Suitability of Banana and Plantain Fruits in Modulating Neurodegenerative Diseases: Implicating the In Vitro and In Vivo Evidence from Neuroactive Narratives of Constituent Biomolecules
Abstract
1. Introduction
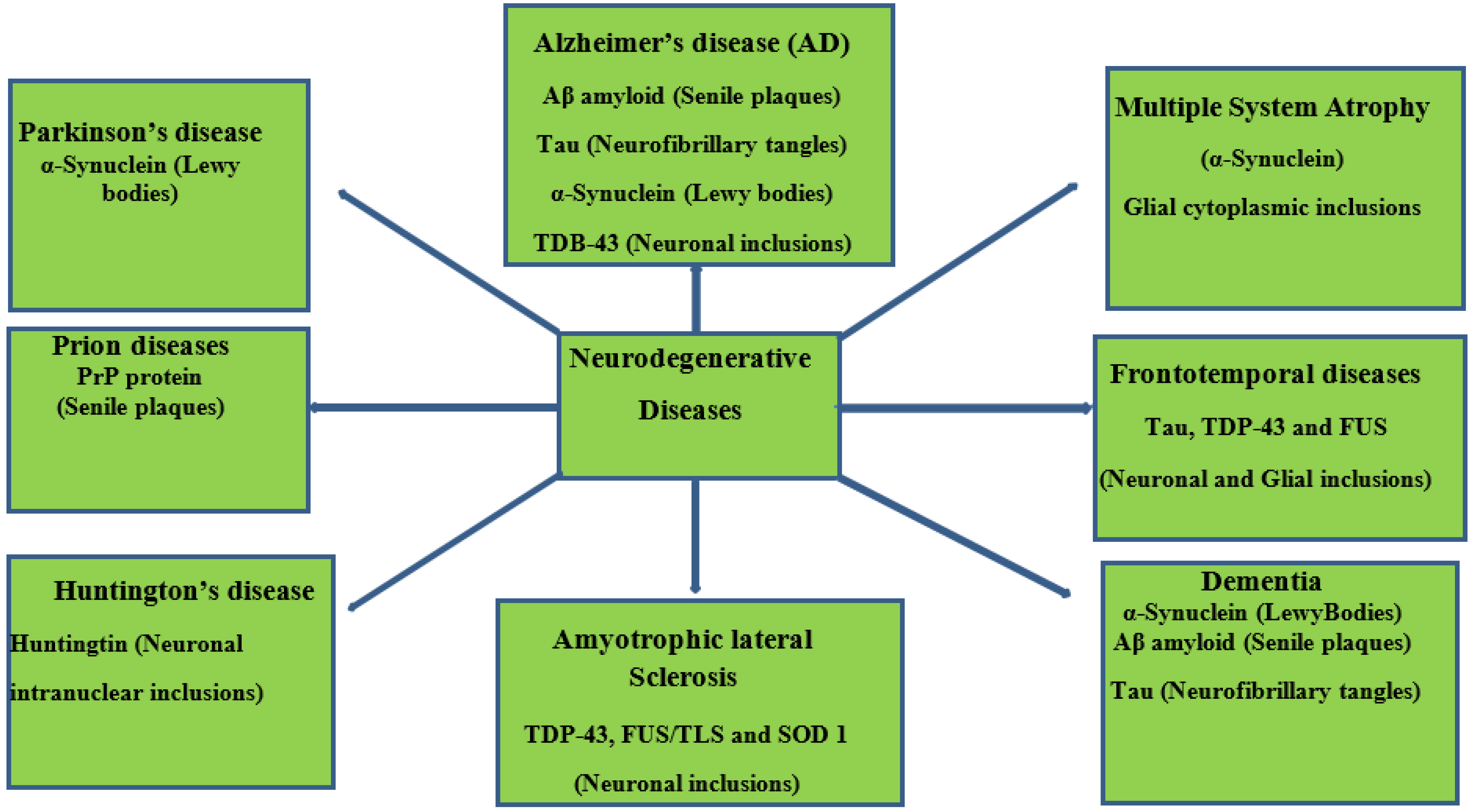
2. Overview and Prevalence of Neurodegenerative Diseases
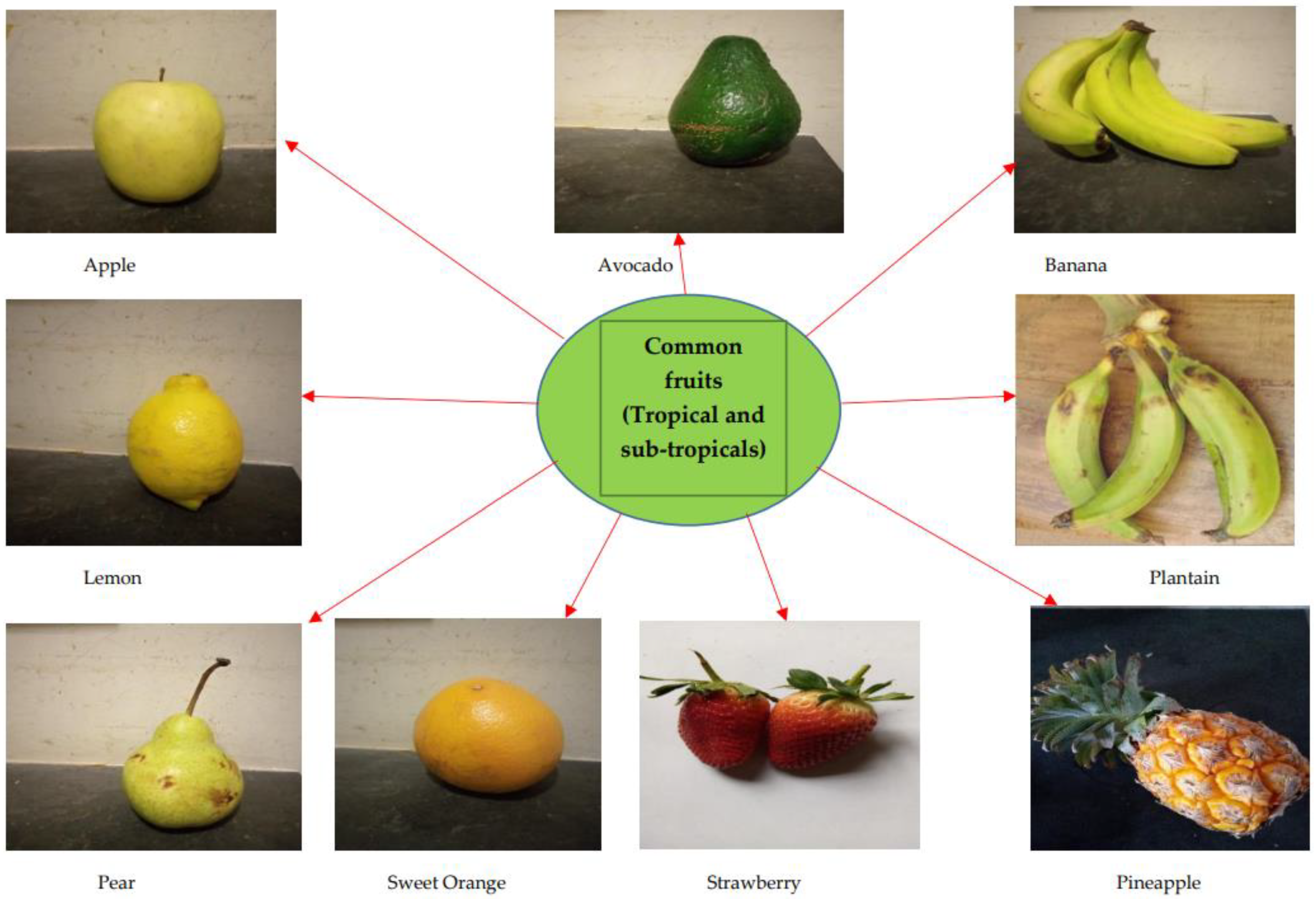
3. Fruits in Neurodegenerative Prevention/Management
4. Neuroprotective Mechanistic Narratives of Active Principles and Mineral Elements Listed in Banana and Plantain fruits
4.1. Tannins
4.2. Phenolic Acid
4.3. Quercetin
4.4. Rutin
4.5. Carbohydrates
4.6. Lipids
4.7. Magnesium
4.8. Zinc
4.9. Copper
4.10. Alkaloid
4.11. Saponin
4.12. Phytate
4.13. Vitamin B
4.14. Vitamin C (Ascorbic Acid)
4.15. Vitamin E (Tocopherol)
4.16. Selenium
4.17. Phytosterol
4.18. Terpenoid
4.19. Glycosides
4.20. Anthraquinones
4.21. Anthocyanins
4.22. Arginine
4.23. β-Carotene
4.24. Lycopene
4.25. β-Cryptoxanthin
4.26. β-Sitosterol
4.27. Sesamin
4.28. Myricetin
4.29. Catechin
5. Concluding Remarks
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| NG108–15 | glioblastoma |
| PGC-1/Nrf-2/HO-1 | peroxisome proliferator-activated receptor coactivator/Nuclear factor2/Heme oxygenase |
| GFAP | glial fibrillary acidic protein |
| BDNF–TrkB-PI3K/Akt | brain-derived neutrophic factor–tropomyosin-related kinase B/ phosphatidylinositol |
| Bax/Bcl2 | Bcl2 associated x/apoptosis regulator |
| TNF-α | tumor necrosis factor-alpha |
| IL-1β | interleukin-1β |
| COX-2 | cyclooxygenase-2 |
| P38MAPK | p38 mitogen-activated protein kinase |
| PRP | Prion Protein Peptide |
| HT22 | hippocampus cell line |
| Aβ | Amyloid Beta Peptide |
| TrKA | tropomyosin receptor kinase A |
| ERK1/2 | extracellular signal-related kinases ½ |
| MPTP | 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine |
| p-JNK | p-Jun N-terminal kinase |
| p-ERK | p-extracellular signal-regulated kinase protein |
| MBP | maltose binding protein |
| DBH | dopamine-β-hydroxylase |
| MAP2 | microtubule-associated protein 2 |
| GAP 43 | growth-associated protein 43 |
| PSD-95 | postsynaptic density protein |
| KLK8 | Kallikrein 8 gene |
| SH-SY5Y | human neuroblastoma cell line |
| P53 | tumor protein |
| β-secretase 1 | Beta secretase 1 |
| γ-secretase | gamma secretase protein |
| NADH | nicotinamide adenine dinucleotide hydrogen |
| NADPH | nicotinamide adenine dinucleotide phosphate |
| PC12 | pheochromocytoma cell line |
| GABA | gamma-aminobutyric acid |
| ERβ | estrogen receptor beta gene |
| NB69 | Cellosaurus cell line |
| NAC | N-acetylcysteine |
| SNAP-25 | synaptosomal-associated protein 25 |
| CA1 | Cortical Area 1 |
| NF-kB | nuclear factor-kappa B cells |
| STAT3 | signal transducer and activator of transcription 3 |
| ERK/MMP-9 | extracellular signal-related kinases/matrix metallopeptidase-9 |
| gp91phox | 91-KD glycoprotein |
| TGF-β1 | transforming growth factor |
| SK-N-SH | neuroblastoma cell line |
| HIF-1α/LDHA | hypoxia-inducible factor-1 alpha/Lactate dehydrogenase A gene |
| Nrf-2/Keap1 | nuclear factor 2/Kelch-like ECH-associated protein 1 |
| GSK-3β | glycogen synthase kinase-3 beta |
| mTOR | mammalian target of rapamycin |
| 6-OHDA | 6-hydroxydopamine |
| SOD-1 | superoxide dismutase 1 |
| TDP-43 | TAR-DNA-binding protein 43 |
| FUS/TLS | fused in sarcoma/translocated in liposarcoma |
| anti-CHE | anti-cholinesterase activity |
| ROS | reactive oxygen species |
| HPLC | High-Performance Liquid Chromatography |
| NMR | Nuclear Magnetic Resonance |
| HNMR | Proton Nuclear Magnetic Resonance |
| FTIR | Fourier-Transform Infrared Spectroscopy |
| GAE | gallic acid equivalent |
| QE | quercetin equivalent |
| CE | catechin equivalent |
References
- González-Aguilar, G.; Robles-Sánchez, R.M.; Martínez-Téllez, M.A.; Olivas, G.I.; Alvarez-Parrilla, E.; de la Rosa, L.A. Bioactive compounds in fruits: Health benefits and effect of storage conditions. Stewart Postharvest Rev. 2008, 4, 1–10. [Google Scholar]
- Dragović-Uzelac, V.; Levaj, B.; Bursać, D.; Pedisić, S.; Radojčić, I.; Biško, A. Total Phenolics and Antioxidant Capacity Assays of Selected Fruits. Agric. Conspec. Sci. 2007, 72, 279–284. [Google Scholar]
- Coats, R.; Martirosyan, D. The effects of bioactive compounds on biomarkers of obesity. Funct. Foods Health Dis. 2015, 5, 365–380. [Google Scholar] [CrossRef]
- Calixto, N.O.; Pinto, M.E.F.; Ramalho, S.D.; Burger, M.C.M.; Bobey, A.F.; Young, M.C.M.; Bolzani, V.S.; Pinto, A.C. The Genus Psychotria: Phytochemistry, Chemotaxonomy, Ethnopharmacology, and Biological Properties. J. Braz. Chem. Soc. 2016, 27, 1355–1378. [Google Scholar]
- Karasawa, M.M.G.; Mohan, C. Fruits as Prospective Reserves of Bioactive Compounds: A Review. Nat. Prod. Bioprospect. 2018, 8, 335–346. [Google Scholar] [CrossRef] [PubMed]
- Rendeiro, C.; Guerreiro, J.D.T.; Williams, C.M.; Spencer, J.P.E. Flavonoids as modulators of memory and learning: Molecular interactions resulting in behavioral effects. Proc. Nutr. Soc. 2012, 71, 246–262. [Google Scholar] [CrossRef]
- Solanki, I.; Parihar, P.; Mansuri, M.L.; Parihar, M.S. Flavonoid-Based Therapies in the Early Management of Neurodegenerative Diseases. Adv. Nutr. 2015, 6, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Aquilano, K.; Baldelli, S.; Rotilio, G.; Ciriolo, M.R. Role of nitric oxide synthases in Parkinson’s disease: A review on the antioxidant and anti-inflammatory activity of polyphenols. Neurochem. Res. 2008, 33, 2416–2426. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.S.; Acharya, A.; Ray, R.S.; Agrawal, R.; Raghuwanshi, R.; Jain, P. Cellular and molecular mechanisms of curcumin in prevention and treatment of disease. Crit. Rev. Food Sci. Nutr. 2019, 60, 887–939. [Google Scholar] [CrossRef]
- Przedborski, S.; Vila, M.; Jackson-Lewis, V. Neurodegeneration: What is it and where are we? J. Clin. Investig. 2003, 111, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Johnston, M. Neurodegenerative disorders of childhood. In Nelson Textbook of Pediatrics, 20th ed.; Behrman, R.E., Kliegman, R.M., Jenson, H.B., Eds.; Elsevier: Philadelphia, PA, USA, 2016; p. 4264. [Google Scholar]
- Hardy, J. Pathways to Primary Neurodegenerative Disease. Mayo Clin. Proc. 1999, 74, 835–837. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.W.; Zhang, X.; Huang, W.J. Role of neuroinflammation in neurodegenerative diseases (Review). Mol. Med. Rep. 2016, 13, 3391–3396. [Google Scholar] [CrossRef] [PubMed]
- Mobbs, C.V.; Hof, P.R. Body Composition and Aging; Interdisciplinary Top Gerontology; Karger: Basel, Switzerland, 2010; Volume 37, pp. 175–192. [Google Scholar]
- Lasonde, M.; Candel, S.; Hacker, J.; Quadrio-Curzio, A.; Onishi, T.; Ramakrishnan, V.; McNutt, M. The Challenge of Neurodegenerative Diseases in an Aging Population; G7 Academies’ Joint Statement; The Royal Society: London, UK, 2017; pp. 1–2. [Google Scholar]
- Taylor, J.P.; Hardy, J.; Fischbeck, K.H. Toxic proteins in neurodegenerative disease. Science 2002, 296, 1991–1995. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Chan, D.C. Mitochondrial dynamics- fusion, fission, movement, and mitophagy in neurodegenerative diseases. Hum. Mol. Genet. 2009, 18, R169–R176. [Google Scholar] [CrossRef] [PubMed]
- Chin-Chan, M.; Navarro-Yepes, J.; Quintanilla-Vega, B. Environmental pollutants as risk factors for neurodegenerative disorders: Alzheimer and Parkinson diseases. Front. Cell. Neurosci. 2015, 124, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Dorsey, E.R.; Holloway, R.G.; Ravina, B.M. Biomarkers in Parkinson’s disease. Expert Rev. Neurother. 2006, 6, 823–831. [Google Scholar] [CrossRef]
- Campbell, I.L.; Krucker, T.; Steffensen, S.; Akwa, Y.; Powell, H.C.; Lane, T.; Carr, D.J.; Gold, L.H.; Henriksen, S.J.; Siggins, G.R. Structural and functional neuropathology in transgenic mice with CNS expression of IFN-alpha. Brain Res. 1999, 835, 46–61. [Google Scholar] [CrossRef]
- Ineichen, B. The epidemiology of dementia in Africa: A review. Soc. Sci. Med. 2000, 50, 1673–1677. [Google Scholar] [CrossRef]
- Okubadejo, N.U.; Bower, J.H.; Rocca, W.A.; Maraganore, D.M. Parkinson’s disease in Africa. A systematic review of epidemiologic and genetic studies. Mov. Disord. 2006, 21, 2150–2156. [Google Scholar] [CrossRef] [PubMed]
- Cilia, R.; Akpalu, A.; Cham, M.; Bonetti, A.; Amboni, M.; Faceli, E.; Pezzoli, G. Parkinson’s disease in sub-Saharan Africa: Step-by-step into the challenge. Neurodegener. Dis. Manag. 2011, 1, 193–202. [Google Scholar] [CrossRef]
- George-Carey, R.; Adeloye, D.; Chan, K.Y.; Paul, A.; Kolcić, I.; Campbell, H.; Rudan, I. An estimate of the prevalence of dementia in Africa: A systematic analysis. J. Glob. Health 2012, 2, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Marin, B.; Kacem, I.; Diagana, M.; Boulesteix, M.; Gouider, R.; Preux, P.M.; Couratier, P. Juvenile, and adult-onset ALS/MND among Africans: Incidence, phenotype, survival: A review. ALS 2012, 13, 276–283. [Google Scholar]
- Onyike, C.U.; Diehl-Schmid, J. The epidemiology of frontotemporal dementia. Int. Rev. Psychiatry 2013, 25, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Bower, J.H.; Maraganore, D.M.; McDonnell, S.K.; Rocca, W.A. Incidence of progressive supranuclear palsy and multiple system atrophy in Olmsted County, Minnesota, 1976 to 1990. Neurology 1997, 49, 1284–1288. [Google Scholar] [CrossRef]
- Coon, E.A.; Sletten, D.M.; Suarez, M.D.; Mandrekar, J.N.; Ahlskog, J.E.; Bower, J.H.; Matsumoto, J.Y.; Silber, M.H.; Benarroch, E.E.; Fealey, R.D.; et al. Clinical features and autonomic testing predict survival in multiple system atrophy. Brain 2015, 138, 3623–3631. [Google Scholar] [CrossRef] [PubMed]
- Köllensperger, M.; Geser, F.; Ndayisaba, J.; Boesch, S.; Seppi, K.; Ostergaard, K.; Dupont, E.; Cardozo, A.; Tolosa, E.; Abele, M.; et al. Presentation, diagnosis, and management of multiple system atrophy in Europe: Final analysis of the European multiple system atrophy registry. Mov. Disord. 2010, 25, 2604–2612. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.J.; Ismail, I.S. Potency of Selected Berries, Grapes, and Citrus Fruit as Neuroprotective Agents. Evid. Based Complement. Alternat. Med. 2020, 2020, 1–12. [Google Scholar] [CrossRef]
- Xia, E.Q.; Deng, G.F.; Guo, Y.J.; Li, H.B. Biological activities of polyphenols from grapes. Int. J. Mol. Sci. 2010, 11, 622–646. [Google Scholar] [CrossRef]
- Fu, L.; Xu, B.T.; Xu, X.R.; Gan, R.Y.; Zhang, Y.; Xia, E.Q.; Li, H.B. Antioxidant capacities and total phenolic contents of 62 fruits. Food Chem. 2011, 129, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Deng, G.F.; Shen, C.; Xu, X.R.; Kuang, R.D.; Guo, Y.J.; Zeng, L.S.; Gao, L.L.; Lin, X.; Xie, J.F.; Xia, E.Q.; et al. Potential of Fruit Wastes as Natural Resources of Bioactive Compounds. Int. J. Mol. Sci. 2012, 13, 8308–8323. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Li, S.; Li, H.B.; Deng, G.F.; Ling, W.H.; Wu, S.; Xu, X.R.; Chen, F. Antiproliferative activity of peels, pulps, and seeds of 61 fruits. J. Funct. Foods 2013, 5, 1298–1309. [Google Scholar] [CrossRef]
- Ishurda, O.; John, F.K. The anti-cancer activity of polysaccharides prepared from Libyan dates (Phoenix dactylifera L.). Carbohydr. Polym. 2005, 59, 531–535. [Google Scholar] [CrossRef]
- Al-Qarawi, A.A.; Abdel-Rahman, H.; Mousa, H.M.; Ali, B.H.; El-Mougy, S.A. Nephroprotective action of Phoenix dactylifera in Gentamicin-Induced Nephrotoxicity. Pharm. Biol. 2008, 46, 227–230. [Google Scholar] [CrossRef]
- Saafi, E.B.; Louedi, M.; Elfeki, A.; Zakhama, A.; Najjar, M.F.; Hammami, M.; Achour, L. Protective effect of date palm fruit extract (Phoenix dactylifera L.) on dimethoate induced oxidative stress in rat liver. Exp. Toxicol. Pathol. 2011, 63, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Kasi, P.B.; Kotormán, M. Avocado juice prevents the formation of Trypsin amyloid-like fibrils in aqueous ethanol. Nat. Prod. Commun. 2019, 14, 1–5. [Google Scholar] [CrossRef]
- Joseph, J.A.; Shukitt-Hale, B.; Denisova, N.A.; Prior, R.L.; Cao, G.; Martin, A.; Taglialatela, G.; Bickford, P.C. Long-term dietary strawberry, spinach, or vitamin E supplementation retards the onset of age-related neuronal signal-transduction and cognitive behavioral deficits. J. Neurosci. 1998, 18, 8047–8055. [Google Scholar] [CrossRef]
- Joseph, J.A.; Shukitt-Hale, B.; Denisova, N.A.; Bielinski, D.; Martin, A.; McEwen, J.J.; Bickford, J.J. Reversals of age-related declines in neuronal signal transduction, cognitive, and motor behavioral deficits with blueberry, spinach, or strawberry dietary supplementation. J. Neurosci. 1999, 19, 8114–8121. [Google Scholar] [CrossRef] [PubMed]
- Gambino, G.; Allegra, M.; Sardo, P.; Attanzio, A.; Tesoriere, L.; Livrea, M.A.; Ferraro, G.; Carletti, F. Brain distribution and modulation of neuronal excitability by indicaxanthin from Opuntia ficus indica administered at nutritionally relevant amounts. Front. Aging Neurosci. 2018, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Oduje, A.A.; Oboh, G.; Ayodele, A.J.; Stephen, A.A. Assessment of the nutritional, antinutritional, and antioxidant capacity of unripe, ripe, and overripe plantain (Musa paradisiaca) peel. Int. J. Curr. Adv. Res. 2015, 3, 63–72. [Google Scholar]
- Ogbonna, O.A.; Izundu, A.I.; Okoye, N.H.; Ikeyi, A.P. Phytochemical compositions of fruits of three Musa species at three stages of development. IOSR J. Pharm. 2016, 11, 48–59. [Google Scholar]
- Aboul-Enein, A.M.; Salama, Z.A.; Gaafar, A.A.; Aly, H.F.; Abou-Elella, F.; Ahmed, H.A. Identification of phenolic compounds from the banana peel (Musa paradisiaca L.) as antioxidant and antimicrobial agents. J. Chem. Pharm. 2016, 8, 46–55. [Google Scholar]
- Agama-Acevedo, E.; Sañudo-Barajas, J.A.; Vélez De La Rocha, R.; González-Aguilar, G.A.; Bello-Peréz, L.A. Potential of plantain peels flour (Musa paradisiaca L.) as a source of dietary fiber and antioxidant compound. CYTA J. Food 2016, 14, 117–123. [Google Scholar] [CrossRef]
- Aina, O.O.; Oyedeji, M.B.; Adegboyega, D.A.; Owoloja, A.O. Phytochemical screening of some selected banana peels of Musa acuminata species. Int. J. Agric. 2019, 4, 68–78. [Google Scholar]
- Boua, B.B.; Ouattara, D.; Traoré, L.; Mamyrbekova-Békro, J.A.; Békro, Y.A. Effect of domestic cooking on the total phenolic, flavonoid and condensed tannin content from plantain of Côte d’Ivoire. J. Mater. Environ. Sci. 2020, 11, 396–403. [Google Scholar]
- Ashafaq, M.; Tabassum, H.; Vishnoi, S.; Salman, M.; Raisuddin, S.; Parvez, S. Tannic acid alleviates lead acetate-induced neurochemical perturbations in rat brain. Neurosci. Lett. 2016, 617, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Salman, M.; Tabassum, H.; Parvez, S. Nrf2/HO-1 mediates the neuroprotective effects of pramipexole by attenuating oxidative damage and mitochondrial perturbation after traumatic brain injury in rats. Dis. Model Mech. 2020, 13, 1–11. [Google Scholar] [CrossRef]
- Sulaiman, S.F.; Yusoff, N.A.M.; Eldeen, I.M.; Seow, E.M.; Sajak, A.A.; Supriatno; Ooi, K.L. Correlation between total phenolic and mineral contents with antioxidant activity of eight Malaysian bananas (Musa sp.). J. Food Compost. Anal. 2011, 24, 1–10. [Google Scholar] [CrossRef]
- Chauhan, A.; Nagar, A.; Bala, K.; Sharma, Y. Comparative study of different parts of fruits of Musa Sp. on the basis of their antioxidant activity. Der Pharm. Lett. 2016, 8, 88–100. [Google Scholar]
- Al Amri, F.S.; Hossain, M.A. Comparison of total phenols, flavonoids, and antioxidant potential of local and imported ripe bananas. Egypt. J. Basic Appl. Sci. 2018, 5, 245–251. [Google Scholar] [CrossRef]
- Kamaayi, F.; Baah, F.A.; Ansah, F.O. Phenolic content, polyphenol oxidase activity, and antioxidant scavenging activity in three species of plantain in Ghana. Int. J. Sci. Res. 2020, 10, 551–557. [Google Scholar] [CrossRef]
- Fatemeh, S.R.; Saifullah, R.; Abbas, F.M.A.; Azhar, M.E. Total phenolics, flavonoids, and antioxidant activity of banana pulp and peel flours: Influence of variety and stage of ripeness. Int. Food Res. J. 2012, 19, 1041–1046. [Google Scholar]
- Oyeyinka, B.O.; Afolayan, A.J. Comparative and Correlational evaluation of the phytochemical constituents and antioxidant activity of Musa sinensis L. and Musa paradisiaca L. fruit compartments (Musaceae). Sci. World J. 2020, 2020, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Ssonko, U.L.; Muranga, F.I. Phenolic compounds identification and antioxidant activity in bananas of AAB and ABB genomes grown in Uganda. J. Health Popul. Nutr. 2020, 4, 1–8. [Google Scholar]
- Kevers, C.; Falkowski, M.; Tabart, J.; Defraigne, J.O.; Dommes, J.; Pincemail, J. Evolution of antioxidant capacity during storage of selected fruits and vegetables. J. Agric. Food Chem. 2007, 55, 8596–8603. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; Irwin, R.; Chen, S.; Hamilton, R.; Cadenas, E.; Brinton, R.D. Ovarian hormone loss induces bioenergetic deficits and mitochondrial β-amyloid. Neurobiol. Aging 2012, 33, 1507–1521. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.; Bahuguna, A.; Bhardwaj, M.; Khaket, T.P.; Kang, S.C. Carvacrol nano-emulsion evokes cell cycle arrest, apoptosis induction, and autophagy inhibition in a doxorubicin-resistant A549 cell line. Artif. Cells Nanomed. Biotechnol. 2018, 46, S664–S675. [Google Scholar] [CrossRef] [PubMed]
- Tsamo, C.V.P.; Herent, M.F.; Tomekpe, K.; Emaga, T.H.; Quetin-Leclercq, J.; Rogez, H.; Larondelle, Y.; Andre, C. Phenolic profiling in the pulp and peel of nine plantain cultivars (Musa sp.). Food Chem. 2015, 167, 197–204. [Google Scholar] [CrossRef]
- Jiménez-Aliaga, K.; Bermejo-Bescós, P.; Benedí, J.; Martín-Aragón, S. Quercetin and rutin exhibit antiamyloidogenic and fibril-disaggregating effects in vitro and potent antioxidant activity in APPswe cells. Life Sci. 2011, 89, 939–945. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Szretter, K.J.; Vermi, W.; Gilfillan, S.; Rossini, C.; Cella, M.; Barrow, A.D.; Diamond, M.S.; Colonna, M. IL-34 is a tissue-restricted ligand of CSF1R required for the development of Langerhans cells and microglia. Nat. Immunol. 2012, 13, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Javed, H.; Khan, M.M.; Ahmad, A.; Vaibhav, K.; Ahmad, M.E.; Khan, A.; Ashafaq, M.; Islam, F.; Siddiqui, M.S.; Safhi, M.M.; et al. Rutin prevents cognitive impairments by ameliorating oxidative stress and neuroinflammation in the rat model of sporadic dementia of Alzheimer’s type. Neuroscience 2012, 210, 340–352. [Google Scholar] [CrossRef]
- Na, J.Y.; Kim, S.; Song, K.; Kwon, J. Rutin alleviates Prion peptide-induced cell death through inhibiting apoptotic pathway activation in dopaminergic neuronal cells. Cell. Mol. Neurobiol. 2014, 34, 1071–1079. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Zhang, M.; Ramos, C.A.; Durett, A.; Liu, E.; Dakhova, O.; Liu, H.; Creighton, C.J.; Gee, A.P.; Heslop, H.E.; et al. Closely related T-memory stem cells correlate with in vivo expansion of CAR.CD19-T cells and are preserved by IL-7 and IL-15. Blood 2014, 123, 3750–3759. [Google Scholar] [CrossRef] [PubMed]
- Ganeshpurkar, A.; Saluja, A.K. The pharmacological potential of Rutin. Saudi Pharm. J. 2017, 25, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Zhang, X.; Wang, W.; Liu, R.; Zhao, K.; Liu, M.; Gong, W.; Ning, B. Neuroprotective mechanisms of rutin for spinal cord injury through anti-oxidation and anti-inflammation and inhibition of the p38 mitogen-activated protein kinase pathway. Neural Regen. Res. 2018, 13, 128–134. [Google Scholar]
- Adepoju, O.T.; Sunday, B.E.; Folaranmi, O.A. Nutrient composition and contribution of plantain (Musa paradisiaca) products to dietary diversity of Nigerian consumers. Afr. J. Biotechnol. 2012, 11, 13601–13605. [Google Scholar] [CrossRef]
- Okareh, O.T.; Adeolu, A.T.; Adepoju, O.T. Proximate and mineral composition of plantain (Musa paradisiaca) wastes flour; a potential nutrients source in the formulation of animal feeds. Afr. J. Food Sci. 2015, 6, 53–57. [Google Scholar] [CrossRef]
- Hapsari, L.; Lestari, D.A. Fruit characteristics and nutrient values of four Indonesian banana cultivars (Musa spp.) at different genomic groups. AGRAVITA J. Agric. Sci. 2016, 38, 303–311. [Google Scholar] [CrossRef][Green Version]
- Siji, S.; Nandini, P.V. Chemical and nutrient composition of selected banana varieties of Kerala. Int. J. Adv. Eng. Manag. Sci. 2017, 3, 401–404. [Google Scholar]
- Ho, Y.S.; Yu, M.S.; Yang, X.F.; So, K.F.; Yuen, W.H.; Chang, R.C.C. Neuroprotective effects of polysaccharides from wolfberry, the fruits of Lycium barbarum, against homocysteine- induced toxicity in rat cortical neurons. J. Alzheimer’s Dis. 2010, 19, 813–827. [Google Scholar] [CrossRef]
- Chiricozzi, A.; De Simone, C.; Fossati, B.; Peris, K. Emerging treatment options for the treatment of moderate to severe plaque psoriasis and psoriatic arthritis: Evaluating bimekizumab and its therapeutic potential. Psoriasis Targets Ther. 2019, 9, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Hasanah, R.; Daningsih, E.; Titin, T. The analysis of nutrient and fiber content of banana (Musa paradisiaca) sold in Pontianak, Indonesia. Biofarmasi J. Nat. Prod. Biochem. 2017, 15, 21–25. [Google Scholar] [CrossRef]
- Abubakar, U.S.; Yusuf, K.M.; Safiyanu, I.; Abdullahi, S.; Saidu, S.R.; Abdu, G.T.; Indee, A.M. Proximate and mineral composition of corn cob, banana, and plantain peels. Int. J. Food Sci. 2016, 1, 25–27. [Google Scholar]
- Adamu, A.S.; Ojo, I.O.; Oyetunde, J.G. Evaluation of nutritional values in ripe, unripe, boiled, and roasted plantain (Musa paradisiaca L.) pulp and peel. Eur. J. Basic Appl. Sci. 2017, 4, 9–12. [Google Scholar]
- Shadrach, I.; Banji, A.; Adebayo, O. Nutraceutical potential of ripe and unripe plantain peels: A comparative study. Chem. Int. 2020, 6, 83–90. [Google Scholar]
- Kirkland, A.E.; Sarlo, G.L.; Holton, K.F. The role of magnesium in neurological disorders. Nutrients 2018, 10, 730. [Google Scholar] [CrossRef] [PubMed]
- Annor, G.A.; Asamoah-Bonti, P.; Sakyi-Dawson, E. Fruit physical characteristics, proximate, mineral and starch characterization of FHIA 19 and FHIA 20 plantain and FHIA 03 cooking banana hybrids. SpringerPlus 2016, 5, 796. [Google Scholar] [CrossRef] [PubMed]
- Seth, R.; Corniola, R.S.; Gower-Winter, S.D.; Morgan, T.J., Jr.; Bishop, B.; Levenson, C.W. Zinc deficiency induces apoptosis via mitochondrial p53- and caspase-dependent pathways in human neuronal precursor cells. J. Trace Elem. Med. Biol. 2015, 30, 59–65. [Google Scholar] [CrossRef]
- Chen, N.N.; Zhao, D.J.; Sun, Y.X.; Wang, D.D.; Ni, H. Long-Term Effects of Zinc Deficiency and Zinc Supplementation on Developmental Seizure-Induced Brain Damage and the Underlying GPR39/ZnT-3 and MBP Expression in the Hippocampus. Front. Neurosci. 2019, 13, 1–10. [Google Scholar] [CrossRef]
- Marasco, D.; Vicidomini, C.; Krupa, P.; Cioffi, F.; Huy, P.D.Q.; Li, M.S.; Florio, D.; Broersen, K.; Francesca De Pandis, M.; Roviello, G.N. Plant isoquinoline alkaloids as potential neuro drugs: A comparative study of the effects of benzo[c]phenanthridine and berberine-based compounds on β-amyloid aggregation. Chem. Biol. Interact. 2021, 334, 109300. [Google Scholar] [CrossRef]
- Konar, A.; Shah, N.; Singh, R.; Saxena, N.; Kaul, S.C.; Wadhwa, R.; Thakur, M.K. Protective Role of Ashwagandha leaf extract and its component Withanone on scopolamine-induced changes in the brain and brain-derived cells. PLoS ONE 2011, 6, e27265. [Google Scholar] [CrossRef]
- Konar, A.; Gupta, R.; Shukla, R.K.; Maloney, B.; Khanna, V.K.; Wadhwa, R.; Lahiri, D.K.; Thakur, M.K. M1 muscarinic receptor is a key target of neuroprotection, neuroregeneration, and memory recovery by i-Extract from Withania somnifera. Sci. Rep. 2019, 9, 13990. [Google Scholar] [CrossRef]
- Oyeyinka, B.O.; Afolayan, A.J. Comparative Evaluation of the Nutritive, Mineral, and Antinutritive Composition of Musa sinensis L. (Banana) and Musa paradisiaca L. (Plantain). Plants 2019, 8, 598. [Google Scholar] [CrossRef] [PubMed]
- Oyeyemi, S.D.; Otoide, J.E.; Obembe, M.O.; Ogunleye, B.J. Nutritional and phytochemical assessment of Musa sapientum var. velutina. The Pharm. Chem. J. 2019, 6, 56–64. [Google Scholar]
- Chen, Y.; Sun, H.; Huang, L.; Li, J.; Zhou, W.; Chang, J. Neuroprotective effect of Radix trichosanthis saponins on subarachnoid hemorrhage. Evid. Based Complement. Alternat. Med. 2015, 2015, 1–10. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Khawas, P.; Das, A.J.; Sit, N.; Badwaik, L.S.; Deka, S.C. Nutritional composition of culinary Musa ABB at Different Stages of Development. Am. J. Food Technol. 2014, 2, 80–87. [Google Scholar] [CrossRef]
- Anekonda, T.S.; Quinn, J.F.; Harris, C.; Frahler, K.; Wadsworth, T.L.; Woltjer, R.L. L-type voltage-gated calcium channel blockade with isradipine as a therapeutic strategy for Alzheimer’s disease. Neurobiol. Dis. 2011, 41, 61–70. [Google Scholar] [CrossRef]
- Thinh, B.B.; Trong, L.V.; Lam, L.T.; Hien, V.T.T. Nutritional value of persimmon, banana, lemon, and longan cultivated in Northern Vietnam. IOP Conf. Ser. Earth Environ. Sci. 2021, 640, 022030. [Google Scholar] [CrossRef]
- Velumani, S. Phytochemical screening and antioxidant activity of banana peel. Int. J. Adv. Res. Innov. Ideas Educ. 2016, 2, 91–102. [Google Scholar]
- Kim, S.M.; Chung, M.J.; Ha, T.J.; Choi, H.N.; Jang, S.J.; Kim, S.O.; Chun, M.H.; Do, S.I.; Choo, Y.K.; Park, Y.I. Neuroprotective effects of black soybean anthocyanins via inactivation of ASK1-JNK/p38 pathways and mobilization of cellular sialic acids. Life Sci. 2012, 90, 874–882. [Google Scholar] [CrossRef]
- Lau, F.C.; Joseph, J.A.; McDonald, J.E.; Kalt, W. Attenuation of iNOS and COX2 by blueberry polyphenols is mediated through the suppression of NF-κB activation. J. Funct. Foods 2009, 1, 274–283. [Google Scholar] [CrossRef]
- Ma, C.; Zhao, L.L.; Zhao, H.J.; Cui, J.W.; Li, W.; Wang, N.Y. Lentivirus-mediated MDA7/IL24 expression inhibits the proliferation of hepatocellular carcinoma cells. Mol. Med. Rep. 2018, 17, 5764–5773. [Google Scholar] [CrossRef]
- Agoreyo, B.O.; Agoreyo, F.O.; Omigie, M.I. Antioxidant activity, phytochemical and antioxidant levels of Musa paradisciaca L. and Musa sapientum L. at various ripening stages. Eur. Food Res. Technol. 2017, 5, 41–59. [Google Scholar]
- Nakaso, K.; Tajima, N.; Horikoshi, Y.; Nakasone, M.; Hanaki, T.; Kamizaki, K.; Matsura, T. The estrogen receptor β-PI3K/Akt pathway mediates the cytoprotective effects of tocotrienol in a cellular Parkinson’s disease model. Biochim. Biophys. Acta BBA Mol. Basis Dis. 2014, 1842, 1303–1312. [Google Scholar] [CrossRef] [PubMed]
- Al-Ahmary, K.M. Selenium content in selected foods from the Saudi Arabia market and estimation of the daily intake. Arab. J. Chem. 2009, 2, 95–99. [Google Scholar] [CrossRef]
- Choi, Y.; Kim, J.; Lee, H.S.; Kim, C.; Hwang, I.K.; Park, H.K.; Oh, C.H. Selenium content in representative Korean foods. J. Food Compos. Anal. 2009, 22, 117–122. [Google Scholar] [CrossRef]
- Al-Othman, A.M.; Al-Othman, Z.A.; El-Desoky, G.E.; Aboul-Soud, M.A.M.; Habila, M.A.; Giesy, J.P. Daily intake of selenium and concentrations in blood of residents of Riyadh City, Saudi Arabia. Environ. Geochem. Health 2012, 34, 417–431. [Google Scholar] [CrossRef]
- Moatkhef, F.; Ismail, H.; Agamy, N.; Aborhyem, S. Quantitative determination of selenium in the most common food items sold in Egypt. J. Egypt. Public Health Assoc. 2020, 95, 1–9. [Google Scholar] [CrossRef]
- Naziroğlu, M.; Kutluhan, S.; Yilmaz, M. Selenium and topiramate modulate brain microsomal oxidative stress values, Ca2+-ATPase activity, and EEG records in pentylenetetrazol-induced seizures in rats. J. Membr. Biol. 2008, 225, 39–49. [Google Scholar] [CrossRef]
- Naziroğlu, M.; Şenol, N.; Ghazizadeh, V.; Yyürüker, V. Neuroprotection induced by N-acetylcysteine and selenium against traumatic brain injury-induced apoptosis and calcium entry in the hippocampus of the rat. Cell. Mol. Neurobiol. 2014, 34, 895–903. [Google Scholar] [CrossRef]
- Klingberg, S.; Andersson, H.; Mulligan, A.; Bhaniani, A.; Welch, A.; Bingham, S.; Khaw, K.T.; Andersson, S.; Ellegard, L. Food sources of plant sterols in the EPIC Norfolk population. Eur. J. Clin. Nutr. 2008, 62, 695–703. [Google Scholar] [CrossRef]
- Rui, X.; Wenfang, L.; Jing, C.; Meng, C.; Chengcheng, D.; Jiqu, X.; Shuang, R. Neuroprotective effects of phytosterol esters against high cholesterol-induced cognitive deficits in the aged rat. Food Funct. 2017, 8, 1323–1332. [Google Scholar] [CrossRef]
- Kopylov, A.T.; Malsagova, K.A.; Stepanov, A.A.; Kaysheva, A.L. Diversity of Plant Sterols Metabolism: The impact on human health, sport, and accumulation of contaminating sterols. Nutrients 2021, 13, 1623. [Google Scholar] [CrossRef] [PubMed]
- Uzairu, S.M.; Kano, M.A. Assessment of phytochemical and mineral composition of unripe and ripe plantain (Musa paradisiaca) peels. Afr. J. Food Sci. 2021, 15, 107–112. [Google Scholar]
- Kasture, V.S.; Deshmukh, V.K.; Chopde, C.T. Anticonvulsant and behavioral actions of triterpene isolated from Rubia cordifolia Linn. Indian J. Exp. Biol. 2000, 38, 675–680. [Google Scholar]
- Yu, L.; Chen, C.; Wang, L.F.; Kuang, X.; Liu, K.; Zhang, H.; Du, J.R. Neuroprotective effect of Kaempferol glycosides against brain injury and neuroinflammation by inhibiting the activation of NF-κB and STAT3 in transient focal stroke. PLoS ONE 2013, 8, e55839. [Google Scholar] [CrossRef]
- Yang, J.; Zeng, Z.; Wu, T.; Yang, Z.; Liu, B.; Lan, T. Emodin attenuates high glucose-induced TGF-β1 and fibronectin expression in mesangial cells through inhibition of the NF-κB pathway. Exp. Cell Res. 2013, 319, 3182–3189. [Google Scholar] [CrossRef]
- Wang, T.; Fan, X.; Tang, T.; Fan, R.; Zhang, C.; Huang, Z.; Peng, W.; Gan, P.; Xiong, X.; Huang, W.; et al. Rhein, and rhubarb similarly protect the blood-brain barrier after experimental traumatic brain injury via gp91phox subunit of NADPH oxidase/ROS/ERK/ MMP-9 signaling pathway. Sci. Rep. 2016, 6, 37098. [Google Scholar] [CrossRef] [PubMed]
- Aurore, G.; Parfait, B.; Fahrasmane, L. Bananas, raw materials for making processed food products. Trends Food Sci. Technol. 2009, 20, 78–91. [Google Scholar] [CrossRef]
- Chen, Y.M.; Li, H.; Chiu, Y.S.; Huang, C.C.; Chen, W.C. Supplementation of L-arginine, L- glutamine, Vitamin C, Vitamin E, folic acid, and green tea extract enhances serum nitric oxide content and anti-fatigue activity in mice. Evid. Based Complement. Alternat. Med. 2020, 2020, 1–10. [Google Scholar] [CrossRef]
- Prakash, A.; Kumar, A. Implicating the role of lycopene in the restoration of mitochondrial enzymes and BDNF levels in β-amyloid induced Alzheimer’s disease. Eur. J. Pharmacol. 2014, 741, 104–111. [Google Scholar] [CrossRef]
- Hong, Y.G.; Roh, S.; Paik, D.; Jeong, S. Development of a reporter system for in vivo monitoring of γ-secretase activity in Drosophila. Mol. Cells 2017, 40, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Heng, Z.; Sheng, O.; Yan, S.; Lu, H.; Motorykin, I.; Gao, H.; Li, C.; Yang, Q.; Hu, C.; Kuang, R.; et al. Carotenoid profiling in the peel and pulp of 36 selected Musa varieties. Food Sci. Technol. Res. 2017, 23, 603–611. [Google Scholar] [CrossRef]
- Villaverde, J.J.; Oliveira, L.; Vilela, C.; Domingues, R.M.; Freitas, N.; Cordeiro, N.; Freire, C.S.R.; Silvestre, A.J.D. High valuable compounds from the unripe peel of several Musa species cultivated in Madeira Island (Portugal). Ind. Crops Prod. 2013, 42, 507–512. [Google Scholar] [CrossRef]
- Shi, C.; Liu, J.; Wu, F.; Zhu, X.; Yew, D.T.; Xu, J. β-sitosterol inhibits high cholesterol-induced platelet β-amyloid release. J. Bioenerg. Biomembr. 2011, 43, 691–697. [Google Scholar] [CrossRef]
- Shi, C.; Wu, F.; Zhu, X.C.; Xu, J. Incorporation of beta-sitosterol into the membrane increases resistance to oxidative stress and lipid peroxidation via estrogen receptor-mediated PI3K/GSK3beta signaling. Biochim. Biophys. Acta 2013, 1830, 2538–2544. [Google Scholar] [CrossRef] [PubMed]
- Mordi, R.C.; Fadiaro, A.E.; Owoeye, T.F.; Olanrewaju, I.O.; Uzoamaka, G.C.; Olorunshola, S.J. Identification by GC-MS of the components of oils of banana peels extract, phytochemical and antimicrobial analyses. Res. J. Phytochem. 2016, 10, 39–44. [Google Scholar] [CrossRef]
- Hsieh, P.F.; Hou, C.W.; Yao, P.W.; Wu, S.P.; Peng, Y.F.; Shen, M.L.; Lin, C.H.; Chao, Y.Y.; Chang, M.H.; Jeng, K.C. Sesamin ameliorates oxidative stress and mortality in kainic acid-induced status epilepticus by inhibition of MAPK and COX-2 activation. J. Neuroinflamm. 2011, 8, 57. [Google Scholar] [CrossRef]
- Xu, Z.; Liu, Y.; Yang, D.; Yuan, F.; Ding, J.; Chen, H.; Tian, H. Sesamin protects SH-SY5Y cells against mechanical stretch injury and promoting cell survival. BMC Neurosci. 2017, 18, 57. [Google Scholar] [CrossRef]
- Udomruk, S.; Kaewmool, C.; Phitak, T.; Pothacharoen, P.; Kongtawelert, P. Sesamin promotes neurite outgrowth under insufficient nerve growth factor condition in PC12 cells through the ERK1/2 pathway and SIRT1 modulation. Evid. Based Complement. Alternat. Med. 2020, 2020, 1–12. [Google Scholar] [CrossRef]
- Singh, B.; Singh, J.P.; Kaur, A.; Singh, N. Bioactive compounds in banana and their associated health benefits—A review. Food Chem. 2016, 206, 1–11. [Google Scholar] [CrossRef]
- Zhang, N.; Feng, H.; Liao, H.H.; Chen, S.; Yang, Z.; Deng, W.; Tang, Q.Z. Myricetin attenuated LPS induced cardiac injury in vivo and in vitro. Phytother. Res. 2018, 32, 459–470. [Google Scholar] [CrossRef] [PubMed]
- Rebello, L.P.G.; Ramos, A.M.; Pertuzatti, P.B.; Barcia, M.T.; Muñoz, N.C.; Hermosín-Gutiérrez, I. Flour of banana (Musa AAA) peel as a source of antioxidant phenolic compounds. Food Res. Int. 2014, 55, 397–403. [Google Scholar] [CrossRef]
- Rinaldo, D. Carbohydrate and bioactive compounds composition of starchy tropical fruits and tubers, in relation to pre and postharvest conditions: A review. J. Food Sci. 2020, 85, 249–259. [Google Scholar] [CrossRef]
- Vlachos, G.S.; Scarmeas, N. Dietary interventions in mild cognitive impairment and dementia. Dialogues Clin. Neurosci. 2019, 21, 69–82. [Google Scholar] [PubMed]
- Singh, P.; Sivanandama, T.M.; Konar, A.; Thakur, M.K. Role of nutraceuticals in cognition during aging and related disorders. Neurochem. Int. 2021, 143, 104928. [Google Scholar] [CrossRef]
- Tan, H.P.; Wong, D.Z.H.; Ling, S.K.; Chuah, C.H.; AbdulKadir, H. Neuroprotective activity of galloylated cyanogenic glucosides and hydrolyzable tannins isolated from leaves of Phyllagathis rotundifolia. Fitoterapia 2012, 83, 223–229. [Google Scholar] [CrossRef]
- Winiarska-Mieczan, A. Protective effect of tannic acid on the brain of adult rats exposed to cadmium and lead. Environ. Toxicol. Pharmacol. 2013, 36, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Calou, I.; Bandeira, M.A.; Aguiar-Galvão, W.; Cerqueira, G.; Siqueira, R.; Neves, K.R.; Brito, G.A.; Viana, G. Neuroprotective properties of a standardized extract from Myracrodruon urundeuva Fr. All. (Aroeira-Do-Sertão), as evaluated by a Parkinson’s disease model in rats. Parkinsons Dis. 2014, 2014, 1–12. [Google Scholar] [CrossRef]
- Zengin, G.; Locatelli, M.; Carradori, S.; Mocan, A.M.; Aktumsek, A. Total phenolics, flavonoids, condensed tannins content of eight Centaurea species and their broad inhibitory activities against cholinesterase, tyrosinase, α-amylase, and α-glucosidase. Not. Bot. Horti Agrobot. 2016, 44, 195–200. [Google Scholar] [CrossRef]
- Liu, X.; Yan, Y.; Li, F.; Zhang, D. Fruit and vegetable consumption and the risk of depression: A meta-analysis. Nutrition 2016, 32, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Türedi, S.; Yuluğ, E.; Alver, A.; Bodur, A.; İnce, I. A morphological and biochemical evaluation of the effects of quercetin on experimental sciatic nerve damage in rats. Exp. Ther. Med. 2018, 15, 3215–3224. [Google Scholar]
- Sharma, D.R.; Wani, W.Y.; Sunkaria, A.; Kandimalla, R.J.L.; Verma, D.; Cameotra, S.S.; Gill, K.D. Quercetin protects against chronic aluminum-induced oxidative stress and ensuing biochemical, cholinergic, and neurobehavioral impairments in rats. Neurotox. Res. 2013, 23, 336–357. [Google Scholar] [CrossRef] [PubMed]
- Wassem, M.; Parvez, S. Neuroprotective activities of curcumin and quercetin with potential relevance to mitochondrial dysfunction induced by oxaliplatin. Protoplasma 2015, 253, 417–430. [Google Scholar] [CrossRef]
- Khan, A.; Ali, T.; Ur Rehman, S.; Khan, M.S.; Alam, S.I.; Ikram, M.; Muhammad, T.; Saeed, K.; Badshah, H.; Kim, K.O. Neuroprotective effect of quercetin against the detrimental effects of LPS in the adult Mouse brain. Front. Pharmacol. 2018, 9, 1–16. [Google Scholar] [CrossRef] [PubMed]
- La Casa, C.; Villegas, I.; Alarcón de la Lastra, C.; Motilva, V.; Martín Calero, M.J. Evidence for protective and antioxidant properties of rutin, a natural flavone, against ethanol-induced gastric lesions. J. Ethnopharmacol. 2000, 71, 45–53. [Google Scholar] [CrossRef]
- Schwedhelm, E.; Maas, R.; Troost, R.; Boger, R.H. Clinical pharmacokinetics of antioxidants and their impact on systemic oxidative stress. Clin. Pharmacokinet. 2003, 42, 437–459. [Google Scholar] [CrossRef] [PubMed]
- Nassiri-Asl, M.; Mortazavi, S.R.; Samiee-Rad, F.; Zangivand, A.A.; Safdari, F.; Saroukhani, S.; Abbasi, E. The effects of rutin on the development of pentylenetetrazole kindling and memory retrieval in rats. Epilepsy Behav. 2010, 18, 50–53. [Google Scholar] [CrossRef]
- Richetti, S.K.; Blank, M.; Capiotti, K.M.; Piato, A.L.; Bogo, M.R.; Vianna, M.R.; Bonan, C.D. Quercetin and rutin prevent scopolamine-induced memory impairment in zebrafish. Behav. Brain Res. 2011, 217, 10–15. [Google Scholar] [CrossRef]
- Ramalingayya, G.V.; Cheruku, S.P.; Nayak, P.G.; Kishore, A.; Shenoy, R.; Rao, C.M.; Krishnadas, N. Rutin protects against neuronal damage in vitro and ameliorates doxorubicin-induced memory deficits in vivo in Wistar rats. Drug Des. Devel. Ther. 2017, 11, 1011–1026. [Google Scholar] [CrossRef]
- Kumar, A.; Rinwa, P.; Dhar, H. Possible nitric oxide modulation in the protective effects of rutin against experimental head trauma-induced cognitive deficits: Behavioral, biochemical, and molecular correlates. J. Surg. Res. 2014, 188, 268–279. [Google Scholar] [CrossRef]
- Robinson, D.T.; Martin, C.R. Fatty acid requirements for the preterm infant. Semin. Fetal Neonatal Med. 2017, 22, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Lo Van, A.; Sakayori, N.; Hachem, M.; Belkouch, M.; Picq, M.; Fourmaux, B.; Lagarde, M.; Osumi, N.; Bernoud-Hubac, N. Targeting the brain with a neuroprotective Omega-3 fatty acid to enhance neurogenesis in the hypoxic condition in culture. Mol. Neurobiol. 2018, 56, 986–999. [Google Scholar] [CrossRef]
- Calderon, F.; Kim, H.Y. Docosahexaenoic acid promotes neurite growth in hippocampal neurons. J. Neurochem. 2004, 90, 979–988. [Google Scholar] [CrossRef] [PubMed]
- Barker-Haliski, M.L.; Dahle, E.J.; Heck, T.D.; Pruess, T.H.; Vanegas, F.; Wilcox, K.S.; White, H.S. Evaluating an etiologically relevant platform for therapy development for temporal lobe epilepsy: Effects of carbamazepine and valproic acid on acute seizures and chronic behavioral comorbidities in the Theiler’s murine encephalomyelitis virus mouse model. J. Pharmacol. Exp. Ther. 2015, 353, 318–329. [Google Scholar] [CrossRef]
- Gröber, U.; Schmidt, J.; Kisters, K. Magnesium in Prevention and Therapy. Nutrients 2015, 7, 8199–8226. [Google Scholar] [CrossRef] [PubMed]
- Clerc, P.; Young, C.A.; Bordt, E.A.; Grigore, A.M.; Fiskum, G.; Polster, B.M. Magnesium sulfate protects against the bioenergetic consequences of chronic glutamate receptor stimulation. PLoS ONE 2013, 8, e79982. [Google Scholar] [CrossRef]
- Lambuk, L.; Jafri, A.J.A.; Arfuzir, N.N.N.; Iezhitsa, I.; Agarwal, R.; Rozali, K.N.B.; Agarwal, P.; Bakar, N.S.; Kutty, M.K.; Yusof, A.P.; et al. Neuroprotective effect of Magnesium acetyltaurate against NMDA-induced excitotoxicity in rat retina. Neurotox. Res. 2017, 31, 31–45. [Google Scholar] [CrossRef]
- Adamo, A.M.; Zago, M.P.; Mackenzie, G.G.; Aimo, L.; Keen, C.L.; Keenan, A.; Oteiza, P.I. The role of zinc in the modulation of neuronal proliferation and apoptosis. Neurotox. Res. 2010, 17, 1–14. [Google Scholar] [CrossRef]
- Osredkar, J.; Sustar, N. Copper and Zinc, Biological Role and Significance of Copper/Zinc Imbalance. J. Clin. Toxicol. S 2011, 3, 1–18. [Google Scholar] [CrossRef]
- Kaler, S.G.; Holmes, C.S.; Goldstein, D.S.; Tang, J.; Godwin, S.C.; Donsante, A.; Liew, C.J.; Sato, S.; Patronas, N. Neonatal diagnosis, and treatment of Menkes disease. N. Engl. J. Med. 2008, 358, 605–614. [Google Scholar] [CrossRef]
- Kaler, S.G. ATP7A-related copper transport diseases-emerging concepts and future trends. Nat. Rev. Neurol. 2011, 7, 15–29. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Osornio, M.; Montes, S.; Perez-Severiano, F.; Aguilera, P.; Floriano-Sanchez, E.; Monroy-Noyola, A.; Rubio, C.; Rios, C. Copper reduces striatal protein nitration and tyrosine hydroxylase inactivation induced by MPP+ in rats. Neurochem. Int. 2009, 54, 447–451. [Google Scholar] [CrossRef]
- Sehgal, N.; Gupta, A.; Valli, R.K.; Joshi, S.D.; Mills, J.T.; Hamel, E.; Khanna, P.; Jain, S.C.; Thakur, S.S.; Ravindranath, V. Withania somnifera reverses Alzheimer’s disease pathology by enhancing low-density lipoprotein receptor-related protein in the liver. Proc. Natl. Acad. Sci. USA 2012, 109, 3510–3515. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, X.; Hua, W.; Wei, Q.; Fang, X.; Zhao, Z.; Chun, G.; Chao, L.; Chen, C.; Yifu, T.; et al. Berberine inhibits macrophage M1 polarization via AKT1/SOCS1/NF-κB signaling pathway to protect against DSS-induced colitis. Int. Immunopharmacol. 2018, 57, 121–131. [Google Scholar] [CrossRef]
- Yan, G.; Li, Y.J.; Zhao, Y.Y.; Guo, J.M.; Zhang, W.H.; Zhang, M.M.; Fua, Y.H.; Liu, Y.P. Neuroprotective Carbazole alkaloids from the stems and leaves of Clausena lenis. Nat. Prod. Res. 2021, 35, 2002–2009. [Google Scholar] [CrossRef]
- Rastogi, M.; Ojha, R.P.; Prabu, P.C.; Parimala Devi, B.; Agrawal, A.; Dubey, G.P. Prevention of age-associated neurodegeneration and promotion of healthy brain aging in female Wistar rats by long-term use of bacosides. Biogerontology 2012, 13, 183–195. [Google Scholar] [CrossRef]
- Son, I.S.; Kim, J.H.; Sohn, H.Y.; Son, K.H.; Kim, J.S.; Kwon, C.S. Antioxidative and hypolipidemic effects of diosgenin, a steroidal saponin of Yam (Dioscorea spp.), on high cholesterol-fed rats. Biosci. Biotechnol. Biochem. 2007, 71, 70471–70479. [Google Scholar] [CrossRef]
- Kondeva-Burdina, M.; Krasteva, I.; Popov, G.; Manov, V. Neuroprotective and antioxidant activities of saponins’ mixture from Astragalus glycyphylloides in a model of 6-hydroxy- dopamine-induced oxidative stress on isolated rat brain synaptosomes. Pharmacia 2019, 66, 233–236. [Google Scholar] [CrossRef][Green Version]
- Abe, T.K.; Taniguchi, M. Identification of Myo-inositol hexakisphosphate (IP6) as a β- secretase 1 (BACE1) inhibitory molecule in rice grain extract and digest. FEBS Open Bio. 2014, 4, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Hernández, D.; Anderson, G.H.; Poon, A.N.; Pannia, E.; Cho, C.E.; Huot, P.S.P.; Kubant, R. Maternal fat-soluble vitamins, brain development, and regulation of feeding behavior: An overview of research. Nutr. Res. Rev. 2016, 36, 1045–1054. [Google Scholar] [CrossRef]
- Mikkelsen, K.; Stojanovska, L.; Tangalakis, K.; Bosevski, M.; Apostolopoulos, V. Cognitive decline: A vitamin B perspective. Maturitas 2016, 93, 108–113. [Google Scholar] [CrossRef]
- Martin, P.R.; Singleton, C.K.; Hiller-Sturmhöfel, S. The role of thiamine deficiency in alcoholic brain disease. Alcohol Res. Health 2003, 27, 134–142. [Google Scholar]
- Williams, A.; Ramsden, D. Nicotinamide: A double-edged sword. Parkinsonism Relat. Disord. 2005, 11, 413–420. [Google Scholar] [CrossRef]
- Griffin, S.M.; Pickard, M.R.; Orme, R.P.; Hawkins, C.P.; Fricker, R.A. Nicotinamide promotes neuronal differentiation of mouse embryonic stem cells in vitro. Neuroreport 2013, 24, 1041–1046. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Tang, L.; Wei, W.; Hong, Y.; Chen, H.; Ying, W.; Chen, S. Nicotinamide mononucleotide improves energy activity and survival rate in an in vitro model of Parkinson’s disease. Exp. Ther. Med. 2014, 8, 943–950. [Google Scholar] [CrossRef]
- Kuypers, N.J.; Hoane, M.R. Pyridoxine administration improves behavioral and anatomical outcomes after unilateral contusion injury in the rat. J. Neurotrauma 2010, 27, 1275–1282. [Google Scholar] [CrossRef]
- Calderón-Ospina, C.A.; Nava-Mesa, M.O. B Vitamins in the nervous system: Current knowledge of the biochemical modes of action and synergies of thiamine, pyridoxine, and cobalamin. CNS Neurosci. Ther. 2020, 26, 5–13. [Google Scholar] [CrossRef]
- Volkov, I. The master key effect of vitamin B12 in the treatment of malignancy- A potential therapy? Med. Hypotheses 2008, 70, 324–328. [Google Scholar] [CrossRef] [PubMed]
- De Queiroz, K.B.; Cavalcante-Silva, V.; Lopes, F.L.; Rocha, G.A.; D’Almeida, V.; Coimbra, R.S. Vitamin B12 is neuroprotective in experimental pneumococcal meningitis through modulation of hippocampal DNA methylation. J. Neuroinflamm. 2020, 17, 1–12. [Google Scholar] [CrossRef]
- Seitz, G.; Gerbhardt, S.; Beck, J.F.; Bohm, W.; Lode, H.N.; Niethammer, D.; Bruchelt, G. Ascorbic acid stimulates DOPA synthesis and tyrosine hydroxylase gene expression in the human neuroblastoma cell line SK-N-SH. Neurosci. Lett. 1998, 244, 33–36. [Google Scholar] [CrossRef]
- Huang, H.O.; Caballero, B.; Chang, S.; Alberg, A.; Semba, R.; Schneyer, C.; Wilson, R.F.; Cheng, T.Y.; Prokopowicz, G.; Barnes, G.J.; et al. Multivitamin/mineral supplements and prevention of chronic disease. Evid. Rep. Technol. Assess. 2006, 139, 1–117. [Google Scholar]
- Sershen, H.; Mason, M.F.; Reith, M.E.A.; Hashim, A.; Lajtha, A. Effect of nicotine and amphetamine on the neurotoxicity of N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) in mice. Neuropharmacology 1985, 25, 1231–1234. [Google Scholar] [CrossRef]
- Pardo, J.V.; Pardo, P.J.; Raichle, M.E. Neural correlates of self-induced dysphoria. Am. J. Psychiatry 1993, 150, 713–719. [Google Scholar]
- Schepers, M.; Martens, N.; Tiane, A.; Vanbrabant, K.; Liu, H.B.; Lütjohann, D.; Mulder, M.; Vanmierlo, T. Edible seaweed-derived constituents: An undisclosed source of neuroprotective compounds. Neural Regen. Res. 2020, 15, 790–795. [Google Scholar] [PubMed]
- Javed, H.; Meeran, M.F.N.; Azimullah, S.; Adem, A.; Sadek, B.; Ojha, S.K. Plant extracts and phytochemicals targeting α-synuclein aggregation in Parkinson’s disease models. Front. Pharmacol. 2019, 9, 1–27. [Google Scholar] [CrossRef]
- Kanhere, R.; Anjana, A.; Anbu, J.; Sumithra, M.; Ahamed, N. Neuroprotective and antioxidant potential of terpenoid fraction from Hygrophila auriculata against transient global cerebral ischemia in rats. Pharm. Biol. 2013, 51, 181–189. [Google Scholar] [CrossRef]
- Choi, S.U.; Park, S.H.; Kim, K.H.; Choi, E.J.; Kim, S.; Park, W.K.; Zhang, Y.H.; Kim, H.S.; Jung, N.P.; Lee, C.O. The bisbenzylisoquinoline alkaloids, tetrandrine, and fangchinoline enhance the cytotoxicity of multidrug resistance-related drugs via modulation of P-glycoprotein. Anticancer Drugs 1998, 9, 255–261. [Google Scholar] [CrossRef]
- Sun, J.; Li, M.; Zou, F.; Bai, S.; Jiang, X.; Tian, L.; Ou, S.; Jiao, R.; Bai, W. Protection of cyanidin-3-O-glucoside against acrylamide and glycidamide-induced reproductive toxicity in Leydig cells. Food Chem. Toxicol. 2018, 119, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Chu, S.; Liu, Y.; Chen, N. Neuroprotective effects of anthraquinones from Rhubarb in central nervous system diseases. Evid. Based Complement. Alternat. Med. 2019, 2019, 1–12. [Google Scholar] [CrossRef]
- Bo, S.; Lai, J.; Lin, H.; Luo, X.; Zeng, Y.; Du, T. Purpurin, an anthraquinone that induces ROS-mediated A549 lung cancer cell apoptosis via inhibition of PI3K/AKT and proliferation. J. Pharm. Pharmacol. 2021, 73, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Xie, J.; Ge, X.; Xu, P.; Wang, A.; Heb, Y.; Zhoub, Q.; Pan, L.; Chen, R. Effects of anthraquinone extract from Rheum officinale Bail on the growth performance and physio- logical responses of Macrobrachium rosenbergii under high-temperature stress. Fish Shellfish Immunol. 2010, 29, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Zhao, L.; Hu, H.; Qin, Y.; Bian, Y.; Jiang, H.; Zhou, H.; Yu, L.; Zeng, S. Interaction of five anthraquinones from rhubarb with human organic anion transporter 1 (SLC22A6) and 3 (SLC22A8) and drug-drug interaction in rats. J. Ethnopharmacol. 2014, 153, 864–871. [Google Scholar] [CrossRef] [PubMed]
- Aldbass, A.; Amina, M.; Al Musayeib, N.M.; Bhat, R.S.; Al-Rashed, S.; Marraiki, N.; Fahmy, R.; El-Ansary, A. Cytotoxic and anti-excitotoxic effects of selected plant and algal extracts using COMET and cell viability assays. Sci. Rep. 2021, 11, 8512. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Xiang, L.; Cao, F.; Li, H.; Wang, X. Research progress on the effects and mechanisms of emodin in Tumor Metastasis. Zhong Liu Yao Xue 2016, 6, 173–177. [Google Scholar]
- Gutierres, J.M.; Carvalho, F.B.; Schetinger, M.R.C.; Rodrigues, M.V.; Schmatz, R.; Pimentel, V.C.; Vieira, J.M.; Rosa, M.M.; Marisco, P.; Ribeiro, D.A.; et al. Protective effects of anthocyanins on the ectonucleotidase activity in the impairment of memory induced by scopolamine in adult rats. Life Sci. 2012, 91, 1221–1228. [Google Scholar] [CrossRef]
- Jeter, C.B.; Hergenroeder, G.W.; Ward, N.H., III; Moore, A.N.; Dash, P.K. L-arginine, and its metabolite levels: A possible link to cerebral blood flow, extracellular matrix remodeling, and energy status. J. Neurotrauma 2012, 29, 119–127. [Google Scholar] [CrossRef]
- Liu, H.; Goodman, J.C.; Robertson, C.S. The effects of L-arginine on cerebral hemodynamics after controlled cortical impact injury in the mouse. J. Neurotrauma 2004, 19, 327–334. [Google Scholar] [CrossRef]
- Cherian, L.; Chacko, G.; Goodman, C.; Robertson, C.S. Neuroprotective effects of L-arginine administration after cortical impact injury in rats: Dose-response and time window. J. Pharmacol. Exp. Ther. 2003, 304, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Bitner, B.R.; Brink, D.C.; Mathew, L.C.; Pautler, R.G.; Robertson, C.S. Impact of arginase II on CBF in experimental cortical impact injury in mice using MRI. J. Cereb. Blood Flow Metab. 2010, 30, 1105–1109. [Google Scholar] [CrossRef]
- De George, M.L.; Marlowe, D.; Werner, E.; Soderstrom, K.E.; Stock, M.; Mueller, A.; Bohn, M.C.; Kozlowski, D.A. Combining glial cell line-derived neurotrophic factor gene delivery (AdGDNF) with L-arginine decreases contusion size but not behavioral deficits after traumatic brain injury. Brain Res. 2011, 1403, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Meloni, B.P.; Craig, A.J.; Milech, N.; Hopkins, R.M.; Watt, P.M.; Knuckey, N.W. The neuroprotective efficacy of cell-penetrating peptides TAT, penetratin, Arg-9, and Pep-1 in glutamic acid, kainic acid, and in vitro ischemia injury models using primary cortical neuronal cultures. Cell. Mol. Neurobiol. 2014, 34, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Meloni, B.P.; Milani, D.; Cross, J.L.; Clark, V.W.; Edwards, A.B.; Anderton, R.S.; Blacker, D.J.; Knuckey, N.W. Assessment of the neuroprotective effects of arginine-rich protamine peptides, poly-arginine peptides (R12-Cyclic, R22), and arginine-tryptophan-containing peptides following in vitro excitotoxicity and/or permanent middle cerebral artery occlusion in rats. Neuromolecular Med. 2017, 19, 271–285. [Google Scholar] [CrossRef]
- Edwards, A.B.; Cross, J.L.; Anderton, R.S.; Knuckey, N.W.; Meloni, B.P. Poly-arginine R18 and R18D (D-enantiomer) peptides reduce infarct volume and improve behavioral outcomes following perinatal hypoxic-ischaemic encephalopathy in the P7 rat. Mol. Brain 2018, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Sachdeva, A.K.; Chopra, K. Lycopene abrogates Aβ(1-42)-mediated neuroinflammatory cascade in an experimental model of Alzheimer’s disease. J. Nutr. Biochem. 2015, 26, 736–744. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, Y.; Azqueta, A.; Luna, L.; Bonilla, F.; Domínguez, G.; Collins, A.R. The carotenoid β-cryptoxanthin stimulates the repair of DNA oxidation damage in addition to acting as an antioxidant in human cells. Carcinogenesis 2008, 30, 308–314. [Google Scholar] [CrossRef]
- Brimson, J.M.; Brimson, S.J.; Brimson, C.A.; Rakkhitawatthana, V.; Tencomnao, T. Rhinacanthus nasutus extracts prevent glutamate and amyloid-β neurotoxicity in HT-22 mouse hippocampal cells: Possible active compounds include lupeol, stigmasterol, and β-sitosterol. Int. J. Mol. Sci. 2012, 13, 5074–5097. [Google Scholar] [CrossRef] [PubMed]
- Hamedi, A.; Ghanbari, A.; Saeidi, V.; Razavipour, R.; Azari, H. Effects of β-sitosterol oral administration on the proliferation and differentiation of neural stem cells. J. Funct. Foods 2014, 8, 252–258. [Google Scholar] [CrossRef]
- Hamedi, A.; Ghanbari, A.; Razavipour, R.; Saeidi, V.; Zarshenas, M.M.; Sohrabpour, M.; Azari, H. Alyssum homolocarpum seeds: Phytochemical analysis and effects of the seed oil on neural stem cell proliferation and differentiation. J. Nat. Med. 2015, 69, 387–396. [Google Scholar] [CrossRef]
- Cheng, F.C.; Jinn, T.R.; Hou, R.C.W.; Tzen, J.T.C. Neuroprotective effects of sesamin and sesamolin on Gerbil brain in cerebral ischemia. Int. J. Biomed. 2006, 2, 284–288. [Google Scholar]
- Shimmyo, Y.; Kihara, T.; Akaike, A.; Niidome, T.; Sugimoto, H. Multifunction of myricetin on A beta: Neuroprotection via a conformational change of A-beta and reduction of A beta via the interference of secretases. J. Neurosci. Res. 2008, 86, 368–377. [Google Scholar] [CrossRef]
- Jones, J.R.; Lebar, M.D.; Jinwal, U.K.; Abisambra, J.K.; Koren, J., III; Blair, L.; O’Leary, J.C.; Davey, Z.; Trotter, J.; Johnson, A.G.; et al. The diarylheptanoid (+)-aR, 11S-myricanol, and two flavones from bayberry (Myrica cerifera) destabilize the microtubule-associated protein Tau. J. Nat. Prod. 2011, 74, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Pinto, N.B.; Alexandre, B.; Neves, K.R.T.; Silva, A.H.; Leal, L.K.A.M.; Viana, G.S.B. Neuroprotective properties of the standardized extract from Camellia sinensis (Green Tea) and its main bioactive components, epicatechin and epigallocatechin gallate, in the 6-OHDA model of Parkinson’s disease. Evid. Based Complement. Alternat. Med. 2015, 2015, 161092. [Google Scholar]
- Cifuentes, J.; Salazar, V.A.; Cuellar, M.; Castellanos, M.C.; Rodríguez, J.; Cruz, J.C.; Muñoz-Camargo, C. Antioxidant and Neuroprotective Properties of Non-Centrifugal Cane Sugar and Other Sugarcane Derivatives in an In Vitro Induced Parkinson’s Model. Antioxidants 2021, 10, 1040. [Google Scholar] [CrossRef]
- Mendonça-Junior, F.J.B.; Scotti, M.T.; Muratov, E.N.; Scotti, L.; Nayarisseri, A. Natural Bioactive Products with Antioxidant Properties Useful in Neurodegenerative Diseases 2020. Oxid. Med. Cell. Longev. 2021, 2021, 1–2. [Google Scholar] [CrossRef]
- Hossain, M.T.; Furhatun-Noor; Asadujjaman; Matin, A.; Tabassum, F.; Ar Rashid, H. A Review study on the pharmacological effects and mechanism of action of Tannins. Eur. J. Pharm. Med. Res. 2021, 8, 5–10. [Google Scholar]
- Soldado, D.; Bessa, R.J.B.; Jeronimo, E. Condensed Tannins as Antioxidants in Ruminants—Effectiveness and Action Mechanisms to Improve Animal Antioxidant Status and Oxidative Stability of Products. Animals 2021, 11, 3243. [Google Scholar] [CrossRef]
- Puiggròs, F.; Llópiz, N.; Ardévol, A.; Bladé, C.; Arola, L.; Salvadó, M.J. Grape Seed Procyanidins Prevent Oxidative Injury by Modulating the Expression of Antioxidant Enzyme Systems. J. Agric. Food Chem. 2005, 53, 6080–6086. [Google Scholar] [CrossRef] [PubMed]
- Ali, T.; Kim, T.; Rehman, S.U.; Khan, M.S.; Amin, F.U.; Khan, M.; Ikram, M.; Kim, M.O. Natural Dietary Supplementation of Anthocyanins via PI3K/Akt/Nrf2/HO-1 Pathways Mitigate Oxidative Stress, Neurodegeneration, and Memory Impairment in a Mouse Model of Alzheimer’s Disease. Mol. Neurobiol. 2018, 55, 6076–6093. [Google Scholar] [CrossRef]
- Echeverry, C.; Arredondo, F.; Abin-Carriquiry, J.A.; Midiwo, J.O.; Ochieng, C.; Kerubo, L.; Dajas, F. Pretreatment with Natural Flavones and Neuronal Cell Survival after Oxidative Stress: A Structure-Activity Relationship Study. J. Agric. Food Chem. 2010, 58, 2111–2115. [Google Scholar] [CrossRef]
- Boesch-Saadatmandi, C.; Pospissil, R.T.; Graeser, A.; Canali, R.; Boomgaarden, I.; Doering, F.; Wolffram, S.; Egert, S.; Mueller, M.J.; Rimbach, G. Effect of Quercetin on Paraoxonase 2 Levels in RAW264.7 Macrophages and in Human Monocytes- Role of Quercetin Metabolism. Int. J. Mol. Sci. 2009, 10, 4168–4177. [Google Scholar] [CrossRef] [PubMed]
- Costa, L.G.; Tait, L.; de Laat, R.; Dao, K.; Giordano, G.; Pellacani, C.; Cole, T.B.; Furlong, C.E. Modulation of paraoxonase 2 (PON2) in mouse brain by the polyphenol quercetin: A mechanism of neuroprotection? Neurochem. Res. 2014, 38, 1809–1818. [Google Scholar] [CrossRef] [PubMed]
- Grewal, A.K.; Singha, T.G.; Sharma, D.; Sharma, V.; Singh, M.; Rahman, H.; Najda, A.; Walasek-Janusz, M.; Kamel, M.; Albadrani, G.M.; et al. Mechanistic insights and perspectives involved in neuroprotective action of quercetin. Biomed. Pharmacother. 2021, 140, 111729. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.J.; Cheang, L.C.V.; Wang, M.W.; Lee, S.M. Quercetin exerts a neuroprotective effect through inhibition of the iNOS/NO system and pro-inflammation gene expression in PC12 cells and in zebrafish. Int. J. Mol. Med. 2011, 27, 195–203. [Google Scholar]
- Ansari, M.A.; Abdul, H.M.; Joshi, G.; Opii, W.O.; Butterfield, D.A. Protective effect of quercetin in primary neurons against Abeta(1-42): Relevance to Alzheimer’s disease. J. Nutr. Biochem. 2009, 20, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Machin, L.; Monzote, L.; Sharifi-Rad, J.; Ezzat, S.M.; Salem, M.A.; Merghany, R.M.; El Mahdy, N.M.; Kılıç, C.S.; Sytar, O.; et al. Therapeutic Potential of Quercetin: New Insights and Perspectives for Human Health. ACS Omega 2020, 5, 11849–11872. [Google Scholar] [CrossRef] [PubMed]
- Khazdair, M.R.; Ghafari, S.; Sadeghi, M. Possible therapeutic effects of Nigella sativa and its thymoquinone on COVID-19. Pharm. Biol. 2021, 59, 696–703. [Google Scholar] [CrossRef]
- Kavya Bhatt, S.; Manjunatha Javagal, R.; Shashirekha Nanjarajurs, M.; Eligar, S.M. In vitro anti-inflammatory property of a Quercetin-3-O-diglucoside-7-O-glucoside characterized from fresh leaves of Trigonella foenum-graecum L. Int. J. Food Prop. 2021, 24, 1438–1452. [Google Scholar] [CrossRef]
- Wang, J.; Fang, X.; Ge, L.; Cao, F.; Zhao, L.; Wang, Z.; Xiao, W. Antitumor, antioxidant and anti-inflammatory activities of kaempferol and its corresponding glycosides and the enzymatic preparation of kaempferol. PLoS ONE 2018, 13, e0197563. [Google Scholar] [CrossRef]
- Isah, T. Anticancer Alkaloids from Trees: Development into Drugs. Pharmacognosy Reviews. Pharmacogn. Rev. 2016, 10, 90–99. [Google Scholar] [CrossRef]
- Huang, Y.; Cui, S.; Cui, X.; Cao, Q.; Ding, H.; Song, J.; Hu, X.; Ye, H.; Yu, B.; Sheng, Z.; et al. Tetrandrine, an alkaloid from S. tetrandra exhibits anti-hypertensive and sleep-enhancing effects in SHR via different mechanisms. Phytomedicine 2016, 23, 1821–1829. [Google Scholar] [CrossRef]
- Yang, Z.; Zhang, D.; Ren, J.; Yang, M. Skimmianine, a furoquinoline alkaloid from Zanthoxylum nitidum as a potential acetylcholinesterase inhibitor. Med. Chem. Res. 2012, 2, 722–725. [Google Scholar] [CrossRef]
- Shimizu, M.; Shirakami, Y.; Moriwaki, H. Targeting Receptor Tyrosine Kinases for Chemoprevention by Green Tea Catechin, EGCG. Int. J. Mol. Sci. 2008, 9, 1034–1049. [Google Scholar] [CrossRef] [PubMed]
- Coșarcă, S.; Tanase, C.; Muntean, D.L. Therapeutic aspects of Catechin and its derivatives—An Update. Acta Biol. Marisiensis 2019, 2, 21–29. [Google Scholar] [CrossRef]
- Yang, W.; Chen, X.; Li, Y.; Guo, S.; Wang, Z.; Yu, X. Advances in Pharmacological Activities of Terpenoids. Nat. Prod. Commun. 2020, 15, 1–13. [Google Scholar] [CrossRef]
- Yoo, K.; Park, S. Terpenoids as potential anti-Alzheimer’s disease therapeutics. Molecules 2012, 17, 3524–3538. [Google Scholar] [CrossRef] [PubMed]
- Kwon, K.H.; Murakami, A.; Tanaka, T.; Ohigashi, H. Dietary rutin, but not its aglycone quercetin, ameliorates dextran sulfate sodium-induced experimental colitis in mice: Attenuation of pro-inflammatory gene expression. Biochem. Pharmacol. 2005, 69, 395–406. [Google Scholar] [CrossRef] [PubMed]
- Shamim, A.; Mahmood, T.; Ahsan, F.; Kumar, A.; Bagga, P. Lipids: An insight into the neurodegenerative disorders. Clin. Nutr. Exp. 2018, 20, 1–19. [Google Scholar] [CrossRef]
| Active Molecules (Nutritive Factors and Secondary Metabolites) | Active Molecule Constituents in Banana and Plantain Fruit Components (Pulp and Peel) | Neuromechanism-Related Protein/Gene Targets |
|---|---|---|
Tannin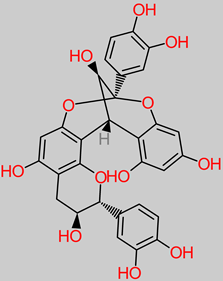 |
| |
Phenolic acid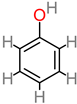 |
| |
Quercetin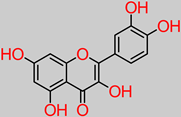 |
| |
Rutin |
| |
Carbohydrates | ||
Lipids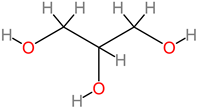 | ||
Magnesium |
|
|
Zinc | ||
Copper | ||
Alkaloid |
| |
Saponin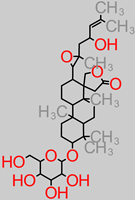 |
| |
Phytate (Phytic acid)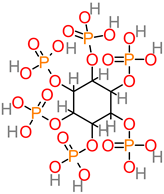 |
| |
Vitamins B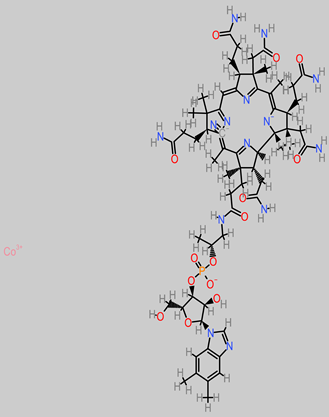 |
| |
Anthocyanin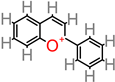 |
|
|
Vitamin E (Tocopherol) |
| |
Selenium | ||
Phytosterol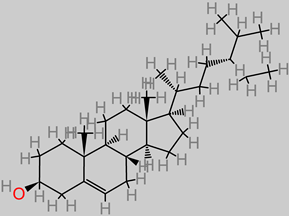 |
| |
Terpenoids |
| |
Glycosides |
| |
Anthraquinone |
| |
Arginine |
|
|
β-carotene |
| |
Lycopene | ||
β-cryptoxanthin |
| |
β-sitosterol |
| |
Sesamin | - M. sapientum/M. acuminata peel extracts (methanol) [119]. | |
Myricetin |
| |
Catechin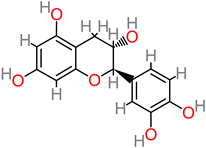 |
| |
Vitamin C (ascorbic)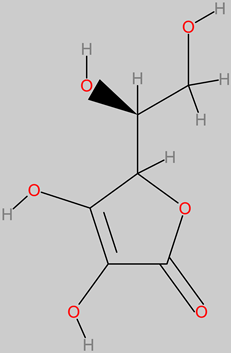 |
|
| Bioactive Compounds | Biological Mechanisms of Action Related to Neuroactivity | Bioactive Compounds | Biological Mechanisms of Action Related to Neuroactivity |
|---|---|---|---|
| TANNINS |
(free-radical scavenging) (metal chelation) (pro-oxidative enzyme inhibition) (endogenous antioxidant system interaction) (inhibition of xanthine oxidase-induced Lipid peroxidation) [208,209]. | ANTHOCYANIN | Delphinidin Cyanidin Anti-inflammatory, anti-Alzheimer, antitumor, and antioxidative properties by depleting the expression of cytokine markers [211]. |
Anti-inflammatory mechanisms, such as depletion of apoptotic cells [208]. | |||
| QUERCETIN | MYRICETIN | Kaempferol Antitumor, anti-inflammation, and antioxidant properties are exercised via an antiproliferative mechanism in cells, attenuation mechanism against inflammation, and tumor growth factors [124,221]. | |
| |||
| ALKALOIDS | Vincristine Antineuroblastoma property exerted via the mechanisms truncating the glutathione metabolism [222]. Tetrandrine Anti-inflammatory and antitumor activities are linked to the calcium-channel blocking mechanism [223]. Skimmianine Anti-inflammatory property via the inhibition of nitric oxide production [224]. | CATECHIN | |
| TERPENOIDS Paeoniflorin, Triptolidenol, Tripterine, Triptonide, Gindenoside, Oleanoic Acid | Anti-inflammatory activity via interleukin-6 inhibition [227]. Anti-nociceptive, antioxidant and anti-inflammatory properties [228]. | RUTIN | |
| LIPID Omega-3 DHA | Anti-inflammatory properties, Cell survival promotors [230]. | Neuroprotectin D1 Lipid | Inhibition of apoptosis-related damage to DNA [230]. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oyeyinka, B.O.; Afolayan, A.J. Suitability of Banana and Plantain Fruits in Modulating Neurodegenerative Diseases: Implicating the In Vitro and In Vivo Evidence from Neuroactive Narratives of Constituent Biomolecules. Foods 2022, 11, 2263. https://doi.org/10.3390/foods11152263
Oyeyinka BO, Afolayan AJ. Suitability of Banana and Plantain Fruits in Modulating Neurodegenerative Diseases: Implicating the In Vitro and In Vivo Evidence from Neuroactive Narratives of Constituent Biomolecules. Foods. 2022; 11(15):2263. https://doi.org/10.3390/foods11152263
Chicago/Turabian StyleOyeyinka, Barnabas Oluwatomide, and Anthony Jide Afolayan. 2022. "Suitability of Banana and Plantain Fruits in Modulating Neurodegenerative Diseases: Implicating the In Vitro and In Vivo Evidence from Neuroactive Narratives of Constituent Biomolecules" Foods 11, no. 15: 2263. https://doi.org/10.3390/foods11152263
APA StyleOyeyinka, B. O., & Afolayan, A. J. (2022). Suitability of Banana and Plantain Fruits in Modulating Neurodegenerative Diseases: Implicating the In Vitro and In Vivo Evidence from Neuroactive Narratives of Constituent Biomolecules. Foods, 11(15), 2263. https://doi.org/10.3390/foods11152263








