Microbial Control of Raw and Cold-Smoked Atlantic Salmon (Salmo salar) through a Microwave Plasma Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Plasma Treatment
2.3. Microbiological Experiments
2.3.1. Storage Tests
2.3.2. Recovery, Detection, and Evaluation of Native Contamination
2.4. Quality Control
2.4.1. Pictures
2.4.2. Texture Analysis
2.4.3. Color Stimulus Specification
2.5. Statistics
2.5.1. General Statistics
2.5.2. Model
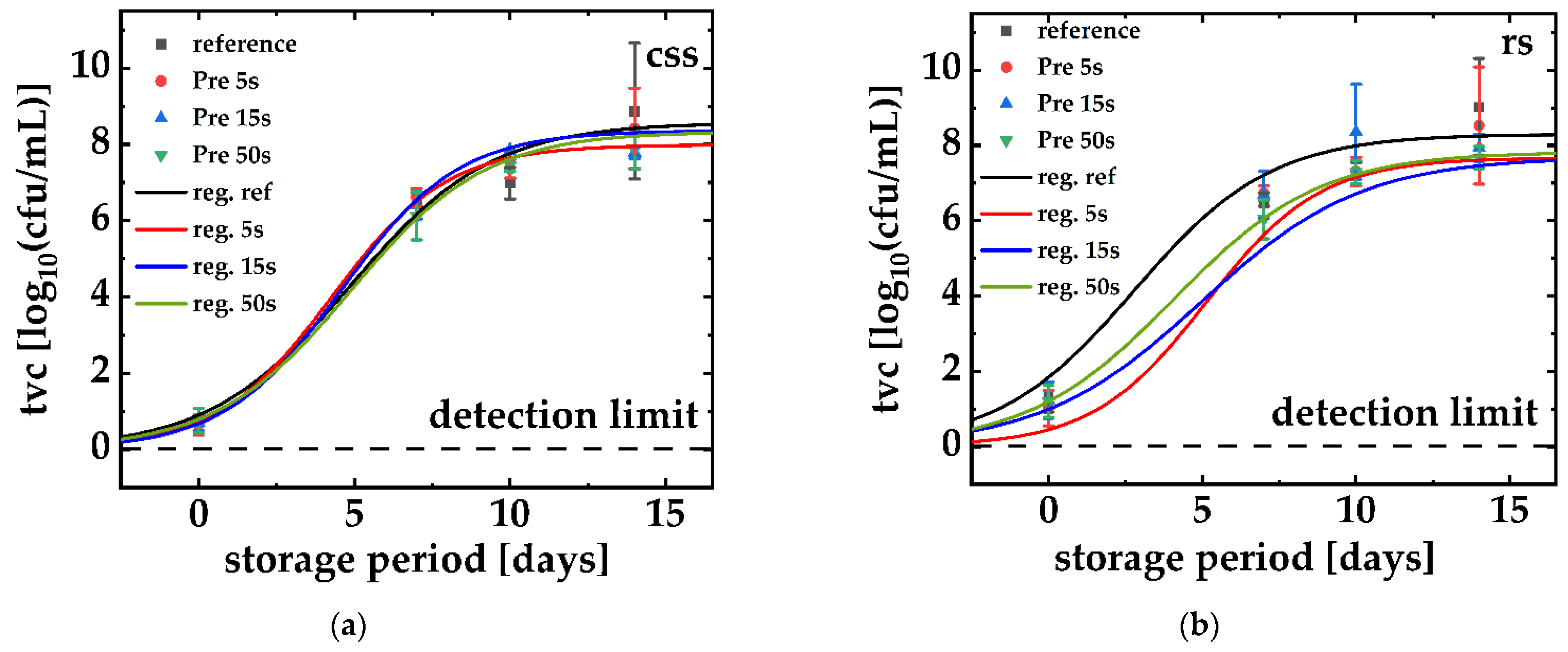
3. Results
3.1. Raw and Smoked Salmon vs. Their PPA-Treated Counterparts
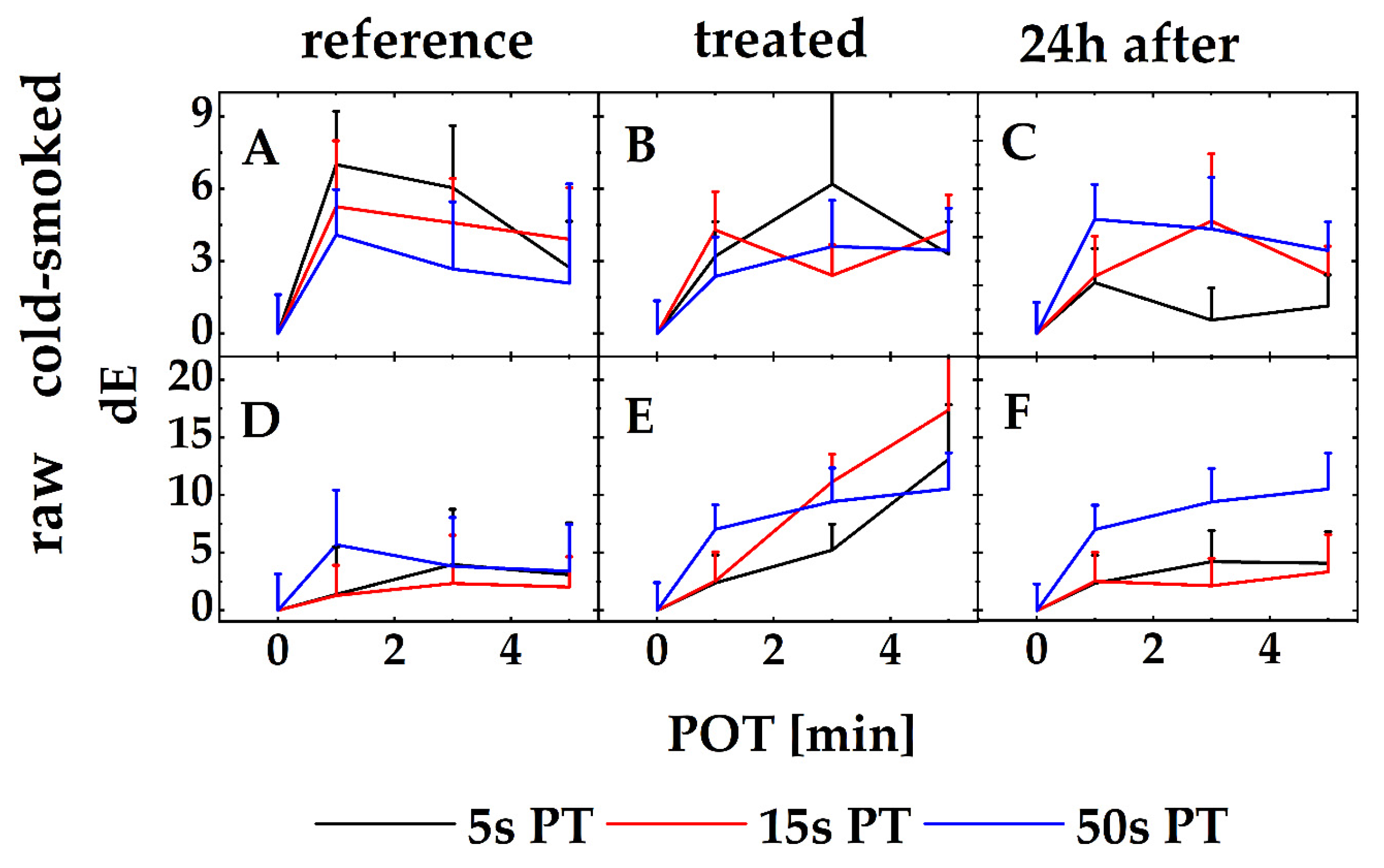
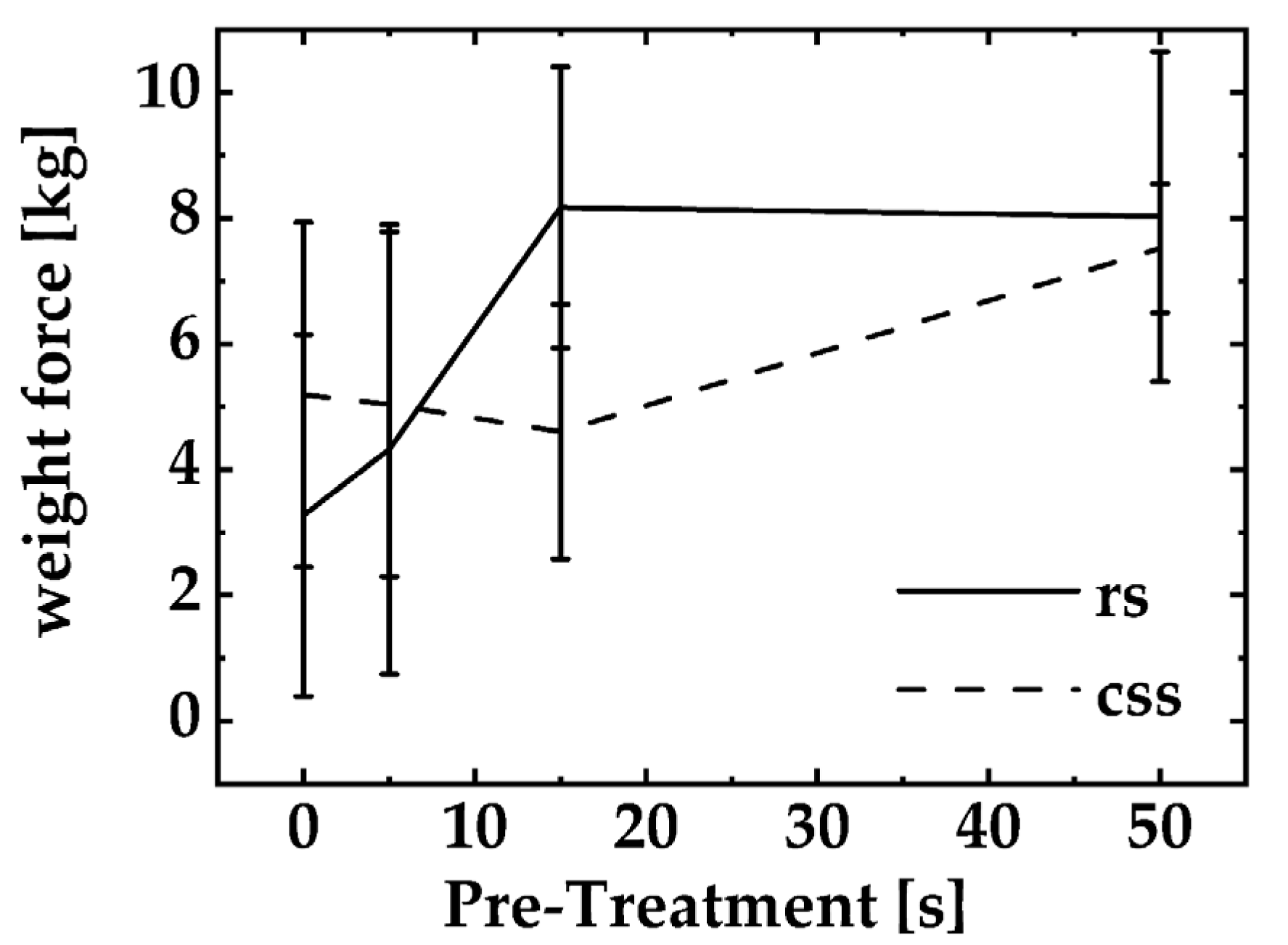
3.2. Quality Control
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sogin, J.H.; Lopez-Velasco, G.; Yordem, B.; Lingle, C.K.; David, J.M.; Çobo, M.; Worobo, R.W. Implementation of ATP and Microbial Indicator Testing for Hygiene Monitoring in a Tofu Production Facility Improves Product Quality and Hygienic Conditions of Food Contact Surfaces: A Case Study. Appl. Environ. Microbiol. 2021, 87, e02278-20. [Google Scholar] [CrossRef] [PubMed]
- Carrascosa, C.; Raheem, D.; Ramos, F.; Saraiva, A.; Raposo, A. Microbial Biofilms in the Food Industry—A Comprehensive Review. Int. J. Environ. Res. Public Health 2021, 18, 2014. [Google Scholar] [CrossRef]
- Garcia Martinez, M.; Fearne, A.; Caswell, J.A.; Henson, S. Co-regulation as a possible model for food safety governance: Opportunities for public–private partnerships. Food Policy 2007, 32, 299–314. [Google Scholar] [CrossRef]
- Bruhn, C.M. Consumer concerns: Motivating to action. Emerg. Infect. Dis. 1997, 3, 511–515. [Google Scholar] [CrossRef]
- Powell, D.A.; Jacob, C.J.; Chapman, B.J. Enhancing food safety culture to reduce rates of foodborne illness. Food Control 2011, 22, 817–822. [Google Scholar] [CrossRef]
- Novotny, L.D.; Dvorska, L.; Lorencova, A.; Beran, V.; Pavlik, I. Fish: A potential source of bacterial pathogenes for human beings. Vet. Med. 2004, 49, 343–358. [Google Scholar] [CrossRef]
- Authority, E.F.S. The European Union One Health 2020 Zoonoses Report. EFSA J. 2021, 19, e06971. [Google Scholar] [CrossRef]
- European Food Safety Authority; European Centre for Disease Prevention and Control. The European Union On Health 2019 Zoonoses Report. EFSA J. 2019, 19, e05926. [Google Scholar]
- Rørvik, L.M. Listeria monocytogenes in the smoked salmon industry. Int. J. Food Microbiol. 2000, 62, 183–190. [Google Scholar] [CrossRef]
- Gillesberg Lassen, S.; Ethelberg, S.; Björkman, J.T.; Jensen, T.; Sørensen, G.; Kvistholm Jensen, A.; Müller, L.; Nielsen, E.M.; Mølbak, K. Two listeria outbreaks caused by smoked fish consumption—Using whole-genome sequencing for outbreak investigations. Clin. Microbiol. Infect. 2016, 22, 620–624. [Google Scholar] [CrossRef]
- Løvdal, T. The microbiology of cold smoked salmon. Food Control 2015, 54, 360–373. [Google Scholar] [CrossRef]
- Dondero, M.; Cisternas, F.; Carvajal, L.; Simpson, R. Changes in quality of vacuum-packed cold-smoked salmon (Salmo salar) as a function of storage temperature. Food Chem. 2004, 87, 543–550. [Google Scholar] [CrossRef]
- Datta, S.; Janes, M.E.; Xue, Q.G.; Losso, J.; La Peyre, J.F. Control of Listeria monocytogenes and Salmonella anatum on the Surface of Smoked Salmon Coated with Calcium Alginate Coating containing Oyster Lysozyme and Nisin. J. Food Sci. 2008, 73, M67–M71. [Google Scholar] [CrossRef] [PubMed]
- Friesema, I.; de Jong, A.; Hofhuis, A.; Heck, M.; van den Kerkhof, H.; de Jonge, R.; Hameryck, D.; Nagel, K.; van Vilsteren, G.; van Beek, P.; et al. Large outbreak of Salmonella Thompson related to smoked salmon in the Netherlands, August to December 2012. Eurosurveillance 2014, 19, 20918. [Google Scholar] [CrossRef] [PubMed]
- Aymerich, T.; Rodríguez, M.; Garriga, M.; Bover-Cid, S. Assessment of the bioprotective potential of lactic acid bacteria against Listeria monocytogenes on vacuum-packed cold-smoked salmon stored at 8 °C. Food Microbiol. 2019, 83, 64–70. [Google Scholar] [CrossRef]
- Gómez-Guillén, M.C.; Gómez-Estaca, J.; Giménez, B.; Montero, P. Alternative fish species for cold-smoking process. Int. J. Food Sci. Technol. 2009, 44, 1525–1535. [Google Scholar] [CrossRef]
- Doe, P.E. Safety and Quality Issues in Fish Processing, 1st ed.; Bremner, H.A., Ed.; Woodhead Publishing in Food Science and Technology; Woodhead Publishing Limited: Cambridge, UK, 2002; Volume 1, p. 507. [Google Scholar]
- Bußler, S.; Herppich, W.B.; Neugart, S.; Schreiner, M.; Ehlbeck, J.; Rohn, S.; Schlüter, O. Impact of cold atmospheric pressure plasma on physiology and flavonol glycoside profile of peas (Pisum sativum ‘Salamanca’). Food Res. Int. 2015, 76 (Pt 1), 132–141. [Google Scholar] [CrossRef]
- Tóth, L.; Potthast, K. Chemical Aspects of the Smoking of Meat and Meat Products. In Advances in Food Research; Chichester, C.O., Mrak, E.M., Schweigert, B.S., Eds.; Academic Press: Cambridge, MA, USA, 1984; Volume 29, pp. 87–158. [Google Scholar]
- Foster, W.W.; Simpson, T.H.; Campbell, D. Studies of the smoking process for foods. II.—The rǒle of smoke particles. J. Sci. Food Agric. 1961, 12, 635–644. [Google Scholar] [CrossRef]
- Foster, W.W.; Simpson, T.H. Studies of the smoking process for foods. I.—The importance of vapours. J. Sci. Food Agric. 1961, 12, 363–374. [Google Scholar] [CrossRef]
- Durek, J.; Fröhling, A.; Bußler, S.; Hase, A.; Ehlbeck, J.; Schlüter, O.K. Pilot-scale generation of plasma processed air and its influence on microbial count, microbial diversity, and selected quality parameters of dried herbs. Innov. Food Sci. Emerg. Technol. 2022, 75, 102890. [Google Scholar] [CrossRef]
- Frohling, A.; Durek, J.; Schnabel, U.; Ehlbeck, J.; Bolling, J.; Schluter, O. Indirect plasma treatment of fresh pork: Decontamination efficiency and effects on quality attributes. Innov. Food Sci. Emerg. Technol. 2012, 16, 381–390. [Google Scholar] [CrossRef]
- Ehlbeck, J.; Schnabel, U.; Polak, M.; Winter, J.; von Woedtke, T.; Brandenburg, R.; von dem Hagen, T.; Weltmann, K.D. Low temperature atmospheric pressure plasma sources for microbial decontamination. J. Phys. D-Appl. Phys. 2011, 44, 013002. [Google Scholar] [CrossRef]
- Schnabel, U.; Schmidt, C.; Stachowiak, J.; Bösel, A.; Andrasch, M.; Ehlbeck, J. Plasma processed air for biological decontamination of PET and fresh plant tissue. Plasma Process. Polym. 2018, 15, 1600057. [Google Scholar] [CrossRef]
- Schnabel, U.; Niquet, R.; Schlüter, O.; Gniffke, H.; Ehlbeck, E. Decontamination and sensory properties of microbiologically contaminated fresh fruits and vegetables by microwave plasma processed air (PPA). J. Food Process. Preserv. 2014, 39, 653–662. [Google Scholar] [CrossRef]
- Andrasch, M.; Stachowiak, J.; Schlüter, O.; Schnabel, U.; Ehlbeck, J. Scale-up to pilot plant dimensions of plasma processed water generation for fresh-cut lettuce treatment. Food Packag. Shelf Life 2017, 14, 40–45. [Google Scholar] [CrossRef]
- Pathare, P.B.; Opara, U.L.; Al-Said, F.A.-J. Colour Measurement and Analysis in Fresh and Processed Foods: A Review. Food Bioprocess Technol. 2013, 6, 36–60. [Google Scholar] [CrossRef]
- Limpert, E.; Stahel, W.A.; Abbt, M. Log-normal Distributions across the Sciences: Keys and Clues: On the charms of statistics, and how mechanical models resembling gambling machines offer a link to a handy way to characterize log-normal distributions, which can provide deeper insight into variability and probability—Normal or log-normal: That is the question. Bioscience 2001, 51, 341–352. [Google Scholar] [CrossRef]
- Egerton, F.N. Modeling Nature: Episodes in the History of Population Ecology. Sharon E. Kingsland. Isis 1986, 77, 355. [Google Scholar] [CrossRef]
- R Core Team (Ed.) R: A Language and Environment for Statistical Computing. In R Foundation for Statistical Computing; R Core Team: Vienna, Austria, 2017. [Google Scholar]
- Tserkezou, I.S.A.a.P. Seafood Processing: Technology, Quality and Safety; Boziaris, I.S., Ed.; Wiley Blackwell: Oxford, UK, 2014; Volume 1, pp. 83–128. [Google Scholar]
- Gysemans, K.P.M.; Bernaerts, K.; Vermeulen, A.; Geeraerd, A.H.; Debevere, J.; Devlieghere, F.; Van Impe, J.F. Exploring the performance of logistic regression model types on growth/no growth data of Listeria monocytogenes. Int. J. Food Microbiol. 2007, 114, 316–331. [Google Scholar] [CrossRef]
- Sprouffske, K.; Wagner, A. Growthcurver: An R package for obtaining interpretable metrics from microbial growth curves. BMC Bioinform. 2016, 17, 172. [Google Scholar] [CrossRef]
- Madigan, M.T.; Bender, K.S.; Buckley, D.H.; Sattley, W.M.; Stahl, D.A. Brock-Microbiology, 15th ed.; Pearson: London, UK, 2020; Volume 15, p. 1440. [Google Scholar]
- Giménez, B.; Dalgaard, P. Modelling and predicting the simultaneous growth of Listeria monocytogenes and spoilage micro-organisms in cold-smoked salmon. J. Appl. Microbiol. 2004, 96, 96–109. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, L.V.; Huss, H.H.; Dalgaard, P. The effect of biogenic amine production by single bacterial cultures and metabiosis on cold-smoked salmon. J. Appl. Microbiol. 2000, 89, 920–934. [Google Scholar] [CrossRef]
- Macé, S.; Joffraud, J.-J.; Cardinal, M.; Malcheva, M.; Cornet, J.; Lalanne, V.; Chevalier, F.; Sérot, T.; Pilet, M.-F.; Dousset, X. Evaluation of the spoilage potential of bacteria isolated from spoiled raw salmon (Salmo salar) fillets stored under modified atmosphere packaging. Int. J. Food Microbiol. 2013, 160, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Joffraud, J.-J.; Cardinal, M.; Cornet, J.; Chasles, J.-S.; Léon, S.; Gigout, F.; Leroi, F. Effect of bacterial interactions on the spoilage of cold-smoked salmon. Int. J. Food Microbiol. 2006, 112, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Ekonomou, S.I.; Parlapani, F.F.; Kyritsi, M.; Hadjichristodoulou, C.; Boziaris, I.S. Preservation status and microbial communities of vacuum-packed hot smoked rainbow trout fillets. Food Microbiol. 2022, 103, 103959. [Google Scholar] [CrossRef]
- Stohr, V.; Joffraud, J.J.; Cardinal, M.; Leroi, F. Spoilage potential and sensory profile associated with bacteria isolated from cold-smoked salmon. Food Res. Int. 2001, 34, 797–806. [Google Scholar] [CrossRef]
- Jérôme, M.; Passerini, D.; Chevalier, F.; Marchand, L.; Leroi, F.; Macé, S. Development of a rapid qPCR method to quantify lactic acid bacteria in cold-smoked salmon. Int. J. Food Microbiol. 2022, 363, 109504. [Google Scholar] [CrossRef]
- Joffraud, J.J.; Leroi, F.; Roy, C.; Berdagué, J.L. Characterisation of volatile compounds produced by bacteria isolated from the spoilage flora of cold-smoked salmon. Int. J. Food Microbiol. 2001, 66, 175–184. [Google Scholar] [CrossRef]
- Maillet, A.; Denojean, P.; Bouju-Albert, A.; Scaon, E.; Leuillet, S.; Dousset, X.; Jaffrès, E.; Combrisson, J.; Prévost, H. Characterization of Bacterial Communities of Cold-Smoked Salmon during Storage. Foods 2021, 10, 362. [Google Scholar] [CrossRef]
- Macé, S.; Cornet, J.; Chevalier, F.; Cardinal, M.; Pilet, M.-F.; Dousset, X.; Joffraud, J.-J. Characterisation of the spoilage microbiota in raw salmon (Salmo salar) steaks stored under vacuum or modified atmosphere packaging combining conventional methods and PCR–TTGE. Food Microbiol. 2012, 30, 164–172. [Google Scholar] [CrossRef]
- Mikš-Krajnik, M.; Yoon, Y.-J.; Ukuku, D.O.; Yuk, H.-G. Volatile chemical spoilage indexes of raw Atlantic salmon (Salmo salar) stored under aerobic condition in relation to microbiological and sensory shelf lives. Food Microbiol. 2016, 53, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Møretrø, T.; Moen, B.; Heir, E.; Hansen, A.Å.; Langsrud, S. Contamination of salmon fillets and processing plants with spoilage bacteria. Int. J. Food Microbiol. 2016, 237, 98–108. [Google Scholar] [CrossRef] [PubMed]
- Uren Webster, T.M.; Rodriguez-Barreto, D.; Castaldo, G.; Gough, P.; Consuegra, S.; Garcia de Leaniz, C. Environmental plasticity and colonisation history in the Atlantic salmon microbiome: A translocation experiment. Mol. Ecol. 2020, 29, 886–898. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.S.; Chatterjee, R.N.; Raju, M.V.; Prakash, B.; Rama Rao, S.V.; Yadav, S.P.; Kannan, A. Gut Microbial Composition Differs Extensively among Indian Native Chicken Breeds Originated in Different Geographical Locations and a Commercial Broiler Line, but Breed-Specific, as Well as Across-Breed Core Microbiomes, Are Found. Microorganisms 2021, 9, 391. [Google Scholar] [CrossRef] [PubMed]
- Ricke, S.C.; Lee, S.I.; Kim, S.A.; Park, S.H.; Shi, Z. Prebiotics and the poultry gastrointestinal tract microbiome. Poult. Sci. 2020, 99, 670–677. [Google Scholar] [CrossRef]
- Ramamurthy, T.; Ghosh, A.; Pazhani, G.P.; Shinoda, S. Current Perspectives on Viable but Non-Culturable (VBNC) Pathogenic Bacteria. Front. Public Health 2014, 2, 103. [Google Scholar] [CrossRef]
- Xin, X.; Dell, K.; Udugama, I.A.; Young, B.R.; Baroutian, S. Transforming biomass pyrolysis technologies to produce liquid smoke food flavouring. J. Clean. Prod. 2021, 294, 125368. [Google Scholar] [CrossRef]
- Liao, X.; Hu, W.; Liu, D.; Ding, T.; Björkroth, J. Stress Resistance and Pathogenicity of Nonthermal-Plasma-Induced Viable-but-Nonculturable Staphylococcus aureus through Energy Suppression, Oxidative Stress Defense, and Immune-Escape Mechanisms. Appl. Environ. Microbiol. 2021, 87, e02380-20. [Google Scholar] [CrossRef] [PubMed]
- Magee, P.N.; Barnes, J.M. Carcinogenic Nitroso Compounds. In Advances in Cancer Research; Haddow, A., Weinhouse, S., Eds.; Academic Press: Cambridge, MA, USA, 1967; Volume 10, pp. 163–246. [Google Scholar]
- Abd Hamid, N.F.H.; Khan, M.M.; Lim, L.H. Assessment of nitrate, nitrite and chloride in selected cured meat products and their exposure to school children in Brunei Darussalam. J. Food Compos. Anal. 2020, 91, 103520. [Google Scholar] [CrossRef]
- Gough, T.A.; McPhail, M.F.; Webb, K.S.; Wood, B.J.; Coleman, R.F. An examination of some foodstuffs for the presence of volatile nitrosamines. J. Sci. Food Agric. 1977, 28, 345–351. [Google Scholar] [CrossRef]
- Iko Afé, O.H.; Kpoclou, Y.E.; Douny, C.; Anihouvi, V.B.; Igout, A.; Mahillon, J.; Hounhouigan, D.J.; Scippo, M.-L. Chemical hazards in smoked meat and fish. Food Sci. Nutr. 2021, 9, 6903–6922. [Google Scholar] [CrossRef] [PubMed]
- Gangolli, S.D.; van den Brandt, P.A.; Feron, V.J.; Janzowsky, C.; Koeman, J.H.; Speijers, G.J.A.; Spiegelhalder, B.; Walker, R.; Wishnok, J.S. Nitrate, nitrite and N-nitroso compounds. Eur. J. Pharmacol. Environ. Toxicol. Pharmacol. 1994, 292, 1–38. [Google Scholar] [CrossRef]
- Genualdi, S.; Matos, M.P.V.; Mangrum, J.B.; DeJager, L. Investigation into the Concentrations and Sources of Nitrates and Nitrites in Milk and Plant-Based Powders. J. Agric. Food Chem. 2020, 68, 1725–1730. [Google Scholar] [CrossRef] [PubMed]
- Blunden, S.; Wallace, T. Tin in canned food: A review and understanding of occurrence and effect. Food Chem. Toxicol. 2003, 41, 1651–1662. [Google Scholar] [CrossRef]
- Schnabel, U.; Handorf, O.; Yarova, K.; Zessin, B.; Zechlin, S.; Sydow, D.; Zellmer, E.; Stachowiak, J.; Andrasch, M.; Below, H.; et al. Plasma-Treated Air and Water—Assessment of Synergistic Antimicrobial Effects for Sanitation of Food Processing Surfaces and Environment. Foods 2019, 8, 55. [Google Scholar] [CrossRef]
- Hultmann, L.; Bencze Rørå, A.M.; Steinsland, I.; Skåra, T.; Rustad, T. Proteolytic activity and properties of proteins in smoked salmon (Salmo salar)—Effects of smoking temperature. Food Chem. 2004, 85, 377–387. [Google Scholar] [CrossRef]
- Bjørnevik, M.; Cardinal, M.; Vallet, J.-L.; Nicolaisen, O.; Arnarson, G.Ö. Effect of salting and cold-smoking procedures on Atlantic salmon originating from pre-or post rigor filleted raw material. Based on the measurement of physiochemical characteristics. LWT 2018, 91, 431–438. [Google Scholar] [CrossRef]
- Mora, M.; Fàbregas, E.; Céspedes, F.; Bartrolí, J.; Puy, N. Production and separation of value-added compounds from pine wood using pyrolysis and biorefinery techniques. Fuel Process. Technol. 2022, 238, 107509. [Google Scholar] [CrossRef]
- Sérot, T.; Baron, R.; Knockaert, C.; Vallet, J.L. Effect of smoking processes on the contents of 10 major phenolic compounds in smoked fillets of herring (Cuplea harengus). Food Chem. 2004, 85, 111–120. [Google Scholar] [CrossRef]
- Yang, Z.; Wang, H.; Wang, W.; Qi, W.; Yue, L.; Ye, Q. Effect of 10MeV E-beam irradiation combined with vacuum-packaging on the shelf life of Atlantic salmon fillets during storage at 4 °C. Food Chem. 2014, 145, 535–541. [Google Scholar] [CrossRef]
- Albert, T.; Braun, P.G.; Saffaf, J.; Wiacek, C. Physical Methods for the Decontamination of Meat Surfaces. Curr. Clin. Microbiol. Rep. 2021, 8, 9–20. [Google Scholar] [CrossRef]
- Keymanesh, K.; Soltani, S.; Sardari, S. Application of antimicrobial peptides in agriculture and food industry. World J. Microbiol. Biotechnol. 2009, 25, 933–944. [Google Scholar] [CrossRef]
- Yildirim, S.; Röcker, B.; Pettersen, M.K.; Nilsen-Nygaard, J.; Ayhan, Z.; Rutkaite, R.; Radusin, T.; Suminska, P.; Marcos, B.; Coma, V. Active Packaging Applications for Food. Compr. Rev. Food Sci. Food Saf. 2018, 17, 165–199. [Google Scholar] [CrossRef] [PubMed]
- Xuan Nguyen, N.T.; Daniel, P.; Pilard, J.-F.; Cariou, R.; Gigout, F.; Leroi, F. Antibacterial activity of plasma-treated polypropylene membrane functionalized with living Carnobacterium divergens in cold-smoked salmon. Food Control 2022, 137, 108903. [Google Scholar] [CrossRef]
- Bekhit, A.E.-D.A.; Holman, B.W.B.; Giteru, S.G.; Hopkins, D.L. Total volatile basic nitrogen (TVB-N) and its role in meat spoilage: A review. Trends Food Sci. Technol. 2021, 109, 280–302. [Google Scholar] [CrossRef]
- Erkan, N.; Üretener, G.; Alpas, H.; Selçuk, A.; Özden, Ö.; Buzrul, S. The effect of different high pressure conditions on the quality and shelf life of cold smoked fish. Innov. Food Sci. Emerg. Technol. 2011, 12, 104–110. [Google Scholar] [CrossRef]
- Truong, B.Q.; Buckow, R.; Stathopoulos, C.E.; Nguyen, M.H. Advances in High-Pressure Processing of Fish Muscles. Food Eng. Rev. 2015, 7, 109–129. [Google Scholar] [CrossRef]
- Oliveira, F.A.d.; Neto, O.C.; Santos, L.M.R.d.; Ferreira, E.H.R.; Rosenthal, A. Effect of high pressure on fish meat quality—A review. Trends Food Sci. Technol. 2017, 66, 1–19. [Google Scholar] [CrossRef]
- Quevedo, R.A.; Aguilera, J.M.; Pedreschi, F. Color of Salmon Fillets By Computer Vision and Sensory Panel. Food Bioprocess Technol. 2010, 3, 637–643. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weihe, T.; Wagner, R.; Schnabel, U.; Andrasch, M.; Su, Y.; Stachowiak, J.; Noll, H.J.; Ehlbeck, J. Microbial Control of Raw and Cold-Smoked Atlantic Salmon (Salmo salar) through a Microwave Plasma Treatment. Foods 2022, 11, 3356. https://doi.org/10.3390/foods11213356
Weihe T, Wagner R, Schnabel U, Andrasch M, Su Y, Stachowiak J, Noll HJ, Ehlbeck J. Microbial Control of Raw and Cold-Smoked Atlantic Salmon (Salmo salar) through a Microwave Plasma Treatment. Foods. 2022; 11(21):3356. https://doi.org/10.3390/foods11213356
Chicago/Turabian StyleWeihe, Thomas, Robert Wagner, Uta Schnabel, Mathias Andrasch, Yukun Su, Jörg Stachowiak, Heinz Jörg Noll, and Jörg Ehlbeck. 2022. "Microbial Control of Raw and Cold-Smoked Atlantic Salmon (Salmo salar) through a Microwave Plasma Treatment" Foods 11, no. 21: 3356. https://doi.org/10.3390/foods11213356
APA StyleWeihe, T., Wagner, R., Schnabel, U., Andrasch, M., Su, Y., Stachowiak, J., Noll, H. J., & Ehlbeck, J. (2022). Microbial Control of Raw and Cold-Smoked Atlantic Salmon (Salmo salar) through a Microwave Plasma Treatment. Foods, 11(21), 3356. https://doi.org/10.3390/foods11213356







