High Precisive Prediction of Aflatoxin B1 in Pressing Peanut Oil Using Raman Spectra Combined with Multivariate Data Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Peanut Oil Samples
2.2. Determination of Aflatoxin B1
2.3. Acquisition of Raman Spectra
2.4. Spectral Pretreatment Methods
- (1)
- Wavelet transform (wavelet transform, WT). WT is a time and frequency manipulation of a class of signals. In the time and frequency domains, the effective signal is continuous, while the noise is discrete [20]. Therefore, after WT, the absolute value of the coefficient of the effective signal is larger, and the threshold method can be used to filter the signal. Since the shoulder widths of the Raman peaks are inconsistent, it is necessary to select appropriate wavelet parameters to achieve a good noise filtering effect. In this study, when using WT to process Raman spectra, the wavelet basis function selected the “sym5” function, and the decomposition level was set to 5. The Raman spectral signal was decomposed and reconstructed using the automatic denoising functions “wden” and “minimaxi” thresholds of the one-dimensional signal to remove the noise signal.
- (2)
- Savitzky–Golay (SG) smoothing method. SG, also known as polynomial smoothing algorithm, was proposed by Savitzky and Golay and applied to data smoothing and noise filtering. When the SG preprocesses the spectral data, after defining the width of the window (which must be an odd number), a polynomial is used to fit the spectral variables in the window to predict the spectral intensity at the central wavelength point of the window [21]. As the window passes over the spectral data, the original value at the center of the window is replaced with the fitted value, resulting in smoothed data [22]. In this study, when processing Raman spectra with the SG, the polynomial order and the size of the smoothing window were set to 3 and 19, respectively.
- (3)
- Adaptive iteratively reweighted penalized least squares (airPLS). The airPLS calculates the spectral baseline by iteratively adjusting the weights of penalized least squares [23]. In the iterative solution, the weight remains unchanged, and the algorithm makes an adaptive adjustment to the fidelity weight based on the difference between the spectrum and the fitting baseline of the previous iteration.
2.5. Variable Selection Method
2.6. Partial Least Square
2.7. Model Evaluation
3. Results
3.1. Results of Spectral Pretreatment
3.2. Division of Calibration Set and Prediction Set Samples
3.3. Results of Feature Selection by the CARS Method
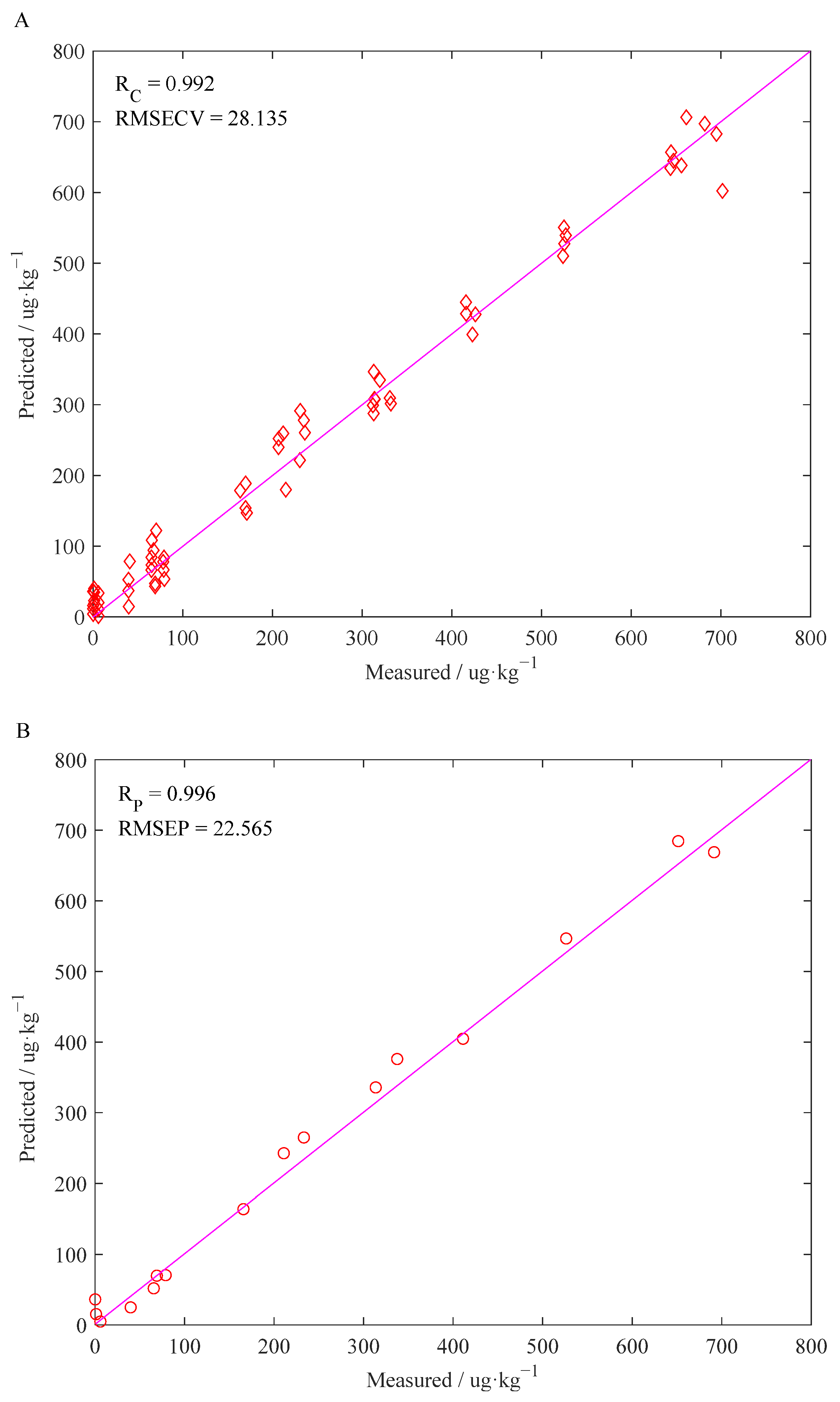
3.4. Results of the PLS Models Built on Optimized Features
4. Discussion
- (1)
- The absorption intensity of the Raman spectrum is related to the number of functional groups. As the degree of mildew increases, it causes a baseline shift in the Raman spectrum. And the more severe the mildew, the stronger the fluorescence effect is and the more serious the baseline drift is. That is to say, the increase of AFB1 content in peanut oil will cause corresponding changes in the Raman spectrum. Much existing literature also theoretically calculates and characterizes the Raman characteristic peaks of the AFB1, but they are all based on trace amounts of pure AFB1 or adding the pure product to non-mold edible oil. In this regard, we compared the theoretical characteristic peaks of the AFB1 calculated by the DFT with the Raman peaks collected by the actual experiment, and used these Raman peaks as input to construct the PLS model, but the model results were not satisfactory. This may be because in the process of mildewing of peanut oil in a high temperature and high humidity environment, the types of moldy fungi are not single (there may also be other molds, such as Salmonella, etc.), and this may result in the presented Raman peaks being a superposition of different mold contaminants, which is quite different from the theoretically calculated Raman spectrum of the AFB1. In addition, the Raman spectra of the same substance collected on different types of instruments will have deviations in the position of the spectral peaks and differences in the heights of the spectral peaks. Moreover, the surrounding environment of the AFB1 molecule in the experimental and theoretical calculations is also different. There is intermolecular interaction in the experiment, while the theoretical calculation simulates gas-phase molecules. Therefore, using the variable selection algorithm to optimize the collected spectral features from the perspective of numerical calculation can effectively reduce the spectral dimension and improve the accuracy and robustness of the subsequent model.
- (2)
- The performance of the CARS–PLS model is better than that of the FULL–PLS model, which directly shows the importance of variable selection in the model calibration process. When Raman spectra are collected, they will be disturbed by environmental, human operation and instrumental factors, resulting in a large amount of useless and irrelevant information in the obtained spectra. The FULL–PLS model construction utilizes full-spectrum variables, and the large amount of useless and irrelevant information will directly affect the generalization performance and stability of the constructed detection model. Therefore, a more reliable detection model can be obtained by optimizing the characteristic wavelengths in the model calibration process.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Khaneghah, A.M.; Es, I.; Raeisi, S.; Fakhri, Y. Aflatoxins in cereals: State of the art. J. Food Saf. 2018, 38, e12532. [Google Scholar]
- Diao, E.; Li, X.; Zhang, Z.; Ma, W.; Ji, N.; Dong, H. Ultraviolet irradiation detoxification of aflatoxins. Trends Food Sci. Technol. 2015, 42, 64–69. [Google Scholar] [CrossRef]
- Adebo, O.A.; Njobeh, P.B.; Gbashi, S.; Nwinyi, O.C.; Mavumengwana, V. Review on microbial degradation of aflatoxins. Crit. Rev. Food Sci. Nutr. 2017, 57, 3208–3217. [Google Scholar] [CrossRef]
- Kowalska, A.; Walkiewicz, K.; Koziel, P.; Muc-Wierzgon, M. Aflatoxins: Characteristics and impact on human health. Postepy Hig. Med. Dosw. 2017, 71, 315–327. [Google Scholar] [CrossRef] [PubMed]
- Shephard, G.S. Aflatoxins in peanut oil: Food safety concerns. World Mycotoxin J. 2018, 11, 149–158. [Google Scholar] [CrossRef]
- Tumukunde, E.; Ma, G.; Li, D.; Yuan, J.; Qin, L.; Wang, S. Current research and prevention of aflatoxins in China. World Mycotoxin J. 2020, 13, 121–138. [Google Scholar] [CrossRef]
- Xie, L.; Chen, M.; Ying, Y. Development of methods for determination of aflatoxins. Crit. Rev. Food Sci. Nutr. 2016, 56, 2642–2664. [Google Scholar] [CrossRef]
- Yan, C.; Wang, Q.; Yang, Q.; Wu, W. Recent advances in aflatoxins detection based on nanomaterials. Nanomaterials 2020, 10, 1626. [Google Scholar] [CrossRef]
- Yao, H.; Hruska, Z.; Di Mavungu, J.D. Developments in detection and determination of aflatoxins. World Mycotoxin J. 2015, 8, 181–191. [Google Scholar] [CrossRef]
- GB 2761-2017; Limits of Mycotoxins in Foods. General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China: Beijing, China, 2017.
- GB 5009.22-2016; Determination of Aflatoxin Group B and Group G in Food. General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China: Beijing, China, 2016.
- Zareef, M.; Arslan, M.; Hassan, M.M.; Ahmad, W.; Ali, S.; Li, H.H.; Qin, O.Y.; Wu, X.Y.; Hashim, M.M.; Chen, Q.S. Recent advances in assessing qualitative and quantitative aspects of cereals using nondestructive techniques: A review. Trends Food Sci. Technol. 2021, 116, 815–828. [Google Scholar] [CrossRef]
- Jiang, H.; He, Y.; Xu, W.; Chen, Q. Quantitative detection of acid value during edible oil storage by Raman spectroscopy: Comparison of the optimization effects of BOSS and VCPA algorithms on the characteristic Raman spectra of edible oils. Food Anal. Methods 2021, 14, 1826–1835. [Google Scholar] [CrossRef]
- Jiang, Y.; Su, M.; Yu, T.; Du, S.; Liao, L.; Wang, H.; Wu, Y.; Liu, H. Quantitative determination of peroxide value of edible oil by algorithm-assisted liquid interfacial surface enhanced Raman spectroscopy. Food Chem. 2021, 344, 128709. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Zhan, Y.; Xu, Z.; Nduwamungu, J.J.; Zhou, Y.; Powers, R.; Xu, C. The application of machine-learning and Raman spectroscopy for the rapid detection of edible oils type and adulteration. Food Chem. 2022, 373, 131471. [Google Scholar] [CrossRef] [PubMed]
- Berghian-Grosan, C.; Magdas, D.A. Raman spectroscopy and machine-learning for edible oils evaluation. Talanta 2020, 218, 121176. [Google Scholar] [CrossRef] [PubMed]
- Hu, R.; He, T.; Zhang, Z.; Yang, Y.; Liu, M. Safety analysis of edible oil products via Raman spectroscopy. Talanta 2019, 191, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Kwofie, F.; Lavine, B.K.; Ottaway, J.; Booksh, K. Differentiation of edible oils by type using Raman spectroscopy and pattern recognition methods. Appl. Spectrosc. 2020, 74, 645–654. [Google Scholar] [CrossRef]
- Ma, X.; Shao, B.; Wang, Z. Gold@silver nanodumbbell based inter-nanogap aptasensor for the surface enhanced Raman spectroscopy determination of ochratoxin A. Anal. Chim. Acta 2021, 1188, 339189. [Google Scholar] [CrossRef]
- Jiang, H.; Liu, G.; Mei, C.; Chen, Q. Qualitative and quantitative analysis in solid-state fermentation of protein feed by FT-NIR spectroscopy integrated with multivariate data analysis. Anal. Methods 2013, 5, 1872–1880. [Google Scholar] [CrossRef]
- Jiang, H.; Xu, W.; Ding, Y.; Chen, Q. Quantitative analysis of yeast fermentation process using Raman spectroscopy: Comparison of CARS and VCPA for variable selection. Spectrochim. Acta Part A-Mol. Biomol. Spectrosc. 2020, 228, 117781. [Google Scholar] [CrossRef]
- Jiang, H.; Xu, W.; Chen, Q. High precision qualitative identification of yeast growth phases using molecular fusion spectra. Microchem. J. 2019, 151, 104211. [Google Scholar] [CrossRef]
- Zhang, Z.-M.; Chen, S.; Liang, Y.-Z. Baseline correction using adaptive iteratively reweighted penalized least squares. Analyst 2010, 135, 1138–1146. [Google Scholar] [CrossRef]
- Li, H.; Liang, Y.; Xu, Q.; Cao, D. Key wavelengths screening using competitive adaptive reweighted sampling method for multivariate calibration. Anal. Chim. Acta 2009, 648, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Chen, X.; Li, L.; Chen, X.; Yuan, L.; Shi, W. Domain adaptive partial least squares regression. Chemom. Intell. Lab. Syst. 2020, 201, 103986. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, Y.A.; He, C.Y.; Xie, M.Y.; Luo, H.T.; Wang, Y.; Zhang, J. Rapid screen of aflatoxin-contaminated peanut oil using Fourier transform infrared spectroscopy combined with multivariate decision tree. Int. J. Food Sci. Technol. 2018, 53, 2386–2393. [Google Scholar] [CrossRef]
- Chen, M.; He, X.M.; Pang, Y.Y.; Shen, F.; Fang, Y.; Hu, Q.H. Laser induced fluorescence spectroscopy for detection of Aflatoxin B1 contamination in peanut oil. J. Food Meas. Charact. 2021, 15, 2231–2239. [Google Scholar] [CrossRef]
- Chen, Q.S.; Yang, M.X.; Yang, X.J.; Li, H.H.; Guo, Z.M.; Rahma, M.H. A large Raman scattering cross-section molecular embedded SERS aptasensor for ultrasensitive Aflatoxin B1 detection using CS-Fe3O4 for signal enrichment. Spectrochim. Acta Part A-Mol. Biomol. Spectrosc. 2018, 189, 147–153. [Google Scholar] [CrossRef] [PubMed]


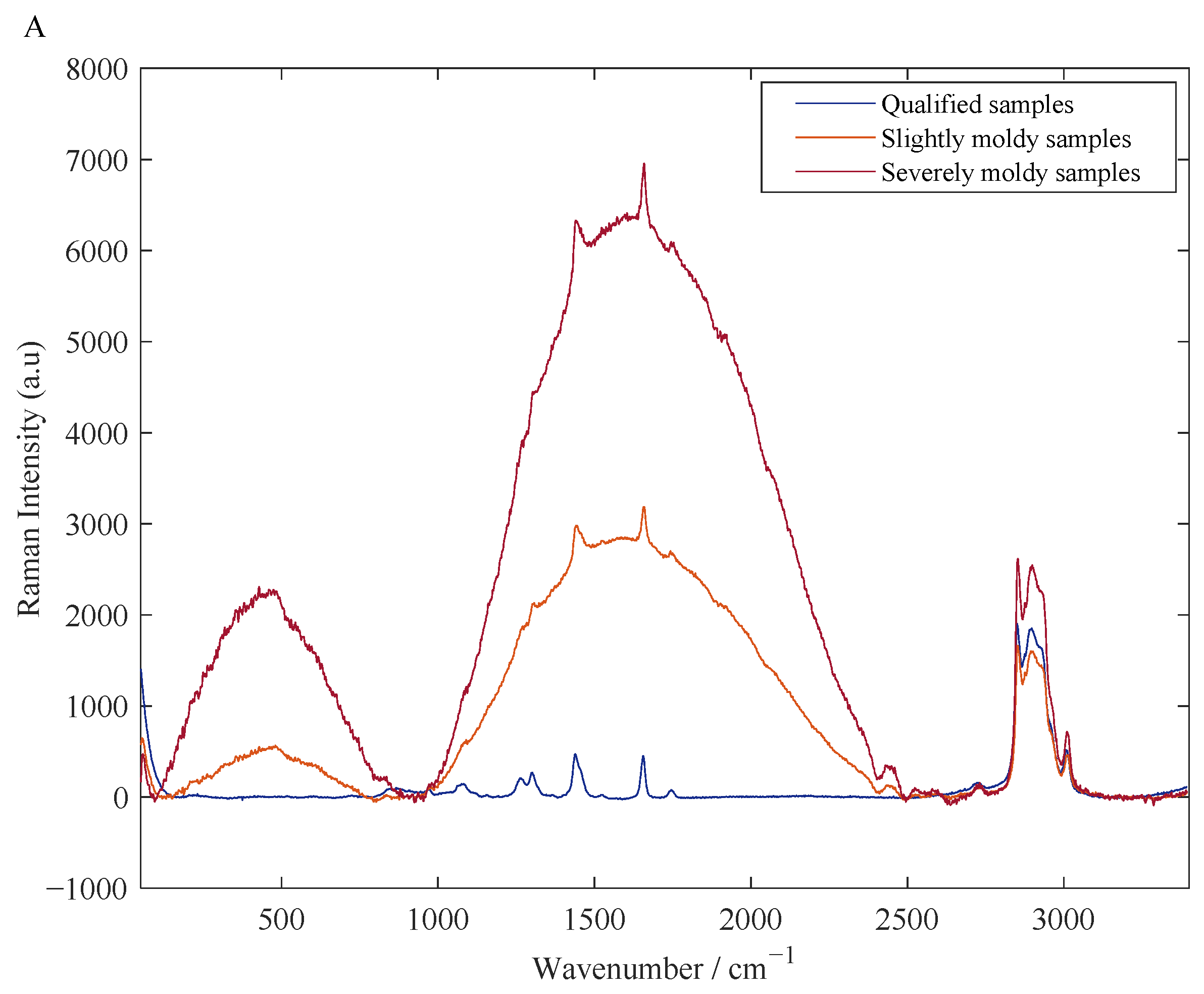
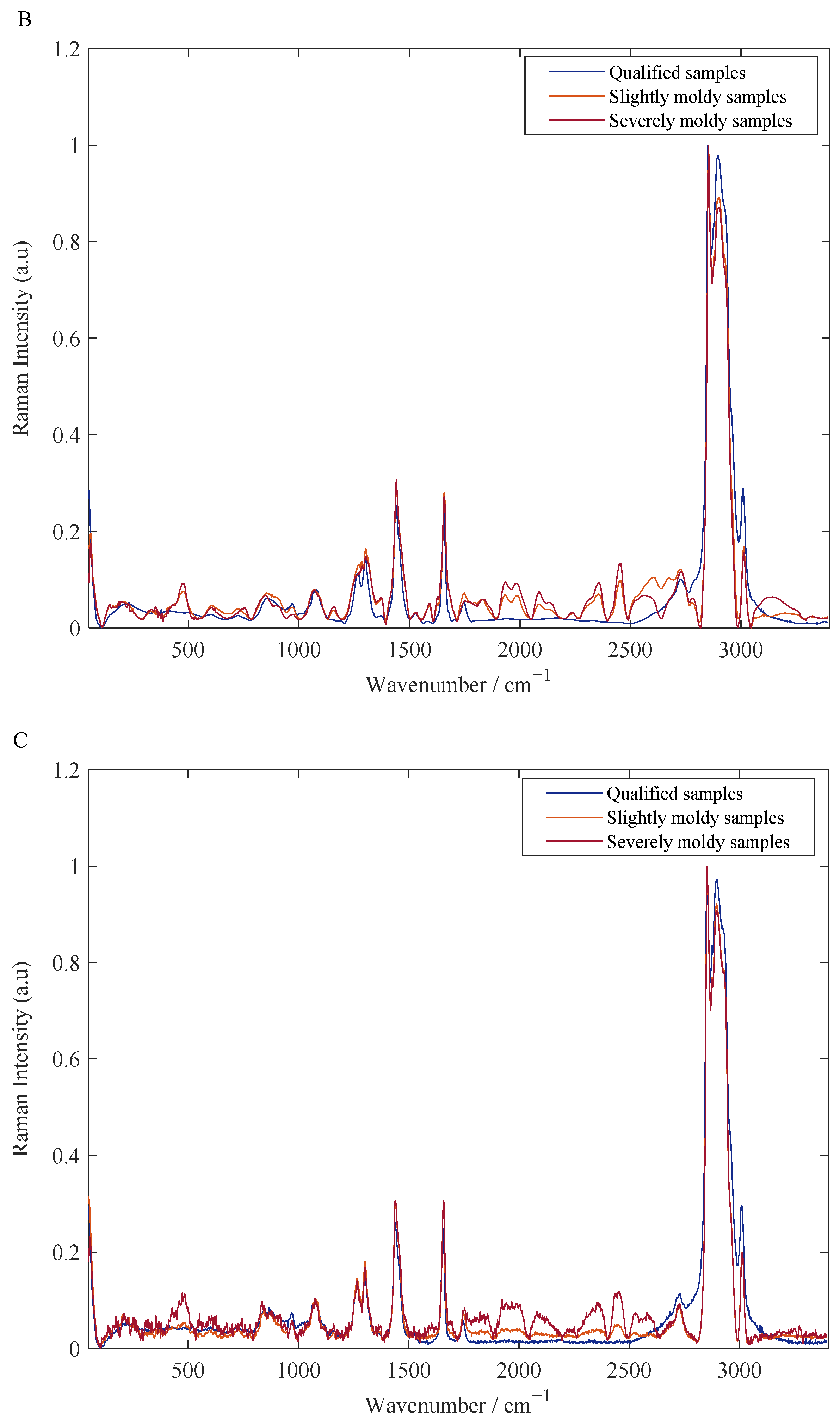


| Sample Sets | Sample Number | Maximum/μg·kg−1 | Minimum/μg·kg−1 | Mean/μg·kg−1 | Standard Deviation/μg·kg−1 |
|---|---|---|---|---|---|
| Calibration set | 64 | 701.7 | 0.097 | 236.8 | 224.2 |
| Prediction set | 16 | 691.5 | 0.11 | 237.6 | 231.0 |
| Raman Spectra Calculated by DFT (cm−1) | Raman Spectra Collected Experimentally (cm−1) | Spectral Attribution |
|---|---|---|
| 686 | 670 | Ring breath(pyrane) |
| 1076 | 1059 | v(C-C-C) |
| 1330 | 1267 | v(C-O-C) |
| 1393 | 1347 | v(C-O)(ring skeleton vibration) |
| 1603 | 1559 | C-H def, v(C-C), v(C=C) |
| 1645 | 1601 | C-H def, v(C-C), v(C=C) (ring skeleton vibration) |
| 1806 | 1701 | v(C=O) (cyclopentene ring) |
| 1883 | 1764 | v(C=O) (pyrane ring) |
| Models | Number of Variables | Parameters | Calibration Set | Prediction Set | ||
|---|---|---|---|---|---|---|
| RMSECV/μg·kg−1 | RMSEP/μg·kg−1 | |||||
| FULL–PLS | 3468 | PCs = 14 | 64.6 | 0.92 | 93.1 | 0.88 |
| DFT–PLS | 8 | PCs = 5 | 107.7 | 0.67 | 124.5 | 0.73 |
| CARS–PLS | 77 | PCs = 15 | 28.1 | 0.98 | 22.6 | 0.99 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, C.; Jiang, H.; Chen, Q. High Precisive Prediction of Aflatoxin B1 in Pressing Peanut Oil Using Raman Spectra Combined with Multivariate Data Analysis. Foods 2022, 11, 1565. https://doi.org/10.3390/foods11111565
Zhu C, Jiang H, Chen Q. High Precisive Prediction of Aflatoxin B1 in Pressing Peanut Oil Using Raman Spectra Combined with Multivariate Data Analysis. Foods. 2022; 11(11):1565. https://doi.org/10.3390/foods11111565
Chicago/Turabian StyleZhu, Chengyun, Hui Jiang, and Quansheng Chen. 2022. "High Precisive Prediction of Aflatoxin B1 in Pressing Peanut Oil Using Raman Spectra Combined with Multivariate Data Analysis" Foods 11, no. 11: 1565. https://doi.org/10.3390/foods11111565
APA StyleZhu, C., Jiang, H., & Chen, Q. (2022). High Precisive Prediction of Aflatoxin B1 in Pressing Peanut Oil Using Raman Spectra Combined with Multivariate Data Analysis. Foods, 11(11), 1565. https://doi.org/10.3390/foods11111565








