Multi-Target Alternative Approaches to Promoting Fresh-Cut Carrots’ Bioactive and Fresh-like Quality
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
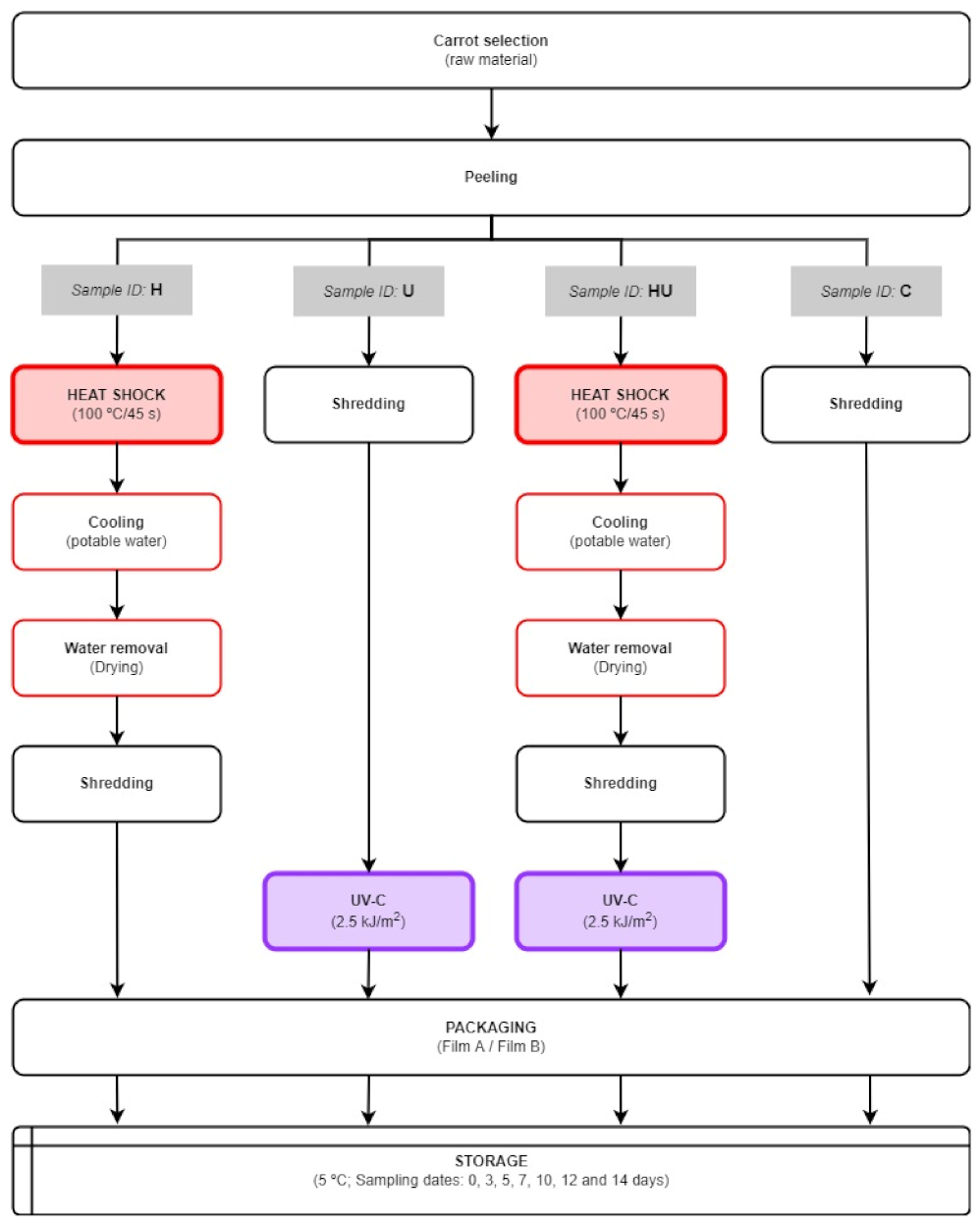
2.2. Sample Preparation
Minimal Processing, Abiotic Stress Treatments, and Storage
2.3. Analytical Procedures
2.3.1. Headspace Gas Analysis
2.3.2. Total Phenolic Content
2.3.3. Chlorogenic Acid Quantification
2.3.4. Antioxidant Capacity
2.3.5. Phenylalanine-Ammonia Lyase (PAL) Activity
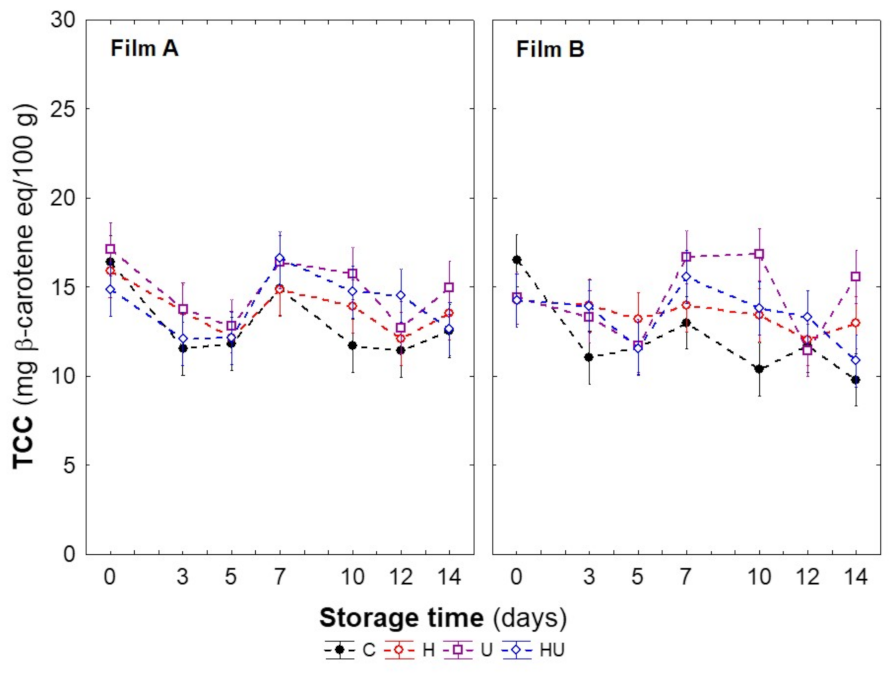
2.3.6. Total Carotenoid Content
2.3.7. pH and Soluble Solids Content
2.3.8. Sensorial Analysis
2.3.9. Microbiological Responses
2.3.10. Statistical Analysis
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. Fruit and Vegetables—Your Dietary Essentials. The International Year of Fruits and Vegetables, 2021, Background Paper; FAO: Rome, Italy, 2020. [Google Scholar] [CrossRef]
- Van Duyn, M.A.; Pivonka, E. Overview of the health benefits of fruit and vegetable consumption for the dietetics professional: Selected literature. J. Am. Diet Assoc. 2000, 100, 1511–1521. [Google Scholar] [CrossRef]
- Rojas-Graü, M.A.; Garner, E.; Martín-Belloso, O. The fresh-cut fruit and vegetables industry: Current situation and market trends. In Advances in Fresh-Cut Fruits and Vegetables Processing, 1st ed.; Martín-Belloso, O., Soliva-Fortuny, R., Eds.; CRC Press: Boca Raton, FL, USA, 2011; pp. 1–12. [Google Scholar] [CrossRef]
- Baselice, A.; Colantuoni, F.; Lass, D.A.; Nardone, G.; Stasi, A. Trends in EU consumers' attitude towards fresh-cut fruit and vegetables. Food Qual. Prefer. 2017, 59, 87–96. [Google Scholar] [CrossRef]
- Raffo, A.; Paoletti, F. Fresh-Cut Vegetables Processing: Environmental Sustainability and Food Safety Issues in a Comprehensive Perspective. Front. Sustain. Food Syst. 2022, 5, 681459. [Google Scholar] [CrossRef]
- Gil, M.I.; Allende, A. Water and wastewater use in the fresh produce industry: Food safety and environmental implications. In Quantitative Methods for Food Safety and Quality in the Vegetable Industry. Food Microbiology and Food Safety; Pérez-Rodríguez, F., Skandamis, P., Valdramidis, V., Eds.; Springer: Cham, Switzerland, 2018; pp. 59–76. [Google Scholar] [CrossRef]
- Barry-Ryan, C.; Pacussi, J.M.; O’Beirne, D. Quality of shredded carrots as affected by packaging film and storage temperature. J. Food Sci. 2000, 65, 726–730. [Google Scholar] [CrossRef]
- Yousuf, B.; Deshi, V.; Ozturk, B.; Siddiqui, M.W. 1-fresh-cut fruits and vegetables: Quality issues and safety concerns. In Fresh-Cut Fruits and Vegetables: Technologies and Mechanisms for Safety Control; Siddiqui, M.W., Ed.; Academic Press: London, UK; San Diego, CA, USA, 2020; pp. 1–15. [Google Scholar]
- Rico, D.; Martín-Diana, A.B.; Barat, J.M.; Barry-Ryan, C. Extending and measuring the quality of fresh-cut fruit and vegetables: A review. Trends Food Sci. Technol. 2007, 18, 373–386. [Google Scholar] [CrossRef]
- Manzocco, L.; Ignat, A.; Anese, M.; Bot, F.; Calligaris, S.; Valoppi, F.; Nicoli, M.C. Efficient management of the water resource in the fresh-cut industry: Current status and perspectives. Trends Food Sci. Technol. 2015, 46, 286–294. [Google Scholar] [CrossRef]
- Piscopo, A.; Zappia, A.; Princi, M.P.; De Bruno, A.; Araniti, F.; Antonio, L.; Abenavoli, M.R.; Poiana, M. Quality of shredded carrots minimally processed by different dipping solutions. J. Food Sci. Technol. 2019, 56, 2584–2593. [Google Scholar] [CrossRef]
- Health Protection Agency. Guidelines for Assessing the Microbiological Safety of Ready-to-Eat Foods; Health Protection Agency: London, UK, 2009. [Google Scholar]
- Seljåsen, R.; Bengtsson, G.B.; Hoftun, H.; Vogt, G. Sensory and chemical changes in five varieties of carrot (Daucus carota L.) in response to mechanical stress at harvest and post-harvest. J. Sci. Food Agric. 2001, 81, 436–447. [Google Scholar] [CrossRef]
- Gastélum-Estrada, A.; Hurtado-Romero, A.; Santacruz, A.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. Sanitizing after fresh-cutting carrots reduces the wound-induced accumulation of phenolic antioxidants compared to sanitizing before fresh-cutting. J. Sci. Food Agric. 2020, 100, 4995–4998. [Google Scholar] [CrossRef] [PubMed]
- Alegria, C.; Pinheiro, J.; Gonçalves, E.M.; Fernandes, I.; Moldão, M.; Abreu, M. Quality attributes of shredded carrot (Daucus carota L. cv. Nantes) as affected by alternative decontamination processes to chlorine. Innov. Food Sci. Emerg. Technol. 2009, 10, 61–69. [Google Scholar] [CrossRef]
- Alegria, C.; Pinheiro, J.; Duthoit, M.; Gonçalves, E.M.; Moldão-Martins, M.; Abreu, M. Fresh-cut carrot (cv. Nantes) quality as affected by abiotic stress (heat shock and UV-C irradiation) pre-treatments. LWT 2012, 48, 197–203. [Google Scholar] [CrossRef]
- Pedreschi, R.; Lurie, S. Advances and current challenges in understanding postharvest abiotic stresses in perishables. Postharvest Biol. Technol. 2015, 107, 77–89. [Google Scholar] [CrossRef]
- Jacobo-Velázquez, D.A.; González-Agüero, M.; Cisneros-Zevallos, L. Cross-talkbetween signaling pathways: The link between plant secondary metabolite production andwounding stress response. Sci. Rep. 2015, 5, 8608. [Google Scholar] [CrossRef] [PubMed]
- Alegria, C.; Gonçalves, E.M.; Moldão-Martins, M.; Cisneros-Zevallos, L.; Abreu, M. Peel removal improves quality without antioxidant loss through wound-induced phenolic biosynthesis in shredded carrot. Postharvest Biol. Technol. 2016, 120, 232–239. [Google Scholar] [CrossRef]
- Cisneros-Zevallos, L. The use of controlled postharvest abiotic stresses as a tool for enhancing the nutraceutical content and adding-value of fresh fruits and vegetables. J. Food Sci. 2003, 68, 1560–1565. [Google Scholar] [CrossRef]
- Aguayo, E.; Escalona, V.H.; Artés, F. Effect of hot water treatment and various calcium salts on quality of fresh-cut ‘Amarillo’ melon. Postharvest Biol. Technol. 2008, 47, 397–406. [Google Scholar] [CrossRef]
- Klaiber, R.G.; Baur, S.; Wolf, G.; Hammes, W.P.; Carle, R. Quality of minimally processed carrots as affected by warm water washing and chlorination. Innov. Food Sci. Emerg. Technol. 2005, 6, 351–362. [Google Scholar] [CrossRef]
- Rux, G.; Efe, E.; Ulrichs, C.; Huyskens-Keil, S.; Hassenberg, K.; Herppich, W.B. Effects of Pre-Processing Short-Term Hot-Water Treatments on Quality and Shelf Life of Fresh-Cut Apple Slices. Foods 2019, 8, 653. [Google Scholar] [CrossRef] [PubMed]
- Rico, D.; Martín-Diana, A.B.; Barry-Ryan, C.; Frías, J.M.; Henehan, G.T.M.; Barat, J.M. Optimisation of steamer jet-injection to extend the shelflife of fresh-cut lettuce. Postharvest Biol. Technol. 2008, 48, 431–442. [Google Scholar] [CrossRef]
- Alegria, C.; Gonçalves, E.M.; Moldão-Martins, M.; Abreu, M. Influence of a heat-shock pre-treatment on wound-induced phenolic biosynthesis as an alternative strategy towards fresh-cut carrot processing. Food Sci. Technol. Int. 2022, 28, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Saltveit, M.E. Wound induced changes in phenolic metabolism and tissue browning are altered by heat shock. Postharvest Biol. Technol. 2000, 21, 61–69. [Google Scholar] [CrossRef]
- Loaiza-Velarde, J.G.; Mangrich, M.E.; Campos-Vargas, R.; Saltveit, M.E. Heat shock reduces browning of fresh-cut celery petioles. Postharvest Biol. Technol. 2003, 27, 305–311. [Google Scholar] [CrossRef]
- Alegria, C.; Pinheiro, J.; Gonçalves, E.M.; Fernandes, I.; Moldão, M.; Abreu, M. (2010). Evaluation of a pre-cut heat treatment as an alternative to chlorine in minimally processed shredded carrot. Innov. Food Sci. Emerg. Technol. 2010, 11, 155–161. [Google Scholar] [CrossRef]
- Castillejo, N.; Martínez-Zamora, L.; Artés-Hernández, F. Postharvest UV radiation enhanced biosynthesis of flavonoids and carotenes in bell peppers. Postharvest Biol. Technol. 2022, 184, 111774. [Google Scholar] [CrossRef]
- Dou, H.; Niu, G.; Gu, M. Pre-Harvest UV-B Radiation and Photosynthetic Photon Flux Density Interactively Affect Plant Photosynthesis, Growth, and Secondary Metabolites Accumulation in Basil (Ocimum basilicum) Plants. Agronomy 2019, 9, 434. [Google Scholar] [CrossRef]
- Moreira-Rodríguez, M.; Nair, V.; Benavides, J.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. UVA, UVB Light, and Methyl Jasmonate, Alone or Combined, Redirect the Biosynthesis of Glucosinolates, Phenolics, Carotenoids, and Chlorophylls in Broccoli Sprouts. Int. J. Mol. Sci. 2017, 18, 2330. [Google Scholar] [CrossRef] [PubMed]
- Pataro, G.; Sinik, M.; Capitoli, M.M.; Donsì, G.; Ferrari, G. The influence of post-harvest UV-C and pulsed light treatments on quality and antioxidant properties of tomato fruits during storage. Innov. Food Sci. Emerg. Technol. 2015, 30, 103–111. [Google Scholar] [CrossRef]
- Adetuyi, F.O.; Karigidi, K.O.; Akintimehin, E.S. Effect of postharvest UV-C treatments on the bioactive components, antioxidant and inhibitory properties of clerodendrum volubile leaves. J. Saudi Soc. Agric. Sci. 2020, 19, 7–13. [Google Scholar] [CrossRef]
- Artés-Hernández, F.; Robles, P.A.; Gómez, P.A.; Tomás-Callejas, A.; Artés, F.; Martínez-Hernández, G.B. Quality Changes of Fresh-Cut Watermelon During Storage as Affected by Cut Intensity and UV-C Pre-treatment. Food Bioprocess Technol. 2021, 14, 505–517. [Google Scholar] [CrossRef]
- Formica-Oliveira, A.C.; Martínez-Hernández, G.B.; Aguayo, E.; Gómes, P.A.; Artés, F.; Artés-Hernandez, F. UV-C and hyperoxia abiotic stresses to improve healthiness of carrots: Study of combined effects. J. Food Sci. Technol. 2016, 53, 3465–3476. [Google Scholar] [CrossRef] [PubMed]
- Formica-Oliveira, A.C.; Martínez-Hernández, G.B.; Díaz-López, V.; Artés, F.; Artés- Hernández, F. Use of postharvest UV-B and UV-C radiation treatments to revalorize broccoli byproducts and edible florets. Innov. Food Sci. Emerg. Technol. 2017, 43, 77–83. [Google Scholar] [CrossRef]
- Artés-Hernández, F.; Martínez-Hernández, G.B.; Aguayo, E.; Gómez, P.A.; Artés, F. Fresh-cut fruit and vegetables: Emerging eco-friendly techniques for sanitation and preserving safety. In Postharvest Handling; Kahramanoglu, I., Ed.; IntechOpen: London, UK, 2017; pp. 7–45. [Google Scholar] [CrossRef]
- Martínez-Hernández, G.B.; Gómez, P.A.; Pradas, I.; Artés, F.; Artés-Hernández, F. Moderate UV-C pretreatment as a quality enhancement tool in fresh-cut Bimi® broccoli. Postharvest Biol. Technol. 2011, 62, 327–337. [Google Scholar] [CrossRef]
- Allende, A.; McEvoy, J.L.; Luo, Y.; Artes, F.; Wang, C.Y. Effectiveness of two-sided UV-C treatments in inhibiting natural microflora and extending the shelf-life of minimally processed ‘Red Oak Leaf’ lettuce. Food Microbiol. 2006, 23, 241–249. [Google Scholar] [CrossRef]
- Surjadinata, B.B.; Jacobo-Velázquez, D.A.; Cisneros-Zevallos, L. UVA, UVB and UVC Light Enhances the Biosynthesis of Phenolic Antioxidants in Fresh-Cut Carrot through a Synergistic Effect with Wounding. Molecules 2017, 22, 668. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Rangel, J.C.; Benavides, J.; Jacobo-Velázquez, D.A. Production of Nutraceuticals in Carrot Bagasse using Abiotic Stresses. Acta Hortic. 2013, 1012, 1475–1479. [Google Scholar] [CrossRef]
- Alegria, C.S.M. Heat Shock and UV-C Abiotic Stress Treatments as Alternative Tools to Promote Fresh-Cut Carrot Quality and Shelf-Life. Ph.D. Thesis, Universidade de Lisboa, Lisbon, Portugal, 24 March 2015. [Google Scholar]
- Petitjean-Freytet, C.; Carnat, A.; Lamaison, J.L. Teneurs en flavonoids et en derives hydroxycinnamiques de la fleurs de Sambacus nigra L. J. Pharm. Belg. 1991, 46, 241–246. [Google Scholar]
- Arnao, M.B.; Cano, A.; Acosta, M. The hydrophilic and lipophilic contribution to total antioxidant activity. Food Chem. 2001, 73, 239–244. [Google Scholar] [CrossRef]
- Ke, D.; Saltveit, M.E. Effect of calcium and auxin on russet spotting and phenylalanine ammonia-lyase activity in iceberg lettuce. HortScience 1986, 21, 1169–1171. [Google Scholar]
- Talcott, S.T.; Howard, L.R. Phenolic autoxidation is responsible for color degradation in processed carrot puree. J. Agric. Food Chem. 1999, 47, 2109–2115. [Google Scholar] [CrossRef] [PubMed]
- EN ISO 4833: 2003; Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Enumeration of Microorganisms—Colony-Count Technique at 30 °C. International Organization for Standardization: Geneva, Switzerland, 2003.
- ISO 15214: 1998; Microbiology of Food and Animal Feeding Stuffs—Horizontal Method for the Enumeration of Mesophilic Lactic Acid Bacteria—Colony-Count Technique at 30 °C. International Organization for Standardization: Geneva, Switzerland, 1998.
- STATISTICA (Data Analysis Software System), Version 8.0; StatSoft, Inc.: Tulsa, OK, USA, 2007.
- Jackson, J.E. A User’s Guide to Principal Components; John Wiley & Sons: New York, NY, USA, 1991. [Google Scholar]
- Larrigaudière, C.; Lentheric, I.; Puy, J.; Pintó, E. Biochemical characterisation of core browning and brown heart disorder in pear by multivariate analysis. Postharvest Biol. Technol. 2004, 31, 29–39. [Google Scholar] [CrossRef]
- Simões, A.; Allende, A.; Tudela, J.A.; Puschmann, R.; Gil, M.I. Optimum controlled atmospheres minimise respiration rate and quality losses while increase phenolic compounds of baby carrots. LWT 2011, 44, 277–283. [Google Scholar] [CrossRef]
- Alasalvar, C.; Al-Farsi, M.; Quantick, P.C.; Shahidi, F.; Wiktorowicz, R. Effect of chill storage and modified atmosphere packaging (MAP) on antioxidant activity, anthocyanins, carotenoids, phenolics and sensory quality of ready to eat shredded orange and purple carrots. Food Chem. 2005, 89, 69–76. [Google Scholar] [CrossRef]
- Izumi, H.; Watada, A.E.; Ko, N.P.; Douglas, W. Controlled atmosphere storage of carrot slices, sticks and shreds. Postharvest Biol. Technol. 1996, 9, 165–172. [Google Scholar] [CrossRef]
- Babic, I.; Amiot, M.J.; Nguyen-the, C.; Aubert, S. Changes in phenolics content in fresh ready-to-use shredded carrots during storage. J. Food Sci. 1993, 58, 351–356. [Google Scholar] [CrossRef]
- Kakiomenou, K.; Tassou, C.; Nychas, G.J. Microbiological, physicochemical and organoleptic changes of shredded carrots stored under modified storage. Int. J. Food Sci. 1996, 31, 359–366. [Google Scholar] [CrossRef]
- Zucker, M.; Levy, C.C. Some Factors which Affect the Synthesis of Chlorogenic Acid in Disks of Potato Tuber. Plant Physiol. 1959, 34, 108–112. [Google Scholar] [CrossRef]
- Allende, A.; Luo, Y.; McEvoy, J.L.; Artes, F.; Wang, C.Y. Microbial and quality changes in minimally processed baby spinach leaves stored under super atmospheric oxygen and modified atmosphere conditions. Postharvest Biol. Technol. 2004, 33, 51–59. [Google Scholar] [CrossRef]
- Montanez, J.; Oliveira, F.; Frías, J.; Pinelo, M.; Mahajan, P.; Cunha, L.; Manso, M.C. Design of perforation-mediated modified atmosphere packaging for shredded carrots: Mathematical modelling and experimental validation. Proc. Fla. State Hort. Soc. 2005, 118, 423–428. [Google Scholar]
- Oliveira, M.; Abadias, M.; Usall, J.; Torres, R.; Teixidó, N.; Vinas, I. (2015). Application of Modified Atmosphere Packaging as a safety approach to fresh-cut fruits and vegetables—A review. Trends Food Sci Technol. 2015, 46, 13–26. [Google Scholar] [CrossRef]
- Caleb, O.J.; Mahajan, P.V.; Al-Said, F.A.; Opara, U.L. Modified Atmosphere Packaging Technology of Fresh and Fresh-cut Produce and the Microbial Consequences-A Review. Food Bioprocess Technol. 2013, 6, 303–329. [Google Scholar] [CrossRef]
- Odumeru, J.A.; Boulter, J.; Knight, K.; Lu, X.; McKellar, R. Assessment of a wash treatment with warm chlorinated water to extend the shelf-life of ready-to-use lettuce. J. Food Qual. 2003, 26, 197–209. [Google Scholar] [CrossRef]
- Klaiber, R.G.; Baur, S.; Koblo, A.; Carle, R. Influence of Washing Treatment and Storage Atmosphere on Phenylalanine Ammonia-Lyase Activity and Phenolic Acid Content of Minimally Processed Carrot Sticks. J. Agric. Food Chem. 2005, 53, 1065–1072. [Google Scholar] [CrossRef] [PubMed]
- Tudela, J.A.; Cantos, E.; Espín, J.C.; Tomás-Barberán, F.A.; Gil, M.I. Induction of antioxidant flavonols biosynthesis in fresh-cut potatoes. Effect of domestic cooking. J. Agric. Food Chem. 2002, 50, 5925–5931. [Google Scholar] [CrossRef] [PubMed]
- Santos, D.I.; Faria, D.L.; Lourenço, S.C.; Alves, V.D.; Saraiva, J.A.; Vicente, A.A.; Moldão-Martins, M. Heat Treatment and Wounding as Abiotic Stresses to Enhance the Bioactive Composition of Pineapple By-Products. Appl. Sci. 2021, 11, 4313. [Google Scholar] [CrossRef]
- Assis, J.S.; Maldonado, R.; Muñoz, T.; Escribano, M.I.; Merodio, C. Effect of high carbon dioxide concentration on PAL activity and phenolic contents in ripening cherimoya fruit. Postharvest Biol. Technol. 2001, 23, 33–39. [Google Scholar] [CrossRef]
- Martínez-Hernández, G.B.; Amodio, M.L.; Colelli, G. Potential use of microwave treatment onfresh-cut carrots: Physical, chemical and microbiological aspects. J. Sci. Food Agric. 2016, 96, 2063–2072. [Google Scholar] [CrossRef]
- Breidt, F., Jr.; Costilow, R.N. Processing and safety. In Acidified Foods: Principles of Handling and Preservation; Fleming, H.P., Costilow, R.N., Eds.; Pickle Packers International, Inc.: St. Charles, IL, USA, 2004; pp. 5-1–5-15. [Google Scholar]
- Manzocco, L.; Plazzotta, S.; Maifreni, M.; Calligaris, S.; Anese, M.; Nicoli, M.C. Impact of UV-C light on storage quality of fresh-cut pineapple in two different packages. LWT 2016, 65, 1138–1143. [Google Scholar] [CrossRef]
- Collado, E.; Klug, T.V.; Martínez-Hernández, G.B.; Artés-Hernández, F.; Martínez-Sánchez, A.; Aguayo, E.; Artés, F.; Fernández, J.; Gómez, P.A. UV-C pretreatment of fresh-cut faba beans (Vicia faba) for shelf life extension: Effects of domestic microwaving for consumption. Food Sci. Technol. Int. 2020, 26, 140–150. [Google Scholar] [CrossRef]


| Quality Measurements | Mean ± SD |
|---|---|
| Bioactive compounds and related responses | |
| Total phenolic content (TPC, mg CAE/100 g) | 85.3 ± 2.2 |
| Chlorogenic acid (CA, mg/100 g) | 12.30 ± 1.17 |
| Total carotenoid content (TCC, mg β-carotene eq/100 g) | 18.2 ± 0.3 |
| Phenylalanine ammonia lyase (PAL, U/100 g) | 28.9 ± 2.5 |
| Antioxidant capacity (AOx, mg TEAC/100 g) | 66.7 ± 6.2 |
| pH/SSC | |
| pH | 6.4 ± 0.1 |
| Soluble solids content (SSC, °Brix) | 8.3 ± 0.1 |
| Microbial load | |
| Total aerobic plate count (TAPC, Log10[CFU/g]) | 5.8 ± 0.1 |
| Lactic acid bacteria (LAB, Log10[CFU/g]) | 3.5 ± 0.1 |
| Yeast and mould (Y&M, Log10[CFU/g]) | 3.2 ± 0.2 |
| Variable | Component 1 | Component 2 |
|---|---|---|
| TPC | 0.179766 | 0.803158 |
| CA | 0.071275 | 0.908997 |
| PAL | 0.083203 | 0.885018 |
| pH | 0.774510 | −0.154230 |
| Rejection | −0.913445 | 0.193772 |
| O2 | 0.788420 | 0.231341 |
| CO2 | −0.889783 | −0.342440 |
| TAPC | −0.889864 | 0.398211 |
| LAB | −0.942574 | 0.126346 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alegria, C.; Gonçalves, E.M.; Moldão-Martins, M.; Abreu, M. Multi-Target Alternative Approaches to Promoting Fresh-Cut Carrots’ Bioactive and Fresh-like Quality. Foods 2022, 11, 2422. https://doi.org/10.3390/foods11162422
Alegria C, Gonçalves EM, Moldão-Martins M, Abreu M. Multi-Target Alternative Approaches to Promoting Fresh-Cut Carrots’ Bioactive and Fresh-like Quality. Foods. 2022; 11(16):2422. https://doi.org/10.3390/foods11162422
Chicago/Turabian StyleAlegria, Carla, Elsa M. Gonçalves, Margarida Moldão-Martins, and Marta Abreu. 2022. "Multi-Target Alternative Approaches to Promoting Fresh-Cut Carrots’ Bioactive and Fresh-like Quality" Foods 11, no. 16: 2422. https://doi.org/10.3390/foods11162422
APA StyleAlegria, C., Gonçalves, E. M., Moldão-Martins, M., & Abreu, M. (2022). Multi-Target Alternative Approaches to Promoting Fresh-Cut Carrots’ Bioactive and Fresh-like Quality. Foods, 11(16), 2422. https://doi.org/10.3390/foods11162422







