Development and Evaluation of an Ultrasonic Humidifier to Control Humidity in a Cold Storage Room for Postharvest Quality Management of Dates
Abstract
1. Introduction
2. Materials and Methods
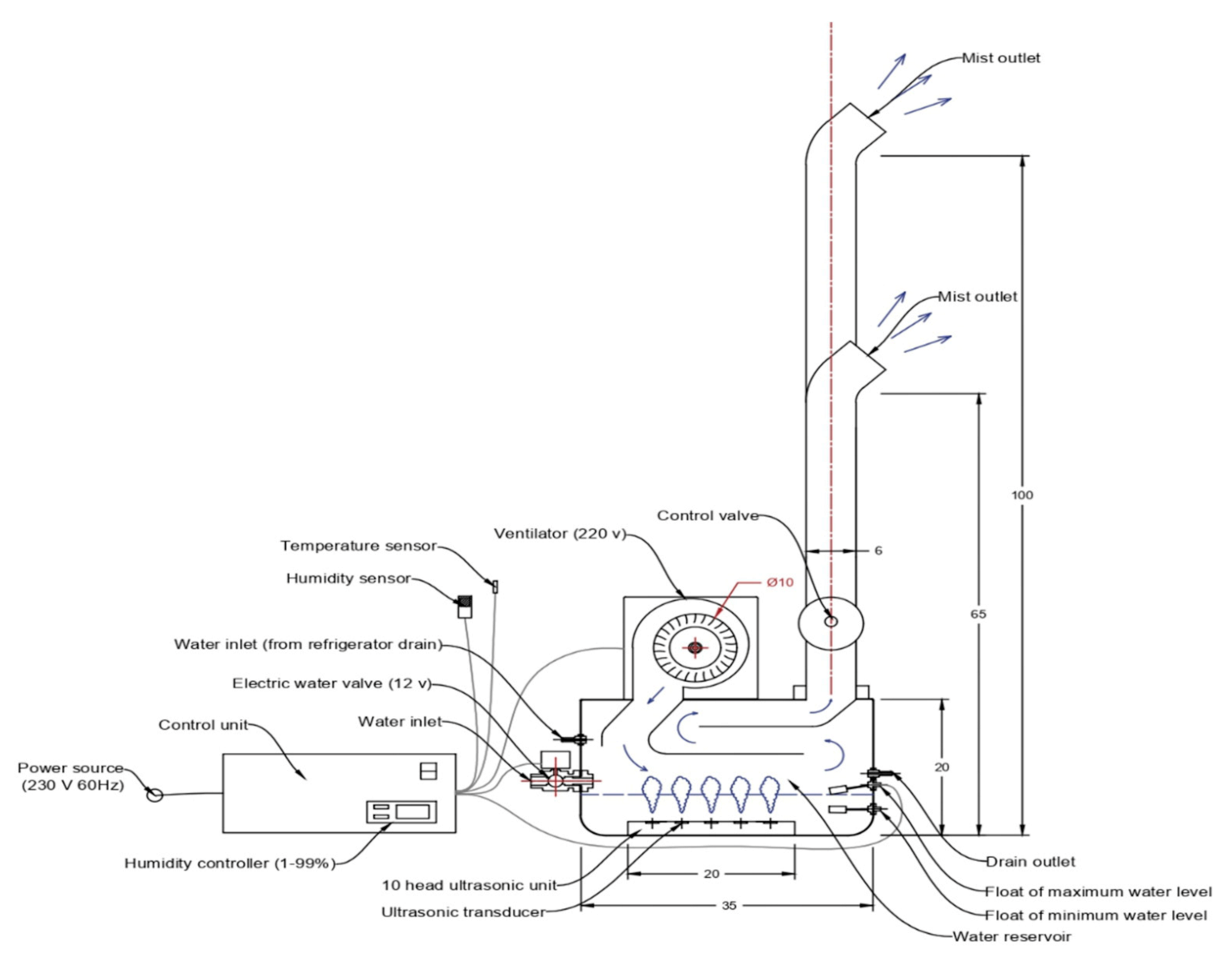
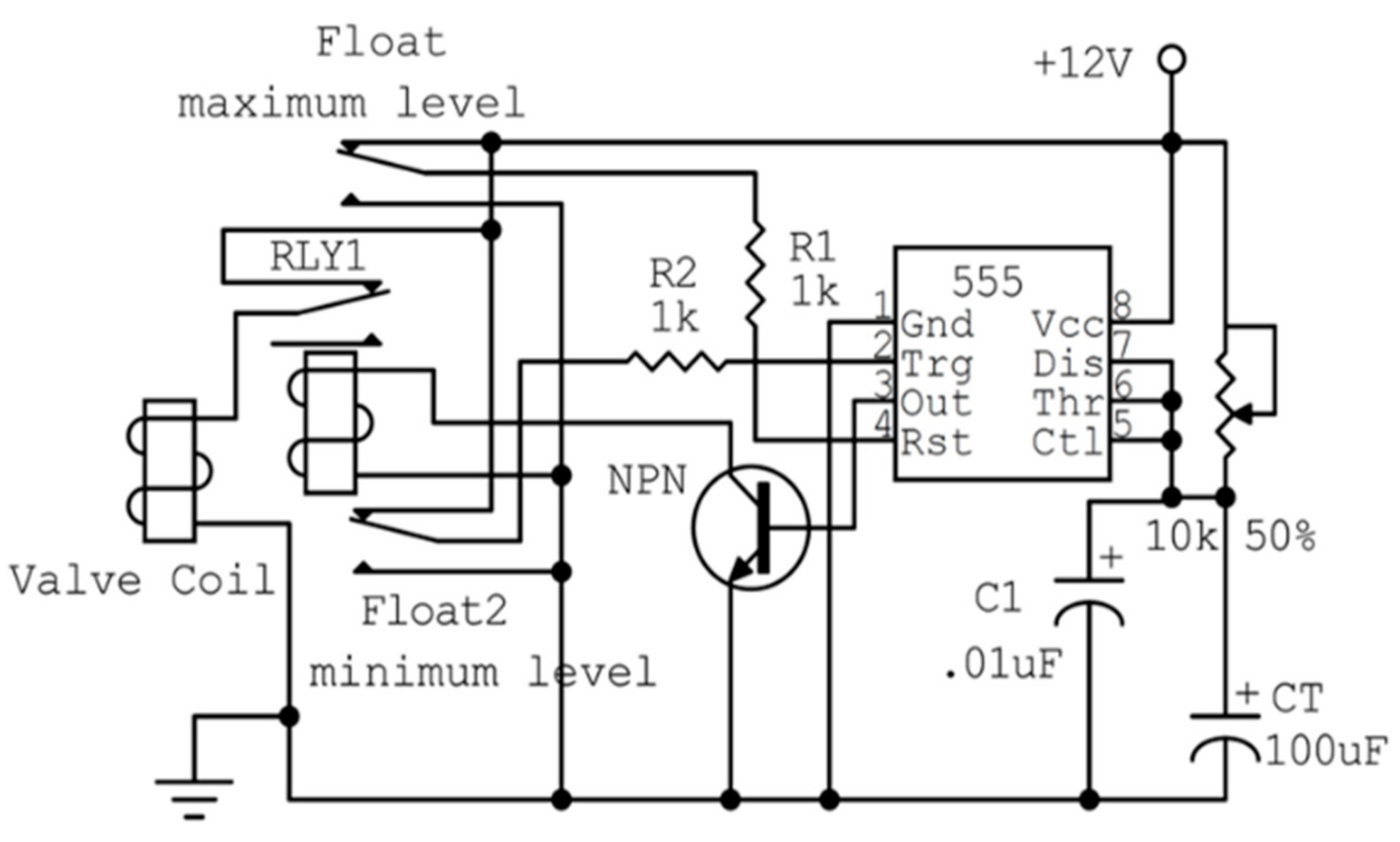
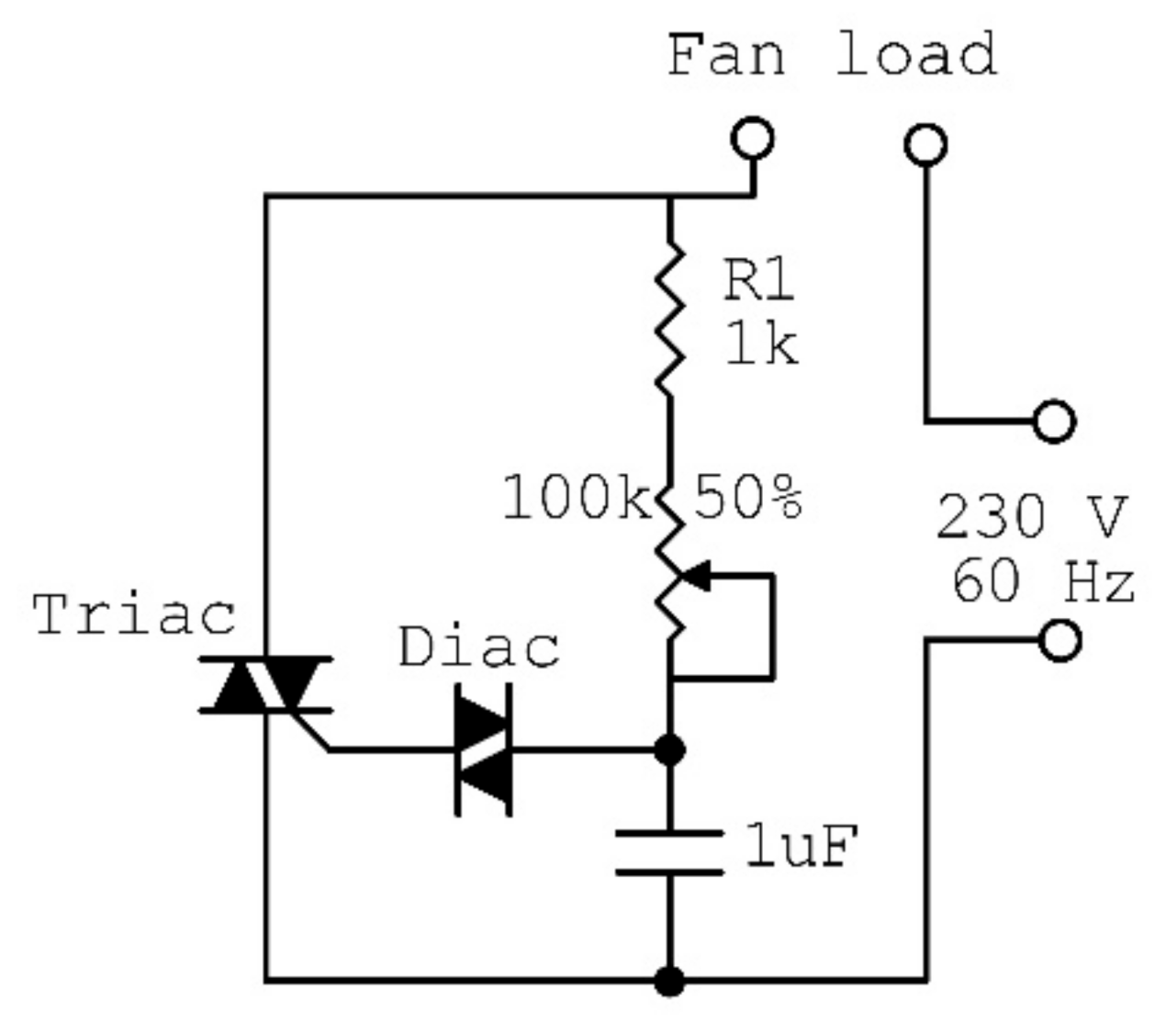
2.1. Description of the Designed Ultrasonic Humidifier
2.2. Experimental Setup
2.3. Tested Date Fruit
2.4. Measurements
2.4.1. Droplet Measurements
2.4.2. Temperature and RH
2.4.3. Air Velocity
2.4.4. Physicochemical Characteristics
- Moisture content
- Shape and size
- Fruit color
- Percentage of weight loss
- Total soluble solids
- Firmness
- Water activity
2.4.5. Microbiological Analysis
2.4.6. Test Insects
2.5. Statistical Analysis
3. Results
3.1. Humidifier Performance
3.2. Management of CSR Atmosphere
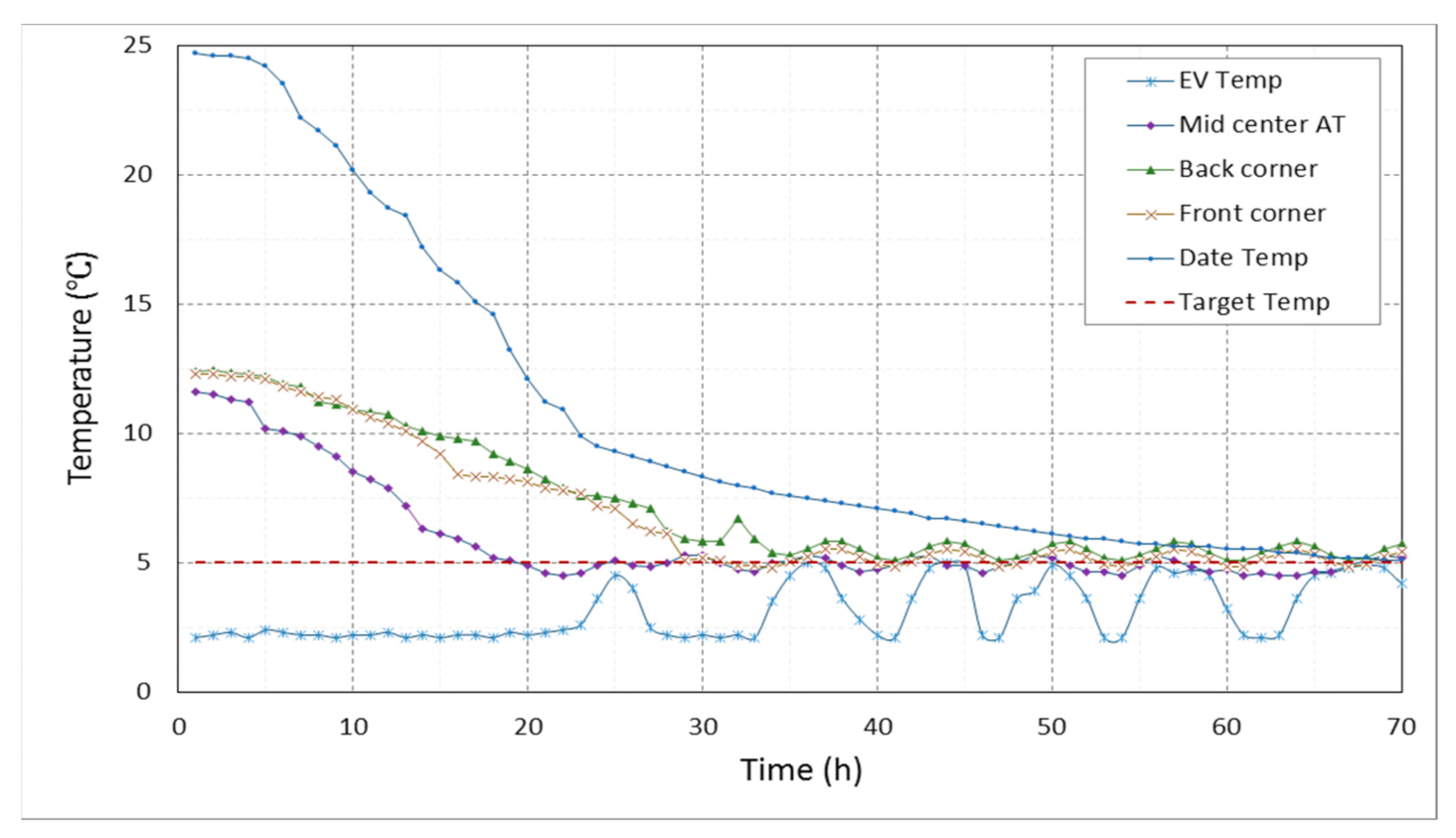
3.2.1. Temperature
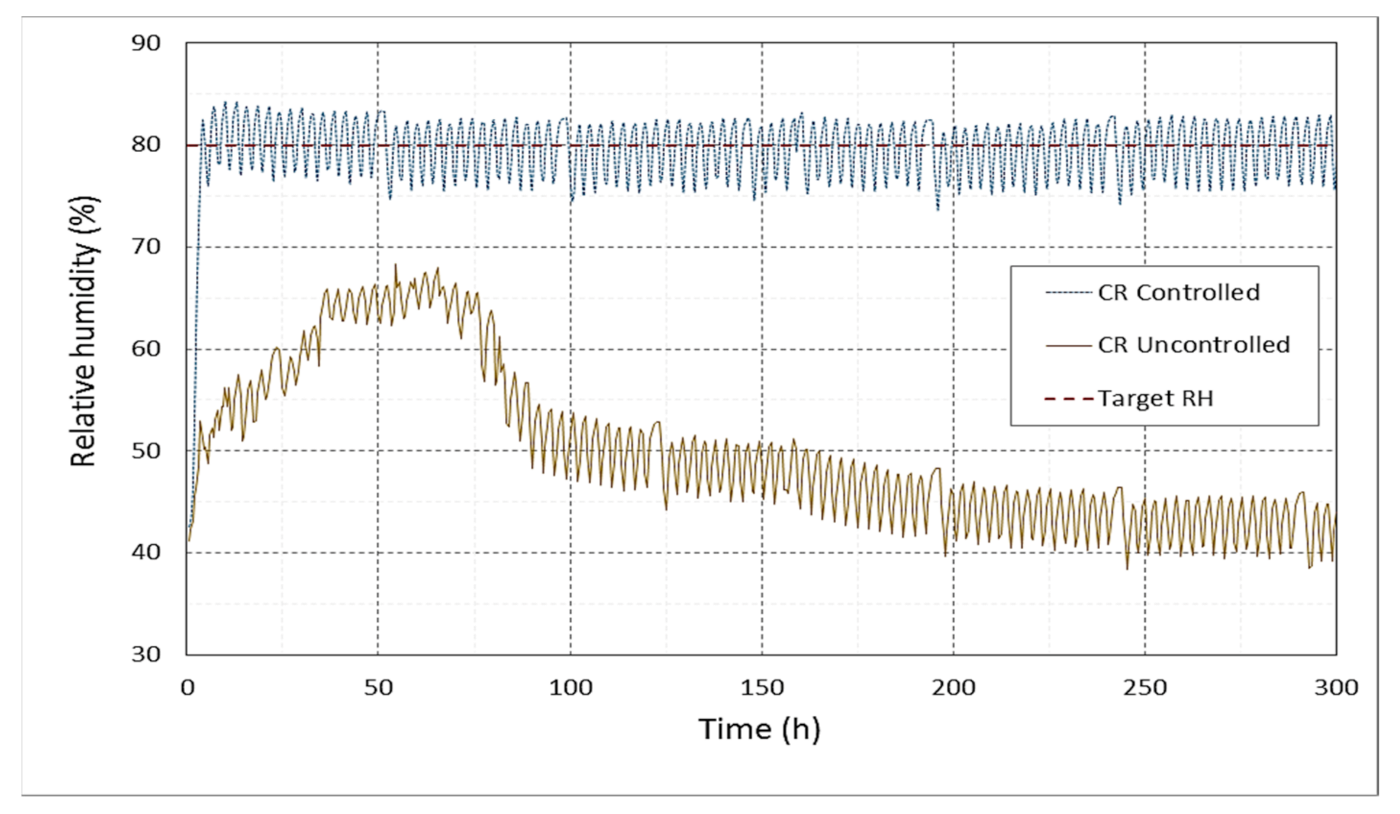
3.2.2. Relative Humidity (RH)
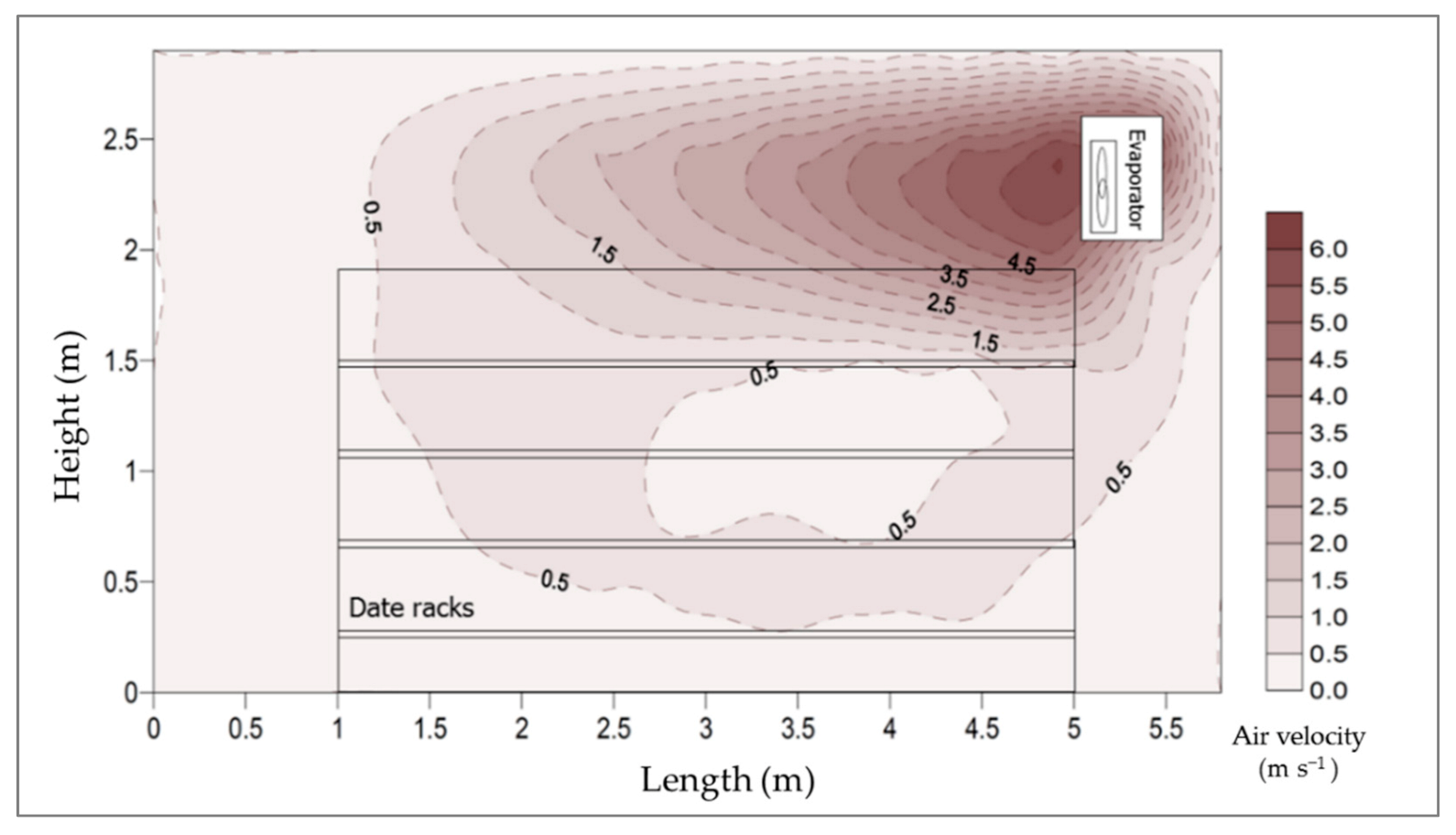
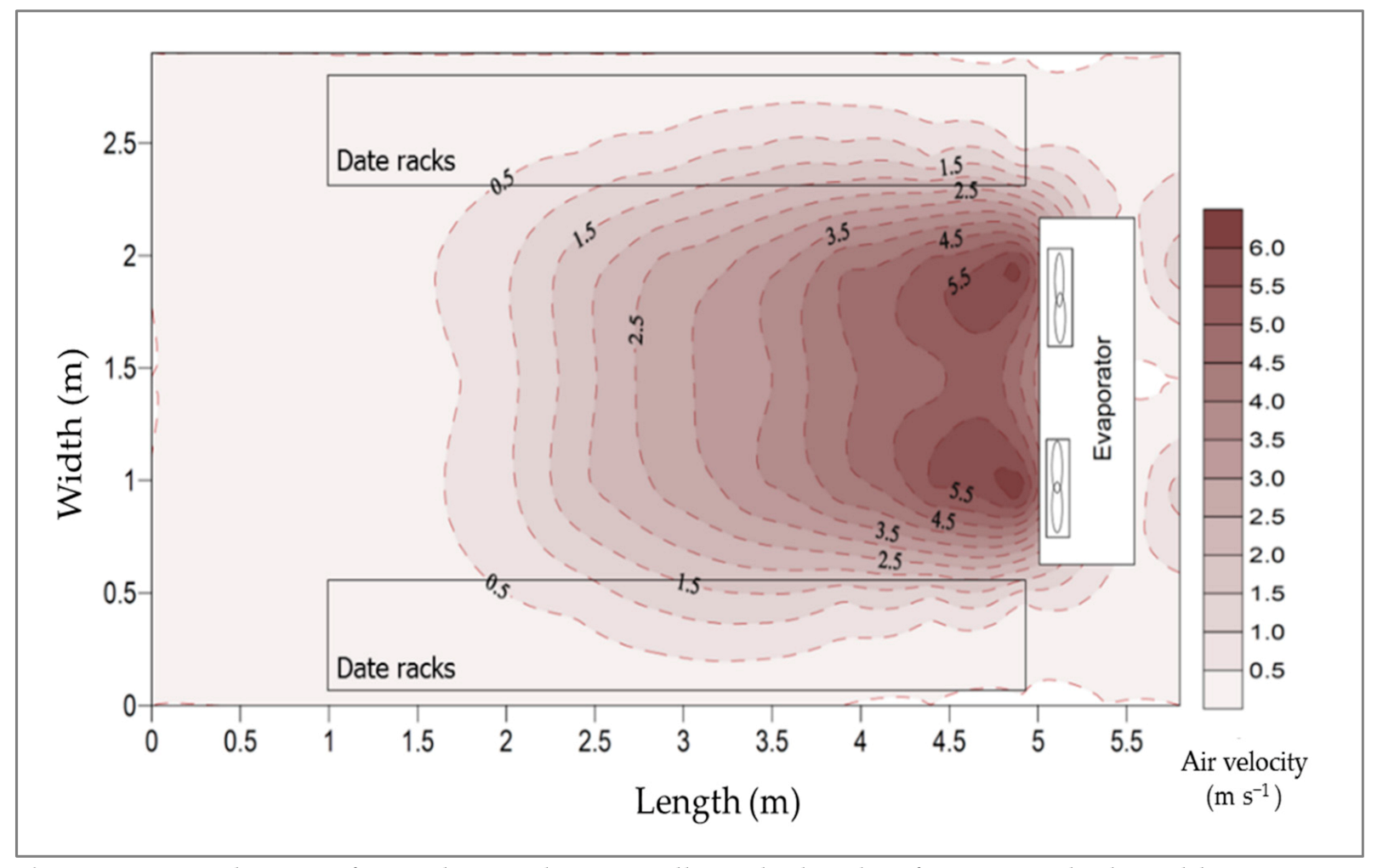
3.2.3. Air Velocity
3.3. Management of Physicochemical Characteristics of Stored Dates
3.3.1. Physicochemical Characteristics of the Tested Dates
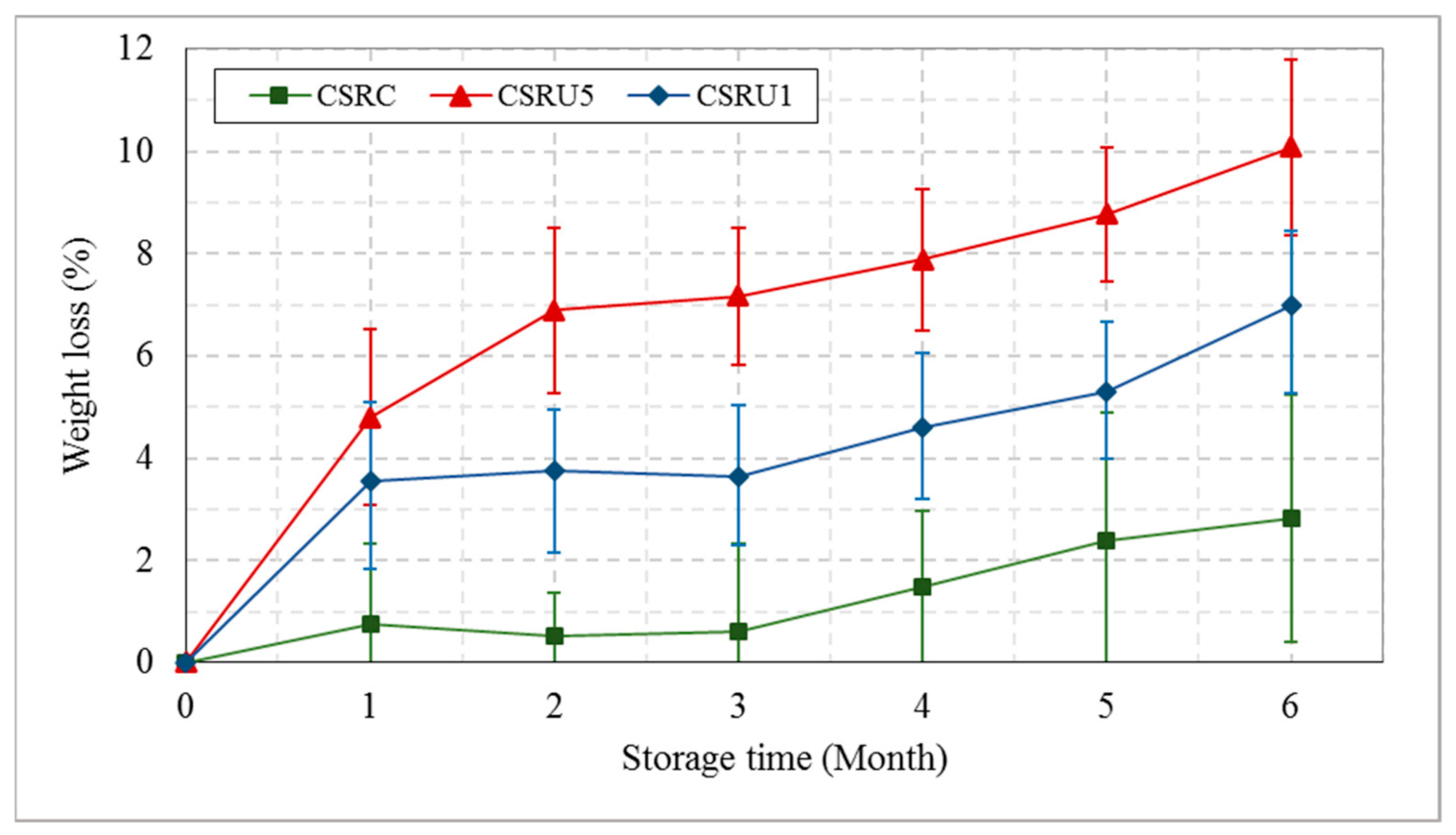
3.3.2. Effect of RH and Storage Time on Date Fruit Quality
3.4. Management of Microorganisms and Insects
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Systems Availability Statement
References
- Bodirsky, B.L.; Rolinski, S.; Biewald, A.; Weindl, I.; Popp, A.; Lotze-Campen, H. Global Food Demand Scenarios for the 21st Century. PLoS ONE 2015, 10, e0139201. [Google Scholar] [CrossRef] [PubMed]
- Anderson, W.; French, C.; Scrimshaw, N.S.; Macnaughton, J.W.; McNaughton, J.W. Meeting the Challenge of Feeding the World. Am. J. Public Health Nations Health 1959, 49, 1364–1372. [Google Scholar] [CrossRef] [PubMed]
- Gustavsson, J.; Cederberg, C.; Sonesson, U.; Van Otterdijk, R.; Meybeck, A. Global Food Losses and Food Waste; FAO: Düsseldorf, Germany, 2011. [Google Scholar]
- Elik, A.; Yanik, D.K.; Istanbullu, Y.; Guzelsoy, N.A.; Yavuz, A.; Gogus, F. Strategies to Reduce Post-Harvest Losses for Fruits and Vegetables. Int. J. Sci. Technol. Res. 2019, 5, 29–39. [Google Scholar] [CrossRef]
- Codex Standards (Codex Stan 143: 1985): Codex Standard for Dates. In Codex Alimentarius; Food and Agriculture Organization of the United Nations: Düsseldorf, Germany, 1985; Available online: http://www.fao.org (accessed on 5 April 2021).
- Adel, A.K.; Awad, M.H. Harvesting and Postharvest Handling of Pineapple. Icarda 2009, 15, 1–15. [Google Scholar]
- Mohammed, M.E.A.; Eissa, A.H.A.; Aleid, S.M. Application of Pulsed Electric Field for Microorganisms Inactivation in Date palm Fruits. J. Food Nutr. Res. 2016, 4, 646–652. [Google Scholar] [CrossRef]
- Arpaia, M.; Kader, A. Lemon: Recommendations for maintaining Postharvest quality. Precambrian Res. 2017, 294, 53–66. [Google Scholar]
- Aleid, S.M.; Hassan, B.H.; Almaiman, S.A.; Al-Kahtani, S.H.; Ismail, S.M. Microbial Loads and Physicochemical Characteristics of Fruits from Four Saudi Date Palm Tree Cultivars: Conformity with Applicable Date Standards. Food Nutr. Sci. 2014, 5, 316–327. [Google Scholar] [CrossRef][Green Version]
- Burks, C.S.; Yasin, M.; El-Shafie, H.A.F.; Wakil, W. Pests of Stored Dates. In Sustainable Pest Management in Date Palm: Current Status and Emerging Challenges; Springer: Dordrecht, The Netherlands, 2015; pp. 237–286. [Google Scholar]
- Mohammed, M.E.; El-Shafie, H.A.; Sallam, A.A. A solar-powered heat system for management of almond moth, Cadra cautella (Lepidoptera: Pyralidae) in stored dates. Postharvest Biol. Technol. 2019, 154, 121–128. [Google Scholar] [CrossRef]
- Mohammed, M.E.; El-Shafie, H.A.; Alhajhoj, M.R. Design and efficacy evaluation of a modern automated controlled atmosphere system for pest management in stored dates. J. Stored Prod. Res. 2020, 89, 101719. [Google Scholar] [CrossRef]
- El-Shafie, H. Arthropod pests of date palm and their management. CAB Rev. Perspect. Agric. Veter Sci. Nutr. Nat. Resour. 2017, 12, 1–18. [Google Scholar] [CrossRef]
- Husain, M.; Sukirno, S.; Mehmood, K.; Tufail, M.; Rasool, K.G.; Alwaneen, W.S.; Aldawood, A. Effectiveness of carbon dioxide against different developmental stages of Cadra cautella and Tribolium castaneum. Environ. Sci. Pollut. Res. 2017, 24, 12787–12795. [Google Scholar] [CrossRef]
- Mohammed, M.E.; Alhajhoj, M.R. Importance and Applications of Ultrasonic Technology to Improve Food Quality. In Food Processing; IntechOpen: London, UK, 2020. [Google Scholar]
- Mohammed, M.E.A.; Amer Eiss, A.H. Pulsed Electric Fields for Food Processing Technology. In Structure and Function of Food Engineering; IntechOpen: London, UK, 2012. [Google Scholar]
- Tang, J.; Mitcham, E.; Wang, S.; Lurie, S. Heat Treatment for Postharvest Pest Control. CAB Int. 2007, 1, 1–366. [Google Scholar]
- Mitchell, F.; Crisosto, C. The Use Cooling and Cold Storage to Stabilize and Preserve Fresh Stone Fruits; IRTA: Leida, Spain, 1995. [Google Scholar]
- Palipane, K.B. Good Practice for Assuring the Post-Harvest Quality of Exotic Tree Fruit Crops Produced in Jamaica—A Technical Guide; Food and Agriculture Organization of The United Nations: Rome, Italy, 2008. [Google Scholar]
- Ahmad, S.A.H.; Ab Rahman, M.N.; Muhamed, A.A. Optimal temperature in cold storage for perishable foods. In Proceedings of the 5th NA International Conference on Industrial Engineering and Operations Management, Detroit, MI, USA, 10–14 August 2020. [Google Scholar]
- Akdemir, S. Designing of Cold Stores and Choosing of Cooling System Elements. J. Appl. Sci. 2008, 8, 788–794. [Google Scholar] [CrossRef]
- Abdul Aly, S.A. Study of the Effect of Storage Temperature on Microbial Stored Dates under Vacuum. In Proceedings of the the Sixth International Date Palm Conference (SIDPC), Abu Dhabi, UAE, 19–21 March 2018. [Google Scholar]
- Akdemir, S.; Bal, E. Quality Changes in Apple in Evaporative Cooling Store. Erwerbs-Obstbau 2019, 62, 61–67. [Google Scholar] [CrossRef]
- Brown, T.; Corry, J.E.; Evans, J.A. Humidification of unwrapped chilled meat on retail display using an ultrasonic fogging system. Meat Sci. 2007, 77, 670–677. [Google Scholar] [CrossRef]
- Brown, T.; Corry, J.E.; James, S.J. Humidification of chilled fruit and vegetables on retail display using an ultrasonic fogging system with water/air ozonation. Int. J. Refrig. 2004, 27, 862–868. [Google Scholar] [CrossRef]
- Mohd-Som, F.; Spomer, L.A.; Martin, S.E.; Schmidt, S.J. Microflora Changes in Misted and Nonmisted Broccoli at Refrigerated Storage Temperatures. J. Food Qual. 1995, 18, 279–293. [Google Scholar] [CrossRef]
- Bittner, B.; Kissel, T. Ultrasonic atomization for spray drying: A versatile technique for the preparation of protein loaded biodegradable microspheres. J. Microencapsul. 1999, 16, 325–341. [Google Scholar] [CrossRef]
- Putra, I.D.G.A.T.; Sunu, P.W.; Temaja, I.W.; Sugiartha, N.; Sugina, I.M.; Suirya, I.W. Investigation on application of ultrasonic humidifier for air conditioning system. J. Phys. Conf. Ser. 2020, 1450, 012050. [Google Scholar] [CrossRef]
- Yao, Y.; Pan, Y.; Liu, S. Power ultrasound and its applications: A state-of-the-art review. Ultrason. Sonochemistry 2020, 62, 104722. [Google Scholar] [CrossRef]
- Al-Jumaily, A.M.; Meshkinzar, A. On the Development of Focused Ultrasound Liquid Atomizers. Adv. Acoust. Vib. 2017, 2017, 7861726. [Google Scholar] [CrossRef]
- Ramisetty, K.A.; Pandit, A.B.; Gogate, P.R. Investigations into ultrasound induced atomization. Ultrason. Sonochem. 2013, 20, 254–264. [Google Scholar] [CrossRef]
- Barreras, F.; Amaveda, H.; Lozano, A. Transient high-frequency ultrasonic water atomization. Exp. Fluids 2002, 33, 405–413. [Google Scholar] [CrossRef]
- Dalmoro, A.; D’Amore, M.; Barba, A.A. Droplet size prediction in the production of drug delivery microsystems by ultrasonic atomization. Transl. Med. UniSa 2019, 7, 6–11. [Google Scholar]
- Yeo, L.Y.; Friend, J.R.; McIntosh, M.P.; Meeusen, E.N.T.; Morton, D.A.V. Ultrasonic nebulization platforms for pulmonary drug delivery. Expert Opin. Drug Deliv. 2010, 7, 663–679. [Google Scholar] [CrossRef]
- Phanphanit, P. Experimental and Computational Study of an Ultrasonic Atomizer; University of Manchester: Manchester, UK, 2011. [Google Scholar]
- Donnelly, T.D.; Hogan, J.; Mugler, A.; Schommer, N.; Schubmehl, M.; Bernoff, A.J.; Forrest, B. An experimental study of micron-scale droplet aerosols produced via ultrasonic atomization. Phys. Fluids 2004, 16, 2843–2851. [Google Scholar] [CrossRef]
- Rahman, M.S. Data and Models of Density, Shrinkage, and Porosity. In Food Properties Handbook; CRC Press: Boca Raton, FL, USA, 2009; pp. 433–516. [Google Scholar] [CrossRef]
- Eissa, A.H.A.; Gamea, G.R.; Aboamera, M.A.; Mohmed, M.E. Machine vision prototype for orange grading using phototransistor. In Proceedings of the Post-Harvest, Food and Process Engineering, International Conference of Agricultural Engineering-CIGR-AgEng 2012: Agriculture and Engineering for a Healthier Life, Valencia, Spain, 8–12 July 2012. [Google Scholar]
- Baines, C.R.; Mohsenin, N.N. Thermal Properties of Foods and Agricultural Materials. Biometrics 1982, 38, 287. [Google Scholar] [CrossRef]
- Gamea, G.R.; Aboamera, M.A.; Mohmed, M.E.; Mohammed, M. Design and Manufacturing of Prototype for Orange Grading Using Phototransistor. Misr J. Agric. Eng. 2011, 28, 505–523. [Google Scholar] [CrossRef]
- Mditshwa, A.; Fawole, O.A.; Opara, U.L. Recent developments on dynamic controlled atmosphere storage of apples—A review. Food Packag. Shelf Life 2018, 16, 59–68. [Google Scholar] [CrossRef]
- Suzannah, S.; Hii, C.L.; Maznah, I. Processing and Drying of Foods, Vegetables and Fruits. In Proceedings of the International Symposia on Processing and Drying of Foods, Vegetables and Fruits (ISPDFVF), Kuala Lumpur, Malaysia, 18–19 June 2012. [Google Scholar]
- Ahmed, J.; Rahman, M.S. Handbook of Food Process Design; John Wiley & Sons: Hoboken, NJ, USA, 2012; ISBN 144433011X. [Google Scholar]
- Chukwu, O. Moisture-sorption study of dried date fruits. AU J. Technol. 2010, 13, 175–180. [Google Scholar]
- Bajpai, S.K.; Tiwari, P. Investigation of moisture sorption behavior of soluble sodium caseinate. Emir. J. Food Agric. 2014, 26, 399. [Google Scholar] [CrossRef]
- De La Cruz, G.; Torres, J.; Martín-Polo, M. Temperature effect on the moisture sorption isotherms for methylcellulose and ethylcellulose films. J. Food Eng. 2001, 48, 91–94. [Google Scholar] [CrossRef]
- Moitte, S.F.A.; Heikal, Y.A. Sorption isotherms behaviour of some Egyptian date fruit varieties. World J. Dairy Food Sci. 2013, 8, 74–81. [Google Scholar]
- Rajan, R.; Pandit, A. Correlations to predict droplet size in ultrasonic atomisation. Ultrasonics 2001, 39, 235–255. [Google Scholar] [CrossRef]
- Yasuda, K.; Honma, H.; Xu, Z.; Asakura, Y.; Koda, S. Ultrasonic atomization amount for different frequencies. Jpn. J. Appl. Phys. 2011, 50, 07HE23. [Google Scholar] [CrossRef]
- AOAC Official Methods of Analysis of AOAC International, 16th ed.; Association of Official Analytical Chemists: Rockville, MD, USA, 1998.
- Aleid, S.; Elansari, A.; Zhen-Xing, T.; Sallam, A. Effect of Cold Storage and Packing Type on Khalas and Sukkary Dates Quality. Adv. J. Food Sci. Technol. 2014, 6, 603–608. [Google Scholar] [CrossRef]
- Aldawood, A.S.; Rasool, K.G.; Alrukban, A.H.; Soffan, A.; Husain, M.; Sutanto, K.D.; Tufail, M. Effects of temperature on the development of ephestia cautella (walker) (pyralidae: Lepidoptera): A case study for its possible control under storage conditions. Pak. J. Zool. 2013, 45, 1573–1578. [Google Scholar]
- Brizio, P.J. Development of a Novel Humidifier for Air Breathing Devices; Auckland University of Technology: Auckland, New Zealand, 2011. [Google Scholar]
- Hadawey, A.; Tassou, S.A.; Chaer, I.; Sundararajan, R. Unwrapped food product display shelf life assessment. Energy Procedia 2017, 123, 62–69. [Google Scholar] [CrossRef]
- Benkeblia, N. Storage Technologies for Date Palm Fruits (Phoenix dazctylifera L.): Current and Potential Techniques. Acta Hortic. 2013, 994, 151–154. [Google Scholar] [CrossRef]
- Chemat, F.; Khan, M.K. Applications of ultrasound in food technology: Processing, preservation and extraction. Ultrason. Sonochem. 2011, 18, 813–835. [Google Scholar] [CrossRef]
- Bermudez-Aguirre, D. Ultrasound: Advances in Food Processing and Preservation; Academic Press: Cambridge, MA, USA; Elsevier: San Diego, CA, USA, 2017. [Google Scholar]
- Al-Dosary, N.H. Role of the Saw-toothed grain beetle (Oryzaephilus surinamensis L.) (Coleoptera: Silvanidae) in date palm fruit decay at different temperatures. Basrah J. Date Palm Res. 2009, 8, 1–14. [Google Scholar]




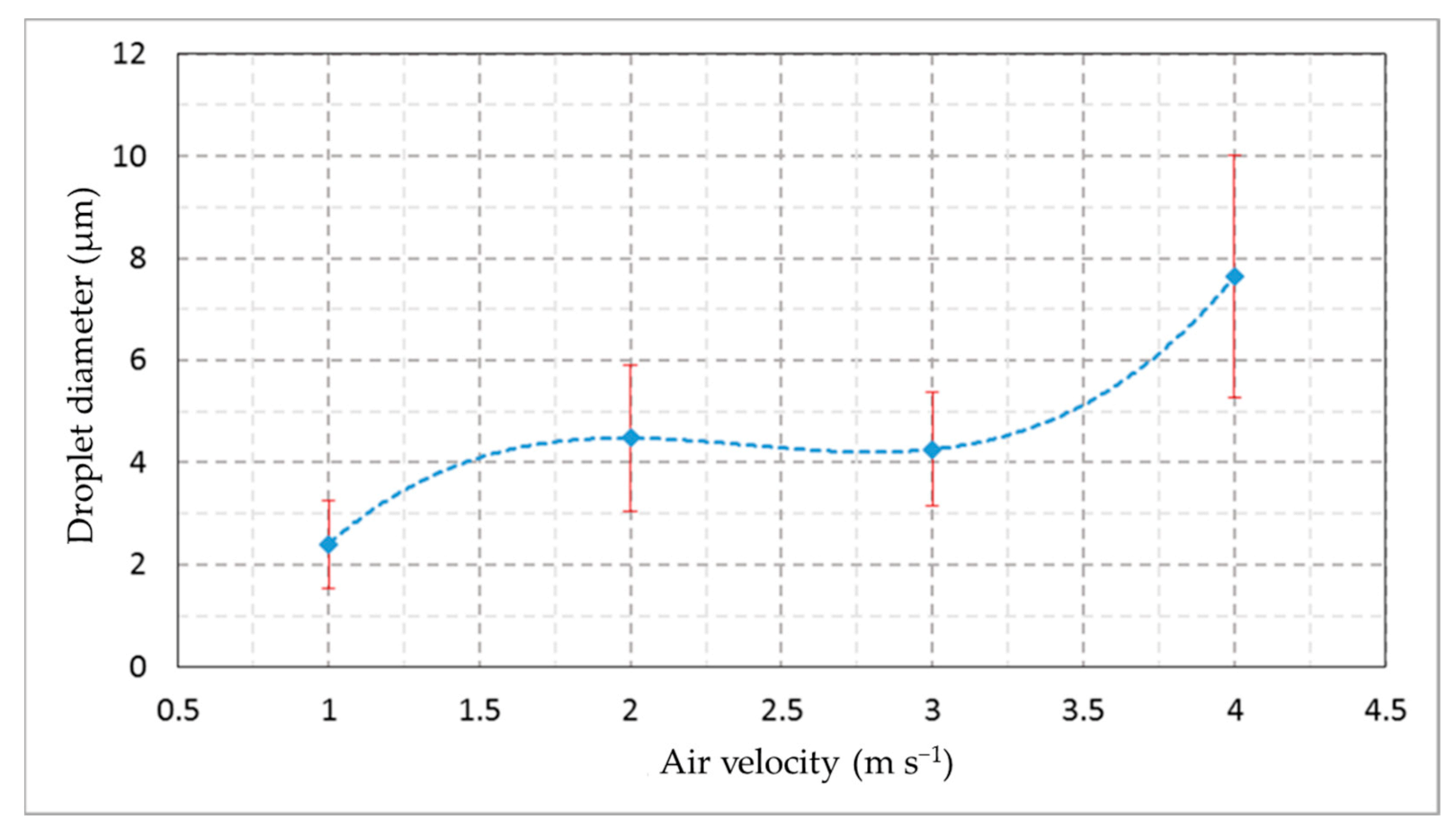
| Flight Parameter | Air Velocity (m s−1) | |||
|---|---|---|---|---|
| 1 | 2 | 3 | 4 | |
| Projected Area (µm2) | 4.61 ± 3.9 c | 17.05 ± 10.59 B | 15.46 ± 8.09 B | 32.67 ± 1.7 A |
| Value | Characteristic | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lf | Df | Sf | Arf | Ff | Mf | MC | TSS | L | a | b | h | C | ||
| Mean | 37.52 | 25.72 | 72.35 | 65.03 | 1.895 | 0.76 | 8.66 | 19.05 | 70.44 | 36.03 | 20.58 | 26.87 | 52.51 | 33.99 |
| St. Dev. | 1.92 | 2.47 | 4.61 | 5.40 | 0.22 | 0.07 | 0.71 | 1.24 | 1.08 | 4.94 | 4.22 | 5.30 | 4.97 | 6.03 |
| Storage Conditions | Storage Time | Characteristic | ||||
|---|---|---|---|---|---|---|
| MC, % | aw | L | ΔE | Mf | ||
| CSRC | C | 19.33 ± 0.6 A | 0.77 ± 0.08 A | 40.2 ± 2.6 A | 0 H | 8.85 ± 0.73 A |
| 1 | 19.15 ± 1.9 A | 0.78 ± 0.08 A | 39.6 ± 2.7 A | 1.09 ± 0.2 G–H | 8.83 ± 0.73 A | |
| 2 | 19.43 ± 1.0 A | 0.76 ± 0.05 AB | 38.9 ± 1.6 AB | 4.35 ± 3.6 E–H | 8.81 ± 0.75 A | |
| 3 | 19.26 ± 0.8 A | 0.78 ± 0.07 A | 36.8 ± 3.0 A–C | 6.37 ± 3.7 E–F | 8.82 ± 0.74 A | |
| 4 | 19.18 ± 1.0 A | 0.76 ± 0.07 AB | 35.4 ± 3.3 A–D | 13.3 ± 2.4 AC | 8.71 ± 0.62 A | |
| 5 | 18.89 ± 0.7 A | 0.78 ± 0.09 A | 31.2 ± 3.0 D–F | 14.5 ± 4.9 AB | 8.65 ± 0.80 A | |
| 6 | 18.86 ± 0.6 A | 0.76 ± 0.07 AB | 30.2 ±2.3 EF | 14.9 ± 4.4 AB | 8.59 ± 0.67 A | |
| CSRU5 | C | 19.20 ± 0.6 A | 0.77 ± 0.05 A | 40.1 ± 2.6 A | 0 H | 8.79 ± 0.72 A |
| 1 | 15.22 ± 2.4 BC | 0.73 ± 0.06 A–C | 37.8 ± 2.6 A–C | 4.85 ± 3.6 E–H | 8.36 ± 0.68 A | |
| 2 | 14.48 ± 1.6 BC | 0.72 ± 0.07 A–C | 37.3 ± 2.3 A–C | 5.94 ± 3.8 E–G | 8.17 ± 0.59 AB | |
| 3 | 13.56 ± 2.0 B–D | 0.73 ± 0.06 A–C | 33.7 ± 3.9 B–E | 8.90 ± 3.2 C–E | 8.15 ± 0.56 AB | |
| 4 | 13.18 ± 2.3 CD | 0.63 ± 0.12 C | 31.1 ± 5.0 D–F | 12.4 ± 4.2 A–C | 8.12 ± 0.61 B | |
| 5 | 11.45 ± 1.4 D–E | 0.65 ± 0.12 BC | 29.7 ± 4.3 EF | 14.8 ± 4.9 AB | 8.01 ± 0.66 BC | |
| 6 | 10.30 ± 0.8 E | 0.63 ± 0.09 C | 28.4 ± 3.0 F | 17.6 ± 5.1 A | 7.83 ± 0.63 C | |
| CSRU1 | C | 19.06 ± 0.6 A | 0.77 ± 0.04 A | 39.8 ± 2.6 A | 0 H | 8.77 ± 0.72 A |
| 1 | 15.69 ± 2.4 BC | 0.78 ± 0.08 A | 38.7 ± 3.0 A–C | 3.65 ± 3.0 F–H | 8.41 ± 0.72 A | |
| 2 | 15.88 ± 1.6 B | 0.78 ± 0.03 A | 35.9 ± 2.5 A–D | 5.66 ± 2.3 E–G | 8.39 ± 0.65 A | |
| 3 | 15.32 ± 2.2 BC | 0.79 ± 0.06 A | 33.6 ± 4.1 C–F | 7.91 ± 2.4 D–F | 8.40 ± 0.62 A | |
| 4 | 14.67 ± 2.3 BC | 0.79 ± 0.05 A | 33.5 ± 3.7 C–F | 8.81 ± 2.8 C–F | 8.37 ± 0.62 A | |
| 5 | 13.49 ± 2.0 B–D | 0.79 ± 0.05 A | 30.0 ± 3.1EF | 12.0 ± 1.3 B–D | 8.26 ± 0.61 AB | |
| 6 | 11.91 ± 1.2 D–E | 0.80 ± 0.02 A | 29.3 ± 4.8 EF | 13.5 ± 2.5 A– C | 8.11 ± 0.66 B | |
| Storage Conditions | Storage Time | Microorganisms | ||
|---|---|---|---|---|
| Total Bacteria, cfu g−1 | Molds and Yeasts, cfu g−1 | Coliforms, cfu g−1 | ||
| CSRC | C | 5.40 × 102 ± 1.71 × 102 A | 0.78 × 102 ± 0.25 × 102 G | n.d |
| 1 | 5.17 × 102 ± 2.44 × 102 A | 1.31 × 102 ± 0.54 × 102 E–G | n.d. | |
| 2 | 5.49 × 102 ± 1.82 × 102 A | 1.86 × 102 ± 0.66 × 102 C–G | n.d. | |
| 3 | 5.85 × 102 ± 1.88 × 102 A | 2.37 × 102 ± 0.92 × 102 B–F | n.d. | |
| 4 | 5.68 × 102 ± 2.41 × 102 A | 2.84 × 102 ± 1.03 × 102 BC | n.d. | |
| 5 | 5.92 × 102 ± 2.06 × 102 A | 3.59 × 102 ± 1.42 × 102 AB | n.d. | |
| 6 | 5.48 × 102 ± 2.45 × 102 A | 4.20 × 102 ± 1.63 × 102 A | n.d. | |
| CSRU5 | C | 5.40 × 102 ± 1.71 × 102 A | 0.78 × 102 ± 0.26 × 102 G | n.d. |
| 1 | 5.34 × 102 ± 2.41 × 102 A | 1.11 × 102 ± 0.50 × 102 FG | n.d. | |
| 2 | 5.48 × 102 ± 2.59 × 102 A | 1.47 × 102 ± 0.72 × 102 D–G | n.d. | |
| 3 | 5.55 × 102 ± 1.84 × 102 A | 1.84 × 102 ± 0.68 × 102 C–G | n.d. | |
| 4 | 5.52 × 102 ± 2.34 × 102 A | 2.59 × 102 ± 1.04 × 102 B–E | n.d. | |
| 5 | 5.49 × 102 ± 2.44 × 102 A | 2.79 × 102 ± 1.15 × 102 B–D | n.d. | |
| 6 | 5.54 × 102 ± 2.36 × 102 A | 2.80 × 102 ± 1.31 × 102 B–D | n.d. | |
| CSRU1 | C | 5.40 × 102 ± 1.71 × 102 A | 0.78 × 102 ± 0.26 × 102 G | n.d. |
| 1 | 5.36 × 102 ± 2.45 × 102 A | 0.81 × 102 ± 0.53 × 102 G | n.d. | |
| 2 | 5.47 × 102 ± 2.63 × 102 A | 0.82 × 102 ± 0.46 × 102 G | n.d. | |
| 3 | 5.49 × 102 ± 1.86 × 102 A | 0.89 × 102 ± 0.33 × 102 G | n.d. | |
| 4 | 5.42 × 102 ± 2.67 × 102 A | 0.88 × 102 ± 0.56 × 102 G | n.d. | |
| 5 | 5.43 × 102 ± 2.92 × 102 A | 1.06 × 102 ± 0.56 × 102 FG | n.d | |
| 6 | 5.49 × 102 ± 2.31 × 102 A | 1.05 × 102 ± 0.49 × 102 FG | n.d | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohammed, M.; Alqahtani, N.; El-Shafie, H. Development and Evaluation of an Ultrasonic Humidifier to Control Humidity in a Cold Storage Room for Postharvest Quality Management of Dates. Foods 2021, 10, 949. https://doi.org/10.3390/foods10050949
Mohammed M, Alqahtani N, El-Shafie H. Development and Evaluation of an Ultrasonic Humidifier to Control Humidity in a Cold Storage Room for Postharvest Quality Management of Dates. Foods. 2021; 10(5):949. https://doi.org/10.3390/foods10050949
Chicago/Turabian StyleMohammed, Maged, Nashi Alqahtani, and Hamadttu El-Shafie. 2021. "Development and Evaluation of an Ultrasonic Humidifier to Control Humidity in a Cold Storage Room for Postharvest Quality Management of Dates" Foods 10, no. 5: 949. https://doi.org/10.3390/foods10050949
APA StyleMohammed, M., Alqahtani, N., & El-Shafie, H. (2021). Development and Evaluation of an Ultrasonic Humidifier to Control Humidity in a Cold Storage Room for Postharvest Quality Management of Dates. Foods, 10(5), 949. https://doi.org/10.3390/foods10050949








