Reusable Plastic Crates (RPCs) for Fresh Produce (Case Study on Cauliflowers): Sustainable Packaging but Potential Salmonella Survival and Risk of Cross-Contamination
Abstract
1. Introduction
2. Materials and Methods
2.1. Transfer and Survival of Salmonella via Different Fresh-Produce Container Materials
2.1.1. Via Polypropylene
Salmonella Strains and Inoculum Preparation
Plant Material and Inoculation
Cross-Contamination
Storage
Sampling and Microbiological Analysis
2.1.2. Effect of the Inoculum Size
2.1.3. Via Cardboard and MDF
2.1.4. Statistical Analysis
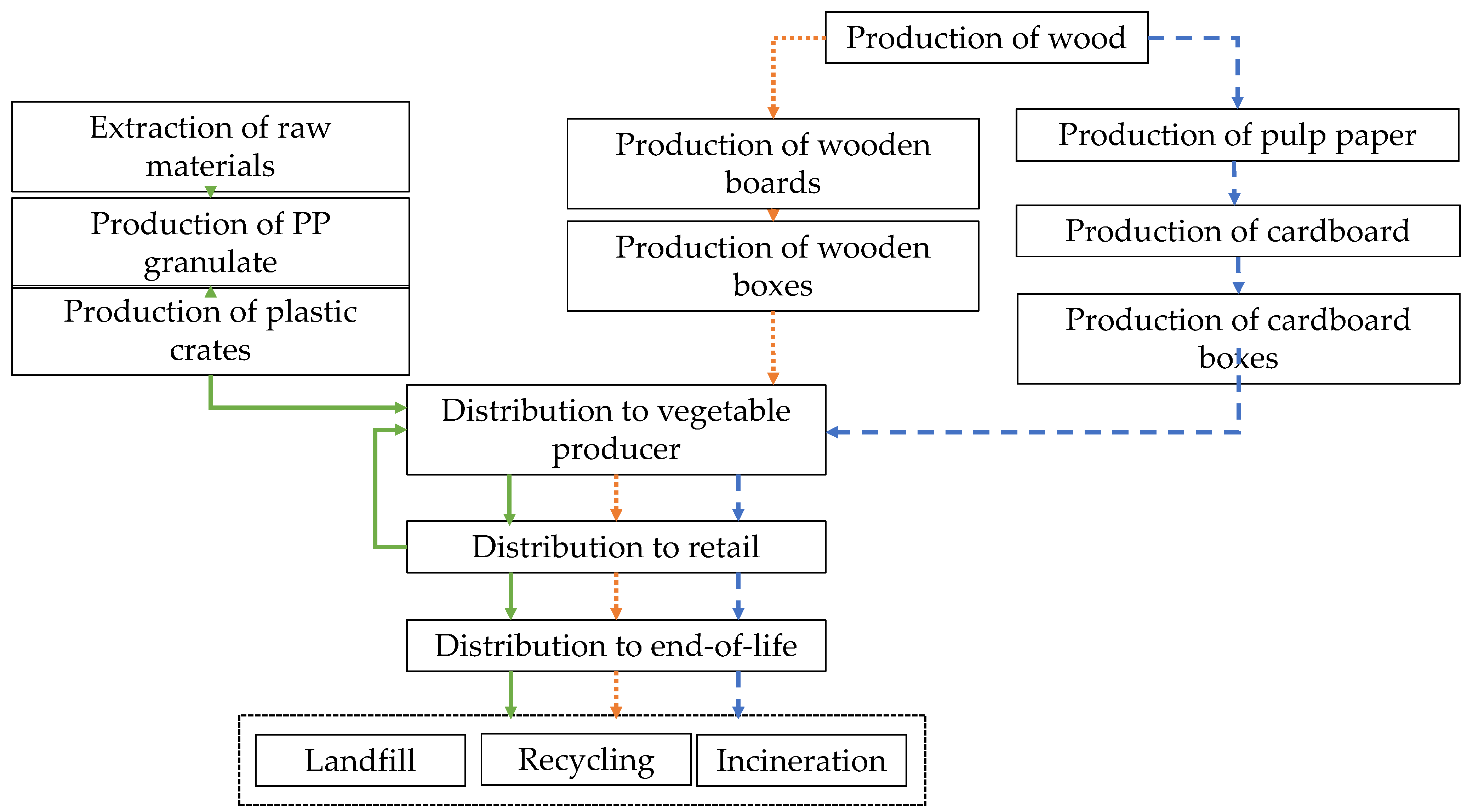
2.2. Environmental Impact of Different Types of Fresh Produce Handling Containers
2.2.1. Goal and Scope
2.2.2. Life Cycle Inventory
2.2.3. Impact Assessment
3. Results
3.1. Transfer and Survival of Salmonella in Cauliflowers via Different Container Materials
3.1.1. Via Polypropylene
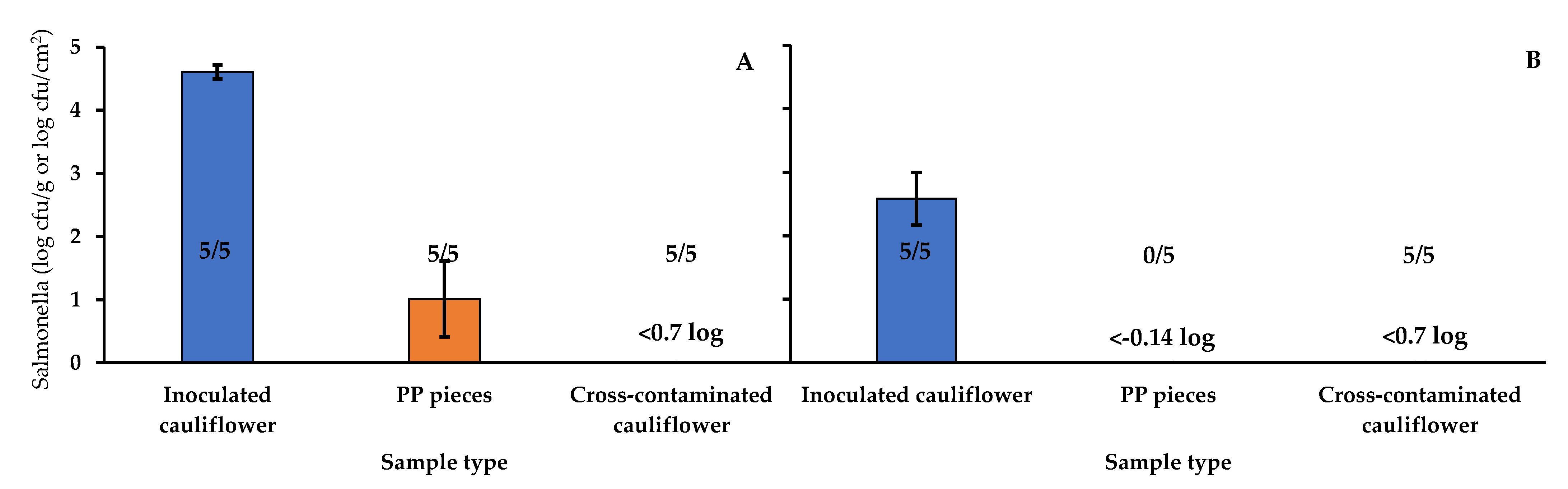
Transfer
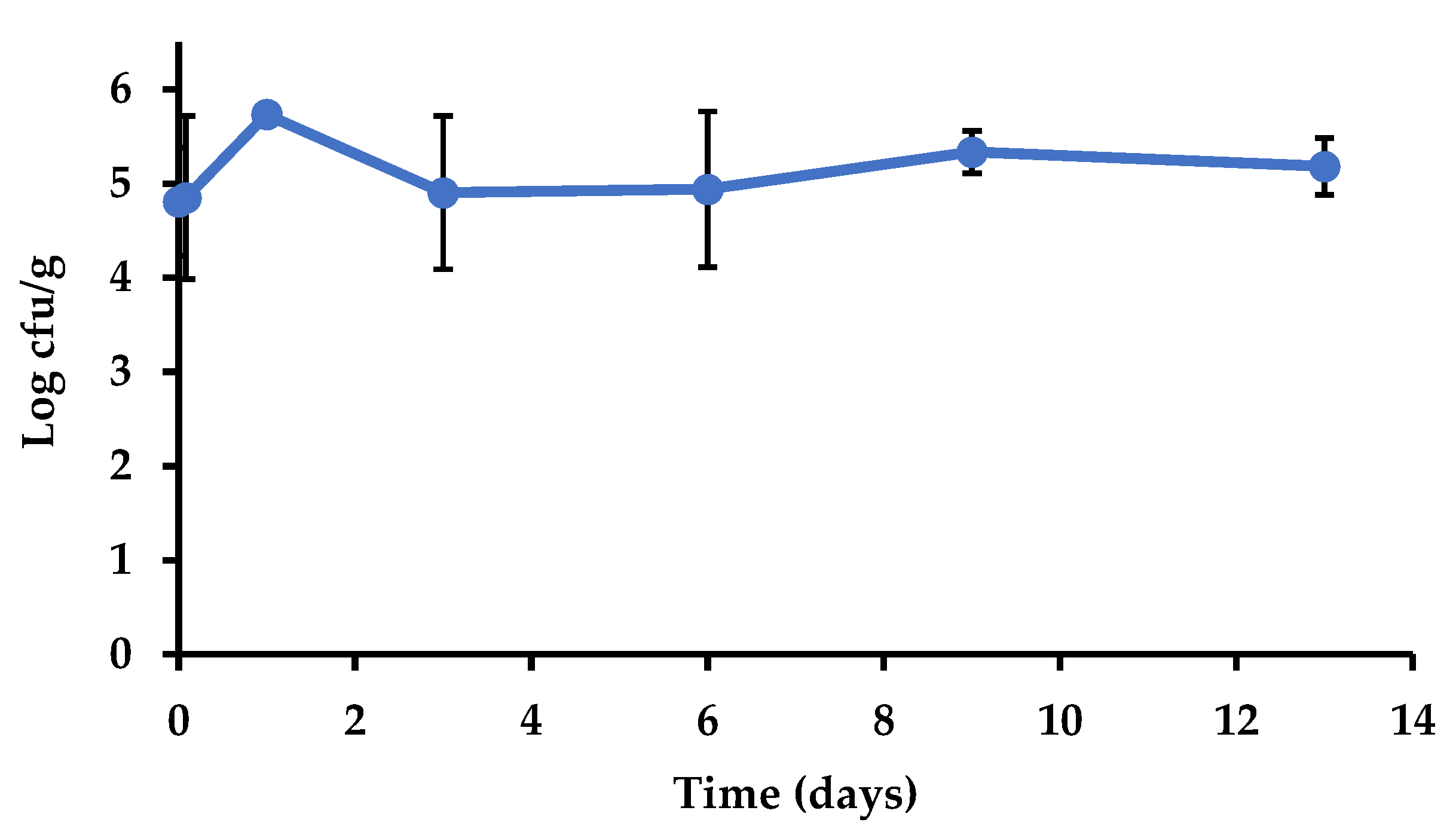
Salmonella Survival
3.1.2. Effect of the Salmonella Inoculum Size
3.1.3. Via Cardboard and MDF
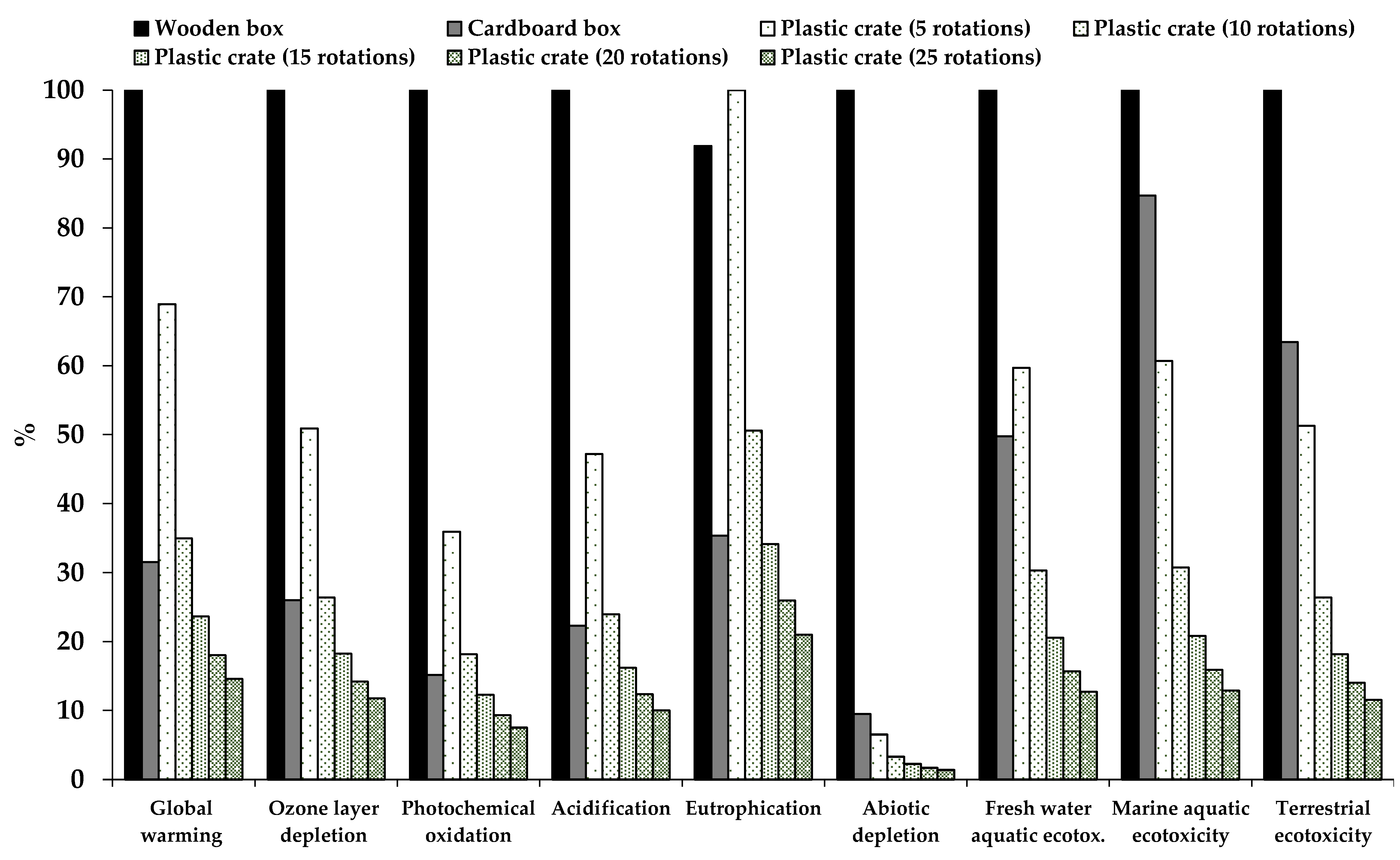
3.2. Environmental Impact of Different Types of Fresh Produce Handling Containers
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Suslow, T.V. Minimizing Risk in Multiple-Use Containers. Food Safety & Quality Magazine: Suslow UC Davis April 2015 Full Version. Available online: https://ucfoodsafety.ucdavis.edu/sites/g/files/dgvnsk7366/files/inline-files/212397.pdf (accessed on 22 March 2021).
- Abejón, R.; Bala, A.; Vázquez-Rowe, I.; Aldaco, R.; Fullana-i-Palmer, P. When plastic packaging should be preferred: Life cycle analysis of packages for fruit and vegetable distribution in the Spanish peninsular market. Resour. Conserv. Recy. 2020, 155, 104666. [Google Scholar] [CrossRef]
- Warriner, K. Microbiological Standards for RPCs within Produce Grower Facilities. University of Guelph, Department of Food Science. 2013. Available online: https://26mvtbfbbnv3ruuzp1625r59-wpengine.netdna-ssl.com/wp-content/uploads/PDFs/Package_Cleanliness/RPC_Sanitation_Testing_and_Research_a.pdf (accessed on 22 March 2021).
- Suslow, T.V. Assessment of General RPC Cleanliness as Delivered for Use in Packing and Distribution of Fresh Produce; Assessment of RPC Cleanliness: Final Report; University of California at Davis, Department of Plant Sciences: Davis, CA, USA, 2014; Available online: https://26mvtbfbbnv3ruuzp1625r59-wpengine.netdna-ssl.com/wp-content/uploads/PDFs/Package_Cleanliness/RPC_Sanitation_Testing_and_Research_c.pdf (accessed on 22 March 2021).
- Zhu, Y.; Wu, F.; Trmcic, A.; Wang, S.; Warriner, K. Microbiological status of RPCs in commercial grower/packer operations and risk of Salmonella cross-contamination between containers and cucumbers. Food Control 2020, 110, 107021. [Google Scholar] [CrossRef]
- Truchado, P.; Allende, A. Relevance of fresh fruits and vegetables in foodborne outbreaks and the significance of the physiological state of bacteria|[La implicación de las frutas y hortalizas en las toxiinfecciones alimentarias y la relevancia del estado fisiológico de las bacterias]. Arbor 2020, 196, 1–9. [Google Scholar] [CrossRef]
- Aworh, O.C. Food safety issues in fresh produce supply chain with particular reference to sub-Saharan Africa. Food Control 2021, 123, 107737. [Google Scholar] [CrossRef]
- Jacxsens, L.; Uyttendaele, M.; Luning, P.; Allende, A. Food safety management and risk assessment in the fresh produce supply chain. IOP Conf. Ser. Mat. Sci. 2017, 193. [Google Scholar] [CrossRef]
- U.S. Food and Drugs Administration. Recalls, Market Withdrawals & Safety Alerts. 2021. Available online: https://www.fda.gov/safety/recalls/ (accessed on 22 March 2021).
- Zhang, H.; Yamamoto, E.; Murphy, J.; Locas, A. Microbiological safety of ready-to-eat fresh-cut fruits and vegetables sold on the Canadian retail market. Int. J. Food Microbiol. 2020, 335, 108855. [Google Scholar] [CrossRef]
- Quiroz-Santiago, C.; Rodas-Suárez, O.R.; Vázquez, C.R.; Fernández, F.J.; Quiñones-Ramírez, E.I.; Vázquez-Salinas, C. Prevalence of Salmonella in vegetables from Mexico. J. Food Prot. 2020, 72, 1279–1282. [Google Scholar] [CrossRef]
- Gkana, E.; Chorianopoulos, N.; Grounta, A.; Koutsoumanis, K.; Nychas, G.-J.E. Effect of inoculum size, bacterial species, type of surfaces and contact time to the transfer of foodborne pathogens from inoculated to non-inoculated beef fillets via food processing surfaces. Food Microbiol. 2017, 62, 51–57. [Google Scholar] [CrossRef]
- Villegas, B.M.; Hall, N.O.; Ryser, E.T.; Marks, B.P. Influence of physical variables on the transfer of Salmonella Typhimurium LT2 between potato (Solanum tuberosum) and stainless steel via static and dynamic contact. Food Microbiol. 2020, 92, 103607. [Google Scholar] [CrossRef] [PubMed]
- Buchholz, A.L.; Davidson, G.R.; Marks, B.P.; Todd, E.C.D.; Ryser, E.T. Transfer of Escherichia coli O157:H7 from equipment surfaces to fresh-cut leafy greens during processing in a model pilot-plant production line with sanitizer-free water. J. Food Prot. 2012, 75, 1920–1929. [Google Scholar] [CrossRef]
- Newman, K.L.; Bartz, F.E.; Johnston, L.; Moe, C.L.; Jaykus, L.-A.; Leon, J.S. Microbial load of fresh produce and paired equipment surfaces in packing facilities near the U.S. and Mexico border. J. Food Prot. 2017, 80, 582–589. [Google Scholar] [CrossRef] [PubMed]
- Cotter, J.; Talbert, J.; Goddard, J.; Autio, W.; McLandsborough, L. Influence of soil particles on the survival of Salmonella on plastic tomato harvest containers. Technical Abstract. In Proceedings of the IAFP Annual Meeting, Providence, RI, USA, 22–25 July 2012. [Google Scholar]
- De Abrew Abeysundara, P.; Dhowlaghar, N.; Nannapaneni, R.; Schilling, M.W.; Mahmoud, B.; Sharma, C.S.; Ma, D.-P. Salmonella enterica growth and biofilm formation in flesh and peel cantaloupe extracts on four food-contact surfaces. Int. J. Food Microbiol. 2018, 280, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Patrignani, F.; Siroli, L.; Gardini, F.; Lanciotti, R. Contribution of two different packaging material to microbial contamination of peaches: Implications in their microbiological quality. Front. Microbiol. 2016, 7, 938. [Google Scholar] [CrossRef] [PubMed]
- Aviat, F.; Le Bayon, I.; Federighi, M.; Montibus, M. Comparative study of microbiological transfer from four materials used in direct contact with apples. Int. J. Food Microbiol. 2020, 333. [Google Scholar] [CrossRef] [PubMed]
- Siroli, L.; Patrignani, F.; Serrazanetti, D.I.; Chiavari, C.; Benevelli, M.; Grazia, L.; Lanciotti, R. Survival of spoilage and pathogenic microorganisms on cardboard and plastic packaging materials. Front. Microbiol. 2017, 8, 2606. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, P.; Singhal, R.S. Broccoli and Cauliflower: Production, Quality, and Processing. In Handbook of Vegetables and Vegetable Processing, 2nd ed.; Siddiq, M., Uebersax, M.A., Eds.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2018; Volume II, pp. 535–558. [Google Scholar]
- Montibus, M.; Ismaïl, R.; Michel, V.; Federighi, M.; Aviat, F.; Le Bayon, I. Assessment of Penicillium expansum and Escherichia coli transfer from poplar crates to apples. Food Control 2016, 60, 95–102. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Albrecht, S.; Brandstetter, P.; Beck, T.; Fullana, I.; Palmer, P.; Grönman, K.; Baitz, M.; Deimling, S.; Sandilands, J.; Fischer, M. An extended life cycle analysis of packaging systems for fruit and vegetable transport in Europe. Int. J. Life Cycle Assess. 2013, 18, 1549–1567. [Google Scholar] [CrossRef]
- Li, K.; Khouryieh, H.; Jones, L.; Etienne, X.; Shen, C. Assessing farmers market produce vendors’ handling of containers and evaluation of the survival of Salmonella and Listeria monocytogenes on plastic, pressed-card, and wood container surfaces at refrigerated and room temperature. Food Control 2018, 94, 116–122. [Google Scholar] [CrossRef]
- ISO. ISO 14000 Collection 2. In ISO 14040:2006: Environmental Management. Life Cycle Assessment. Principles and Framework; International Organization for Standardization: Geneva, Switzerland, 2006. [Google Scholar]
- ISO. ISO 14000 Collection 2. In ISO14044:2006: Life Cycle Assessment. Requirements and Guidelines; International Organization for Standardization: Geneva, Switzerland, 2006. [Google Scholar]
- Environdec. Produc Category Rules: Crates for Food; Environtec Limited: Chelmsford, UK, 2020; pp. 1–29. [Google Scholar]
- Boletín Oficial del Estado. Real Decreto 888/1988, de 29 de Julio, por el que se Aprueba la Norma General Sobre Recipientes que Contengan Productos Alimenticios Frescos, de Carácter Perecedero, no Envasados o Envueltos. «BOE» núm. 1988, pp. 187, 24293–24294. Available online: https://www.boe.es/diario_boe/txt.php?id=BOE-A-1988-19396 (accessed on 31 May 2021).
- Betelgeux. Lavadora de Cajas EKW 2500—Betelgeux. 2021. Available online: https://www.betelgeux.es/equipos/maquinas-lavadoras/lavadora-cajas-ekw-2500/ (accessed on 31 May 2021).
- TEMIC. Technical Sheet of Crates Washing Tunnels TR-42-TEMIC S.L. Available online: http://www.temicsl.com/downloads/tUnel-lavado-de-cajas-tr-424.pdf (accessed on 31 May 2021).
- MITECO. Memoria Anual de Generación y Gestión de Residuos de Competencia Municipal. 2018. Available online: https://www.miteco.gob.es/es/calidad-y-evaluacion-ambiental/publicaciones/memoriaresiduosmunicipales2018_tcm30-521965.pdf (accessed on 22 April 2021).
- European Federation of Corrugated Board Manufacturers (FEFCO); CEPI ContainerBoard (CCB). European Database for Corrugated Board Life Cycle Studies. 2018. Available online: https://www.fefco.org/lca (accessed on 6 May 2021).
- Agence de L’environnement et de la Maîtrise de L’énergie (ADEME). Analyse du Cycle de vie Des Caisses en Bois, Carton Ondulé et Plastique Pour Pommes. 2000. Available online: https://www.ademe.fr/sites/default/files/assets/documents/28246_acvs.pdf (accessed on 6 May 2021).
- Accorsi, R.; Cascini, A.; Cholette, S.; Manzini, R.; Mora, C. Economic and environmental assessment of reusable plastic containers: A food catering supply chain case study. Int. J. Prod. Econ. 2014, 152, 88–101. [Google Scholar] [CrossRef]
- SimaPro|The World’s Leading LCA Software. (n.d.). Available online: https://simapro.com/ (accessed on 18 March 2021).
- Ecoinvent. (n.d.). Ecoinvent Database 3.7.1. Available online: https://www.ecoinvent.org/ (accessed on 22 March 2021).
- Jones, S.L.; Ricke, S.C.; Keith Roper, D.; Gibson, K.E. Swabbing the surface: Critical factors in environmental monitoring and a path towards standardization and improvement. Crit. Rev. Food Sci. 2020, 60, 225–243. [Google Scholar] [CrossRef] [PubMed]
- Keeratipibul, S.; Laovittayanurak, T.; Pornruangsarp, O.; Chaturongkasumrit, Y.; Takahashi, H.; Techaruvichit, P. Effect of swabbing techniques on the efficiency of bacterial recovery from food contact surfaces. Food Control 2017, 77, 139–144. [Google Scholar] [CrossRef]
- Jensen, D.A.; Friedrich, L.M.; Harris, L.J.; Danyluk, M.D.; Schaffner, D.W. Quantifying transfer rates of Salmonella and Escherichia coli O157:H7 between fresh-cut produce and common kitchen surfaces. J. Food Prot. 2013, 76, 1530–1538. [Google Scholar] [CrossRef]
- Kroupitski, Y.; Pinto, R.; Brandl, M.T.; Belausov, E.; Sela, S. Interactions of Salmonella enterica with lettuce leaves. J. Appl. Microbiol. 2009, 106, 1876–1885. [Google Scholar] [CrossRef] [PubMed]
- Delbeke, S.; Ceuppens, S.; Jacxsens, L.; Uyttendaele, M. Survival of Salmonella and Escherichia coli O157:H7 on strawberries, basil, and other leafy greens during storage. J. Food Prot. 2015, 78, 652–660. [Google Scholar] [CrossRef]
- Pinton, S.C.; Bardsley, C.A.; Marik, C.M.; Boyer, R.R.; Strawn, L.K. Fate of Listeria monocytogenes on broccoli and cauliflower at different storage temperatures. J. Food Prot. 2020, 83, 858–864. [Google Scholar] [CrossRef]
- Ma, C.; Li, J.; Zhang, Q. Behavior of Salmonella spp. on fresh-cut tropical fruits. Food Microbiol. 2016, 54, 133–141. [Google Scholar] [CrossRef]
- Strawn, L.K.; Danyluk, M.D. Fate of Escherichia coli O157:H7 and Salmonella spp. on fresh and frozen cut mangoes and papayas. Int. J. Food Microbiol. 2010, 138, 78–84. [Google Scholar] [CrossRef]
- Montville, R.; Schaffner, D.W. Inoculum size influences bacterial cross contamination between surfaces. Appl. Environ. Microb. 2003, 69, 7188–7193. [Google Scholar] [CrossRef]
- Lo-Iacono-Ferreira, V.G.; Viñoles-Cebolla, R.; Bastante-Ceca, M.J.; Capuz-Rizo, S.F. Transport of Spanish fruit and vegetables in cardboard boxes: A carbon footprint analysis. J. Clean. Prod. 2020, 244, 118784. [Google Scholar] [CrossRef]
- Del Borghi, A.; Parodi, S.; Moreschi, L.; Gallo, M. Sustainable packaging: An evaluation of crates for food through a life cycle approach. Int. J. Life Cycle Assess. 2021, 26, 753–766. [Google Scholar] [CrossRef]
- Tua, C.; Biganzoli, L.; Grosso, M.; Rigamonti, L. Life cycle assessment of reusable plastic crates (RPCs). Resources 2019, 8, 110. [Google Scholar] [CrossRef]
| Sample Type | Sampling Time (Days) | ||||||
|---|---|---|---|---|---|---|---|
| 0 | 0.1 | 1 | 3 | 6 | 9 | 13 | |
| IC a before contact with PP pieces | X | ||||||
| IC after contact with PP pieces | X | X | X | X | X | X | X |
| PP pieces after contact with IC | X | ||||||
| PP pieces after contact with IC and non-IC | X | X | X | X | X | X | X |
| Non-IC before contact with PP pieces | X | ||||||
| Cross-contaminated cauliflower b | X | X | X | X | X | X | X |
| Polyethylene film from IC | X | X | X | X | X | X | |
| Polyethylene film from cross-contaminated cauliflower | X | X | X | X | X | X | |
| Type of Box | Materials | Weight (kg) |
|---|---|---|
| Plastic | Polypropylene | 1.550 |
| Cardboard | Corrugated Cardboard | 0.440 |
| Wooden | All | 1.586 |
| MDF | 1.032 | |
| Pinewood | 0.330 | |
| Poplar wood | 0.221 | |
| Stainless Steel | 0.003 |
| Type of Box | Phase | Mean Distance (km) | Reference |
|---|---|---|---|
| RPCs | Material–manufacturing plant | 1000 | [2] |
| RPCs | Manufacturing–packaging center | 500 | [2] |
| Cardboard boxes | Material–manufacturing plant | 467 | [33] |
| Cardboard boxes | Manufacturing–packaging center | 50 | [33] |
| Wooden boxes | Material–manufacturing plant | 400 | [34] |
| Wooden boxes | Manufacturing–packaging center | 100 | [34] |
| All | Packaging center–logistics platform | 400 | [2] |
| All | Logistics platform–retailer | 100 | [2] |
| All | Retailer–logistics platform | 100 | [2] |
| RPCs | Logistics platform–washing center | 100 | [2] |
| RPCs | Washing center–packaging center | 200 | [2] |
| RPCs | Washing center–End-of-life | 650 | [2] |
| Cardboard boxes | Logistics platform–End-of-life | 100 | [2] |
| Wooden boxes | Logistics platform–End-of-life | 100 | [35] |
| Sample Type | Sampling Time (Days) | Total (*) | ||||||
|---|---|---|---|---|---|---|---|---|
| 0 | 0.1 | 1 | 3 | 6 | 9 | 13 | ||
| Inoculated cauliflower | 3/3 | 3/3 | 3/3 | 3/3 | 3/3 | 3/3 | 3/3 | 21/21 (100%) |
| PP pieces | 3/3 | 3/3 | 3/3 | 3/3 | 3/3 | 2/3 | 1/3 | 18/21 (33%) |
| Cross-contaminated cauliflower | 3/3 | 2/3 | 3/3 | 3/3 | 2/3 | 2/3 | 2/3 | 17/21 (35%) |
| Polyethylene film from inoculated cauliflower | NA | 3/3 | 3/3 | 3/3 | 3/3 | 3/3 | 2/3 | 17/18 (71%) |
| Polyethylene film from cross-contaminated cauliflower | NA | 0/3 | 1/3 | 0/3 | 0/3 | 0/3 | 0/3 | 1/18 (0%) |
| Storage Time (days) | PP | Cardboard | Fiberboard | Cross-Contaminated Cauliflower (PP) | Cross-Contaminated Cauliflower (Cardboard) | Cross-Contaminated Cauliflower (Fiberboard) |
|---|---|---|---|---|---|---|
| 0 | 0.49 ± 0.71 (3/3) | 1.14 ± 0.83 (3/3) | 0.10 ± 0.40 (3/3) | <0.7 (3/3) | <0.7 (2/3) | <0.7 (2/3) |
| 1 | 0.40 ± 0.92 (3/3) | 0.71 ± 0.15 (3/3) | <−0.14 (3/3) | 0.85 ± 0.21 (3/3) | <0.7 (0/3) | <0.7 (0/3) |
| 6 | 0.20 ± 0.35 (3/3) | <−0.14 (3/3) | <−0.14 (3/3) | <0.7 (2/3) | <0.7 (0/3) | <0.7 (0/3) |
| 13 | <−0.14 (1/3) | <−0.14 (3/3) | <−0.14 (3/3) | 0.90 ± 0.17 (2/3) | <0.7 (0/3) | <0.7 (0/3) |
| Impact Category | Unit | Total | Plastic Crate Production | Cleaning | Transport | End-of-Life * |
|---|---|---|---|---|---|---|
| Global warming | kg CO2 eq | 6.02 × 10−3 | 73% | 22% | 5% | −25% |
| Ozone layer depletion | kg CFC-11 eq | 1.03 × 10−9 | 39% | 54% | 6% | 1% |
| Photochemical oxidation | kg C2H4 eq | 1.30 × 10−6 | 77% | 20% | 3% | −23% |
| Acidification | kg SO2 eq | 2.59 × 10−5 | 70% | 26% | 4% | −16% |
| Eutrophication | kg PO43− eq | 1.22 × 10−5 | 64% | 26% | 3% | 7% |
| Abiotic depletion | kg Sb eq | 7.82 × 10−8 | 65% | 26% | 9% | −20% |
| Fresh water aquatic ecotoxicity | kg 1,4-DB eq | 3.23 × 10−3 | 67% | 31% | 2% | −3% |
| Marine aquatic ecotoxicity | kg 1,4-DB eq | 7.27 × 100 | 69% | 30% | 2% | −2% |
| Terrestrial ecotoxicity | kg 1,4-DB eq | 1.55 × 10−5 | 48% | 49% | 3% | 0% |
| Human toxicity | kg 1,4-DB eq | 3.46 × 10−3 | 69% | 27% | 4% | −17% |
| Total energy (non-renewable and renewable) | MJ | 1.77 × 10−1 | 70% | 28% | 2% | −35% |
| Total non-renewable | MJ | 1.48 × 10−1 | 75% | 22% | 2% | −39% |
| Non-renewable, fossil fuels | MJ | 1.09 × 10−1 | 84% | 13% | 3% | −45% |
| Non-renewable, nuclear | MJ | 3.92 × 10−2 | 35% | 64% | 0% | −8% |
| Non-renewable, biomass | MJ | 3.68 × 10−6 | 64% | 32% | 4% | 4% |
| Total renewable | MJ | 1.80 × 10−2 | 39% | 61% | 0% | −3% |
| Renewable, biomass | MJ | 4.89 × 10−3 | 61% | 39% | 1% | −7% |
| Renewable, wind, solar, geothermal | MJ | 6.13 × 10−3 | 20% | 80% | 0% | −1% |
| Renewable, water | MJ | 7.00 × 10−3 | 40% | 59% | 1% | 0% |
| Impact Category | Unit | Total | Cardboard Box Production | Transport | End-of-Life * |
|---|---|---|---|---|---|
| Global warming | kg CO2 eq | 5.88 × 10−2 | 92% | 8% | −26% |
| Ozone layer depletion | kg CFC−11 eq | 7.54 × 10−9 | 88% | 12% | −24% |
| Photochemical oxidation | kg C2H4 eq | 1.22 × 10−5 | 95% | 5% | −36% |
| Acidification | kg SO2 eq | 2.59 × 10−4 | 92% | 8% | −23% |
| Eutrophication | kg PO43− eq | 9.62 × 10−5 | 97% | 3% | −50% |
| Abiotic depletion | kg Sb eq | 2.35 × 10−6 | 97% | 3% | −48% |
| Fresh water aquatic ecotoxicity | kg 1,4-DB eq | 5.56 × 10−2 | 99% | 1% | −49% |
| Marine aquatic ecotoxicity | kg 1,4-DB eq | 2.15 × 102 | 99% | 1% | −47% |
| Terrestrial ecotoxicity | kg 1,4-DB eq | 3.04 × 10−4 | 98% | 2% | −44% |
| Human toxicity | kg 1,4-DB eq | 8.29 × 10−1 | 97% | 3% | −47% |
| Total energy (non-renewable and renewable) | MJ | 5.53 × 10−1 | 94% | 6% | −71% |
| Total non-renewable | MJ | 9.32 × 10−1 | 92% | 8% | −26% |
| Non-renewable, fossil fuels | MJ | 8.29 × 10−1 | 91% | 9% | −26% |
| Non-renewable, nuclear | MJ | 1.02 × 10−1 | 99% | 1% | −26% |
| Non-renewable, biomass | MJ | 2.40 × 10−4 | 100% | 0% | −62% |
| Total renewable | MJ | 1.38 × 10−1 | 100% | 0% | −79% |
| Renewable, biomass | MJ | 1.08 × 10−1 | 100% | 0% | −82% |
| Renewable, wind, solar, geothermal | MJ | 7.73 × 10−3 | 99% | 1% | −45% |
| Renewable, water | MJ | 2.15 × 10−2 | 98% | 2% | −33% |
| Impact Category | Unit | Total | Wooden Box Production | Transport | End-of-Life | |||
|---|---|---|---|---|---|---|---|---|
| MDF | Pine | Poplar | Stainless Steel | |||||
| Global warming | kg CO2 eq | 1.86 × 10−1 | 51% | 9% | 3% | 1% | 14% | 23% |
| Ozone layer depletion | kg CFC−11 eq | 2.90 × 10−8 | 42% | 7% | 3% | 1% | 16% | 32% |
| Photochemical oxidation | kg C2H4 eq | 8.07 × 10−5 | 55% | 6% | 5% | 1% | 4% | 28% |
| Acidification | kg SO2 eq | 1.16 × 10−3 | 42% | 7% | 2% | 1% | 9% | 40% |
| Eutrophication | kg PO43− eq | 2.50 × 10−4 | 70% | 11% | 4% | 2% | 10% | 5% |
| Abiotic depletion | kg Sb eq | 2.47 × 10−5 | 89% | 2% | 1% | 0% | 2% | 5% |
| Fresh water aquatic ecotoxicity | kg 1,4-DB eq | 1.12 × 10−1 | 57% | 5% | 1% | 3% | 4% | 30% |
| Marine aquatic ecotoxicity | kg 1,4-DB eq | 2.53 × 102 | 45% | 5% | 1% | 5% | 3% | 40% |
| Terrestrial ecotoxicity | kg 1,4-DB eq | 4.80 × 10−4 | 67% | 11% | 7% | 2% | 8% | 6% |
| Human toxicity | kg 1,4-DB eq | 2.47 × 10−1 | 41% | 4% | 1% | 1% | 5% | 48% |
| Total energy (non-renewable and renewable) | MJ | 7.63 × 100 | 41% | 28% | 17% | 0% | 5% | 8% |
| Total non-renewable | MJ | 4.54 × 100 | 39% | 5% | 2% | 1% | 9% | 45% |
| Non-renewable, fossil fuels | MJ | 4.05 × 100 | 40% | 5% | 2% | 0% | 10% | 43% |
| Non-renewable, nuclear | MJ | 4.86 × 10−1 | 30% | 3% | 3% | 1% | 1% | 61% |
| Non-renewable, biomass | MJ | 1.35 × 10−4 | 49% | 22% | 26% | 0% | 2% | −65% |
| Total renewable | MJ | 3.09 × 100 | 31% | 43% | 26% | 0% | 0% | −31% |
| Renewable, biomass | MJ | 2.96 × 100 | 30% | 43% | 26% | 0% | 0% | −33% |
| Renewable, wind, solar, geothermal | MJ | 3.79 × 10−2 | 53% | 5% | 2% | 1% | 2% | 38% |
| Renewable, water | MJ | 9.87 × 10−2 | 38% | 8% | 10% | 1% | 3% | 41% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-Gálvez, F.; Rasines, L.; Conesa, E.; Gómez, P.A.; Artés-Hernández, F.; Aguayo, E. Reusable Plastic Crates (RPCs) for Fresh Produce (Case Study on Cauliflowers): Sustainable Packaging but Potential Salmonella Survival and Risk of Cross-Contamination. Foods 2021, 10, 1254. https://doi.org/10.3390/foods10061254
López-Gálvez F, Rasines L, Conesa E, Gómez PA, Artés-Hernández F, Aguayo E. Reusable Plastic Crates (RPCs) for Fresh Produce (Case Study on Cauliflowers): Sustainable Packaging but Potential Salmonella Survival and Risk of Cross-Contamination. Foods. 2021; 10(6):1254. https://doi.org/10.3390/foods10061254
Chicago/Turabian StyleLópez-Gálvez, Francisco, Laura Rasines, Encarnación Conesa, Perla A. Gómez, Francisco Artés-Hernández, and Encarna Aguayo. 2021. "Reusable Plastic Crates (RPCs) for Fresh Produce (Case Study on Cauliflowers): Sustainable Packaging but Potential Salmonella Survival and Risk of Cross-Contamination" Foods 10, no. 6: 1254. https://doi.org/10.3390/foods10061254
APA StyleLópez-Gálvez, F., Rasines, L., Conesa, E., Gómez, P. A., Artés-Hernández, F., & Aguayo, E. (2021). Reusable Plastic Crates (RPCs) for Fresh Produce (Case Study on Cauliflowers): Sustainable Packaging but Potential Salmonella Survival and Risk of Cross-Contamination. Foods, 10(6), 1254. https://doi.org/10.3390/foods10061254










