Carob: A Sustainable Opportunity for Metabolic Health
Abstract
1. Introduction
2. Carob; Composition in Nutrients and Non-Nutrient Bioactive Compounds
3. A Sustainable Source for Human Nutrition in Both Developing and Developed Countries
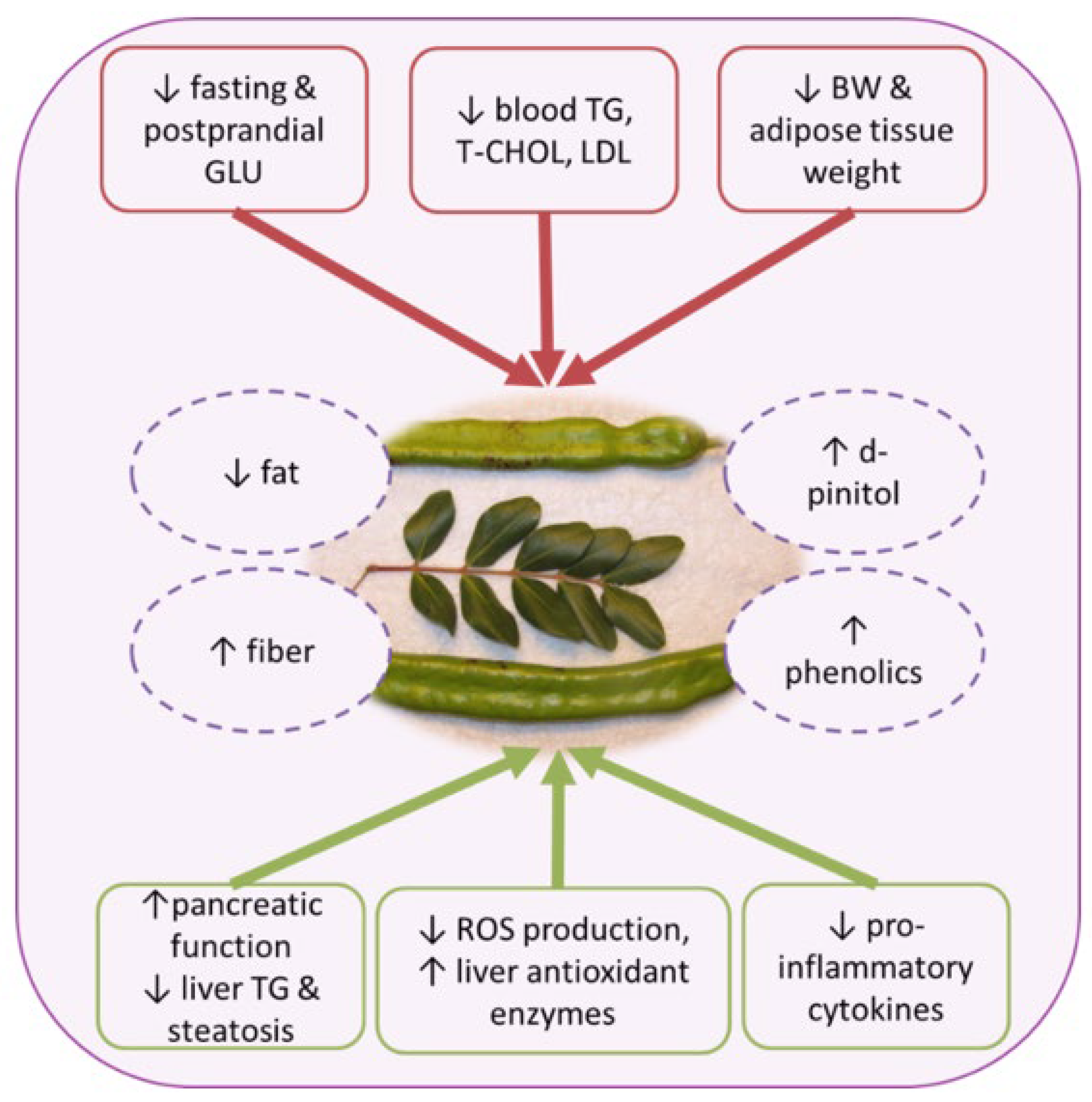
4. Effects on Metabolic Health
4.1. Obesity, Meta-Inflammation and Dyslipidemia
4.2. Effects on Glycaemia and T2DM
4.3. Other Metabolic Disorders
5. Conclusions and Perspectives
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tosti, V.; Bertozzi, B.; Fontana, L. Health Benefits of the Mediterranean Diet: Metabolic and Molecular Mechanisms. J. Gerontol. Ser. A 2018, 73, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Eurostat. Over Half of Adults in the EU Are Overweight. Available online: https://ec.europa.eu/eurostat/web/products-eurostat-news/-/ddn-20210721-2 (accessed on 21 July 2021).
- Pineda, E.; Sanchez-Romero, L.M.; Brown, M.; Jaccard, A.; Jewell, J.; Galea, G.; Webber, L.; Breda, J. Forecasting Future Trends in Obesity across Europe: The Value of Improving Surveillance. Obes. Facts 2018, 11, 360–371. [Google Scholar] [CrossRef] [PubMed]
- Prentice, A.M. The Emerging Epidemic of Obesity in Developing Countries. Int. J. Epidemiol. 2006, 35, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Nyberg, S.T.; Batty, G.D.; Pentti, J.; Virtanen, M.; Alfredsson, L.; Fransson, E.I.; Goldberg, M.; Heikkilä, K.; Jokela, M.; Knutsson, A.; et al. Obesity and Loss of Disease-Free Years Owing To Major Non-Communicable Diseases: A Multicohort Study. Lancet Public Health 2018, 3, e490–e497. [Google Scholar] [CrossRef]
- Huai, P.; Liu, J.; Ye, X.; Li, W.Q. Association of Central Obesity With All Cause and Cause-Specific Mortality in US Adults: A Prospective Cohort Study. Front. Cardiovasc. Med. 2022, 9, 816144. [Google Scholar] [CrossRef]
- Mongraw-Chaffin, M.; Foster, M.C.; Anderson, C.A.; Burke, G.L.; Haq, N.; Kalyani, R.R.; Ouyang, P.; Sibley, C.T.; Tracy, R.; Woodward, M.; et al. Metabolically Healthy Obesity, Transition to Metabolic Syndrome, and Cardiovascular Risk. J. Am. Coll. Cardiol. 2018, 71, 1857–1865. [Google Scholar] [CrossRef]
- Hwalla, N.; Jaafar, Z. Dietary Management of Obesity: A Review of the Evidence. Diagnostics 2020, 11, 24. [Google Scholar] [CrossRef]
- Wang, Z.; Nakayama, T. Inflammation, a Link between Obesity and Cardiovascular Disease. Mediat. Inflamm. 2010, 2010, 535918. [Google Scholar] [CrossRef]
- Nani, A.; Murtaza, B.; Sayed Khan, A.; Khan, N.A.; Hichami, A. Antioxidant and Anti-Inflammatory Potential of Polyphenols Contained in Mediterranean Diet in Obesity: Molecular Mechanisms. Molecules 2021, 26, 985. [Google Scholar] [CrossRef]
- Stefanson, A.L.; Bakovic, M. Dietary Regulation of Keap1/Nrf2/ARE Pathway: Focus on Plant-Derived Compounds and Trace Minerals. Nutrients 2014, 6, 3777–3801. [Google Scholar] [CrossRef]
- Carrasco-Pozo, C.; Cires, M.J.; Gotteland, M. Quercetin and Epigallocatechin Gallate in the Prevention and Treatment of Obesity: From Molecular to Clinical Studies. J. Med. Food 2019, 22, 753–770. [Google Scholar] [CrossRef] [PubMed]
- Serra-Majem, L.; Tomaino, L.; Dernini, S.; Berry, E.M.; Lairon, D.; Ngo de la Cruz, J.; Bach-Faig, A.; Donini, L.M.; Medina, F.-X.; Belahsen, R.; et al. Updating the Mediterranean Diet Pyramid towards Sustainability: Focus on Environmental Concerns. Int. J. Environ. Res. Public Health 2020, 17, 8758. [Google Scholar] [CrossRef] [PubMed]
- Swinburn, B.A.; Kraak, V.I.; Allender, S.; Atkins, V.J.; Baker, P.I.; Bogard, J.R.; Brinsden, H.; Calvillo, A.; De Schutter, O.; Devarajan, R.; et al. The Global Syndemic of Obesity, Undernutrition, and Climate Change: The Lancet Commission report. Lancet 2019, 393, 791–846. [Google Scholar] [CrossRef]
- Batlle, I.; Tous, J. Carob Tree: Ceratonia Siliqua L. Promoting the Conservation and Use of Underutilized and Neglected Crops, 1st ed.; Batlle, I., Ed.; Leibniz Institute of Plant Genetics and Crop Plant Research (IPK): Gatersleben, Germany, 1997; pp. 1–92. ISBN 10:92-9043-328-X. [Google Scholar]
- Rodríguez-Solana, R.; Romano, A.; Moreno-Rojas, J.M. Carob Pulp: A Nutritional and Functional By-Product Worldwide Spread in the Formulation of Different Food Products and Beverages. A Review. Processes 2021, 9, 1146. [Google Scholar] [CrossRef]
- Brassesco, M.E.; Brandão, T.R.S.; Silva, C.L.M.; Pintado, M. Carob Bean (Ceratonia Siliqua L.): A New Perspective for Functional Food. Trends Food Sci. Technol. 2021, 114, 310–322. [Google Scholar] [CrossRef]
- Goulas, V.; Georgiou, E. Utilization of Carob Fruit as Sources of Phenolic Compounds with Antioxidant Potential: Extraction Optimization and Application in Food Models. Foods 2019, 9, 20. [Google Scholar] [CrossRef] [PubMed]
- Goulas, V.; Stylos, E.; Chatziathanasiadou, M.V.; Mavromoustakos, T.; Tzakos, A.G. Functional Components of Carob Fruit: Linking the Chemical and Biological Space. Int. J. Mol. Sci. 2016, 17, 1875. [Google Scholar] [CrossRef]
- Fidan, H.; Stankov, S.; Petkova, N.; Petkova, Z.; Iliev, A.; Stoyanova, M.; Ivanova, T.; Zhelyazkov, N.; Ibrahim, S.; Stoyanova, A.; et al. Evaluation of Chemical Composition, Antioxidant Potential and Functional Properties of Carob (Ceratonia Siliqua L.) Seeds. J. Food Sci. Technol. 2020, 57, 2404–2413. [Google Scholar] [CrossRef]
- Tous, J.; Romero, A.; Batlle, I. The Carob Tree: Botany, Horticulture, and Genetic Resources. In Horticultural Reviews Volume 41, 1st ed.; Jules Janick, J., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013; pp. 385–446. [Google Scholar] [CrossRef]
- Tous, J.; Romero, A.; Hermoso, J.F.; Ninot, A.; Plana, J.; Batlle, I. Agronomic and Commercial Performance of Four Spanish Carob Cultivars. HortTechnology 2009, 19, 465–470. [Google Scholar] [CrossRef]
- Panteleitchouk, A.V.; Cruz, L.; Silva, L.I.B.; Rocha-Santos, T.A.P.; Duarte, A.C.; Lopes, M.L.; Canhoto, J.M. Differentiation of Carob tree (Ceratonia Siliqua L.) Cultivars by Elemental Fingerprinting of Leaves. Acta Hortic. 2012, 938, 453–457. [Google Scholar] [CrossRef]
- Farag, M.A.; El-Kersh, D.M.; Ehrlich, A.; Choucry, M.A.; El-Seedi, H.; Frolov, A.; Wessjohann, L.A. Variation in Ceratonia Siliqua Pod Metabolome in Context of Its Different Geographical Origin, Ripening Stage and Roasting Process. Food Chem. 2019, 283, 675–687. [Google Scholar] [CrossRef] [PubMed]
- Kyratzis, A.C.; Antoniou, C.; Papayiannis, L.C.; Graziani, G.; Rouphael, Y.; Kyriacou, M.C. Pod Morphology, Primary and Secondary Metabolite Profiles in Non-grafted and Grafted Carob Germplasm are Configured by Agro-Environmental Zone, Genotype, and Growing Season. Front. Plant Sci. 2021, 11, 612376. [Google Scholar] [CrossRef] [PubMed]
- Viruel, J.; Le Galliot, N.; Pironon, S.; Nieto Feliner, G.; Suc, J.-P.; Lakhal-Mirleau, F.; Juin, M.; Selva, M.; Bou Dagher Kharrat, M.; Ouahmane, L.; et al. A Strong East—West Mediterranean Divergence Supports a New Phylogeographic History of the Carob Tree (Ceratonia Siliqua Leguminosae) and Multiple Domestications From Native Populations. J. Biogeogr. 2020, 47, 460–471. [Google Scholar] [CrossRef]
- Avallone, R.; Plessi, M.; Baraldi, M.; Monzani, A. Determination of Chemical Composition of Carob (Ceratonia Siliqua): Protein, Fat, Carbohydrates, and Tannins. J. Food Compos. Anal. 1997, 10, 166–172. [Google Scholar] [CrossRef]
- Nasar-Abbas, S.M.; E-Huma, Z.; Vu, T.-H.; Khan, M.K.; Esbenshade, H.; Jayasena, V. Carob Kibble: A Bioactive-Rich Food Ingredient. Compr. Rev. Food Sci. Food Saf. 2016, 15, 63–72. [Google Scholar] [CrossRef]
- Papaefstathiou, E.; Agapiou, A.; Giannopoulos, S.; Kokkinofta, R. Nutritional Characterization of Carobs and Traditional Carob Products. Food Sci. Nutr. 2018, 6, 2151–2161. [Google Scholar] [CrossRef]
- Ruiz-Aceituno, L.; Rodríguez-Sánchez, S.; Ruiz-Matute, A.I.; Ramos, L.; Soria, A.C.; Sanz, M.L. Optimisation of a Biotechnological Procedure for Selective Fractionation of Bioactive Inositols in Edible Legume Extracts. J. Sci. Food Agric. 2013, 93, 2797–2803. [Google Scholar] [CrossRef]
- Zhu, B.J.; Zayed, M.Z.; Zhu, H.X.; Zhao, J.; Li, S.P. Functional Polysaccharides of Carob Fruit: A Review. Chin. Med. 2019, 14, 1–10. [Google Scholar] [CrossRef]
- Makris, D.P.; Boskou, G.; Andrikopoulos, N.K. Polyphenolic Content and In Vitro Antioxidant Characteristics of Wine Industry and Other Agri-Food Solid Waste Extracts. J. Food Compos. Anal. 2007, 20, 125–132. [Google Scholar] [CrossRef]
- Turhan, I.; Tetik, N.; Aksu, M.; Karhan, M.; Certel, M. Liquid-solid Extraction of Soluble Solids and Total Phenolic Compounds of Carob Bean (Ceratonia siliqua L.). J. Food Proc. Engr. 2006, 29, 498–507. [Google Scholar] [CrossRef]
- Turhan, I. Optimization of Extraction of D-Pinitol and Phenolics from Cultivated and Wild Types of Carob Pods Using Response Surface Methodology. Int. J. Food Eng. 2011, 7, 7. [Google Scholar] [CrossRef]
- Owen, R.W.; Haubner, R.; Hull, W.E.; Erben, G.; Spiegelhalder, B.; Bartsch, H.; Haber, B. Isolation and Structure Elucidation of the Major Individual Polyphenols in Carob Fibre. Food Chem. Toxicol. 2003, 41, 1727–1738. [Google Scholar] [CrossRef]
- Dakia, P.A.; Wathelet, B.; Paquot, M. Isolation and Chemical Evaluation of Carob (Ceratonia Siliqua L.) Seed Germ. Food Chem. 2007, 102, 1368–1374. [Google Scholar] [CrossRef]
- Simsek, S.; Ozcan, M.M.; Al Juhaimi, F.; ElBabiker, E.; Ghafoor, K. Amino Acid and Sugar Contents of Wild and Cultivated Carob (Ceratonia Siliqua) Pods Collected in Different Harvest Periods. Chem. Nat. Compd. 2017, 53, 1008–1009. [Google Scholar] [CrossRef]
- Ozcan, M.M.; Arslan, D.; Gökçalik, H. Some Compositional Properties and Mineral Contents of Carob (Ceratonia Siliqua) Fruit, Flour and Syrup. Int. J. Food Sci. Nutr. 2009, 58, 652–658. [Google Scholar] [CrossRef]
- Oziyci, H.R.; Tetik, N.; Turhan, I.; Yatmaz, E.; Ucgun, K.; Akgul, H.; Gubbuk, H.; Karhan, M. Mineral Composition of Pods and Seeds of Wild and Grafted Carob (Ceratonia Siliqua L.) Fruits. Sci. Hortic. 2014, 167, 149–152. [Google Scholar] [CrossRef]
- Loullis, A.; Pinakoulaki, E. Carob as Cocoa Substitute: A Review on Composition, Health Benefits and Food Applications. Eur. Food Res. Technol. 2018, 244, 959–977. [Google Scholar] [CrossRef]
- Yousif, A.K.; Alghzawi, H.M. Processing and Characterization of Carob Powder. Food Chem. 2000, 69, 283–287. [Google Scholar] [CrossRef]
- Racolta, E.; Muste, S.; Muresan, A.E.; Muresan, C.C.; Bota, M.M.; Muresan, V. Characterization of Confectionery Spreadable Creams Based on Roasted Sunflower Kernels and Cocoa or Carob Powder. Bull. UASVM Food Sci. Technol. 2014, 71, 62–67. [Google Scholar] [CrossRef][Green Version]
- Tetik, N.; Turhan, I.; Oziyci, H.R.; Karhan, M. Determination of D-Pinitol in Carob Syrup. Int. J. Food Sci. Nutr. 2011, 62, 572–576. [Google Scholar] [CrossRef]
- Martins, V.; Dias, C.; Caldeira, J.; Duarte, L.C.; Reis, A.; da Silva, T.L. Carob Pulp Syrup: A Potential Mediterranean Carbon Source for Carotenoids Production by Rhodosporidium Toruloides NCYC 921. Bioresour. Technol. Rep. 2018, 3, 177–184. [Google Scholar] [CrossRef]
- Santonocito, D.; Granata, G.; Geraci, C.; Panico, A.; Siciliano, E.A.; Raciti, G.; Puglia, C. Carob Seeds: Food Waste Or Source of Bioactive Compounds? Pharmaceutics 2020, 12, 1090. [Google Scholar] [CrossRef] [PubMed]
- Mahtout, R.; Ortiz-Martínez, V.; Salar-García, M.; Gracia, I.; Hernández-Fernández, F.; Pérez de los Ríos, A.; Zaidia, F.; Sanchez-Segado, S.; Lozano-Blanco, L. Algerian Carob Tree Products: A Comprehensive Valorization Analysis and Future Prospects. Sustainability 2018, 10, 90. [Google Scholar] [CrossRef]
- Stavrou, I.J.; Christou, A.; Kapnissi-Christodoulou, C.P. Polyphenols in Carobs: A Review on Their Composition, Antioxidant Capacity and Cytotoxic Effects, and Health Impact. Food Chem. 2018, 269, 355–374. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, M.; Kapoor, V.P. Seed Galactomannans: An Overview. Chem. Biodivers. 2005, 2, 295–317. [Google Scholar] [CrossRef]
- Barak, S.; Mudgil, D. Locust bean gum: Processing, Properties and Food Applications—A Review. Int. J. Biol. Macromol. 2014, 66, 74–80. [Google Scholar] [CrossRef]
- Smith, B.M.; Bean, S.R.; Schober, T.J.; Tilley, M.; Herald, T.J.; Aramouni, F.; Dakia, P.A.; Wathelet, B.; Paquot, M.; Bengoechea, C.; et al. Composition and Molecular Weight Distribution of Carob Germprotein Fractions. Food Chem. 2008, 102, 675–683. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G. Monounsaturated Fatty Acids and Risk of Cardiovascular Disease: Synopsis of the Evidence Available From Systematic Reviews and Meta-Analyses. Nutrients 2012, 4, 1989–2007. [Google Scholar] [CrossRef]
- Picariello, G.; Sciammaro, L.; Siano, F.; Volpe, M.G.; Puppo, M.C.; Mamone, G. Comparative Analysis of C-Glycosidic Flavonoids from Prosopis Spp. and Ceratonia Siliqua Seed Germ Flour. Food Res. Int. 2017, 99, 730–738. [Google Scholar] [CrossRef]
- Siano, F.; Sciammaro, L.; Volpe, M.G.; Mamone, G.; Puppo, M.C.; Picariello, G. Integrated Analytical Methods to Characterize Lipids from Prosopis spp. and Ceratonia Siliqua Seed Germ Flour. Food Anal. Methods 2018, 11, 3471–3480. [Google Scholar] [CrossRef]
- Lakkab, I.; El, H.; Lachkar, N.; Lefter, R.; Ciobica, A.; El, B.; Lachkar, M. Ceratonia Siliqua L. Seed Peels: Phytochemical Profile, Antioxidant Activity, and Effect on Mood Disorders. J. Funct. Foods 2019, 54, 457–465. [Google Scholar] [CrossRef]
- European Commission. Climate Action—Climate Change: Causes of Climate Change. Available online: https://ec.europa.eu/clima/climate-change/causes-climate-change_en (accessed on 11 April 2022).
- Intergovernmental Panel on Climate Change (IPCC). Climate Change 2021: The Physical Science Basis-Summary of Policymakers. Working Group I Contribution to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. Available online: https://www.ipcc.ch/report/ar6/wg1/downloads/report/IPCC_AR6_WGI_SPM_final.pdf (accessed on 11 April 2022).
- Smith, P.; Bustamante, M.; Ahammad, H.; Clark, H.; Dong, H.; Elsiddig, E.A.; Haberl, H.; Harper, R.; House, J.; Jafari, M.; et al. Agriculture, Forestry and Other Land Use (AFOLU). In Climate Change 2014: Mitigation of Climate Change. Contribution of Working Group III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Edenhofer, O., Pichs-Madruga, R., Sokona, Y., Minx, J.C., Farahami, E., Kadner, S., Seyboth, K., Adler, A., Baum, I., Brunner, S., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA.
- Laborde, D.; Mamun, A.; Martin, W.; Piñeiro, V.; Vos, R. Agricultural subsidies and global greenhouse gas emissions. Nat. Commun 2021, 12, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization of the United Nations (FAO). Emissions Due to Agriculture Global, Regional and Country Trends 2000–2018. In FAOSTAT Analytical Brief 18; FAO: Roma, Italy, 2021; ISSN 2709-0078. [Google Scholar]
- Food and Agriculture Organization of the United Nations (FAO). Climate Change and Food Security: Risks and Responses; FAO: Roma, Italy, 2015; ISBN 978-92-5-108998-9. [Google Scholar]
- Horn, B.; Ferreira, C.; Kalantari, Z. Links between Food Trade, Climate Change and Food Security in Developed Countries: A Case Study of Sweden. Ambio 2022, 51, 943–954. [Google Scholar] [CrossRef] [PubMed]
- Leisner, C.P. Climate Change Impacts on Food Security-Focus on Perennial Cropping Systems and Nutritional Value. Plant Sci. 2020, 293, 110412. [Google Scholar] [CrossRef]
- Medek, D.E.; Schwartz, J.; Myers, S.S. Estimated Effects of Future Atmospheric CO2 Concentrations on Protein Intake and the Risk of Protein Deficiency by Country and Region. Environ. Health Perspect. 2017, 125, 087002. [Google Scholar] [CrossRef]
- Myers, S.S.; Smith, M.R.; Guth, S.; Golden, C.D.; Vaitla, B.; Mueller, N.D.; Dangour, A.D.; Huybers, P. Climate Change and Global Food Systems: Potential Impacts on Food Security and Undernutrition. Annu. Rev. Public Health 2017, 38, 259–277. [Google Scholar] [CrossRef]
- Springmann, M.; Mason-D’Croz, D.; Robinson, S.; Garnett, T.; Godfray, H.C.; Gollin, D.; Rayner, M.; Ballon, P.; Scarborough, P. Global and Regional Health Effects of Future Food Production Under Climate Change: A Modelling Study. Lancet 2016, 387, 1937–1946. [Google Scholar] [CrossRef]
- Phalkey, R.K.; Aranda-Jan, C.; Marx, S.; Höfle, B.; Sauerborn, R. Systematic Review of Current Efforts to Quantify the Impacts of Climate Change on Undernutrition. Proc. Natl. Acad. Sci. USA 2015, 112, E4522–E4529. [Google Scholar] [CrossRef]
- Friel, S.; Bowen, K.; Campbell-Lendrum, D.; Frumkin, H.; McMichael, A.J.; Rasanathan, K. Climate Change, Noncommunicable Diseases, and Development: The Relationships and Common Policy Opportunities. Annu. Rev. Public Health 2011, 32, 133–147. [Google Scholar] [CrossRef]
- Kjellstrom, T.; Butler, A.J.; Lucas, R.M.; Bonita, R. Public Health Impact of Global Heating due to Climate Change: Potential Effects on Chronic Non-Communicable Diseases. Int. J. Public Health 2010, 55, 97–103. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Fact Sheets: Climate Change and Health. Available online: https://www.who.int/news-room/fact-sheets/detail/climate-change-and-health (accessed on 30 October 2021).
- Santos, M.; Fraga, H.; Belo-Pereira, M.; Santos, J.A. Assessment of Growing Thermal Conditions of Main Fruit Species in Portugal Based on Hourly Records From a Weather Station Network. Appl. Sci. 2019, 9, 3782. [Google Scholar] [CrossRef]
- Palacios-Rodríguez, G.; Quinto, L.; Lara-Gómez, M.A.; Pérez-Romero, J.; Recio, J.M.; Álvarez-Romero, M.; Cachinero-Vivar, A.M.; Hernández-Navarro, S.; Navarro-Cerrillo, R.M. Carbon Sequestration in Carob (Ceratonia Siliqua L.) Plantations under the EU Afforestation Program in Southern Spain Using Low-Density Aerial Laser Scanning (ALS) Data. Forests 2022, 13, 285. [Google Scholar] [CrossRef]
- Mhirit, O.; Et-Tobi, M. Trees Outside Forests: Morocco. In Trees Outside Forests. Towards a Better Awareness. FAO CONSERVATION GIUDE; Bellefontaine, R., Petit, S., Pain-Orcet, M., Deleporte, P., Bertault., J.-G., Eds.; FAO: Roma, Italy, 2002; ISSN 92-5-104656-5. Available online: https://www.fao.org/3/y2328e/y2328e22.htm (accessed on 11 April 2022).
- FAOSTAT. Crops and Livestock Products: Crops Primary-Carobs. Available online: https://www.fao.org/faostat/en/#data/QCL (accessed on 11 April 2022).
- Correia, P.J.; Pestana, M. Exploratory Analysis of the Productivity of Carob Tree (Ceratonia siliqua) Orchards Conducted under Dry-Farming Conditions. Sustainability 2018, 10, 2250. [Google Scholar] [CrossRef]
- Martínez-Villaluenga, C.; Peñas, E.; Rico, D.; Martin-Diana, A.B.; Portillo, M.P.; Macarulla, M.T.; De Luis, D.A.; Miranda, J. Potential Usefulness of a Wakame/Carob Functional Snack for the Treatment of Several Aspects of Metabolic Syndrome: From In Vitro to In Vivo Studies. Mar. Drugs 2018, 16, 512. [Google Scholar] [CrossRef]
- Rico, D.; Martín-Diana, A.B.; Martínez-Villaluenga, C.; Aguirre, L.; Silván, J.M.; Dueñas, M.; De Luis, D.A.; Lasa, A. In vitro Approach for Evaluation of Carob By-Products as Source Bioactive Ingredients with Potential to Attenuate Metabolic Syndrome (MetS). Heliyon 2019, 5, e01175. [Google Scholar] [CrossRef]
- Aboura, I.; Nani, A.; Belarbi, M.; Murtaza, B.; Fluckiger, A.; Dumont, A.; Benammar, C.; Tounsi, M.S.; Ghiringhelli, F.; Rialland, M.; et al. Protective Effects of Polyphenol-Rich Infusions from Carob (Ceratonia Siliqua) Leaves and Cladodes of Opuntia Ficus-Indica Against Inflammation Associated with Diet-Induced Obesity and DSS-Induced Colitis in Swiss Mice. Biomed. Pharmacother. 2017, 96, 1022–1035. [Google Scholar] [CrossRef]
- Fujita, K.; Norikura, T.; Matsui-Yuasa, I.; Kumazawa, S.; Honda, S.; Sonoda, T.; Kojima-Yuasa, A. Carob Pod Polyphenols Suppress the Differentiation of Adipocytes through Posttranscriptional Regulation of C/EBPβ. PLoS ONE 2021, 16, e0248073. [Google Scholar] [CrossRef]
- Sour, S.; Chahinez, F.; Taif, A. Beneficial Effects of Carob Pulp (Ceratonia Siliqua) on Lipids Profile and Oxidant/Antioxidant Status in Obese Rats. Rev. Agrobiol. 2019, 9, 1200–1206, e-ISSN 2507-7627. [Google Scholar]
- Hassanein, K.; Youssef, M.K.E.; Ali, H.M.; El-Manfaloty, M.M. The Influence of Carob Powder on Lipid Profile and Histopathology of Some Organs in Rats. Comp. Clin. Pathol. 2015, 24, 1509–1513. [Google Scholar] [CrossRef]
- El Rabey, H.A.; Al-Seeni, M.N.; Al-Ghamdi, H.B. Comparison between the Hypolipidemic Activity of Parsley and Carob in Hypercholesterolemic Male Rats. BioMed Res. Int. 2017, 2017, 1–9. [Google Scholar] [CrossRef]
- Al-Seeni, M.N.; El Rabey, H.A.; Al-Ghamdi, H.; Al-Sieni, A.I.; Sakran, M.I.; Mohammed, G.M. Assessment of the Antioxidant Activity of Parsley and Carob in Hypercholesterolemic Male Rats. BioMed Res. 2018, 29, 3370–3377. [Google Scholar] [CrossRef]
- Süzek, H.; Celik, I.; Doğan, A. Nephroprotective Hepatoprotective Potential and Antioxidant Role of Carob Pods (Cerotonia Siliqua L). Against Carbon Tetrachloride-Induced Toxicity in Rats. IJPER 2017, 51, 312–320. [Google Scholar] [CrossRef]
- Martić, N.; Zahorec, J.; Stilinović, N.; Andrejić-Višnjić, B.; Pavlić, B.; Kladar, N.; Šoronja-Simović, D.; Šereš, Z.; Vujčić, M.; Horvat, O.; et al. Hepatoprotective Effect of Carob Pulp Flour (Ceratonia siliqua L.) Extract Obtained by Optimized Microwave-Assisted Extraction. Pharmaceutics 2022, 14, 657. [Google Scholar] [CrossRef] [PubMed]
- de la Fuente-Fernández, M.; González-Hedström, D.; Amor, S.; Tejera-Muñoz, A.; Fernández, N.; Monge, L.; Almodóvar, P.; Andrés-Delgado, L.; Santamaría, L.; Prodanov, M.; et al. Supplementation with a Carob (Ceratonia Siliqua L.) Fruit Extract Attenuates the Cardiometabolic Alterations Associated with Metabolic Syndrome in Mice. Antioxidants 2020, 9, 339. [Google Scholar] [CrossRef] [PubMed]
- Valero-Muñoz, M.; Ballesteros, S.; Ruiz-Roso, B.; Pérez-Olleros, L.; Martín-Fernández, B.; Lahera, V.; de Las Heras, N. Supplementation with an Insoluble Fiber Obtained from Carob Pod (Ceratonia Siliqua L.) Rich in Polyphenols Prevents Dyslipidemia in Rabbits Through SIRT1/PGC-1α Pathway. Eur. J. Nutr. 2019, 58, 357–366. [Google Scholar] [CrossRef]
- Würsch, P. Influence of Tannin-Rich Carob Pod Fiber on the Cholesterol Metabolism in the Rat. J. Nutr. 1979, 109, 685–692. [Google Scholar] [CrossRef]
- Macho-González, A.; Garcimartín, A.; Naes, F.; López-Oliva, M.E.; Amores-Arrojo, A.; González-Muñoz, M.J.; Bastida, S.; Benedí, J.; Sánchez-Muniz, F.J. Effects of Fiber Purified Extract of Carob Fruit on Fat Digestion and Postprandial Lipemia in Healthy Rats. J. Agric. Food Chem. 2018, 66, 6734–6741. [Google Scholar] [CrossRef]
- Pérez-Olleros, L.; García-Cuevas, M.; Ruiz-Roso, B.; Requejo, A. Comparative study of natural carob fibre and psyllium husk in rats. Influence on some aspects of nutritional utilisation and lipidaemia. J. Sci. Food Agric. 1999, 79, 173–178. [Google Scholar] [CrossRef]
- El-Haskoury, R.; Al-Waili, N.; El-Hilaly, J.; Al-Waili, W.; Lyoussi, B. Antioxidant, Hypoglycemic, and Hepatoprotective Effect of Aqueous and Ethyl Acetate Extract of Carob Honey in Streptozotocin-Induced Diabetic Rats. Vet. World 2019, 12, 1916–1923. [Google Scholar] [CrossRef]
- Souli, A.; Sebai, H.; Chehimi, L.; Rtibi, K.; Tounsi, H.; Boubaker, S.; Sakly, M.; El-Benna, J.; Amri, M. Hepatoprotective Effect of Carob Against Acute Ethanol-Induced Oxidative Stress in Rat. Toxicol. Ind. Health 2015, 31, 802–810. [Google Scholar] [CrossRef]
- Temiz, M.A.; Temur, A.; Çelik, I. Antioxidant Role and Hepatoprotective Effects of Carob (Ceratonia Siliqua L.) Seeds against Ethanol-Induced Oxidative Stress in Rats. J. Food Nutr. Res. 2015, 3, 57–61. [Google Scholar] [CrossRef]
- Papakonstantinou, E.; Orfanakos, N.; Farajian, P.; Kapetanakou, A.E.; Makariti, I.P.; Grivokostopoulos, N.; Ha, M.A.; Skandamis, P.N. Short-Term Effects of a Low Glycemic Index Carob-Containing Snack on Energy Intake, Satiety, and Glycemic Response in Normal-Weight, Healthy Adults: Results from Two Randomized Trials. Nutrition 2017, 42, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Jaffari, H.; Abedi, B.; Fatolah, H. The Effect of 8 Weeks of Carob Supplementation and Resistance Training on Lipid Profile and Irisin in Obese Men. IJSEHR 2020, 4, 91–95. [Google Scholar] [CrossRef]
- Zunft, H.; Lüder, W.; Harde, A.; Haber, B.; Graubaum, H.J.; Gruenwald, J. Carob Pulp Preparation for Treatment of Hypercholesterolemia. Adv. Ther. 2001, 18, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Zunft, H.J.; Lüder, W.; Harde, A.; Haber, B.; Graubaum, H.J.; Koebnick, C.; Grünwald, J. Carob Pulp Preparation Rich in Insoluble Fibre Lowers Total and LDL Cholesterol in Hypercholesterolemic Patients. Eur. J. Nutr. 2003, 42, 235–242. [Google Scholar] [CrossRef]
- Ruiz-Roso, B.; Quintela, J.C.; de la Fuente, E.; Haya, J.; Pérez-Olleros, L. Insoluble Carob Fiber Rich in Polyphenols Lowers Total and LDL Cholesterol in Hypercholesterolemic Subjects. Plant Foods Hum. Nutr. 2010, 65, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Forestieri, A.M.; Galati, E.M.; Trovato, A.; Tumino, G. Effects of Guar and Carob Gums on Glucose, Insulin and Cholesterol Plasma Levels in the Rat. Phytother. Res. 1989, 3, 1–4. [Google Scholar] [CrossRef]
- Macho-González, A.; Garcimartín, A.; López-Oliva, M.E.; Bertocco, G.; Naes, F.; Bastida, S.; Sánchez-Muniz, F.J.; Benedí, J. Fiber Purified Extracts of Carob Fruit Decrease Carbohydrate Absorption. Food Funct. 2017, 8, 2258–2265. [Google Scholar] [CrossRef]
- Macho-González, A.; Garcimartín, A.; López-Oliva, M.E.; Celada, P.; Bastida, S.; Benedí, J.; Sánchez-Muniz, F.J. Carob-Fruit-Extract-Enriched Meat Modulates Lipoprotein Metabolism and Insulin Signaling in Diabetic Rats Induced by High-Saturated-Fat Diet. J. Funct. Foods 2020, 64, 103600. [Google Scholar] [CrossRef]
- Macho-González, A.; López-Oliva, M.E.; Merino, J.J.; García-Fernández, R.A.; Garcimartín, A.; Redondo-Castillejo, R.; Bastida, S.; Sánchez-Muniz, F.J.; Benedí, J. Carob Fruit Extract-Enriched Meat Improves Pancreatic Beta-Cell Dysfunction, Hepatic Insulin Signaling and Lipogenesis in Late-Stage Type 2 Diabetes Mellitus Model. J. Nutr. Biochem. 2020, 84, 108461. [Google Scholar] [CrossRef]
- Macho-González, A.; Garcimartín, A.; Redondo, N.; Cofrades, S.; Bastida, S.; Nova, E.; Benedí, J.; Sánchez-Muniz, F.J.; Marcos, A.; Elvira López-Oliva, M. Carob Fruit Extract-Enriched Meat, as Preventive and Curative Treatments, Improves Gut Microbiota and Colonic Barrier Integrity in A Late-Stage T2DM Model. Food Res. Int. 2021, 141, 110124. [Google Scholar] [CrossRef] [PubMed]
- Macho-González, A.; Garcimartín, A.; López-Oliva, M.E.; Ruiz-Roso, B.; Martín de la Torre, I.; Bastida, S.; Benedí, J.; Sánchez-Muniz, F.J. Can Carob-Fruit-Extract-Enriched Meat Improve the Lipoprotein Profile, VLDL-Oxidation, and LDL Receptor Levels Induced by an Atherogenic Diet in STZ-NAD-Diabetic Rats? Nutrients 2019, 11, 332. [Google Scholar] [CrossRef] [PubMed]
- Qasem, M.A.; Noordin, M.I.; Arya, A.; Alsalahi, A.; Jayash, S.N. Evaluation of the Glycemic Effect of Ceratonia Siliqua Pods (Carob) on a Streptozotocin-Nicotinamide Induced Diabetic Rat Model. PeerJ 2018, 6, e4788. [Google Scholar] [CrossRef] [PubMed]
- Rtibi, K.; Selmi, S.; Grami, D.; Saidani, K.; Sebai, H.; Amri, M.; Eto, B.; Marzouki, L. Ceratonia Siliqua L. (Immature Carob Bean) Inhibits Intestinal Glucose Absorption, Improves Glucose Tolerance and Protects Against Alloxan-Induced Diabetes in Rat. J. Sci. Food Agric. 2017, 97, 2664–2670. [Google Scholar] [CrossRef] [PubMed]
- Lambert, C.; Cubedo, J.; Padró, T.; Vilahur, G.; López-Bernal, S.; Rocha, M.; Hernández-Mijares, A.; Badimon, L. Effects of a Carob-Pod-Derived Sweetener on Glucose Metabolism. Nutrients 2018, 10, 271. [Google Scholar] [CrossRef] [PubMed]
- Navarro, J.A.; Decara, J.; Medina-Vera, D.; Tovar, R.; Suarez, J.; Pavón, J.; Serrano, A.; Vida, M.; Gutierrez-Adan, A.; Sanjuan, C.; et al. D-Pinitol from Ceratonia Siliqua is an Orally Active Natural Inositol that Reduces Pancreas Insulin Secretion and Increases Circulating Ghrelin Levels in Wistar Rats. Nutrients 2020, 12, 2030. [Google Scholar] [CrossRef]
- Abidar, S.; Yildiz, O.; Degirmenci, A.; Amakran, A.; El Maadoudi, M.; Nhiri, M. Glucose-Mediated Protein Glycation: Contribution of Methanolic Extract of Ceratonia Siliqua L. In Protection and In Vitro Potential Inhibition of Acetylcholinesterase. J. Food Biochem. 2019, 43, e13009. [Google Scholar] [CrossRef]
- Rtibi, K.; Jabri, M.A.; Selmi, S.; Souli, A.; Sebai, H.; El-Benna, J.; Amri, M.; Marzouki, L. Carob Pods (Ceratonia Siliqua L.) Inhibit Human Neutrophils Myeloperoxidase and In Vitro ROS-Scavenging Activity. RSC Adv. 2015, 5, 84207–84215. [Google Scholar] [CrossRef]
- Sassi, A.; Bouhlel, I.; Mustapha, N.; Mokdad-Bzeouich, I.; Chaabane, F.; Ghedira, K.; Chekir-Ghedira, L. Assessment In Vitro of the Genotoxicity, Antigenotoxicity and Antioxidant of Ceratonia Siliqua L. Extracts in Murine Leukaemia Cells L1210 by Comet assay. Regul. Toxicol. Pharmacol. 2016, 77, 117–124. [Google Scholar] [CrossRef]
- Papakonstantinou, E.; Chaloulos, P.; Papalexi, A.; Mandala, I. Effects of Bran Size and Carob Seed Flour of Optimized Bread Formulas on Glycemic Responses in Humans: A Randomized Clinical Trial. J. Funct. Foods 2018, 46, 345–355. [Google Scholar] [CrossRef]
- Gruendel, S.; Otto, B.; Garcia, A.L.; Wagner, K.; Mueller, C.; Weickert, M.O.; Heldwein, W.; Koebnick, C. Carob pulp Preparation Rich in Insoluble Dietary Fibre and Polyphenols Increases Plasma Glucose and Serum Insulin Responses in Combination with a Glucose Load in Humans. Br. J. Nutr. 2007, 98, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Gruendel, S.; Garcia, A.L.; Otto, B.; Wagner, K.; Bidlingmaier, M.; Burget, L.; Weickert, M.O.; Dongowski, G.; Speth, M.; Katz, N.; et al. Increased Acylated Plasma Ghrelin, But Improved Lipid Profiles 24-h after Consumption of Carob Pulp Preparation Rich in Dietary Fibre And Polyphenols. Br. J. Nutr. 2007, 98, 1170–1177. [Google Scholar] [CrossRef]
- Milek Dos Santos, L.; Tomzack Tulio, L.; Fuganti Campos, L.; Ramos Dorneles, M.; Carneiro Hecke Krüger, C. Glycemic Response to Carob (Ceratonia Siliqua L.) in Healthy Subjects and With The in Vitro Hydrolysis Index. Nutr. Hosp. 2014, 31, 482–487. [Google Scholar] [CrossRef]
- Bañuls, C.; Rovira-Llopis, S.; Falcón, R.; Veses, S.; Monzó, N.; Víctor, V.M.; Rocha, M.; Hernández-Mijares, A. Chronic Consumption of an Inositol-Enriched Carob Extract Improves Postprandial Glycaemia and Insulin Sensitivity in Healthy Subjects: A Randomized Controlled Trial. Clin. Nutr. 2016, 35, 600–607. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Mijares, A.; Bañuls, C.; Rovira-Llopis, S.; Álvarez, Á.; Orden, S.; Rubio-Puchol, O.; Víctor, V.M.; Rocha, M. Chronic Consumption of an Inositol-Enriched Beverage Ameliorates Endothelial Dysfunction and Oxidative Stress in Type 2 Diabetes. J. Funct. Foods 2015, 18, 598–607. [Google Scholar] [CrossRef]
- Bañuls, C.; Rovira-Llopis, S.; López-Doménech, S.; Veses, S.; Víctor, V.M.; Rocha, M.; Hernández-Mijares, A. Effect of Consumption of a Carob Pod Inositol-Enriched Beverage on Insulin Sensitivity and Inflammation in Middle-Aged Prediabetic Subjects. Food Funct. 2016, 7, 4379–4387. [Google Scholar] [CrossRef] [PubMed]
- López-Sánchez, J.; Moreno, D.A.; García-Viguer, C. D-pinitol, A Highly Valuable Product From Carob Pods: Health-Promoting Effects and Metabolic Pathways of This Natural Super-Food Ingredient and Its Derivatives. AIMS Agric. Food 2018, 3, 41–63. [Google Scholar] [CrossRef]
- Rico, D.; Martin-Diana, A.B.; Lasa, A.; Aguirre, L.; Milton-Laskibar, I.; De Luis, D.A.; Miranda, J. Effect of Wakame and Carob Pod Snacks on Non-Alcoholic Fatty Liver Disease. Nutrients 2019, 11, 86. [Google Scholar] [CrossRef]
- El Haskoury, R.; Zizi, S.; Touzani, S.; Al Waili, N.; AlGhamdi, A.; Lyoussi, B. Diuretic Activity of Carob (Ceratonia Silique L.) Honey: Comparison with Furosemide. Afr. J. Tradit. Complement. Altern. Med. 2015, 12, 128–133. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gioxari, A.; Amerikanou, C.; Nestoridi, I.; Gourgari, E.; Pratsinis, H.; Kalogeropoulos, N.; Andrikopoulos, N.K.; Kaliora, A.C. Carob: A Sustainable Opportunity for Metabolic Health. Foods 2022, 11, 2154. https://doi.org/10.3390/foods11142154
Gioxari A, Amerikanou C, Nestoridi I, Gourgari E, Pratsinis H, Kalogeropoulos N, Andrikopoulos NK, Kaliora AC. Carob: A Sustainable Opportunity for Metabolic Health. Foods. 2022; 11(14):2154. https://doi.org/10.3390/foods11142154
Chicago/Turabian StyleGioxari, Aristea, Charalampia Amerikanou, Irini Nestoridi, Eleni Gourgari, Harris Pratsinis, Nick Kalogeropoulos, Nikolaos K. Andrikopoulos, and Andriana C. Kaliora. 2022. "Carob: A Sustainable Opportunity for Metabolic Health" Foods 11, no. 14: 2154. https://doi.org/10.3390/foods11142154
APA StyleGioxari, A., Amerikanou, C., Nestoridi, I., Gourgari, E., Pratsinis, H., Kalogeropoulos, N., Andrikopoulos, N. K., & Kaliora, A. C. (2022). Carob: A Sustainable Opportunity for Metabolic Health. Foods, 11(14), 2154. https://doi.org/10.3390/foods11142154









