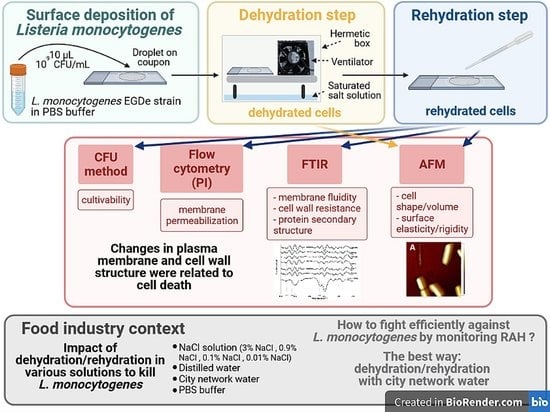
Management of Listeria monocytogenes on Surfaces via Relative Air Humidity: Key Role of Cell Envelope
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strain and Growth Conditions
2.2. Preparation of Environmental Drying Chambers
2.3. Desiccation and Rehydration Bacteria Processes
2.4. Measurement of Cell Cultivability
2.5. Determination of Changes in Cell Component Structure by FTIR
2.6. Determination of Cell Integrity by Flow Cytometry
2.7. Observation of L. monocytogenes by AFM
2.8. Measurement of Bacterial Cell Envelope Elasticity by AFM in Force Volume Mode and Young’s Modulus
2.9. Statistical Analysis
3. Results and Discussion
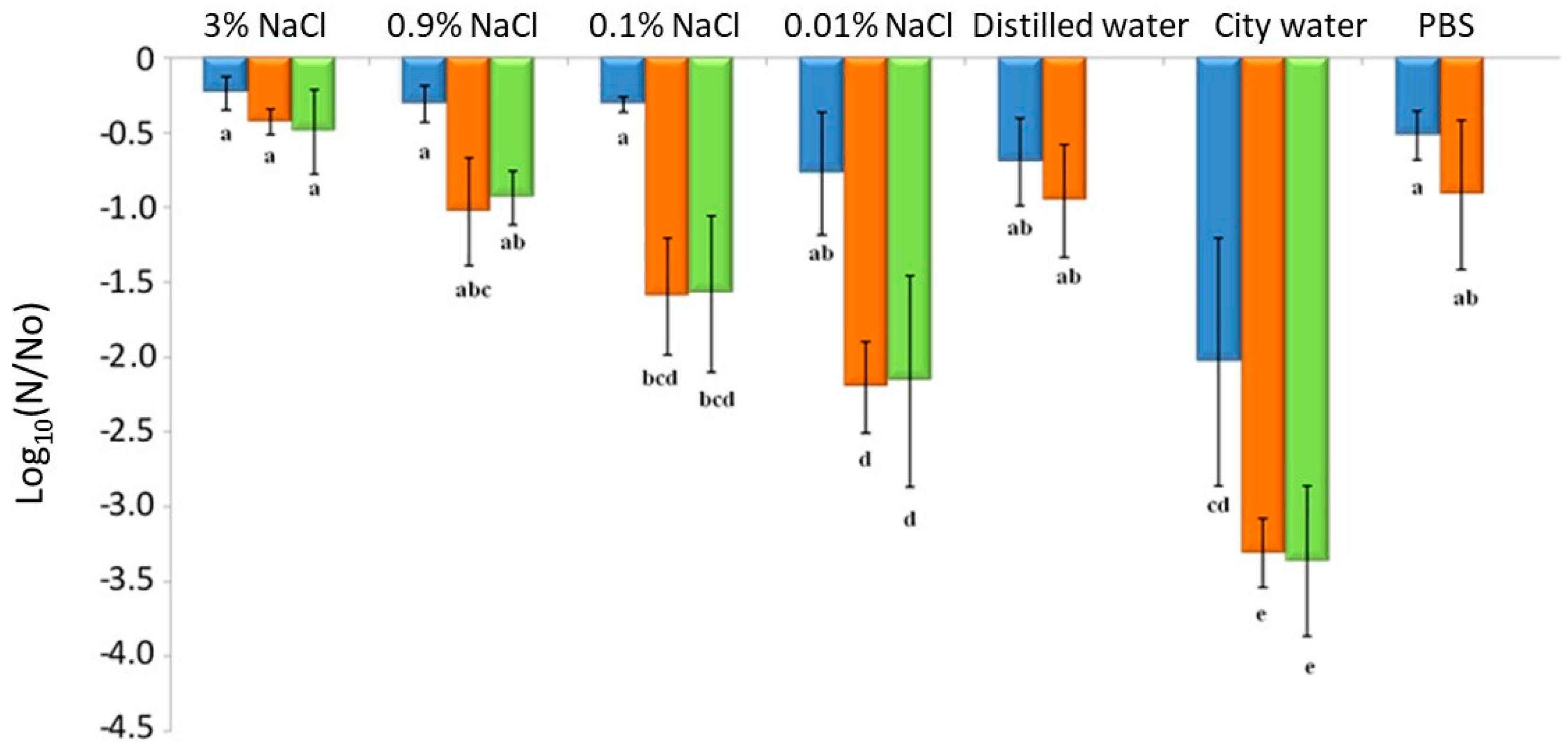
3.1. Impact of Drying and Rehydration in PBS on Plasma Membrane Permeability
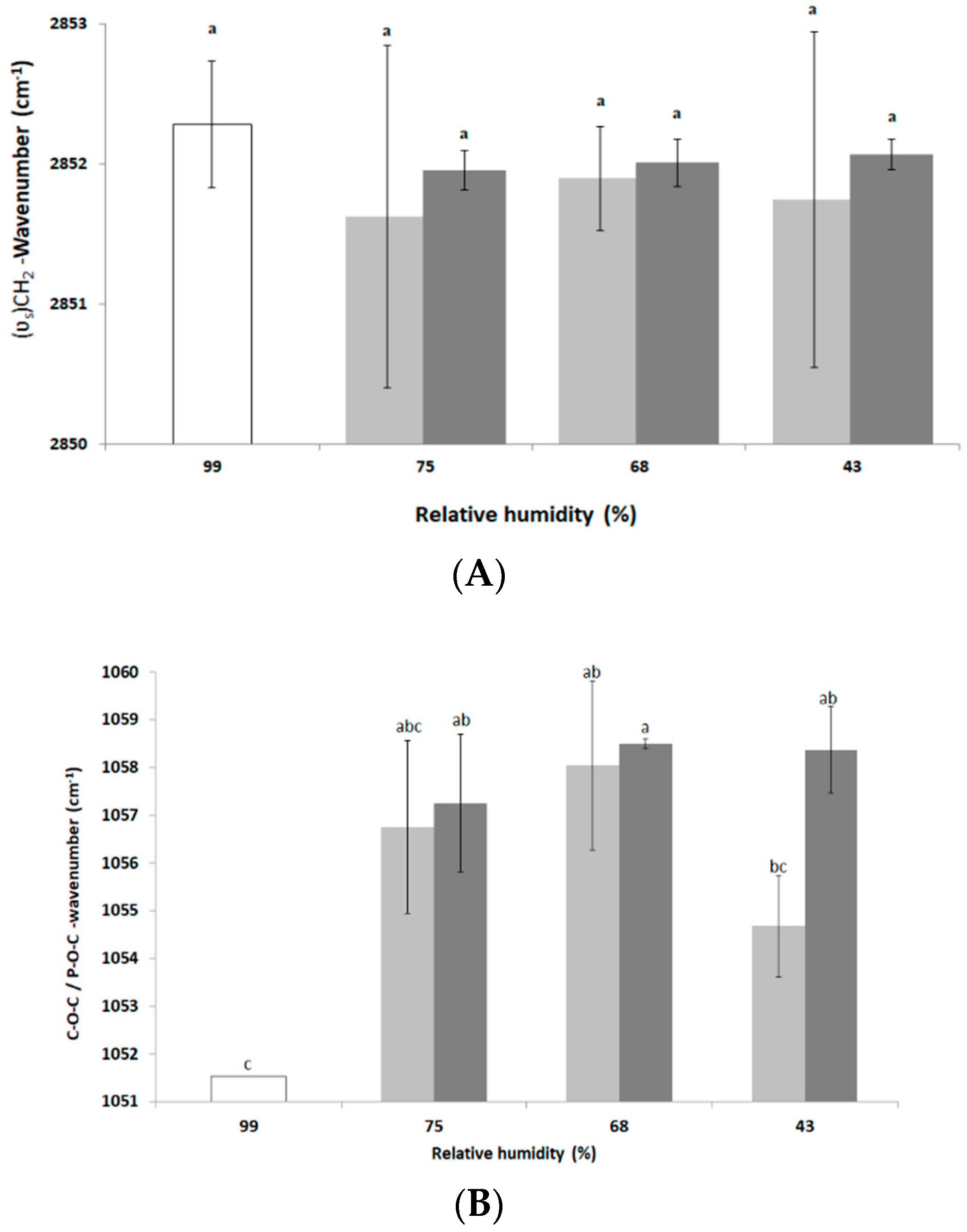
3.2. Impact of Drying and Rehydration in PBS on the Cell Envelope
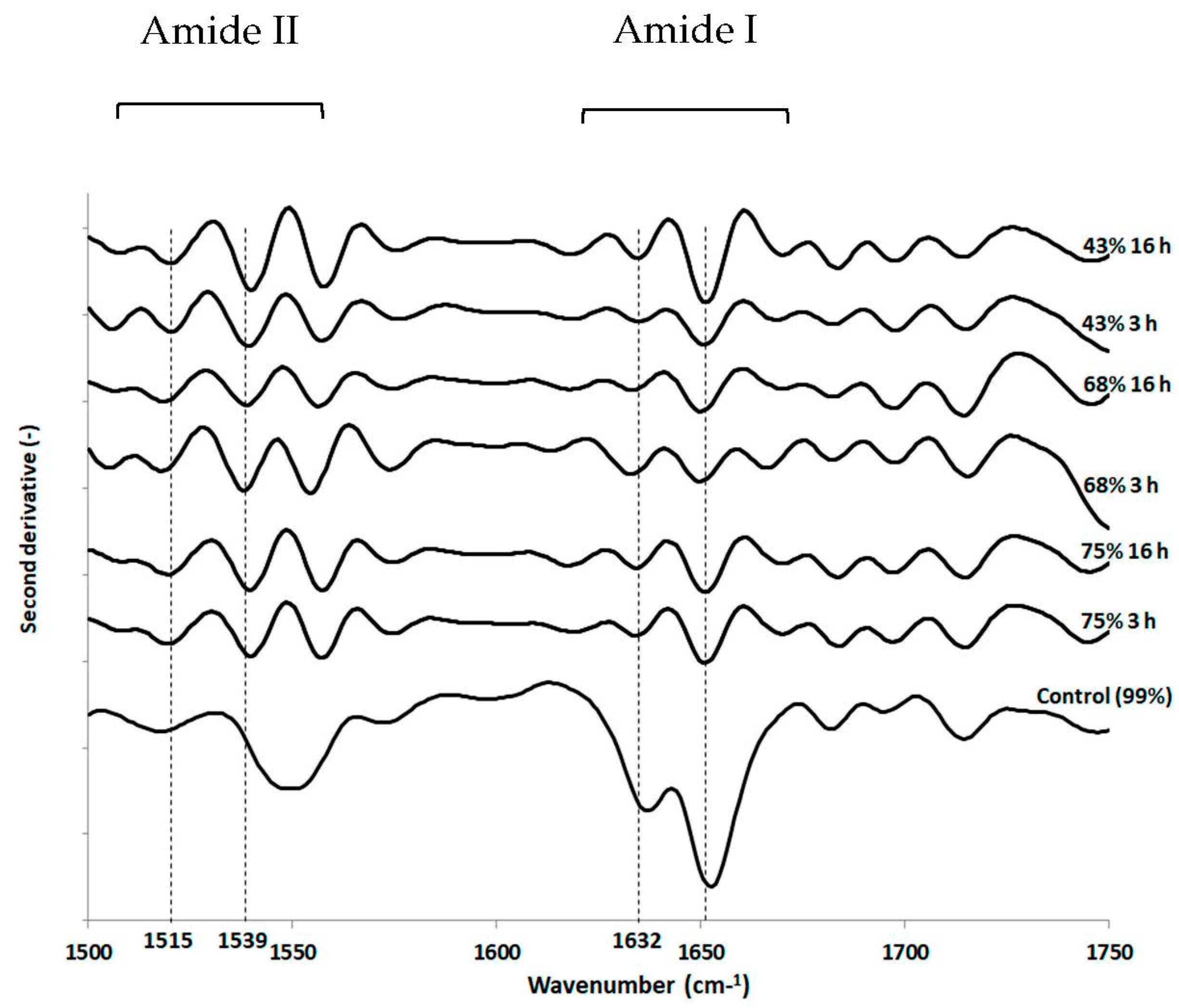
3.3. Impact of Drying and Rehydration in PBS on Protein Secondary Structure
3.4. Impacts of Drying and Rehydration on L. monocytogenes in the Food Industry Context
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Donovan, S. Listeriosis: A rare but deadly disease. Clin. Microbiol. Newsl. 2015, 37, 135–140. [Google Scholar] [CrossRef]
- Leclercq, A.; Kooh, P.; Augustin, J.-C.; Guillier, L.; Thébault, A.; Cadavez, V.; Gonzales-Barron, U.; Sanaa, M. Risk factors for sporadic listeriosis: A systematic review and meta-analysis. Microb. Risk Anal. 2021, 17, 100128. [Google Scholar] [CrossRef]
- De Noordhout, C.M.; Devleesschauwer, B.; Angulo, F.J.; Verbeke, G.; Haagsma, J.; Kirk, M.; Havelaar, A.; Speybroeck, N. The global burden of listeriosis: A systematic review and meta-analysis. Lancet Infect. Dis. 2014, 14, 1073–1082. [Google Scholar] [CrossRef]
- European Food Safety Authority; European Centre for Disease Prevention and Control (EFSA and ECDC). The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2017. EFSA J. 2018, 16, e05500. [Google Scholar] [CrossRef]
- Buchanan, R.L.; Gorris, L.G.M.; Hayman, M.M.; Jackson, T.C.; Whiting, R.C. A review of Listeria monocytogenes: An update on outbreaks, virulence, dose-response, ecology, and risk assessments. Food Control 2017, 75, 1–13. [Google Scholar] [CrossRef]
- Rodríguez-López, P.; Rodríguez-Herrera, J.J.; Cabo, M.L. Tracking bacteriome variation over time in Listeria monocytogenes-positive foci in food industry. Int. J. Food Microbiol. 2020, 315, 108439. [Google Scholar] [CrossRef]
- Cox, L.J.; Kleiss, T.; Cordier, J.L.; Cordellana, C.; Konkel, P.; Pedrazzini, C.; Beumer, R.; Siebenga, A. Listeria spp. in food processing, non-food and domestic environments. Food Microbiol. 1989, 6, 49–61. [Google Scholar] [CrossRef]
- Chasseignaux, E.; Gérault, P.; Toquin, M.-T.; Salvat, G.; Colin, P.; Ermel, G. Ecology of Listeria monocytogenes in the environment of raw poultry meat and raw pork meat processing plants. FEMS Microbiol. Lett. 2002, 210, 271–275. [Google Scholar] [CrossRef]
- Parisi, A.; Latorre, L.; Fraccalvieri, R.; Miccolupo, A.; Normanno, G.; Caruso, M.; Santagada, G. Occurrence of Listeria spp. in dairy plants in southern Italy and molecular subtyping of isolates using AFLP. Food Control 2013, 29, 91–97. [Google Scholar] [CrossRef]
- Kramarenko, T.; Roasto, M.; Meremäe, K.; Kuningas, M.; Põltsama, P.; Elias, T. Listeria monocytogenes prevalence and serotype diversity in various foods. Food Control 2013, 30, 24–29. [Google Scholar] [CrossRef]
- Silva, I.M.M.; Almeida, R.C.C.; Alves, M.A.O.; Almeida, P.F. Occurrence of Listeria spp. in critical control points and the environment of Minas Frescal cheese processing. Int. J. Food Microbiol. 2003, 81, 241–248. [Google Scholar] [CrossRef]
- Chambel, L.; Sol, M.; Fernandes, I.; Barbosa, M.; Zilhão, I.; Barata, B.; Jordan, S.; Perni, S.; Shama, G.; Adrião, A.; et al. Occurrence and persistence of Listeria spp. in the environment of ewe and cow’s milk cheese dairies in Portugal unveiled by an integrated analysis of identification, typing and spatial–temporal mapping along production cycle. Int. J. Food Microbiol. 2007, 116, 52–63. [Google Scholar] [CrossRef]
- Fox, E.; Hunt, K.; O’Brien, M.; Jordan, K. Listeria monocytogenes in Irish farmhouse cheese processing environments. Int. J. Food Microbiol. 2011, 145, S39–S45. [Google Scholar] [CrossRef] [PubMed]
- Montero, D.; Bodero, M.; Riveros, G.; Lapierre, L.; Gaggero, A.; Vidal, R.M.; Vidal, M. Molecular epidemiology and genetic diversity of Listeria monocytogenes isolates from a wide variety of ready-to-eat foods and their relationship to clinical strains from listeriosis outbreaks in Chile. Front. Microbiol. 2015, 6, 384. [Google Scholar] [CrossRef]
- Mazaheri, T.; Cervantes-Huamán, B.R.H.; Bermúdez-Capdevila, M.; Ripolles-Avila, C.; Rodríguez-Jerez, J.J. Listeria monocytogenes biofilms in the food industry: Is the current hygiene program sufficient to combat the persistence of the pathogen? Microorganisms 2021, 9, 181. [Google Scholar] [CrossRef]
- Silva, S.; Teixeira, P.; Oliveira, R.; Azeredo, J. Adhesion to and viability of Listeria monocytogenes on food contact surfaces. J. Food Prot. 2008, 71, 1379–1385. [Google Scholar] [CrossRef]
- Carpentier, B.; Cerf, O. Persistence of Listeria monocytogenes in food industry equipment and premises. Int. J. Food Microbiol. 2011, 145, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Jordan, K.; Hunt, K.; Lourenco, A.; Pennone, V. Listeria monocytogenes in the food processing environment. Curr. Clin. Microbiol. Rep. 2018, 5, 106–119. [Google Scholar] [CrossRef]
- Lundén, J.M.; Autio, T.J.; Sjöberg, A.M.; Korkeala, H.J. Persistent and nonpersistent Listeria monocytogenes contamination in meat and poultry processing plants. J. Food Prot. 2003, 66, 2062–2069. [Google Scholar] [CrossRef] [PubMed]
- Fagerlund, A.; Heir, E.; Møretrø, T.; Langsrud, S. Listeria monocytogenes biofilm removal using different commercial cleaning agents. Molecules 2020, 25, 792. [Google Scholar] [CrossRef] [PubMed]
- Zoz, F.; Iaconelli, C.; Lang, E.; Iddir, H.; Guyot, S.; Grandvalet, C.; Gervais, P.; Beney, L. Control of relative air humidity as a potential means to improve hygiene on surfaces: A preliminary approach with Listeria monocytogenes. PLoS ONE 2016, 11, e0148418. [Google Scholar] [CrossRef]
- Zoz, F.; Grandvalet, C.; Lang, E.; Iaconelli, C.; Gervais, P.; Firmesse, O.; Guyot, S.; Beney, L. Listeria monocytogenes ability to survive desiccation: Influence of serotype, origin, virulence, and genotype. Int. J. Food Microbiol. 2017, 248, 82–89. [Google Scholar] [CrossRef]
- Lang, E.; Zoz, F.; Iaconelli, C.; Guyot, S.; Alvarez-Martin, P.; Beney, L.; Perrier-Cornet, J.M.; Gervais, P. Recovery estimation of dried foodborne pathogens is directly related to rehydration kinetics. PLoS ONE 2016, 11, e0160844. [Google Scholar] [CrossRef]
- Lang, E.; Iaconelli, C.; Zoz, F.; Guyot, S.; Alvarez-Martin, P.; Beney, L.; Perrier-Cornet, J.-M.; Gervais, P. Drying parameters greatly affect the destruction of Cronobacter sakazakii and Salmonella Typhimurium in standard buffer and milk. Food Microbiol. 2017, 62, 82–91. [Google Scholar] [CrossRef]
- Laguerre, O.; Lecoq, L.; Zoz, F.; Guyot, S.; Beney, L.; Flick, D. Influence of the air humidity on the drying of a liquid droplet on a solid plate and on bacterial destruction. J. Food Eng. 2017, 212, 76–86. [Google Scholar] [CrossRef]
- Burke, T.P.; Loukitcheva, A.; Zemansky, J.; Wheeler, R.; Boneca, I.G.; Portnoy, D.A. Listeria monocytogenes is resistant to lysozyme through the regulation, not the acquisition, of cell wall-modifying enzymes. J. Bacteriol. 2014, 196, 3756–3767. [Google Scholar] [CrossRef]
- Hingston, P.A.; Piercey, M.J.; Truelstrup Hansen, L. Genes associated with desiccation and osmotic stress in Listeria monocytogenes as revealed by insertional mutagenesis. Appl. Environ. Microbiol. 2015, 81, 5350–5362. [Google Scholar] [CrossRef]
- Durack, J.; Burke, T.P.; Portnoy, D.A. A Prl mutation in SecY suppresses secretion and virulence defects of Listeria monocytogenes SecA2 mutants. J. Bacteriol. 2015, 197, 932–942. [Google Scholar] [CrossRef]
- Lang, E.; Guyot, S.; Peltier, C.; Alvarez-Martin, P.; Perrier-Cornet, J.-M.; Gervais, P. Cellular injuries in Cronobacter sakazakii CIP 103183T and Salmonella enterica exposed to drying and subsequent heat treatment in milk powder. Front. Microbiol. 2018, 9, 475. [Google Scholar] [CrossRef]
- Lemetais, G.; Dupont, S.; Beney, L.; Gervais, P. Air-drying kinetics affect yeast membrane organization and survival. Appl. Microbiol. Biotechnol. 2012, 96, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Simonin, H.; Beney, L.; Gervais, P. Sequence of occurring damages in yeast plasma membrane during dehydration and rehydration: Mechanisms of cell death. Biochim. Biophys. Acta Biomembr. 2007, 1768, 1600–1610. [Google Scholar] [CrossRef]
- Heins, A.L.; Weuster-Botz, D. Population heterogeneity in microbial bioprocesses: Origin, analysis, mechanisms, and future perspectives. Bioprocess. Biosyst. Eng. 2018, 41, 889–916. [Google Scholar] [CrossRef]
- Hewitt, C.J.; Nebe-Von-Caron, G. The application of multi-parameter flow cytometry to monitor individual microbial cell physiological state. In Physiological Stress Responses in Bioprocesses; Scheper, T., Enfors, S.-O., Eds.; Springer: Berlin, Germany, 2004; Volume 89, pp. 197–223. ISBN 978-3-540-39669-7. [Google Scholar]
- Beney, L.; Gervais, P. Influence of the fluidity of the membrane on the response of microorganisms to environmental stresses. Appl. Microbiol. Biotechnol. 2001, 57, 34–42. [Google Scholar] [CrossRef]
- Mille, Y.; Beney, L.; Gervais, P. Viability of Escherichia coli after combined osmotic and thermal treatment: A plasma membrane implication. Biochim. Biophys. Acta Biomembr. 2002, 1567, 41–48. [Google Scholar] [CrossRef]
- Simonin, H.; Beney, L.; Gervais, P. Cell death induced by mild physical perturbations could be related to transient plasma membrane modifications. J. Membr. Biol. 2007, 216, 37–47. [Google Scholar] [CrossRef]
- Leslie, S.B.; Israeli, E.; Lighthart, B.; Crowe, J.H.; Crowe, L.M. Trehalose and sucrose protect both membranes and proteins in intact bacteria during drying. Appl. Environ. Microbiol. 1995, 61, 3592–3597. [Google Scholar] [CrossRef] [PubMed]
- Laroche, C.; Simonin, H.; Beney, L.; Gervais, P. Phase transitions as a function of osmotic pressure in Saccharomyces cerevisiae whole cells, membrane extracts and phospholipid mixtures. Biochim. Biophys. Acta Biomembr. 2005, 1669, 8–16. [Google Scholar] [CrossRef][Green Version]
- Kardas, M.; Gozen, A.G.; Severcan, F. FTIR spectroscopy offers hints towards widespread molecular changes in cobalt-acclimated freshwater bacteria. Aquat. Toxicol. 2014, 155, 15–23. [Google Scholar] [CrossRef]
- Vollmer, W.; Blanot, D.; De Pedro, M.A. Peptidoglycan structure and architecture. FEMS Microbiol. Rev. 2008, 32, 149–167. [Google Scholar] [CrossRef]
- Liu, Q.; Yang, H. Application of atomic force microscopy in food microorganisms. Trends Food Sci. Technol. 2019, 87, 73–83. [Google Scholar] [CrossRef]
- Thwaites, J.J.; Mendelson, N.H. Mechanical behaviour of bacteria cell walls. Adv. Microb. Physiol. 1991, 32, 173–222. [Google Scholar] [CrossRef]
- Kurek, A.; Grudniak, A.M.; Szwed, M.; Klicka, A.; Samluk, L.; Wolska, K.I.; Janiszowska, W.; Popowska, M. Oleanolic acid and ursolic acid affect peptidoglycan metabolism in Listeria monocytogenes. Antonie Leeuwenhoek 2010, 97, 61–68. [Google Scholar] [CrossRef]
- Francius, G.; Polyakov, P.; Merlin, J.; Abe, Y.; Ghigo, J.-M.; Merlin, C.; Beloin, C.; Duval, J.F.L. Bacterial surface appendages strongly impact nanomechanical and electrokinetic properties of Escherichia coli cells subjected to osmotic stress. PLoS ONE 2011, 6, e20066. [Google Scholar] [CrossRef]
- Saar Dover, R.; Bitler, A.; Shimoni, E.; Trieu-Cuot, P.; Shai, Y. Multiparametric AFM reveals turgor-responsive net-like peptidoglycan architecture in live Streptococci. Nat. Commun. 2015, 6, 7193. [Google Scholar] [CrossRef]
- Marquis, R.E. Salt-induced contraction of bacterial cell walls. J. Bacteriol. 1968, 95, 775–781. [Google Scholar] [CrossRef] [PubMed]
- Dupont, S.; Beney, L.; Ritt, J.-F.; Lherminier, J.; Gervais, P. Lateral reorganization of plasma membrane is involved in the yeast resistance to severe dehydration. Biochim. Biophys. Acta Biomembr. 2010, 1798, 975–985. [Google Scholar] [CrossRef]
- Mille, Y.; Beney, L.; Gervais, P. Magnitude and kinetics of rehydration influence the viability of dehydrated E. coli K-12. Biotechnol. Bioeng. 2003, 83, 578–582. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.D.; Guyot, S.; Pénicaud, C.; Passot, S.; Sandt, C.; Fonseca, F.; Saurel, R.; Husson, F. Understanding the responses of Saccharomyces cerevisiae yeast strain during dehydration processes using synchrotron infrared spectroscopy. Analyst 2017, 142, 3620–3628. [Google Scholar] [CrossRef]
- Vogel, B.F.; Hansen, L.T.; Mordhorst, H.; Gram, L. The survival of Listeria monocytogenes during long term desiccation is facilitated by sodium chloride and organic material. Int. J. Food Microbiol. 2010, 140, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Dreux, N.; Albagnac, C.; Sleator, R.D.; Hill, C.; Carlin, F.; Morris, C.E.; Nguyen-the, C. Glycine betaine improves Listeria monocytogenes tolerance to desiccation on parsley leaves independent of the osmolyte transporters BetL, Gbu and OpuC. J. Appl. Microbiol. 2008, 104, 1221–1227. [Google Scholar] [CrossRef]
- Hingston, P.A.; Stea, E.C.; Knøchel, S.; Hansen, T. Role of initial contamination levels, biofilm maturity and presence of salt and fat on desiccation survival of Listeria monocytogenes on stainless steel surfaces. Food Microbiol. 2013, 36, 46–56. [Google Scholar] [CrossRef]
- Bergholz, T.M.; Bowen, B.; Wiedmann, M.; Boor, K.J. Listeria monocytogenes shows temperature-dependent and -independent responses to salt stress, including responses that induce cross-protection against other stresses. Appl. Environ. Microbiol. 2012, 78, 2602–2612. [Google Scholar] [CrossRef]
- Luu, P.; Chhetri, V.S.; Janes, M.E.; King, J.M.; Adhikari, A. Effectiveness of aqueous chlorine dioxide in minimizing food safety risk associated with Salmonella, E. coli O157:H7, and Listeria monocytogenes on sweet potatoes. Foods 2020, 9, 1259. [Google Scholar] [CrossRef]
- Vaid, R.; Linton, R.H.; Morgan, M.T. Comparison of inactivation of Listeria monocytogenes within a biofilm matrix using chlorine dioxide gas, aqueous chlorine dioxide and sodium hypochlorite treatments. Food Microbiol. 2010, 27, 979–984. [Google Scholar] [CrossRef] [PubMed]
- Berg, J.D.; Roberts, P.V.; Matin, A. Effect of chlorine dioxide on selected membrane functions of Escherichia coli. J. Appl. Bacteriol. 1986, 60, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Aieta, E.M.; Berg, J.D. A review of chlorine dioxide in drinking water treatment. J. Am. Water Work Assoc. 1986, 78, 62–72. [Google Scholar] [CrossRef]
- Ghanbari, H.A.; Wheeler, W.B.; Kirk, J.R. Reactions of aqueous chlorine and chlorine dioxide with lipids: Chlorine incorporation. J. Food Sci. 1982, 47, 482–485. [Google Scholar] [CrossRef]
- Sharma, V.K.; Sohn, M. Reactivity of chlorine dioxide with amino acids, peptides, and proteins. Environ. Chem. Lett. 2012, 10, 255–264. [Google Scholar] [CrossRef]
- Da Silva Pedrini, M.R.; Dupont, S.; de Anchieta Câmara, A.; Beney, L.; Gervais, P. Osmoporation: A simple way to internalize hydrophilic molecules into yeast. Appl. Microbiol. Biotechnol. 2014, 98, 1271–1280. [Google Scholar] [CrossRef] [PubMed]


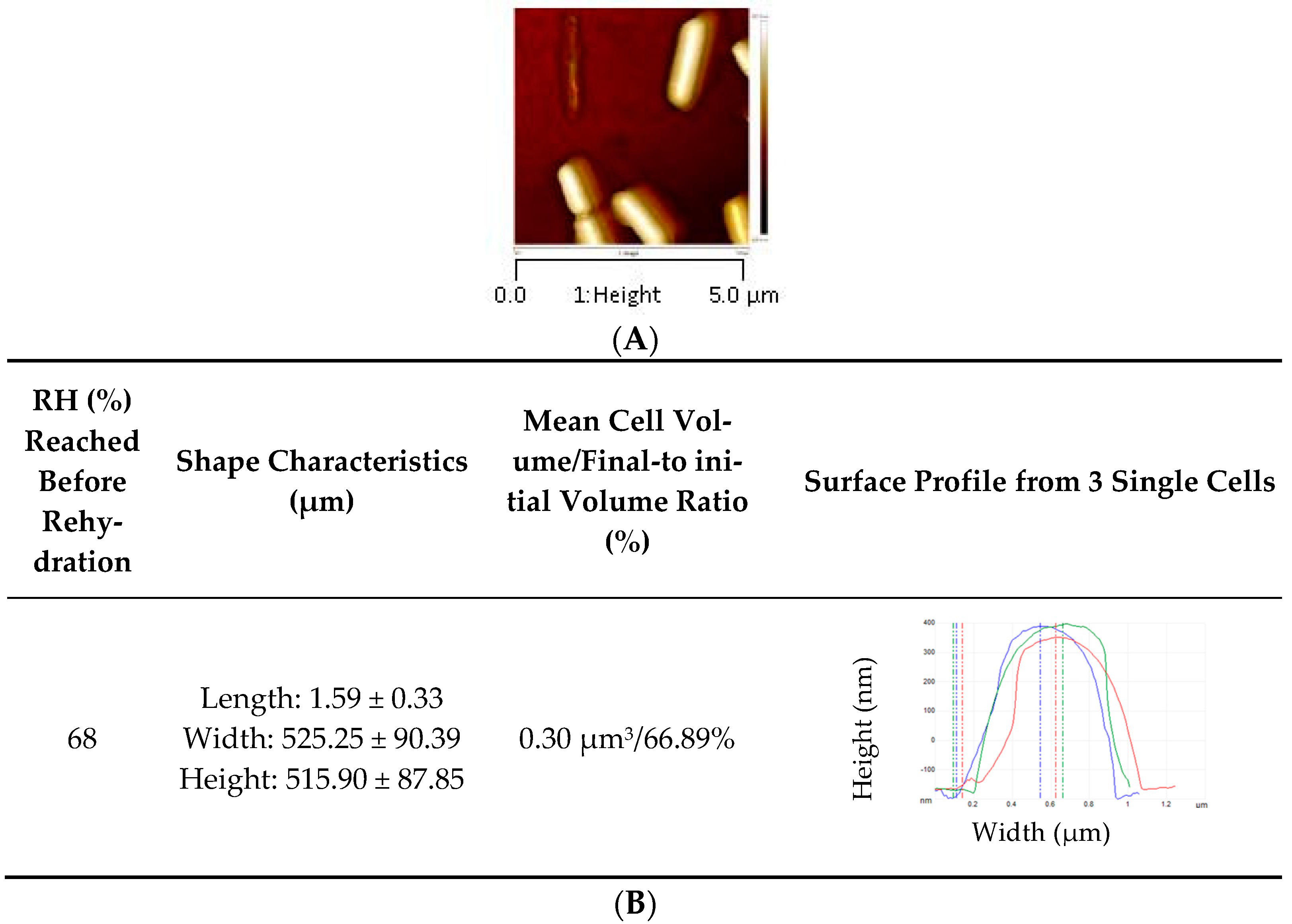
| RH (%) | Shape Characteristics (µm) | Mean Cell Volume/Final-to Initial Volume Ratio (%) | Mean Surface/Surface Excess (%) | Surface Profile from 3 Single Cells | Young Modulus (MPa) |
|---|---|---|---|---|---|
| 99 | Length: 1.78 ± 0.33 Width: 653.08 ± 121.27 Height: 555.56 ± 18.48 | 0.45 µm3/100% | 3.38 µm2/0.00% | 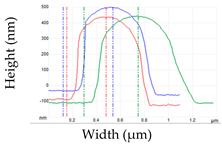 | 2.48 ± 0.57 c |
| 75 | Length: 1.38 ± 0.47 Width: 433.09 ± 76.24 Height: 375.30 ± 32.60 | 0.15 µm3/35.28% | 1.75 µm2/48.15% | 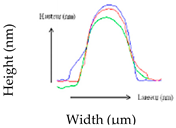 | 152.63 ± 11.54 a |
| 68 | Length: 1.42 ± 0.37 Width: 495.27 ± 67.13 Height: 323.38 ± 59.14 | 0.16 µm3/37.50% | 1.83 µm2/45.70% | 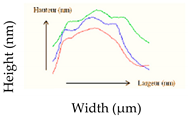 | 41. 58 ± 16.10 b |
| 43 | Length: 1.32 ± 0.12 Width: 409.50 ± 31.94 Height: 382.16 ± 7.98 | 0.14 µm3/32.31% | 1.64 µm2/51.38% |  | 26. 73 ± 14.86 bc |
| Food Item | Average NaCl Content (g/100 g) | Confidence Code * |
|---|---|---|
| Salty snacks, crackers, reduced fat | 3 | A |
| Braised ham on the bone | 2.97 | C |
| American-style sauce, prepacked | 2.94 | A |
| Lardoons, raw | 2.72 | D |
| Ketchup, prepacked | 2.59 | A |
| Salad dressing, reduced fat, prepacked | 2.57 | A |
| Atlantic herring, marinated, or rollmops | 2.56 | C |
| Caribbean-style fish fritters, fish acras | 1.24 | A |
| Tabbouleh, prepacked | 1 | A |
| Seafood, cooked, frozen | 0.99 | C |
| Marine fish, white, without skin, cooked | 0.98 | D |
| Mackerel, smoked | 0.96 | C |
| Vegetable dish for baby, w meat/fish and starch, from 6–8 months | 0.2 | A |
| European whitefish, raw | 0.13 | B |
| Carrot juice, pure juice | 0.1 | A |
| Pork, roast, raw | 0.1 | A |
| Yogurt, ewe’s milk, wholemilk, plain | 0.1 | A |
| Jam, strawberry or cherry | 0.018 | A |
| Fruit juice, from concentrate (average) | 0.012 | B |
| Fruit juice, mixed, juice and fruit puree | 0.01 | A |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zoz, F.; Guyot, S.; Grandvalet, C.; Ragon, M.; Lesniewska, E.; Dupont, S.; Firmesse, O.; Carpentier, B.; Beney, L. Management of Listeria monocytogenes on Surfaces via Relative Air Humidity: Key Role of Cell Envelope. Foods 2021, 10, 2002. https://doi.org/10.3390/foods10092002
Zoz F, Guyot S, Grandvalet C, Ragon M, Lesniewska E, Dupont S, Firmesse O, Carpentier B, Beney L. Management of Listeria monocytogenes on Surfaces via Relative Air Humidity: Key Role of Cell Envelope. Foods. 2021; 10(9):2002. https://doi.org/10.3390/foods10092002
Chicago/Turabian StyleZoz, Fiona, Stéphane Guyot, Cosette Grandvalet, Mélanie Ragon, Eric Lesniewska, Sébastien Dupont, Olivier Firmesse, Brigitte Carpentier, and Laurent Beney. 2021. "Management of Listeria monocytogenes on Surfaces via Relative Air Humidity: Key Role of Cell Envelope" Foods 10, no. 9: 2002. https://doi.org/10.3390/foods10092002
APA StyleZoz, F., Guyot, S., Grandvalet, C., Ragon, M., Lesniewska, E., Dupont, S., Firmesse, O., Carpentier, B., & Beney, L. (2021). Management of Listeria monocytogenes on Surfaces via Relative Air Humidity: Key Role of Cell Envelope. Foods, 10(9), 2002. https://doi.org/10.3390/foods10092002