (WGJ) | 10 mL juice |
| Apple | |
| Orange | |
| Beet | |
| Carrot | |
| Lemon | |
| Apple + WGJ | 6 mL juice + 4 mL WGJ |
| Orange + WGJ | |
| Beet + WGJ | |
| Carrot + WGJ | |
| Lemon + WGJ | |
| Apple + WGJ + Lemon | 5.75 mL juice + 3.75 mL WGJ + 0.5 mL lemon |
| Orange + WGJ + Lemon | |
| Beet + WGJ + Lemon | |
| Carrot + WGJ + Lemon |
| Total Concentrations (mg L−1) | Bioaccessible Concentrations (mg L−1) | |||||
|---|---|---|---|---|---|---|
| K | Ca | Mg | K | Ca | Mg | |
| WGJ | 3262 ± 66.6 b | 431 ± 1.28 a | 325 ± 3.65 a | 2828 ± 19.1 b | 305 ± 1.94 a | 248 ± 0.43 a |
| Apple | 806 ± 75.5 e | 14.4 ± 1.71 d | 21.0 ± 0.81 c | 585 ± 18.0 d | 0.00 * | 11.0 ± 0.52 c |
| Beet | 4067 ± 187.3 a | 30.3 ± 0.55 d | 286 ± 19.7 a | 3908 ± 217.6 a | 3.66 ± 0.08 d | 218 ± 14.05 a |
| Carrot | 2109 ± 126.5 c | 59.4 ± 0.96 c | 33.4 ± 1.48 c | 1845 ± 88.0 c | 16.9 ± 0.85 c | 20.5 ± 11.5 c |
| Orange | 1620 ± 156 cd | 90 ± 7.65 b | 100 ± 12.2 b | 1177 ± 84.2 d | 40.1 ± 1.70 b | 69.2 ± 7.66 b |
| Lemon | 1257 ± 73.1 de | 63.5 ± 1.32 c | 69.2 ± 4.02 bc | 1097 ± 36.8 d | 34.9 ± 3.32 b | 49.9 ± 4.70 bc |
| F value | 102.92 | 2238.96 | 185.29 | 145.6 | 4511.88 | 227.79 |
| p | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 |
| Total Concentrations (mg L−1) | Bioaccessible Concentrations (mg L−1) | |||||
|---|---|---|---|---|---|---|
| Mn | Fe | Zn | Mn | Fe | Zn | |
| WGJ | 6.40 ± 0.07 a | 4.64 ± 0.11 a | 2.80 ± 0.03 a | 4.93 ± 0.09 a | 2.66 ± 0.08 a | 1.05 ± 0.02 b |
| Apple | 0.108 ± 0.01 c | 0.149 ± 0.02 d | 0.303 ± 0.07 c | 0.075 ± 0.007 c | 0.00 * | 0.13 ± 0.016 e |
| Beet | 0.72 ± 0.001 b | 3.04 ± 0.01 b | 2.75 ± 0.11 a | 0.13 ± 0.004 bc | 1.27 ± 0.13 b | 1.43 ± 0.04 a |
| Carrot | 0.70 ± 0.02 b | 1.64 ± 0.01 c | 0.80 ± 0.07 b | 0.35 ± 0.03 b | 1.20 ± 0.03 b | 0.58 ± 0.04 c |
| Orange | 0.115 ± 0.002 c | 2.57 ± 0.17 b | 0.34 ± 0.02 c | 0.093 ± 0.01 c | 0.43 ± 0.04 c | 0.32 ± 0.02 d |
| Lemon | 0.099 ± 0.01 c | 0.36 ± 0.03 d | 0.57 ± 0.06 bc | 0.083 ± 0.01 c | 0.18 ± 0.00 cd | 0.38 ± 0.04 d |
| F value | 6260.78 | 400.48 | 420.82 | 2740.94 | 223.4 | 266.92 |
| p | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 |
| Total Concentrations (mg L−1) | Bioaccessible Concentrations (mg L−1) | |||||
|---|---|---|---|---|---|---|
| K | Ca | Mg | K | Ca | Mg | |
| Apple | 806 ± 75.5 b | 14.4 ± 1.71 b | 21.0 ± 0.81 b | 585 ± 18.0 b | 0.00 b * | 11.0 ± 0.52 b |
| Apple + WGJ | 1760 ± 56.4 a | 181 ± 3.97 a | 143 ± 2.65 a | 1486 ± 70.7 a | 116 ± 12.3 a | 104 ± 2.94 a |
| Apple + WGJ + Lemon | 1679 ± 28.5 a | 165 ± 3.10 a | 129 ± 4.11 a | 1489 ± 54.6 a | 120 ± 8.2 a | 104 ± 5.45 a |
| F value | 88.02 | 900.69 | 542.35 | 98.13 | 63.62 | 224,48 |
| p | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 |
| Beet | 4067 ± 187.3 | 165 ± 3.10 b | 286 ± 19.7 | 3908 ± 217.6 a | 3.66 ± 0.08 b | 218 ± 14.05 |
| Beet + WGJ | 3683 ± 79.0 | 182 ± 2.10 a | 292 ± 25.4 | 3240 ± 191.6 ab | 40.7 ± 2.13 a | 221 ± 6.44 |
| Beet + WGJ + Lemon | 3584 ± 83.7 | 175 ± 2.65 a | 275 ± 21.7 | 2296 ± 106.9 b | 33.8 ± 1.77 a | 202 ± 12.7 |
| F value | 4.03 | 1873.39 | 0.16 | 20.61 | 151.71 | 0.16 |
| p | 0.0776 | <0.0001 | 0.8577 | 0.0021 | <0.0001 | 0.8577 |
| Carrot | 2109 ± 126.5 | 59.4 ± 0.96 b | 33.4 ± 1.48 b | 1845 ± 88.0 | 16.9 ± 0.85 b | 20.5 ± 11.5 b |
| Carrot + WGJ | 2496 ± 70.3 | 189 ± 14.1 a | 131 ± 11.7 a | 2319 ± 22.6 | 124 ± 13.6 a | 112 ± 11.8 a |
| Carrot + WGJ + Lemon | 2504 ± 25.6 | 194 ± 4.99 a | 140 ± 6.05 a | 2082 ± 162.6 | 127 ± 114.8 a | 105 ± 18.1 a |
| F value | 7.09 | 78.15 | 229.41 | 4.85 | 50.64 | 59.14 |
| p | 0.0263 | <0.0001 | 0.0001 | 0.0559 | 0.0002 | 0.0001 |
| Orange | 1620 ± 155.7 | 90 ± 7.65 b | 100 ± 12.2 b | 1177 ± 84.2 | 40.1 ± 1.70 b | 69.2 ± 7.66 b |
| Orange + WGJ | 2227 ± 82.3 | 227 ± 1.15 a | 189 ± 7.33 a | 1751 ± 84.8 | 161 ± 0.16 a | 137 ± 3.00 a |
| Orange + WGJ + Lemon | 2135 ± 111.0 | 202 ± 4.05 a | 170 ± 10.36 a | 1721 ± 125.7 | 164 ± 2.00 a | 130 ± 2.04 a |
| F value | 7.42 | 1783.21 | 21.26 | 10.42 | 210.02 | 229.41 |
| p | 0.0239 | <0.0001 | 0.0019 | 0.0112 | <0.0001 | 0.0001 |
| Lemon | 1257 ± 73.1 b | 63.5 ± 1.32 b | 69.2 ± 4.02 b | 1097 ± 36.8 b | 34.9 ± 3.32 b | 49.9 ± 4.70 b |
| Lemon + WGJ | 2019 ± 17.4 a | 207 ± 3.12 a | 164 ± 4.77 a | 1786 ± 74.5 a | 151 ± 2.27 a | 123 ± 0.50 a |
| F value | 102.57 | 1783.21 | 21.26 | 68.8 | 834.85 | 236.99 |
| p | 0.0005 | <0.0001 | 0.0019 | 0.0012 | <0.0001 | 0.0001 |
| Total Concentrations (mg L−1) | Bioaccessible Concentrations (mg L−1) | |||||
|---|---|---|---|---|---|---|
| Mn | Fe | Zn | Mn | Fe | Zn | |
| Apple | 0.11 ± 0.01 b | 0.15 ± 0.02 b | 0.30 ± 0.07 b | 0.08 ± 0.007 b | 0.00 * | 0.13 ± 0.02 c |
| Apple + WGJ | 2.63 ± 0.067 a | 1.83 ± 0.01 a | 1.26 ± 0.00 a | 2.29 ± 0.02 a | 0.41 ± 0.02 b | 0.52 ± 0.02 b |
| Apple + WGJ + Lemon | 2.46 ± 0.069 a | 1.69 ± 0.06 a | 1.16 ± 0.01 a | 2.32 ± 0.04 a | 0.74 ± 0.08 a | 0.90 ± 0.01 a |
| F value | 638.08 | 688.43 | 855.57 | 2367.13 | 56.8 | 855.57 |
| p | <0.0001 | <0.0001 | <0.0001 | <0.0001 | 0.0001 | <0.0001 |
| Beet | 0.72 ± 0.001 b | 3.04 ± 0.01 | 2.75 ± 0.11 | 0.13 ± 0.004 b | 1.27 ± 0.13 | 1.43 ± 0.04 |
| Beet + WGJ | 2.89 ± 0.04 a | 3.46 ± 0.18 | 2.76 ± 0.03 | 1.93 ± 0.10 a | 1.69 ± 0.08 | 1.43 ± 0.07 |
| Beet + WGJ + Lemon | 2.73 ± 0.05 a | 3.20 ± 0.11 | 2.57 ± 0.02 | 2.02 ± 0.13 a | 1.62 ± 0.11 | 1.29 ± 0.18 |
| F value | 937.99 | 3.11 | 2.53 | 129.66 | 4.38 | 0.52 |
| p | <0.0001 | 0.1185 | 0.1595 | <0.0001 | 0.0671 | 0.6167 |
| Carrot | 0.70 ± 0.02 b | 1.64 ± 0.01 | 0.80 ± 0.07 b | 0.35 ± 0.03 b | 1.20 ± 0.03 b | 0.58 ± 0.04 |
| Carrot + WGJ | 2.76 ± 0.29 a | 3.17 ± 0.44 | 1.51 ± 0.08 a | 2.06 ± 0.03 a | 1.81 ± 0.06 a | 0.78 ± 0.06 |
| Carrot + WGJ + Lemon | 2.85 ± 0.04 a | 2.73 ± 0.09 | 1.51 ± 0.01 a | 2.45 ± 0.12 a | 1.85 ± 0.13 a | 0.96 ± 0.08 |
| F value | 51.92 | 9.38 | 43.68 | 250.8 | 18.46 | 9.83 |
| p | 0.0002 | 0.0142 | 0.0003 | 0.0002 | 0.0027 | 0.0128 |
| Orange | 0.12 ± 0.002 b | 2.57 ± 0.17 | 0.34 ± 0.02 b | 0.09 ± 0.01 b | 0.43 ± 0.04 b | 0.32 ± 0.02 b |
| Orange + WGJ | 2.63 ± 0.02 a | 1.95 ± 0.01 | 1.28 ± 0.03 a | 2.32 ± 0.06 a | 1.49 ± 0.07 a | 0.94 ± 0.04 a |
| Orange + WGJ + Lemon | 2.39 ± 0.08 a | 1.83 ± 0.13 | 1.18 ± 0.06 a | 2.15 ± 0.10 a | 1.44 ± 0.11 a | 1.04 ± 0.02 a |
| F value | 964.85 | 10.37 | 160.61 | 315.64 | 56.68 | 174.48 |
| p | <0.0001 | 0.0113 | <0.0001 | <0.0001 | 0.0001 | <0.0001 |
| Lemon | 0.10 ± 0.01 b | 0.36 ± 0.03 b | 0.57 ± 0.06 b | 0.08 ± 0.01 b | 0.18 ± 0.00 b | 0.38 ± 0.04 b |
| Lemon + WGJ | 2.66 ± 0.082 a | 2.01 ± 0.04 a | 1.38 ± 0.01 a | 2.28 ± 0.03 a | 1.47 ± 0.04 a | 1.07 ± 0.02 a |
| F value | 757.72 | 1049 | 209.28 | 3878.23 | 1103.83 | 222.8 |
| p | <0.0001 | <0.0001 | 0.0001 | <0.0001 | <0.0001 | 0.0001 |
| Vitamin C | Phenolic Content | Flavonoid Content | DPPH | |
|---|---|---|---|---|
| Apple | 3.93 ± 0.024 b | 677.38 ± 4.32 | 19.88 ± 0.91 b | 0.29 ± 0.002 |
| Apple + WGJ | 25.47 ± 0.787 ab | 826.82 ± 11.03 | 143.77 ± 7.74 a | 0.34 ± 0.018 |
| Apple + WGJ + Lemon | 36.40 ± 0.279 a | 858.65 ± 41.34 | 90.11 ± 2.28 ab | 0.31 ± 0.005 |
| Beet | 2.46 ± 0.174 b | 861.27 ± 17.52 | 237.68 ± 2.73 | 0.42 ± 0.066 |
| Beet + WGJ | 16.73 ± 0.627 ab | 1005.47 ± 10.81 | 244.78 ± 6.83 | 0.30 ± 0.023 |
| Beet + WGJ + Lemon | 30.96 ± 0.858 a | 961.02 ± 34.32 | 141.41 ± 4.56 | 0.22 ± 0.003 |
| Carrot | 8.15 ± 1.135 b | 154.53 ± 5.67 b | 18.83 ± 0.53 b | 3.14 ± 0.061 a |
| Carrot + WGJ | 27.12 ± 0.881 ab | 634.68 ± 23.23 ab | 169.02 ± 0.46 a | 0.49 ± 0.034 ab |
| Carrot + WGJ + Lemon | 37.13 ± 0.733 a | 796.48 ± 12.11 a | 92.48 ± 1.82 ab | 0.35 ± 0.005 b |
| Orange | 37.41 ± 1.726 | 942.17 ± 36.98 | 85.38 ± 4.1 b | 0.15 ± 0.006 |
| Orange + WGJ | 44.66 ± 2.436 | 1122.32 ± 31.07 | 128.78 ± 6.38 a | 0.22 ± 0.020 |
| Orange + WGJ + Lemon | 44.90 ± 1.906 | 1033.93 ± 63.84 | 100.37 ± 0.91 ab | 0.21 ± 0.006 |
| Lemon | 33.79 ± 0.461 | 754.28 ± 28.24 | 53.03 ± 1.82 b | 0.18 ± 0.008 |
| Lemon + WGJ | 45.15 ± 1.095 | 1020.82 ± 7.14 | 115.37 ± 4.1 a | 0.25 ± 0.010 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grubišić, S.; Kristić, M.; Lisjak, M.; Mišković Špoljarić, K.; Petrović, S.; Vila, S.; Rebekić, A. Effect of Wheatgrass Juice on Nutritional Quality of Apple, Carrot, Beet, Orange and Lemon Juice. Foods 2022, 11, 445. https://doi.org/10.3390/foods11030445
Grubišić S, Kristić M, Lisjak M, Mišković Špoljarić K, Petrović S, Vila S, Rebekić A. Effect of Wheatgrass Juice on Nutritional Quality of Apple, Carrot, Beet, Orange and Lemon Juice. Foods. 2022; 11(3):445. https://doi.org/10.3390/foods11030445
Chicago/Turabian StyleGrubišić, Sanja, Marija Kristić, Miroslav Lisjak, Katarina Mišković Špoljarić, Sonja Petrović, Sonja Vila, and Andrijana Rebekić. 2022. "Effect of Wheatgrass Juice on Nutritional Quality of Apple, Carrot, Beet, Orange and Lemon Juice" Foods 11, no. 3: 445. https://doi.org/10.3390/foods11030445
APA StyleGrubišić, S., Kristić, M., Lisjak, M., Mišković Špoljarić, K., Petrović, S., Vila, S., & Rebekić, A. (2022). Effect of Wheatgrass Juice on Nutritional Quality of Apple, Carrot, Beet, Orange and Lemon Juice. Foods, 11(3), 445. https://doi.org/10.3390/foods11030445






