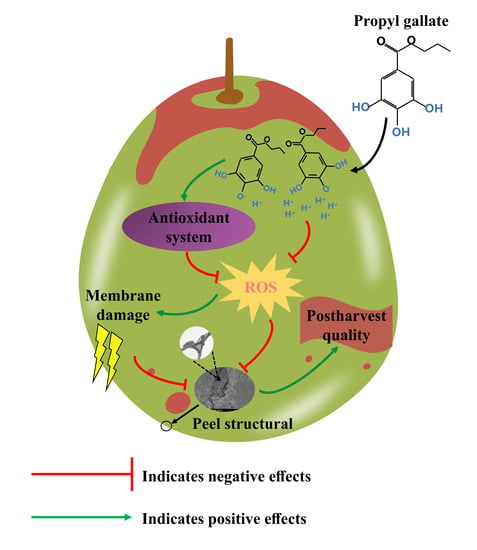
Propyl Gallate Treatment Improves the Postharvest Quality of Winter Jujube (Zizyphus jujuba Mill. cv. Dongzao) by Regulating Antioxidant Metabolism and Maintaining the Structure of Peel
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Postharvest Treatments
2.3. Reddening Index, Reddish Peel Area, Chromatic Value, Firmness, Total Soluble Solids (TSS), and Titratable Acid (TA)
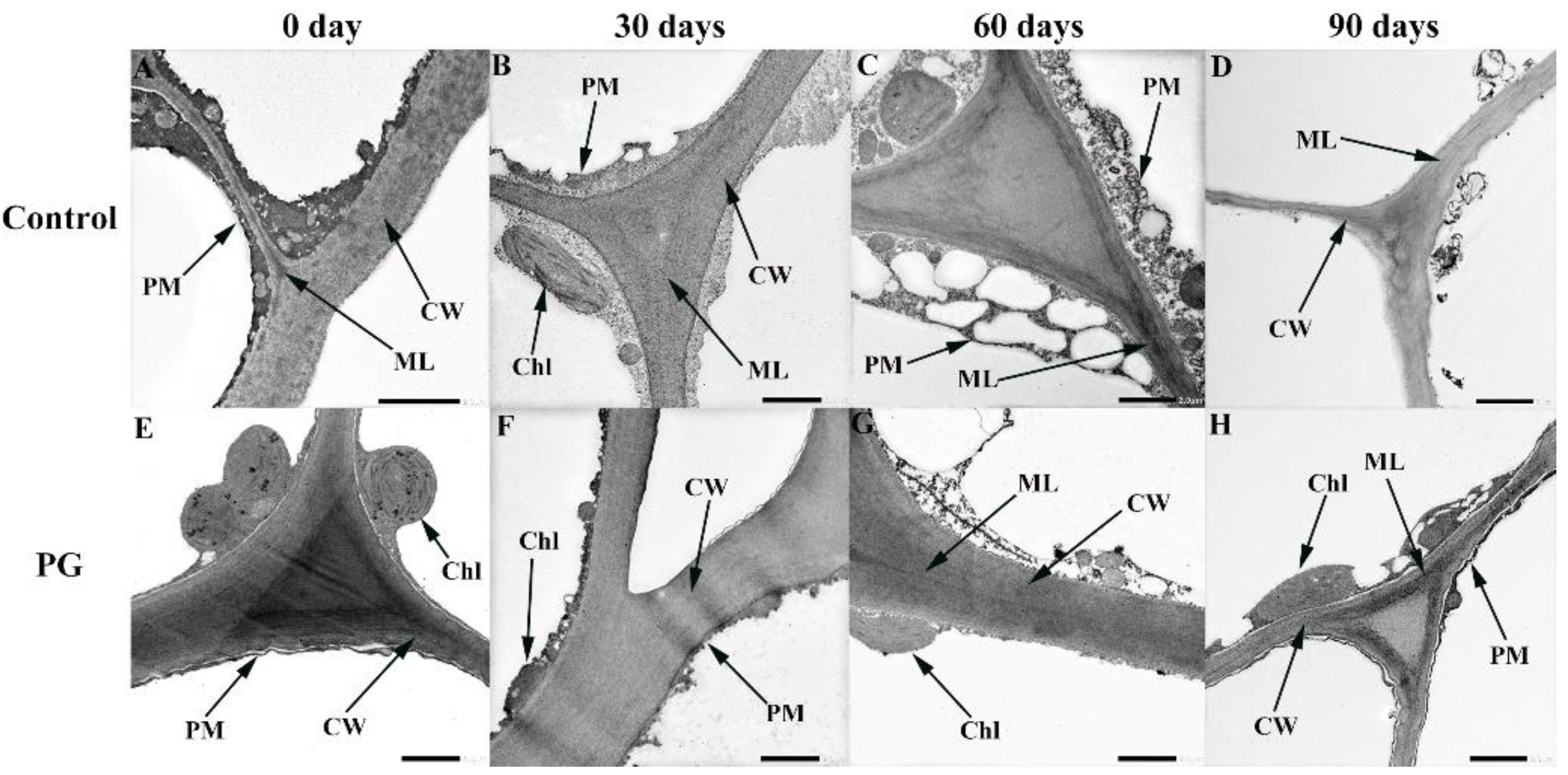
2.4. The Microstructure and Ultrastructure of Winter Jujube Peel
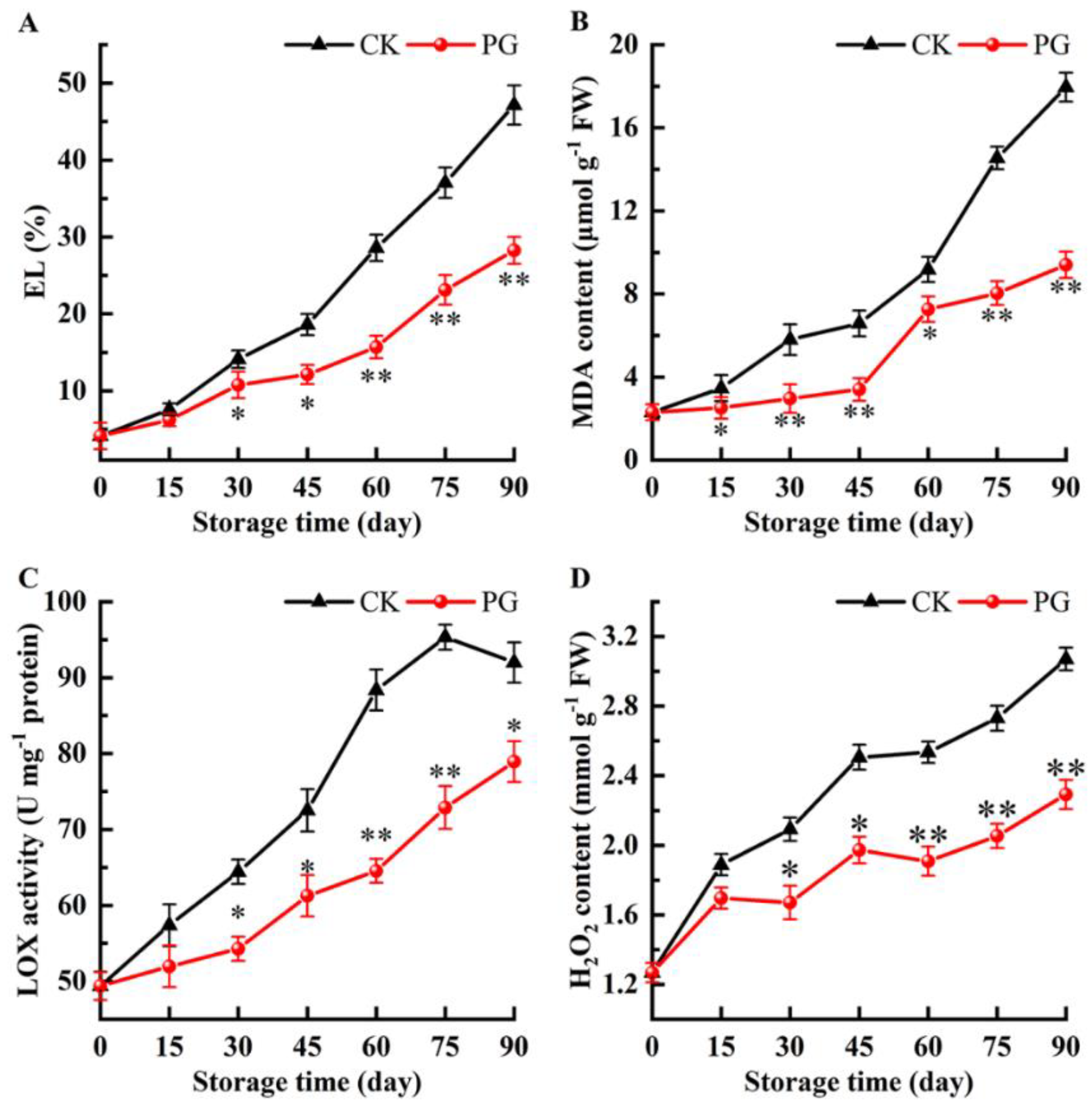
2.5. Electrolyte Leakage (EL), MDA, Lipoxygenase (LOX) and H2O2
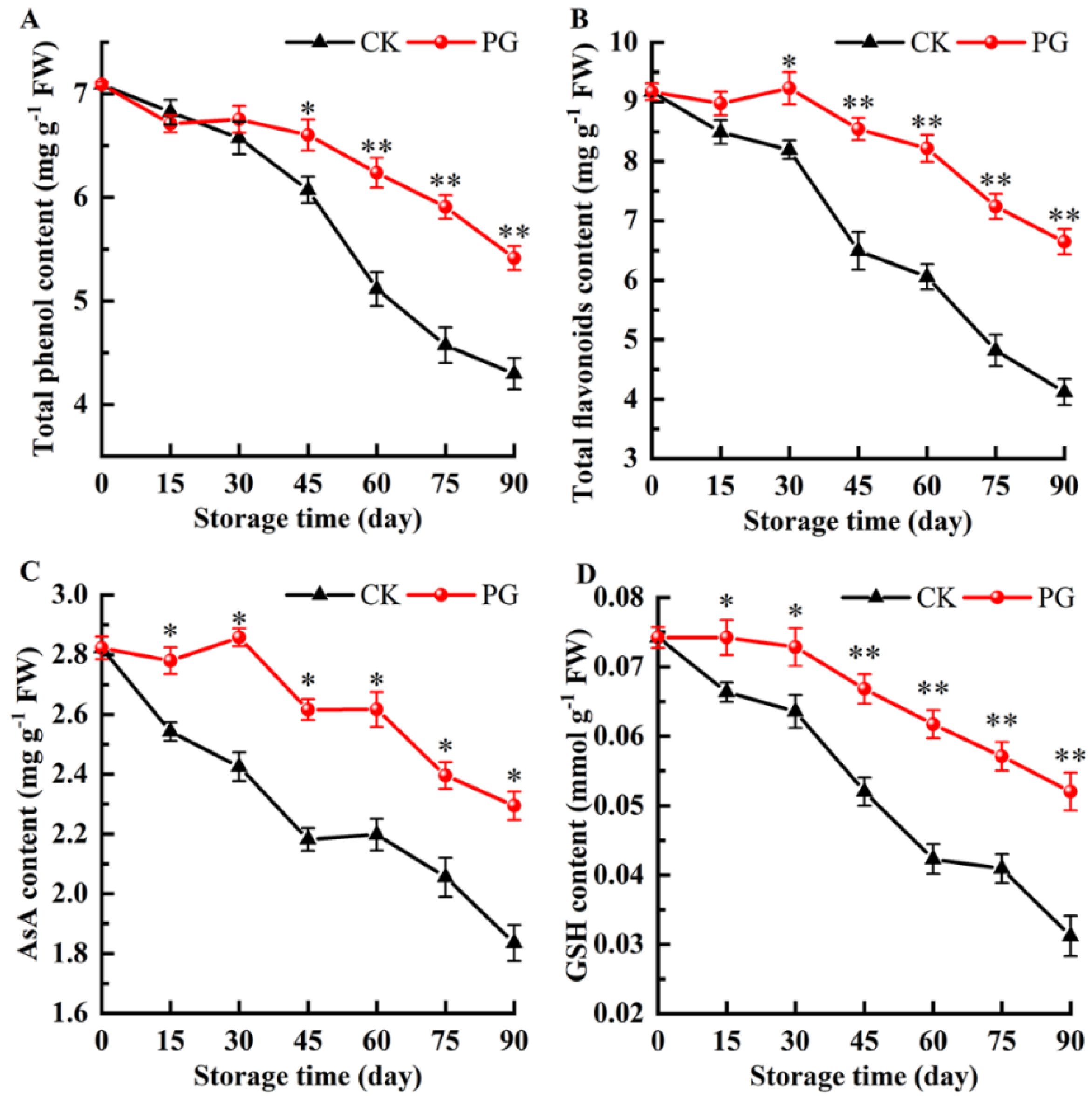
2.6. Total Phenols, Total Flavonoids, Ascorbic Acid (AsA), and Reduced Glutathione (GSH)
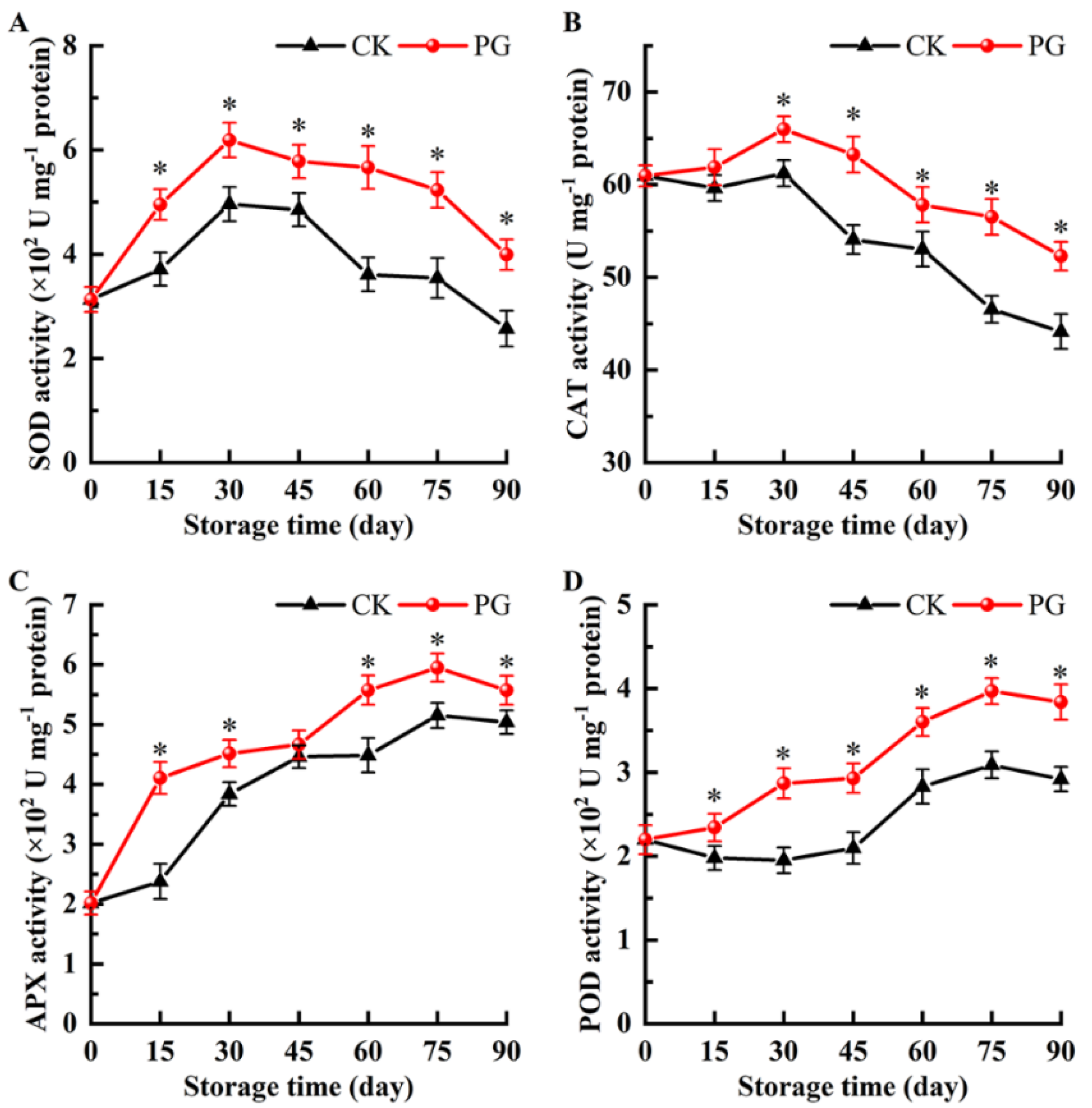
2.7. Superoxide Dismutase (SOD), Catalase (CAT), Ascorbate Peroxidase (APX), and Peroxidase (POD) Activity
2.8. Statistical Analysis
3. Results
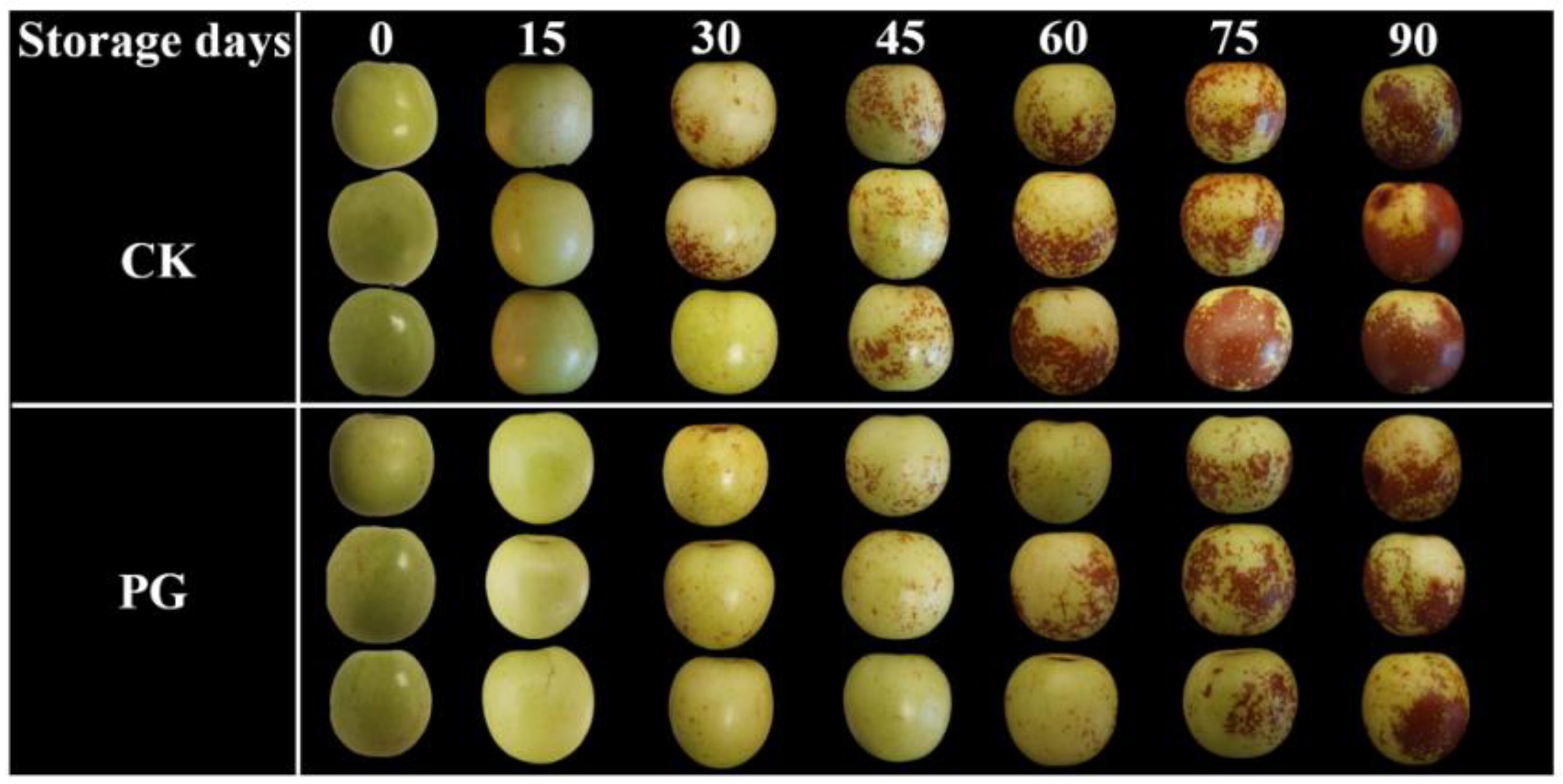
3.1. Changes of Visual Appearance, Reddish Peel Area, and Chromatic Value in the Winter Jujube Fruit after PG Treatment
3.2. Results of Two-Way Analysis of ANOVA
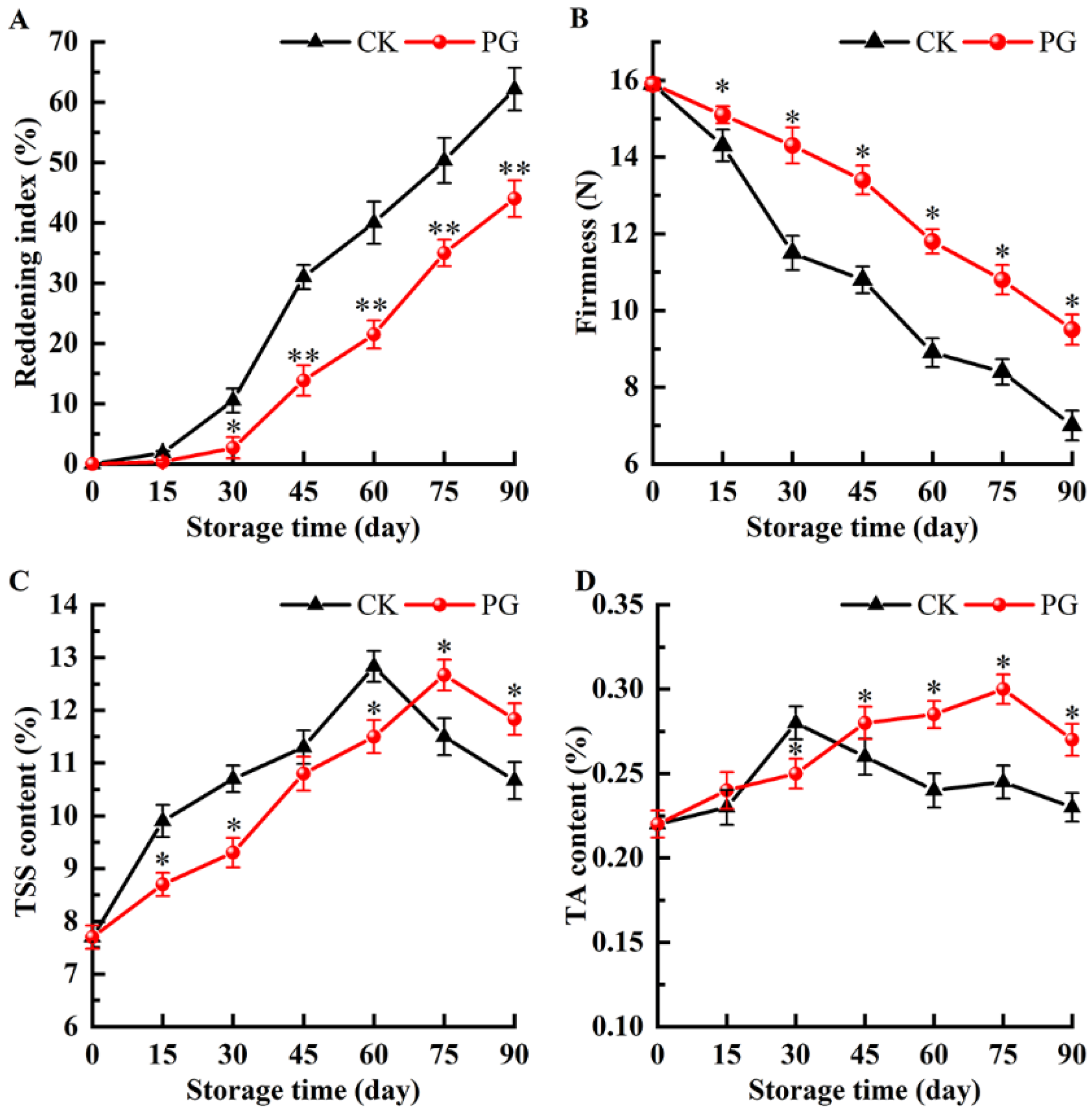
3.3. Changes of Reddening Index, Firmness, TSS, and TA in the Winter Jujube Fruit after PG Treatment
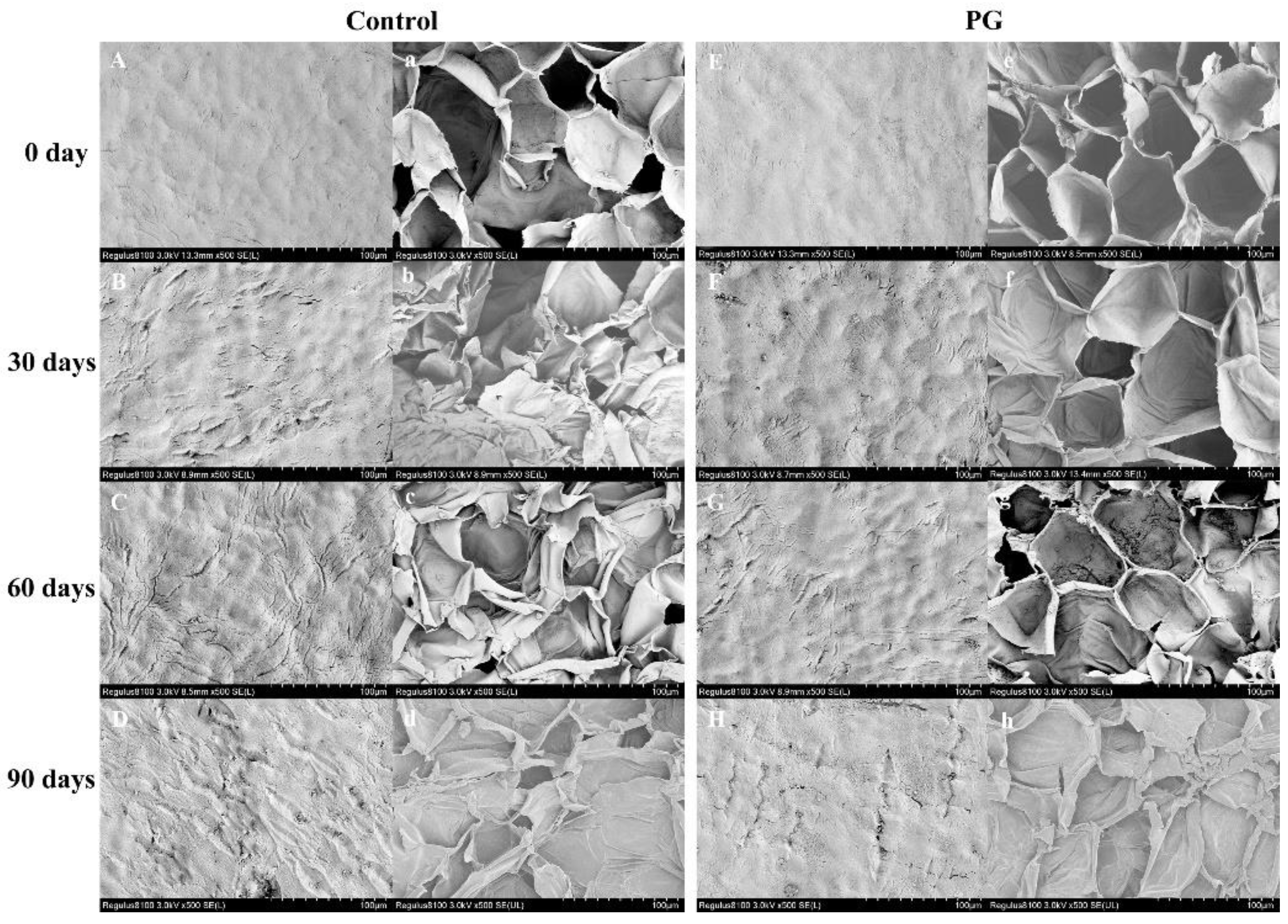
3.4. Changes of Microstructure and Ultrastructure in the Winter Jujube Peel after PG Treatment
3.5. Changes of EL, H2O2, LOX, and MDA in the Winter Jujube Peel after PG Treatment
3.6. Changes of Total Phenols, Total Flavonoids, AsA, and GSH in the Winter Jujube Peel after PG Treatment
3.7. Changes of the Activity of SOD, CAT, APX, and POD in the Winter Jujube Peel after PG Treatment
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sun, L.N.; Liu, M.C.; Zhu, S.H.; Zhou, J.; Wang, M.L. Effect of Nitric Oxide on Alcoholic Fermentation and Qualities of Chinese Winter Jujube During Storage. Agric. Sci. China 2007, 7, 849–856. [Google Scholar] [CrossRef]
- Zhang, L.H.; Li, S.F.; Dong, Y.; Zhi, H.H.; Zong, W. Tea polyphenols incorporated into alginate-based edible coating for quality maintenance of Chinese winter jujube under ambient temperature. Lwt-Food Sci. Technol. 2016, 70, 155–161. [Google Scholar] [CrossRef]
- Zhao, Y.T.; Zhu, X.; Hou, Y.Y.; Wang, X.Y.; Li, X.H. Postharvest nitric oxide treatment delays the senescence of winter jujube (Zizyphus jujuba Mill. cv. Dongzao) fruit during cold storage by regulating reactive oxygen species metabolism. Sci. Hortic. 2020, 261, 109009. [Google Scholar] [CrossRef]
- Cheng, S.B.; Yu, Y.; Guo, J.Y.; Chen, G.G.; Guo, M.R. Effect of 1-methylcyclopropene and chitosan treatment on the storage quality of jujube fruit and its related enzyme activities. Sci. Hortic. 2020, 265, 109281. [Google Scholar] [CrossRef]
- Kou, X.H.; He, Y.L.; Li, Y.F.; Chen, X.Y.; Feng, Y.C.; Xue, Z.H. Effect of abscisic acid (ABA) and chitosan/nano-silica/sodium alginate composite film on the color development and quality of postharvest Chinese winter jujube (Zizyphus jujuba Mill. cv. Dongzao). Food Chem. 2019, 270, 385–394. [Google Scholar] [CrossRef]
- Wang, L.; Shan, T.M.; Xie, B.; Ling, C.; Shao, S.; Jin, P.; Zheng, Y.H. Glycine betaine reduces chilling injury in peach fruit by enhancing phenolic and sugar metabolisms. Food Chem. 2019, 272, 530–538. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, J.W.; Zhou, B.; Li, G.D.; Liu, Y.F.; Xia, X.L.; Xiao, Z.G.; Lu, F.; Ji, S.J. Calcium inhibited peel browning by regulating enzymes in membrane metabolism of ‘Nanguo’ pears during post-ripeness after refrigerated storage. Sci. Hortic. 2019, 244, 15–21. [Google Scholar] [CrossRef]
- Fan, M.C.; Li, W.X.; Hu, X.L.; Sun, Y.; Yu, G.; Zhang, X. Effect of micro-vacuum storage on active oxygen metabolism, internal browning and related enzyme activities in Laiyang pear (Pyrus bretschneideri Reld). Lwt-Food Sci. Technol. 2016, 72, 467–474. [Google Scholar] [CrossRef]
- Kou, X.H.; Chai, L.P.; Yang, S.; He, Y.L.; Wu, C.E.; Liu, Y.F.; Zhou, J.Q.; Xue, Z.H.; Wang, Z.Z. Physiological and metabolic analysis of winter jujube after postharvest treatment with calcium chloride and a composite film. J. Sci. Food Agric. 2021, 101, 703–717. [Google Scholar] [CrossRef]
- Lin, Y.F.; Hu, Y.H.; Lin, H.T.; Liu, X.; Chen, Y.H.; Zhang, S.; Chen, Q.X. Inhibitory Effects of Propyl Gallate on Tyrosinase and Its Application in Controlling Pericarp Browning of Harvested Longan Fruits. J. Agric. Food Chem. 2013, 61, 2889–2895. [Google Scholar] [CrossRef]
- Lin, Y.F.; Lin, Y.X.; Lin, H.T.; Zhang, S.; Chen, Y.H.; Shi, J. Inhibitory effects of propyl gallate on browning and its relationship to active oxygen metabolism in pericarp of harvested longan fruit. Lwt-Food Sci. Technol. 2015, 60, 1122–1128. [Google Scholar] [CrossRef]
- Lin, Y.F.; Lin, Y.X.; Lin, H.T.; Shi, J.; Chen, Y.H.; Wang, H. Inhibitory effects of propyl gallate on membrane lipids metabolism and its relation to increasing storability of harvested longan fruit. Food Chem. 2017, 217, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Sogvar, O.B.; Saba, M.K.; Emamifar, A. Aloe vera and ascorbic acid coatings maintain postharvest quality and reduce microbial load of strawberry fruit. Postharvest Biol. Technol. 2016, 114, 29–35. [Google Scholar] [CrossRef]
- Kaur, R.; Kaur, N.; Singh, H. Pericarp and pedicel anatomy in relation to fruit cracking in lemon (Citrus limon L Burm.). Sci. Hortic. 2019, 246, 462–468. [Google Scholar] [CrossRef]
- Zhao, Y.T.; Zhu, X.; Hou, Y.Y.; Wang, X.Y.; Li, X.H. Effects of nitric oxide fumigation treatment on retarding cell wall degradation and delaying softening of winter jujube (Ziziphus jujuba Mill. cv. Dongzao) fruit during storage. Postharvest Biol. Technol. 2019, 156, 110954. [Google Scholar] [CrossRef]
- Liu, H.; Song, L.L.; You, Y.L.; Li, Y.B.; Duan, X.W.; Jiang, Y.M.; Joyce, D.C.; Ashraf, M.; Lu, W.J. Cold storage duration affects litchi fruit quality, membrane permeability, enzyme activities and energy charge during shelf time at ambient temperature. Postharvest Biol. Technol. 2011, 60, 24–30. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, K.S.; Zhang, S.L.; Ferguson, I. The role of salicylic acid in postharvest ripening of kiwifruit. Postharvest Biol. Technol. 2003, 28, 67–74. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Zhang, Z.; Xu, J.; Chen, Y.; Wei, J.; Wu, B. Nitric oxide treatment maintains postharvest quality of table grapes by mitigation of oxidative damage. Postharvest Biol. Technol. 2019, 152, 9–18. [Google Scholar] [CrossRef]
- Xu, F.X.; Liu, S.Y.; Liu, Y.F.; Xu, Q.; Wang, S.H. The combined effect of ultraviolet-C irradiation and lysozyme coatings treatment on control of brown heart in Huangguan pears. Sci. Hortic. 2019, 256, 108634. [Google Scholar] [CrossRef]
- Wootton-Beard, P.C.; Moran, A.; Ryan, L. Stability of the total antioxidant capacity and total polyphenol content of 23 commercially available vegetable juices before and after in vitro digestion measured by FRAP, DPPH, ABTS and Folin-Ciocalteu methods. Food Res. Int. 2011, 44, 217–224. [Google Scholar] [CrossRef]
- Kou, X.H.; Chen, Q.; Li, X.H.; Li, M.F.; Kan, C.; Chen, B.R.; Zhang, Y.; Xue, Z.H. Quantitative assessment of bioactive compounds and the antioxidant activity of 15 jujube cultivars. Food Chem. 2015, 173, 1037–1044. [Google Scholar] [CrossRef]
- Ge, Y.H.; Bi, Y.; Guest, D.I. Defence responses in leaves of resistant and susceptible melon (Cucumis melo L.) cultivars infected with Colletotrichum lagenarium. Physiol. Mol. Plant Pathol. 2013, 81, 13–21. [Google Scholar] [CrossRef]
- Kou, X.; Wu, M.; Li, L.; Wang, S.; Xue, Z.; Liu, B.; Fei, Y. Effects of CaCl2 dipping and pullulan coating on the development of brown spot on ‘Huangguan’ pears during cold storage. Postharvest Biol. Technol. 2015, 99, 63–72. [Google Scholar] [CrossRef]
- Zhang, J.H.; Li, C.Y.; Wei, M.L.; Ge, Y.H.; Tang, Q.; Xue, W.J.; Zhang, S.Y.; Wang, W.H.; Lv, J.Y. Effects of trisodium phosphate treatment after harvest on storage quality and sucrose metabolism in jujube fruit. J. Sci. Food Agric. 2019, 99, 5526–5532. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, F.; Noguera-Artiaga, L.; Burlo, F.; Wojdylo, A.; Carbonell-Barrachina, A.A.; Legua, P. Physico-chemical, nutritional, and volatile composition and sensory profile of Spanish jujube (Ziziphus jujuba Mill.) fruits. J. Sci. Food Agric. 2016, 96, 2682–2691. [Google Scholar] [CrossRef] [PubMed]
- Juan, X.U.; Zhang, X.L.; Peng, L.I.; Jiang, L.I.; Chun-Sheng, L.U. Effects of Propyl Gallate Treatment on the Fruit Quality Related Indexes of Korla Fragrant Pear during the Storage Period. Xinjiang Agric. Sci. 2016, 53, 125–134. [Google Scholar]
- Cao, Z.; Lulu, Y.U.; Fei, X.U. Effect of N-propyl Gallate Treatment on Postharvest Storage of Thin-skinned Melon (Cucumis melo L.). North Hortic. 2016, 5, 148–152. [Google Scholar]
- Bordoloi, A.; Kaur, L.; Singh, J. Parenchyma cell microstructure and textural characteristics of raw and cooked potatoes. Food Chem. 2012, 133, 1092–1100. [Google Scholar] [CrossRef]
- Alquezar, B.; Mesejo, C.; Alferez, F.; Agusti, M.; Zacarias, L. Morphological and ultrastructural changes in peel of ‘Navelate’ oranges in relation to variations in relative humidity during postharvest storage and development of peel pitting. Postharvest Biol. Technol. 2010, 56, 163–170. [Google Scholar] [CrossRef]
- Pietrysiak, E.; Ganjyal, G.M. Apple peel morphology and attachment of Listeria innocua through aqueous environment as shown by scanning electron microscopy. Food Control 2018, 92, 362–369. [Google Scholar] [CrossRef]
- Tamer, C.; Isci, A.; Kutlu, N.; Sakiyan, O.; Sahin, S.; Sumnu, G. Effect of Drying on Porous Characteristics of Orange Peel. Int. J. Food Eng. 2016, 12, 921–928. [Google Scholar] [CrossRef]
- Khaliq, G.; Mohamed, M.T.M.; Ghazali, H.M.; Ding, P.; Ali, A. Influence of gum arabic coating enriched with calcium chloride on physiological, biochemical and quality responses of mango (Mangifera indica L.) fruit stored under low temperature stress. Postharvest Biol. Technol. 2016, 111, 362–369. [Google Scholar] [CrossRef]
- Sun, H.J.; Luo, M.L.; Zhou, X.; Zhou, Q.; Ji, S.J. Influence of Melatonin Treatment on Peel Browning of Cold-Stored “Nanguo” Pears. Food Bioprocess Technol. 2020, 13, 1478–1490. [Google Scholar] [CrossRef]
- Huang, Q.H.; Qian, X.C.; Jiang, T.J.; Zheng, X.L. Effect of chitosan and guar gum based composite edible coating on quality of mushroom (Lentinus edodes) during postharvest storage. Sci. Hortic. 2019, 253, 382–389. [Google Scholar] [CrossRef]
- Zhang, Y.; Jin, P.; Huang, Y.P.; Shan, T.M.; Wang, L.; Li, Y.Y.; Zheng, Y.H. Effect of hot water combined with glycine betaine alleviates chilling injury in cold-stored loquat fruit. Postharvest Biol. Technol. 2016, 118, 141–147. [Google Scholar] [CrossRef]
- Li, T.T.; Shi, D.D.; Wu, Q.X.; Zhang, Z.K.; Qu, H.X.; Jiang, Y.M. Sodium para-aminosalicylate delays pericarp browning of litchi fruit by inhibiting ROS-mediated senescence during postharvest storage. Food Chem. 2019, 278, 552–559. [Google Scholar] [CrossRef]
- Sun, H.; Luo, M.L.; Zhou, X.; Zhou, Q.; Sun, Y.Y.; Ge, W.Y.; Wei, B.D.; Cheng, S.C.; Ji, S.J. Exogenous glycine betaine treatment alleviates low temperature-induced pericarp browning of ‘Nanguo’ pears by regulating antioxidant enzymes and proline metabolism. Food Chem. 2020, 306, 125626. [Google Scholar] [CrossRef]
- Ge, Y.; Duan, B.; Li, C.; Qi, T.; Xue, L.; Wei, M.; Chen, Y.; Li, J. γ-Aminobutyric acid delays senescence of blueberry fruit by regulation of reactive oxygen species metabolism and phenylpropanoid pathway. Sci. Hortic. 2018, 240, 303–309. [Google Scholar] [CrossRef]
- Mittler, R. ROS Are Good. Trends Plant Sci. 2017, 22, 11–19. [Google Scholar] [CrossRef] [Green Version]
- Ji, Y.R.; Hu, W.Z.; Liao, J.; Jiang, A.L.; Xiu, Z.L.; Gaowa, S.; Guan, Y.G.; Yang, X.Z.; Feng, K.; Liu, C.H. Effect of atmospheric cold plasma treatment on antioxidant activities and reactive oxygen species production in postharvest blueberries during storage. J. Sci. Food Agric. 2020, 100, 5586–5595. [Google Scholar] [CrossRef]
- Jin, P.; Wu, X.; Xu, F.; Wang, X.L.; Wang, J.; Zheng, Y.H. Enhancing Antioxidant Capacity and Reducing Decay of Chinese Bayberries by Essential Oils. J. Agric. Food Chem. 2012, 60, 3769–3775. [Google Scholar] [CrossRef] [PubMed]
- Pandey, P.; Singh, J.; Achary, V.M.M.; Reddy, M.K. Redox homeostasis via gene families of ascorbate-glutathione pathway. Front. Environ. Sci.-Switz. 2015, 3, 25. [Google Scholar] [CrossRef] [Green Version]
- Meyer, A.J. The integration of glutathione homeostasis and redox signaling. J. Plant Physiol. 2008, 165, 1390–1403. [Google Scholar] [CrossRef] [PubMed]
- Chotikakham, S.; Faiyue, B.; Uthaibutra, J.; Saengnil, K. Exogenous methyl salicylate alleviates senescent spotting by enhancing the activity of antioxidative ascorbate-glutathione cycle in harvested ‘Sucrier’ bananas. Sci. Hortic. 2020, 267, 109324. [Google Scholar] [CrossRef]
- Lin, Y.X.; Lin, H.T.; Fan, Z.Q.; Wang, H.; Lin, M.S.; Chen, Y.H.; Hung, Y.C.; Lin, Y.F. Inhibitory effect of propyl gallate on pulp breakdown of longan fruit and its relationship with ROS metabolism. Postharvest Biol. Technol. 2020, 168, 111272. [Google Scholar] [CrossRef]
- Zhou, Q.; Ma, C.; Cheng, S.C.; Wei, B.D.; Liu, X.Y.; Ji, S.J. Changes in antioxidative metabolism accompanying pitting development in stored blueberry fruit. Postharvest Biol. Technol. 2014, 88, 88–95. [Google Scholar] [CrossRef]
- Garrido, J.; Garrido, E.M.; Borges, F. Studies on the Food Additive Propyl Gallate: Synthesis, Structural Characterization, and Evaluation of the Antioxidant Activity. J. Chem. Educ. 2012, 89, 130–133. [Google Scholar] [CrossRef]
| Variable | Treatments (A) | Storage Times (B) | Interaction (A × B) |
|---|---|---|---|
| F | F | F | |
| Reddening index | 303.061 ** | 603.204 ** | 22.112 ** |
| Firmness | 148.836 ** | 163.285 ** | 6.755 ** |
| TSS | 4.605 * | 81.618 ** | 9.595 ** |
| TA | 93.143 ** | 48.421 ** | 26.053 ** |
| EL | 262.602 ** | 341.015 ** | 29.288 ** |
| MDA | 438.375 ** | 404.521 ** | 50.161 ** |
| LOX | 86.234 ** | 66.957 ** | 5.974 ** |
| H2O2 | 137.947 ** | 79.388 ** | 7.208 ** |
| Total phenol | 111.385 ** | 127.260 ** | 15.868 ** |
| Total flavonoids | 183.257 ** | 94.247 ** | 11.512 ** |
| ASA | 621.014 ** | 226.845 ** | 21.975 ** |
| GSH | 264.355 ** | 141.382 ** | 12.812 ** |
| SOD | 165.602 ** | 54.523 ** | 6.664 ** |
| CAT | 152.018 ** | 86.204 ** | 9.528 ** |
| APX | 96.392 ** | 169.214 ** | 8.785 ** |
| POD | 684.640 ** | 297.156 ** | 27.418 ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, C.; Chen, C.; Zhao, X.; Wu, C.; Kou, X.; Xue, Z. Propyl Gallate Treatment Improves the Postharvest Quality of Winter Jujube (Zizyphus jujuba Mill. cv. Dongzao) by Regulating Antioxidant Metabolism and Maintaining the Structure of Peel. Foods 2022, 11, 237. https://doi.org/10.3390/foods11020237
Wang C, Chen C, Zhao X, Wu C, Kou X, Xue Z. Propyl Gallate Treatment Improves the Postharvest Quality of Winter Jujube (Zizyphus jujuba Mill. cv. Dongzao) by Regulating Antioxidant Metabolism and Maintaining the Structure of Peel. Foods. 2022; 11(2):237. https://doi.org/10.3390/foods11020237
Chicago/Turabian StyleWang, Chao, Cunkun Chen, Xiaoyang Zhao, Caie Wu, Xiaohong Kou, and Zhaohui Xue. 2022. "Propyl Gallate Treatment Improves the Postharvest Quality of Winter Jujube (Zizyphus jujuba Mill. cv. Dongzao) by Regulating Antioxidant Metabolism and Maintaining the Structure of Peel" Foods 11, no. 2: 237. https://doi.org/10.3390/foods11020237
APA StyleWang, C., Chen, C., Zhao, X., Wu, C., Kou, X., & Xue, Z. (2022). Propyl Gallate Treatment Improves the Postharvest Quality of Winter Jujube (Zizyphus jujuba Mill. cv. Dongzao) by Regulating Antioxidant Metabolism and Maintaining the Structure of Peel. Foods, 11(2), 237. https://doi.org/10.3390/foods11020237