Abstract
Algae have been consumed for millennia in several parts of the world as food, food supplements, and additives, due to their unique organoleptic properties and nutritional and health benefits. Algae are sustainable sources of proteins, minerals, and fiber, with well-balanced essential amino acids, pigments, and fatty acids, among other relevant metabolites for human nutrition. This review covers the historical consumption of algae in Europe, developments in the current European market, challenges when introducing new species to the market, bottlenecks in production technology, consumer acceptance, and legislation. The current algae species that are consumed and commercialized in Europe were investigated, according to their status under the European Union (EU) Novel Food legislation, along with the market perspectives in terms of the current research and development initiatives, while evaluating the interest and potential in the European market. The regular consumption of more than 150 algae species was identified, of which only 20% are approved under the EU Novel Food legislation, which demonstrates that the current legislation is not broad enough and requires an urgent update. Finally, the potential of the European algae market growth was indicated by the analysis of the trends in research, technological advances, and market initiatives to promote algae commercialization and consumption.
Keywords:
algae market; novel food; microalgae; seaweed; macroalgae; EU novel food legislation; food ‡ These authors contributed equally to this work.
1. Introduction
In Europe, animals are today’s major source of protein to address dietary needs in response to the rapid growth of the world’s population. However, concerns regarding the environmental impact of the current food production systems, together with health and animal welfare issues, have driven the need to develop healthier and more sustainable food sources [1].
Algae—microalgae (including cyanobacteria) and seaweed (also known as macroalgae and sea vegetables)—have been used as food since medieval times, with a consolidated market in Asia and a growing market in Europe, being driven by a rising awareness in consumers regarding sustainable and healthy foods [2,3,4,5]. In addition, algae have a wide application potential beyond merely a food source, such as animal feed, biofertilizers, bioremediation, or the extraction of added-value biochemical compounds [6]. The global market of algae was valued at EUR 594 million (2018) and is projected to reach EUR 1131 million by 2027 [7]. Notably, by 2050, food production has to increase by at least 60% to meet the demands of the growing world population (around 9 billion), according to the Food and Agriculture Organization (FAO) [8]. Algae can be part of the solution by providing a good alternative to traditional crops as they do not require arable land and are able to grow on minimal nutrients [9].
In 2009, the European Algae Biomass Association (EABA) was founded to promote the algae industry by establishing synergies between academia, industry, and decision-makers in the field [10]. After that, regional associations started to be created, such as the Fédération des Spiruliniers de France and PROALGA, the Associação Portuguesa de Produtores de Algas. Currently, around 420 companies, distributed over 23 countries, are producing microalgae and seaweed in Europe, 46% of which produce Spirulina, 36% seaweed, and 10% microalgae. The remaining 8% produce both Spirulina and microalgae [11,12,13].
Algae can be consumed as food or as ingredients in prepared foods, in a fresh, fermented, dried, or frozen format, either whole or milled into differently sized flakes, granules, or powders [14,15]. Algae foods are receiving more attention since they are a natural source of micro- and macronutrients, as well as trace elements, increasing their nutritional and pharmacological value [16,17,18]. Product launches of traditional foods with algae ingredients, such as cookies, pasta, bread, and beverages, among others, are increasing in the European market, holding a 1.34% share of the new European foods and drinks launched in 2017 [19,20,21,22].
Moreover, algae are a sustainable source of natural high-value bioactive compounds, with the potential to manufacture new products for human nutrition. Lipids, in the form of polyunsaturated fatty acids (PUFA), ω-3 and ω-6 PUFA, and phytosterols are major compounds in algae that can be applied in several contexts, due to their beneficial properties for the cardiovascular system, anti-cholesterol activity, and others [23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38]. Algal ω-3 PUFA, such as docosahexaenoic (DHA) and eicosapentaenoic (EPA) acids, are also earning market relevance, replacing the traditional intake of these compounds via fish consumption [39]. Proteinogenic amino acids and peptides from both microalgae and seaweeds can be applied as antioxidants, antihypertensives, and anticoagulants, and as antiproliferative and immunostimulant agents [2,35,37,38,40,41,42,43,44,45,46,47]. Polysaccharides, whose content in seaweed species ranges from 4 to 76% of their total dry weight, can also be consumed as they display numerous health-promoting properties, such as antitumoral, antiviral, and antioxidant activities [48,49,50,51,52,53,54]. Their derivates (phycocolloids: alginate, carrageenans, and agar) are also widely used as thickening, emulsifying, stabilizing, and gelling agents in the drink and food industries [55,56,57,58,59,60]. Natural pigments, such as chlorophylls, phycobiliproteins, and carotenoids, are among the most sought-after, high-value metabolites produced by algae. Carotenoids such as lutein, β-carotene, lycopene, and astaxanthin are mainly used as dietary supplements, food fortifiers, and beverage natural colorants, as the new generation of consumers chooses natural over synthetic products (particularly in response to allergic reactions and health concerns) [18,61,62,63,64,65]. Therefore, algae biomass and its extracts are used as dietary supplements and food additives, such as flavor enhancers, color additives, preservatives, emulsifiers, and antioxidants, among others.
Each category of algae-based products is regulated by separate legislation. For food and food supplements, novel food regulation (EU 2017/2470) is covered in the Novel Food Catalog, a list of authorized novel foods that are safe for human consumption [66].
This review paper addresses the applications of algae (microalgae and seaweed) as a food in Europe, with an overview of the consumption habits, market, and legislation. Approved and consumed algae species are listed and the challenges of incorporating them into the European market are evaluated, together with the analysis of research and development initiatives.
2. Algae as Food, Not as a Novelty
2.1. Historical Perspective
Algae have been included historically in the human diet worldwide. According to archaeological findings in Monte Verde (Chile), people were collecting seaweeds for food and medicine 14,000 years ago [3]. Seaweeds have been used for centuries in Asian cuisine for their nutritional properties and unique flavors, particularly in Korea and Japan. In China, seaweed consumption has been recorded since 500 BCE [5]. In Chile and northeast Atlantic countries, there are historical records of seaweed consumption [3,67,68]. People living on islands, where conventional agriculture is difficult to adopt, have relied on the sea as their main source of food, making seaweed an important element in their diet [69]. Nowadays, seaweeds are seen as major coastal resources, valuable to human consumption and good for the environment, and are consumed globally [35,70,71].
In Europe, seaweeds are likely to have been harvested for human consumption for centuries, although they are a vastly underexploited resource when compared with consumption in Asia [58,72]. Archaeological evidence is scarce, due to seaweed’s easily degradable nature; however, their use can be traced back to the era of Hippocrates, when burned seaweeds were consumed as dietary supplements for medicinal purposes [14,73,74,75,76]. Seaweeds have played an important part in the livelihoods of coastal-dwelling peoples, in countries such as Portugal, Spain, France, the UK, Ireland, Denmark, Norway, and Iceland, being gathered by hand without any mechanization or equipment [5,68,74,76,77,78,79,80,81,82,83,84]. Seaweeds were included in the diet and were also collected to fertilize agricultural fields and supplement cattle feed, playing an indirect but important role in the production of food for centuries as storm-cast mineral-rich seaweed [58,60,76,78,82,83,85,86,87]. Apart from the current increase in its popularity as a gourmet food and ingredient, historically, seaweed has been vital to ensure survival in times of starvation and poverty [14,76,86,88]. In England, Ireland, and Scotland, Palmaria palmata (Dulse, “Creathnach” or “Dillisk”, in Gaelic) was traditionally eaten raw, toasted with hot irons, cooked in a thick oatmeal broth, served boiled, tossed in butter, or fried as crisped seaweed “bacon” for sandwiches; later, it was used for chewing tobacco, as well as ingested for deworming and to cure “women’s longing” [14,72,77,89,90,91,92]. Vikings and the Icelandic Inuits gathered seaweed to eat raw or to be dried in old fish barrels [68,76,78,79,80,86,87,91]. Other traditionally harvested seaweeds include the Atlantic species of Porphyra sp. (Atlantic Nori or Purple Laver), baked into different dishes, such as the popular Welsh laverbread (“bara lawr”) from Wales, or fried as an ingredient in omelets or pies (“Tortas de Erva do Calhau”), in the Azores Islands (Portugal) [58,83,84,85,93,94]. In the Mediterranean, Ulva lactuca (sea lettuce) and Chondracanthus teedei (“Mauru” in Italian) were traditionally eaten fresh by fishermen and beachgoers, with a squeeze of lemon and salt, in coastal regions such as Catania (Sicily) [95,96,97]. These species are still consumed, dried as a snack, mixed into salads, bread dough, curds, or consumed with fish in local restaurants [5,21].
In Western countries, seaweeds have been mainly associated with industrial applications focused on the extraction of phycocolloids for the food industry [70,98]. In the first half of the 20th century, the main commercialized species were: wracks and kelps used for alginate extraction (Ascophyllum nodosum, known as Rockweed, in Norway; Laminaria digitata, or Oarweed, in France and Ireland; Laminaria hyperborea, or Tangle, in Britain, Ireland, and Norway); harvested Irish Moss in coastal areas (Chondrus crispus and Mastocarpus stellatus, particularly in France, Brittany, and Normandy, as well as western Ireland) and black carrageenan or “Danish Agar” (Furcellaria lumbricalis, in Denmark) for carrageenan and furcellaran extraction [58,82,86,88,99]. Moreover, during this period, George Ohsawa, a Japanese philosopher, popularized the macrobiotic lifestyle movement in Europe, which introduced the notion of a regular intake of sea vegetables in the diet, mainly Nori (Porphyra sp.), Wakame (Undaria pinnatifida), Kombu (Laminaria sp.), Agar, Arame (Eisenia bicyclis) and Hijiki (Hizikia fusiforme), which are still largely imported from Asia to this day [100,101,102]. In fact, macroalgae are an essential part of the food pyramid in the macrobiotic dietary guidelines, and people are encouraged to consume them as locally as possible [100,101]. By the mid-20th century, the need for bacteriology and penicillin production from agar, during World War II, spiked an urgent search for alternative agar sources to the Japanese and US pre-war imports [58,60]. The phycocolloid industry followed in Britain (Chondrus sp. and Mastocarpus sp.), in mainland Portugal, Spain, and France (Gelidium corneum, previously known as Gelidium sesquipedale), the Azorean Islands (Pterocladiella capillacea) and Italy (Gracilariopsis longissima (or Gracilaria verrucosa) [58,60,103,104,105,106,107,108,109,110]. Toward the end of the 20th century, harvesting techniques were mechanized in France and Norway, in order to meet the increasing industrial demand and to improve access to fresh macroalgae, beyond storm-cast supplies [111,112]. Furthermore, the harvest of Gelidium corneum and Pterocladiella capillacea peaked in 1980, with Portugal leading as the world’s fifth-largest agar producer and European macroalgae contributing to 34% of the world’s alginate supply [58,81,110,113].
For microalgae, the oldest documented use as food was 1500 years ago in China [16,114]. Nostoc species are traditional foods in China (called “Fa cai”), Mongolia, and South America (called “Cushuru”) [115,116,117]. Arthrospira (also known as “Spirulina”) was historically harvested by the Aztecs living near Lake Texcoco, as recorded in the 14th century as an ingredient for a dry cake called “Tecuitlatl” [5,118]. In Africa, at the same time or even earlier (since the 9th century), the local population in Chad was also harvesting Spirulina, locally known as “Dihé”, from Lake Kossorom; Spirulina was sundried for use in dishes such as meat and vegetable broths and sold in local markets or to wholesalers [115,119,120]. These discoveries were the premise for a series of studies that followed from the end of the 1960s onward, which would boost global interest in Spirulina in terms of mass production. In fact, as it started to gain a promising reputation for potential food applications, in 1967, Spirulina was branded as a “wonderful future food source” by the International Association of Applied Microbiology and was later acknowledged as “the best food for the future” by the United Nations (UN) World Food Conference of 1974 [120,121]. The commercial exploitation of microalgae is only a few decades old. After World War II, the focus was set on a possible insufficient protein supply to respond to the rapid increase of the world population [16]. Since then, algae have been considered a good source of protein with a well-balanced profile of essential amino acids [40]. Recently, there is an increased interest in algae due to their unique bioactive metabolites, giving them great potential as a source of food and functional molecules, to be used as supplements, functional ingredients, or nutraceuticals [43,122,123,124].
Microalgae have only recently been consumed in Europe. From the historical perspective, it is important to highlight that although microalgae were under active research for many decades, especially for the human diet, the product of this research has failed to materialize commercially in the EU space, at least until the publication of Regulation (EC) No. 258/97 of the European Parliament and of the Council of 27 January 1997, concerning novel foods and novel food ingredients [6,22,125]. Unlike in EU countries, in global markets, such as the US or some countries in Asia, commercial development around Spirulina has drastically increased since the 1980s, not only as a food supplement in the form of capsules, tablets, or powder but also as a food ingredient for the formulation of novel healthy foods [6,81].
The historical role of microalgae [6,111,112,126,127,128,129,130,131,132,133,134,135,136,137,138,139,140,141,142,143,144,145,146,147,148,149,150] and seaweed [14,58,59,60,68,72,73,74,75,76,77,78,79,80,81,82,85,86,87,88,91,93,99,100,101,102,103,104,106,107,108,109,110,113,151,152,153,154,155,156] for consumption in food, supplements and the phycocolloid industry is represented in Figure 1.
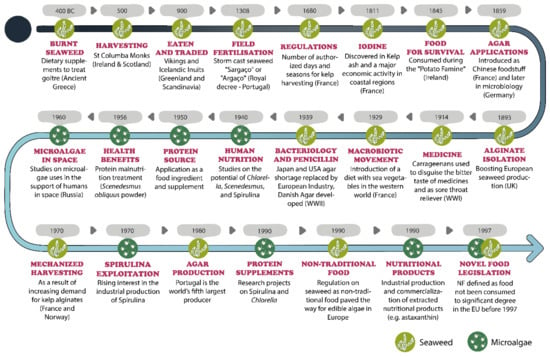
Figure 1.
European historical marks on algae consumption as a food and food supplements (image not scaled) [6,14,58,59,60,68,72,73,74,75,76,77,78,79,80,81,82,85,86,87,88,91,93,99,100,101,102,103,104,106,107,108,109,111,112,126,127,128,129,130,131,132,133,134,135,136,137,138,139,140,141,142,143,144,145,146,147,148,149,150,151,152,153,154,155,156].
2.2. Current EU Market Data
The European algae industry, with the production of around 500 t algae dry weight per year, remains a niche market, mainly directed to food and food-related applications and concentrated on the production of large quantities of biomass using a limited number of species [10,12]. Many decades have passed since the beginning of the exploitation of algae as a sustainable source of protein; however, they are yet to gain greater worldwide prominence in the fight against the lack of food and in the consumption habits of the Western population. Nevertheless, commercial and industrial interest has been growing remarkably in recent years [121,157,158]. Consequently, food and beverage markets are evolving rapidly to meet this increasing demand, presenting end-consumers with novel eco-friendly organic food products, adjusted to new diets as well as food consumption and purchasing habits [22]. New cooking habits and the formulation of functional foods and the associated organoleptic characteristics increased the importance of algae as a food ingredient [76,157]. In fact, in line with this growing trend, over the past few years, there has been a greater share of new launches of algae-based products for the food and drink markets in Europe [22]. Furthermore, this category of food products has been achieving increasing commercial representation in retail sales, particularly in Western Europe, where retail sales of fungi/algae meat substitutes were valued at EUR 223.65 million in 2015 [159]. More recently, in 2018, algae meat substitute products, such as algae burgers and grills, accounted for EUR 1.82 million in Europe [160].
Despite the promising figures, the high cost of production, the complexity of European regulations concerning cultivation licenses, and the synchronization of guidelines for organic certification between countries limit the algae market in Europe [11,21,81,82,161,162]. Additionally, the lack of effective algae production and distribution is perhaps due to algae not being traditionally consumed in Europe, compared to Asian countries. Dried seaweed consumption in Japan is around 2 kg per capita−1 year−1, which may be compared to the consumption of salad in Europe (3 kg per capita−1 year−1) [161]. Data regarding European algae consumption is very limited but is expected to be significantly lower (< 50%) than in Asian countries, where the average daily intake of, for instance, seaweed by adults ranges from 4 to 8.5 g (Japan, China, and South Korea, in ascending order) [163,164]. Interestingly, the recommended maximum daily algae intake in European products (i.e., Tok de Mar, Algaessence) is 5 g of dried biomass [165].
European algae producers face heavy competition from Asian producers, mainly due to higher production costs and product prices, which may be discouraging for the producers. However, Asian producers are frequently criticized for the environmental impact caused by their aquaculture systems in eutrophic waters. As this is viewed negatively by an ever more eco-conscious market, European production might excel in the global market of retailers and consumers who demand higher-quality, sustainable algae-based products [166].
The estimated value of the European algae products market is expected to grow by around 43% between 2016 and 2023 (Figure 2), forecasting a value of EUR 1240 million by 2023 and a worldwide total value of EUR 4810 million [167]. North America and the Asia Pacific will be the regions with the highest market representation [167]. More specifically, projections show that the global algae protein market size will approximately double between 2016 and 2023 and is estimated to be worth around EUR 928 million [167]. These increases in market value could be explained not only by advances in algae technology but also by recent trends showing a higher demand for eco-friendly products among Western industrialized societies, which have the benefit of higher education levels and resources to promote the growth of new market segments. The influence of Asian cuisine and consumers’ choice of more natural, sustainable, plant-based, and local ingredients encourage chefs to create new dishes, along with the publication of cookbooks featuring recipes with these versatile ingredients, part of a movement known as phycogastronomy [14,81,92,168]. For example, in the gourmet luxury gastronomy sector, algae, in particular seaweeds, are nowadays starring in the menus of world-renowned Michelin-acclaimed restaurants and inventive chefs, such as René Redzep from Noma (Denmark), João Rodrigues of Feitoria and Project Matéria, José Avillez of Belcanto, Leonel Pereira of Checkin Faro, Ricardo Costa from The Yeatman (Portugal) and Ángel León of Aponiente (Spain), among many others [169,170,171].
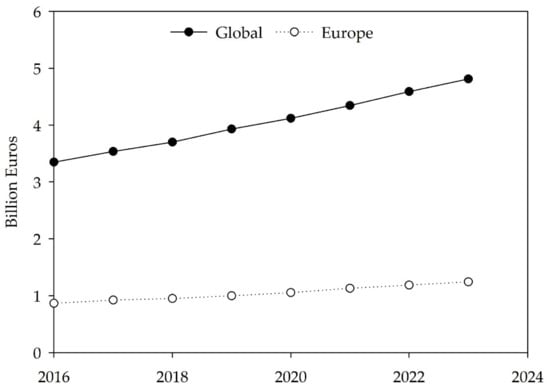
Figure 2.
Global and European algae market trends from 2016 to 2023. Data from Statista (2020) [167].
2.2.1. Microalgae
In Europe, microalgae are commonly produced in photobioreactors in closed and controlled conditions. Germany, France, and Spain host the largest number of producers; however, it is in Portugal that we can find the oldest and the largest microalgae production companies in the EU, Necton and Allmicroalgae, respectively.
One of the key microalgae applications is in the nutraceutical market, in terms of dietary supplementation, where they are seen as valuable natural sources of macro- and micronutrients of great commercial potential [172,173]. In Europe, dietary supplements accounted for EUR 30 billion in 2020, with an expected growth of 50.6% by 2026 [174], the Western market being worth almost three times more than the Eastern one [175]. Moreover, the retail sales forecasts of vitamins and dietary supplements have demonstrated the same continuous commercial trend since the previous decade [176], reaching EUR 182 billion by 2022 [174]. As such, these added-value compounds, extracted from microalgae, are designed to be applied and marketed as ingredients or additives in food products. In that regard, Spain and Italy are the two main European countries driving the microalgae nutraceuticals research field [172]. Currently, depending on their legal status and biochemical profile, a restricted selection of microalgae can be considered nutraceuticals and used for this purpose in the EU, Spirulina sp. and Chlorella sp. being the most popular. Nevertheless, a wide range of algae has the potential to also be considered for this purpose [173]. Carotenoids of commercial value, like α- and β-carotene, astaxanthin, fucoxanthin, or lutein can be obtained from microalgae, such as Dunaliella salina, Haematococcus pluvialis, Chlorella vulgaris, Microchloropsis gaditana (also called Nannochloropsis gaditana), Auxenochlorella protothecoides, Phaeodactylum tricornutum, Tisochrysis lutea and Spirulina sp. [173,177,178]. Furthermore, microalgal fatty acids can be extracted from P. tricornutum, Chlorella sorokiniana or Nannochloropsis oceanica [173,179]. In several European countries, microalgae-based dietary supplements are available on the open market for end-consumers, mostly in capsules; they are also available in powder format. Some examples include astaxanthin, extracted from H. pluvialis, and retinol or β-carotene obtained from D. salina [180,181,182,183,184,185].
Microalgae can be found either as an ingredient in food products or as a whole-cell powder that can be used freely in cooking recipes, smoothies, drinks, or simple snacks [186,187,188,189]. The majority of the microalgae powders are marketed typically as Chlorella and Spirulina [186]. In a 2017 survey, these two algae were identified as being purchased regularly by 2.5–3.8% of the surveyed German population [190]. It is also important to mention that other species have been commercialized in Europe, even though some of them have not as yet been authorized for widespread sale in EU countries. Some examples of such microalgae are Nannochloropsis gaditana and Nannochloropsis oculata, while others, such as Chlorella sorokiniana and Auxenochlorella protothecoides, were being sold without any authorization until recently, when they were approved [47,66,191,192,193]. As for commercial food products, they are formulated with different microalgae, such as Euglena gracilis, Spirulina, and Chlorella, among others, mostly for use in traditional food products such as pasta, ready-to-eat bars, dairy and fermented products, candies, and pâtés [187,188,189].
2.2.2. Seaweed
The European seaweed industry is still in its early development and has been almost exclusively based on wild stock harvesting, unlike the extensive farming that takes place in Asia [11]. However, although small in the global context, the European Atlantic coastline is privileged in the sense that it harbors more than 3000 seaweed species in high-quality waters, capable of sustaining a wide range of industrial demands. According to the FAO’s global production dataset, in 2019, European seaweed production was 96% wild-harvested (275,907 tons, wet weight) and 4% cultivated (11,125 tons, wet weight) [194]. In the same year, Norway was the third-largest worldwide seaweed harvester, while France, Ireland, Iceland, the Faroe Islands, and Portugal were significant in Europe [21,82,195,196,197]. Cultivation is seen as a sustainable solution, as opposed to the overexploitation of wild seaweed resources; this is necessary to meet the increasing demand from the processing industry for traceable, high-quality, and predictable yields of biomass [11]. Additionally, seaweed can be grown in a sustained manner when developed in combination with fed and extractive aquaculture species of different trophic levels in integrated multi-trophic aquaculture (IMTA) systems, currently promoted at both European and national levels [11,21,81,198]. Despite the increase in seaweed aquaculture in Europe, it remains a small portion (0.03%) of the globally produced biomass [194].
The value of European seaweed is roughly estimated as EUR 500–600 million per annum (representing around 8% of the global market size), but it is highly dependent on species, whether wild-harvested or farmed, and the targeted market. Leading markets for seaweed are as plant biostimulants for agriculture and the phycocolloid industry, which is focused on the extraction of single products, such as alginic acid, laminaran, and colorants. Europe is the top food and pharma-grade alginate producer worldwide, with a minor share of global carrageenan and agar production [21,81,82,99,163]. The main commercially exploited species are brown (more than 99%), such as Ascophyllum nodosum, Alaria esculenta, Laminaria hyperborea, L. digitata, Saccharina latissima and Undaria pinnatifida [5,21,81,82,99,195,197].
The European seaweed market is developing at a fast pace, with an annual growth rate of 7–10% and a wholesale value of around EUR 24 million, focusing only on A. esculenta, S. latissima, Porphyra sp., P. palmata, and Ulva sp. [21,199,200]; it is projected to reach EUR 700 to EUR 2100 million by 2030 [201]. The production of selected edible seaweed for culinary uses and ingredients is still a minimal activity, but the market is promising and has been growing, with a huge variety of products such as algae mayonnaise [21,22,202]. The dominant market for edible seaweed is the domestic food market, with supplies sold directly to health-food stores, specialist retailers, and supermarkets, as well as restaurants or factory outlets [200,203].
In 2016, the EU imports of seaweed products were almost twice the size of its exports (178,467 tons vs. 101,594 tons), making the EU the world’s second-largest importer in terms of volume, valued at EUR 506 million [99]. Markets are segmented into four categories: seaweed for human consumption (8.5% of imports; 4.5% of exports); carrageenan (~40% import/exports); agar (~2% import/exports); lastly, seaweed that is not for human consumption (49.6% of imports; 53% of exports), with all data from 2016 [99]. Food products include nori sheets for sushi, other dried seaweeds and wakame mixed salads, with the main suppliers being from Japan, China, and South Korea. In 2018, import values were estimated at EUR 94 million and exports at EUR 42 million [99,204]. Notably, 60% of the consumed algae in Europe is of Pyropia sp. (Nori, imported from Asia). Europe has a nominal supply of Atlantic Nori, producing only 1% of what is consumed. On the other hand, 90% of the P. palmata consumed in Europe is regionally wild-harvested in France. In this case, Europe is becoming more proactively competitive, relying on imports to satisfy 10% of the European consumer demand [204].
Another use of seaweed in food production is its application in packaging biomaterials, such as for edible pouches or pods to replace drinking bottles [205].
3. Challenges of Algae as Food
Despite the historical consumption of algae as a food and their associated nutritional and health benefits, there are still bottlenecks blocking its progress from the current niche markets to larger ones. The main reasons are production constraints, high costs, environmental concerns, health and safety, legality, and consumers’ perception of the product (Table 1).

Table 1.
Challenges to the consumption of algae as food.
3.1. Production Constraints
The growth of algae can be affected not only by abiotic factors (e.g., light, temperature, pH, nutrients and dissolved oxygen content) but also by biological factors (e.g., bacteria, virus, fungi, epiphytes, biofouling, and life cycles) and operational factors (e.g., dilution rate, shear stress, and harvesting methods) [206,207,208,209,210]. At a laboratory scale, these factors are easily controlled, and it is possible to achieve high volumetric and areal production rates [211]. The bigger challenges start with the need to scale up production. Many culture systems have been designed and optimized to control the external and internal factors inherent to production. Examples of culture systems are open ponds, closed photobioreactors (PBRs), and immobilized culture systems [177], and each culture system has its advantages and constraints and must be chosen according to the desired species [206]. A list of the existing production technologies, along with their advantages and disadvantages, is presented in the Supplementary Material (Table S1).
In order to move to a larger-scale operation, an increase in qualified human resources, mechanization, and automation in the operations (such as hatcheries, sea or land cultivation, and harvesting) is required. The scaling-up of photobioreactors from the laboratory to an industrial scale, for example, is pricey not only due to the high material costs but also because of high operational costs, especially if the algae are being cultivated in a different climate from their original habitat. The temperature and light control in this case significantly increases the energy costs of the operation [212].
The appearance of biological contaminants during algae cultivation is also a constraint that can compromise cultures and lead to enormous economic losses. Like most other farming activities, algae cultivation suffers from contamination by parasites, epiphytes, epizootics, competitors, grazers, and predators [208,213,214,215,216,217,218]. Industrially produced algae cultures may share optimal conditions for the growth of these species, which can also lead to a decrease in the biomass quality, or, in the worst-case scenario, a culture collapse.
In the case of microalgae, another constraint arises: due to their microscopic size, the sensitivity of the cell walls for most species, and their low culture concentrations (usually, less than 2 g/L), the downstream process is costly and difficult [211]. There are high process costs associated with the harvesting, dewatering, and drying of the microalgae. According to the literature, the downstream processing of microalgae can account for 40% of the total costs of microalgae production [206].
Additional challenges in seaweed production emerge, for instance, the complexity of their life cycles and domestication constraints for some of the most highly demanded species, such as Dulse (P. palmata) and Atlantic Nori (Porphyra sp.). Successful production and domestication are, in many ways, dependent on the manipulation of different life stages and on monitoring the factors that trigger reproductive events, such as sporulation, which may result in large biomass losses [92,219,220]. Seaweed farming, as in the case of kelp farming, can also be in competition with other sea activities, such as fish farming, fisheries, and tourism [21].
3.2. Food Safety and Health
Algae’s potential for human nutrition is strongly related to their biochemical composition and bioactive properties, which are known to vary widely among the different classes and even among strains. Some highlights include isolated polysaccharides, (e.g., alginate and fucoidan), proteins (phycobiliproteins), polyphenols (e.g., phlorotannins), carotenoids (e.g., fucoxanthin) and n-3 long-chain polyunsaturated fatty acids (e.g., eicosapentaenoic acid) [221]. Over the last few decades, the biochemical composition and functional properties of algae feedstocks have been extensively scrutinized, greatly supported by the improvement and development of new techniques that allowed the high-resolution profiling of proteins, lipids, polysaccharides, pigments, and others [20,222]. Overall, algae are a sustainable source of natural high-value bioactive compounds, with the potential to provide new products for human nutrition.
3.2.1. Toxicity and Allergens
Toxicological factors also need to be considered, as algae can produce or accumulate several contaminants that impact the consumer’s health. In early studies, human experiments with microalgae were scarce, and they were limited to certain countries, like Japan, the US, and Russia [128]. It was only during the 1970s that larger studies started to be promoted and microalgae consumption in small doses was approved, as overall consumption did not cause any metabolic changes [223]. One example is the major research projects promoted by the German government, conducted in India, Peru, and Thailand [146]. However, the number of diet trials conducted directly within European countries remained small [140,141,224]. There were also some discussions concerning the high nucleic acid content of microalgae and the potential problems in terms of human nutrition. Nevertheless, the Protein-Caloric Advisory Group (PAG) of the United Nations determined that the consumption of microalgae would be safe within certain limits [146]. In three studies, microalgae were incorporated into the diets of up to five volunteers, with dosages varying from 100 to 210 g of dry weight per day of biomass, for a maximum of 30 days. No changes were found in the metabolic indices of the subjects when consuming up to 100 g of microalgae; however, at higher dosages, changes in health and alterations to the assimilation of certain micronutrients, such as calcium and magnesium, were apparent [140,141,224].
Natural toxins, such as cyanotoxins, are produced by cyanobacteria and are a common chemical contaminant in large-scale cultures [225]. Cyanotoxins are mostly of cylindrospermopsin, microcystin-LR, -RR, and -LA [226]. In fact, commercially available Chlorella sp., sold as a food supplement, was found to be contaminated with anatoxin [227], a compound produced by cyanobacteria that, in high concentrations, can damage the liver or nervous system and cause human and animal death; therefore, toxicological analysis for product control is required [226]. The cyanobacterium Aphanizomenon sp., which naturally occurs in the Upper Klamath Lake, Oregon, USA, can also produce these toxins [228]; despite the intoxication events related to the consumption of these algae, products from this lake are still commercialized today [227,229]. Although seaweed is not known to be toxic, there are a few exceptions. Desmarestia sp. (Coarse Acid Kelp) is a genus that concentrates sulfuric acid in cell vacuoles and should be avoided since its consumption causes gastrointestinal problems [230]. Another example is the toxic epiphytic dinoflagellate, Coolia monotis, which produces cooliatoxin (a neurotoxin) and was detected in drifting seaweed in New Zealand [231,232].
Algae-related food allergies are uncommon and are poorly documented, with only one clinic case documented, which was associated with Nori [233]. Microalgae from various genera were identified from household dust, including Chlorella sp., and some cyanobacteria were reported to produce a compound responsible for dermatitis and inflammations of the human respiratory system, but this is when the microalgae were in their natural form and not after being processed as a food [234].
Allergens in algae are usually associated with the environment in which they grow. Seaweeds are typically cultivated and are harvested in the open ocean or estuary environments, which are inhabited by crustaceans such as gammarids (amphipods), shrimp, and crabs (Decapoda). Commercial edible crustaceans are frequently involved in immunoglobulin E (IgE)-mediated food allergies [235]. In a study of allergenicity in Nori sheets, tropomyosin from amphipods was identified as the main allergen; however, its presence in each sheet was so low that it is suggested that ingestion would only rarely cause a hypersensitive reaction [235]. Although cultivation and processing practices decrease the risks of contamination, these organisms may still find themselves in seaweed accidentally and may potentially cause allergic reactions in people with high sensitivities to shellfish, behaving as hidden allergens. Therefore, it is common to find mentions of shellfish allergens in the labeling of seaweed, such as in Japan, where the government made it mandatory to have a shrimp label on the allergen details of Nori products [236].
3.2.2. Mineral Contaminants
Although heavy metals are not usually found in algae at values surpassing the legal limits, frequent and excessive algae consumption could pose risks that are associated with the bioaccumulation of these compounds. Heavy metal accumulation depends largely on species, as well as on environmental conditions like light, temperature, pH, salinity, or nitrogen levels [237,238]. This can result in variable metal composition over different commercial products and sometimes in different batches if production is not controlled.
In microalgae, the toxic heavy metals, arsenic, nickel, and lead, were found at very different concentrations among 10 commercial products, although at levels below the safety limits, according to the European Food Safety Authority (EFSA) [227]. In seaweed, the accumulation of heavy metals is a concern, especially for wild-harvested biomass. In the case of arsenic, cadmium, and lead, some brown species may adsorb more than 80% of the soluble metals in water and so need to be monitored closely [239,240,241]. Studies have reported that a few seaweeds, especially Hizikia fusiforme, may have high levels of total and inorganic arsenic [242,243]. In a Danish study, for a weekly single-serving size of 5 g of freeze-dried biomass per adult (Fucus vesiculosus, F. serratus, F. spiralis, F. evanescens, S. latissima, U. lactuca, and Cladophora sp.), there were low-level concerns regarding the presence of mercury, cadmium, and lead [164]. In another study, the impact of an increased seaweed consumption (10% replacement with seaweed products) among Dutch and Portuguese adults revealed no consequences on human health regarding the intake of sodium and exposure to cadmium, lead, and mercury; however, the connection between a higher intake of iodine and arsenic exposure suggests a need for further research [244]. Health assessments of algae consumption are scarce in Europe, but it is urgently required that the authorities should set up regulations and provide accurate recommendations for consumption [164,245].
Iodine is another common concern as it can occur in seaweed at high concentrations. Although it is essential to humans and its deficiency is a major public health challenge, excessive intake can have harmful effects, for instance, thyroid dysfunctions, goiter, and hyperthyroidism [244,246,247]. This is especially true for some kelp species, where iodine concentrations above 45 mg/kg dry weight can be found and can be health-threatening [241,248]. Thus, the identification and control of these compounds, particularly in iodine-rich species (wracks and kelps) are recommended, particularly for high-risk subgroups (pregnant women, children, and individuals with thyroid dysfunction) [164].
Mitigation strategies can be employed to minimize the risk of contamination from algae products. Water quality control is one way to avoid these problems, by minimizing the components that are adsorbed in algae [249]. Hence, choosing the water supply for algae cultivation or at a farming site of naturally occurring algae is crucial [21]. In recent years, an approach using circular culturing systems has been considered, not just as an economic and ecological measure but also as a way of controlling water quality, improving its sustainability, and avoiding the risk of contamination [250].
The specificity and complexity of algae contaminants are sometimes not taken into account by the current food legislation. For instance, the quantification of arsenic could be differentiated as organic and inorganic, as health risks are much lower when consuming the organic form [251]. If these were detailed, seaweed products containing mostly organic arsenic could ensure product safety and build higher customer trust. As algae contamination is product-dependent, frequent chemical analyses, coupled with legal quantification limits, could ensure product safety. These new legislations could open the door to introducing and categorizing algae products to the market and promoting the development of the algae food industry.
3.2.3. Microbiological Contaminants
The processing and manipulation of the algae, such as their handling and packaging, can also be a hotspot for cross-contamination with viruses, bacteria, fungi, protozoa, organic molecules, such as prions, natural toxins, and persistent organic pollutants [226]. The proliferation of contaminant organisms may occur if preservation methods are not adequate, for instance, after harvesting the algae. However, guidelines to prevent these contaminations are extensively described in food security standards, such as regarding hazard analysis critical control points (HACCP).
Biomass manipulation after harvesting could prevent the degradation of the product while ensuring a low level of contaminants. A conventional example of this manipulation is washing and boiling seaweed, as this can reduce its arsenic and iodine contents, achieving decreases of around 22% in inorganic arsenic and an almost complete reduction of iodine in Laminaria sp. [164,242,243,252,253,254].
In the case of microalgae, boiling can have both beneficial and harmful effects. For example, boiling decreases the concentration of microcystin by 97% after 5 min [255], but increases the harmful characteristics of brevetoxin [256,257]. Other uncertainties remain, due to the lack of data on the characterization of these compounds, their bioavailability, and variability among species, and their cultivation, season, or geographic location [164]. Therefore, assessing the risk exposure in the case of unprocessed biomass can lead to inaccuracies in the final product evaluation.
3.3. Consumer’s Perception
Another important factor to be highlighted regarding the introduction of algae to the market is their organoleptic characteristics, in terms of their color, odor, texture, and flavor [128]. In terms of the public’s perceptions of algae consumption, the sensorial aspects of the food have a direct impact on diminishing the perceived risk and uncertainty, which could have a direct impact on the behavioral intention to consume algae products [14,80,84,99,111,133,136,162,203,220,223,258,259,260,261,262,263,264,265,266,267]. Since the first studies involving humans, the acceptability and palatability of algae have been doubtful because of their naturally unpleasant flavors, dark, strange colors, and strong odors [111,112,128,134,223,268]. For example, Chlorella sp. was defined as having a strong vegetable-like flavor and aroma that would taste like powdered green tea; in the case of some dried seaweeds, they have an intense green color that could be considered limiting or off-putting when mixed in foods [111,133,136]. Similar observations were noted in green-colored Scenedesmus-based food, where the color was not well-received by testers. In France, tests on the nutritional value and customer acceptability of Spirulina-based foods concluded that algae were not very enjoyable to eat, due to their strong taste, smell, and even color [223]. French consumer studies identified the main reasons that may prevent potential consumers from purchasing algae-based food products; namely, a lack of consumer knowledge regarding the possibility of using them in food, their unavailability in stores, unappealing taste and image, and consumers’ overall apprehension [161].
The unique flavor of “umami” makes algae stand out gastronomically [162,267,269]. The term means “delicious essence”, and it is considered the “fifth taste”, derived from the glutamic acid present in kelp (kombu) [162,267]. In addition, given that algae are a diverse group, they display a variety of pleasant properties among species, which are not only limited to “umami”, for instance, a green-tea aroma and flavor in kelp species [260].
The processing methods, as well as the storage conditions, could influence the flavor [94]. Nevertheless, it was predicted and demonstrated that the general acceptance of algae in the human diet would increase through its incorporation in foods, given the general acceptance of the flavor when mixed in low dosages in traditional foods, such as cookies or chocolate [111,133,136,223]. Other strategies have been proposed to improve the sensorial characteristics during the processing of algae, such as developing mutant algae with low chlorophyll [134,177,223], but their acceptability is dependent on the food product. Thus, besides sushi or snacks, algae are now present in a wide range of European food products, including soups, salads, jellies, canned foods, pickles, dips, and sauces (mustard, marinades, pates, rillettes, and pesto), burgers, pasta, cakes, spices, salts, cheeses, bread, cookies, chips, and condiments, and drinks (juices, gin, liquor, ales, and beer) [14,80,99,203,261,262,263,264,265].
Finally, and in a transversal way, a general lack of awareness and accessibility regarding algae is evidenced [137,270,271,272]. Socio-demographic factors, such as age, occupation, education, and even social status appear to explain the differences in terms of the level of knowledge [270,272]. Additionally, it is also pointed out that the lack of available information contributes to less comprehensive knowledge about the subject, which, consequently, leads to lower consumption interest [270,272]. Internet is cited as the most used source of information, followed by relatives and friends [273]. While familiarity is suggested as an important factor that promotes consumption, the nutritional and health benefits are pointed out as the main reason why consumers included algae, such as Spirulina, in their diet [270,272,273,274,275,276,277].
On the other hand, environmental concerns are quite different among countries; for instance, while Spanish consumers consider microalgae as being environmentally friendly, the French have contrastingly double perceptions, and the Belgians do not see this as an incentive to motivate consumption [272,275,277]. Interestingly, a German survey indicates that even though the population wishes microalgae to contribute to world sustainability, they are more immediately concerned with their own healthy and balanced diet [278]. Other apparent obstacles to algae consumption seem to be the lack of consuming habits, pricing, and availability on the market, along with neophobia [272,275,277]. Furthermore, in the context of some studies, different target audiences were identified based on psychographic factors, such as sports participants, vegetarians, vegans, and foodies, who presented a greater predisposition for algae-based functional foods [274,275]. Similar target groups were identified based on their diets, like lacto-ovo-vegetarians, vegans, or simply consumers who do not feel the need to eat meat [273,276].
Over the last two decades, changes in the consumers’ mindset have been happening; now, the end consumers are more informed and receptive to sustainable behaviors. This aligns with both the new and improved support policies for research projects and programs, leading to the continuous expressive growth of algae applications in the European market [125,167,172,272,279]. The topmost motive of Western societies to follow the green consumerism trend is the belief that new environmentally friendly techniques pose an alternative to existing unsustainable processes. This leads to a higher social status and the prosocial reputation of the society, also referred to as conspicuous conservation [280,281].
Italian consumers have a high level of willingness to eat seaweed, according to studies, given the familiarity with seaweed gastronomy in both national and Asian traditional dishes [95]. In Nordic cuisine, seaweeds are seen as having great potential for food applications, given their sensory qualities (flavor, texture, and color) and overall health benefits. However, consumers are still very apprehensive about the idea of their inclusion as sea vegetables, due to traditional cultural frameworks and prejudices regarding rapid changes in Nordic cuisine [260]. To invert this tendency, research shows that it is crucial to eliminate consumer hesitation toward trying new food products and to focus on product differentiation by the use of labels and certifications (quality, safety, traceability, and organic status) [161].
4. Legislation on Edible Algae Species
4.1. Legislation for Algae Consumption in Some Algae-Consuming Countries
Some specific legislation and food standards entities regulate how algae can be used as food for human consumption worldwide. Usually, algae and algae-based products are regulated individually, as in the EU. The categories applied to algae are in foods including food supplements and food additives, feed and feed additives, nutraceuticals, cosmetics, packaging materials, fertilizers, biostimulants, and biofuels.
In the USA, the safety of food items, including algae products, is under the regulation of the Food and Drug Administration (FDA), which grants the status of being generally recognized as safe (GRAS) to any substance that is considered to be safe for human consumption [173,282]. GRAS status is required for any organism that is going to be used as a food or a food ingredient and can be achieved in two ways: (a) through documented evidence of consumption by humans over a long period, and (b) by having scientific proofs showing that a substance is safe under the conditions of its intended use. Nevertheless, whenever any new substance being introduced to food is considered as a food additive, it has to be subjected to a premarket review and approval by the FDA, which determines the safety of the ingredient, unless a consensus among qualified experts has already recognized the substance as being safe [39,283,284]. Canada has similar standards to those of the USA; the organization known as Health Canada oversees food safety supervision and describes novel foods as any food product that is new or has been changed compared to the existing foods. Products classified as novel foods, including genetically modified food, must be assessed by Health Canada and reviewed for safety before being sold in the country [39,285,286].
Similar approaches in the regulations of novel foods are also found in Australia and New Zealand, where the Food Standards in Australia and New Zealand (FSANZ) are the entity responsible for regulating the use of new ingredients. According to FSANZ, the novel food and novel food ingredients are considered all non-traditional food and/or its derivatives requiring an assessment of the public health under the Novel Foods in the Food Standards Code. A novel food cannot be sold as food or used as a food ingredient unless expressly permitted by the Code in Standard 1.5.1 [173,287].
The growth of novel food regulation in China is justified by the development of the food industries and the long-lasting food culture [288,289,290]. The Chinese administrative measures for the safety of the review of new food raw materials (2013) rule that under the new regulations, “novel foods” were replaced by “new food raw materials” in the Food Safety Law (2009). The term “new food raw materials” refers to: (a) animals, plants, and microorganisms; (b) ingredients extracted from animals, plants, and microorganisms; (c) food ingredients where the original composition has been changed; and (d) other newly developed food raw materials, which are not part of traditional eating habits in China. The National Health Commission (NHC) is responsible for the review of materials for the safety evaluation of new food raw materials, for the issuing of a license [291], and for conducting the pre-market approval of novel food products. In China, the legislation of algae products mainly focuses on microalgae since seaweed such as Laminaria, Gracilaria, Porphyra, Undaria, and Eucheuma have been accepted as a traditional food instead of a novel food by the Chinese for many years. Moreover, algae products could also be approved as “Food for Special Medical Purposes”, with a 5-year validation certificate being issued by the Center for Food Evaluation in the State Administration for the Market Regulation of China. When compared with the EU, both have established a similar system to regulate novel foods, within some common perspectives.
Table 2 shows the legislation in Europe compared to other countries. Some similarities between countries regarding the regulation of novel foods were noted, such as the requirement of a premarket safety assessment so that these new foods can be commercialized. However, some divergences were also found; these vary between countries, such as the identification of these new foods, the requirements needed to bring such foods to the market, and their regulatory levels.

Table 2.
Legislation on edible algae species as food products.
4.2. EU Legislation
Algae, as food products in the EU, are subject to the General Food Law Regulation (EU) No. 178/2002 which is implemented in all member countries. Furthermore, the entry of novel algae species into the EU market is regulated by the Novel Food Regulation (EU), No. 2015/2283. The application procedure for authorizing the placing on the market within the EU of novel food and for updating the Union List may be found in the Official Journal of the EU (L327/1).
Consumption history affects the regulatory status, meaning that the entry of novel algae species that have not been used as food in the EU before 15 May 1997 need prior authorization, to ensure their safety for human consumption. Additionally, a notification system is available to offer an easier route to the EU market for some species that have not been used in Europe but that are considered traditional foods in developing countries. This “traditional food” status is given if a history of safe food use for at least 25 years can be proven. This new Regulation facilitates the introduction of new and innovative foods to the EU market, provided that specifications, such as labeling and usage conditions, are respected, thus guaranteeing food safety for European consumers.
The rules enforced on algae products are regulated by Directive 2002/46/EC, which aims to protect consumers against the potential health risks associated with toxicity or misinformation. The SANCO/2006/E4/018 report assessed the use of substances, other than vitamins and minerals, with a nutritional or physiological effect in food supplements, which includes amino acids, enzymes, pre- and probiotics, essential fatty acids, botanicals (within which algae are included), botanical extracts, and other bioactive substances. In addition, the EU delivered several reports (SEC (2008) 2976 and SEC (2008) 2977) stating that specific rules being made applicable to substances other than vitamins and minerals for use in FS are not justified.
Food additives, such as preservatives, colorants, or sweeteners, which are normally used during food preparation are regulated by (EC) 1333/2008 and (EU) 231/2012 legislations. The safety of these products and the authorization assessment are carried out by the Scientific Committee on Food (SCF) and the EFSA. The concept of functional food, derived from Japan and the USA, goes beyond the nutritional and health benefits of traditional nutritional effects and differs from food supplements and nutraceuticals. Although this market is gaining extensive popularity in the last few years, the EU still lacks a regulatory framework for functional food, hindering economic competitiveness in this sector.
Legislation inadequacies remain regarding the use of algae as a raw material for food. Therefore, the European Committee for Standardization (CEN) developed a technical committee for algae and algae products, including cyanobacteria and Thraustochytrids (CEN/TC 454) in 2017, which focuses on standardizing the specification, classification, terminology, algae processing, and determination methods for algae biomass, extracts or purified compounds. It is noteworthy that as these guidelines are updated, the legislation is changed accordingly.
Because of limited European regulation, some EU countries are implementing their specific regulations regarding the use of algae as a food source. Non-approved algae species are being commercialized for food purposes in several European countries. For example, in 1990, France was the first European country to establish a specific regulation concerning the use of seaweed for human consumption as a non-traditional food substance, authorizing the consumption of algae for food other than what is considered to be a novel food, according to the EU [21,156]. Therefore, legislation may differ slightly between the European Member States.
4.3. Edible Algae Species in Europe
Evidence for the consumption of more than 150 edible species of algae was identified in Europe, of which 14% are considered microalgae and cyanobacteria and 86% are considered seaweed (Figure 3). Interestingly, from these 150 species, only 30 species are approved as novel foods by the EU. For seaweed alone, more than 650 species are considered edible and are consumed worldwide, particularly in Asian countries; therefore, they have entered the EU market, despite not being listed in the European novel food catalog [5]. This indicates that the market still relies heavily on imports to satisfy the demand. Nevertheless, there is an evident growth potential for this sector. Recently, a technical report was published by Araújo, compiling the latest updates on algae items and species in official lists (the Novel Food Catalog, the Union List of Authorized Novel Foods, and official Member States’ lists of food and food supplements) as well as unofficial lists, including the extensive research performed in the current review [292].

Figure 3.
Algae species consumed in Europe, subdivided into Microalgae and Seaweeds (the Chlorophyta, Phaeophyceae, and Rhodophyta groups). Highlighted novel food (NF) species are presented in the Novel Food Catalog; food supplements (FS) are those Food Supplements listed on the European Union list. Species with consumption evidence before 1997 are highlighted *. A detailed table with the common names and referenced evidence is given in the Supplementary Material (Table S2).
5. Perspectives of the Consumption of New Species
5.1. Research and Development
Research and development to mature the production process (from cultivation to packaging) have been encouraged to introduce new algae species to the market [10].
Microalgae biotechnology R&D dates back to the 19th century; since then, technologies have evolved, leading to the continuous improvement of production systems [293]. Today, microalgae biotechnology is a major component of the blue economy, which had an estimated value of EUR 2.4 billion globally in 2011. In the EU, the blue economy has a gross added value of EUR 0.8 billion, which represents ~30% of global activities [294]. At present, microalgae biotechnology is a niche market in Europe, due to high production costs of about EUR 5 kg−1, which are planned to decrease to EUR 0.5 kg−1 in the best-case scenario within the next decade [295].
Seaweed wild harvesting is a well-established industry, and the cultivation process has been promoted [11]. Specifically, for the European seaweed-based food industry, the main identified challenges and research recommendations presented by the Phycomorph European Guidelines for the Sustainable Aquaculture of Seaweeds were listed as:
- i.
- To increase the risk-benefit seaweed analyses, with added knowledge on the speciation of iodine/chemical form, and bioavailability;
- ii.
- To standardize and define the chemical compound classes, activities, traceability, methods, and species identification;
- iii.
- To further investigate the domestication of new species, the effects of preservation methods and treatments on biomass, and to define the best storage procedures and best practices for the evaluation of product shelf-life; and
- iv.
- To implement sensory evaluation panels [65].
Scientists successfully brought a green fingerprint to algae as an alternative, sustainable feedstock for food and other applications (feed, cosmetics, fertilizers, fuel, etc.), which led to an increase in the funding rate for algae-related projects within the European Union over the last few decades (Figure 4a).
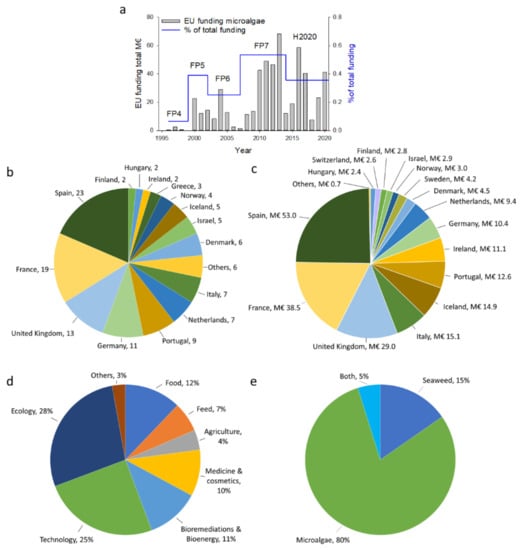
Figure 4.
Overview regarding EU funding for algae-related projects. (a) Funding per year for microalgae-related projects in the European Commission, within the framework programs FP4-H2020; (b) the number of H2020 projects led, per country; (c) EC projects funding per leading country; (d) main areas of the projects funded; and (e) the proportion of projects connected to microalgae or seaweed [296].
The eco-friendly image of algae industries is currently moving this sector toward larger markets, bringing with it the creation of new, skilled jobs, study programs, and economic value. Most EU projects use the green fingerprint and sustainability aspects of algae products or processes to promote a more sustainable way of living. The driving arguments for food-related algae projects and products are usually related to green consumerism. Indeed, algae technologies pose advantages compared to traditional approaches for a given application. For example, algae can be a source of food in areas where no arable lands or potable water is available and, thus, do not necessarily compete with agriculture. In addition, unsustainable fish- and soy-based lipids or proteins in food and feeds are replaceable by algae products, reducing overfishing and deforestation. This approach reduces the eutrophication and contamination of the environment and allows the recirculation of limited and valuable resources (e.g., phosphorus) [297].
Despite this green image, algae industries generally receive low recognition among society and decision-makers. During the Seventh Framework Program (FP7), 2007–2013, only ~0.5% of the EUR 44 billion of total funding was spent on algae-related projects, which was the highest percentage so far. Notably, approximately ten times more money was invested in agriculture research. Nonetheless, algae biotechnology is earning its position in the research world in Europe.
For the Horizon 2020 Research and Innovation program, EUR 220.8 million was invested in 124 projects that focused mainly on microalgae and seaweed (see the Supplementary Material, Table S3). Most of the projects (n = 23) were coordinated by Spanish companies and institutions, accounting for 19% of the projects, followed by those in France (n = 19), the United Kingdom (n = 13), Germany (n = 11), and Portugal (n = 9; Figure 4b). Likewise, projects coordinated by companies and institutions in Spain, France, and the UK received the most funding (24%, 17%, and 13%, respectively), consisting of 55% of the total funding for the algae projects within the H2020 framework (Figure 4c).
The H2020 projects are classified into major topics. The area of ecology, which includes research about ecosystems and the interactions of organisms with their environment, incorporated most of the H2020 projects (28% of all projects; Figure 4d). This area is mostly scientific and ecocentric in nature, with limited anthropocentric applications. However, the second-largest area is technology, comprising 25% of the total projects, and includes the development of biorefinery, photobioreactors, and scale-ups, or white biotechnology. Many projects incorporate elements of this field to reach the final goal of efficient cultivation, the separation of compounds, and the creation of new products. Food applications are the third-largest area (12% of all projects), which comes as no surprise. The idea of feeding the rising human population with algae-based foods, the increasing trend toward green consumerism, and the shift from meat-based diets to vegetarianism or veganism drive the demand for protein, fiber, or PUFA-rich “superfoods”, including algae. It is expected that with the development of the existing technologies for culturing algae, food-related projects will earn a larger share of future research funding.
About 80% of the H2020 projects in algae R&D are focusing on microalgae (Figure 4e); this interest was piqued by their faster growth rate compared to seaweed and their promising applications in foods, feeds, and other sectors (such as biofertilizers and bioenergy), which are, as yet, underexplored [295]. When compared with seaweed, the novelty of microalgae, which have only been cultivated on a commercial scale since the 1970s [293], promotes high expectations in terms of microalgae and must be understood and improved upon to reveal their true potential.
The main challenges to be tackled for microalgae biotechnology are the cost reduction for the mass production of metabolites and primary components, such as lipids, sugars, polymers, or proteins for food, feeds, and the chemical industries. The most promising breakthrough technologies in the sector are the discovery and development of high-added-value products and bioactive compounds, as well as downstream processing and drug discoveries [293,295].
The low number of seaweed-related projects is reflected by a drop in publications and patents between the 1990s and the 2000s [298]. In 2016, Spain and France were represented among the top 10 global researchers on the topic of seaweed farming, mostly dedicated to the subjects of bioremediation, environmental impacts, and the development of farming technologies [299]. Additionally, the UK, Ireland, and Italy show a significant production of seaweed and the research output of seaweed aquaculture [298]. The top seaweed producers and patent holders are the Asian nations (China, Japan, and South Korea) where not only does the activity have traditional importance but also where large investments and efforts are made in research [300]. Scientific research for biotechnology is the main driver for seaweed production innovation and the rate at which new species become domesticated, yielding a significant return in terms of intellectual property [219,300]. Research gaps include the need for the development of cryopreservation methods, the assessment of environmental factors on targeted phenotypic traits, and work unraveling fertility, reproduction, and genetic compatibility [21,219].
5.2. Advances in Technology Production
A relevant advance factor, in terms of the algae market as food, is nanotechnology. The concept of nanotechnology involves the creation, characterization, and/or manipulation of materials that have a length of between 1 and 100 nm [301].
Algae are able to synthesize multiple types of nanoparticles (NPs). By choosing species that are able to produce NPs, the downstream process of algae cultivation—especially microalgae—becomes easier and cheaper. This happens because metallic NPs have the capability to clump together, allowing a faster, cleaner, and easier separation of the algae from the culture media (2). Biological sensors made from nanoparticles are also being developed to identify the presence of hostile microorganisms. The incorporation of this technology into the cultivation of algae—especially microalgae—would enable a possible way of controlling the contamination of algal cultures, since this could become an accurate and fast technique by which to signal the presence of contaminants and even kill them, due to their bioactive properties [301].
Metallic NPs have also been reported to have anti-bacterial and anti-fungal properties, especially silver and gold NP, AgNP, and AuNP, respectively [302]. By including these particles in their packaging, the shelf life of a product is increased: not only do they prevent biological contaminants but also provide a better way to control the humidity and oxygen by increasing the gas diffusion length (compared to normal polymer packaging) of the protecting layer separating the inside from the outside of the package.
5.3. Algae Food Products
With the increase in the research for food products, an increase in the market for algae products is expected to make space for new products and brands. Algae-based products for the foods, cosmetics, and nutraceuticals markets can benefit compared to existing products, if the manufacturing companies advertise their positive properties, including the richness of their essential nutrients or the product’s green fingerprint. A “green” product will drive consumers to spend more money, even if it is of lower quality or performance than a regular product [281]; interestingly, this behavior is triggered by status competition among citizens. Thus, the promotion of algae products for green consumerism will be more fruitful if they are destined to be consumed in public; this behavior has been stimulated by the brand designers.
The design of algae foods is essential for market integration because algae are usually not part of Western cuisine and may have a distasteful or fishy taste and smell. Novel algae food designs are supported by EU projects. Commonly, these projects aim to position small- and medium-sized enterprises (SMEs) in the algae health food market and usually include strategies for promoting and establishing algae products in these markets. Indeed, the evolving green consumerism market serves as a stepping-stone for many companies toward the full market integration of algae foods, which is key for the movement from the development bench to the market and away from the dependency on public funding. Once established, algae products will probably spread quickly among communities via peer-to-peer influence and social norms.
5.4. Future Perspectives
With legislation amendment and investment in the research and development of production systems and algae products, the algae market has the potential to grow in Europe (Figure 5). The identification of novel algae species with properties desired by the markets (e.g., biochemicals, odors, or haptic traits), their approval as novel foods, and the final commercial production are required to foster the positioning of algae in the food market. Notably, while multiple studies have previously identified promising algae as novel foods and the algae-producing companies are eager to commercialize this valuable bioresource, the current legislation procedures are too lengthy to adapt to these rapidly changing markets and discoveries, and urgently require revision for a quicker, cheaper and safe approval process for novel algal strains.
Figure 5.
Schematic summary of this review.
6. Conclusions
This review has highlighted the historical consumption of microalgae and seaweed species diversity, and their applications as foodstuffs, food supplements, and food additives in Europe. Concerns regarding establishing algae products in the market, such as production constraints (such as large-scale production technology limitations and domestication challenges), health impacts (caused by the toxicology of species, allergens, or contaminants), and consumers’ perceptions (acceptance and knowledge) have been addressed, as well as the legislation process followed to submit algae to the EU Novel Food Catalog. The current legislation is not broad enough for the algae sector, with specific regulations within each country and several species being produced, consumed, and commercialized beyond the approved catalog. Therefore, an update of the authorized species list is urgently required. It is evident that the algae market has strong motivations and huge potential, due to algae’s nutritional and health benefits, the likelihood of sustainable production, and especially, the need to address the rise in food demand by the growing population. The development of new projects and products has been encouraged, especially regarding microalgae cultivation technology, as seaweed products are becoming more established in the global market.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/foods11131871/s1, Table S1: Algae production technologies. Table S2: List of algae species consumed in Europe. Table S3: Projects funded through the Horizon 2020 Research and Innovation program, divided by country (major role). References [5,9,11,14,15,21,35,47,66,68,69,70,73,80,81,83,84,85,89,92,93,96,97,98,100,102,103,104,113,140,141,142,143,144,148,149,155,156,158,179,181,182,184,185,191,192,193,194,206,223,224,262,263,292,303,304,305,306,307,308,309,310,311,312,313,314,315,316,317,318,319,320,321,322,323,324,325,326,327,328,329,330,331,332,333,334,335,336,337,338,339,340,341,342,343,344,345,346,347,348,349,350,351,352,353,354,355,356,357,358,359,360,361,362,363,364,365,366,367,368,369,370,371,372,373,374,375,376,377,378,379,380,381,382,383,384,385] are cited in the supplementary materials.
Author Contributions
Conceptualization, P.S.C.S., H.P., L.G., M.H.A., J.L.S., J.N., J.C.S.V. and L.G.S.; writing—original draft preparation, M.C.M., S.N., A.F., C.P., D.F., D.S., F.G. (Florinda Gama), G.B., R.J., S.S.A., P.S.C.S., A.T.G., H.P., L.G. and L.G.S.; writing—review and editing, M.C.M., S.N., F.G. (Fengzheng Gao), P.S.C.S., A.T.G., H.P., L.G., R.F.P., M.H.A., J.L.S., J.N., J.C.S.V. and L.G.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was partially funded by the ProFuture project (2019–2023 “Microagae protein-rich ingredients for the food and feed of the future”). ProFuture has received funding from the European Union’s Horizon 2020 research and innovation program, under grant agreement No. 862980. Funding was also partially provided from the National Funds through FCT—Fundação para a Ciência e Tecnologia, within the projects UID/BIA/00329/2020–2023 and UID/Multi/04326/2020. R.F.P. was supported by an FCT Science and Technology Management Fellowship grant SFRH/BGCT/135478/2018. Alice Ferreira was supported by an FCT Ph.D. grant SFRH/BD/144122/2019.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Acknowledgments
The authors are grateful to the Instituto Macrobiótico de Portugal, Ricardo Melo, the Biblioteca da Faculdade de Ciências da Universidade de Lisboa, and Ângelo Rocha for providing data and knowledge about seaweed consumption before 1997 in Portugal. The authors are grateful to the late Ana I. Neto for her helpful comments on the Azorean seaweed historical data, and to Afonso C. L. Prestes for his support in the compilation of the historical data before 1997, in the Azores.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Caporgno, M.P.; Mathys, A. Trends in Microalgae Incorporation Into Innovative Food Products With Potential Health Benefits. Front. Nutr. 2018, 5, 58. [Google Scholar] [CrossRef] [PubMed]
- Bleakley, S.; Hayes, M. Algal Proteins: Extraction, Application, and Challenges Concerning Production. Foods 2017, 6, 33. [Google Scholar] [CrossRef] [PubMed]
- Dillehay, T.D.; Ramírez, C.; Pino, M.; Collins, M.B.; Rossen, J.; Pino-Navarro, J.D. Monte Verde: Seaweed, Food, Medicine, and the Peopling of South America. Science 2008, 320, 784–786. [Google Scholar] [CrossRef] [PubMed]
- Khalilieh, H.S.; Boulos, A. A glimpse on the uses of seaweeds in islamic science and daily life during the classical period. Arab. Sci. Philos. 2006, 16, 91–101. [Google Scholar] [CrossRef]
- Pereira, L. Edible Seaweeds of the World; CRC Press: Boca Raton, FL, USA, 2016; ISBN 9780429154041. [Google Scholar]
- Richmond, A.E.; Soeder, C.J. Microalgaculture. Crit. Rev. Biotechnol. 1986, 4, 369–438. [Google Scholar] [CrossRef]
- Boukid, F.; Castellari, M. Food and Beverages Containing Algae and Derived Ingredients Launched in the Market from 2015 to 2019: A Front-of-Pack Labeling Perspective with a Special Focus on Spain. Foods 2021, 10, 173. [Google Scholar] [CrossRef]
- Alexandratos, N.; Bruinsma, J. World Agriculture towards 2030/2050: The 2012 Revision; ESA Working Paper No. 12-03; FAO: Rome, Italy, 2012. [Google Scholar]
- Norsker, N.-H.; Barbosa, M.J.; Vermuë, M.H.; Wijffels, R.H. Microalgal production—A close look at the economics. Biotechnol. Adv. 2011, 29, 24–27. [Google Scholar] [CrossRef]
- Smith, A.G.; Tredici, M.R.; Boussiba, S.; Verdelho, V.; Cadoret, J.-P.; Davey, M.P.; Huete-Ortega, M.; Acien, F.G.; Schmid-Staiger, U.; Rodriguez, H.; et al. EABA—Position Paper—What Are Algae? EABA: Florence, Italy, 2020. [Google Scholar]
- Araújo, R.; Vázquez Calderón, F.; Sánchez López, J.; Azevedo, I.C.; Bruhn, A.; Fluch, S.; Garcia Tasende, M.; Ghaderiardakani, F.; Ilmjärv, T.; Laurans, M.; et al. Current Status of the Algae Production Industry in Europe: An Emerging Sector of the Blue Bioeconomy. Front. Mar. Sci. 2021, 7, 626389. [Google Scholar] [CrossRef]
- Fernández, F.G.A.; Reis, A.; Wijffels, R.H.; Barbosa, M.; Verdelho, V.; Llamas, B. The role of microalgae in the bioeconomy. New Biotechnol. 2021, 61, 99–107. [Google Scholar] [CrossRef]
- Fédération des Spiruliniers de France. Available online: https://www.spiruliniersdefrance.fr/ (accessed on 9 May 2022).
- Mouritsen, O.G.; Rhatigan, P.; Pérez-Lloréns, J.L. The rise of seaweed gastronomy: Phycogastronomy. Bot. Mar. 2019, 62, 195–209. [Google Scholar] [CrossRef]
- ALGAplus Products. Available online: https://www.algaplus.pt/produtos/ (accessed on 10 April 2021).
- Spolaore, P.; Joannis-Cassan, C.; Duran, E.; Isambert, A. Commercial applications of microalgae. J. Biosci. Bioeng. 2006, 101, 87–96. [Google Scholar] [CrossRef]
- Peñalver, R.; Lorenzo, J.M.; Ros, G.; Amarowicz, R.; Pateiro, M.; Nieto, G. Seaweeds as a functional ingredient for a healthy diet. Marine Drugs. Mar. Drugs 2020, 18, 301. [Google Scholar] [CrossRef]
- Holdt, S.L.; Kraan, S. Bioactive compounds in seaweed: Functional food applications and legislation. J. Appl. Phycol. 2011, 23, 543–597. [Google Scholar] [CrossRef]
- Nova, P.; Martins, A.P.; Teixeira, C.; Abreu, H.; Silva, J.G.; Silva, A.M.; Freitas, A.C.; Gomes, A.M. Foods with microalgae and seaweeds fostering consumers health: A review on scientific and market innovations. J. Appl. Phycol. 2020, 32, 1789–1802. [Google Scholar] [CrossRef]
- Ścieszka, S.; Klewicka, E. Algae in food: A general review. Crit. Rev. Food Sci. Nutr. 2019, 59, 3538–3547. [Google Scholar] [CrossRef]
- Barbier, M.; Charrier, B.; Araujo, R.; Holdt, S.L.; Bertrand, J.; Céline, R. PEGASUS-PHYCOMORPH European Guidelines for a Sustainable Aquaculture of Seaweeds. COST Action FA1406; Barbier, M., Charrier, B., Eds.; HAL Open Science: Lyon, France, 2019. [Google Scholar]
- Mintel. Seaweed-flavoured food and drink launches increased by 147% in Europe between 2011 and 2015. Available online: http://www.mintel.com/press-centre/food-and-drink/seaweed-flavoured-food-and-drink-launches-increased-by-147-in-europe-between-2011-and-2015 (accessed on 9 March 2016).
- Kumari, P.; Kumar, M.; Reddy, C.R.K.; Jha, B. Algal lipids, fatty acids and sterols. In Functional Ingredients from Algae for Foods and Nutraceuticals; Elsevier: Amsterdam, The Netherlands, 2013; pp. 87–134. [Google Scholar]
- Wielgosz-Collin, G.; Kendel, M.; Couzinet-Mossion, A. Lipids, fatty acids, glycolipids, and phospholipids. In Seaweed in Health and Disease Prevention; Fleurence, J., Levine, I., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 185–221. [Google Scholar]
- Marik, P.E.; Varon, J. Omega-3 Dietary Supplements and the Risk of Cardiovascular Events: A Systematic Review. Clin. Cardiol. 2009, 32, 365–372. [Google Scholar] [CrossRef]
- Huang, H.-L.; Wang, B.-G. Antioxidant Capacity and Lipophilic Content of Seaweeds Collected from the Qingdao Coastline. J. Agric. Food Chem. 2004, 52, 4993–4997. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Ascherio, A.; Hu, F.B.; Stampfer, M.J.; Willett, W.C.; Siscovick, D.S.; Rimm, E.B. Interplay Between Different Polyunsaturated Fatty Acids and Risk of Coronary Heart Disease in Men. Circulation 2005, 111, 157–164. [Google Scholar] [CrossRef]
- Sakamoto, A.; Saotome, M.; Iguchi, K.; Maekawa, Y. Marine-derived omega-3 polyunsaturated fatty acids and heart failure: Current understanding for basic to clinical relevance. Int. J. Mol. Sci. 2019, 20, 4025. [Google Scholar] [CrossRef]
- Freitas, R.; Campos, M.M. Protective Effects of Omega-3 Fatty Acids in Cancer-Related Complications. Nutrients 2019, 11, 945. [Google Scholar] [CrossRef]
- Rennie, K.L.; Hughes, J.; Lang, R.; Jebb, S.A. Nutritional management of rheumatoid arthritis: A review of the evidence. J. Hum. Nutr. Diet. 2003, 16, 97–109. [Google Scholar] [CrossRef]
- SanGiovanni, J.P.; Parra-Cabrera, S.; Colditz, G.A.; Berkey, C.S.; Dwyer, J.T. Meta-analysis of Dietary Essential Fatty Acids and Long-Chain Polyunsaturated Fatty Acids as They Relate to Visual Resolution Acuity in Healthy Preterm Infants. Pediatrics 2000, 105, 1292–1298. [Google Scholar] [CrossRef]
- Barros, R.; Moreira, A.; Fonseca, J.; Delgado, L.; Graça Castel-Branco, M.; Haahtela, T.; Lopes, C.; Moreira, P. Dietary intake of α-linolenic acid and low ratio of n-6:n-3 PUFA are associated with decreased exhaled NO and improved asthma control. Br. J. Nutr. 2011, 106, 441–450. [Google Scholar] [CrossRef]
- Tsai, C.-J.; Sun Pan, B. Identification of Sulfoglycolipid Bioactivities and Characteristic Fatty Acids of Marine Macroalgae. J. Agric. Food Chem. 2012, 60, 8404–8410. [Google Scholar] [CrossRef]
- da Costa, E.; Melo, T.; Moreira, A.; Bernardo, C.; Helguero, L.; Ferreira, I.; Cruz, M.; Rego, A.; Domingues, P.; Calado, R.; et al. Valorization of Lipids from Gracilaria sp. through Lipidomics and Decoding of Antiproliferative and Anti-Inflammatory Activity. Mar. Drugs 2017, 15, 62. [Google Scholar] [CrossRef]
- Paiva, L.; Lima, E.; Patarra, R.F.; Neto, A.I.; Baptista, J. Edible Azorean macroalgae as source of rich nutrients with impact on human health. Food Chem. 2014, 164, 128–135. [Google Scholar] [CrossRef]
- Patarra, R.F.; Leite, J.; Pereira, R.; Baptista, J.; Neto, A.I. Fatty acid composition of selected macrophytes. Nat. Prod. Res. 2013, 27, 665–669. [Google Scholar] [CrossRef]
- Paiva, L.; Lima, E.; Neto, A.I.; Marcone, M.; Baptista, J. Health-promoting ingredients from four selected Azorean macroalgae. Food Res. Int. 2016, 89, 432–438. [Google Scholar] [CrossRef]
- Paiva, L.; Lima, E.; Neto, A.I.; Marcone, M.; Baptista, J. Nutritional and functional bioactivity value of selected Azorean macroalgae: Ulva compressa, Ulva rigida, Gelidium microdon, and Pterocladiella capillacea. J. Food Sci. 2017, 82, 1757–1764. [Google Scholar] [CrossRef]
- Torres-Tiji, Y.; Fields, F.J.; Mayfield, S.P. Microalgae as a future food source. Biotechnol. Adv. 2020, 41, 107536. [Google Scholar] [CrossRef]
- Becker, E.W. Micro-algae as a source of protein. Biotechnol. Adv. 2007, 25, 207–210. [Google Scholar] [CrossRef] [PubMed]
- Koyande, A.K.; Chew, K.W.; Rambabu, K.; Tao, Y.; Chu, D.-T.; Show, P.-L. Microalgae: A potential alternative to health supplementation for humans. Food Sci. Hum. Wellness 2019, 8, 16–24. [Google Scholar] [CrossRef]
- Samarakoon, K.; Jeon, Y.-J. Bio-functionalities of proteins derived from marine algae—A review. Food Res. Int. 2012, 48, 948–960. [Google Scholar] [CrossRef]
- Admassu, H.; Gasmalla, M.A.A.; Yang, R.; Zhao, W. Bioactive Peptides Derived from Seaweed Protein and Their Health Benefits: Antihypertensive, Antioxidant, and Antidiabetic Properties. J. Food Sci. 2018, 83, 6–16. [Google Scholar] [CrossRef] [PubMed]
- Ursu, A.-V.; Marcati, A.; Sayd, T.; Sante-Lhoutellier, V.; Djelveh, G.; Michaud, P. Extraction, fractionation and functional properties of proteins from the microalgae Chlorella vulgaris. Bioresour. Technol. 2014, 157, 134–139. [Google Scholar] [CrossRef]
- Ovando, C.A.; de Carvalho, J.C.; Vinícius de Melo Pereira, G.; Jacques, P.; Soccol, V.T.; Soccol, C.R. Functional properties and health benefits of bioactive peptides derived from Spirulina: A review. Int. Food Res. J. 2018, 34, 34–51. [Google Scholar] [CrossRef]
- Fleurence, J. Seaweed proteins. Trends Food Sci. Technol. 1999, 10, 25–28. [Google Scholar] [CrossRef]
- Canelli, G.; Tarnutzer, C.; Carpine, R.; Neutsch, L.; Bolten, C.J.; Dionisi, F.; Mathys, A. Biochemical and nutritional evaluation of Chlorella and Auxenochlorella biomasses relevant for food application. Front. Nutr. 2020, 7, 168. [Google Scholar] [CrossRef]
- Usman, A.; Khalid, S.; Usman, A.; Hussain, Z.; Wang, Y. Algal Polysaccharides, Novel Application, and Outlook. In Algae Based Polymers, Blends, and Composites: Chemistry, Biotechnology and Materials Science; Elsevier: Amsterdam, The Netherlands, 2017; pp. 115–153. ISBN 9780128123607. [Google Scholar]
- Xu, S.-Y.; Huang, X.; Cheong, K.-L. Recent Advances in Marine Algae Polysaccharides: Isolation, Structure, and Activities. Mar. Drugs 2017, 15, 388. [Google Scholar] [CrossRef]
- Hayes, M.; Tiwari, B. Bioactive Carbohydrates and Peptides in Foods: An Overview of Sources, Downstream Processing Steps and Associated Bioactivities. Int. J. Mol. Sci. 2015, 16, 22485–22508. [Google Scholar] [CrossRef]
- Garcia-Vaquero, M.; Rajauria, G.; O’Doherty, J.V.; Sweeney, T. Polysaccharides from macroalgae: Recent advances, innovative technologies and challenges in extraction and purification. Int. Food Res. J. 2017, 99, 1011–1020. [Google Scholar] [CrossRef]
- MacArtain, P.; Gill, C.; Brooks, M.; Campbell, R.; Rowland, I.R. Nutritional Value of Edible Seaweeds. Nutr. Rev. 2007, 65, 535–543. [Google Scholar] [CrossRef]
- Devillé, C.; Gharbi, M.; Dandrifosse, G.; Peulen, O. Study on the effects of laminarin, a polysaccharide from seaweed, on gut characteristics. J. Sci. Food Agric. 2007, 87, 1717–1725. [Google Scholar] [CrossRef]
- Walsh, A.M.; Sweeney, T.; O’Shea, C.J.; Doyle, D.N.; O’Doherty, J.V. Effect of dietary laminarin and fucoidan on selected microbiota, intestinal morphology and immune status of the newly weaned pig. Br. J. Nutr. 2013, 110, 1630–1638. [Google Scholar] [CrossRef]
- Koyande, A.K.; Show, P.L.; Guo, R.; Tang, B.; Ogino, C.; Chang, J.S. Bio-processing of algal bio-refinery: A review on current advances and future perspectives. Bioengineered 2019, 10, 574–592. [Google Scholar] [CrossRef]
- Abe, J.-I.; Kotla, S.; Yu, J.; Baker, A.B.; Patil, N.P.; Le, V.; Sligar, A.D.; Mei, L.; Chavarria, D.; Yang, E.Y. Algal Polysaccharides as Therapeutic Agents for Atherosclerosis. Front. Cardiovasc. Med. 2018, 5, 153. [Google Scholar] [CrossRef]
- Shanura Fernando, I.P.; Kim, K.-N.; Kim, D.; Jeon, Y.-J. Algal polysaccharides: Potential bioactive substances for cosmeceutical applications. Crit. Rev. Biotechnol. 2019, 39, 99–113. [Google Scholar] [CrossRef]
- Kain (Jones), J.M.; Dawes, C.P. Useful European seaweeds: Past hopes and present cultivation. Hydrobiologia 1987, 151–152, 173–181. [Google Scholar] [CrossRef]
- Glicksman, M. Utilization of seaweed hydrocolloids in the food industry. In Twelfth International Seaweed Symposium; Ragan, M.A., Bird, C.J., Eds.; Springer: Dordrecht, The Netherlands, 1987; pp. 31–47. [Google Scholar]
- Melo, R.A. Exploração dos Recursos Algológicos em Portugal. In Fragmentos em Ecologia; Martins-Loução, A., Ed.; FCUL e Livraria Escolar Editora: Lisbon, Portugal, 2002; pp. 45–65. [Google Scholar]
- Bogacz-Radomska, L.; Harasym, J.; Piwowar, A. Commercialization aspects of carotenoids. In Carotenoids: Properties, Processing and Applications; Elsevier: Amsterdam, The Netherlands, 2020; pp. 327–357. [Google Scholar]
- Cotas, J.; Leandro, A.; Pacheco, D.; Gonçalves, A.M.M.; Pereira, L. A Comprehensive Review of the Nutraceutical and Therapeutic Applications of Red Seaweeds (Rhodophyta). Life 2020, 10, 19. [Google Scholar] [CrossRef]
- Vílchez, C.; Forján, E.; Cuaresma, M.; Bédmar, F.; Garbayo, I.; Vega, J.M. Marine Carotenoids: Biological Functions and Commercial Applications. Mar. Drugs 2011, 9, 319–333. [Google Scholar] [CrossRef]
- Catarina Guedes, A.; Barbosa, C.R.; Amaro, H.M.; Pereira, C.I.; Xavier Malcata, F. Microalgal and cyanobacterial cell extracts for use as natural antibacterial additives against food pathogens. Int. J. Food Sci. Technol. 2011, 46, 862–870. [Google Scholar] [CrossRef]
- Pangestuti, R.; Kim, S.-K. Biological activities and health benefit effects of natural pigments derived from marine algae. J. Funct. Foods 2011, 3, 255–266. [Google Scholar] [CrossRef]
- European Comission EU Novel Food Catalogue. Available online: https://ec.europa.eu/food/safety/novel_food/catalogue/search/public/index.cfm (accessed on 1 March 2022).
- Mouritsen, O.G.; Dawczynski, C.; Duelund, L.; Jahreis, G.; Vetter, W.; Schröder, M. On the human consumption of the red seaweed dulse (Palmaria palmata (L.) Weber & Mohr). J. Appl. Phycol. 2013, 25, 1777–1791. [Google Scholar] [CrossRef]
- Hallsson, S. The uses of seaweeds in iceland. In Proceedings of the Fourth International Seaweed Symposium, Biarritz, France, 18–24 September 1961. [Google Scholar]
- Johnston, H. The Biological and Economic Importance of Algae, Part 2. Tuatara 1966, 14, 4–6. [Google Scholar]
- Klnc, B.; Cirik, S.; Turan, G.; Tekogul, H.; Koru, E. Seaweeds for Food and Industrial Applications. In Food Industry; InTech: London, UK, 2013; pp. 735–748. [Google Scholar]
- Haroun, R.; Gil-Rodríguez, M.C.; Neto, A.I.; Machín-Sánchez, M.; Viera-Rodríguez, M.A. A review of current uses and potential biotechnological applications of seaweeds from the Macaronesian region (Central-East Atlantic Ocean). J. Appl. Phycol. 2019, 31, 3777–3790. [Google Scholar] [CrossRef]
- Kraan, S.; Lyons, H.J.; Steele, S.; Guiry, M. How Seaweed Is Used in Ireland: The Seaweed of Ireland’s Coastline; An Chomhairle Oidhreachta The Heritage Council: Kilkenny, Ireland, 2010. [Google Scholar]
- Pereira, L. Seaweed Flora of the European North Atlantic and Mediterranean. In Hb25_Springer Handbook of Marine Biotechnology; Springer: Berlin/Heidelberg, Germany, 2015; pp. 65–178. [Google Scholar] [CrossRef]
- Teas, J.; Pino, S.; Critchley, A.; Braverman, L.E. Variability of iodine content in common commercially available edible seaweeds. Thyroid 2004, 14, 836–841. [Google Scholar] [CrossRef] [PubMed]
- McDowell, L.R. Iodine. In Minerals in Animal and Human Nutrition; McDowell, L.R., Ed.; Academic Press: San Diego, CA, USA, 1992; pp. 224–245. [Google Scholar]
- Pérez-Lloréns, J.L.; Mouritsen, O.G.; Rhatigan, P.; Cornish, M.L.; Critchley, A.T. Seaweeds in mythology, folklore, poetry, and life. J. Appl. Phycol. 2020, 32, 3157–3182. [Google Scholar] [CrossRef]
- (BioMara) The Importance of Seaweed across the Ages. Available online: http://www.biomara.org/understanding-seaweed/the-importance-of-seaweed-across-the-ages.html (accessed on 1 March 2021).
- Baldwin, J.R. Working with Seaweed in North-West Sutherland. In The Province of Strathnaver; Baldwin, J.R., Thomson, W.P.L., Eds.; Scottish Society for Northern Studies: Edinburgh, Scotland, 2000; pp. 116–141. [Google Scholar]
- (Nordic Seaweed Tang) Nordic Seaweed: Nutrition in Seaweed. Available online: http://nordicseaweed.com/engelsk/eng_nutrition.html (accessed on 5 April 2021).
- Lloréns, J.L.P.; Carrero, I.H.; Oñate, J.J.V.; Murillo, F.G.B.; González, Á.L. ¿Las Algas Se Comen? Un Periplo por La Biología, La Historia, Las Curiosidades y La Gastronomía (Ceimar), 1st ed.; Servicio de Publicaciones de la Universidad de Cádiz: Cádiz, Spain, 2016. [Google Scholar]
- Patarra, A.R.F. Culture Studies of Economically Important Seaweeds; University of Azores: Ponta Delgada, Portugal, 2018. [Google Scholar]
- (Netalgae) Overview of the Seaweed Industry by Country Ireland, France, Norway, Portugal, Spain, United Kingdom; Neatalgae Report; European Union, European Regional Development Fund (EDRF), Nordland County Council: Nordland, Norway, 2012.
- Patarra, R.F.; Paiva, L.; Neto, A.I.; Lima, E.; Baptista, J. Nutritional value of selected macroalgae. J. Appl. Phycol. 2011, 23, 205–208. [Google Scholar] [CrossRef]
- Neto, A.I.; Tittley, I.; Raposeiro, P.M. Flora Marinha do Litoral dos Açores = Rocky Shore Marine Flora of the Azores; Secretaria Regional do Ambiente e do Mar: Horta, Portugal, 2005. [Google Scholar]
- Sousa-Pinto, I. The Seaweed Resources of Portugal. In Seaweed Resources of the World; JICA: Tokyo, Japan, 1998; pp. 176–184. [Google Scholar]
- Michanek, G. Seaweed Resources of the Ocean, 138th–, 139th ed.; Food and Agriculture Organization of the United Nations: Rome, Italy, 1975; Volume 138. [Google Scholar]
- Seaver, K.A. The Last Vikings: The Epic Story of the Great Norse Voyages; I.B.Tauris & Co., Ltd.: London, UK, 2010. [Google Scholar]
- Mouritsen, O.G.; Rhatigan, P.; Cornish, M.L.; Critchley, A.T.; Pérez-Lloréns, J.L. Saved by seaweeds: Phyconomic contributions in times of crises. J. Appl. Phycol. 2021, 33, 443–458. [Google Scholar] [CrossRef] [PubMed]
- Dickens, C.; Robertson, J.; Dickens, C. The Purple Shore. Househ. Words 1856, XIV, 391–395. [Google Scholar]
- (Mara Seaweed) A Short History of Dulse Seaweed. Available online: https://maraseaweed.com/blogs/news/a-short-history-of-dulse-seaweed (accessed on 28 March 2021).
- Pereira, L. MACOI, Portuguese Seaweeds Website; World-wide Electronic Publication, MARE UC, Department of Life Sciences, University of Coimbra: Coimbra, Portugal, 2021; Available online: http://macoi.ci.uc.pt/spec_list_detail.php?spec_id=125 (accessed on 2 April 2021).
- McHugh, D.J. Seaweeds uses as Human Foods. In A Guide to the Seaweed Industry; FAO: Rome, Italy, 2003; p. 105. [Google Scholar]
- Walsh, M.; Watson, L. A Market Analysis towards the Further Development of Seaweed Aquaculture in Ireland; Irish Sea Fisheries Board: Dublin, Ireland, 2011; Volume 1. [Google Scholar]
- Gaspar, R.; Pereira, L.; Sousa-Pinto, I. The seaweed resources of Portugal. Bot. Mar. 2019, 62, 499–525. [Google Scholar] [CrossRef]
- Palmieri, N.; Forleo, M.B. The potential of edible seaweed within the western diet. A segmentation of Italian consumers. Int. J. Gastron. Food Sci. 2020, 20, 100202. [Google Scholar] [CrossRef]
- Lentini, F.; Venza, F. Wild food plants of popular use in Sicily. J. Ethnobiol. Ethnomed. 2007, 3, 15. [Google Scholar] [CrossRef]
- Cappellani, S. Il Mauru Ossia Alghe Rosse Commestibili Nella Sicilia Centro-Orientale; CITEM: Metro Manila, Philippines, 1960. [Google Scholar]
- Subba Rao, P.V.; Ganesan, K.; Suresh Kumar, K. Seaweeds as a Human Diet: An Emerging Trend in the New Millennium. Adv. Appl. Phycol. 2007, 1, 85–96. [Google Scholar] [CrossRef]
- Ferdouse, F.; Lovstad Holdt, S.; Smith, R.; Murúa, P.; Yang, Z.; (FAO). The global status of seaweed production, trade and utilization. Globefish Res. Programme 2018, 124, 120. [Google Scholar]
- Calado, R.M. Macrobiótica Como e Porquê; Grafitécnica: Lisbon, Portugal, 1976. [Google Scholar]
- Kushi, L.H.; Cunningham, J.E.; Hebert, J.R.; Lerman, R.H.; Bandera, E.V.; Teas, J. The Macrobiotic Diet in Cancer. J. Nutr. 2001, 131, 3056S–3064S. [Google Scholar] [CrossRef]
- Varatojo, F. As Algas-Os Legumes do Mar. Available online: https://www.institutomacrobiotico.com/pt-pt/imp/artigos/alimentacao/algas (accessed on 1 March 2021).
- Fralick, R.A.; Andrade, F. The growth, reproduction, harvesting and management of Pterocladia pinnata (Rhodophyceae) in the Azores, Portugal. In Proceedings of the 10th International Seaweed Symposium, Goteborg, Sweden, 11–15 August 1980; Levring, T., Ed.; Walter de Gruyter & Co.: Gothenburg, Sweden, 1981; pp. 289–295. [Google Scholar]
- Fralick, R.A. Algas agarófitas. In Exposição Oral Sobre Desenvolvimento das Algas nos Açores; Presidência do Governo, Departamento Regional de Estudos e Planeamento, Região Autónoma dos Açores: Horta, Portugal, 1980. [Google Scholar]
- Palminha, F. Exploração e utilização de Algas Marinhas na Plataforma Portuguesa e nas Ilhas do Arquipélago dos Açores. Junta Nac. Fom. Das Pescas 1971, 7, 25–36. [Google Scholar]
- Palminha, F.; Melo, R.; Santos, R. A existência de Gelidium sesquipedale (Clem.) Born et Thur. na costa sul do Algarve. I. Distribuição local. Bol. INIP. 1982, 8, 93–105. [Google Scholar]
- Palminha, F.; Melo, R.; Santos, R. A existência de Gelidium sesquipedale (Clem.) Born et Thur. na costa sul do Algarve. II. Biomassa total. Bol. INIP. 1985, 13, 77–91. [Google Scholar]
- Santos, R.; Duarte, P. Marine plant harvest in Portugal. J. Appl. Phycol. 1991, 3, 11–18. [Google Scholar] [CrossRef]
- Marshall, S.N.; Newton, L.; Orr, A.P. A Study of Certain British Seaweeds and Their Utilisation in the Preparation of Agar., 1st ed.; His Majesty’s Stationary Office: London, UK, 1949. [Google Scholar]
- Patarra, R.F.; Iha, C.; Pereira, L.; Neto, A.I. Concise review of the species Pterocladiella capillacea (S.G. Gmelin) Santelices & Hommersand. J. Appl. Phycol. 2020, 32, 787–808. [Google Scholar]
- Burlew, J.S. Algal Culture from Laboratory to Pilot Plant. AIBS Bull. 1953, 3, 11. [Google Scholar] [CrossRef]
- Powell, R.C.; Nevels, E.M.; McDowell, M.E. Algae Feeding in Humans. J. Nutr. 1961, 75, 7–12. [Google Scholar] [CrossRef]
- Melo, R.A. Gelidium commercial exploitation: Natural resources and cultivation. J. Appl. Phycol. 1998, 10, 303–314. [Google Scholar] [CrossRef]
- Gao, F.Z.; Ge, B.S.; Xiang, W.Z.; Qin, S. Development of microalgal industries in the past 60 years due to biotechnological research in China: A review. Sci. Sin. Vitae 2021, 51, 26–39. [Google Scholar] [CrossRef]
- Gantar, M.; Svirčev, Z. Microalgae and cyanobacteria: Food for thought. J. Phycol. 2008, 44, 260–268. [Google Scholar] [CrossRef]
- Takenaka, H.; Yamaguchi, Y.; Sakaki, S.; Watarai, K.; Tanaka, N.; Hori, M.; Seki, H.; Tsuchida, M.; Yamada, A.; Nishimori, T.; et al. Safety evaluation of Nostoc flagelliforme (nostocales, cyanophyceae) as a potential food. Food Chem. Toxicol. 1998, 36, 1073–1077. [Google Scholar] [CrossRef]
- Aaronson, S. A role for algae as human food in antiquity. Food Foodways 1986, 1, 311–315. [Google Scholar] [CrossRef]
- Farrar, W.V. Tecuitlatl; A Glimpse of Aztec Food Technology. Nature 1966, 211, 341–342. [Google Scholar] [CrossRef]
- Abdulqader, G.; Barsanti, L.; Tredici, M.R. Harvest of Arthrospira platensis from Lake Kossorom (Chad) and its household usage among the Kanembu. J. Appl. Phycol. 2000, 12, 493–498. [Google Scholar] [CrossRef]
- Ciferri, O. Spirulina, the edible microorganism. Microbiol. Rev. 1983, 47, 551–578. [Google Scholar] [CrossRef] [PubMed]
- Piccolo, A. Spirulina A Livehood and A Business Venture; FAO/IOC: Rome, Italy, 2012; Volume 45. [Google Scholar]
- Chacón-Lee, T.L.; González-Mariño, G.E. Microalgae for “Healthy” Foods-Possibilities and Challenges. Compr. Rev. Food Sci. Food Saf. 2010, 9, 655–675. [Google Scholar] [CrossRef] [PubMed]
- Sathasivam, R.; Radhakrishnan, R.; Hashem, A.; Abd_Allah, E.F. Microalgae metabolites: A rich source for food and medicine. Saudi J. Biol. Sci. 2019, 26, 709–722. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.M.; Allsopp, P.J.; Magee, P.J.; Gill, C.I.; Nitecki, S.; Strain, C.R.; McSorley, E.M. Seaweed and human health. Nutr. Rev. 2014, 72, 205–216. [Google Scholar] [CrossRef]
- Enzing, C.; Ploeg, M.; Barbosa, M.; Sijtsma, L. Microalgae-Based Products for the Food and Feed Sector: An Outlook for Europe; Vigani, M., Parisi, C., Cerezo, E.R., Eds.; Publications Office of the European Union: Luxembourg, 2014. [Google Scholar]
- Santillan, C. Mass production of Spirulina. Experientia 1982, 38, 40–43. [Google Scholar] [CrossRef]
- Geoghegan, M.J. Unicellular Algae as a Source of Food. Nature 1951, 168, 426–427. [Google Scholar] [CrossRef]
- Algae as a Source of Human Food. Nutr. Rev. 1959, 17, 238–240. [CrossRef]
- Soeder, C.J. Zur Verwendung von Mikroalgen Für Ernährungszwecke. Naturwissenschaften 1976, 63, 131–138. [Google Scholar] [CrossRef]
- Krauss, R.W. Mass Culture of Algae for Food and Other Organic Compounds. Am. J. Bot. 1962, 49, 425. [Google Scholar] [CrossRef]
- Kihlberg, R. The Microbe as a Source of Food. Annu. Rev. Microbiol. 1972, 26, 427–466. [Google Scholar] [CrossRef]
- Soeder, C.J. Massive cultivation of microalgae: Results and prospects. Hydrobiologia 1980, 72, 197–209. [Google Scholar] [CrossRef]
- Tamiya, H. Growing Chlorella for Food and Feed. In Proceedings of the World Symposium on Applied Solar Energy, Phoenix, AZ, USA; 1955; pp. 231–241. [Google Scholar]
- Tamura, E.; Baba, H.; Tamura, A.; Kobatake, Y. Nutritional Studies on Chlorella. Jpn. J. Nutr. Diet. 1959, 17, 197–198. [Google Scholar] [CrossRef]
- Tamura, E.; Matsuno, N.; Morimoto, K. Nutritional Studies on Chlorella VII Absorption Experiment of Dried Chlorella on Human Subject. Jpn. J. Nutr. Diet 1959, 17, 91–93. [Google Scholar] [CrossRef][Green Version]
- Tamura, E.; Morimoto, K.; Tsuchida, M. Nutritional Studies on Chlorella II. Practical Use of Chlorella on Cooking. Jpn. J. Nutr. Diet 1959, 17, 21–22. [Google Scholar] [CrossRef][Green Version]
- Soeder, C.J.; Pabst, W. Gesichtspunkte Für Die Verwendung von Mikroalgen in Der Ernährung von Mensch Und Tier 1. Ber. Dtsch. Bot. Ges. 1970, 83, 607–625. [Google Scholar]
- Space Medicine. Scott. Med. J. 1959, 4, 462–465. [CrossRef]
- Helvey, T.C. Nutrition in Space and Related Waste Problems. In Proceedings of the Conference on Nutrition in Space and Related Waste problems, Universtiy of South Florida, Tampa, FL, USA, 27–30 April 1964. [Google Scholar]
- Kondrat’ev, I.I.; Bychkov, V.P.; Ushakov, A.S.; Boĭko, N.N.; Kliushkina, N.S.; Abaturova, E.A.; Terpilovskiĭ, A.M.; Korneeva, N.A.; Beliakova, M.I.; Kasatkina, A.G. The aus der Alge Dunaliella of dried biomass of unicellular algae in human food rations. Vopr. Pitan. 1966, 25, 9–14. [Google Scholar]
- Kondrat’ev, I.I.; Bychkov, V.P.; Ushakov, A.S.; Boĭko, N.N.; Kliushkina, N.S.; Abaturova, E.A.; Terpilovskiĭ, A.M.; Korneeva, N.W.; Beliakova, M.I.; Vorob’eva, E.S.; et al. The use of 150 g dry blomass of unicellular algae in human diet. Vopr. Pitan. 1966, 25, 14–19. [Google Scholar]
- Kofrányi, E.; Jekat, F. Zur Bestimmung der biologischen Wertigkeit von Nahrungsproteinen, XII. Die Mischung von Ei mit Reis, Mais, Soja, Algen. Hoppe-Seyler´S Z. Physiol. Chem. 1967, 348, 84–88. [Google Scholar] [CrossRef]
- Müller-Wecker, H.; Kofrányi, E. Zur Bestimmung der biologischen Wertigkeit von Nahrungsproteinen, XVIII. Einzeller als zusätzliche Nahrungsquelle. Hoppe-Seyler´S Z. Physiol. Chem. 1973, 354, 1034–1042. [Google Scholar] [CrossRef]
- Fink, H.; Herold, E. Über den Biologischen Wert der Einzelligen Grünalge Scenedesmus Obliquus—Frisch Und Verschieden Getrocknet—Und Ihre Diätetischen Und Therapeutischen Eigenschaften; Schlie, I., Ed.; VS Verlag für Sozialwissenschaften: Wiesbaden, Germany, 1963; ISBN 978-3-663-06425-1. [Google Scholar]
- Berkhoff, G.; Kramer, G. 156a. Untersuchungen über den Einfluß der Algennahrung auf den Eiweißhaushalt bei Intensivpflegefällen. Langenbecks Arch. Chir. 1971, 329, 523. [Google Scholar] [CrossRef][Green Version]
- Shelef, G.; Moraine, R.; Oron, G.; Binsack, R.; Heussler, P.; Castillo, J.; Merino, F.; Becker, E.W.; Payer, H.J.; Pithakpol, B.; et al. Microalgae for food and feed. In Proceedings of the German-Israeli Workshop, Neuherberg, Germany, 17–18 October 1977; Soeder, C.J., Binsack, R., Eds.; FAO: Rome, Italy, 1979; p. 300, ISBN 978-3-510-47009-9. [Google Scholar]
- Ahsan, M.; Habib, B.; Parvin, M.; Huntington, T.C.; Hasan, M. A Review on Culture, Production and Use of Spirulina as Food for Humans and Feeds for Domestic Animals and Fish; FAO: Rome, Italy, 2008; Volume 1034, ISBN 9789251061060. [Google Scholar]
- Oliveira, J.C. Utilisation of Macro-Algae as Vegetables and Food Additives in Portugal; Instituto Nacional de Investigação das Pescas: Lisbon, Portugal, 1992; p. 7. [Google Scholar]
- AstaReal AB The World´s Leading Producer and Innovator of Natural Astaxanthin. Available online: http://www.astareal.se/about-us (accessed on 20 April 2021).
- Fuji Chemical Industries Co. Satisfying the Global Demand for Natural Astaxanthin. Available online: https://www.fujichemical.co.jp/english/whatsnew/archive/200609/whatsnew.html (accessed on 10 April 2021).
- Indergaard, M.; Minsaas, J. Animal and human nutrition. In Seaweed Resources in Europe: Uses and Potential; Guiry, M.D., Blunden, G., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 1991; pp. 21–64. ISBN 0 471 929 476. [Google Scholar]
- Blake, W. Jerusalem the Emanation of the Giant Albion; William Blake: London, UK, 1821. [Google Scholar]
- McIrvine, D. An Erne in Erin: William Blake Depicts a “Sea Eagle” and Dillisk from County Antrim’s ‘Giants Caus[e]way’ in Plate 78 of *Jerusalem*, and Japanese Women Abalone-Diving in Plate 89; 2019; Volume 1. Available online: https://www.academia.edu/41485060/An_Erne_in_Erin_William_Blake_Depicts_a_Sea_Eagle_and_Dillisk_from_County_Antrims_Giants_caus_e_way_in_Plate_78_of_Jerusalem_and_Japanese_Women_Abalone_Diving_in_Plate_89 (accessed on 10 April 2021).
- Kelly, F.C. Iodine in Medicine and Pharmacy Since its Discovery-1811–1961. Proc. R. Soc. Med. 1961, 54, 831–836. [Google Scholar] [CrossRef][Green Version]
- Instituto Nacional de Estatística. Estatísticas da Pesca Continente e Ilhas Adjacentes. Junta Nac. Fom. Pescas 1973, 26–36. [Google Scholar]
- CEVA Edible Seaweed and Microalgae—Regulatory Status in France and Europe; 2019 Update; CEVA: Pleubian, France, 2019.
- Wells, M.L.; Potin, P.; Craigie, J.S.; Raven, J.A.; Merchant, S.S.; Helliwell, K.E.; Smith, A.G.; Camire, M.E.; Brawley, S.H. Algae as nutritional and functional food sources: Revisiting our understanding. J. Appl. Phycol. 2017, 29, 949–982. [Google Scholar] [CrossRef]
- Pérez-Lloréns, J.L. Microalgae: From staple foodstuff to avant-garde cuisine. Int. J. Gastron. Food Sci. 2020, 21, 100221. [Google Scholar] [CrossRef]
- Statista. Retail Sales Value of Fungi/Algae Meat Substitutes in Western Europe in 2015 and 2018, with a Forecast for 2019 and 2022. Available online: https://www.statista.com/statistics/1076190/retail-sales-value-of-fungi-algae-meat-substitutes-in-western-europe/ (accessed on 10 April 2021).
- Statista. Retail Sales Value of Fungi/Algae Meat Substitutes in Western Europe in 2018, by Category. Available online: https://www.statista.com/statistics/1076194/retail-sales-value-of-fungi-algae-meat-substitutes-in-western-europe/i-algae-meat-substitutes-in-western-europe/ (accessed on 10 April 2021).
- Lucas, S.; Gouin, S.; Lesueur, M. Seaweed Consumption and Label Preferences in France. Mar. Resour. Econ. 2019, 34, 143–162. [Google Scholar] [CrossRef]
- Mouritsen, O.G.; Williams, L.; Bjerregaard, R.; Duelund, L. Seaweeds for umami flavour in the New Nordic Cuisine. Flavour 2012, 1, 4. [Google Scholar] [CrossRef]
- Roleda, M.Y.; Marfaing, H.; Desnica, N.; Jónsdóttir, R.; Skjermo, J.; Rebours, C.; Nitschke, U. Variations in polyphenol and heavy metal contents of wild-harvested and cultivated seaweed bulk biomass: Health risk assessment and implication for food applications. Food Control 2019, 95, 121–134. [Google Scholar] [CrossRef]
- National Food Institute Denmark; Sá Monteiro, M.; Sloth, J.; Holdt, S.; Hansen, M. Analysis and Risk Assessment of Seaweed. EFSA J. 2019, 17, e170915. [Google Scholar] [CrossRef]
- (ALGAplus) Fava-Do-Mar Desidratada, 100g. Available online: https://algaplus.lojasonlinectt.pt/product/fava-do-mar-desidratada-100g (accessed on 10 May 2021).
- Li, X.; Li, J.; Wang, Y.; Fu, L.; Fu, Y.; Li, B.; Jiao, B. Aquaculture Industry in China: Current State, Challenges, and Outlook. Rev. Fish. Sci. 2011, 19, 187–200. [Google Scholar] [CrossRef]
- Statista. Estimated Value of Algae Market Worldwide from 2016 to 2023, by Region. Available online: https://www.statista.com/statistics/1032615/global-market-value-of-algae-by-region/ (accessed on 10 April 2021).
- Turan, G.; Cırık, S. Sea Vegetables. In Vegetables—Importance of Quality Vegetables to Human Health; InTech: London, UK, 2018. [Google Scholar]
- (MICHELIN Guide UK Editorial Team) Explore the World of the MICHELIN Guide. Available online: https://guide.michelin.com/en (accessed on 24 May 2021).
- Goulding, M.; Seeding the Ocean: Inside a Michelin-Starred Chef’s Revolutionary Quest to Harvest Rice From the Sea. Available online: https://time.com/5926780/chef-angel-leon-sea-rice/ (accessed on 10 April 2021).
- Rodrigues, J. Projecto Matéria. Available online: https://www.projectomateria.pt (accessed on 1 February 2022).
- Rumin, J.; Nicolau, E.; Gonçalves de Oliveira Junior, R.; Fuentes-Grünewald, C.; Picot, L. Analysis of Scientific Research Driving Microalgae Market Opportunities in Europe. Mar. Drugs 2020, 18, 264. [Google Scholar] [CrossRef] [PubMed]
- García, J.L.; de Vicente, M.; Galán, B. Microalgae, old sustainable food and fashion nutraceuticals. Microb. Biotechnol. 2017, 10, 1017–1024. [Google Scholar] [CrossRef] [PubMed]
- PwC. Vitamins & Dietary Supplements Market trends—Overview. Euromonitor International 2019, Press. Available online: https://www.pwc.com/it/it/publications/assets/docs/Vitamins-Dietary-Supplements-Market-Overview.pdf (accessed on 25 May 2022).
- Statista. Value of the Dietary Supplements Market in Europe in 2015 and 2020, by Region. Available online: https://www.statista.com/statistics/589374/dietary-supplements-market-value-europe/ (accessed on 10 April 2021).
- Statista. Retail Sales Forecast of Vitamin and Dietary Supplements in Europe from 2010 to 2020. Available online: https://www.statista.com/statistics/646795/retail-sales-forecast-of-supplements-european-union-eu/ (accessed on 10 April 2021).
- Schüler, L.; Greque de Morais, E.; Trovão, M.; Machado, A.; Carvalho, B.; Carneiro, M.; Maia, I.; Soares, M.; Duarte, P.; Barros, A.; et al. Isolation and Characterization of Novel Chlorella vulgaris Mutants With Low Chlorophyll and Improved Protein Contents for Food Applications. Front. Bioeng. Biotechnol. 2020, 8, 469. [Google Scholar] [CrossRef] [PubMed]
- Molino, A.; Iovine, A.; Casella, P.; Mehariya, S.; Chianese, S.; Cerbone, A.; Rimauro, J.; Musmarra, D. Microalgae Characterization for Consolidated and New Application in Human Food, Animal Feed and Nutraceuticals. Int. J. Environ. Res. Public Health 2018, 15, 2436. [Google Scholar] [CrossRef]
- Niccolai, A.; Chini Zittelli, G.; Rodolfi, L.; Biondi, N.; Tredici, M.R. Microalgae of interest as food source: Biochemical composition and digestibility. Algal Res. 2019, 42, 101617. [Google Scholar] [CrossRef]
- (Foodfrog) Astaxanthin Powder [Vacuumed] [1.2%] [5%] & [10%] Haematococcus pluvialis without Additives, Laboratory Tested. Available online: https://www.amazon.de/-/en/Astaxanthin-Haematococcus-laboratory-Resealable-03-01-2020/dp/B079G735CJ/ref=sr_1_6?dchild=1&keywords=Haematococcus&qid=1619540791&s=drugstore&sr=1-6 (accessed on 22 April 2021).
- (Vita World) Astaxantina 8mg 60 Cápsulas Vita World Producción en Farmacia en Alemania—Antioxidante—Haematococcus Pluvialis. Available online: https://www.amazon.es/Astaxantina-Cápsulas-Vita-World-Producción/dp/B0183YGDC8/ref=sr_1_1?__mk_es_ES=ÅMÅŽÕÑ&dchild=1&keywords=Haematococcus+pluvialis&qid=1619546645&sr=8-1 (accessed on 22 April 2021).
- (Igennus Healthcare) Nutrition Pure & Essential Astaxanthin Complex, 42 mg Astapure Providing 4 mg H. Pluvialis Astaxanthin, 90 Vegan Capsules. Available online: https://www.amazon.co.uk/Essential-Astaxanthin-Astapure-Delivering-Capsules/dp/B07BGHKG77/ref=sr_1_7?dchild=1&keywords=Haematococcus+pluvialis&qid=1619543962&s=drugstore&sr=1-7 (accessed on 22 April 2021).
- (Ekopura) Vegan Oméga 3—Huile d’Algues, 90 Capsules (250mg DHA/Capsule). Available online: https://www.amazon.fr/Vegan-Oméga-dAlgues-Capsules-Capsule/dp/B076PVW2LV/ref=sr_1_9?__mk_fr_FR=ÅMÅŽÕÑ&dchild=1&keywords=Haematococcus+pluvialis&qid=1619543956&sr=8-9 (accessed on 22 April 2021).
- NOVERG Antioxydant|Vitamines C & E, Zinc, CoEnzyme Q10, Sélénium, Pépins de Raisin OPC, Thé Vert, Dunaliella|Anti-Age & Belle Peau|Vegan|90 Gélules|Cure de 45 Jours|Fabriqué en France. Available online: https://www.amazon.fr/Antioxydant-Vitamines-CoEnzyme-Sélénium-Dunaliella/dp/B08RHY3J4X/ref=sr_1_10?__mk_fr_FR=ÅMÅŽÕÑ&dchild=1&keywords=Dunaliella&qid=1619543512&sr=8-10 (accessed on 22 April 2021).
- Anastore Vitamine A (Rétinol) * 16 mg/60 Kapseln * Trockenextrakt aus der Alge Dunaliella, zu 30 % aus β-Karotin titriert * Augen, Haut, Immun, Vegan. Available online: https://www.amazon.de/-/en/DN65/dp/B01IR7HDPM/ref=sr_1_3?dchild=1&keywords=Dunaliella&qid=1619540972&s=drugstore&sr=1-3 (accessed on 22 April 2021).
- Amazon Microalgae Powders. Available online: https://www.amazon.de/s?k=mikroalgaen+pulver&ref=nb_sb_noss (accessed on 22 April 2021).
- VeganBio Spirulina Products. Available online: https://www.veganobio.it/search-results/?action=search&productids=1559,4227,2355,2163,5669,463,2559,5578,1463,813,5824,2332,2555,2562,2558,5064,2575,5881,5828,4783,4775,5458 (accessed on 27 April 2021).
- Biosklep Mikroalgen Pulver Products. Available online: https://biosklep.com.pl/pl/searchquery/spirulina/1/phot/5?url=spirulina (accessed on 27 April 2021).
- Planeta Huerto Mikroalgen Pulver Products. Available online: https://www.planetahuerto.es/articulos-buscar?k=spirulina (accessed on 21 February 2021).
- Statista. Which of the Following Superfoods Do You Regularly Buy Because of Their Special Effect? Available online: https://www.statista.com/statistics/788851/superfoods-purchase-germany/ (accessed on 10 April 2021).
- Mattisson Chlorella sorokiniana Product of. Available online: https://www.mattisson.nl/product/absolute-chlorella-poeder-nederlands/ (accessed on 21 February 2021).
- MySuperfoods Marine Phytoplankton Powder (100 Grams), MySuperFoods, Purest Food on Earth, Cultivated from The Deep Sea, Rich in Micronutrients, Add to Juices, Smoothies, Shakes. Available online: https://www.amazon.nl/-/en/dp/B00M8F6QP8/ref=pd_sim_1?pd_rd_w=EerIM&pf_rd_p=14f79929-97cc-4510-b8a0-227891619eb7&pf_rd_r=9S9VQF5ZAYZ3WM8PEAMT&pd_rd_r=279be38e-7370-43d3-af05-3d08ddf36662&pd_rd_wg=ARZAK&pd_rd_i=B00M8F6QP8&psc=1 (accessed on 27 April 2021).
- Algova Algova Nannochloropsis Freeze Dried Phytoplankton—Food for Artemia, Daphnia, Whederanimals, Mussels, Shrimp Corals, Copepodes High Vitamin, Fatty Acid & Mineral Content. Available online: https://www.amazon.de/-/en/Nannochloropsis-Freeze-Dried-Phytoplankton-Artemia/dp/B00TVARE3G/ref=sr_1_3?dchild=1&keywords=nannochloropsis&qid=1619543022&sr=8-3 (accessed on 27 April 2021).
- Cai, J.; Lovatelli, A.; Gamarro, E.G.; Geehan, J.; Lucente, D.; Mair, G.; Miao, W.; Reantaso, M.; Roubach, R.; Yuan, X.; et al. Seaweeds and Microalgae: An Overview for Unlocking Their Potential in Global Aquaculture Development; FAO: Rome, Italy, 2021; ISBN 978-92-5-134710-2. [Google Scholar]
- Dos Santos Fernandes de Araujo, R. Brief on Algae Biomass Production; Lusser, M., Sanchez Lopez, J., Avraamides, M., Eds.; Publications Office of the European Union: Luxembourg, 2019; ISBN 978-92-76-12271-5. [Google Scholar]
- FAO. Fishery and Aquaculture Statistics 2019/FAO annuaire. Statistiques des pêches et de l’aquaculture 2019/FAO anuario. Estadísticas de pesca y acuicultura 2019; FAO: Rome, Italy, 2021. [Google Scholar] [CrossRef]
- FAO. Global Production Statistics Database. Available online: http://www.fao.org/fishery/topic/16140/en (accessed on 10 April 2021).
- Chopin, T.; Buschmann, A.H.; Halling, C.; Troell, M.; Kautsky, N.; Neori, A.; Kraemer, G.P.; Zertuche-González, J.A.; Yarish, C.; Neefus, C. Integrating Seaweeds into Marine Aquaculture Systems: A Key toward Sustainability. J. Phycol. 2001, 37, 975–986. [Google Scholar] [CrossRef]
- Bronswijk, L.; Vlottes, M.; Draisma, M.; Brouwers, E.; van Baelen, F. D4.1.1 Study on Existing Market for Algal Food Applications Part A: Seaweed. North Sea Farm Foundation: The Hague, Amsterdam, 2019. [Google Scholar]
- Organic Monitor. The European Market for Sea Vegetables; Report prepared for Bord Iascaigh Mhara (BIM). 2014. Available online: https://bim.ie/wp-content/uploads/2021/02/The,European,Market,for,Sea,Vegetables,-,2015.pdf (accessed on 10 April 2021).
- Vincent, A.; Stanley, A.; Ring, J. Hidden Champion of the Ocean: Seaweed as a Growth Engine for a Sustainable European Future: Seaweed for Europe. 2020. Available online: https://www.seaweedeurope.com/wp-content/uploads/2020/10/Seaweed_for_Europe-Hidden_Champion_of_the_ocean-Report.pdf (accessed on 10 April 2021).
- Paladin Maionese Do Mar. Available online: https://www.paladin.pt/pt-pt/produto/maionese-mar-225ml (accessed on 11 February 2022).
- Le Bras, Q.; Ritter, L.; Fasquel, D.; Lesueur, M.; Lucas, S. Etude de la consommation des algues alimentaires en France; Programme IDEALG Phase 1. Etude nationale. [Rapport Technique]; Agrocampus Ouest: Rennes, France, 2014. [Google Scholar]
- Simoes, J.G.; Hidalgo, C.A. Seaweeds and Other Algae: Not Fit for Human Consumption, Fresh, Chilled, Frozen or Dried, Whether or Not Ground. The Economic Complexity Observatory. Datawheel. Available online: https://oec.world/en/profile/hs/seaweeds-and-other-algae-not-fit-for-human-consumption-fresh-chilled-frozen-or-dried-whether-or-not-ground?countryComparisonMeasureSelector=Trade%20Value&countryComparisonGeoSelector=eu (accessed on 10 April 2021).
- Notpla Notpla Limited—We Make Packaging Disappear. Available online: https://www.notpla.com (accessed on 25 April 2021).
- Shen, Y.; Yuan, W.; Pei, Z.J.; Wu, Q.; Mao, E. Microalgae Mass Production Methods. Trans. ASABE 2009, 52, 1275–1287. [Google Scholar] [CrossRef]
- Larsson, C.; Axelsson, L. Bicarbonate uptake and utilization in marine macroalgae. Eur. J. Phycol. 1999, 34, 79–86. [Google Scholar] [CrossRef]
- Lüning, K.; Pang, S. Mass cultivation of seaweeds: Current aspects and approaches. J. Appl. Phycol. 2003, 15, 115–119. [Google Scholar] [CrossRef]
- Hurd, C.L.; Harrison, P.J.; Bischof, K.; Lobban, C.S. Seaweed Ecology and Physiology; Cambridge University Press: Cambridge, UK, 2014; ISBN 1139952013. [Google Scholar]
- Kim, J.K.; Yarish, C.; Hwang, E.K.; Park, M.; Kim, Y. Seaweed aquaculture: Cultivation technologies, challenges and its ecosystem services. ALGAE 2017, 32, 1–13. [Google Scholar] [CrossRef]
- Grobbelaar, J.U. Microalgae mass culture: The constraints of scaling-up. J. Appl. Phycol. 2012, 24, 315–318. [Google Scholar] [CrossRef]
- Borowitzka, M.A. Microalgae for Aquaculture: Opportunities and Constraints. J. Appl. Phycol. 1997, 9, 393–401. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, W.; Chen, L.; Wang, J.; Liu, T. The contamination and control of biological pollutants in mass cultivation of microalgae. Bioresour. Technol. 2013, 128, 745–750. [Google Scholar] [CrossRef]
- Day, J.G.; Gong, Y.; Hu, Q. Microzooplanktonic grazers—A potentially devastating threat to the commercial success of microalgal mass culture. Algal Res. 2017, 27, 356–365. [Google Scholar] [CrossRef]
- Masó, M.; Garcés, E. Harmful microalgae blooms (HAB); problematic and conditions that induce them. Mar. Pollut. Bull. 2006, 53, 620–630. [Google Scholar] [CrossRef]
- Buschmann, A.H.; Retamales, C.A.; Figueroa, C. Ceramialean epiphytism in an intertidal Gracilaria chilensis (Rhodophyta) bed in southern Chile. J. Appl. Phycol. 1997, 9, 129–135. [Google Scholar] [CrossRef]
- Redmond, S.; Green, L.; Yarish, C.; Kim, J.; Neefus, C. New England Seaweed Culture Handbook. Seaweed Cultiv. 2014, 1–92. Available online: https://opencommons.uconn.edu/seagrant_weedcult/1 (accessed on 10 April 2021).
- Patarra, R.F.; Carreiro, A.S.; Lloveras, A.A.; Abreu, M.H.; Buschmann, A.H.; Neto, A.I. Effects of light, temperature and stocking density on Halopteris scoparia growth. J. Appl. Phycol. 2017, 29, 405–411. [Google Scholar] [CrossRef]
- Liu, X.; Bogaert, K.; Engelen, A.H.; Leliaert, F.; Roleda, M.Y.; De Clerck, O. Seaweed reproductive biology: Environmental and genetic controls. Bot. Mar. 2017, 60, 89–108. [Google Scholar] [CrossRef]
- Wang, H.; Lin, A.; Gu, W.; Huan, L.; Gao, S.; Wang, G. The sporulation of the green alga Ulva prolifera is controlled by changes in photosynthetic electron transport chain. Sci. Rep. 2016, 6, 24923. [Google Scholar] [CrossRef]
- Cherry, P.; O’Hara, C.; Magee, P.J.; McSorley, E.M.; Allsopp, P.J. Risks and benefits of consuming edible seaweeds. Nutr. Rev. 2019, 77, 307–329. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, E.; Silva, J.; Mendonça, S.; Abreu, M.; Domingues, M. Lipidomic Approaches towards Deciphering Glycolipids from Microalgae as a Reservoir of Bioactive Lipids. Mar. Drugs 2016, 14, 101. [Google Scholar] [CrossRef] [PubMed]
- Sautier, C.; Trémolières, J.; Billion, J.; Flament, C.; Poivre, R. Valeur alimentaire des algues spirulines chez l’homme. Ann. Nutr. Aliment. 1975, 29, 517–534. [Google Scholar] [PubMed]
- Pokrovskaia, Y.E.I.; Tereshchenko, A.P.; Volynets, V.M. Effect of a Vegetable Diet Including a Biomass of Unicellular Algae on the Excretion and Balance of Mineral Elements (Vegetable Diet, Including 210 g of Dry Chlorella Biomass, Decreases Effect on Calcium and Magnesium Assimilation to Produce Insignificant. Kosm. Biol. I Meditsina 1968, 2, 78–81. [Google Scholar]
- Pereira, H.; Silva, J.; Santos, T.; Gangadhar, K.N.; Raposo, A.; Nunes, C.; Coimbra, M.A.; Gouveia, L.; Barreira, L.; Varela, J. Nutritional potential and toxicological evaluation of Tetraselmis sp. CtP4 microalgal biomass produced in industrial photobioreactors. Molecules 2019, 24, 3192. [Google Scholar] [CrossRef]
- World Health Organization. Cyanobacterial Toxins: Anatoxin-a Background Document for Development of WHO Guidelines for Drinking-Water Quality and Guidelines for Safe Recreational Water Environments; WHO: Geneva, Switzerland, 2019; Available online: https://apps.who.int/iris/handle/10665/338060. (accessed on 11 April 2021).
- Sánchez-Parra, E.; Boutarfa, S.; Aboal, M. Are Cyanotoxins the Only Toxic Compound Potentially Present in Microalgae Supplements? Results from a Study of Ecological and Non-Ecological Products. Toxins 2020, 12, 552. [Google Scholar] [CrossRef]
- Zastepa, A.; Chemali, C. Bloom announcement: Late season cyanobacterial blooms co-dominated by Microcystis flos-aquae, Lyngbya birgei, and Aphanizomenon flos-aquae complex in Hamilton Harbour (Lake Ontario), an area of concern impacted by industrial effluent and residential waste. Data Br. 2021, 35, 106800. [Google Scholar] [CrossRef]
- (Quackwatch) Cell Tech Sued for Wrongful Death. Available online: https://quackwatch.org/cases/civil/celltech/complaint/ (accessed on 10 April 2021).
- Pelletreau, K.; Muller-Parker, G. Sulfuric acid in the phaeophyte alga Desmarestia munda deters feeding by the sea urchin Strongylocentrotus droebachiensis. Mar. Biol. 2002, 141, 1–9. [Google Scholar] [CrossRef]
- Rhodes, L.L.; Thomas, A.E. Coolia monotis (Dinophyceae): A toxic epiphytic microalgal species found in New Zealand (Note). N. Z. J. Mar. Freshw. Res. 1997, 31, 139–141. [Google Scholar] [CrossRef]
- AlgaeBase Coolia monotis Meunier 1919. Available online: https://www.algaebase.org/search/species/detail/?species_id=52206 (accessed on 10 May 2021).
- Thomas, I.; Siew, L.Q.C.; Watts, T.J.; Haque, R. Seaweed allergy. J. Allergy Clin. Immunol. Pract. 2019, 7, 714–715. [Google Scholar] [CrossRef]
- Fleurence, J.; Levine, I.A. Antiallergic and Allergic Properties. In Microalgae in Health and Disease Prevention; Levine, I.A., Fleurence, J., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 307–315. ISBN 978-0-12-811405-6. [Google Scholar]
- Motoyama, K.; Hamada, Y.; Nagashima, Y.; Shiomi, K. Allergenicity and allergens of amphipods found in nori (dried laver). Food Addit. Contam. 2007, 24, 917–922. [Google Scholar] [CrossRef]
- Hashimoto, H.; Hongo, T.; Hayashi, C.; Nakamura, K.; Nakanishi, K.; Ikeda, M.; Adachi, R.; Akiyama, H.; Teshima, R.; Yano, T. A method for the detection of shrimp/prawn and crab DNAs to identify allergens in dried seaweed products. Jpn. J. Food Chem. Saf. 2015, 22, 1–10. [Google Scholar] [CrossRef]
- Arulkumar, A.; Nigariga, P.; Paramasivam, S.; Rajaram, R. Metals accumulation in edible marine algae collected from Thondi coast of Palk Bay, Southeastern India. Chemosphere 2019, 221, 856–862. [Google Scholar] [CrossRef]
- Lin, Z.; Li, J.; Luan, Y.; Dai, W. Application of algae for heavy metal adsorption: A 20-year meta-analysis. Ecotoxicol. Environ. Saf. 2020, 190, 110089. [Google Scholar] [CrossRef]
- Packer, M.A.; Harris, G.C.; Adams, S.L. Food and Feed Applications of Algae. In Algae Biotechnology; Springer International Publishing: Cham, Switzerland, 2016; pp. 217–247. [Google Scholar]
- Arunakumara, K.K.I.U.; Zhang, X. Heavy Metal Bioaccumulation and Toxicity with Special Reference to Microalgae. J. Ocean Univ. China 2008, 7, 60–64. [Google Scholar] [CrossRef]
- Desideri, D.; Cantaluppi, C.; Ceccotto, F.; Meli, M.A.; Roselli, C.; Feduzi, L. Essential and toxic elements in seaweeds for human consumption. J. Toxicol. Environ. Health Part A 2016, 79, 112–122. [Google Scholar] [CrossRef]
- Rose, M.; Lewis, J.; Langford, N.; Baxter, M.; Origgi, S.; Barber, M.; MacBain, H.; Thomas, K. Arsenic in seaweed—Forms, concentration and dietary exposure. Food Chem. Toxicol. 2007, 45, 1263–1267. [Google Scholar] [CrossRef]
- Fu, Y.; Yin, N.; Cai, X.; Du, H.; Wang, P.; Sultana, M.S.; Sun, G.; Cui, Y. Arsenic speciation and bioaccessibility in raw and cooked seafood: Influence of seafood species and gut microbiota. Environ. Pollut. 2021, 280, 116958. [Google Scholar] [CrossRef]
- Vellinga, R.E.; Sam, M.; Verhagen, H.; Jakobsen, L.S.; Ravn-Haren, G.; Sugimoto, M.; Torres, D.; Katagiri, R.; Thu, B.J.; Granby, K.; et al. Increasing Seaweed Consumption in the Netherlands and Portugal and the Consequences for the Intake of Iodine, Sodium, and Exposure to Chemical Contaminants: A Risk-Benefit Study. Front. Nutr. 2022, 8, 792923. [Google Scholar] [CrossRef]
- Lähteenmäki-Uutela, A.; Rahikainen, M.; Camarena-Gómez, M.T.; Piiparinen, J.; Spilling, K.; Yang, B. European Union legislation on macroalgae products. Aquac. Int. 2021, 29, 487–509. [Google Scholar] [CrossRef]
- Banach, J.L.; Hoek-van den Hil, E.F.; Fels-Klerx, H.J. Food safety hazards in the European seaweed chain. Compr. Rev. Food Sci. Food Saf. 2020, 19, 332–364. [Google Scholar] [CrossRef]
- González, A.; Paz, S.; Rubio, C.; Gutiérrez, Á.J.; Hardisson, A. Human Exposure to Iodine from the Consumption of Edible Seaweeds. Biol. Trace Elem. Res. 2020, 197, 361–366. [Google Scholar] [CrossRef]
- Phaneuf, D.; Côté, I.; Dumas, P.; Ferron, L.A.; LeBlanc, A. Evaluation of the Contamination of Marine Algae (Seaweed) from the St. Lawrence River and Likely to Be Consumed by Humans. Environ. Res. 1999, 80, S175–S182. [Google Scholar] [CrossRef]
- Malea, P.; Chatziapostolou, A.; Kevrekidis, T. Trace element seasonality in marine macroalgae of different functional-form groups. Mar. Environ. Res. 2015, 103, 18–26. [Google Scholar] [CrossRef]
- Han, P.; Lu, Q.; Fan, L.; Zhou, W. A Review on the Use of Microalgae for Sustainable Aquaculture. Appl. Sci. 2019, 9, 2377. [Google Scholar] [CrossRef]
- Fabricant, D. Arsenic in Herbal Kelp Supplements: Concentration, Regulations, and Labeling. Environ. Health Perspect. 2007, 115, A574. [Google Scholar] [CrossRef][Green Version]
- Mirzayeva, A.; Castro, R.; Barroso, C.G.; Durán-Guerrero, E. Characterization and differentiation of seaweeds on the basis of their volatile composition. Food Chem. 2021, 336, 127725. [Google Scholar] [CrossRef]
- Hou, X.; Chai, C.; Qian, Q.; Yan, X.; Fan, X. Determination of chemical species of iodine in some seaweeds (I). Sci. Total Environ. 1997, 204, 215–221. [Google Scholar] [CrossRef]
- Zava, T.T.; Zava, D.T. Assessment of Japanese iodine intake based on seaweed consumption in Japan: A literature-based analysis. Thyroid Res. 2011, 4, 14. [Google Scholar] [CrossRef]
- Badar, M.; Batool, F.; Idrees, M.; Iqbal, H.R.; Zia, M.A. Effect of Boiling on Removing of Botulinum Toxins from Drinking Water Samples. Int. J. Sci. Res. 2016, 5, 1969–1973. [Google Scholar] [CrossRef]
- Abraham, A.; Flewelling, L.J.; El Said, K.R.; Odom, W.; Geiger, S.P.; Granholm, A.A.; Jackson, J.T.; Bodager, D. An occurrence of neurotoxic shellfish poisoning by consumption of gastropods contaminated with brevetoxins. Toxicon 2021, 191, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Wu, S.; Yin, R.; Bai, X.; Bhunia, A.K.; Liu, C.; Zheng, Y.; Wang, F.; Blatchley, E.R. Effects of fulvic acid size on microcystin-LR photodegradation and detoxification in the chlorine/UV process. Water Res. 2021, 193, 116893. [Google Scholar] [CrossRef] [PubMed]
- Al-Thawadi, S. Public perception of algal consumption as an alternative food in the Kingdom of Bahrain. Arab J. Basic Appl. Sci. 2018, 25, 1–12. [Google Scholar] [CrossRef]
- Lee, J.; Chambers IV, E.; Chambers, D.H.; Chun, S.S.; Oupadissakoon, C.; Johnson, D.E. Consumer Acceptance for Green Tea by Consumers in the United States, Korea and Thailand. J. Sens. Stud. 2010, 25, 109–132. [Google Scholar] [CrossRef]
- Chapman, A.S.; Stévant, P.; Larssen, W.E. Food or fad? Challenges and opportunities for including seaweeds in a Nordic diet. Bot. Mar. 2015, 58, 423–433. [Google Scholar] [CrossRef]
- (Bord à Bord) La Cuisine Aux Algues. Available online: https://www.bord-a-bord.fr/-La-cuisine-aux-algues-.html?lang=fr (accessed on 10 April 2021).
- Milne, X. The Seaweed Cookbook; Joseph, M., Ed.; Penguin UK: London, UK, 2016; ISBN 978-0718183660. [Google Scholar]
- Morel, H. Vive Les Algues! Saveurs Iodées Pour Recettes Gourmandes, 1st ed.; Trop Mad: Lorient, France, 2019; ISBN 9782918068136. [Google Scholar]
- van den Burg, S.W.K.; Dagevos, H.; Helmes, R.J.K. Towards sustainable European seaweed value chains: A triple P perspective. ICES J. Mar. Sci. 2021, 78, 443–450. [Google Scholar] [CrossRef]
- Kulsdom, M.; Hanekroot, D.; van Langelaar, K.; Kreischer, L. The Dutch Weed Burger. Available online: https://dutchweedburger.com/en/ (accessed on 19 April 2021).
- Miyaki, T.; Retiveau-Krogmann, A.; Byrnes, E.; Takehana, S. Umami Increases Consumer Acceptability, and Perception of Sensory and Emotional Benefits without Compromising Health Benefit Perception. J. Food Sci. 2016, 81, S483–S493. [Google Scholar] [CrossRef]
- Ikeda, K. New Seasonings. Chem. Senses 2002, 27, 847–849. [Google Scholar] [CrossRef]
- Mitsuda, H.; Shikanai, T. Studies on the Utilization of Chlorella for Food: Studies on the Nutritive Values of Cell Free Algal Proteins (Commemoration Issue Dedicated to Professor Sankichi Takei On the Occasion of his Retirement). Bull. Inst. Chem. Res. Kyoto Univ. 1960, 38, 40–58. [Google Scholar]
- Dewi, E.N.; Amalia, U.; Mel, M. The Effect of Different Treatments to the Amino Acid Contents of Micro Algae Spirulina sp. Aquat. Procedia 2016, 7, 59–65. [Google Scholar] [CrossRef]
- Kamenidou, I.; Aggelopoulos, S.; Batzios, A.C. Natural Medical Attributes and Benefits of Spirulina: Segmentation Based on Consumers Knowledge. J. Med. Plants Res. 2011, 5, 3192–3199. [Google Scholar]
- Kamenidou, I.; Priporas, C.-V. Factors Predicting Consumers’ Knowledge of Spirulina Health Benefits. J. Food Agric. Environ. 2010, 8, 16–20. [Google Scholar]
- Lafarga, T.; Rodríguez-Bermúdez, R.; Morillas-España, A.; Villaró, S.; García-Vaquero, M.; Morán, L.; Sánchez-Zurano, A.; González-López, C.V.; Acién-Fernández, F.G. Consumer knowledge and attitudes towards microalgae as food: The case of Spain. Algal Res. 2021, 54, 102174. [Google Scholar] [CrossRef]
- Rzymski, P.; Jaśkiewicz, M. Microalgal food supplements from the perspective of Polish consumers: Patterns of use, adverse events, and beneficial effects. J. Appl. Phycol. 2017, 29, 1841–1850. [Google Scholar] [CrossRef]
- Moons, I.; De Pelsmacker, P.; Barbarossa, C. The Drivers of the Usage Intention of Spirulina Algae in Food in Different Market Segments. In Proceedings of the 16th International Marketing Trends Conference, Madrid, Spain, 26–28 January 2017; pp. 1–22. [Google Scholar]
- Moons, I.; Barbarossa, C.; De Pelsmacker, P. The Determinants of the Adoption Intention of Eco-friendly Functional Food in Different Market Segments. Ecol. Econ. 2018, 151, 151–161. [Google Scholar] [CrossRef]
- Weinrich, R.; Elshiewy, O. Preference and willingness to pay for meat substitutes based on micro-algae. Appetite 2019, 142, 104353. [Google Scholar] [CrossRef]
- Thomas, C.; Symoneaux, R.; Pantin-Sohier, G.; Picouet, P.; Maître, I. Perceptions of Spirulina from French Consumers of Organic Products; hal-02615769; HAL Open Science: Lyon, France, 2020. [Google Scholar]
- Roßmann, M.; Rösch, C. Key-Narratives of Microalgae Nutrition: Exploring futures through a public Delphi survey in Germany. Sci. Public Policy 2020, 47, 137–147. [Google Scholar] [CrossRef]
- Ricci, E.C.; Banterle, A.; Stranieri, S. Trust to Go Green: An Exploration of Consumer Intentions for Eco-friendly Convenience Food. Ecol. Econ. 2018, 148, 54–65. [Google Scholar] [CrossRef]
- Sachdeva, S.; Jordan, J.; Mazar, N. Green consumerism: Moral motivations to a sustainable future. Curr. Opin. Psychol. 2015, 6, 60–65. [Google Scholar] [CrossRef]
- Griskevicius, V.; Tybur, J.M.; Van den Bergh, B. Going green to be seen: Status, reputation, and conspicuous conservation. J. Personal. Soc. Psychol. 2010, 98, 392–404. [Google Scholar] [CrossRef]
- Burdock, G.A.; Carabin, I.G. Generally recognized as safe (GRAS): History and description. Toxicol. Lett. 2004, 150, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Office of Food Additive Safety Determining Regulatory Status Food Ingredientg the Regulatory Status of a Food Ingredient. Available online: https://www.fda.gov/food/food-ingredients-packaging/determining-regulatory-status-food-ingredient (accessed on 20 April 2021).
- FDA Generally Recognized as Safe (GRAS). Available online: https://www.fda.gov/food/food-ingredients-packaging/generally-recognized-safe-gras (accessed on 20 April 2021).
- (Government of Canada) About Novel and Genetically-Modified (GM) Foods. Available online: https://www.canada.ca/en/health-canada/services/food-nutrition/genetically-modified-foods-other-novel-foods.html (accessed on 20 April 2021).
- (Health Canada) Natural Health Products Ingredients Database. Available online: http://webprod.hc-sc.gc.ca/nhpid-bdipsn/search-rechercheReq.do?lang=eng (accessed on 20 April 2021).
- Standard 1.5.1; (Federal Register of Legislation) Australia New Zealand Food Standards Code—Standard 1.5.1—Novel Foods. Available online: https://www.legislation.gov.au/Details/F2017C00324 (accessed on 20 April 2021).
- Shen, R. China Regulations on New Food Raw Materials. Available online: https://food.chemlinked.com/foodpedia/china-regulations-new-food-raw-materials#C6 (accessed on 20 April 2021).
- China, S.C. of the N.P.C.—Food Safety Law of the People’s Republic of China. Available online: http://www.lawinfochina.com/Display.aspx?LookType=3&Lib=law&Cgid=113981&Id=7344&SearchKeyword=&SearchCKeyword=&paycode=# (accessed on 20 April 2021).
- Sun, J. The Regulation of “Novel Food” in China: The Tendency of Deregulation. Eur. Food Feed Law Rev. 2015, 10, 442–448. [Google Scholar]
- (Instrumentalities of the State Council) China Administrative Measures for the Safety Review of New Food Raw Materials. Available online: http://www.lawinfochina.com/display.aspx?lib=law&id=15358&CGid&EncodingName=big5 (accessed on 20 April 2021).
- Dos Santos Fernandes De Araujo, R.; Peteiro, C. Algae as Food and Food Supplements in Europe; Publications Office of the European Union: Luxembourg, 2021; ISBN 978-92-76-40548-1. [Google Scholar]
- Pulz, O.; Gross, W. Valuable products from biotechnology of microalgae. Appl. Microbiol. Biotechnol. 2004, 65, 635–648. [Google Scholar] [CrossRef]
- European Commission. Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions. In A Blueprint to Safeguard Europe’s Water Resources; European Commission: Brussels, Belgium, 2012. [Google Scholar]
- Ruiz, J.; Olivieri, G.; de Vree, J.; Bosma, R.; Willems, P.; Reith, J.H.; Eppink, M.H.M.; Kleinegris, D.M.M.; Wijffels, R.H.; Barbosa, M.J. Towards industrial products from microalgae. Energy Environ. Sci. 2016, 9, 3036–3043. [Google Scholar] [CrossRef]
- EU Open Data Portal. The Official Portal for European Data. Available online: https://data.europa.eu/en (accessed on 10 April 2021).
- Schulze, P.S.C.; Carvalho, C.F.M.; Pereira, H.; Gangadhar, K.N.; Schüler, L.M.; Santos, T.F.; Varela, J.C.S.; Barreira, L. Urban wastewater treatment by Tetraselmis sp. CTP4 (Chlorophyta). Bioresour. Technol. 2017, 223, 175–183. [Google Scholar] [CrossRef]
- Mazarrasa, I.; Olsen, Y.S.; Mayol, E.; Marbà, N.; Duarte, C.M. Global Unbalance in Seaweed Production, Research Effort and Biotechnology Markets. Biotechnol Adv 2014, 32, 1028–1036. [Google Scholar] [CrossRef]
- Buschmann, A.H.; Camus, C.; Infante, J.; Neori, A.; Israel, Á.; Hernández-González, M.C.; Pereda, S.V.; Gomez-Pinchetti, J.L.; Golberg, A.; Tadmor-Shalev, N.; et al. Seaweed production: Overview of the global state of exploitation, farming and emerging research activity. Eur. J. Phycol. 2017, 52, 391–406. [Google Scholar] [CrossRef]
- Mazarrasa, I.; Olsen, Y.S.; Mayol, E.; Marbà, N.; Duarte, C.M. Rapid growth of seaweed biotechnology provides opportunities for developing nations. Nat. Biotechnol. 2013, 31, 591–592. [Google Scholar] [CrossRef]
- Duncan, T.V. Applications of Nanotechnology in Food Packaging and Food Safety: Barrier Materials, Antimicrobials and Sensors. J. Colloid Interface Sci. 2011, 363, 1–24. [Google Scholar] [CrossRef]
- Koynde, A.K.; Chew, K.W.; Manickam, S.; Chang, J.S.; Show, P.L. Emerging Algal Nanotechnology for High-Value Compounds: A Direction to Future Food Production. Trends Food Sci. Technol. 2021, 116, 290–302. [Google Scholar] [CrossRef]
- Carvalho, A.P.; Meireles, L.A.; Malcata, F.X. Microalgal Reactors: A Review of Enclosed System Designs and Performances. Biotechnol. Prog. 2006, 22, 1490–1506. [Google Scholar] [CrossRef] [PubMed]
- Cuaresma, M.; Janssen, M.; van den End, E.J.; Vílchez, C.; Wijffels, R.H. Luminostat operation: A tool to maximize microalgae photosynthetic efficiency in photobioreactors during the daily light cycle? Bioresour. Technol. 2011, 102, 7871–7878. [Google Scholar] [CrossRef] [PubMed]
- SCHOTT Algae cultivation in flat panel reactors compared to tubular glass PBRs; 2017. Available online: https://www.schott.com/en-gb/applications/photobioreactors (accessed on 1 March 2021).
- Sierra, E.; Acién, F.G.; Fernández, J.M.; García, J.L.; González, C.; Molina, E. Characterization of a flat plate photobioreactor for the production of microalgae. Chem. Eng. J. 2008, 138, 136–147. [Google Scholar] [CrossRef]
- Ministério da Marinha Portaria no 22.559; Diário da República: Lisbon, Portugal, 1967; p. 264.
- D’Oliveira, M.L. Sobre a utilização de algumas algas marinhas da nossa costa. Brotéria Ciências Nat. 1947, 16, 77–82. [Google Scholar]
- Viana, C.E. Argaço; AO NORTE: Viana do Castelo, Portugal, 2012; Available online: http://www.ao-norte.com/argaco.php (accessed on 1 March 2022).
- Un Océano de Algas Multiuso. Available online: https://elpais.com/diario/2008/03/21/tendencias/1206054003_850215.html (accessed on 1 March 2022).
- Varatojo, F. Alimentação Macrobiótica Padrão. Available online: https://www.institutomacrobiotico.com/pt-pt/imp/artigos/alimentacao-macrobiotica-padrao (accessed on 1 March 2021).
- Rahikainen, M.; Yang, B. Macroalgae as food and feed ingredients in the Baltic Sea region—Regulation by the European Union. 2020, pp. 1–20. Available online: https://www.submariner-network.eu/images/grass/GRASS_O3.4a_EU_regulation_of_seaweed_food_and_feed.pdf (accessed on 19 February 2021).
- (Provida) Provida, Portugal. Available online: https://www.provida.pt/produtos/#!/pesquisa/alga/?search=prods&query=alga (accessed on 19 February 2021).
- Nutrialga, Portugal. Available online: https://www.nutrialga.com/ (accessed on 6 February 2021).
- Loja das Algas, Portugal. Available online: https://www.lojadasalgas.pt/ (accessed on 6 February 2021).
- Drennan, F. Seaweed Bushcraft guide: Seaweed in Season. Bushcr. J. 2016. [Google Scholar]
- Rhatigan, P. Prannie Rhatigan’s Irish Seaweed Christmas Kitchen: Christmastide by the Coast; Inishmurray Ink Publishing: London, UK, 2018; ISBN 1916493602. [Google Scholar]
- Oliveira, J. 1o Encontro Nacional sobre Macroalgas Marinhas; LNETI INIP: Lisbon, Portugal, 1990. [Google Scholar]
- Chapman, V.J.; Chapman, D.J. Seaweeds and their Uses; Springer: Dordrecht, The Netherlands, 1980; ISBN 978-94-009-5808-1. [Google Scholar]
- (Ocean Rainforest) Ocean Rainforest Products (Sustainable Nordic Seaweed). Available online: https://www.oceanrainforest.com/full-range (accessed on 1 March 2022).
- McKenna, S. Extreme Greens: Understanding Seaweeds: Cooking, Foraging, Cosmetic, 1st ed.; McKenna, J., Ed.; Estragon Press: Cork, Ireland, 2013; ISBN 1906927197. [Google Scholar]
- Quéva, R.; Le Joncour, C. Algues gourmandes; Flammarion: Paris, France, 2017; ISBN 978-2-08-140848-7. [Google Scholar]
- (CBI) The European Market Potential for Edible Seaweed. Available online: https://www.cbi.eu/market-information/natural-ingredients-health-products/seaweed/market-potential (accessed on 10 April 2021).
- (DGRM) Algas: Denominação Comercial para as Diferentes Macroalgas e Microalgas que Podem ser Comercializadas em Portugal. 27 November 2019. Available online: https://www.dgrm.mm.gov.pt/documents/20143/56136/ALGAS+27.Nov.2019.pdf/32aefb8d-6fc3-54b8-d4dd-89e465390495 (accessed on 10 April 2021).
- Guiry, M.D. Use of seaweed as food in Ireland. Available online: https://www.seaweed.ie/uses_ireland/irishseaweedfood.php (accessed on 1 February 2021).
- (AlgAran) Organic Seaveg & Seaweed Supplements. Available online: https://www.seaweedproducts.ie/product-category/organic-seaveg-seaweed-supplements/ (accessed on 14 March 2022).
- (Seaweed Solutions) Seaweed Farming—The Future of Cultivation is at Sea. Available online: https://seaweedsolutions.com (accessed on 1 March 2022).
- (PORTO-MUIÑOS) Las verduras del mar. Available online: https://www.portomuinos.com (accessed on 1 March 2022).
- (AlgAran) Algaran Organic Vegan Kelp Supplements. Available online: https://www.seaweedproducts.ie/product/algaran-organic-vegan-kelp-supplements-70-days/ (accessed on 14 March 2022).
- Williams, M. Edible Seaweed Foraging—Galloway Wild Foods, Ireland. Available online: https://gallowaywildfoods.com/category/edible-wild-seaweeds/ (accessed on 11 February 2021).
- Pires, P. Receitas UBQ Madeira, Portugal. Available online: https://www.ubqmadeira.com/receitas/ (accessed on 21 February 2021).
- Ornelas, B. Receitas: Omelete de Espinafres com a Alga Asparagopsis taxiformis. Available online: https://www.ubqmadeira.com/receitas/ (accessed on 21 February 2021).
- Alga SeaExpert, Portugal. Available online: https://seaexpert-azores.com/ (accessed on 17 February 2021).
- Milinovic, J.; Campos, B.; Mata, P.; Diniz, M.; Noronha, J.P. Umami free amino acids in edible green, red, and brown seaweeds from the Portuguese seashore. J. Appl. Phycol. 2020, 32, 3331–3339. [Google Scholar] [CrossRef]
- (El Bulli) El Bulli Restaurant menu (2006–2009). Available online: http://www.elbulli.com/catalogo/catalogo/index.php?lang=en (accessed on 7 February 2021).
- Ziino, G.; Nibali, V.; Panebianco, A. Bacteriological investigation on “Mauro” sold in Catania. Vet. Res. Commun. 2010, 34, 157–161. [Google Scholar] [CrossRef][Green Version]
- Araujo, G.S.; Cotas, J.; Morais, T.; Leandro, A.; García-Poza, S.; Gonçalves, A.M.M.M.; Pereira, L. Calliblepharis jubata cultivation potential—A comparative study between controlled and semi-controlled aquaculture. Appl. Sci. 2020, 10, 7553. [Google Scholar] [CrossRef]
- Albe, M. Mauru: La ricetta dell’insalta con l’alga siciliana. Available online: https://www.greenme.it/mangiare/altri-alimenti/mauru-ricetta-alga/ (accessed on 11 February 2021).
- Black, W.A.P. Seaweeds and their value in foodstuffs. Proc. Nutr. Soc. 1953, 12, 32–39. [Google Scholar] [CrossRef]
- Zemke-White, W.L.; Ohno, M. World seaweed utilisation: An end-of-century summary. J. Appl. Phycol. 1999, 11, 369–376. [Google Scholar] [CrossRef]
- CEVA Réglementation Algues Alimentaires Synthèse CEVA au 10/02/2014. 2014. Available online: https://www.foodplanet.fr/app/download/5806453856/R3A9glementation+algues+alimentaires+2014.pdf (accessed on 11 February 2021).
- García Tasende, M.; Peteiro, C. Explotación de las macroalgas marinas: Galicia como caso de estudio hacia una gestión sostenible de los recursos. Ambienta 2015, 111, 116–132. [Google Scholar]
- MacLean, R. Eat Your Greens: An Examination of the Potential Diet Available in Ireland during the Mesolithic. Ulster J. Archaeol. 1993, 56, 1–8. [Google Scholar]
- (ALGAplus) Tok de Mar Products by ALGAplus. Available online: https://algaplus.lojasonlinectt.pt/catalog (accessed on 10 April 2021).
- (DanskTANG) Seaweed from Denmark. Available online: https://www.danish-seaweed.com/seaweed-types/ (accessed on 14 March 2022).
- Rhatigan, P. Irish Seaweed Kitchen (The comprehensive Guide to Healthy Everyday Cooking with Seaweeds); Booklink: Northampton, MA, USA, 2009; ISBN 1906886229. [Google Scholar]
- Weinberger, F.; Paalme, T.; Wikström, S.A. Seaweed resources of the Baltic Sea, Kattegat and German and Danish North Sea coasts. Bot. Mar. 2020, 63, 61–72. [Google Scholar] [CrossRef]
- WoRMS Taxon Details: Coccotylus truncatus. Available online: https://www.marinespecies.org/aphia.php?p=taxdetails&id=145654#links (accessed on 17 March 2022).
- Hairon, D. Edible seaweeds in Jersey. Available online: https://jerseywalkadventures.co.uk/press-office/edible-seaweeds-in-jersey/ (accessed on 17 February 2021).
- Algamar, Portugal. Available online: https://www.algamar.com/pt-pt/quem-somos/ (accessed on 6 February 2021).
- ALGA4FOOD Gastronomia: O Alimento que vem do mar. Available online: https://alga4food.wixsite.com/page/gastronomia (accessed on 11 February 2021).
- Commission Implementing Regulation (EU). Commission Implementing Regulation (EU) 2018/460 of 20 March 2018 authorising the placing on the market of Ecklonia cava phlorotannins as a novel food under Regulation (EU) 2015/2283 of the European Parliament and of the Council and amending Commission Im. Off. J. Eur. Union. 2018, 78, 2–6. [Google Scholar]
- Gabriel, P. (ALGA4FOOD) Algas, Onde o mar Começa. Available online: https://dd4d232f-da72-4e83-b936-ede1885a4984.filesusr.com/ugd/baccf3_06402b38772549dd88c708818d5b0e32.pdf (accessed on 11 April 2021).
- Dias, N. My Name is Best, José Besteiro! 50 anos, 50 Receitas; 1st ed.; Âncora Editora: Lisbon, Portugal, 2016; ISBN 9789727805778. [Google Scholar]
- Younes, M.; Aggett, P.; Aguilar, F.; Crebelli, R.; Filipič, M.; Frutos, M.J.; Galtier, P.; Gott, D.; Gundert-Remy, U.; Kuhnle, G.G.; et al. Re-evaluation of carrageenan (E 407) and processed Eucheuma seaweed (E 407a) as food additives. EFSA J. 2018, 16. [Google Scholar] [CrossRef]
- Pereira, J. A Treatise on Food and Diet with Observation on the Dietetic Regimen suited for Disordered States of the Digestive Organs.; Fowler & Wells Publisher: New York, NY, USA, 1843; ISBN 1247549038. [Google Scholar]
- Patarra, R.F.; Buschmann, A.H.; Abreu, M.H.; Neto, A.I. Cultivo de macroalgas nos Açores… Oportunidades e desafios. In Boletim de Biotecnologia; Sociedade Portuguesa de Biotecnologia: Braga, Portugal, 2014; Volume 2. [Google Scholar]
- (Celeiro) Celeiro, Portugal. Available online: https://www.celeiro.pt/ (accessed on 19 February 2021).
- EC Commission implementing Regulation (EU) 2017/2470 of 20 December 2017 establishing the Union list of novel foods in accordance with Regulation (EU) 2015/2283 of the European Parliament and of the Council on novel foods. Off. J. Eur. Union 2017, 351, 1–188.
- Fralick, R.A.; Baldwin, H.P.; Neto, A.I.; Hehre, E.J. Physiological responses of Pterocladia and Gelidium (Gelidiales, Rhodophyta) from the Azores, Portugal. Hydrobiologia 1990, 204–205, 479–482. [Google Scholar] [CrossRef]
- Fralick, R. Algas agarófitas nos Açores. Relatório Final (R/5/77); Centro de Documentação Tradução e. Autónoma dos Açores: Açores, Portugal, 1977. [Google Scholar]
- Abreu, H. Exploração de Macroalgas: Regulamentação; Aveiro, Portugal, 2014. [Google Scholar]
- Rodrigues, D.; Freitas, A.C.; Pereira, L.; Rocha-Santos, T.A.P.; Vasconcelos, M.W.; Roriz, M.; Rodríguez-Alcalá, L.M.; Gomes, A.M.P.; Duarte, A.C. Chemical composition of red, brown and green macroalgae from Buarcos bay in Central West Coast of Portugal. Food Chem. 2015, 183, 197–207. [Google Scholar] [CrossRef]
- Largo, D.; Critchley, A.; Hurtado, A.; Paul, N.; Pereira, L.; Cornish, M. Seaweed resources of the world: A 2020 vision. Part 4. Bot. Mar. 2020, 63, 299–301. [Google Scholar] [CrossRef]
- Denis, C.; Morançais, M.; Li, M.; Deniaud, E.; Gaudin, P.; Wielgosz-Collin, G.; Barnathan, G.; Jaouen, P.; Fleurence, J. Study of the chemical composition of edible red macroalgae Grateloupia turuturu from Brittany (France). Food Chem. 2010, 119, 913–917. [Google Scholar] [CrossRef]
- Munier, M.; Dumay, J.; Morançais, M.; Jaouen, P.; Fleurence, J. Variation in the Biochemical Composition of the Edible Seaweed Grateloupia turuturu Yamada Harvested from Two Sampling Sites on the Brittany Coast (France): The Influence of Storage Method on the Extraction of the Seaweed Pigment R-Phycoerythrin. J. Chem. 2013, 2013, 568548. [Google Scholar] [CrossRef]
- Pereira, L. University of C. Algas à mesa. Workshop Sobre Algas Comestíveis. IMAR-CMA/Universidade de Coimbra; Museu do Traje: Lisbon, Portugal, 2010. [Google Scholar]
- Palminha, F. As algas marinhas nos costumes faialenses. 1958; pp. 113–116.
- Pateira, M.L. Algas—Recursos e Investigação em Portugal. 1992.
- Rodrigues, F. “Legumes Do Mar” Saltam para as Suas Receitas. Available online: http://achadosmaracores.blogspot.com/2014/03/legumes-do-mar-saltam-para-as-suas.html (accessed on 3 February 2021).
- Pimenta, I. Macroalgas na Alimentação Humana—A Congelação como Processo de Conservação. 2010, pp. 1–110. Available online: http://hdl.handle.net/20.500.11960/2075 (accessed on 19 February 2021).
- Neves, B.; Real, H.; Carvalho, T. Algas a gosto: Considerações nutricionais e de saúde; Craveiro, C., Ed.; Multitema: Porto, Portugal, 2019; ISBN 978-989-8631-41-1. [Google Scholar]
- Rey, F.; Cartaxana, P.; Melo, T.; Calado, R.; Pereira, R.; Abreu, H.; Domingues, P.; Cruz, S.; Domingues, M.R. Domesticated Populations of Codium tomentosum Display Lipid Extracts with Lower Seasonal Shifts than Conspecifics from the Wild—Relevance for Biotechnological Applications of this Green Seaweed. Mar. Drugs 2020, 18, 188. [Google Scholar] [CrossRef]
- C-, C.; Gmbh, N.; Nordrhein-westfalen, L.; German, T.; Drink, S.; Land, T.; Rhine-westphalia, N.; Bundesverwaltungsgericht, T.; Court, F.A. Court of Justice of the European Union PRESS RELEASE No 69/21 EU Law Prohibits the Addition of the alga Lithothamnium Calcareum in the Processing of Organic Foodstuffs Such as rice- and Soya-Based Organic Drinks for the Purpose of Their Enrichment with Calcium; Court of Justice of the European Union: Luxembourg, 2021; Volume 2018. [Google Scholar]
- (Violey) Natumi Organic Rice Drink Calcium from seaweed Lithoathamnium. Available online: https://www.violey.com/en/natumi-organic-rice-drink-calcium_p_24785.html (accessed on 4 August 2021).
- (Vecteur Santé) Lithothamne de mer de Norvège 500 mg. Available online: https://www.naturitas.fr/p/complements/algues/lithothamne-de-mer-de-norvege-500mg-80-capsules-vecteur-sante (accessed on 1 March 2022).
- Nauticorvo Erva do Calhau—Corvo Açores 2008. Available online: https://youtu.be/fbXqBwoWtxU (accessed on 1 February 2021).
- (Expolab Centro Ciência Viva) Workshop “Alimentação do Oceano: Algas Como Fonte Alternativa de Proteína”. Available online: https://www.youtube.com/watch?v=wvMHcSjKHPc&ab_channel=ExpolabCentroCiênciaViva (accessed on 1 February 2021).
- Matos, S. Designing Food Cultures: Propagating the Consumption of Seaweed in the Azores Islands Through Recipes. Iridescent 2012, 2, 24–33. [Google Scholar] [CrossRef]
- Patarra, A.R.F. Fatty Acids of Selected Azorean Seaweeds. Pesquisa de Ácidos Gordos em Macroalgas Marinhas do litoral dos Açores. Master Thesis, University of Porto, Porto, Portugal, 2008. [Google Scholar]
- Algas comercializadas Flying Fish Azores. Available online: https://www.flyingfishazores.com/algas-pt (accessed on 17 March 2022).
- European Commission Commission Decision 2009/778/EC concerning the extension of uses of algal oil from the micro-algae Schizochytrium sp. as a novel food ingredient under Regulation (EC) No 258/97 of the European Parliament and of the Council. Off. J. Eur. Union 2009, L 278, 56–57.
- Dias, C. “Erva Patinha” tem Elevado teor de Proteínas e Fibras e o seu Consumo Regular Reduziria o Número de Acidentes Cardiovasculares. Available online: http://correiodosacores.pt/NewsDetail/ArtMID/383/ArticleID/25396/“Erva-patinha”-tem-elevado-teor-de-prote237nas-e-fibras-e-o-seu-consumo-regular-reduziria-o-n250mero-de-acidentes-cardiovasculares (accessed on 21 February 2021).
- Agenda Açores Top Azores: 19 Sabores açorianos que tens mesmo de provar. Available online: https://agendacores.pt/19-sabores-acorianos-que-tens-mesmo-de-provar/ (accessed on 16 February 2021).
- Lahaye, M.; Gomez-Pinchetti, J.-L.; del Rio, M.J.; Garcia-Reina, G. Natural decoloration, composition and increase in dietary fibre content of an edible marine algae, Ulva rigida (Chlorophyta), grown under different nitrogen conditions. J. Sci. Food Agric. 1995, 68, 99–104. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).