Potential Uses of Spent Coffee Grounds in the Food Industry
Abstract
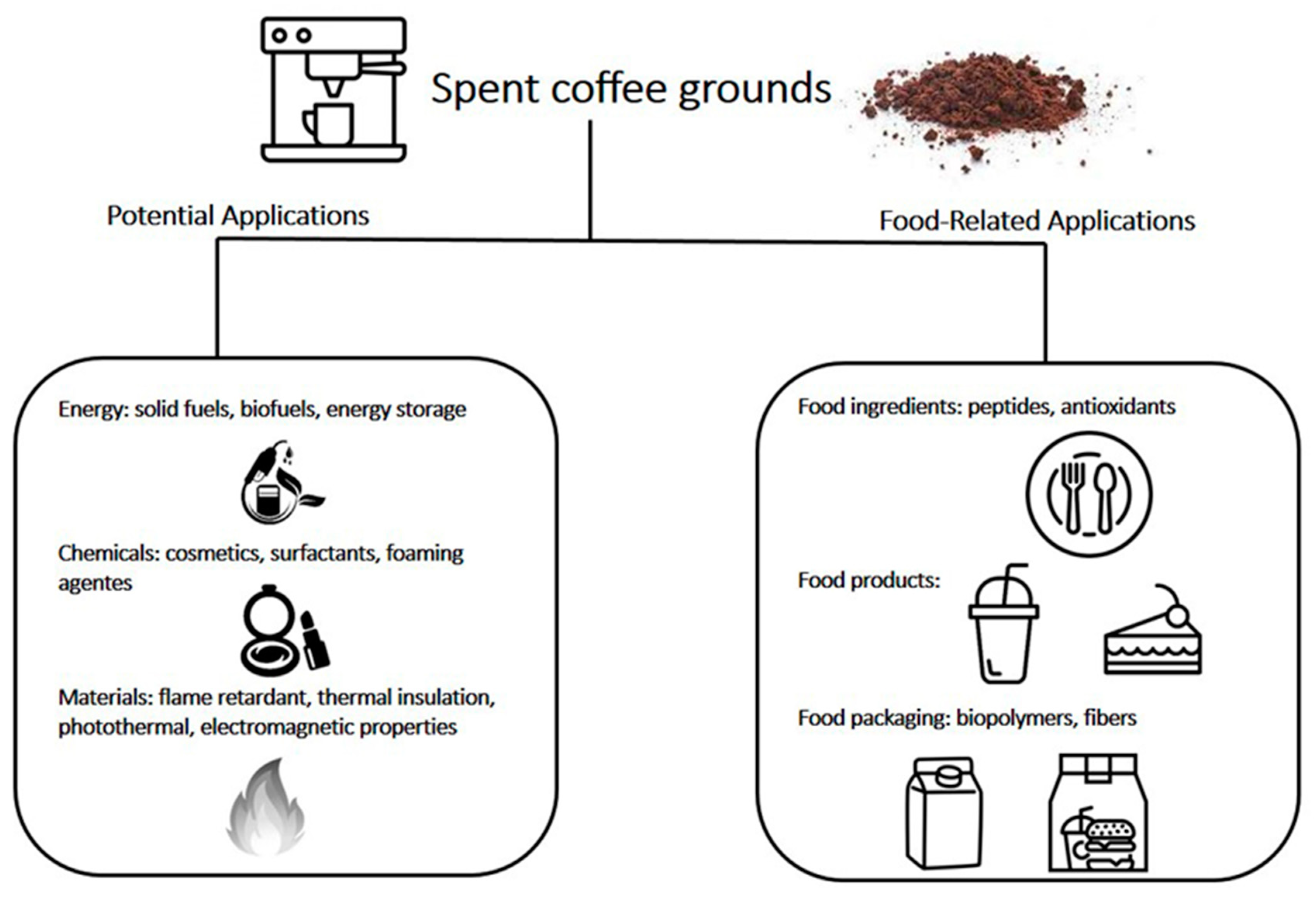
:1. Introduction
| Starting Material | Technology | Product/Application | Ref. |
|---|---|---|---|
| Energy | |||
| wood sawdust and SCG | pellet pressing | solid fuel pellets | [14] |
| SCG and larch sawdust or spruce shavings | high-pressure hydraulic briquetting press | solid fuel briquettes | [15] |
| SCG | pyrolysis | biochar/carbon cloth electrode/electricity generation and storage | [16] |
| SCG and reduced graphene oxide | vacuum-assisted impregnation | composite phase change material/solar energy storage | [17] |
| SCG | drying + oil extraction + transesterification | biodiesel | [18] |
| SCG | carbonization + CO2 activation | energy storage | [19] |
| Chemicals | |||
| SCG extract and Xanthophyllomyces dendrorhous | drying, sterilization and water extraction | astaxanthin/cosmetics, supplements and food | [20] |
| SCG extract and Millerozyma farinosa | drying, sterilization and water extraction | glutathione/medicine, food supplements and cosmetics | [21] |
| SCG | defatting + drying + hot water extraction + incubation with ammonium sulfate + centrifugation + ultra-filtration + vacuum drying | surfactants | [22] |
| SCG | solvent extraction (ultrasound, microwave or β-cyclodextrin-assisted) | phenolics | [23] |
| SCG | high-pressure temperature extraction | chlorogenic acids and caffein | [24] |
| SCG | subsequent extractions (hexane, water and ammonium sulfate) + centrifugation+ ultrafiltration + vacuum drying | foaming agent | [25] |
| Adsorbents | |||
| SCG | mixture with KOH and carbonization | biochar/ammonia removal from water | [26] |
| SCG | torrefaction | biochar/removal of diesel mixed in water | [27] |
| SCG | activation with NaOH, CaCO3 and carbonization | activated carbon/removal of methylene blue and methyl orange from water | [28] |
| SCG | no treatment | biosorbent/heavy metal (Cd) removal from aqueous solution | [29] |
| SCG | H3PO4 pyrolysis | microporous AC/removal of explosives from water | [30] |
| SCG | pyrolysis at 500 °C | biochar/removal of Norfloxacin (antibiotic) from water | [31] |
| SCG | washing with NaOH following by drying | biosorbent/recovery of dissolved metals (Fe, Al, Ca, Co, Mn, Ni, and Zn) from acid sulfate soil drainage | [32] |
| Materials | |||
| SCG and epoxy resin | mixing and curing | Flame retardant in polymer | [33] |
| SCG and Polylactide (PLA) powder | decolorization, micro- and nano-processing and extrusion | polylactic acid composite for 3D-printing | [34] |
| SCG and pectin | continuous casting | biocomposite pectin film | [35] |
| SCG and plaster | mixture with water and drying | thermal insulation material | [36] |
| SCG and hydrogels | near infra-red laser irradiation and incorporation into poly(N-isopropylacrylamide) hydrogels | photothermal materials | [37] |
| SCG | carbonization + composite mixture (cyanate ester, graphene nanoplates and epoxy resin) +hot pressing | composite with electromagnetic interference (EMI) shielding properties | [38] |
| Food products and ingredients | |||
| SCG | acid extraction followed by precipitation | bioactive peptides with antihypertensive and antioxidant potentials | [39] |
| SCG | microwave-assisted extraction of antioxidants, fermentation (Saccharomyces cerevisiae) and distillation | fermented and distilled alcoholic beverages | [40] |
| SCG | hydrolysis followed by fermentation with wine yeasts | alcoholic beverages | [41] |
| SCG | ethanolic extraction followed by addition of caramel, water, glucose syrup and vanillin | coffee-flavored liquor | [42] |
| SCG | extraction with isopropanol | antimycotic and anti-ochratoxigenic material; potential food ingredient with moderate cytotoxic and antibacterial activities | [43] |
2. Chemical Composition
3. Applications
3.1. SCG as a Source of Bioactive Compounds
3.2. SCG in Food Products
3.3. SCG in Polymers
4. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Franca, A.S.; Oliveira, L.S. Coffee. In Integrated Processing Technologies for Food and Agricultural By-Products; Pan, Z., Zhang, R., Zicari, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 413–438. [Google Scholar] [CrossRef]
- Reis, N.; Franca, A.S.; Oliveira, L.S. Performance of diffuse reflectance infrared Fourier transform spectroscopy and chemometrics for detection of multiple adulterants in roasted and ground coffee. LWT 2013, 53, 395–401. [Google Scholar] [CrossRef] [Green Version]
- Reis, N.; Botelho, B.G.; Franca, A.S.; Oliveira, L.S. Simultaneous detection of multiple adulterants in ground roasted coffee by ATR-FTIR spectroscopy and data fusion. Food Anal. Methods 2017, 10, 2700–2709. [Google Scholar] [CrossRef]
- Sodhi, A.S.; Sharma, N.; Bhatia, S.; Verma, A.; Soni, S.; Batra, N. Insights on sustainable approaches for production and applications of value added products. Chemosphere 2022, 286, 131623. [Google Scholar] [CrossRef] [PubMed]
- Banu, J.R.; Kavitha, S.; Kannah, R.Y.; Kumar, M.D.; Preethi; Atabani, A.E.; Kumar, G. Biorefinery of spent coffee grounds waste: Viable pathway towards circular bioeconomy. Bioresour. Technol. 2020, 302, 122821. [Google Scholar] [CrossRef] [PubMed]
- Cruz, R.; Morais, S.; Mendes, E.; Pereira, J.A.; Baptista, P.; Casal, S. Improvement of vegetables elemental quality by espresso coffee residues. Food Chem. 2014, 148, 294–299. [Google Scholar] [CrossRef]
- Cruz, R.; Mendes, E.; Torrinha, Á.; Morais, S.; Pereira, J.A.; Baptista, P.; Casal, S. Revalorization of spent coffee residues by a direct agronomic approach. Food Res. Int. 2015, 73, 190–196. [Google Scholar] [CrossRef]
- Cruz, R.; Gomes, T.; Ferreira, A.; Mendes, E.; Baptista, P.; Cunha, S.; Pereira, J.A.; Ramalhosa, E.; Casal, S. Antioxidant activity and bioactive compounds of lettuce improved by espresso coffee residues. Food Chem. 2014, 145, 95–101. [Google Scholar] [CrossRef]
- Hanc, A.; Hrebeckova, T.; Grasserova, A.; Cajthaml, T. Conversion of spent coffee grounds into vermicompost. Bioresour. Technol. 2021, 341, 125925. [Google Scholar] [CrossRef]
- Cervera-Mata, A.; Fernández-Arteaga, A.; Navarro-Alarcón, M.; Hinojosa, D.; Pastoriza, S.; Delgado, G.; Rufián-Henares, J.Á. Spent coffee grounds as a source of smart biochelates to increase Fe and Zn levels in lettuces. J. Clean. Prod. 2021, 328, 129548. [Google Scholar] [CrossRef]
- Sikka, S.S.; Bakshi, M.P.; Ichhponani, J.S. Evaluation in vitro of spent coffee grounds as a livestock feed. Agric. Wastes 1985, 13, 315–317. [Google Scholar] [CrossRef]
- Xu, C.C.; Cai, Y.; Zhang, J.G.; Ogawa, M. Fermentation quality and nutritive value of a total mixed ration silage containing coffee grounds at ten or twenty percent of dry matter. J. Anim. Sci. 2007, 85, 1024–1029. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Otálora, X.D.; Ruiz, R.; Goiri, I.; Rey, J.; Atxaerandio, R.; Martin, D.S.; Orive, M.; Iñarra, B.; Zufia, J.; Urkiza, J.; et al. Valorisation of spent coffee grounds as functional feed ingredient improves productive performance of Latxa dairy ewes. Anim. Feed Sci. Technol. 2020, 264, 114461. [Google Scholar] [CrossRef]
- Nosek, R.; Tun, M.M.; Juchelkova, D. Energy utilization of spent coffee grounds in the form of pellets. Energies 2020, 13, 1235. [Google Scholar] [CrossRef] [Green Version]
- Brunerová, A.; Roubík, H.; Brožek, M.; Haryanto, A.; Hasanudin, U.; Iryani, D.; Herák, D. Valorization of bio-briquette fuel by using spent coffee ground as an external additive. Energies 2020, 13, 54. [Google Scholar] [CrossRef] [Green Version]
- Andrade, T.S.; Vakros, J.; Mantzavinos, D.; Lianos, P. Biochar obtained by carbonization of spent coffee grounds and its application in the construction of an energy storage device. Chem. Eng. J. Adv. 2020, 4, 100061. [Google Scholar] [CrossRef]
- Hu, X.; Huang, H.; Hu, Y.; Lu, X.; Qin, Y. Novel bio-based composite phase change materials with reduced graphene oxide-functionalized spent coffee grounds for efficient solar-to-thermal energy storage. Sol. Energy Mater. Sol. Cells 2021, 219, 110790. [Google Scholar] [CrossRef]
- Kamil, M.; Ramadan, K.M.; Awad, O.I.; Ibrahim, T.K.; Inayat, A.; Ma, X. Environmental impacts of biodiesel production from waste spent coffee grounds and its implementation in a compression ignition engine. Sci. Total Environ. 2019, 675, 13–30. [Google Scholar] [CrossRef]
- Hsieh, T.-H.; Wang, H.-L.; Yu, G.-T.; Huang, G.-M.; Lin, J.-H. Meso-pore dominant activated carbon from spent coffee grounds for high-performance electrochemical capacitors in organic electrolyte. J. Environ. Chem. Eng. 2021, 9, 106418. [Google Scholar] [CrossRef]
- Hirono-Hara, Y.; Mizutani, Y.; Murofushi, K.; Iwahara, K.; Sakuragawa, S.; Kikukawa, H.; Hara, K.Y. Effect of spent coffee grounds extract on astaxanthin production by Xanthophyllomyces dendrorhous. Bioresour. Technol. Rep. 2022, 17, 100953. [Google Scholar] [CrossRef]
- Hirono-Hara, Y.; Mizutani, Y.; Murofushi, K.; Iwahara, K.; Sakuragawa, S.; Kikukawa, H.; Hara, K.Y. Glutathione fermentation by Millerozyma farinosa using spent coffee grounds extract and seawater. Bioresour. Technol. Rep. 2021, 15, 100777. [Google Scholar] [CrossRef]
- Ishwarya, S.P.; Nisha, P. Insights into the composition, structure-function relationship, and molecular organization of surfactants from spent coffee grounds. Food Hydrocoll. 2022, 124, 107204. [Google Scholar] [CrossRef]
- Solomakou, N.; Loukri, A.; Tsafrakidou, P.; Michaelidou, A.-M.; Mourtzinos, I.; Goula, A.M. Recovery of phenolic compounds from spent coffee grounds through optimized extraction processes. Sustain. Chem. Pharm. 2022, 25, 100592. [Google Scholar] [CrossRef]
- da Silva, M.F.; Pettinato, M.; Casazza, A.A.; Maciel, M.I.; Perego, P. Design and evaluation of non-conventional extraction for bioactive compounds recovery from spent coffee (Coffea arabica L.) grounds. Chem. Eng. Res. Des. 2022, 177, 418–430. [Google Scholar] [CrossRef]
- Ishwarya, S.P.; Nisha, P. Foaming agents from spent coffee grounds: A mechanistic understanding of the modes of foaming and the role of coffee oil as antifoam. Food Hydrocoll. 2021, 112, 106354. [Google Scholar] [CrossRef]
- Nguyen, V.-T.; Vo, T.-D.-H.; Tran, T.; Nguyen, T.-N.; Le, T.-N.-C.; Bui, X.-T.; Bach, L.-G. Biochar derived from the spent coffee ground for ammonium adsorption from aqueous solution. Case Stud. Chem. Environ. Eng. 2021, 4, 100141. [Google Scholar] [CrossRef]
- Lee, K.-T.; Cheng, C.-L.; Lee, D.-S.; Chen, W.-H.; Vo, D.-V.N.; Ding, L.; Lam, S.S. Spent coffee grounds biochar from torrefaction as a potential adsorbent for spilled diesel oil recovery and as an alternative fuel. Energy 2022, 239, 122467. [Google Scholar] [CrossRef]
- Block, I.; Günter, C.; Rodrigues, A.D.; Paasch, S.; Hesemann, P.; Taubert, A. Carbon adsorbents from spent coffee for removal of methylene blue and methyl orange from water. Materials 2021, 14, 3996. [Google Scholar] [CrossRef]
- Kim, M.-S.; Kim, J.-G. Adsorption characteristics of spent coffee grounds as an alternative adsorbent for cadmium in solution. Environments 2020, 7, 24. [Google Scholar] [CrossRef] [Green Version]
- Charmas, B.; Zięzio, M.; Tomaszewski, W.; Kucio, K. Smart preparation of microporous carbons from spent coffee grounds. Comprehensive characterization and application in explosives removal from water samples. Colloids Surf. A Physicochem. Eng. Asp. 2022, 645, 128889. [Google Scholar] [CrossRef]
- Nguyen, V.-T.; Vo, T.-D.-H.; Nguyen, T.-B.; Dat, N.D.; Huu, B.T.; Nguyen, X.-C.; Tran, T.; Le, T.-N.-C.; Duong, T.-G.-H.; Bui, M.-H.; et al. Adsorption of norfloxacin from aqueous solution on biochar derived from spent coffee ground: Master variables and response surface method optimized adsorption process. Chemosphere 2022, 288, 132577. [Google Scholar] [CrossRef]
- Shahabi-Ghahfarokhi, S.; Rahmati-Abkenar, M.; Matson, J.G.; Karimi, H.; Yu, C.; Hogland, W.; Klavinš, M.; Ketzer, M. Removal and potential recovery of dissolved metals from acid sulfate soil drainage by spent coffee-grounds and dissolved organic carbon. Environ. Adv. 2022, 8, 100193. [Google Scholar] [CrossRef]
- Vahabi, H.; Jouyandeh, M.; Parpaite, T.; Saeb, M.R.; Ramakrishna, S. Coffee Wastes as Sustainable Flame Retardants for Polymer Materials. Coatings 2021, 11, 1021. [Google Scholar] [CrossRef]
- Li, S.; Shi, C.; Sun, S.; Chan, H.; Lu, H.; Nilghaz, A.; Tian, J.; Cao, R. From brown to colored: Polylactic acid composite with micro/nano-structured white spent coffee grounds for three-dimensional printing. Int. J. Biol. Macromol. 2021, 174, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Mendes, J.F.; Martins, J.T.; Manrich, A.; Neto, A.R.; Pinheiro, A.C.; Mattoso, L.H.; Martins, M.A. Development and physical-chemical properties of pectin film reinforced with spent coffee grounds by continuous casting. Carbohydr. Polym. 2019, 210, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Lachheb, A.; Allouhi, A.; Marhoune, M.E.; Saadani, R.; Kousksou, T.; Jamil, A.; Rahmoune, M.; Oussouaddi, O. Thermal insulation improvement in construction materials by adding spent coffee grounds: An experimental and simulation study. J. Clean. Prod. 2019, 209, 1411–1419. [Google Scholar] [CrossRef]
- Chen, X.-E.; Mangindaan, D.; Chien, H.-W. Green sustainable photothermal materials by spent coffee grounds. J. Taiwan Inst. Chem. Eng. 2022, 346, 104259. [Google Scholar] [CrossRef]
- Guo, Z.; Ren, P.; Zhang, Z.; Dai, Z.; Lu, Z.; Jin, Y.; Ren, F. Fabrication of carbonized spent coffee grounds/graphene nanoplates/cyanate ester composites for superior and highly absorbed electromagnetic interference shielding performance. J. Mater. Sci. Technol. 2022, 102, 123–131. [Google Scholar] [CrossRef]
- Ribeiro, E.; de Souza Rocha, T.; Prudencio, S.H. Potential of green and roasted coffee beans and spent coffee grounds to provide bioactive peptides. Food Chem. 2021, 348, 129061. [Google Scholar] [CrossRef]
- Machado, E.; Mussatto, S.; Teixeira, J.; Vilanova, M.; Oliveira, J. Increasing the sustainability of the coffee agro-industry: Spent coffee grounds as a source of new beverages. Beverages 2018, 4, 105. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Lu, Y.; Liu, S.Q. The potential of spent coffee grounds hydrolysates fermented with Torulaspora delbrueckii and Pichia kluyveri for developing an alcoholic beverage: The yeasts growth and chemical compounds modulation by yeast extracts. Curr. Res. Food Sci. 2021, 4, 489–498. [Google Scholar] [CrossRef]
- Masino, F.; Montevecchi, G.; Calvini, R.; Foca, G.; Antonelli, A. Sensory evaluation and mixture design assessment of coffee-flavored liquor obtained from spent coffee grounds. Food Qual. Prefer. 2022, 96, 104427. [Google Scholar] [CrossRef]
- Badr, A.N.; El-Attar, M.M.; Ali, H.S.; Elkhadragy, M.F.; Yehia, H.M.; Farouk, A. Spent coffee grounds valorization as bioactive phenolic source acquired antifungal, anti-mycotoxigenic, and anti-cytotoxic activities. Toxins 2022, 14, 109. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.-H.; Park, J.-H.; Im, S.-S.; Song, D.-K. Coffee and health. Integr. Med. Res. 2014, 3, 189–191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Costabile, A.; Sarnsamak, K.; Hauge-Evans, A.C. Coffee, type 2 diabetes and pancreatic islet function—A mini-review. J. Funct. Foods 2018, 45, 409–416. [Google Scholar] [CrossRef] [Green Version]
- Sedaghat, G.; Mirshekar, M.A.; Amirpour, M.; Montazerifar, F.; Miri, S.; Shourestani, S. Sub-chronic administration of brewed coffee on rat behavior and cognition and oxidative stress Alzheimer\textquotesingles disease model. Clin. Nutr. Exp. 2019, 28, 62–73. [Google Scholar] [CrossRef] [Green Version]
- AlAmri, O.D.; Albeltagy, R.S.; Akabawy, A.M.; Mahgoub, S.; Abdel-Mohsen, D.M.; Moneim, A.E.; Amin, H.K. Investigation of antioxidant and anti-inflammatory activities as well as the renal protective potential of green coffee extract in high fat-diet/streptozotocin-induced diabetes in male albino rats. J. Funct. Foods 2020, 71, 103996. [Google Scholar] [CrossRef]
- Grzelczyk, J.; Budryn, G.; Peña-García, J.; Szwajgier, D.; Gałązka-Czarnecka, I.; Oracz, J.; Pérez-Sánchez, H. Evaluation of the inhibition of monoamine oxidase A by bioactive coffee compounds protecting serotonin degradation. Food Chem. 2021, 348, 129108. [Google Scholar] [CrossRef] [PubMed]
- Montenegro, J.; dos Santos, L.S.; de Souza, R.G.; Lima, L.G.; Mattos, D.S.; Viana, B.P.; Fonseca Bastos, A.C.S.; Muzzi, L.; Conte-Júnior, C.A.; Gimba, E.R.P.; et al. Bioactive compounds, antioxidant activity and antiproliferative effects in prostate cancer cells of green and roasted coffee extracts obtained by microwave-assisted extraction (MAE). Food Res. Int. 2021, 140, 110014. [Google Scholar] [CrossRef]
- Zhu, M.; Jatoi, A. Colorectal Cancer, Crohn-Like Lymphoid Reactions, Survival—And the Power of a Good Cup of Coffee! Mayo Clin. Proc. 2022, 97, 15–17. [Google Scholar] [CrossRef] [PubMed]
- Açıkalın, B.; Sanlier, N. Coffee and its effects on the immune system. Trends Food Sci. Technol. 2021, 114, 625–632. [Google Scholar] [CrossRef]
- Lara-Guzmán, O.J.; Álvarez, R.; Muñoz-Durango, K. Changes in the plasma lipidome of healthy subjects after coffee consumption reveal potential cardiovascular benefits: A randomized controlled trial. Free Radic. Biol. Med. 2021, 176, 345–355. [Google Scholar] [CrossRef] [PubMed]
- LIczbiński, P.; Bukowska, B. Tea and coffee polyphenols and their biological properties based on the latest in vitro investigations. Ind. Crops Prod. 2022, 175, 114265. [Google Scholar] [CrossRef] [PubMed]
- Hall, S.; Desbrow, B.; Anoopkumar-Dukie, S.; Davey, A.K.; Arora, D.; McDermott, C.; Schubert, M.M.; Perkins, A.V.; Kiefel, M.J.; Grant, G.D. A review of the bioactivity of coffee, caffeine and key coffee constituents on inflammatory responses linked to depression. Food Res. Int. 2015, 76, 626–636. [Google Scholar] [CrossRef] [PubMed]
- Franca, A.S.; Oliveira, L.S. Chemistry of Defective Coffee Beans. In Progress in Food Chemistry; Koeffer, E.N., Ed.; Nova Publishers: New York, NY, USA, 2008; pp. 105–138. [Google Scholar]
- Ballesteros, L.F.; Teixeira, J.A.; Mussatto, S.I. Chemical, Functional, and Structural Properties of Spent Coffee Grounds and Coffee Silverskin. Food Bioprocess Technol. 2015, 7, 3493–3503. [Google Scholar] [CrossRef] [Green Version]
- Bijla, L.; Aissa, R.; Bouzid, H.A.; Sakar, E.H.; Ibourki, M.; Gharby, S. Spent Coffee Ground Oil as a Potential Alternative for Vegetable Oil Production: Evidence from Oil Content, Lipid Profiling, and Physicochemical Characterization. Biointerface Res. Appl. Chem. 2021, 12, 6308–6320. [Google Scholar] [CrossRef]
- Nzekoue, K.; Khamitova, G.; Angeloni, S.; Sempere, A.N.; Tao, J.; Maggi, F.; Xiao, J.; Sagratini, G.; Vittori, S.; Caprioli, G. Spent coffee grounds: A potential commercial source of phytosterols. Food Chem. 2020, 325, 126836. [Google Scholar] [CrossRef]
- Marx, S.; Venter, R.; Karmee, S.K.; Louw, J.; Truter, C. Biofuels from spent coffee grounds: Comparison of processing routes. Biofuels 2020, 13, 537–543. [Google Scholar] [CrossRef]
- Bijla, L.; Aissa, R.; Laknifli, A.; Bouyahya, A.; Harhar, H.; Gharby, S. Spent coffee grounds: A sustainable approach toward novel perspectives of valorization. J. Food Biochem. 2022, e14190. [Google Scholar] [CrossRef]
- Devasagayam, T.P.; Kamat, J.P.; Mohan, H.; Kesavan, P.C. Caffeine as an antioxidant: Inhibition of lipid peroxidation induced by reactive oxygen species. Biochim. Biophys. Acta (BBA)-Biomembr. 1996, 1282, 63–70. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, L.S.; Franca, A.S. Potential of spent coffee grounds as sources of dietary fiber with antioxidant activity. In Food Waste: Practices, Management and Challenges; Riley, G.L., Ed.; Nova Publishers: New York, NY, USA, 2016; pp. 51–70. [Google Scholar]
- Franca, A.S. Coffee: Decaffeination. In Encyclopedia of Food and Health; Elsevier: Amsterdam, The Netherlands, 2016; pp. 232–236. [Google Scholar] [CrossRef]
- Campos-Vega, R.; Loarca-Piña, G.; Vergara-Castañeda, H.A.; Oomah, B.D. Spent coffee grounds: A review on current research and future prospects. Trends Food Sci. Technol. 2015, 45, 24–36. [Google Scholar] [CrossRef]
- Oosterveld, A.; Voragen, A.G.; Schols, H.A. Effect of roasting on the carbohydrate composition of Coffea arabica beans. Carbohydr. Polym. 2003, 54, 183–192. [Google Scholar] [CrossRef]
- Mussatto, S.I.; Carneiro, L.M.; Silva, J.P.; Roberto, I.C.; Teixeira, J.A. A study on chemical constituents and sugars extraction from spent coffee grounds. Carbohydr. Polym. 2011, 83, 368–374. [Google Scholar] [CrossRef] [Green Version]
- Simões, J.; Nunes, F.M.; Domingues, M.R.; Coimbra, M.A. Extractability and structure of spent coffee ground polysaccharides by roasting pre-treatments. Carbohydr. Polym. 2013, 97, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Passos, C.P.; Coimbra, M.A. Microwave superheated water extraction of polysaccharides from spent coffee grounds. Carbohydr. Polym. 2013, 94, 626–633. [Google Scholar] [CrossRef] [PubMed]
- Moreira, A.S.; Nunes, F.M.; Domingues, M.R.; Coimbra, M.A. Galactomannans in Coffee. In Coffee in Health and Disease Prevention; Preedy, V.R., Ed.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 173–182. [Google Scholar] [CrossRef]
- Vardon, D.R.; Moser, B.R.; Zheng, W.; Witkin, K.; Evangelista, R.L.; Strathmann, T.J.; Rajagopalan, K.; Sharma, B.K. Complete utilization of spent coffee grounds to produce biodiesel, bio-oil, and biochar. ACS Sustain. Chem. Eng. 2013, 1, 1286–1294. [Google Scholar] [CrossRef]
- Vilela, W.F.; Leão, D.P.; Franca, A.S.; Oliveira, L.S. Effect of peroxide treatment on functional and technological properties of fiber-rich powders based on spent coffee grounds. ETP Int. J. Food Eng. 2016, 2, 42–47. [Google Scholar] [CrossRef]
- Murthy, P.S.; Naidu, M.M. Recovery of phenolic antioxidants and functional compounds from coffee industry by-products. Food Bioprocess Technol. 2010, 5, 897–903. [Google Scholar] [CrossRef]
- Tejada-Ortigoza, V.; García-Amezquita, L.E.; Serna-Saldívar, S.O.; Welti-Chanes, J. The dietary fiber profile of fruit peels and functionality modifications induced by high hydrostatic pressure treatments. Food Sci. Technol. Int. 2017, 23, 396–402. [Google Scholar] [CrossRef]
- dos Santos Opuski de Almeida, J.; Dias, C.O.; Arriola, N.D.; de Freitas, B.S.; de Francisco, A.; Petkowicz, C.L.; Araujo, L.; Guerra, M.P.; Nodari, R.O.; Amboni, R.D. Feijoa (Acca sellowiana) peel flours: A source of dietary fibers and bioactive compounds. Food Biosci. 2020, 38, 100789. [Google Scholar] [CrossRef]
- Elleuch, M.; Bedigian, D.; Roiseux, O.; Besbes, S.; Blecker, C.; Attia, H. Dietary fibre and fibre-rich by-products of food processing: Characterisation, technological functionality and commercial applications: A review. Food Chem. 2011, 124, 411–421. [Google Scholar] [CrossRef]
- Leão, D.P.; Franca, A.S.; Oliveira, L.S.; Bastos, R.; Coimbra, M.A. Physicochemical characterization, antioxidant capacity, total phenolic and proanthocyanidin content of flours prepared from pequi (Caryocar brasilense Camb.) fruit by-products. Food Chem. 2017, 225, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, R.C.; Esposito, F.; Napolitano, A.; Ritieni, A.; Fogliano, V. Characterization of a New Potential Functional Ingredient: Coffee Silverskin. J. Agric. Food Chem. 2014, 52, 1338–1343. [Google Scholar] [CrossRef] [PubMed]
- Cangussu, L.B.; Melo, J.C.; Franca, A.S.; Oliveira, L.S. Chemical Characterization of Coffee Husks, a By-Product of Coffea arabica Production. Foods 2021, 10, 3125. [Google Scholar] [CrossRef] [PubMed]
- Resende, L.M.; Franca, A.S.; Oliveira, L.S. Buriti (Mauritia flexuosa L. f.) fruit by-products flours: Evaluation as source of dietary fibers and natural antioxidants. Food Chem. 2019, 270, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Viuda-Martos, M.; Ruiz-Navajas, Y.; Martin-Sánchez, A.; Sánchez-Zapata, E.; Fernández-López, J.; Sendra, E.; Sayas-Barberá, E.; Navarro, C.J.; Pérez-Álvarez, J.A. Chemical, physico-chemical and functional properties of pomegranate (Punica granatum L.) bagasses powder co-product. J. Food Eng. 2012, 110, 220–224. [Google Scholar] [CrossRef]
- Gawlik-Dziki, U.; Świeca, M.; Dziki, D.; Kowalska, I.; Pecio, Ł.; Durak, A.; Sęczyk, Ł. Lipoxygenase inhibitors and antioxidants from green coffee—Mechanism of action in the light of potential bioaccessibility. Food Res. Int. 2014, 61, 48–55. [Google Scholar] [CrossRef]
- Yin, P.; Xie, S.; Zhuang, Z.; Fang, H.; Tian, L.; Liu, Y.; Niu, J. Chlorogenic acid improves health in juvenile largemouth bass (Micropterus salmoides) fed high-fat diets: Involvement of lipid metabolism, antioxidant ability, inflammatory response, and intestinal integrity. Aquaculture 2011, 545, 737169. [Google Scholar] [CrossRef]
- Suzuki, A.; Kagawa, D.; Ochiai, R.; Tokimitsu, I.; Saito, I. Green coffee bean extract and its metabolites have a hypotensive effect in spontaneously hypertensive rats. Hypertens. Res. 2002, 25, 99–107. [Google Scholar] [CrossRef] [Green Version]
- Macheiner, L.; Schmidt, A.; Mayer, H.K. A novel basis for monitoring the coffee roasting process: Isomerization reactions of 3-caffeoylquinic and 4-caffeoylquinic acids. LWT 2021, 152, 112343. [Google Scholar] [CrossRef]
- McNutt, J.; He, Q. Spent coffee grounds: A review on current utilization. J. Ind. Eng. Chem. 2019, 71, 78–88. [Google Scholar] [CrossRef]
- Saberian, M.; Li, J.; Donnoli, A.; Bonderenko, E.; Oliva, P.; Gill, B.; Lockrey, S.; Siddique, R. Recycling of spent coffee grounds in construction materials: A review. J. Clean. Prod. 2021, 289, 125837. [Google Scholar] [CrossRef]
- dos Santos, É.M.; de Macedo, L.M.; Tundisi, L.L.; Ataide, J.A.; Camargo, G.A.; Alves, R.C.; Oliveira, M.B.P.P.; Mazzola, P.G. Coffee by-products in topical formulations: A review. Trends Food Sci. Technol. 2021, 111, 280–291. [Google Scholar] [CrossRef]
- Kovalcik, A.; Obruca, S.; Marova, I. Valorization of spent coffee grounds: A review. Food Bioprod. Process. 2018, 110, 104–119. [Google Scholar] [CrossRef]
- Saratale, G.D.; Bhosale, R.; Shobana, S.; Banu, J.R.; Pugazhendhi, A.; Mahmoud, E.; Bhatia, S.K.; Atabani, A.E.; Mulone, V.; Yoon, J.-J.; et al. A review on valorization of spent coffee grounds (SCG) towards biopolymers and biocatalysts production. Bioresour. Technol. 2020, 314, 123800. [Google Scholar] [CrossRef] [PubMed]
- Hejna, A. Potential applications of by-products from the coffee industry in polymer technology–Current state and perspectives. Waste Manag. 2021, 121, 296–330. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, G.; Passos, C.P.; Ferreira, P.; Coimbra, M.A.; Gonçalves, I. Coffee By-Products and Their Suitability for Developing Active Food Packaging Materials. Foods 2021, 10, 683. [Google Scholar] [CrossRef]
- Sisti, L.; Celli, A.; Totaro, G.; Cinelli, P.; Signori, F.; Lazzeri, A.; Bikaki, M.; Corvini, P.; Ferri, M.; Tassoni, A.; et al. Monomers, materials and energy from coffee by-products: A review. Sustainability 2021, 13, 6921. [Google Scholar] [CrossRef]
- Karmee, S.K. A spent coffee grounds based biorefinery for the production of biofuels, biopolymers, antioxidants and biocomposites. Waste Manag. 2018, 72, 240–254. [Google Scholar] [CrossRef]
- Mata, T.M.; Martins, A.A.; Caetano, N.S. Bio-refinery approach for spent coffee grounds valorization. Bioresour. Technol. 2018, 247, 1077–1084. [Google Scholar] [CrossRef]
- Battista, F.; Zuliani, L.; Rizzioli, F.; Fusco, S.; Bolzonella, D. Biodiesel, biogas and fermentable sugars production from Spent coffee Grounds: A cascade biorefinery approach. Bioresour. Technol. 2021, 342, 125952. [Google Scholar] [CrossRef]
- Araujo, M.N.; dos Santos, K.C.; do Carmo Diniz, N.; de Carvalho, J.C.; Corazza, M.L. A biorefinery approach for spent coffee grounds valorization using pressurized fluid extraction to produce oil and bioproducts: A systematic review. Bioresour. Technol. Rep. 2022, 18, 101013. [Google Scholar] [CrossRef]
- Atabani, A.E.; Ali, I.; Naqvi, S.R.; Badruddin, I.A.; Aslam, M.; Mahmoud, E.; Almomani, F.; Juchelková, D.; Atelge, M.R.; Khan, T.M. A state-of-the-art review on spent coffee ground (SCG) pyrolysis for future biorefinery. Chemosphere 2022, 286, 131730. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.J.; Lee, Y.G.; Song, Y.; Nguyen, D.-T.; Bae, H.-J. An integrated process for conversion of spent coffee grounds into value-added materials. Bioresour. Technol. 2022, 346, 126618. [Google Scholar] [CrossRef]
- Zabaniotou, A.; Kamaterou, P. Food waste valorization advocating Circular Bioeconomy-A critical review of potentialities and perspectives of spent coffee grounds biorefinery. J. Clean. Prod. 2019, 211, 1553–1566. [Google Scholar] [CrossRef]
- Bhaturiwala Rizwan, A.; Modi Hasmukh, A. Extraction of oligosaccharides and phenolic compounds by roasting pretreatment and enzymatic hydrolysis from spent coffee ground. J. Appl. Biol. 2020, 8, 75–81. [Google Scholar] [CrossRef]
- Zhang, S.; Yang, J.; Wang, S.; Rupasinghe, H.P.; He, Q. Experimental exploration of processes for deriving multiple products from spent coffee grounds. Food Bioprod. Process. 2021, 128, 21–29. [Google Scholar] [CrossRef]
- Wongsiridetchai, C.; Jonjaroen, V.; Sawangwan, T.; Charoenrat, T.; Chantorn, S. Evaluation of prebiotic mannooligosaccharides obtained from spent coffee grounds for nutraceutical application. LWT 2021, 148, 111717. [Google Scholar] [CrossRef]
- Panusa, A.; Zuorro, A.; Lavecchia, R.; Marrosu, G.; Petrucci, R. Recovery of Natural Antioxidants from Spent Coffee Grounds. J. Agric. Food Chem. 2013, 61, 4162–4168. [Google Scholar] [CrossRef]
- Severini, C.; Derossi, A.; Fiore, A.G. Ultrasound-assisted extraction to improve the recovery of phenols and antioxidants from spent espresso coffee ground: A study by response surface methodology and desirability approach. Eur. Food Res. Technol. 2016, 243, 835–847. [Google Scholar] [CrossRef]
- Abrahão, F.R.; Rocha, L.C.; Santos, T.A.; do Carmo, E.L.; Pereira, L.A.; Borges, S.V.; Pereira, R.G.F.A.; Botrel, D.A. Microencapsulation of bioactive compounds from espresso spent coffee by spray drying. LWT 2019, 103, 116–124. [Google Scholar] [CrossRef]
- Mayanga-Torres, P.C.; Lachos-Perez, D.; Rezende, C.A.; Prado, J.M.; Ma, Z.; Tompsett, G.T.; Timko, M.T.; Forster-Carneiro, T. Valorization of coffee industry residues by subcritical water hydrolysis: Recovery of sugars and phenolic compounds. J. Supercrit. Fluids 2019, 120, 75–85. [Google Scholar] [CrossRef]
- Shang, Y.-F.; Xu, J.-L.; Lee, W.-J.; Um, B.-H. Antioxidative polyphenolics obtained from spent coffee grounds by pressurized liquid extraction. S. Afr. J. Bot. 2017, 109, 75–80. [Google Scholar] [CrossRef]
- Torres-Valenzuela, L.S.; Ballesteros-Gómez, A.; Sanin, A.; Rubio, S. Valorization of spent coffee grounds by supramolecular solvent extraction. Sep. Purif. Technol. 2019, 228, 115759. [Google Scholar] [CrossRef]
- Pettinato, M.; Casazza, A.A.; Perego, P. The role of heating step in microwave-assisted extraction of polyphenols from spent coffee grounds. Food Bioprod. Process. 2019, 114, 227–234. [Google Scholar] [CrossRef]
- Ramón-Gonçalves, M.; Gómez-Mejía, E.; Rosales-Conrado, N.; León-González, M.E.; Madrid, Y. Extraction, identification and quantification of polyphenols from spent coffee grounds by chromatographic methods and chemometric analyses. Waste Manag. 2019, 96, 15–24. [Google Scholar] [CrossRef]
- Balzano, M.; Loizzo, M.R.; Tundis, R.; Lucci, P.; Nunez, O.; Fiorini, D.; Giardinieri, A.N.; Frega, G.; Pacetti, D. Spent espresso coffee grounds as a source of anti-proliferative and antioxidant compounds. Innov. Food Sci. Emerg. Technol. 2020, 59, 102254. [Google Scholar] [CrossRef]
- Arauzo, P.J.; Lucian, M.; Du, L.; Olszewski, M.P.; Fiori, L.; Kruse, A. Improving the recovery of phenolic compounds from spent coffee grounds by using hydrothermal delignification coupled with ultrasound assisted extraction. Biomass Bioenergy 2020, 139, 105616. [Google Scholar] [CrossRef]
- Bitencourt, R.G.; Mello, F.M.; Cabral, F.A.; Meirelles, A.J. High-pressure fractionation of spent coffee grounds oil using green solvents. J. Supercrit. Fluids 2020, 157, 104689. [Google Scholar] [CrossRef]
- Zengin, G.; Sinan, K.I.; Mahomoodally, M.F.; Angeloni, S.; Mustafa, A.M.; Vittori, S.; Maggi, F.; Caprioli, G. Chemical composition, antioxidant and enzyme inhibitory properties of different extracts obtained from spent coffee ground and coffee silverskin. Foods 2020, 9, 713. [Google Scholar] [CrossRef]
- Díaz-Hernández, G.C.; Alvarez-Fitz, P.; Maldonado-Astudillo, Y.I.; Jiménez-Hernández, J.; Parra-Rojas, I.; Flores-Alfaro, E.; Salazar, R. Ramírez, M. Antibacterial, Antiradical and Antiproliferative Potential of Green, Roasted, and Spent Coffee Extracts. Appl. Sci. 2022, 12, 1938. [Google Scholar] [CrossRef]
- Rathnakumar, K.; Osorio-Arias, J.C.; Krishnan, P.; Martínez-Monteagudo, S.I. Fractionation of spent coffee ground with tertiary amine extraction. Sep. Purif. Technol. 2021, 274, 119111. [Google Scholar] [CrossRef]
- Valdés, A.; Castro-Puyana, M.; Marina, M.L. Isolation of proteins from spent coffee grounds. Polyphenol removal and peptide identification in the protein hydrolysates by RP-HPLC-ESI-Q-TOF. Food Res. Int. 2020, 137, 109368. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, K.; Pineda-Hidalgo, K.V.; Rochín-Medina, J.J. Fermentation of spent coffee grounds by Bacillus clausii induces release of potentially bioactive peptides. LWT 2021, 138, 110685. [Google Scholar] [CrossRef]
- Kim, J.-H.; Ahn, D.; Eun, J.; Moon, S. Antioxidant effect of extracts from the coffee residue in raw and cooked meat. Antioxidants 2016, 5, 21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sampaio, A.; Dragone, G.; Vilanova, M.; Oliveira, J.M.; Teixeira, J.A.; Mussatto, S.I. Production, chemical characterization, and sensory profile of a novel spirit elaborated from spent coffee ground. LWT 2013, 54, 557–563. [Google Scholar] [CrossRef] [Green Version]
- Martinez-Saez, N.; García, A.T.; Pérez, I.D.; Rebollo-Hernanz, M.; Mesías, M.; Morales, F.J.; Martín-Cabrejas, M.A.; del Castillo, M.D. Use of spent coffee grounds as food ingredient in bakery products. Food Chem. 2017, 216, 114–122. [Google Scholar] [CrossRef]
- Sharma, A.; Ray, A.; Singhal, R.S. A biorefinery approach towards valorization of spent coffee ground: Extraction of the oil by supercritical carbon dioxide and utilizing the defatted spent in formulating functional cookies. Future Foods 2021, 4, 100090. [Google Scholar] [CrossRef]
- Hussein, A.; Ali, H.; Bareh, G.; Farouk, A. Influence of spent coffee ground as fiber source on chemical, rheological and sensory properties of sponge cake. Pak. J. Biol. Sci. 2013, 22, 273–282. [Google Scholar] [CrossRef] [Green Version]
- Meerasri, J.; Sothornvit, R. Novel development of coffee oil extracted from spent coffee grounds as a butter substitute in bakery products. J. Food Process. Preserv. 2022, e16687. [Google Scholar] [CrossRef]
- Zhang, M.; Biesold, G.M.; Choi, W.; Yu, J.; Deng, Y.; Silvestre, C.; Lin, Z. Recent advances in polymers and polymer composites for food packaging. Mater. Today 2022, 53, 134–161. [Google Scholar] [CrossRef]
- Han, J.-W.; Ruiz-Garcia, L.; Qian, J.-P.; Yang, X.-T. Food Packaging: A Comprehensive Review and Future Trends. Compr. Rev. Food Sci. 2018, 17, 860–877. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Batista, M.J.; Ávila, A.F.; Franca, A.S.; Oliveira, L.S. Polysaccharide-rich fraction of spent coffee grounds as promising biomaterial for films fabrication. Carbohydr. Polym. 2020, 233, 115851. [Google Scholar] [CrossRef] [PubMed]
- Coelho, G.O.; Batista, M.J.; Ávila, A.F.; Franca, A.S.; Oliveira, L.S. Development and characterization of biopolymeric films of galactomannans recovered from spent coffee grounds. J. Food Eng. 2021, 289, 110083. [Google Scholar] [CrossRef]
- Wu, H.; Hu, W.; Zhang, Y.; Huang, L.; Zhang, J.; Tan, S.; Cai, X.; Liao, X. Effect of oil extraction on properties of spent coffee ground–plastic composites. J. Mater. Sci. 2016, 51, 10205–10214. [Google Scholar] [CrossRef]
- Moustafa, H.; Guizani, C.; Dupont, C.; Martin, V.; Jeguirim, M.; Dufresne, A. Utilization of Torrefied Coffee Grounds as Reinforcing Agent To Produce High-Quality Biodegradable PBAT Composites for Food Packaging Applications. ACS Sustain. Chem. Eng. 2017, 5, 1906–1916. [Google Scholar] [CrossRef]
- Tarazona, E.R.; Oliveira, L.S.; Rubio, J.C.; Franca, A.S. Preparation, preliminary characterization and mechanical properties of epoxy composites reinforced with spent coffee grounds. In Proceedings of the 8th International Conference on Mechanical and Aerospace Engineering (ICMAE), Prague, Czech Republic, 22–25 July 2017. [Google Scholar] [CrossRef]
- Thiagamani, S.M.; Nagarajan, R.; Jawaid, M.; Anumakonda, V.; Siengchin, S. Utilization of chemically treated municipal solid waste (spent coffee bean powder) as reinforcement in cellulose matrix for packaging applications. Waste Manag. 2017, 69, 445–454. [Google Scholar] [CrossRef] [PubMed]
- Essabir, H.; Raji, M.; Laaziz, S.A.; Rodrique, D.; Bouhfid, R.; el kacem Qaiss, A. Thermo-mechanical performances of polypropylene biocomposites based on untreated, treated and compatibilized spent coffee grounds. Compos. B Eng. 2018, 149, 1–11. [Google Scholar] [CrossRef]
- Senthil Muthu Kumar, T.; Yorseng, K.; Rajini, N.; Siengchin, S.; Ayrilmis, N.; Varada Rajulu, A. Mechanical and thermal properties of spent coffee bean filler/poly(3-hydroxybutyrate-co-3-hydroxyvalerate) biocomposites: Effect of recycling. Process. Saf. Environ. Prot. 2019, 124, 187–195. [Google Scholar] [CrossRef]
- Gazonato, E.C.; Maia, A.A.; da Silva Moris, V.A.; de Paiva, J.M. Thermomechanical Properties of Corn Starch Based Film Reinforced with Coffee Ground Waste as Renewable Resource. Mater. Res. 2019, 22. [Google Scholar] [CrossRef]
- Terroba-Delicado, E.; Fiori, S.; Gomez-Caturla, J.; Montanes, N.; Sanchez-Nacher, L.; Torres-Giner, S. Valorization of Liquor Waste Derived Spent Coffee Grains for the Development of Injection-Molded Polylactide Pieces of Interest as Disposable Food Packaging and Serving Materials. Foods 2022, 11, 1162. [Google Scholar] [CrossRef]
- Gupta, A.; Mohanty, A.K.; Misra, M. Biocarbon from spent coffee ground and their sustainable biocomposites with recycled water bottle and bale wrap: A new life for waste plastics and waste food residues for industrial uses. Compos. Part A Appl. Sci. Manuf. 2022, 154, 106759. [Google Scholar] [CrossRef]
- da Silva, A.P.; de Paula Pereira, M.; Passador, F.R.; Montagna, L.S. PLA/coffee grounds composites: A study of photodegradation and biodegradation in soil. In Macromolecular Symposia; Wiley-VCH: Weinheim, Germany, 2020; Volume 394, p. 2000091. [Google Scholar] [CrossRef]
- Ounkaew, A.; Kasemsiri, P.; Kamwilaisak, K.; Saengprachatanarug, K.; Mongkolthanaruk, W.; Souvanh, M.; Pongsa, U.; Chindaprasirt, P. Polyvinyl alcohol (pva)/starch bioactive packaging film enriched with antioxidants from spent coffee ground and citric acid. J. Polym. Environ. 2018, 26, 3762–3772. [Google Scholar] [CrossRef]
- Cacciotti, I.; Mori, S.; Cherubini, V.; Nanni, F. Eco-sustainable systems based on poly(lactic acid), diatomite and coffee grounds extract for food packaging. Int. J. Biol. Macromol. 2018, 112, 567–575. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Getachew, A.T.; Ahmad, R.; Park, J.-S.; Chun, B.-S. Fish skin gelatin based packaging films functionalized by subcritical water extract from spent coffee ground. Food Packag. Shelf Life 2021, 29, 100735. [Google Scholar] [CrossRef]
- Papadaki, A.; Kachrimanidou, V.; Lappa, I.K.; Andriotis, H.; Eriotou, E.; Mandala, I.; Kopsahelis, N. Tuning the physical and functional properties of whey protein edible films: Effect of pH and inclusion of antioxidants from spent coffee grounds. Sustain. Chem. Pharm. 2022, 27, 100700. [Google Scholar] [CrossRef]
- Bomfim, A.; Oliveira, D.; Voorwald, H.; Benini, K.; Dumont, M.-J.; Rodrigue, D. Valorization of Spent Coffee Grounds as Precursors for Biopolymers and Composite Production. Polymers 2022, 14, 437. [Google Scholar] [CrossRef] [PubMed]
| Green Arabica and Robusta | Roasted Arabica | Roasted Robusta | Spent Coffee Grounds | |
|---|---|---|---|---|
| Protein | 13–17 | 12–15 | 13–15 | 10–17 |
| Lipids | 9–15 | 15–20 | 11–16 | 22–27 |
| Minerals | 4–5 | 5 | 0.1–1 | |
| Carbohydrate | 40–79 | 64–71 | 45–89 | |
| Caffeine | ~1 | ~2 | 0.07–0.4 | |
| TDF | - | 45–51 | ||
| Cellulose | 6.7–8.7 | - | - | 16–25 |
| Galactomannans | 25 | 12–13 | - | ~23 |
| Arabinogalactans | 17 | - | - | ~11 |
| Protein | 13–17 | 12–15 | 13–15 | 10–17 |
| Lipids | 9–15 | 15–20 | 11–16 | 22–27 |
| Minerals | 4–5 | 5 | 0.1–1 |
| SCG Treatment | Polymer/Treatment | Main Effects of SCG Addition | Ref. |
|---|---|---|---|
| Removal of coffee oil by hexane extraction by ultrasonication | Polypropylene/extrusion | Improvement in mechanical and thermal properties | [129] |
| Torrefaction | poly(butylene adipate-coterephthalate)/extrusion | Enhancement in the thermo-mechanical properties and increased hydrophobicity | [130] |
| Alkali treatment | Epoxidized soybean oil/heating and curing | Significant improvement in mechanical properties | [131] |
| Alkali treatment | Cellulose/casting | Increase in tensile strength and thermal stability | [132] |
| Alkali treatment, bleaching and mixture with coupling agent | Polypropylene/extrusion | Improvement in mechanical properties | [133] |
| none | Poly(3-hydroxybutyrate-co-3-hydroxyvalerate)/casting | Decrease in tensile strength, slight increase in elongation | [134] |
| none | Pectin/continuous casting | Increase in water permeability/improvement in thermal properties | [35] |
| none | Starch/microwave heating | Increase in tensile strength/no effect on thermal properties | [135] |
| Milling (SCG from ethanolic extraction) | Polylactide + lactic acid oligomers/extrusion | Increase in ductility | [136] |
| Slow pyrolysis | Polyethylene terephthalate + linear low-density polyethylene/extrusion | improvements in the flexural modulus and thermal properties | [137] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franca, A.S.; Oliveira, L.S. Potential Uses of Spent Coffee Grounds in the Food Industry. Foods 2022, 11, 2064. https://doi.org/10.3390/foods11142064
Franca AS, Oliveira LS. Potential Uses of Spent Coffee Grounds in the Food Industry. Foods. 2022; 11(14):2064. https://doi.org/10.3390/foods11142064
Chicago/Turabian StyleFranca, Adriana S., and Leandro S. Oliveira. 2022. "Potential Uses of Spent Coffee Grounds in the Food Industry" Foods 11, no. 14: 2064. https://doi.org/10.3390/foods11142064
APA StyleFranca, A. S., & Oliveira, L. S. (2022). Potential Uses of Spent Coffee Grounds in the Food Industry. Foods, 11(14), 2064. https://doi.org/10.3390/foods11142064







