Effects of Three Different Withering Treatments on the Aroma of White Tea
Abstract
:1. Introduction
2. Materials and Methods
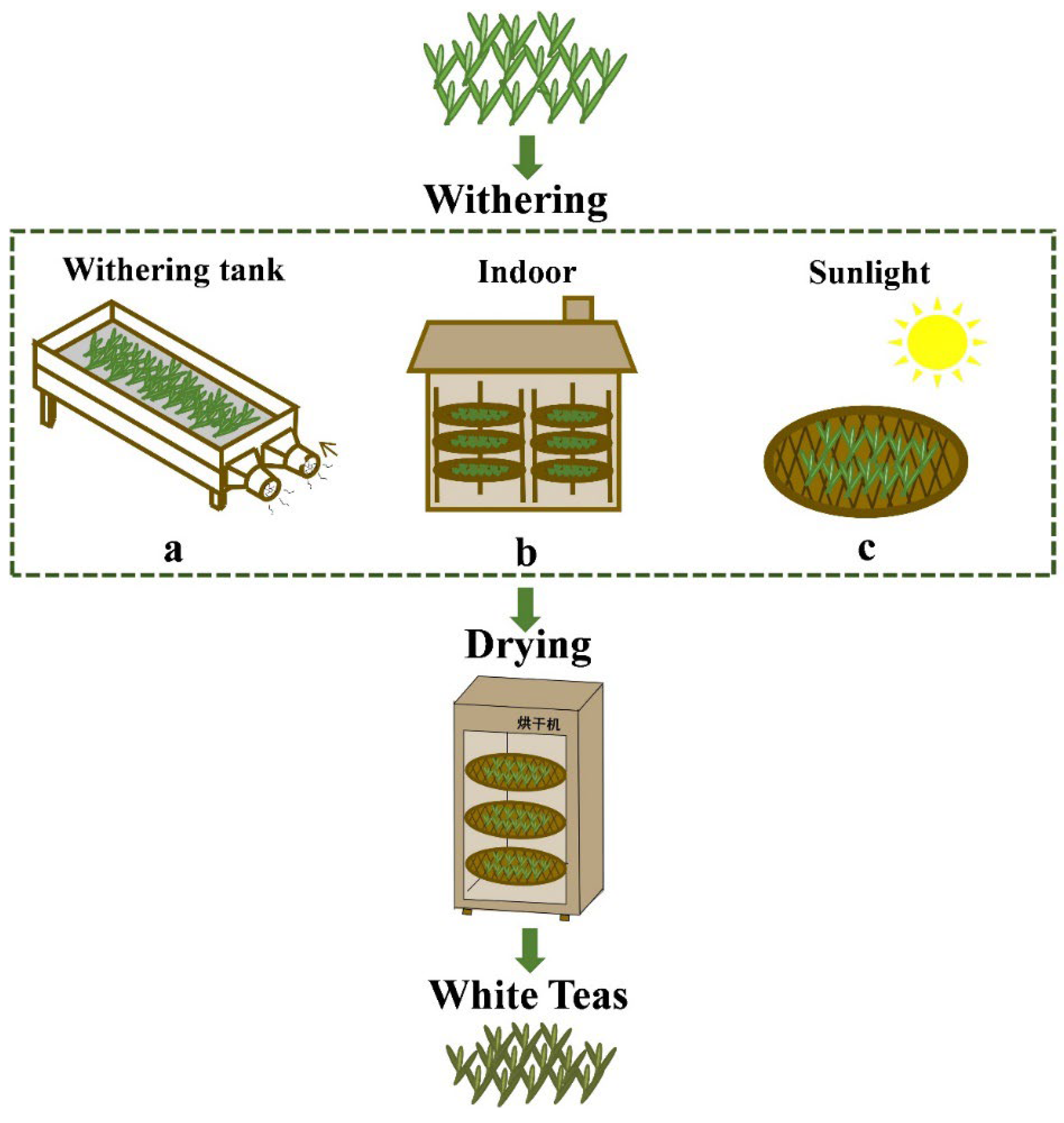
2.1. Chemicals and Samples of Different Withering White Teas
2.2. Quantitative Descriptive Analysis
2.3. Solvent Assisted Flavor Evaporation
2.4. Headspace Solid-Phase Microextraction
2.5. Gas Chromatography-Mass Spectrometry
2.6. Gas Chromatography-Olfactometry and Aroma Extract Dilution Analysis
2.7. Calculation of Odor Activity Values
2.8. Compound Identification and Quantitative Analysis
2.9. Statistical Analysis
3. Results and Discussion
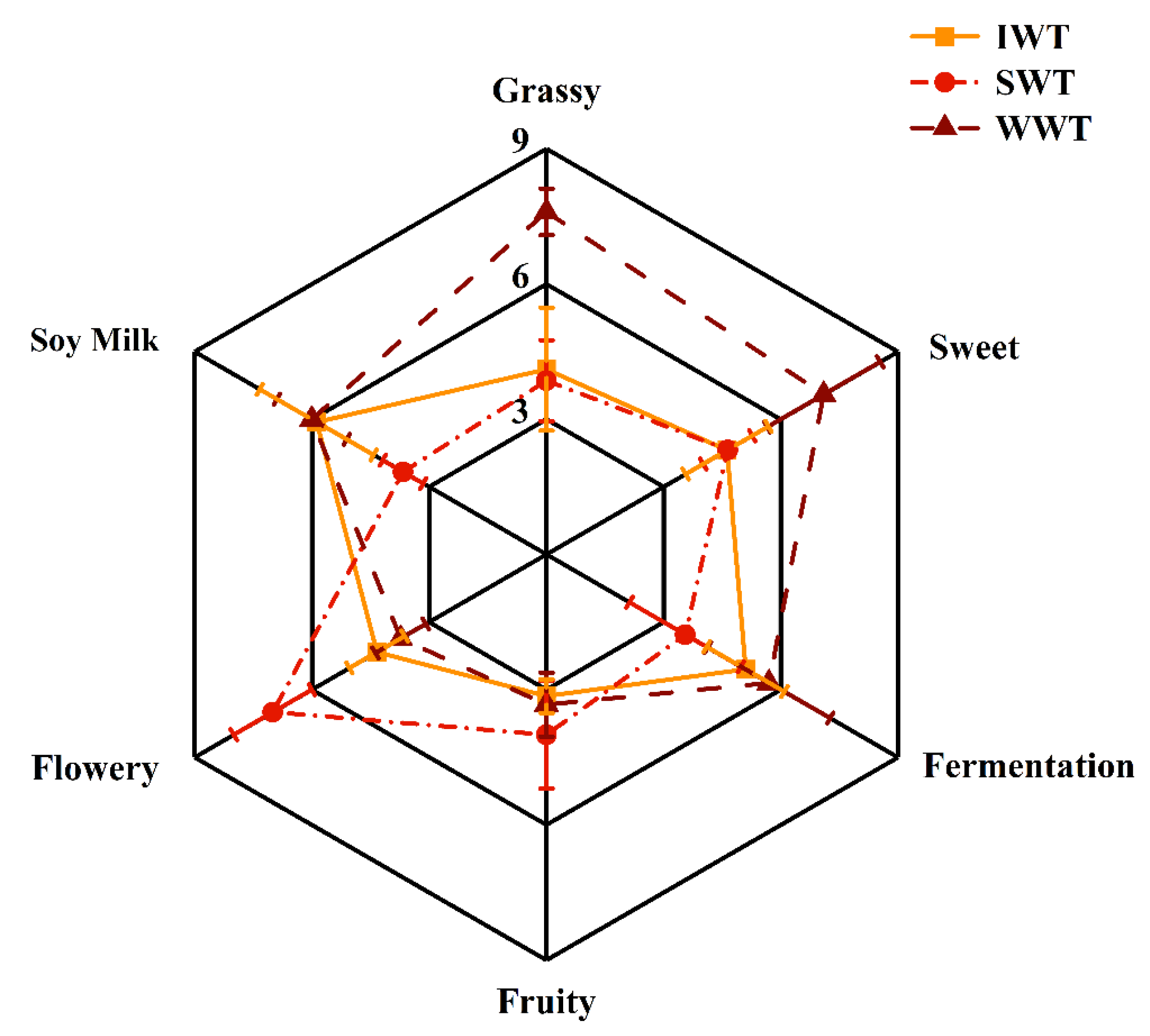
3.1. Aroma Characteristics of White Teas Using Different Withering Treatments
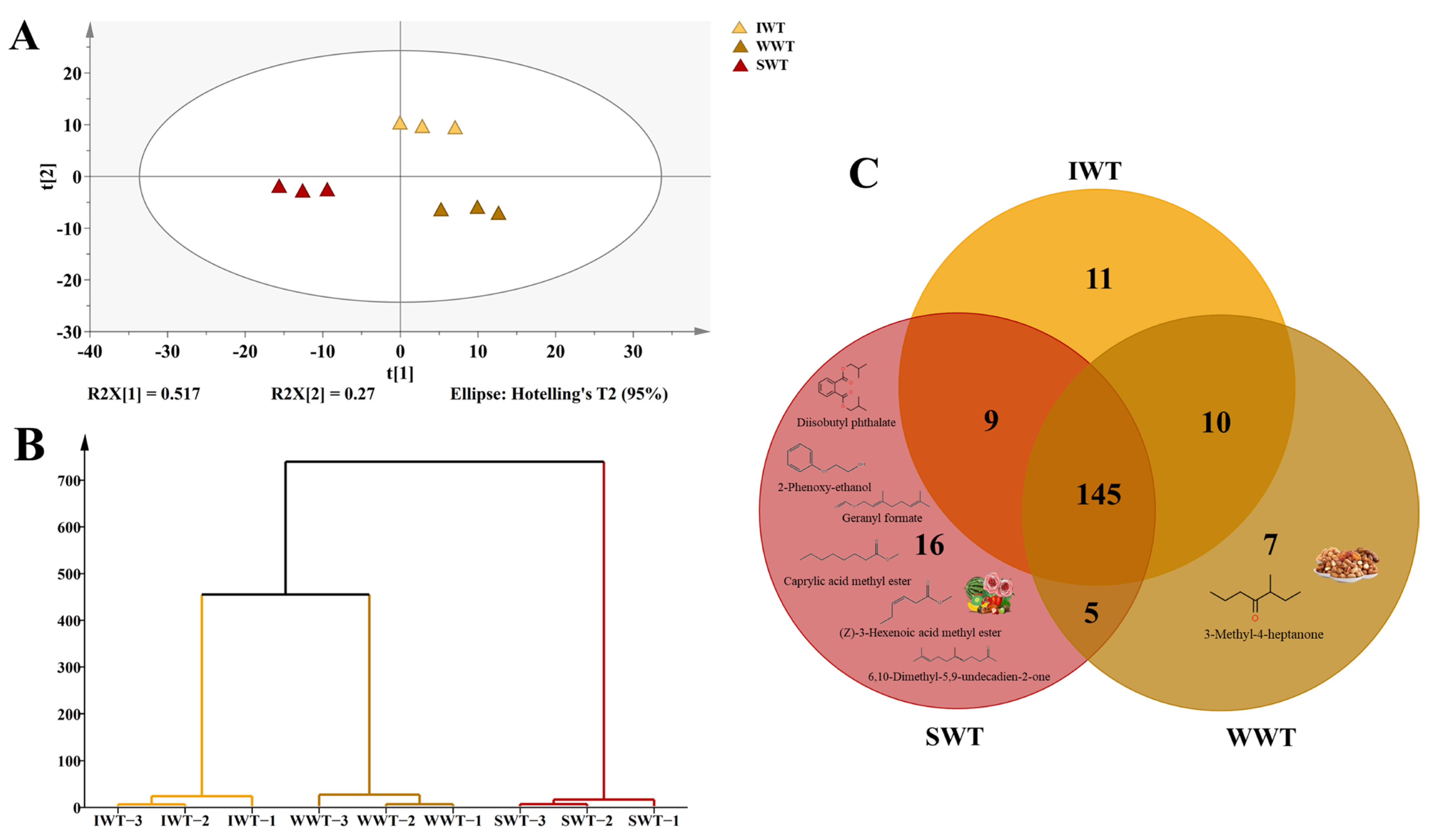
3.2. Comparison of the Differences in the Overall Volatile Compounds of White Teas
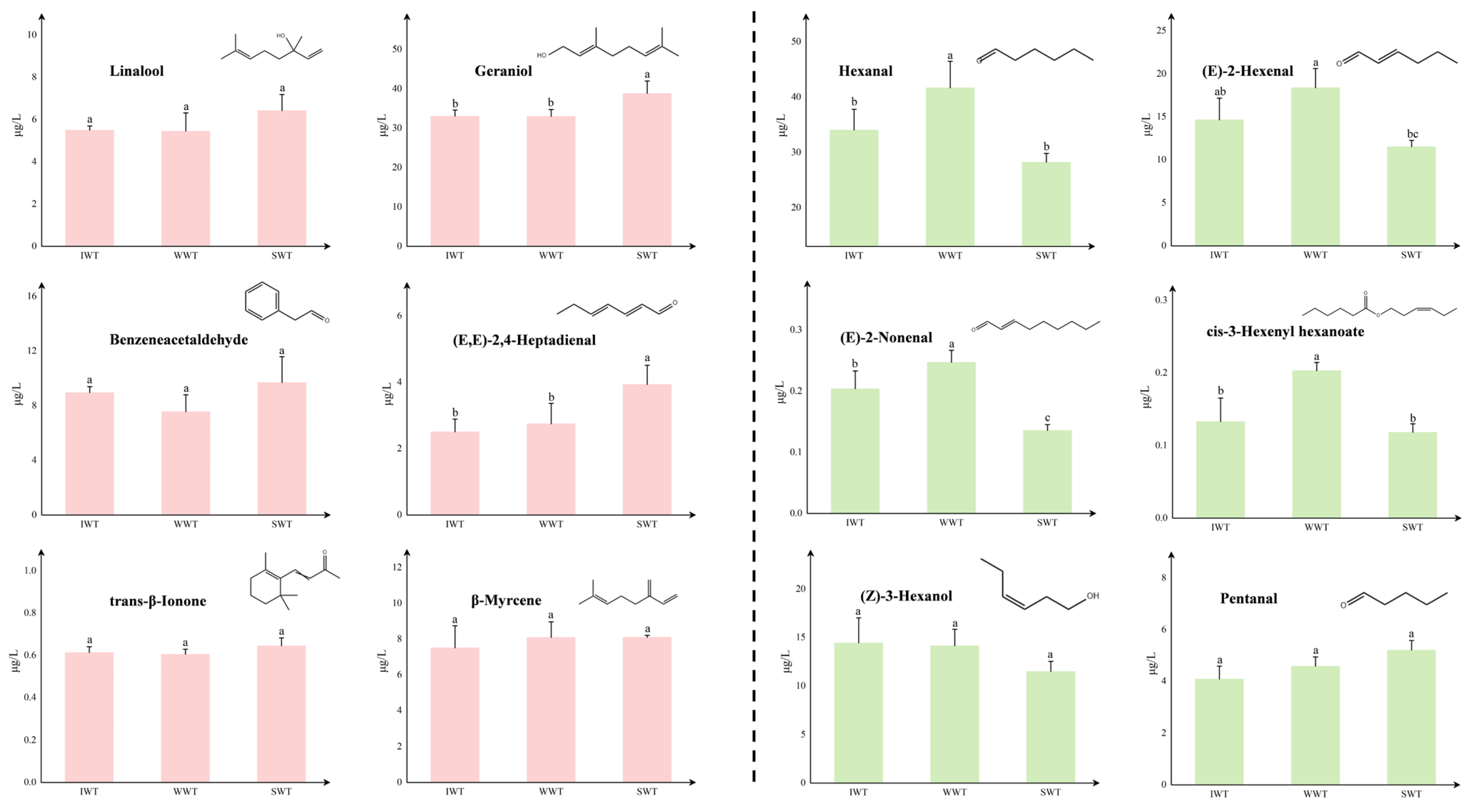
3.3. Comparison of the Differences in Key Odor-Active Compounds of White Teas
3.4. Formation Mechanism of Key Odor-Active Compounds between Different Withering Treatments
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, L.; Ho, C.-T.; Zhou, J.; Santos, J.S.; Armstrong, L. Chemistry and Biological Activities of Processed Camellia sinensis Teas: A Comprehensive Review. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1474–1495. [Google Scholar] [CrossRef] [PubMed]
- Dai, W.; Xie, D.; Lu, M.; Li, P.; Lv, H.; Yang, C.; Peng, Q.; Zhu, Y.; Guo, L.; Zhang, Y. Characterization of white tea metabolome: Comparison against green and black tea by a nontargeted metabolomics approach. Food Res. Int. 2017, 96, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Zhu, C.; Li, X.; Chen, L.; Xie, S.; Chen, G.; Zhang, H.; Lai, Z.; Lin, Y.; Guo, Y. Transcriptome and phytochemical analyses reveal the roles of characteristic metabolites in the taste formation of white tea during the withering process. J. Integr. Agric. 2022, 21, 862–877. [Google Scholar] [CrossRef]
- Chen, L.; Song, D.; Pan, Z.; Chen, J. Overview of white tea withering process research. Acta Tea Sin. 2010, 3, 4–8. [Google Scholar] [CrossRef]
- Yu, X.; Yao, Y.; Zhu, W.; Zhu, L.; Huang, Y. Weathering Technology of Processing Black Tea Leaves in Summer or Autumn. Hubei Agric. Sci. 2015, 54, 424–427. [Google Scholar] [CrossRef]
- Li, Y.; He, C.; Yu, X.; Zhou, J.; Ntezimana, B.; Yu, Z.; Chen, Y.; Ni, D.J. Study on improving aroma quality of summer-autumn black tea by red-light irradiation during withering. LWT 2022, 154, 112597. [Google Scholar] [CrossRef]
- Deng, H.; Chen, S.; Zhou, Z.; Li, X.; Chen, S.; Hu, J.; Lai, Z.; Sun, Y. Transcriptome analysis reveals the effect of short-term sunlight on aroma metabolism in postharvest leaves of oolong tea (Camellia sinensis). Food Res. Int. 2020, 137, 109347. [Google Scholar] [CrossRef]
- Wang, L.; Cai, L.; Lin, Z.; Zhong, Q.; Lv, H.; Tan, J.; Guo, L. Analysis of Aroma Compounds in White Tea Using Headspace Solid-phase Micro-extraction and GC-MS. Tea Sci. 2010, 30, 115–123. [Google Scholar] [CrossRef]
- Chen, Q.; Zhu, Y.; Dai, W.; Lv, H.; Mu, B.; Li, P.; Tan, J.; Ni, D.; Lin, Z. Aroma formation and dynamic changes during white tea processing. Food Chem. 2019, 274, 915–924. [Google Scholar] [CrossRef]
- Mu, L.; Li, T.; Tang, J.; Liu, L.; Wang, R. Effects of LED Light Withering on the Quality of White Tea. Earth Environ. Sci. 2021, 792, 012018. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, Y.; Jing, Y.; Yang, X. Study on the Withering Process and Quality of White Tea in Summer and Autumn in Zhejiang Province. China Tea Proc. 2018, 3, 55–59. [Google Scholar] [CrossRef]
- Feng, Z.; Li, M.; Li, Y.; Yin, J.; Wan, X.; Yang, X. Characterization of the key aroma compounds in infusions of four white teas by the sensomics approach. Eur. Food Res. Technol. 2022, 248, 1299–1309. [Google Scholar] [CrossRef]
- Qi, D.; Miao, A.; Cao, J.; Wang, W.; Chen, W.; Pang, S.; He, X.; Ma, C. Study on the effects of rapid aging technology on the aroma quality of white tea using GC-MS combined with chemometrics: In comparison with natural aged and fresh white tea. Food Chem. 2018, 265, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.Y.; Baldermann, S.; Watanabe, N. Recent studies of the volatile compounds in tea. Food Res. Int. 2013, 53, 585–599. [Google Scholar] [CrossRef]
- Engel, W.; Bahr, W.; Schieberle, P. Solvent assisted flavour evaporation—A new and versatile technique for the careful and direct isolation of aroma compounds from complex food matrices. Eur. Food Res. Technol. 1999, 209, 237–241. [Google Scholar] [CrossRef]
- Corral, S.; Salvador, A.; Flores, M. Elucidation of key aroma compounds in traditional dry fermented sausages using different extraction techniques. J. Sci. Food Agric. 2015, 95, 1350–1361. [Google Scholar] [CrossRef]
- Chen, S.; Cheng, W.; Qian, M.; Li, Z.; Xu, Y. Characterization of the Key Aroma Compounds in Aged Chinese Rice Wine by Comparative Aroma Extract Dilution Analysis, Quantitative Measurements, Aroma Recombination, and Omission Studies. J. Agric. Food Chem. 2019, 67, 4876–4884. [Google Scholar] [CrossRef]
- Xu, S.; Zeng, X.; Wu, H.; Shen, S.; Yang, X.; Deng, W.-W.; Ning, J. Characterizing Volatile Metabolites in Raw Pu’er Tea Stored in Wet-Hot or Dry-Cold Environments by Performing Metabolomic Analysis and using the Molecular Sensory Science Approach. Food Chem. 2021, 350, 129186. [Google Scholar] [CrossRef]
- Niu, H.; Yuan, L.; Zhou, H.; Yun, Y.; Li, J.; Tian, J.; Zhong, K.; Zhou, L. Comparison of the Effects of High Pressure Processing, Pasteurization and High Temperature Short Time on the Physicochemical Attributes, Nutritional Quality, Aroma Profile and Sensory Characteristics of Passion Fruit Purée. Foods 2022, 11, 632. [Google Scholar] [CrossRef]
- Cao, Q.; Wang, F.; Wang, J.; Chen, J.; Yin, J.; Li, L.; Meng, F.; Cheng, Y.; Xu, Y. Effects of brewing water on the sensory attributes and physicochemical properties of tea infusions. Food Chem. 2021, 364, 130235. [Google Scholar] [CrossRef]
- Wei, Y.; Yin, X.; Wu, H.; Zhao, M.; Huang, J.; Zhang, J.; Li, T.; Ning, J. Improving the flavor of summer green tea (Camellia sinensis L.) using the yellowing process. Food Chem. 2022, 388, 132982. [Google Scholar] [CrossRef] [PubMed]
- Zhai, X.; Zhang, L.; Michael, G.; Ho, C.-T.; Wan, X. Flavor of tea (Camellia sinensis): A review on odorants and analytical techniques. Compr. Rev. Food Sci. Food Saf. 2022, 1–43. [Google Scholar] [CrossRef] [PubMed]
- Gemert, L.J. Compilations of Odour Threshold Values in Air, Water and Other Media, 2nd ed.; Oliemans Punter & Partners BV: Zeist, The Netherlands, 2011; pp. 207–359. [Google Scholar]
- Guo, X.; Ho, C.-T.; Schwab, W.; Wan, X. Aroma profiles of green tea made with fresh tea leaves plucked in summer. Food Chem. 2021, 363, 130328. [Google Scholar] [CrossRef] [PubMed]
- Leibniz Institute for Food Systems Biology at the Technical University of Munich: Freising, Germany; Kreissl, J.; Mall, V.; Steinhaus, P.; Steinhaus, M. Leibniz-LSB@TUM Odorant Database, Version 1.2. Available online: https://www.leibniz-lsb.de/en/databases/leibniz-lsbtum-odorant-database (accessed on 13 April 2022).
- Meng, T.; Zhipeng, X.; Ai, H.; Jiayu, C.; Tianji, Y.; Zhengquan, L. Effect of 1–20 years storage on volatiles and aroma of Keemun congou black tea by solvent extraction-solid phase extraction-gas chromatography-mass spectrometry. LWT 2021, 136, 110278. [Google Scholar] [CrossRef]
- Zeng, L.; Fu, Y.; Huang, J.; Wang, J.; Jin, S.; Yin, J.; Xu, Y. Comparative Analysis of Volatile Compounds in Tieguanyin with Different Types Based on HS–SPME–GC–MS. Foods 2022, 11, 1530. [Google Scholar] [CrossRef]
- Granato, D.; Santos, J.; Escher, G.; Ferreira, B.; Maggio, R. Use of principal component analysis (PCA) and hierarchical cluster analysis (HCA) for multivariate association between bioactive compounds and functional properties in foods: A critical perspective. Trends Food Sci. Technol. 2018, 72, 83–90. [Google Scholar] [CrossRef]
- Zhu, Y.; Dong, J.; Jin, J.; Liu, J.; Zheng, X.; Lu, J.; Liang, Y.; Ye, J. Roasting process shaping the chemical profile of roasted green tea and the association with aroma features. Food Chem. 2021, 353, 129428. [Google Scholar] [CrossRef]
- Zhai, X.T.; Granvogl, M. Characterization of the Key Aroma Compounds in Two Differently Dried Toona sinensis (A. Juss.) Roem. by Means of the Molecular Sensory Science Concept. J. Agric. Food Chem. 2019, 67, 9885–9894. [Google Scholar] [CrossRef]
- Jiang, S.; Xia, D.; Wang, X.; Zhu, Y.; Chen, G.; Liu, Y. Analysis of aroma-active compounds in four Chinese dry-cured hams based on GC-O combined with AEDA and frequency detection methods. LWT 2022, 153, 112497. [Google Scholar] [CrossRef]
- Song, H.; Liu, J. GC-O-MS technique and its applications in food flavor analysis. Food Res. Int. 2018, 114, 187–198. [Google Scholar] [CrossRef]
- Ma, L.; Gao, M.; Zhang, L.; Qiao, Y.; Li, J.; Du, L.; Zhang, H.; Wang, H. Characterization of the key aroma-active compounds in high-grade Dianhong tea using GC-MS and GC-O combined with sensory-directed flavor analysis. Food Chem. 2022, 378, 132058. [Google Scholar] [CrossRef]
- Gu, F.; Chen, Y.; Hong, Y.; Fang, Y.; Tan, L. Comparative metabolomics in vanilla pod and vanilla bean revealing the biosynthesis of vanillin during the curing process of vanilla. AMB Express 2017, 7, 116. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Li, Q.; Xiang, L.; Liang, Y. Recent Advances in Volatiles of Teas. Molecules 2016, 21, 338. [Google Scholar] [CrossRef] [PubMed]
- Schwab, W.; Davidovich-Rikanati, R.; Lewinsohn, E. Biosynthesis of plant-derived flavor compounds. Plant J. 2008, 54, 712–732. [Google Scholar] [CrossRef]
- Xu, Q.; He, Y.; Yan, X.; Zhao, S.; Zhu, J.; Wei, C. Unraveling a crosstalk regulatory network of temporal aroma accumulation in tea plant (Camellia sinensis) leaves by integration of metabolomics and transcriptomics. Environ. Exp. Bot. 2018, 149, 81–94. [Google Scholar] [CrossRef]
- Nagegowda, D.A. Plant volatile terpenoid metabolism: Biosynthetic genes, transcriptional regulation and subcellular compartmentation. FEBS Lett. 2010, 584, 2965–2973. [Google Scholar] [CrossRef]
- Hu, C.; Li, D.; Ma, Y.; Zhang, W.; Lin, C.; Zheng, X.; Liang, Y.; Lu, J. Formation mechanism of the oolong tea characteristic aroma during bruising and withering treatment. Food Chem. 2018, 269, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Contreras, C.; Tjellström, H.; Beaudry, R.M. Relationships between free and esterified fatty acids and LOX-derived volatiles during ripening in apple. Postharvest Biol. Technol. 2016, 112, 105–113. [Google Scholar] [CrossRef]
- Salas, J.; Sanchez, C.; Garciia-Gonzalez, D.; Aparicio, R. Impact of the suppression of lipoxygenase and hydroperoxide lyase on the quality of the green odor in green leaves. J. Agric. Food Chem. 2005, 53, 1648–1655. [Google Scholar] [CrossRef]
| No. | RI A | CAS | Compound B | Category C | Odor Characteristics | OT (μg/kg) D | OAV E | FD/Odor Intensity F | OAV | FD | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IWT | WWT | SWT | IWT | WWT | SWT | |||||||||
| 1 | <600 | 75-18-3 | Dimethyl sulfide | AADVs | cabbage-like | 0.07 | 357.3 | 290.5 | 210.9 | 1.8 | 64/2.7 | 2.0 | √ | √ |
| 2 | 647 | 96-17-3 | 2-Methyl-butanal | AADVs | malty | 1.5 | 5.4 | 4.0 | 3.6 | 2.2 | 64/3.0 | 2.3 | √ | √ |
| 3 | 652 | 1629-58-9 | 1-Penten-3-one | FADVs | pungent, fish | 0.9 | 4.1 | 3.5 | 3.0 | 2.2 | 4/2.8 | 2.2 | √ | √ |
| 4 | 800 | 66-25-1 | Hexanal | FADVs | green, grassy | 1.09 | 31.2 | 38.2 | 25.9 | 2.3 | 16/3.3 | 2.8 | √ | √ |
| 5 | 897 | 6728-31-0 | (Z)-4-Heptenal | AADVs | fatty, fish | 0.0087 | 21.0 | 23.5 | 14.5 | 2.2 | 128/3.7 | 2.5 | √ | √ |
| 6 | 994 | 123-35-3 | β-Myrcene | VTs | sweet balsamic aroma | 1.2 | 6.3 | 6.7 | 6.7 | 1.3 | 8/1.7 | 2.2 | √ | √ |
| 7 | 1101 | 78-70-6 | Linalool | VTs/GDVs | citrus-like, flowery | 0.58 | 9.5 | 9.4 | 11.1 | 128 | 128 | 512 | √ | √ |
| 8 | 1252 | 106-24-1 | Geraniol | VTs/GDVs | rose-like, citrus-like | 1.1 | 30.0 | 30.0 | 35.3 | 16 | 16 | 256 | √ | √ |
| 9 | 1479 | 79-77-6 | trans-β-Ionone | CDVs | flowery, violet-like | 0.021 | 29.2 | 28.8 | 30.7 | - | - | 64 | √ | √ |
| 10 | 632 | 590-86-3 | 3-Methyl-butanal | AADVs | fruity, fatty, animal | 1.44 | 3.1 | 2.6 | 2.2 | √ | ||||
| 11 | 852 | 928-96-1 | (Z)-3-hexanol | FADVs | green leaves | 3.9 | 3.7 | 3.6 | 2.9 | √ | ||||
| 12 | 928 | 15726-15-5 | 3-Methyl-4-heptanone | nutty | 0.05 | - | 2.6 | - | √ | |||||
| 14 | 1012 | 4313-3-5 | (E,E)-2,4-Heptadienal | FADVs | fatty, flowery | 0.032 | 78.3 | 85.8 | 122.8 | √ | ||||
| 15 | 1045 | 122-78-1 | Benzeneacetaldehyde | AADVs | fatty, flowery | 5.2 | 1.7 | 1.5 | 1.9 | √ | ||||
| 16 | 1081 | 586-62-9 | 1-Methyl-4-(1-methylethylidene)-cyclohexene | sweet-piney, oily, pleasant aroma | 0.2 | 2.1 | 2.4 | 1.5 | √ | |||||
| 17 | 1150 | 10340-23-5 | (Z)-3-Nonen-1-ol | waxy green melon aroma | 0.209 | 1.5 | 1.4 | 0.9 | √ | |||||
| 18 | 1156 | 18829-56-6 | (E)-2-Nonenal | FADVs | fatty, green | 0.19 | 1.1 | 1.3 | 0.7 | √ | ||||
| 19 | 1202 | 116-26-7 | 2,6,6-Trimethyl-1,3-cyclohexadiene-1-carboxaldehyde | CDVs | saffron tea | 0.0455 | 2.2 | 2.7 | 4.6 | √ | ||||
| 20 | 1377 | 31501-11-8 | cis-3-Hexenyl hexanoate | FADVs | fruity green odour | 0.195 | 0.7 | 1.0 | 0.6 | √ | ||||
| 21 | <600 | 534-22-5 | 2-Methyl-Furan | spicy smoky aroma | n.d. | - | - | - | 1.5 | 8/2.2 | 1.3 | √ | ||
| 22 | 671 | 110-62-3 | Pentanal | FADVs | green, fatty, moldy | 8 | 0.5 | 0.6 | 0.7 | 1.5 | 8/2.3 | 1.0 | √ | |
| 23 | 847 | 6728-26-3 | (E)-2-Hexenal | FADVs | green apple-like | 110 | 0.1 | 0.2 | 0.1 | 0.8 | 8/1.3 | 1.0 | √ | |
| 24 | 1047 | 695-06-7 | 5-Ethyldihydro-2(3H)-furanone | FADVs | coconut-like, fruity | 260 | 0.0 | 0.0 | 0.0 | 4 | - | 8 | √ | |
| 25 | 1061 | 98-85-1 | α-methyl-Benzenemethanol | mlid floral odor | 479 | - | 0.0 | 0.0 | - | 2 | 4 | √ | ||
| 26 | 1080 | 111-14-8 | Heptanoic acid | FADVs | rancid, sweaty | 640 | 0.0 | 0.0 | 0.0 | - | - | 16 | √ | |
| 27 | 1115 | 1960-12-8 | Phenylethyl Alcohol | AADVs/GBVs | floral, honey-like | 140 | 0.2 | 0.2 | 0.2 | 8 | 32 | 64 | √ | |
| 28 | 1238 | 106-26-3 | Neral | VTs | citrus-like, soapy | 5.5 | 0.1 | 0.1 | 0.1 | 8 | 64 | 32 | √ | |
| 29 | 1262 | 141-27-5 | α-Citral | VTs | citrus-like | 12 | 0.1 | - | - | - | 2 | 32 | √ | |
| 30 | 1365 | 459-80-3 | Geranic acid | apple-like fruit and vegetable aroma | n.d. | - | - | - | - | 4 | 8 | √ | ||
| 31 | 1398 | 121-33-5 | Vanillin | vanilla-like, sweet | 53 | 0.0 | 0.0 | 0.0 | 16 | 128 | 128 | √ | ||
| 32 | 1439 | 91-64-5 | Coumarin | GBVs | woodruff-like, almond paste-like | 11 | 0.2 | 0.2 | 0.2 | 8 | 4 | 256 | √ | |
| 33 | 1495 | 25524-95-2 | Jasmine lactone | coconut, fatty, fruity | 350 | 0.0 | 0.0 | 0.0 | - | 2 | 4 | √ | ||
| 34 | 1521 | 96-76-4 | 2,4-Di-tert-butylphenol | phenolic-like, leather-like | 500 | 0.0 | 0.0 | 0.0 | - | - | 8 | √ | ||
| 35 | 1530 | 17092-92-1 | Dihydroactindiolide | CDVs | musky or coumarin-like aroma | n.d. | - | - | - | - | 2 | 4 | √ | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, H.; Chen, Y.; Feng, W.; Shen, S.; Wei, Y.; Jia, H.; Wang, Y.; Deng, W.; Ning, J. Effects of Three Different Withering Treatments on the Aroma of White Tea. Foods 2022, 11, 2502. https://doi.org/10.3390/foods11162502
Wu H, Chen Y, Feng W, Shen S, Wei Y, Jia H, Wang Y, Deng W, Ning J. Effects of Three Different Withering Treatments on the Aroma of White Tea. Foods. 2022; 11(16):2502. https://doi.org/10.3390/foods11162502
Chicago/Turabian StyleWu, Huiting, Yuyu Chen, Wanzhen Feng, Shanshan Shen, Yuming Wei, Huiyan Jia, Yujie Wang, Weiwei Deng, and Jingming Ning. 2022. "Effects of Three Different Withering Treatments on the Aroma of White Tea" Foods 11, no. 16: 2502. https://doi.org/10.3390/foods11162502
APA StyleWu, H., Chen, Y., Feng, W., Shen, S., Wei, Y., Jia, H., Wang, Y., Deng, W., & Ning, J. (2022). Effects of Three Different Withering Treatments on the Aroma of White Tea. Foods, 11(16), 2502. https://doi.org/10.3390/foods11162502








