Maternal Diet Quality and the Health Status of Newborns
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.3. Dietary Assessment
2.4. Persistent Organic Pollutant Analysis
2.5. Oxidative Stress Analysis in Mothers and Newborns
2.6. Statistical Analysis
3. Results
3.1. Participants
3.2. Diet Quality
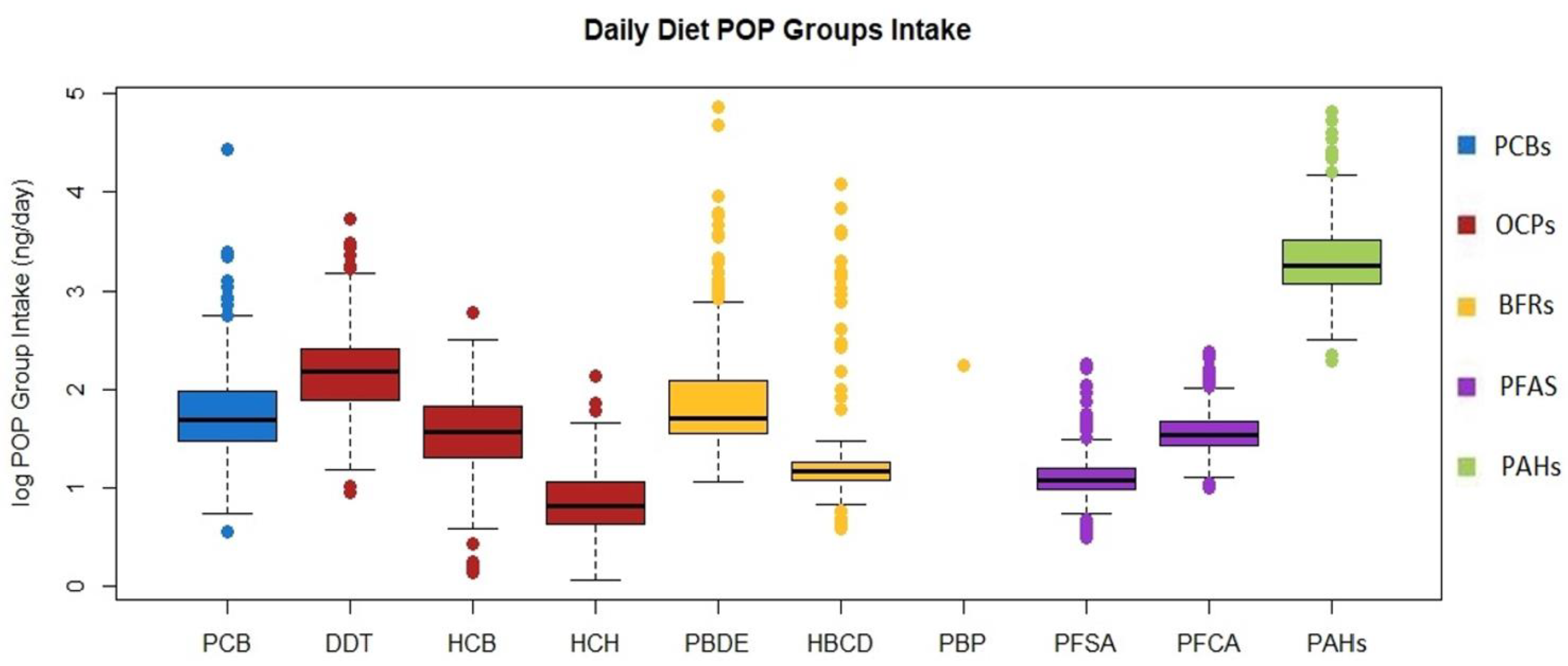
3.3. Diet POPs
3.4. Birth Weight
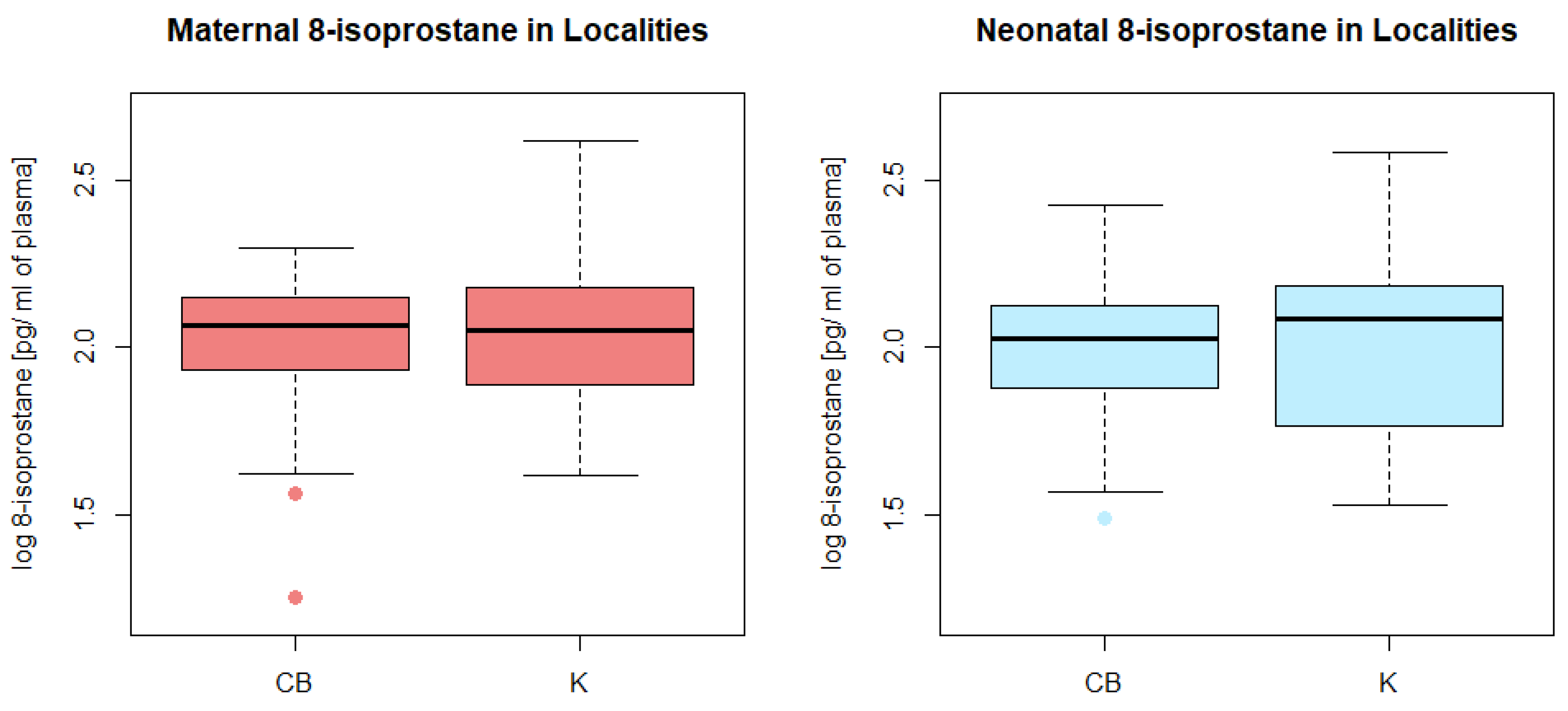
3.5. Plasma 8-Isoprostane
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hales, C.N.; Barker, D.J. Type 2 (non-insulin-dependent) diabetes mellitus: The thrifty phenotype hypothesis. Diabetologia 1992, 35, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Poulsen, P.; Vaag, A.A.; Kyvik, K.O.; Møller Jensen, D.; Beck-Nielsen, H. Low birth weight is associated with NIDDM in discordant monozygotic and dizygotic twin pairs. Diabetologia 1997, 40, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Simmons, R.A. Developmental origins of diabetes: The role of epigenetic mechanisms. Curr. Opin. Endocrinol. Diabetes Obes. 2007, 14, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Anway, M.D.; Cupp, A.S.; Uzumcu, M.; Skinner, M.K. Epigenetic transgenerational actions of endocrine disruptors and male fertility. Science 2005, 308, 1466–1469. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Chillaron, J.C.; Isganaitis, E.; Charalambous, M.; Gesta, S.; Pentinat-Pelegrin, T.; Faucette, R.R.; Otis, J.P.; Chow, A.; Diaz, R.; Ferguson-Smith, A.; et al. Intergenerational transmission of glucose intolerance and obesity by in utero undernutrition in mice. Diabetes 2009, 58, 460–468. [Google Scholar] [CrossRef]
- Hanafi, M.Y.; Saleh, M.M.; Saad, M.I.; Abdelkhalek, T.M.; Kamel, M.A. Transgenerational effects of obesity and malnourishment on diabetes risk in F2 generation. Mol. Cell Biochem. 2016, 412, 269–280. [Google Scholar] [CrossRef]
- Aye, I.L.; Rosario, F.J.; Powell, T.L.; Jansson, T. Adiponectin supplementation in pregnant mice prevents the adverse effects of maternal obesity on placental function and fetal growth. Proc. Natl. Acad. Sci. USA 2015, 13, 12858–12863. [Google Scholar] [CrossRef]
- Cerf, M.E.; Herrera, E. High fat diet administration during specific periods of pregnancy alters maternal fatty acid profiles in the near-term rat. Nutrients 2016, 4, 25. [Google Scholar] [CrossRef]
- Rebelato, H.J.; Esquisatto, M.A.; de Sousa Righi, E.F.; Catisti, R. Gestational protein restriction alters cell proliferation in rat placenta. J. Mol. Histol. 2016, 47, 203–211. [Google Scholar] [CrossRef]
- DuBois, B.N.; O’Tierney-Ginn, P.; Pearson, J.; Friedman, J.E.; Thornburg, K.; Cherala, G. Maternal obesity alters feto-placental cytochrome P4501A1 activity. Placenta 2012, 33, 1045–1051. [Google Scholar] [CrossRef]
- Średnicka, P.; Juszczuk-Kubiak, E.; Wójcicki, M.; Akimowicz, M.; Roszko, M.Ł. Probiotics as a biological detoxification tool of food chemical contamination: A review. Food Chem. Toxicol. 2021, 153, 112306. [Google Scholar] [CrossRef] [PubMed]
- Gálvez-Ontiveros, Y.; Páez, S.; Monteagudo, C.; Rivas, A. Endocrine disruptors in food: Impact on gut microbiota and metabolic miseases. Nutrients 2020, 12, 1158. [Google Scholar] [CrossRef] [PubMed]
- Gallardo, J.M.; Klünder-Klünder, M.; Sánchez-Urbina, R. Diet and maternal obesity are associated with increased oxidative stress in newborns: A cross-sectional study. Nutrients 2022, 10, 746. [Google Scholar] [CrossRef]
- Ambroz, A.; Vlkova, V.; Rossner, P., Jr.; Rossnerova, A.; Svecova, V.; Milcova, A.; Pulkrabova, J.; Hajslova, J.; Veleminsky, M., Jr.; Solansky, I.; et al. Impact of air pollution on oxidative DNA damage and lipid peroxidation in mothers and their newborns. Int. J. Hyg. Environ. Health 2016, 219, 545–556. [Google Scholar] [CrossRef]
- D’Angelo, G.; Chimenz, R.; Reiter, R.J.; Gitto, E. Use of melatonin in oxidative stress related neonatal diseases. Antioxidants 2020, 2, 477. [Google Scholar] [CrossRef]
- Available online: https://www.kaloricketabulky.cz/ (accessed on 17 September 2022).
- Brazdova, Z. Nutrition of Pregnant and Lactating Women: A Guide.2. vyd; Department of Preventive Medicine, M.U.: Brno, Czech Republic, 2004; ISBN 80-901-4277-X. (In Czech) [Google Scholar]
- Polachova, A.; Gramblicka, T.; Parizek, O.; Sram, R.J.; Stupak, M.; Hajslova, J.; Pulkrabova, J. Estimation of human exposure to polycyclic aromatic hydrocarbons (PAHs) based on the dietary and outdoor atmospheric monitoring in the Czech Republic. Environ. Res. 2020, 182, 108977. [Google Scholar] [CrossRef]
- Rossner, P., Jr.; Rossnerova, A.; Spatova, M.; Beskid, O.; Uhlirova, K.; Libalova, H.; Solansky, I.; Topinka, J.; Sram, R.J. Analysis of biomarkers in a Czech population exposed to heavy air pollution. Part II: Chromosomal aberrations and oxidative stress. Mutagenesis 2013, 28, 97–106. [Google Scholar] [CrossRef]
- Agostoni, C.; Bresson, J.; Fairweather-Tait, S.; Flynn, A.; Golly, I.; Korhonen, I.; Lagiou, P.; Løvik, M.; Marchelli, R.; Martin, A.; et al. Scientific opinion on dietary reference values for protein. EFSA J. 2012, 10, 2557. [Google Scholar] [CrossRef]
- EFSA. Scientific opinion on dietary reference values for carbohydrates and dietary fibre. EFSA J. 2010, 8, 1462. [Google Scholar]
- Mousa, A.; Naqash, A.; Lim, S. Macronutrient and micronutrient intake during pregnancy: An overview of recent evidence. Nutrients 2019, 11, 443. [Google Scholar] [CrossRef]
- Cucó, G.; Arija, V.; Iranzo, R.; Vilà, J.; Prieto, M.T.; Fernández-Ballart, J. Association of maternal protein intake before conception and throughout pregnancy with birth weight. Acta Obstet. Gynecol. Scand. 2006, 85, 413–421. [Google Scholar] [CrossRef]
- Lowensohn, R.I.; Stadler, D.D.; Naze, C. Current concepts of maternal nutrition. Obstet. Gynecol. Surv. 2016, 71, 413–426. [Google Scholar] [CrossRef] [PubMed]
- Zulyniak, M.A.; de Souza, R.J.; Shaikh, M.; Desai, D.; Lefebvre, D.L.; Gupta, M.; Wilson, J.; Wahi, G.; Subbarao, P.; Becker, A.B.; et al. Does the impact of a plant-based diet during pregnancy on birth weight differ by ethnicity? A dietary pattern analysis from a prospective Canadian birth cohort alliance. BMJ Open 2017, 7, e017753. [Google Scholar] [CrossRef] [PubMed]
- Vargas-Terrones, M.; Nagpal, T.S.; Barakat, R. Impact of exercise during pregnancy on gestational weight gain and birth weight: An overview. Braz. J. Phys. Ther. 2019, 23, 164–169. [Google Scholar] [CrossRef]
- Gul, R.; Iqbal, S.; Anwar, Z.; Ahdi, S.G.; Ali, S.H.; Pirzada, S. Pre-pregnancy maternal BMI as predictor of neonatal birth weight. PLoS ONE 2020, 15, e0240748. [Google Scholar] [CrossRef] [PubMed]
- Navajas-Porras, B.; Pérez-Burillo, S.; Valverde-Moya, Á.J.; Hinojosa-Nogueira, D.; Pastoriza, S.; Rufián-Henares, J.Á. Effect of cooking methods on the antioxidant capacity of plant foods submitted to in vitro digestion-fermentation. Antioxidants 2020, 9, 1312. [Google Scholar] [CrossRef] [PubMed]
- Han, J.H.; Lee, H.J.; Cho, M.R.; Chang, N.; Kim, Y.; Oh, S.Y.; Kang, M.H. Total antioxidant capacity of the Korean diet. Nutr. Res. Pract. 2014, 8, 183–191. [Google Scholar] [CrossRef]
- Sun, Y.; Ge, X.; Li, X.; He, J.; Wei, X.; Du, J.; Sun, J.; Li, X.; Xun, Z.; Liu, W.; et al. High-fat diet promotes renal injury by inducing oxidative stress and mitochondrial dysfunction. Cell Death Dis. 2020, 11, 914. [Google Scholar] [CrossRef]
- Muñoz, A.; Costa, M. Nutritionally mediated oxidative stress and inflammation. Oxid. Med. Cell Longev. 2013, 2013, 610950. [Google Scholar] [CrossRef]
- Najjar, R.S.; Moore, C.E.; Montgomery, B.D. A defined, plant-based diet utilized in an outpatient cardiovascular clinic effectively treats hypercholesterolemia and hypertension and reduces medications. Clin. Cardiol. 2018, 41, 307–313. [Google Scholar] [CrossRef]
- EFSA Panel on Contaminants in the Food Chain (EFSA CONTAM Panel); Schrenk, D.; Bignami, M.; Bodin, L.; Chipman, J.K.; Del Mazo, J.; Grasl-Kraupp, B.; Hogstrand, C.; Hoogenboom, L.; Leblanc, J.; et al. Risk to human health related to the presence of perfluoroalkyl substances in food. EFSA J. 2020, 18, e06223. [Google Scholar] [CrossRef] [PubMed]
- Pasecnaja, E.; Bartkevics, V.; Zacs, D. Occurrence of selected per- and polyfluorinated alkyl substances (PFASs) in food available on the European market—A review on levels and human exposure assessment. Chemosphere 2022, 287, 132378. [Google Scholar] [CrossRef] [PubMed]
- Lallas, P. The Stockholm Convention on Persistent Organic Pollutants. Am. J. Int. Law 2001, 95, 692–708. [Google Scholar] [CrossRef]
- Kirman, C.R.; Aylward, L.L.; Hays, S.M.; Krishnan, K.; Nong, A. Biomonitoring equivalents for DDT/DDE. Regul. Toxicol. Pharmacol. 2011, 60, 172–180. [Google Scholar] [CrossRef]
- Cirillo, P.M.; La Merrill, M.A.; Krigbaum, N.Y.; Cohn, B.A. Grandmaternal perinatal serum DDT in relation to granddaughter early menarche and adult obesity: Three generations in the child health and development studies cohort. Cancer Epidemiol. Biomark. Prev. 2021, 30, 1480–1488. [Google Scholar] [CrossRef] [PubMed]
- Harada, T.; Takeda, M.; Kojima, S.; Tomiyama, N. Toxicity and carcinogenicity of dichlorodiphenyltrichloroethane (DDT). Toxicol. Res. 2016, 32, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Marouani, N.; Hallegue, D.; Sakly, M.; Benkhalifa, M.; Ben Rhouma, K.; Tebourbi, O. p,p′-DDT induces testicular oxidative stress-induced apoptosis in adult rats. Reprod. Biol. Endocrinol. 2017, 26, 40. [Google Scholar] [CrossRef]
- Thompson, L.A.; Ikenaka, Y.; Sobhy Darwish, W.; Nakayama, S.M.M.; Mizukawa, H.; Ishizuka, M. Effects of the organochlorine p,p′-DDT on MCF-7 cells: Investigating metabolic and immune modulatory transcriptomic changes. Environ. Toxicol. Pharmacol. 2019, 72, 103249. [Google Scholar] [CrossRef]
- Waliszewski, S.M.; Aguirre, A.A.; Infanzón, R.M.; Siliceo, J. Carry-over of persistent organochlorine pesticides through placenta to fetus. Salud. Publica Mex. 2000, 42, 384–390. [Google Scholar] [CrossRef]
- Tang, Z.R.; Xu, X.L.; Deng, S.L.; Lian, Z.X.; Yu, K. Oestrogenic Endocrine disruptors in the placenta and the fetus. Int. J. Mol. Sci. 2020, 21, 1519. [Google Scholar] [CrossRef]
- Mrema, E.J.; Rubino, F.M.; Brambilla, G.; Moretto, A.; Tsatsakis, A.M.; Colosio, C. Persistent organochlorinated pesticides and mechanisms of their toxicity. Toxicology 2013, 10, 74–88. [Google Scholar] [CrossRef] [PubMed]
- Obersby, D.; Chappell, D.C.; Dunnett, A.; Tsiami, A.A. Plasma total homocysteine status of vegetarians compared with omnivores: A systematic review and meta-analysis. Br. J. Nutr. 2013, 14, 785–794. [Google Scholar] [CrossRef] [PubMed]
- Salter, A.M. The effects of meat consumption on global health. Rev. Sci. Tech. 2018, 37, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Ford, T.C.; Downey, L.A.; Simpson, T.; McPhee, G.; Oliver, C.; Stough, C. The effect of a high-dose vitamin B multivitamin supplement on the relationship between brain metabolism and blood biomarkers of oxidative stress: A randomized control trial. Nutrients 2018, 10, 1860. [Google Scholar] [CrossRef]
- Jeon, Y.M.; Kwon, Y.; Lee, S.; Kim, S.; Jo, M.; Lee, S.; Kim, S.R.; Kim, K.; Kim, H.J. Vitamin B12 reduces TDP-43 toxicity by alleviating oxidative stress and mitochondrial dysfunction. Antioxidants 2021, 11, 82. [Google Scholar] [CrossRef]
- Wimalawansa, S.J. Vitamin D deficiency: Effects on oxidative stress, epigenetics, gene regulation, and aging. Biology 2019, 8, 30. [Google Scholar] [CrossRef]
- Javanbakht, M.H.; Mohammady, H.; Fooladsaz, K.; Razzaghi, M.; Zarei, M.; Djalali, M. Are serum levels of F2-isoprostane and oxidized-LDL related to vitamin d status in type 2 diabetic patients? a case-control study. Rep. Biochem. Mol. Biol. 2016, 5, 26–32. [Google Scholar]
- Darnerud, P.O. Toxic effects of brominated flame retardants in man and in wildlife. Environ. Int. 2003, 29, 841–853. [Google Scholar] [CrossRef]
- Logerova, H.; Tuma, P.; Stupak, M.; Pulkrabova, J.; Dlouha, P. Evaluation of the burdening on the Czech population by brominated flame retardants. Int. J. Environ. Res. Public Health 2019, 16, 4105. [Google Scholar] [CrossRef]
- Schreiberova, M.; Skachova, H.; Vlasakova, L. Air quality in the Czech Republic in 2020 Preliminary evaluation, I.I. part Evaluation of concentrations of benzo[a]pyrene, benzene and heavy metals (As, Cd, Ni, Pb). CHMI 2021, 10, 5. Available online: https://www.chmi.cz/files/portal/docs/uoco/interaktivni/zpravy/2021-04_manualniStanice2020/#chapter6 (accessed on 2 February 2022). (In Czech).
- Hladky, D.; Volna, V.; Krejci, B. Evaluation of measurements at monitoring stations Vernovice and Mizerov in 2020. CHMI 2021, 31, 3. Available online: https://www.chmi.cz/files/portal/docs/poboc/OS/OCO/prehledy/mizerov_vernovice/zprava_MSK_2020.pdf (accessed on 2 February 2022). (In Czech).
- European Union. Commission Regulation (EU) 2020/1255 of 7 September 2020. Off. J. Eur. Union 2020, 8, 9. Available online: http://data.europa.eu/eli/reg/2020/1255/oj (accessed on 2 February 2022).

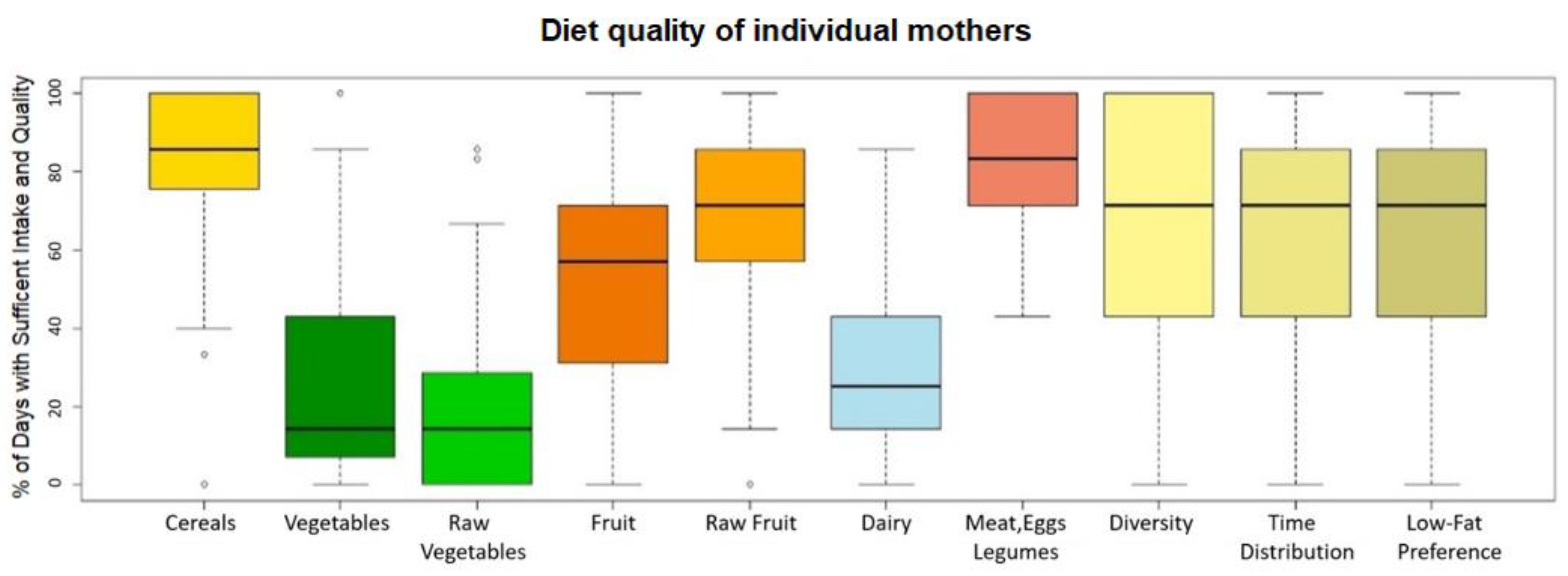


| Diet Factor | Cut Point |
|---|---|
| Cereals | 3 servings/day |
| Vegetables | 300 g/day |
| Raw vegetables | 200 g/day |
| Fruit | 200 g/day |
| Raw fruit | 100 g/day |
| Dairy | 3 servings/day |
| Meat, eggs, legumes | 1 serving/day |
| Diversity | 3 different foods in 3 different food groups/day |
| Time distribution | 5 nutritional value meals/day |
| Low-fat preference | predominance of low-fat foods over high-fat foods in a period of one day |
| Karvina | Ceske Budejovice | ||||||
|---|---|---|---|---|---|---|---|
| N | Mean ± SD | Median (Min.–Max.) | N | Mean ± SD | Median (Min.–Max.) | p | |
| Mothers | |||||||
| Age (years) | 26 | 30.50 ± 5.05 | 30 (22–43) | 28 | 31.50 ± 3.95 | 31 (25–39) | 0.424 |
| Gestation (weeks) | 26 | 39.38 ± 1.06 | 40 (37–41) | 28 | 39.32 ± 1.19 | 39 (37–41) | 0.795 |
| Weight (before pregnancy) | 26 | 64.88 ± 16.66 | 60.50 (43–115) | 28 | 70.82 ± 12.79 | 69 (48–98) | 0.035 |
| WHtR (before pregnancy) | 26 | 0.45 ± 0.09 | 0.44 (0.36–0.80) | 28 | 0.49 ± 0.08 | 0.47 (0.36–0.65) | 0.090 |
| BMI (before pregnancy) | 26 | 23.57 ± 5.36 | 22.27 (17.21–41.23) | 28 | 24.44 ± 4.67 | 22.63 (17.42–36.89) | 0.350 |
| Cotinine (ng/mg creatinine) | 26 | 3.75 ± 7.78 | 1.13 (0.22–38.78) | 27 | 1.47 ± 2.40 | 0.77 (0.18–12.34) | 0.039 |
| Education (Basic/ High school/University) | 2/10/14 | N/A | N/A | 2/8/18 | N/A | N/A | 0.511 |
| Newborns | |||||||
| Birth weight (g) | 26 | 3329 ± 411.28 | 3275 (2730–4170) | 28 | 3548 ± 445.83 | 3540 (2650–4580) | 0.067 |
| Apgar score (value) | 26 | 9.80 ± 0.57 | 10 (8–10) | 28 | 9.79 ± 0.42 | 10 (9–10) | 0.430 |
| Gender (M/F) | 16/10 | N/A | N/A | 13/15 | N/A | N/A | 0.401 |
| Time interval between diet sampling and cord blood sampling (days) | 26 | 12.77 ± 7.66 | 12 (1–29) | 26 | 10.54 ±7.49 | 8.5 (1.5–27) | 0.245 |
| University-Educated Mothers (n = 33) | Lower Educated Mothers (n = 21) | ||||
|---|---|---|---|---|---|
| Mean ± SD | Median (Min.–Max.) | Mean ± SD | Median (Min.–Max.) | p | |
| Average daily intake | |||||
| Protein (g) | 77.8 ± 14.6 | 76.9 (52.3–107.7) | 68.0 ± 18.0 | 69.0 (31.7–103.9) | 0.050 |
| Carbohydrate (g) | 258 ± 63.6 | 244 (147–391) | 217 ± 66.6 | 217 (107–325) | 0.036 |
| Fiber (g) | 18.7 ± 5.1 | 17.8 (10.6–37.6) | 15.2 ± 7.5 | 13.1 (6.3–36.4) | 0.012 |
| % of days with sufficient intake | |||||
| Cereal (%) | 86.9 ± 21.7 | 85.7 (0.0–100) | 73.3 ± 24.0 | 77.4 (33.3–100) | 0.024 |
| % of days with sufficient quality | |||||
| Food intake distribution (%) | 72.0 ± 28.5 | 71.4 (0.0–100) | 54.6 ± 32.9 | 61.9 (0.0–100) | 0.046 |
| Low-fat food preference (%) | 72.2 ± 25.7 | 75.0 (0.0–100) | 53.3 ± 28.3 | 46.4 (0.0–100) | 0.015 |
| Average diet quality (max. 10 points) | 5.9 ± 1.0 | 6.0 (4.3–7.9) | 5.0 ± 1.7 | 5.0 (2.3–9.0) | 0.038 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pavlikova, J.; Ambroz, A.; Honkova, K.; Chvojkova, I.; Sram, R.J.; Rossner, P., Jr.; Topinka, J.; Gramblicka, T.; Parizek, O.; Parizkova, D.; et al. Maternal Diet Quality and the Health Status of Newborns. Foods 2022, 11, 3893. https://doi.org/10.3390/foods11233893
Pavlikova J, Ambroz A, Honkova K, Chvojkova I, Sram RJ, Rossner P Jr., Topinka J, Gramblicka T, Parizek O, Parizkova D, et al. Maternal Diet Quality and the Health Status of Newborns. Foods. 2022; 11(23):3893. https://doi.org/10.3390/foods11233893
Chicago/Turabian StylePavlikova, Jitka, Antonin Ambroz, Katerina Honkova, Irena Chvojkova, Radim J. Sram, Pavel Rossner, Jr., Jan Topinka, Tomas Gramblicka, Ondrej Parizek, Denisa Parizkova, and et al. 2022. "Maternal Diet Quality and the Health Status of Newborns" Foods 11, no. 23: 3893. https://doi.org/10.3390/foods11233893
APA StylePavlikova, J., Ambroz, A., Honkova, K., Chvojkova, I., Sram, R. J., Rossner, P., Jr., Topinka, J., Gramblicka, T., Parizek, O., Parizkova, D., Schmuczerova, J., Pulkrabova, J., & Rossnerova, A. (2022). Maternal Diet Quality and the Health Status of Newborns. Foods, 11(23), 3893. https://doi.org/10.3390/foods11233893








