Influence of Sample Matrix on Determination of Histamine in Fish by Surface Enhanced Raman Spectroscopy Coupled with Chemometric Modelling
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Fish Samples
2.3. Determination of Histamine by Reference HPLC Method
2.4. Preparation of Citrate Reduced Silver Nanoparticles (AGC)
2.5. SERS Measurements
2.6. Instrumentation
2.7. Spectral Analysis and Chemometric Modelling
2.7.1. Partial Least Squares Regression
2.7.2. Principal Component Analysis
2.7.3. Artificial Neural Networks
3. Results and Discussion
3.1. Histamine Content and Spectral Features of Fish Samples
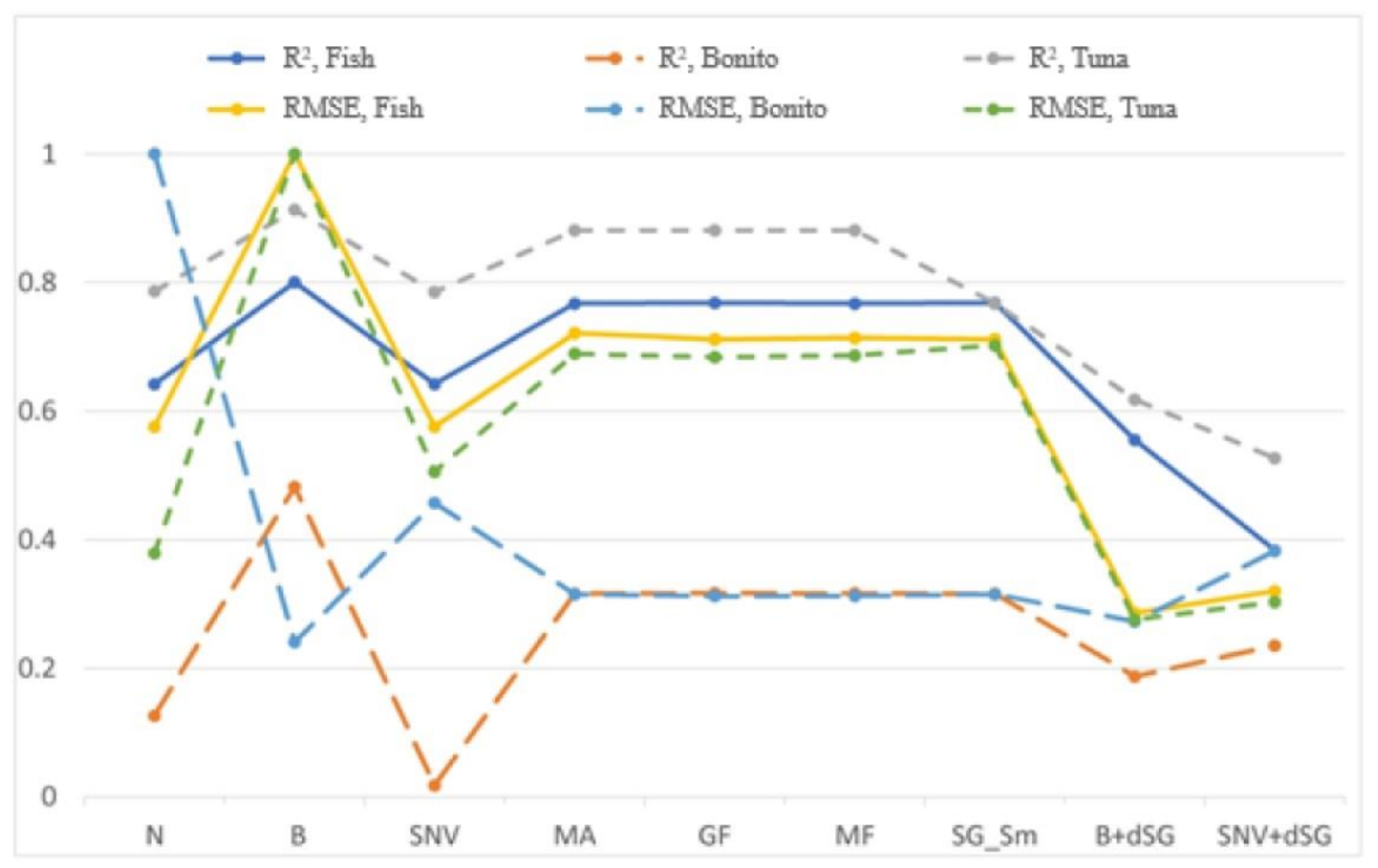
3.2. PLSR Models
3.3. Artificial Neural Network Models
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Vidaček, S.; Janči, T. Chapter 5—Safety of fish products. In Regulating Safety of Traditional and Ethnic Foods; Prakash, V., Martín-Belloso, O., Keener, L., Astley, S., Braun, S., McMahon, H., Lelieveld, H., Eds.; Academic Press: San Diego, CA, USA, 2016; pp. 79–97. [Google Scholar]
- Hungerford, J.M. Scombroid poisoning: A review. Toxicon 2010, 56, 231–243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feng, C.; Teuber, S.; Gershwin, M.E. Histamine (scombroid) fish poisoning: A comprehensive review. Clin. Rev. Allerg. Immunol. 2016, 50, 64–69. [Google Scholar] [CrossRef] [PubMed]
- FDA. Scombrotoxin (histamine) formation. In Fish and Fishery Products Hazards and Controls Guidance, 4th ed.; Department of Health and Human Services: Washington, DC, USA; Public Health Service: Washington, DC, USA; Food and Drug Administration: Silver Spring, MD, USA; Center for Food Safety and Applied Nutrition: College Park, MD, USA; Office of Food Safety: Washington, DC, USA, 2011; pp. 113–152. [Google Scholar]
- Commission regulation (EC) No 2073/2005 of 15 November 2005 on microbiological criteria for foodstuffs. OJEU 2005, 338, 1. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=CELEX:02005R2073-20200308&from=EN#tocld2 (accessed on 21 February 2021).
- Lehane, L.; Olley, J. Histamine fish poisoning revisited. Int. J. Food Microbiol. 2000, 58, 1–37. [Google Scholar] [CrossRef]
- Hussain, M.A.; Saputra, T.; Szabo, E.A.; Nelan, B. An overview of seafood supply, food safety and regulation in new South Wales, Australia. Foods 2017, 6, 52. [Google Scholar] [CrossRef] [Green Version]
- Barrett, K.A.; Nakao, J.H.; Taylor, E.V.; Eggers, C.; Gould, L.H. Fish-associated foodborne disease outbreaks: United States, 1998–2015. Foodborne Pathog. Dis. 2017, 14, 537–543. [Google Scholar] [CrossRef]
- EFSA. The European Union one health 2018 zoonoses report. EFSA J. 2019, 17, e05926. [Google Scholar] [CrossRef] [Green Version]
- Altieri, I.; Semeraro, A.; Scalise, F.; Calderari, I.; Stacchini, P. European official control of food: Determination of histamine in fish products by a HPLC–UV-DAD method. Food Chem. 2016, 211, 694–699. [Google Scholar] [CrossRef]
- Li, Y.-F.; Lin, Z.-Z.; Hong, C.-Y.; Huang, Z.-Y. Histamine detection in fish samples based on indirect competitive ELISA method using iron-cobalt co-doped carbon dots labeled histamine antibody. Food Chem. 2021, 345, 128812. [Google Scholar] [CrossRef]
- Yu, H.; Zhuang, D.; Hu, X.; Zhang, S.; He, Z.; Zeng, M.; Fang, X.; Chen, J.; Chen, X. Rapid determination of histamine in fish by thin-layer chromatography-image analysis method using diazotized visualization reagent prepared with p-nitroaniline. Anal. Methods 2018, 10, 3386–3392. [Google Scholar] [CrossRef]
- Nadeem, M.; Naveed, T.; Rehman, F.; Xu, Z. Determination of histamine in fish without derivatization by indirect reverse phase-HPLC method. Microchem. J. 2019, 144, 209–214. [Google Scholar] [CrossRef]
- Kounnoun, A.; EL Maadoudi, M.; Cacciola, F.; Mondello, L.; Bougtaib, H.; Alahlah, N.; Amajoud, N.; El Baaboua, A.; Louajri, A. Development and validation of a high-performance liquid chromatography method for the determination of histamine in fish samples using fluorescence detection with pre-column derivatization. Chromatographia 2020, 83, 893–901. [Google Scholar] [CrossRef]
- Huang, J.; Gan, N.; Lv, F.; Cao, Y.; Ou, C.; Tang, H. Environmentally friendly solid-phase microextraction coupled with gas chromatography and mass spectrometry for the determination of biogenic amines in fish samples. J. Sep. Sci. 2016, 39, 4384–4390. [Google Scholar] [CrossRef]
- Koçoğlu, İ.O.; Erden, P.E.; Kılıç, E. Disposable biogenic amine biosensors for histamine determination in fish. Anal. Methods 2020, 12, 3802–3812. [Google Scholar] [CrossRef]
- Veseli, A.; Vasjari, M.; Arbneshi, T.; Hajrizi, A.; Švorc, Ľ.; Samphao, A.; Kalcher, K. Electrochemical determination of histamine in fish sauce using heterogeneous carbon electrodes modified with rhenium(IV) oxide. Sens. Actuat. B Chem. 2016, 228, 774–781. [Google Scholar] [CrossRef]
- Serrano, V.M.; Cardoso, A.R.; Diniz, M.; Sales, M.G.F. In-situ production of histamine-imprinted polymeric materials for electrochemical monitoring of fish. Sens. Actuat. B Chem. 2020, 311, 127902. [Google Scholar] [CrossRef]
- Torre, R.; Costa-Rama, E.; Lopes, P.; Nouws, H.P.A.; Delerue-Matos, C. Amperometric enzyme sensor for the rapid determination of histamine. Anal. Methods 2019, 11, 1264–1269. [Google Scholar] [CrossRef]
- Lee, M.-Y.; Wu, C.-C.; Sari, M.I.; Hsieh, Y.-H. A disposable non-enzymatic histamine sensor based on the nafion-coated copper phosphate electrodes for estimation of fish freshness. Electrochim. Acta 2018, 283, 772–779. [Google Scholar] [CrossRef]
- Tan, A.; Zhao, Y.; Sivashanmugan, K.; Squire, K.; Wang, A.X. Quantitative TLC-SERS detection of histamine in seafood with support vector machine analysis. Food Control. 2019, 103, 111–118. [Google Scholar] [CrossRef] [Green Version]
- Janči, T.; Valinger, D.; Gajdoš Kljusurić, J.; Mikac, L.; Vidaček, S.; Ivanda, M. Determination of histamine in fish by surface enhanced Raman spectroscopy using silver colloid SERS substrates. Food Chem. 2017, 224, 48–54. [Google Scholar] [CrossRef]
- Pochanagone, S.; Rittiron, R. Preliminary study on the determination of ppm-level concentration of histamine in tuna fish using a dry extract system for infrared coupled with near-infrared spectroscopy. ACS Omega 2019, 4, 19164–19171. [Google Scholar] [CrossRef] [Green Version]
- Ghidini, S.; Chiesa, L.M.; Panseri, S.; Varrà, M.O.; Ianieri, A.; Pessina, D.; Zanardi, E. Histamine control in raw and processed tuna: A rapid tool based on NIR spectroscopy. Foods 2021, 10, 885. [Google Scholar] [CrossRef]
- Pauline, O.; Chang, H.-T.; Tsai, I.L.; Lin, C.-H.; Chen, S.; Chuang, Y.-K. Intelligent assessment of the histamine level in mackerel (Scomber australasicus) using near-infrared spectroscopy coupled with a hybrid variable selection strategy. LWT 2021, 145, 111524. [Google Scholar] [CrossRef]
- Gao, F.; Grant, E.; Lu, X. Determination of histamine in canned tuna by molecularly imprinted polymers-surface enhanced Raman spectroscopy. Anal. Chim. Acta 2015, 901, 68–75. [Google Scholar] [CrossRef]
- Zhou, T.; Fan, M.; You, R.; Lu, Y.; Huang, L.; Xu, Y.; Feng, S.; Wu, Y.; Shen, H.; Zhu, L. Fabrication of Fe3O4/Au@ATP@Ag Nanorod sandwich structure for sensitive SERS quantitative detection of histamine. Anal. Chim. Acta 2020, 1104, 199–206. [Google Scholar] [CrossRef]
- Lin, Z.; He, L. Recent advance in SERS techniques for food safety and quality analysis: A brief review. Curr. Opin. Food Sci. 2019, 28, 82–87. [Google Scholar] [CrossRef]
- McNay, G.; Eustace, D.; Smith, W.E.; Faulds, K.; Graham, D. Surface-enhanced Raman scattering (SERS) and surface-enhanced resonance Raman scattering (SERRS): A review of applications. Appl. Spectrosc. 2011, 65, 825–837. [Google Scholar] [CrossRef]
- Aroca, R. Surface-enhanced Raman scattering. In Surface-Enhanced Vibrational Spectroscopy; Aroca, R., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2006; pp. 73–106. [Google Scholar]
- Le Ru, E.C.; Blackie, E.; Meyer, M.; Etchegoin, P.G. Surface enhanced Raman scattering enhancement factors: A comprehensive study. J. Phys. Chem. C 2007, 111, 13794–13803. [Google Scholar] [CrossRef]
- Bizzarri, A.R.; Cannistraro, S. Surface-enhanced resonance Raman spectroscopy signals from single myoglobin molecules. Appl. Spectrosc. 2002, 56, 1531–1537. [Google Scholar] [CrossRef]
- Habuchi, S.; Cotlet, M.; Gronheid, R.; Dirix, G.; Michiels, J.; Vanderleyden, J.; De Schryver, F.C.; Hofkens, J. Single-molecule surface enhanced resonance Raman spectroscopy of the enhanced green fluorescent protein. J. Am. Chem. Soc. 2003, 125, 8446–8447. [Google Scholar] [CrossRef]
- Mao, P.; Liu, C.; Favraud, G.; Chen, Q.; Han, M.; Fratalocchi, A.; Zhang, S. Broadband single molecule SERS detection designed by warped optical spaces. Nat. Commun. 2018, 9, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Huynh, K.-H.; Pham, X.-H.; Hahm, E.; An, J.; Kim, H.-M.; Jo, A.; Seong, B.; Kim, Y.-H.; Son, B.S.; Kim, J.; et al. Facile histamine detection by surface-enhanced Raman scattering using SiO2@Au@Ag alloy nanoparticles. Int. J. Mol. Sci. 2020, 21, 4048. [Google Scholar] [CrossRef] [PubMed]
- Janči, T.; Mikac, L.; Ivanda, M.; Marušić Radovčić, N.; Medić, H.; Vidaček, S. Optimization of parameters for histamine detection in fish muscle extracts by surface-enhanced Raman spectroscopy using silver colloid SERS substrates. J. Raman Spectrosc. 2017, 48, 64–72. [Google Scholar] [CrossRef]
- Malle, P.; Vallé, M.; Bouquelet, S. Assay of biogenic amines involved in fish decomposition. J. AOAC Int. 1996, 79, 43–49. [Google Scholar] [CrossRef] [Green Version]
- Lee, P.C.; Meisel, D. Adsorption and surface-enhanced Raman of dyes on silver and gold sols. J. Phys. Chem. 1982, 86, 3391–3395. [Google Scholar] [CrossRef]
- Collado, J.A.; Ramírez, F.J. Infrared and Raman spectra of histamine-Nh4 and histamine-Nd4 monohydrochlorides. J. Raman Spectrosc. 1999, 30, 391–397. [Google Scholar] [CrossRef]
- Valinger, D.; Jurina, T.; Šain, A.; Matešić, N.; Panić, M.; Benković, M.; Gajdoš Kljusurić, J.; Jurinjak Tušek, A. Development of ANN models based on combined UV-vis-NIR spectra for rapid quantification of physical and chemical properties of industrial hemp extracts. Phytochem. Anal. 2021, 32, 326–338. [Google Scholar] [CrossRef]
- Mukherjee, V.; Yadav, T. Conformational study of neutral histamine monomer and their vibrational spectra. Spectrochim. Acta A 2016, 165, 167–175. [Google Scholar] [CrossRef]
- Valinger, D.; Kušen, M.; Tušek, A.; Panić, M.; Jurina, T.; Benković, M.; Redovniković, I.R.; Kljusurić, J.G. Development of near infrared spectroscopy models for quantitative prediction of the content of bioactive compounds in olive leaves. Chem. Biochem. Eng. Q 2019, 32, 535–543. [Google Scholar] [CrossRef]
- Jurinjak Tušek, A.; Benković, M.; Malešić, E.; Marić, L.; Jurina, T.; Gajdoš Kljusurić, J.; Valinger, D. Rapid quantification of dissolved solids and bioactives in dried root vegetable extracts using near infrared spectroscopy. Spectrochim. Acta A 2021, 120074. [Google Scholar] [CrossRef]
- Stoyanova, R.; Brown, T.R. NMR spectral quantitation by principal component analysis. NMR Biomed. 2001, 14, 271–277. [Google Scholar] [CrossRef]
- Stoyanova, R.; Arias-Mendoza, F.; Brown, T.R. Making multi-dimensional spectroscopy practical: Application of principal-component analysis to CSI data. In High-Power Gradient MR-Imaging; Oudkerk, M., Edelman, R.R., Eds.; Blackwell: Berlin, Germany, 1997. [Google Scholar]
- Ajiboye, A.; Abdullah-Arshah, R.; Qin, H.; Isah-Kebbe, H. Evaluating the effect of dataset size on predictive model using supervised learning technique. IJSECS 2015, 1, 75–84. [Google Scholar] [CrossRef]
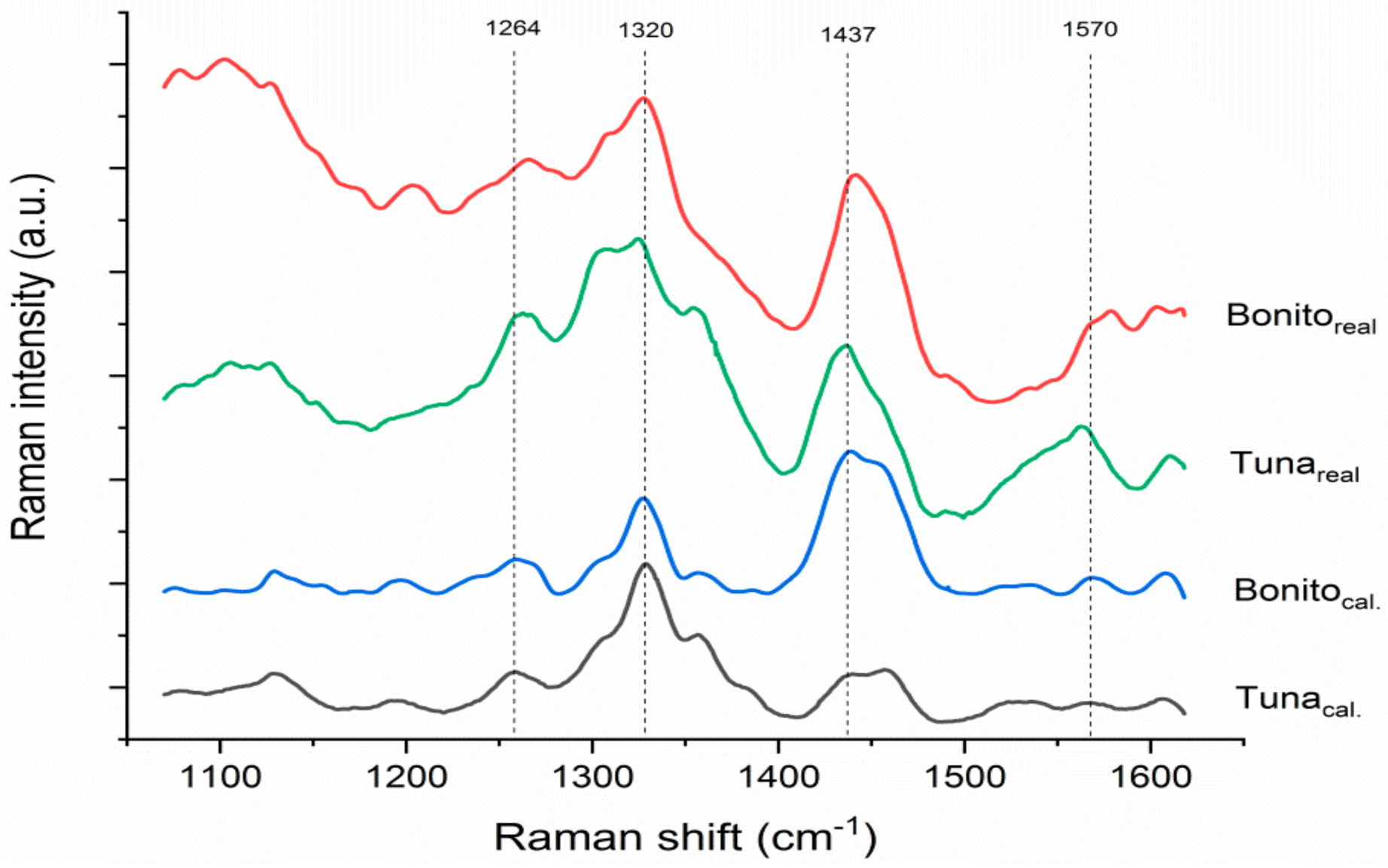
| Sample | Histamine Content (mg/kg) | Sample | Histamine Content (mg/kg) |
|---|---|---|---|
| T1 | 8.6 ± 3.3 | T7 | 248.1 ± 11.7 |
| T2 | 9.6 ± 1.4 | B1 | 33.6 ± 0.6 |
| T3 | 23.0 ± 6.3 | B2 | 41.1 ± 1.0 |
| T4 | 33.3 ± 0.8 | B3 | 76.1 ± 0.5 |
| T5 | 81.8 ± 6.2 | B4 | 136.2 ± 0.4 |
| T6 | 173.7 ± 10.1 | B5 | 184.9 ± 10.2 |
| Calibration | ||
|---|---|---|
| Pre-Processing Method | Rc2 | RMSEC |
| Normalize, N | 0.966 | 16.072 |
| Baseline, B | 0.978 | 13.169 |
| Standard Normal Variate, SNV | 0.984 | 11.127 |
| Moving Average, MA | 0.966 | 16.125 |
| Gaussian filter, GF | 0.971 | 14.969 |
| Median filter, MF | 0.967 | 15.904 |
| Savitzky-Golay Smoothing, SG_Sm | 0.966 | 16.129 |
| Baseline + der. S-G, B+dSG | 0.959 | 17.805 |
| SNV + der. S-G, SNV+dSG | 0.972 | 14.747 |
| Pre-processing method | 0.966 | 16.072 |
| Model | Calibration | Fish | Bonito | Tuna | ||||
|---|---|---|---|---|---|---|---|---|
| Rc2 | RMSEC | Rv2 | RMSEV | Rv2 | RMSEV | Rv2 | RMSEV | |
| M1 | 0.978 | 13.169 | 0.800 | 237.741 | 0.482 | 52.578 | 0.913 | 242.606 |
| M2 | 0.945 | 20.701 | 0.786 | 140.529 | 0.722 | 43.262 | 0.912 | 131.231 |
| M3 | 0.764 | 42.842 | 0.691 | 67.589 | 0.3775 | 56.698 | 0.879 | 68.901 |
| M4 | 0.504 | 73.525 | 0.409 | 68.045 | 0.4022 | 50.376 | 0.422 | 71.555 |
| Fish | Model | Network Architecture | Training Perf. | Test Perf. | Validation Perf. | Training Error | Test Error | Validation Error | Hidden Activation | Output Activation |
|---|---|---|---|---|---|---|---|---|---|---|
| Fish | M1 | 10-12-1 | 0.9975 | 0.9828 | 0.9754 | 6.3774 | 15.3578 | 20.4546 | Tanh | Exponential |
| M2 | 10-6-1 | 0.9960 | 0.9911 | 0.9906 | 7.8974 | 11.2420 | 13.2226 | Tanh | Identity | |
| M3 | 10-10-1 | 0.9897 | 0.9614 | 0.9585 | 12.6903 | 23.3557 | 28.1661 | Exponential | Logistic | |
| M4 | 10-9-1 | 0.8045 | 0.7117 | 0.6644 | 52.6201 | 58.9764 | 71.8243 | Logistic | Logistic | |
| Bonito | M1 | 10-12-1 | 0.9964 | 0.9955 | 0.9910 | 7.6300 | 9.0898 | 13.0946 | Exponential | Logistic |
| M2 | 10-5-1 | 0.9981 | 0.9954 | 0.9916 | 5.6370 | 9.2371 | 10.1250 | Tanh | Exponential | |
| M3 | 10-7-1 | 0.9964 | 0.9939 | 0.9870 | 7.8028 | 10.8345 | 12.6816 | Logistic | Exponential | |
| M4 | 10-8-1 | 0.9047 | 0.8928 | 0.8507 | 39.4613 | 44.2791 | 49.3624 | Tanh | Exponential | |
| Tuna | M1 | 10-13-1 | 0.9981 | 0.9953 | 0.9945 | 5.3512 | 9.2580 | 9.3494 | Tanh | Exponential |
| M2 | 10-11-1 | 0.9979 | 0.9908 | 0.9902 | 6.0746 | 11.6041 | 13.0318 | Tanh | Logistic | |
| M3 | 10-6-1 | 0.9959 | 0.9946 | 0.9919 | 7.8603 | 9.7427 | 11.2788 | Logistic | Exponential | |
| M4 | 10-13-1 | 0.9295 | 0.8648 | 0.8269 | 31.8624 | 47.3664 | 49.4626 | Logistic | Exponential |
| Fish | Model | Network Architecture | Training Perf. | Test Perf. | Validation Perf. | Training Error | Test Error | Validation Error | Hidden Activation | Output Activation |
|---|---|---|---|---|---|---|---|---|---|---|
| Bonito | M1 | 10-10-1 | 0.9965 | 0.9906 | 0.9874 | 7.5415 | 11.8646 | 12.7808 | Logistic | Exponential |
| M2 | 10-7-1 | 0.9964 | 0.9926 | 0.9883 | 7.6440 | 10.7651 | 13.4342 | Tanh | Exponential | |
| M3 | 10-6-1 | 0.9931 | 0.9839 | 0.9797 | 10.6157 | 15.4946 | 16.6087 | Exponential | Exponential | |
| M4 | 10-5-1 | 0.9350 | 0.8755 | 0.8564 | 32.2163 | 42.3773 | 49.1198 | Logistic | Logistic | |
| Tuna | M1 | 10-10-1 | 0.9977 | 0.9933 | 0.9920 | 6.3520 | 8.8020 | 11.6935 | Logistic | Exponential |
| M2 | 10-12-1 | 0.9981 | 0.9931 | 0.9930 | 5.8671 | 9.0178 | 10.6760 | Tanh | Exponential | |
| M3 | 10-10-1 | 0.9980 | 0.9933 | 0.9925 | 6.0089 | 9.0942 | 11.3809 | Tanh | Exponential | |
| M4 | 10-11-1 | 0.9399 | 0.8698 | 0.8630 | 32.2323 | 39.1596 | 48.9777 | Exponential | Tanh |
| Fish | Model | Network Architecture | Training Perf. | Test Perf. | Validation Perf. | Training Error | Test Error | Validation Error | Hidden Activation | Output Activation |
|---|---|---|---|---|---|---|---|---|---|---|
| Fish | M1 | 10-7-1 | 0.9988 | 0.9937 | 0.9936 | 3.9688 | 8.8349 | 9.7735 | Tanh | Identity |
| M2 | 10-8-1 | 0.9993 | 0.9973 | 0.9935 | 2.9749 | 5.8620 | 9.6090 | Exponential | Tanh | |
| M3 | 10-10-1 | 0.9978 | 0.9939 | 0.9935 | 5.4714 | 9.0641 | 10.4907 | Logistic | Tanh | |
| M4 | 10-5-1 | 0.9831 | 0.9653 | 0.9602 | 15.1824 | 21.7015 | 23.3168 | Logistic | Exponential | |
| Bonito | M1 | 10-10-1 | 0.9990 | 0.9976 | 0.9955 | 2.4070 | 5.3407 | 5.8885 | Logistic | Exponential |
| M2 | 10-10-1 | 0.9992 | 0.9972 | 0.9958 | 2.4093 | 4.8894 | 8.1239 | Exponential | Logistic | |
| M3 | 10-4-1 | 0.9996 | 0.9989 | 0.9713 | 1.6074 | 5.1973 | 16.2568 | Logistic | Identity | |
| M4 | 10-9-1 | 0.9982 | 0.9698 | 0.9603 | 3.8024 | 17.4051 | 21.3636 | Tanh | Logistic | |
| Tuna | M1 | 10-6-1 | 0.9995 | 0.9990 | 0.9988 | 2.8103 | 3.6519 | 4.3021 | Exponential | Logistic |
| M2 | 10-12-1 | 0.9998 | 0.9990 | 0.9989 | 1.6327 | 3.6420 | 4.6434 | Tanh | Exponential | |
| M3 | 10-11-1 | 0.9999 | 0.9996 | 0.9987 | 1.2763 | 2.3012 | 4.5641 | Tanh | Identity | |
| M4 | 10-4-1 | 0.9959 | 0.9924 | 0.9912 | 7.7512 | 10.0073 | 12.8127 | Tanh | Tanh |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Filipec, S.V.; Valinger, D.; Mikac, L.; Ivanda, M.; Kljusurić, J.G.; Janči, T. Influence of Sample Matrix on Determination of Histamine in Fish by Surface Enhanced Raman Spectroscopy Coupled with Chemometric Modelling. Foods 2021, 10, 1767. https://doi.org/10.3390/foods10081767
Filipec SV, Valinger D, Mikac L, Ivanda M, Kljusurić JG, Janči T. Influence of Sample Matrix on Determination of Histamine in Fish by Surface Enhanced Raman Spectroscopy Coupled with Chemometric Modelling. Foods. 2021; 10(8):1767. https://doi.org/10.3390/foods10081767
Chicago/Turabian StyleFilipec, Sanja Vidaček, Davor Valinger, Lara Mikac, Mile Ivanda, Jasenka Gajdoš Kljusurić, and Tibor Janči. 2021. "Influence of Sample Matrix on Determination of Histamine in Fish by Surface Enhanced Raman Spectroscopy Coupled with Chemometric Modelling" Foods 10, no. 8: 1767. https://doi.org/10.3390/foods10081767
APA StyleFilipec, S. V., Valinger, D., Mikac, L., Ivanda, M., Kljusurić, J. G., & Janči, T. (2021). Influence of Sample Matrix on Determination of Histamine in Fish by Surface Enhanced Raman Spectroscopy Coupled with Chemometric Modelling. Foods, 10(8), 1767. https://doi.org/10.3390/foods10081767








