Comparison of Selected Phenotypic Features of Persistent and Sporadic Strains of Listeria monocytogenes Sampled from Fish Processing Plants
Abstract
1. Introduction
2. Materials and Methods
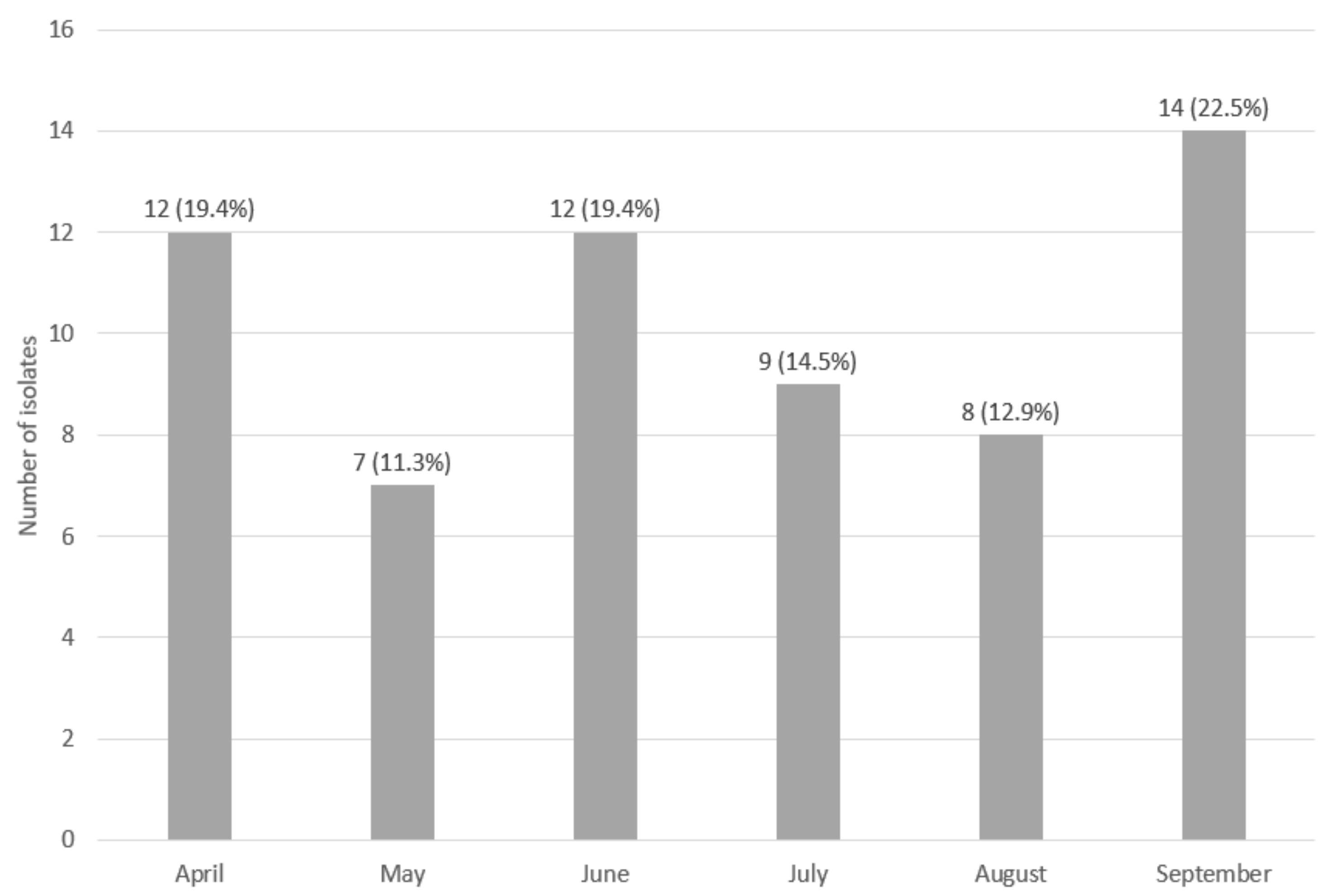
2.1. Sampling Procedure
2.2. Sampling and Identification of L. monocytogenes Isolates
2.3. Assessment of the Genetic Similarity of the Collected Isolates
2.4. Isolation of Genomic DNA
2.5. Determination of Serological Groups
2.6. Assessment of Drug Susceptibility of the Tested L. monocytogenes Strains
2.7. Comparison of the Selected Properties of Sporadic and Persistent L. monocytogenes Strains
2.7.1. Assessment of Invasiveness against HT-29 Eukaryotic Cells
2.7.2. Determination of the Minimum Bactericidal Concentration (MBC) of Selected Disinfectants against Persistent and Sporadic Strains of L. monocytogenes
2.7.3. Assessment of the Rate of Initiation of Biofilm Formation
2.7.4. Assessment of the Intensity of Biofilm Formation
2.8. Statistical Analysis
3. Results
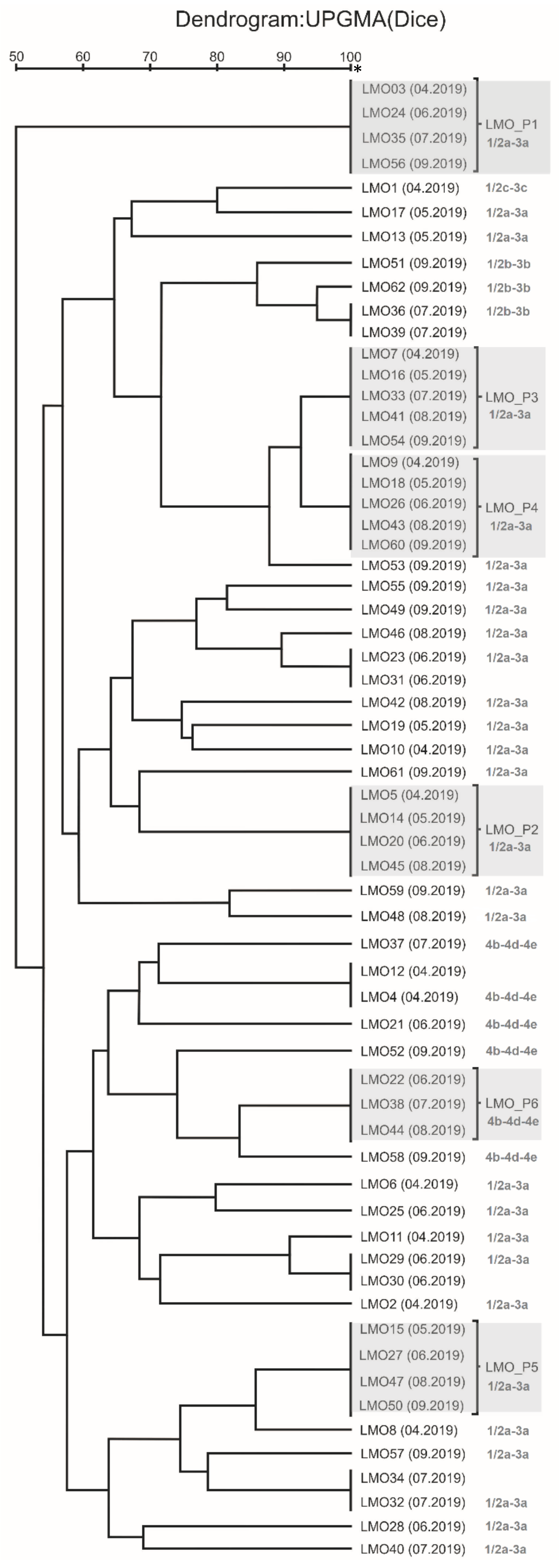
3.1. Assessment of the Genetic Similarity of Isolate Strains Classified as Persistent
- LMO-P1 (LMO 3 (April 2019), LMO 24 (June 2019), LMO 35 (July 2019), and LMO 56 (September 2019))—isolated from the trim conveyor rollers.
- LMO-P2 (LMO 5 (April 2019), LMO 14 (May 2019), LMO 20 (June 2019), and LMO 45 (August 2019))—isolated from the cutting element of the head remover machine.
- LMO-P3 (LMO 7 (April 2019), LMO 16 (May 2019), LMO 33 (July2019), LMO 41 (August 2019), and LMO 54 (September 2019))—isolated from the knives of the filleting machine.
- LMO-P4 (LMO 9 (April 2019), LMO 18 (May 2019), LMO 26 (June 2019), LMO 43 (August 2019), and LMO 60 (September 2019))—isolated from the knives of the filleting machine.
- LMO-P5 (LMO 15 (May 2019), LMO 27 (June 2019), LMO 47 (August 2019), and LMO 50 (September 2019))—isolated from the skin remover machine.
- LMO-P6 (LMO 22 (June 2019), LMO 38 (July 2019), and LMO 44 (August 2019))—isolated from the trommel of the pin bone remover.
3.2. Molecular Serotyping of L. monocytogenes Strains
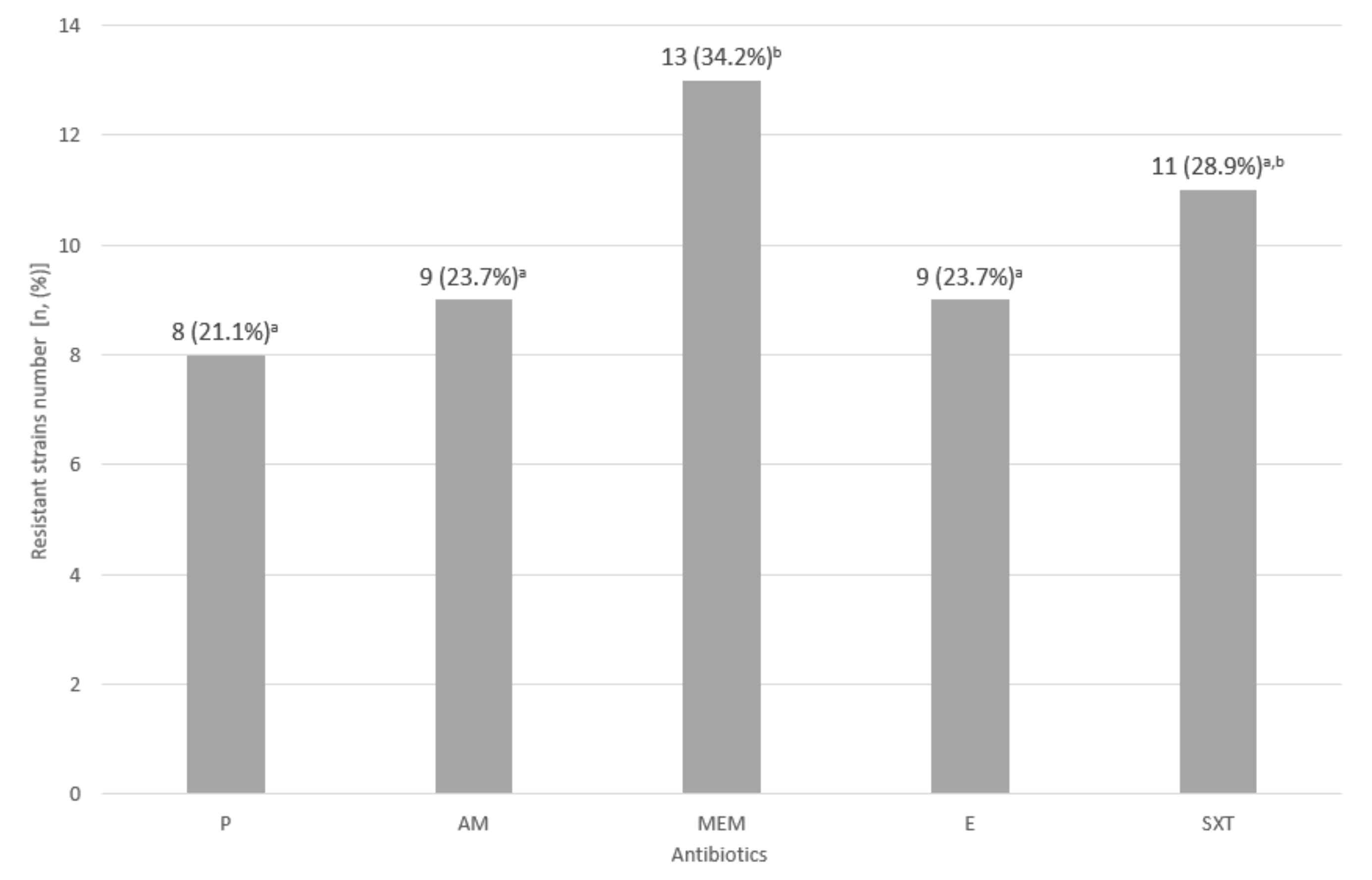
3.3. Assessment of Drug Susceptibility of the Tested L. monocytogenes Strains
3.4. Assessment of Invasiveness against HT-29 Eukaryotic Cells
3.5. Minimum Bactericidal Concentrations (MBC) of the Selected Disinfectants against the Persistent and Sporadic Strains of L. monocytogenes
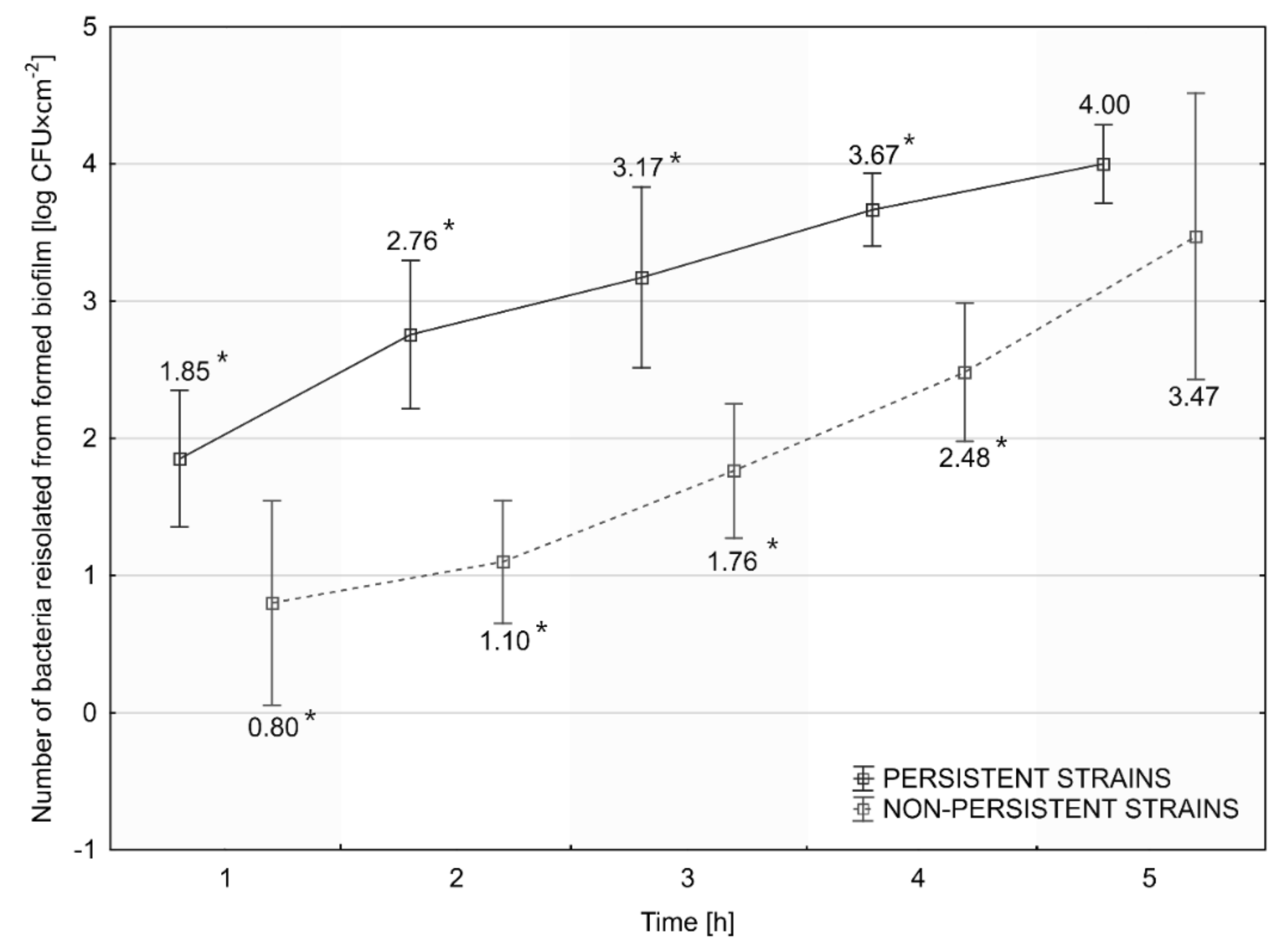
3.6. Assessment of the Rate of Initiation of Biofilm Formation
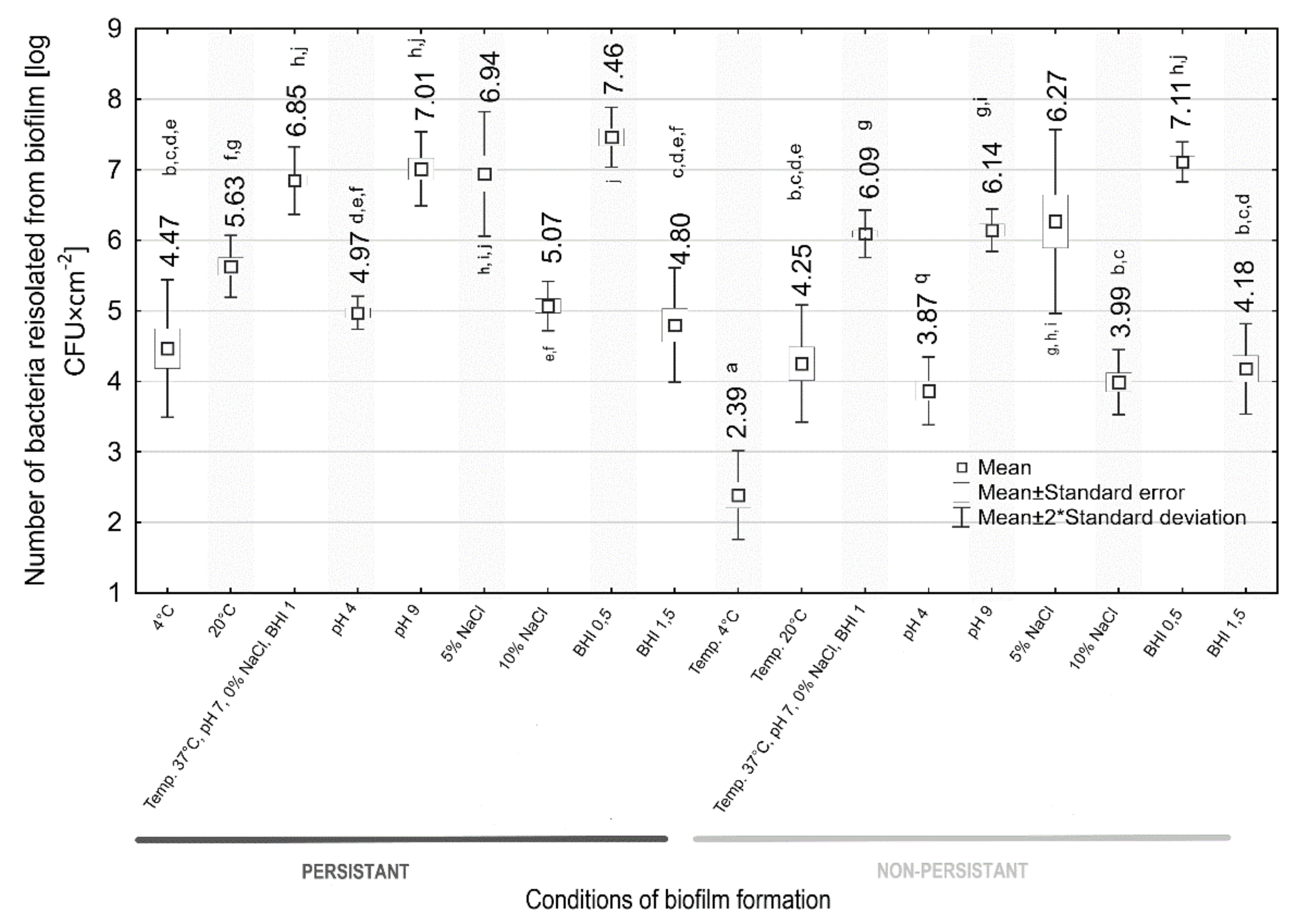
3.7. Assessment of the Intensity of Biofilm Formation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Carpentier, B.; Cerf, O. Review—Persistence of Listeria monocytogenes in food industry equipment and premises. Int. J. Food Microbiol. 2011, 145, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Lundén, J. Persistent Listeria monocytogenes Contamination in Food Processing Plants. Ph.D. Thesis, University of Helsinki, Helsinki, Finland, January 2004. Available online: http://urn.fi/URN:ISBN:952-10-1507-1 (accessed on 12 November 2021).
- Larsen, M.H.; Dalmasso, M.; Ingmer, H.; Langsrud, S.; Malakauskas, M.; Mader, A.; Møretrø, T.; Možina, S.S.; Rychli, K.; Wagner, M.; et al. Persistence of foodborne pathogens and their control in primary and secondary food production chains. Food Control 2014, 44, 92–109. [Google Scholar] [CrossRef]
- Unrath, N.; McCabe, E.; Macori, G.; Fanning, S. Application of Whole Genome Sequencing to Aid in Deciphering the Persistence Potential of Listeria monocytogenes in Food Production Environments. Microorganisms 2021, 9, 1856. [Google Scholar] [CrossRef] [PubMed]
- Axelsson, L.; Bjerke, G.A.; McLeod, A.; Berget, I.; Holck, A.L. Growth Behavior of Listeria monocytogenes in a Traditional Norwegian Fermented Fish Product (Rakfisk), and Its Inhibition through Bacteriophage Addition. Foods 2020, 9, 119. [Google Scholar] [CrossRef]
- Shamloo, E.; Hosseini, H.; Moghadam, Z.A.; Larsen, M.H.; Haslberger, A.; Alebouyeh, M. Importance of Listeria monocytogenes in food safety: A review of its prevalence, detection, and antibiotic resistance. Iran. J. Vet. Res. 2019, 20, 241–254. [Google Scholar]
- Szymczak, B.; Szymczak, M.; Trafiałek, J. Prevalence of Listeria species and L. monocytogenes in ready-to-eat foods in the West Pomeranian region of Poland: Correlations between the contamination level, serogroups, ingredients, and producers. Food Microbiol. 2020, 91, 103532. [Google Scholar] [CrossRef]
- Kurpas, M.; Osek, J.; Moura, A.; Leclercq, A.; Lecuit, M.; Wieczorek, K. Genomic Characterization of Listeria monocytogenes Isolated from Ready-to-Eat Meat and Meat Processing Environments in Poland. Front. Microbiol. 2020, 11, 1412. [Google Scholar] [CrossRef]
- Kurpas, M.; Wieczorek, K.; Osek, J. Ready-to-eat Meat Products As a Source of Listeria monocytogenes. J. Veter. Res. 2018, 62, 49–55. [Google Scholar] [CrossRef]
- Cherifi, T.; Arsenault, J.; Pagotto, F.; Quessy, S.; Côté, J.-C.; Neira, K.; Fournaise, S.; Bekal, S.; Fravalo, P. Distribution, diversity and persistence of Listeria monocytogenes in swine slaughterhouses and their association with food and human listeriosis strains. PLoS ONE 2020, 15, e0236807. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Listeriosis. Available online: https://www.who.int/news-room/fact-sheets/detail/listeriosis (accessed on 21 April 2022).
- Smith, A.M.; Tau, N.P.; Smouse, S.L.; Allam, M.; Ismail, A.; Ramalwa, N.R.; Disenyeng, B.; Ngomane, M.; Thomas, J. Outbreak of Listeria monocytogenes in South Africa, 2017–2018: Laboratory Activities and Experiences Associated with Whole-Genome Sequencing Analysis of Isolates. Foodborne Pathog. Dis. 2019, 16, 524–530. [Google Scholar] [CrossRef]
- Abdallah, M.; Benoliel, C.; Drider, D.; Dhulster, P.; Chihib, N.-E. Biofilm formation and persistence on abiotic surfaces in the context of food and medical environments. Arch. Microbiol. 2014, 196, 453–472. [Google Scholar] [CrossRef]
- Fagerlund, A.; Langsrud, S.; Møretrø, T. In-Depth Longitudinal Study of Listeria monocytogenes ST9 Isolates from the Meat Processing Industry: Resolving Diversity and Transmission Patterns Using Whole-Genome Sequencing. Appl. Environ. Microbiol. 2020, 86, e00579-20. [Google Scholar] [CrossRef]
- Commission Regulation (EC) No 2073/2005. Commission Regulation (EC) No 2073/2005 of 15 November 2005 on Microbiological Criteria for Foodstuffs (Text with EEA Relevance); European Commission: Brussels, Belgium, 2005. [Google Scholar]
- Balaban, N.Q.; Helaine, S.; Lewis, K.; Ackermann, M.; Aldridge, B.; Andersson, D.I.; Brynildsen, M.P.; Bumann, D.; Camilli, A.; Collins, J.J.; et al. Definitions and guidelines for research on antibiotic persistence. Nat. Rev. Genet. 2019, 17, 441–448. [Google Scholar] [CrossRef]
- Fagerlund, A.; Langsrud, S.; Schirmer, B.C.; Møretrø, T.; Heir, E. Genome Analysis of Listeria monocytogenes Sequence Type 8 Strains Persisting in Salmon and Poultry Processing Environments and Comparison with Related Strains. PLoS ONE 2016, 11, e0151117. [Google Scholar] [CrossRef]
- Leong, D.; Alvarez-Ordonez, A.; Jordan, K. Monitoring occurrence and persistence of Listeria monocytogenes in foods and food processing environments in the Republic of Ireland. Front. Microbiol. 2014, 5, 436. [Google Scholar] [CrossRef]
- Costa, A.; Bertolotti, L.; Brito, L.; Civera, T. Biofilm Formation and Disinfectant Susceptibility of Persistent and Nonpersistent Listeria monocytogenes Isolates from Gorgonzola Cheese Processing Plants. Foodborne Pathog. Dis. 2016, 13, 602–609. [Google Scholar] [CrossRef]
- Fox, E.M.; Leonard, N.; Jordan, K. Physiological and Transcriptional Characterization of Persistent and Nonpersistent Listeria monocytogenes Isolates. Appl. Environ. Microbiol. 2011, 77, 6559–6569. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control. Multi-country outbreak of Listeria monocytogenes clonal complex 8 infections linked to consumption of cold-smoked fish products. EFSA Support. Publ. 2019, 16, 1665E. [Google Scholar] [CrossRef][Green Version]
- Lundén, J.; Tolvanen, R.; Korkeala, H. Acid and heat tolerance of persistent and nonpersistent Listeria monocytogenes food plant strains. Lett. Appl. Microbiol. 2008, 46, 276–280. [Google Scholar] [CrossRef]
- Magalhães, R.; Ferreira, V.; Brandão, T.; Palencia, R.C.; Almeida, G.; Teixeira, P. Persistent and non-persistent strains of Listeria monocytogenes: A focus on growth kinetics under different temperature, salt, and pH conditions and their sensitivity to sanitizers. Food Microbiol. 2016, 53, 103–108. [Google Scholar] [CrossRef]
- Martínez-Suárez, J.V.; Ortiz, S.; López-Alonso, V. Potential Impact of the Resistance to Quaternary Ammonium Disinfectants on the Persistence of Listeria monocytogenes in Food Processing Environments. Front. Microbiol. 2016, 7, 638. [Google Scholar] [CrossRef]
- Bucur, F.I.; Grigore-Gurgu, L.; Crauwels, P.; Riedel, C.U.; Nicolau, A.I. Resistance of Listeria monocytogenes to Stress Conditions Encountered in Food and Food Processing Environments. Front. Microbiol. 2018, 9, 2700. [Google Scholar] [CrossRef]
- Lundén, J.M.; Miettinen, M.K.; Autio, T.J.; Korkeala, H. Persistent Listeria monocytogenes Strains Show Enhanced Adherence to Food Contact Surface after Short Contact Times. J. Food Prot. 2000, 63, 1204–1207. [Google Scholar] [CrossRef]
- Nowak, J.; Visnovsky, S.B.; Pitman, A.R.; Cruz, C.D.; Palmer, J.; Fletcher, G.C.; Flint, S. Biofilm Formation by Listeria monocytogenes 15G01, a Persistent Isolate from a Seafood-Processing Plant, Is Influenced by Inactivation of Multiple Genes Belonging to Different Functional Groups. Appl. Environ. Microbiol. 2021, 87, e02349-20. [Google Scholar] [CrossRef]
- Lundén, J.M.; Autio, T.J.; Sjöberg, A.-M.; Korkeala, H.J. Persistent and Nonpersistent Listeria monocytogenes Contamination in Meat and Poultry Processing Plants. J. Food Prot. 2003, 66, 2062–2069. [Google Scholar] [CrossRef]
- ISO 11290-1; Microbiology of the food chain—Horizontal method for the detection and enumeration of Listeria mono-cytogenes and of Listeria spp.—Part 1: Detection method. ISO: Geneva, Switzerland.
- Dalmasso, M.; Jordan, K. PFGE as a Tool to Track Listeria monocytogenes in Food Processing Facilities: Case Studies. Pulse Field Gel Electrophoresis. In Methods in Molecular Biology; Jordan, K., Dalmasso, M., Eds.; Humana Press: New York, NY, USA, 2015; Volume 1301, Available online: https://doi.org/10.1007/978-1-4939-2599-5_3 (accessed on 20 November 2021).
- PNL04. Standard Operating Procedure for PulseNet PFGE of Listeria monocytogenes. Available online: https://pulsenetinternational.org/assets/PulseNet/uploads/pfge/PNL04_ListeriaPFGEProtocol.pdf (accessed on 25 November 2019).
- Doumith, M.; Buchrieser, C.; Glaser, P.; Jacquet, C.; Martin, P. Differentiation of the Major Listeria monocytogenes Serovars by Multiplex PCR. J. Clin. Microbiol. 2004, 42, 3819–3822. [Google Scholar] [CrossRef]
- Wałecka-Zacharska, E.; Kosek-Paszkowska, K.; Bania, J.; Karpíšková, R.; Stefaniak, T. Salt stress-induced invasiveness of major Listeria monocytogenes serotypes. Lett. Appl. Microbiol. 2013, 56, 216–221. [Google Scholar] [CrossRef]
- Kuch, A.; Goc, A.; Belkiewicz, K.; Filipello, V.; Ronkiewicz, P.; Gołębiewska, A.; Wróbel, I.; Kiedrowska, M.; Waśko, I.; Hryniewicz, W.; et al. Molecular diversity and antimicrobial susceptibility of Listeria monocytogenes isolates from invasive infections in Poland (1997–2013). Sci. Rep. 2018, 8, 14562. [Google Scholar] [CrossRef]
- European Committee on Antimicrobial Susceptibility Testing (EUCAST). Breakpoints Tables for Interpretation of MICs and Zones Diameters. Version 12.0. 2022. Available online: http://www.eucast.org (accessed on 15 April 2022).
- Skowron, K.; Wałecka-Zacharska, E.; Grudlewska, K.; Białucha, A.; Wiktorczyk, N.; Bartkowska, A.; Kowalska, M.; Kruszewski, S.; Gospodarek-Komkowska, E. Biocidal Effectiveness of Selected Disinfectants Solutions Based on Water and Ozonated Water against Listeria monocytogenes Strains. Microorganisms 2019, 7, 127. [Google Scholar] [CrossRef]
- Di Ciccio, P.; Meloni, D.; Festino, A.R.; Conter, M.; Zanardi, E.; Ghidini, S.; Vergara, A.; Mazzette, R.; Ianieri, A. Longitudinal study on the sources of Listeria monocytogenes contamination in cold-smoked salmon and its processing environment in Italy. Int. J. Food Microbiol. 2012, 158, 79–84. [Google Scholar] [CrossRef]
- Leong, D.; NicAogáin, K.; Luque-Sastre, L.; McManamon, O.; Hunt, K.; Alvarez-Ordóñez, A.; Scollard, J.; Schmalenberger, A.; Fanning, S.; O’Byrne, C.; et al. A 3-year multi-food study of the presence and persistence of Listeria monocytogenes in 54 small food businesses in Ireland. Int. J. Food Microbiol. 2017, 249, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Miettinen, H.; Wirtanen, G. Ecology of Listeria spp. in a fish farm and molecular typing of Listeria monocytogenes from fish farming and processing companies. Int. J. Food Microbiol. 2006, 112, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Ramires, T.; Kleinubing, N.R.; Iglesias, M.A.; Vitola, H.R.S.; Núncio, A.S.P.; Kroning, I.S.; Moreira, G.M.S.G.; Fiorentini, M.; da Silva, W.P. Genetic diversity, biofilm and virulence characteristics of Listeria monocytogenes in salmon sushi. Food Res. Int. 2020, 140, 109871. [Google Scholar] [CrossRef] [PubMed]
- Aalto-Araneda, M.; Lundén, J.; Markkula, A.; Hakola, S.; Korkeala, H. Processing plant and machinery sanitation and hygiene practices associate with Listeria monocytogenes occurrence in ready-to-eat fish products. Food Microbiol. 2019, 82, 455–464. [Google Scholar] [CrossRef]
- Mędrala, D.; Dąbrowski, W.; Czekajło-Kołodziej, U.; Daczkowska-Kozon, E.; Koronkiewicz, A.; Augustynowicz, E.; Manzano, M. Persistence of Listeria monocytogenes strains isolated from products in a Polish fish-processing plant over a 1-year period. Food Microbiol. 2003, 20, 715–724. [Google Scholar] [CrossRef]
- Cruz, C.D.; Fletcher, G. Prevalence and biofilm-forming ability of Listeria monocytogenes in New Zealand mussel (Perna canaliculus) processing plants. Food Microbiol. 2011, 28, 1387–1393. [Google Scholar] [CrossRef]
- Skowron, K.; Wałecka-Zacharska, E.; Grudlewska, K.; Gajewski, P.; Wiktorczyk, N.; Wietlicka-Piszcz, M.; Dudek, A.; Skowron, K.J.; Gospodarek-Komkowska, E. Disinfectant Susceptibility of Biofilm Formed by Listeria monocytogenes under Selected Environmental Conditions. Microorganisms 2019, 7, 280. [Google Scholar] [CrossRef]
- Jensen, A.; Larsen, M.H.; Ingmer, H.; Vogel, B.F.; Gram, L. Sodium Chloride Enhances Adherence and Aggregation and Strain Variation Influences Invasiveness of Listeria monocytogenes Strains. J. Food Prot. 2007, 70, 592–599. [Google Scholar] [CrossRef]
- Taylor, A.; Stasiewicz, M.J. Persistent and sporadic Listeria monocytogenes strains do not differ when growing at 37 °C, in planktonic state, under different food associated stresses or energy sources. BMC Microbiol. 2019, 19, 257. [Google Scholar] [CrossRef]
- Nowak, J.; Cruz, C.D.; Tempelaars, M.; Abee, T.; van Vliet, A.; Fletcher, G.C.; Hedderley, D.; Palmer, J.; Flint, S. Persistent Listeria monocytogenes strains isolated from mussel production facilities form more biofilm but are not linked to specific genetic markers. Int. J. Food Microbiol. 2017, 256, 45–53. [Google Scholar] [CrossRef]
- Assisi, C.; Forauer, E.; Oliver, H.F.; Etter, A.J. Genomic and Transcriptomic Analysis of Biofilm Formation in Persistent and Transient Listeria monocytogenes Isolates from the Retail Deli Environment Does Not Yield Insight into Persistence Mechanisms. Foodborne Pathog. Dis. 2020, 18, 179–188. [Google Scholar] [CrossRef]
- Wang, J.; Ray, A.J.; Hammons, S.R.; Oliver, H.F. Persistent and Transient Listeria monocytogenes Strains from Retail Deli Environments Vary in Their Ability to Adhere and Form Biofilms and Rarely HaveinlAPremature Stop Codons. Foodborne Pathog. Dis. 2015, 12, 151–158. [Google Scholar] [CrossRef]
- Gambarin, P.; Magnabosco, C.; Losio, M.N.; Pavoni, E.; Gattuso, A.; Arcangeli, G.; Favretti, M. Listeria monocytogenes in Ready-to-Eat Seafood and Potential Hazards for the Consumers. Int. J. Microbiol. 2012, 2012, 497635. [Google Scholar] [CrossRef]
- Skowron, K.; Kwiecińska-Piróg, J.; Grudlewska, K.; Świeca, A.; Paluszak, Z.; Bauza-Kaszewska, J.; Walecka-Zacharska, E.; Gospodarek-Komkowska, E. The occurrence, transmission, virulence and antibiotic resistance of Listeria monocytogenes in fish processing plant. Int. J. Food Microbiol. 2018, 282, 71–83. [Google Scholar] [CrossRef]
- Pizarro-Cerdá, J.; Kühbacher, A.; Cossart, P. Entry of Listeria monocytogenes in Mammalian Epithelial Cells: An Updated View. Cold Spring Harb. Perspect. Med. 2012, 2, a010009. [Google Scholar] [CrossRef]
- Povolyaeva, O.; Chalenko, Y.; Kalinin, E.; Kolbasova, O.; Pivova, E.; Kolbasov, D.; Yurkov, S.; Ermolaeva, S. Listeria monocytogenes Infection of Bat Pipistrellus nathusii Epithelial cells Depends on the Invasion Factors InlA and InlB. Pathogens 2020, 9, 867. [Google Scholar] [CrossRef]
- Moroni, O.; Kheadr, E.; Boutin, Y.; Lacroix, C.; Fliss, I. Inactivation of Adhesion and Invasion of Food-Borne Listeria monocytogenes by Bacteriocin-Producing Bifidobacterium Strains of Human Origin. Appl. Environ. Microbiol. 2006, 72, 6894–6901. [Google Scholar] [CrossRef]
- Porsby, C.H.; Vogel, B.F.; Mohr, M.; Gram, L. Influence of processing steps in cold-smoked salmon production on survival and growth of persistent and presumed non-persistent Listeria monocytogenes. Int. J. Food Microbiol. 2008, 122, 287–295. [Google Scholar] [CrossRef]
| Group of Disinfectants | Trade Name | Active Substances | Manufacturer | Working Solution Concentration |
|---|---|---|---|---|
| Quaternary ammonium compounds | Sansept 0200 | Didecyldimethylammonium chloride, benzyl-C12-16-alkyldimethyl chlorides | Sanechem | 3 mL/L |
| Oxidizing agents | Peroxat | Peracetic acid, hydrogen peroxide | Agro-trade | 5 mL/L |
| Chlorine compounds | Calcium hypochlorite | Hypochlorous acid calcium salt | Chem Point | 2 g/L |
| Iodine compounds | Rapicid | Iodine | Pfizer | 10 mL/L |
| Environment Parameter | Experimental Conditions Set for Biofilm Formation | Temperature (°C) | pH | Salinity (% NaCl) | Nutrient Availability (BHI) |
|---|---|---|---|---|---|
| Temperature (°C) | 1 | 4 | 7 | 0 | 1.0 |
| 2 | 20 | 7 | 0 | 1.0 | |
| 3 | 37 | 7 | 0 | 1.0 | |
| pH | 4 | 37 | 4 | 0 | 1.0 |
| 5 | 37 | 7 | 0 | 1.0 | |
| 6 | 37 | 9 | 0 | 1.0 | |
| Salinity (% NaCl) | 7 | 37 | 7 | 0 | 1.0 |
| 8 | 37 | 7 | 5 | 1.0 | |
| 9 | 37 | 7 | 10 | 1.0 | |
| Nutrient availability (BHI) | 10 | 37 | 7 | 0 | 0.5 * |
| 11 | 37 | 7 | 0 | 1.0 * | |
| 12 | 37 | 7 | 0 | 1.5 * |
| Element of the Processing Line | Number (%) of All Isolates | Isolates * | |
|---|---|---|---|
| Box pallets in the raw material warehouse | 3 (4.8) | LMO 36, LMO 39, LMO 62 | |
| Head remover machine (cutting element) | 7 (11.3) | LMO-P2 (LMO 5, LMO 14, LMO 20, LMO 45) LMO 48, LMO 55, LMO 61 | |
| Filleting machine | Conveyor | 3 (4.8) | LMO 40, LMO 49, LMO 52 |
| Knives | 14 (22.6) | LMO-P3 (LMO 7, LMO 16, LMO 33, LMO 41, LMO 54), LMO-P4 (LMO 9, LMO 18, LMO 26, LMO 43, LMO 60) LMO 2, LMO 13, LMO 51, LMO 53 | |
| Trim conveyor | Conveyor rollers | 7 (11.3) | LMO-P1 (LMO 3, LMO 24, LMO 35, LMO 56) LMO 17, LMO 25, LMO 59 |
| Worktops | 3 (4.8) | LMO 23, LMO 31, LMO 46 | |
| Pin bone remover | Conveyor | 3 (4.8) | LMO 6, LMO 21, LMO 42 |
| Trommel | 4 (6.5) | LMO-P6 (LMO 22, LMO 38, LMO 44) LMO 58 | |
| Skin remover machine | 8 (12.9) | LMO-P5 (LMO 15, LMO 27, LMO 47, LMO 50) LMO 8, LMO 32, LMO 34, LMO 57 | |
| Fillet washer | 3 (4.8) | LMO 11, LMO 29, LMO 30 | |
| Portion machine | Conveyor | 4 (6.5) | LMO 1, LMO 10, LMO 19, LMO 28 |
| Knives | 3 (4.8) | LMO4, LMO 12, LMO 37 | |
| Profile Number | Antibiotic Resistance Profile | Number (%) of Strains | Isolates and Strains Representing the Profile |
|---|---|---|---|
| 1 | R: --- S: P, AM, MEM, E, SXT | 18 (47.4) a | LMO 2, LMO 6, LMO 8, LMO 10, LMO 11, LMO 19, LMO 21, LMO 25, LMO 28, LMO 29, LMO 32, LMO 36, LMO 37, LMO 40, LMO 42, LMO 51, LMO 57, LMO 62 |
| 2 | R: AM S: P, MEM, E, SXT | 5 (13.2) b | LMO 1, LMO 13, LMO 17, LMO 49, LMO 55 |
| 3 | R: MEM, E, SXT S: P, AM | 5 (13.2) b | LMO-P1 *, LMO-P2 LMO 23, LMO 46, LMO 61 |
| 4 | R: P, MEM S: AM, E, SXT | 4 (10.5) b | LMO-P6 LMO 4, LMO 52, LMO 58 |
| 5 | R: P, AM, MEM, E, SXT S: --- | 4 (10.5) b | LMO-P3, LMO-P4, LMO-P5 LMO 53 |
| 6 | R: SXT S: P, AM, MEM, E | 2 (5.3) b | LMO 48, LMO 59 |
| Persistent Strains | Sporadic Strains | ||
|---|---|---|---|
| Strain | % | Strain | % |
| LMO-P1 | 3.42 ± 0.70 a,b | LMO 4 | 1.42 ± 0.17 a |
| LMO-P2 | 2.05 ± 0.35 a | LMO 23 | 5.59 ± 1.14 b |
| LMO-P3 | 9.55 ± 4.37 c | LMO 46 | 4.25 ± 0.93 b |
| LMO-P4 | 11.21 ± 3.19 d | LMO 52 | 2.10 ± 0.69 a |
| LMO-P5 | 5.71 ± 1.66 b | LMO 53 | 6.22 ± 1.84 b,e |
| LMO-P6 | 1.07 ± 0.39 a | LMO 61 | 7.99 ± 1.11 c,e |
| Mean | 5.50 ± 3.71 * | Mean | 4.60 ± 2.29 * |
| Disinfectant | PERSISTENT STRAINS | SPORADIC STRAINS | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| LMO-P1 | LMO-P2 | LMO-P3 | LMO-P4 | LOM-P5 | LMO-P6 | MEAN | LMO 4 | LMO 23 | LMO 46 | LMO 52 | LMO 53 | LMO 61 | MEAN | |
| Sansept 0200 [mL/L] | 0.81 ± 0.11 (27%) | 0.81 ± 0.18 (27%) | 1.17 ± 0.27 (39%) | 1.47 ± 0.31 (49%) | 0.93 ± 0.09 (31%) | 0.72 ± 0.15 (24%) | 0.99 ± 0.28 a (33%) | 0.15 ± 0.04 (5%) | 0.21 ± 0.10 (7%) | 0.21 ± 0.06 (7%) | 0.18 ± 0.07 (6%) | 0.27 ± 0.02 (9%) | 0.30 ± 0.12 (10%) | 0.22 ± 0.06 b (7%) |
| Peroxat [mL/L] | 3.50 ± 1.02 (70%) | 3.10 ± 0.68 (62%) | 4.10 ± 0.91 (82%) | 4.60 ± 1.16 (92%) | 3.85 ± 0.74 (77%) | 3.10 ± 1.00 (62%) | 3.71 ± 0.59 a (74%) | 1.75 ± 0.51 (35%) | 2.10 ± 0.43 (42%) | 2.05 ± 1.40 (41%) | 1.85 ± 0.82 (37%) | 2.50 ± 0.99 (50%) | 2.90 ± 0.38 (58%) | 2.19 ± 0.43 b (44%) |
| Calcium hypochlorite [g/L] | 0.68 ± 0.23 (34%) | 0.54 ± 0.10 (27%) | 0.78 ± 0.31 (39%) | 0.90 ± 0.09 (45%) | 0.80 ± 0.66 (40%) | 0.60 ± 0.17 (30%) | 0.72 ± 0.14 a (36%) | 0.30 ± 0.08 (15%) | 0.34 ± 0.13 (17%) | 0.38 ± 0.20 (19%) | 0.34 ± 0.14 (17%) | 0.48 ± 0.07 (24%) | 0.40 ± 0.23 (20%) | 0.37 ± 0.06 b (19%) |
| Rapicid [mL/L] | 3.10 ± 0.71 (31%) | 3.20 ± 0.52 (32%) | 3.80 ± 0.84 (38%) | 4.00 ± 1.11 (40%) | 3.80 ± 0.95 (38%) | 2.70 ± 0.36 (27%) | 3.43 ± 0.51 a (34%) | 2.80 ± 0.29 (28%) | 3.10 ± 0.41 (31%) | 3.10 ± 0.37 (31%) | 3.00 ± 0.19 (30%) | 3.10 ± 0.28 (31%) | 3.60 ± 0.37 (36%) | 3.12 ± 0.26 a (31%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wiktorczyk-Kapischke, N.; Wałecka-Zacharska, E.; Skowron, K.; Kijewska, A.; Bernaciak, Z.; Bauza-Kaszewska, J.; Kraszewska, Z.; Gospodarek-Komkowska, E. Comparison of Selected Phenotypic Features of Persistent and Sporadic Strains of Listeria monocytogenes Sampled from Fish Processing Plants. Foods 2022, 11, 1492. https://doi.org/10.3390/foods11101492
Wiktorczyk-Kapischke N, Wałecka-Zacharska E, Skowron K, Kijewska A, Bernaciak Z, Bauza-Kaszewska J, Kraszewska Z, Gospodarek-Komkowska E. Comparison of Selected Phenotypic Features of Persistent and Sporadic Strains of Listeria monocytogenes Sampled from Fish Processing Plants. Foods. 2022; 11(10):1492. https://doi.org/10.3390/foods11101492
Chicago/Turabian StyleWiktorczyk-Kapischke, Natalia, Ewa Wałecka-Zacharska, Krzysztof Skowron, Agnieszka Kijewska, Zuzanna Bernaciak, Justyna Bauza-Kaszewska, Zuzanna Kraszewska, and Eugenia Gospodarek-Komkowska. 2022. "Comparison of Selected Phenotypic Features of Persistent and Sporadic Strains of Listeria monocytogenes Sampled from Fish Processing Plants" Foods 11, no. 10: 1492. https://doi.org/10.3390/foods11101492
APA StyleWiktorczyk-Kapischke, N., Wałecka-Zacharska, E., Skowron, K., Kijewska, A., Bernaciak, Z., Bauza-Kaszewska, J., Kraszewska, Z., & Gospodarek-Komkowska, E. (2022). Comparison of Selected Phenotypic Features of Persistent and Sporadic Strains of Listeria monocytogenes Sampled from Fish Processing Plants. Foods, 11(10), 1492. https://doi.org/10.3390/foods11101492







