Retarding Oxidative and Enzymatic Degradation of Phenolic Compounds Using Large-Ring Cycloamylose
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Analysis of Produced Cycloamylose
2.3. Stability of PCs with CA against Oxidation
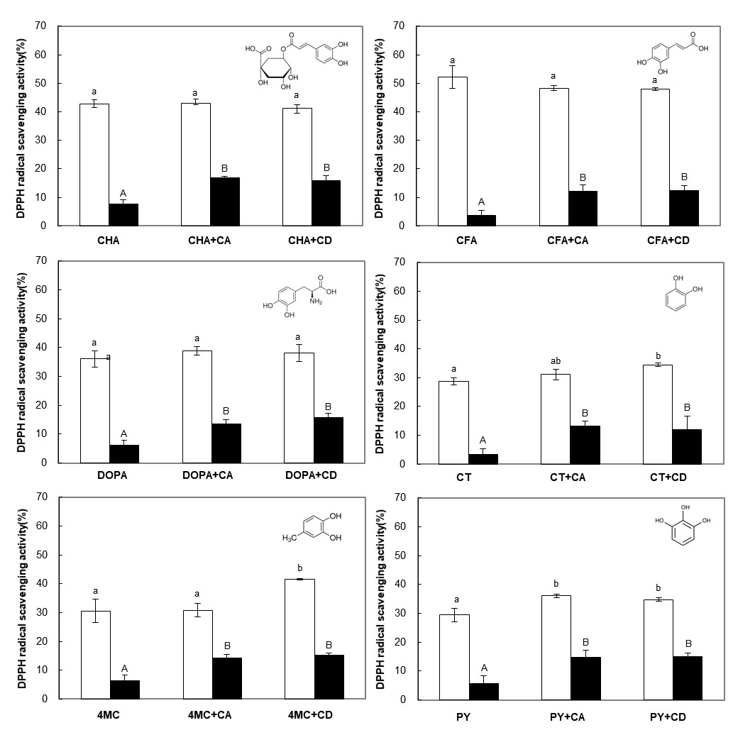
2.3.1. DPPH Radical Scavenging Activity
2.3.2. Oxidative Stability of PCs with CA
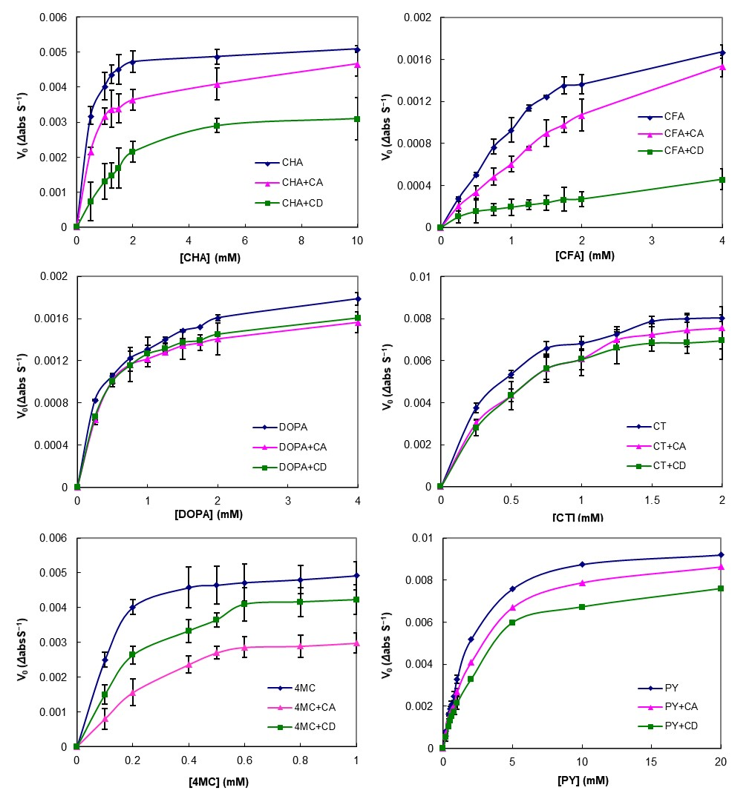
2.4. Stability of PCs with CA against Enzymatic Oxidation
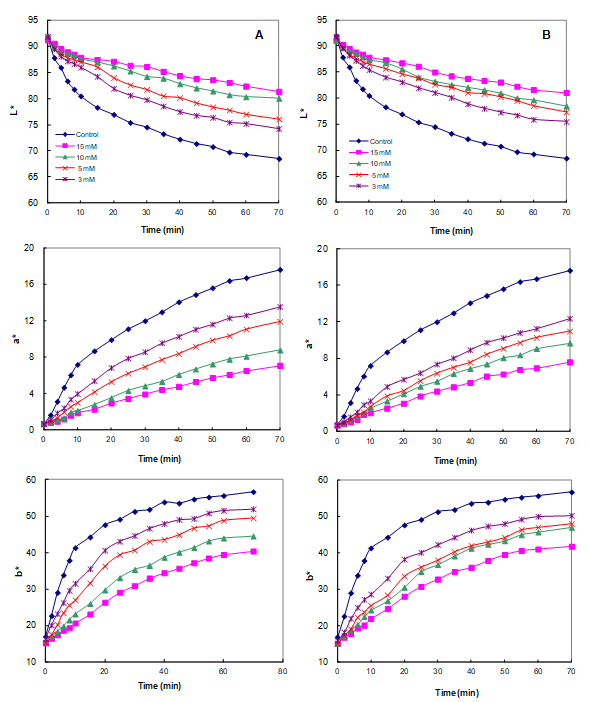
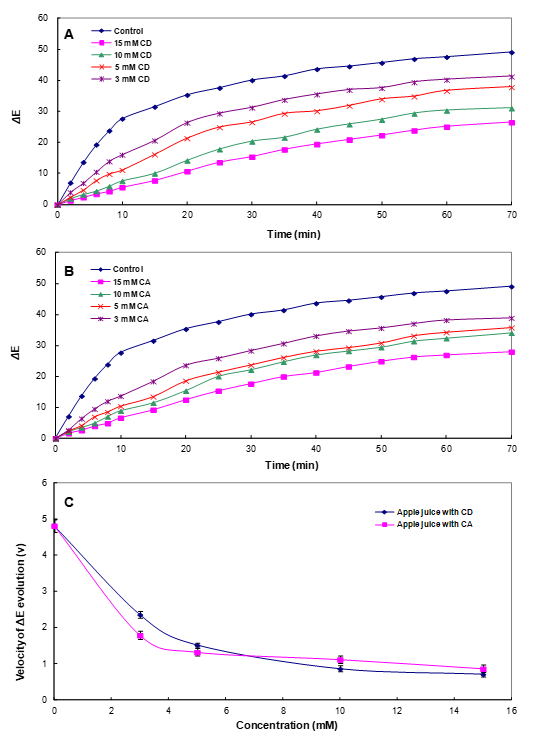
2.5. Color Evolution Assessment
2.6. Statistical Analysis
3. Results and Discussion
3.1. Synthesis of CA
3.2. Effect of CA on the Oxidative Stability of PCs during Storage
3.3. Effect of CA on PCs Stability against Enzymatic Oxidation
3.4. Effect of CA on the Color Stability of Apple Juice
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Croguennec, T. Enzymatic browning. In Handbook of Food Science and Technology 1: Food Alteration and Food Quality, 1st ed.; Jeantet, R., Croguennec, T., Schuck, P., Brulé, G., Eds.; John Wiley & Sons Inc: Hoboken, NJ, USA, 2016; pp. 159–181. [Google Scholar]
- Ghasemzadeh, A.; Ghasemzadeh, N. Flavonoids and phenolic acids: Role and biochemical activity in plants and human. J. Med. Plants Res. 2011, 5, 6697–6703. [Google Scholar] [CrossRef]
- Rice-Evans, C.; Miller, N.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- Robards, K.; Prenzler, P.D.; Tucker, G.; Swatsitang, P.; Glover, W. Phenolic compounds and their role in oxidative processes in fruits. Food Chem. 1999, 66, 401–436. [Google Scholar] [CrossRef]
- Burton, S.G. Biocatalysis with polyphenol oxidase: A review. Catal. Today. 1994, 22, 459–487. [Google Scholar] [CrossRef]
- Queiroz, C.; Mendes Lopes, M.L.; Fialho, E.; Valente-Mesquita, V.L. Polyphenol oxidase: Characteristics and mechanisms of browning control. Food Rev. Int. 2008, 24, 361–375. [Google Scholar] [CrossRef]
- Luo, Y.; Barbosa-Cánovas, G.V. Enzymatic browning and its inhibition in new apple cultivars slices using 4-hexylresorcinol in combination with ascorbic acid. Food Sci. Technol. Int. 1997, 3, 195–201. [Google Scholar] [CrossRef]
- Nicolas, J.J.; Richard-Forget, F.C.; Goupy, P.M.; Amiot, M.J.; Aubert, S.Y. Enzymatic browning reactions in apple and apple products. Crit. Rev. Food Sci. Nutr. 1994, 34, 109–157. [Google Scholar] [CrossRef]
- McEvily, A.J.; Iyengar, R.; Otwell, W.S. Inhibition of enzymatic browning in foods and beverages. Crit. Rev. Food Sci. Nutr. 1992, 32, 253–273. [Google Scholar] [CrossRef]
- Cao, X.; Cai, C.; Wang, Y.; Zheng, X. The inactivation kinetics of polyphenol oxidase and peroxidase in bayberry juice during thermal and ultrasound treatments. Innov. Food Sci. Emerg. Technol. 2018, 45, 169–178. [Google Scholar] [CrossRef]
- Moon, K.M.; Kwon, E.B.; Lee, B.; Kim, C.Y. Recent trends in controlling the enzymatic browning of fruit and vegetable products. Molecules 2020, 25, 2754. [Google Scholar] [CrossRef]
- Muñoz-Pina, S.; Ros-Lis, J.V.; Argüelles, Á.; Coll, C.; Martínez-Máñez, R.; Andrés, A. Full inhibition of enzymatic browning in the presence of thiol-functionalised silica nanomaterial. Food Chem. 2018, 241, 199–205. [Google Scholar] [CrossRef]
- Sikora, M.; Świeca, M. Effect of ascorbic acid postharvest treatment on enzymatic browning, phenolics and antioxidant capacity of stored mung bean sprouts. Food Chem. 2018, 239, 1160–1166. [Google Scholar] [CrossRef]
- Zhou, L.; Liu, W.; Xiong, Z.; Zou, L.; Chen, J.; Liu, J.; Zhong, J. Different modes of inhibition for organic acids on polyphenoloxidase. Food Chem. 2016, 199, 439–446. [Google Scholar] [CrossRef]
- Crouzet, J.; Bastier, P.; Signoret, A.; Mentink, L. Enzymatic browning inhibition by cyclodextrins. Food Ingred. Eur. 1991, 1991, 293–295. [Google Scholar]
- Le Bourvellec, C.; Renard, C.M.G.C. Interactions between polyphenols and macromolecules: Quantification methods and mechanisms. Crit. Rev. Food Sci. Nutr. 2012, 52, 213–248. [Google Scholar] [CrossRef]
- López-Nicolás, J.M.; Núñez-Delicado, E.; Sánchez-Ferrer, Á.; García-Carmona, F. Kinetic model of apple juice enzymatic browning in the presence of cyclodextrins: The use of maltosyl-β-cyclodextrin as secondary antioxidant. Food Chem. 2007, 101, 1164–1171. [Google Scholar] [CrossRef]
- López-Nicolás, J.M.; Pérez-López, A.J.; Carbonell-Barrachina, Á.; García-Carmona, F. Use of natural and modified cyclodextrins as inhibiting agents of peach juice enzymatic browning. J. Agric. Food Chem. 2007, 55, 5312–5319. [Google Scholar] [CrossRef]
- López-Nicolás, J.M.; García-Carmona, F. Use of cyclodextrins as secondary antioxidants to improve the color of fresh pear juice. J. Agric. Food Chem. 2007, 55, 6330–6338. [Google Scholar] [CrossRef]
- Singh, V.; Jadhav, S.B.; Singhal, R.S. Interaction of polyphenol oxidase of Solanum tuberosum with β-cyclodextrin: Process details and applications. Int. J. Biol. Macromol. 2015, 80, 469–474. [Google Scholar] [CrossRef]
- Alvarez-Parrilla, E.; Laura, A.; Torres-Rivas, F.; Rodrigo-Garcia, J.; González-Aguilar, G.A. Complexation of apple antioxidants: Chlorogenic acid, quercetin and rutin by β-cyclodextrin (β-CD). J. Incl. Phenom. Macrocycl. Chem. 2005, 53, 121–129. [Google Scholar] [CrossRef]
- Billaud, C.; Regaudie, E.; Fayad, N.; Richard-Forget, F.; Nicolas, J. Effect of cyclodextrins on polyphenol oxidation catalyzed by apple polyphenol oxidase. In Enzymatic Browning and its Prevention; Lee, C.Y., Whitaker, J.R., Eds.; ACS: Washington, DC, USA, 1995; pp. 295–312. [Google Scholar] [CrossRef]
- Fayad, N.; Marchal, L.; Billaud, C.; Nicolas, J. Comparison of β-cyclodextrin effect on polyphenol oxidation catalyzed by purified polyphenol oxidase from different sources. J. Agric. Food Chem. 1997, 45, 2442–2446. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, Y.; Gao, X.; Fu, J.; Hu, L. Application of cyclodextrin in food industry. Crit. Rev. Food Sci. Nutr. 2020, 1–15. [Google Scholar] [CrossRef]
- Astray, G.; Gonzalez-Barreiro, C.; Mejuto, J.C.; Rial-Otero, R.; Simal-Gandara, J. A review on the use of cyclodextrins in foods. Food Hydrocoll. 2009, 23, 1631–1640. [Google Scholar] [CrossRef]
- Stella, V.J.; He, Q. Cyclodextrins. Toxicol. Pathol. 2008, 36, 30–42. [Google Scholar] [CrossRef]
- Takaha, T.; Yanase, M.; Takata, H.; Okada, S.; Smith, S.M. Potato D-enzyme catalyzes the cyclization of amylose to produce cycloamylose, a novel cyclic glucan. J. Biol. Chem. 1996, 271, 2901–2908. [Google Scholar] [CrossRef]
- Baek, H.H.; Kim, D.H.; Kwon, S.Y.; Rho, S.J.; Kim, D.W.; Choi, H.G.; Kim, Y.R.; Yong, C.S. Development of novel ibuprofen-loaded solid dispersion with enhanced bioavailability using cycloamylose. Arch. Pharm. Res. 2012, 35, 683–689. [Google Scholar] [CrossRef]
- Mun, S.; Rho, S.J.; Kim, Y.R. Study of inclusion complexes of cycloamylose with surfactants by isothermal titration calorimetry. Carbohydr. Polym. 2009, 77, 223–230. [Google Scholar] [CrossRef]
- Toita, S.; Morimoto, N.; Akiyoshi, K. Functional cycloamylose as a polysaccharide-based biomaterial: Application in a gene delivery system. Biomacromolecules 2009, 11, 397–401. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, P.M.; Jaime, C. Insights into the structure of large-ring cyclodextrins through molecular dynamics simulations in solution. J. Phys. Chem. B 2004, 108, 6261–6274. [Google Scholar] [CrossRef] [PubMed]
- Gotsev, M.G.; Ivanov, P.M. Large-ring cyclodextrins. Further support for the preferred conformations of CD26 in water solution: Molecular dynamics studies on CD26-derived conformations of CDn (n= 24, 25, 27, 28, 29, 30). Int. J. Quantum Chem. 2007, 107, 1657–1672. [Google Scholar] [CrossRef]
- Larsen, K.L. Large cyclodextrins. J. Incl. Phenom. Macrocycl. Chem. 2002, 43, 1–13. [Google Scholar] [CrossRef]
- Taira, H.; Nagase, H.; Endo, T.; Ueda, H. Isolation, purification and characterization of large-ring cyclodextrins (CD36–CD39). J. Incl. Phenom. Macrocycl. Chem. 2006, 56, 23–28. [Google Scholar] [CrossRef]
- Ueda, H. Large ring cyclodextrins-Recent progress. FABAD J. Pharm. Sci. 2004, 29, 27–38. [Google Scholar]
- Rho, S.J.; Mun, S.; Hong, J.S.; Kim, Y.L.; Do, H.V.; Kim, Y.W.; Han, S.I.; Kim, Y.R. Physicochemical interactions of cycloamylose with phenolic compounds. Carbohydr. Polym. 2017, 174, 980–989. [Google Scholar] [CrossRef]
- Blois, M.S. Antioxidant determinations by the use of a stable free radical. Nature 1985, 181, 1199–1200. [Google Scholar] [CrossRef]
- Takaha, T.; Smith, S.M. The functions of 4-α-glucanotransferases and their use for the production of cyclic glucans. Biotechnol. Genet. Eng. Rev. 1999, 16, 257–280. [Google Scholar] [CrossRef]
- Park, J.H.; Kim, H.J.; Kim, Y.H.; Cha, H.; Kim, Y.W.; Kim, T.J.; Kim, Y.R.; Park, K.H. The action mode of Thermus aquaticus YT-1 4-α-glucanotransferase and its chimeric enzymes introduced with starch-binding domain on amylose and amylopectin. Carbohydr. Polym. 2007, 67, 164–173. [Google Scholar] [CrossRef]
- Leemhuis, H.; Kelly, R.M.; Dijkhuizen, L. Engineering of cyclodextrin glucanotransferases and the impact for biotechnological applications. Appl. Microbiol. Biotechnol. 2010, 85, 823–835. [Google Scholar] [CrossRef]
- Goh, K.M.; Mahadi, N.M.; Hassan, O.; Rahman, R.N.Z.R.A.; Illias, R.M. A predominant β-CGTase G1 engineered to elucidate the relationship between protein structure and product specificity. J. Mol. Catal. B Enzym. 2009, 57, 270–277. [Google Scholar] [CrossRef]
- van der Veen, B.A.; Uitdehaag, J.C.; Penninga, D.; van Alebeek, G.J.W.; Smith, L.M.; Dijkstra, B.W.; Dijkhuizen, L. Rational design of cyclodextrin glycosyltransferase from Bacillus circulans strain 251 to increase α-cyclodextrin production. J. Mol. Biol. 2000, 296, 1027–1038. [Google Scholar] [CrossRef]
- Del Valle, E.M. Cyclodextrins and their uses: A review. Process Biochem. 2004, 39, 1033–1046. [Google Scholar] [CrossRef]
- Stražišar, M.; Andrenšek, S.; Šmidovnik, A. Effect of β-cyclodextrin on antioxidant activity of coumaric acids. Food Chem. 2008, 110, 636–642. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C.L.W.T. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Cos, P.; Rajan, P.; Vedernikova, I.; Calomme, M.; Pieters, L.; Vlietinck, A.J.; Augustyns, K.; Haemers, A.; Berghe, D.V. In vitro antioxidant profile of phenolic acid derivatives. Free Radic. Res. 2002, 36, 711–716. [Google Scholar] [CrossRef]
- Cao, G.; Sofic, E.; Prior, R.L. Antioxidant and prooxidant behavior of flavonoids: Structure-activity relationships. Free Radic. Biol. Med. 1997, 22, 749–760. [Google Scholar] [CrossRef]
- Yuan, E.; Liu, B.; Li, W.; Li, Q. Characterization and antioxidant activity of the complex of phloridzin and hydroxypropyl-β-cyclodextrin. Trop. J. Pharm. Res. 2012, 11, 545–551. [Google Scholar] [CrossRef][Green Version]
- Zhao, M.; Wang, H.; Yang, B.; Tao, H. Identification of cyclodextrin inclusion complex of chlorogenic acid and its antimicrobial activity. Food Chem. 2010, 120, 1138–1142. [Google Scholar] [CrossRef]
- Lu, Z.; Cheng, B.; Hu, Y.; Zhang, Y.; Zou, G. Complexation of resveratrol with cyclodextrins: Solubility and antioxidant activity. Food Chem. 2009, 113, 17–20. [Google Scholar] [CrossRef]
- Bocharova, O.; Bocharova, M. The dual role of phenolic compounds in oxidative changes in fruit products. Int. Food Res. J. 2017, 24, 1261–1269. [Google Scholar]
- Jiang, S.; Penner, M.H. The nature of β-cyclodextrin inhibition of potato polyphenol oxidase-catalyzed reactions. Food Chem. 2019, 298, 125004. [Google Scholar] [CrossRef]
- Aachmann, F.L.; Larsen, K.L.; Wimmer, R. Interactions of cyclodextrins with aromatic amino acids: A basis for protein interactions. J. Incl. Phenom. Macrocycl. Chem. 2012, 73, 349–357. [Google Scholar] [CrossRef]
- Alvarez-Parrilla, E.; De la Rosa, L.A.; Rodrigo-García, J.; Escobedo-González, R.; Mercado-Mercado, G.; Moyers-Montoya, E.; Vázquez-Flores, A.; González-Aguilar, G.A. Dual effect of β-cyclodextrin (β-CD) on the inhibition of apple polyphenol oxidase by 4-hexylresorcinol (HR) and methyl jasmonate (MJ). Food Chem. 2007, 101, 1346–1356. [Google Scholar] [CrossRef]
- Campo, A.F.; Sancho, M.I.; Melo, G.; Dávila, Y.A.; Gasull, E. In vitro and in vivo inhibition of Hass avocado polyphenol oxidase enzymatic browning by paeonol, β-cyclodextrin, and paeonol: β-cyclodextrin inclusion complex. J. Biosci. Bioeng. 2019, 127, 703–709. [Google Scholar] [CrossRef]
- Friedman, M. Food browning and its prevention: An overview. J. Agric. Food Chem. 1996, 44, 631–653. [Google Scholar] [CrossRef]
- Shimada, J.; Handa, S.; Kaneko, H.; Takada, T. Conformation of novel cycloamylose: Topological aspects and simulations. Macromolecules 1996, 29, 6408–6421. [Google Scholar] [CrossRef]

| Host Molecules 1 | kd (Day−1) | |||||
|---|---|---|---|---|---|---|
| CHA 2 | CFA | DOPA | CT | 4MC | PY | |
| Control | 0.075 ± 0.0081 b | 0.161 ± 0.0024 b | 0.089 ± 0.0026 b | 0.102 ± 0.0020 c | 0.079 ± 0.0018 c | 0.061 ± 0.0016 b |
| CA | 0.037 ± 0.0015 a | 0.046 ± 0.0025 a | 0.036 ± 0.0015 a | 0.024 ± 0.0016 a | 0.026 ± 0.0013 a | 0.030 ± 0.0015 a |
| CD | 0.036 ± 0.0017 a | 0.054 ± 0.0026 a | 0.029 ± 0.0017 a | 0.040 ± 0.0017 b | 0.043 ± 0.0017 b | 0.037 ± 0.0018 a |
| Host Molecules | t1/2 (Day) | |||||
| CHA | CFA | DOPA | CT | 4MC | PY | |
| Control | 9.34 ± 1.424 a | 4.30 ± 0.091 a | 7.79 ± 0.322 a | 6.80 ± 0.185 a | 8.81 ± 0.29 a | 11.39 ± 0.430 a |
| CA | 18.97 ± 1.099 b | 15.04 ± 1.151 b | 19.50 ± 1.146 b | 29.24 ± 2.728 b | 27.14 ± 1.979 b | 23.31 ± 1.649 b |
| CD | 19.57 ± 1.325 b | 12.79 ± 0.866 b | 23.65 ± 1.934 b | 17.58 ± 1.038 c | 16.14 ± 0.908 c | 18.88 ± 1.284 b |
| Guest Molecules 2 | Vmax 3 | Km | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Non-Linear Fitting 4 | Linear Fitting 5 | Non-Linear Fitting | Linear Fitting | |||||||||
| C 1 | CA | CD | C | CA | CD | C | CA | CD | C | CA | CD | |
| CHA | 5.3 | 4.8 | 3.8 | 5.4 | 4.8 | 4.4 | 0.30 | 0.56 | 1.81 | 0.33 | 0.59 | 2.45 |
| CFA | 2.5 | 2.4 | 0.7 | 3.3 | 2.3 | 0.5 | 1.56 | 2.75 | 2.90 | 2.67 | 2.12 | 1.68 |
| DOPA | 1.9 | 1.7 | 1.7 | 1.8 | 1.7 | 1.8 | 0.36 | 0.38 | 0.38 | 0.30 | 0.41 | 0.40 |
| CT | 9.8 | 9.7 | 9.0 | 9.7 | 9.8 | 9.5 | 0.40 | 0.56 | 0.50 | 0.40 | 0.57 | 0.59 |
| 4MC | 5.5 | 4.1 | 5.3 | 5.7 | 5.3 | 5.7 | 0.10 | 0.32 | 0.22 | 0.12 | 0.55 | 0.27 |
| PY | 10.5 | 10 | 8.9 | 10.8 | 9.1 | 9.3 | 2.22 | 2.74 | 3.02 | 2.41 | 2.41 | 3.28 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rho, S.-J.; Mun, S.; Park, J.; Kim, Y.-R. Retarding Oxidative and Enzymatic Degradation of Phenolic Compounds Using Large-Ring Cycloamylose. Foods 2021, 10, 1457. https://doi.org/10.3390/foods10071457
Rho S-J, Mun S, Park J, Kim Y-R. Retarding Oxidative and Enzymatic Degradation of Phenolic Compounds Using Large-Ring Cycloamylose. Foods. 2021; 10(7):1457. https://doi.org/10.3390/foods10071457
Chicago/Turabian StyleRho, Shin-Joung, Saehun Mun, Jiwoon Park, and Yong-Ro Kim. 2021. "Retarding Oxidative and Enzymatic Degradation of Phenolic Compounds Using Large-Ring Cycloamylose" Foods 10, no. 7: 1457. https://doi.org/10.3390/foods10071457
APA StyleRho, S.-J., Mun, S., Park, J., & Kim, Y.-R. (2021). Retarding Oxidative and Enzymatic Degradation of Phenolic Compounds Using Large-Ring Cycloamylose. Foods, 10(7), 1457. https://doi.org/10.3390/foods10071457







