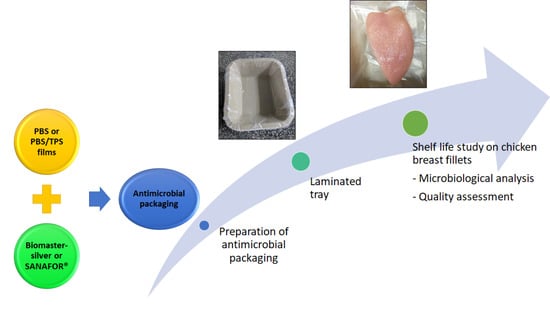
Efficacy of Biopolymer/Starch Based Antimicrobial Packaging for Chicken Breast Fillets
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Fabrication of PBS and PBS/TPS Films
2.3. Sampling and Storage
2.4. Microbiological Analysis
2.5. Quality Assessment
2.5.1. pH Analysis
2.5.2. Weight Loss
2.5.3. Texture Analysis
2.5.4. Color Analysis
2.5.5. Overall Visual Quality
2.6. Statistical Analysis
3. Results
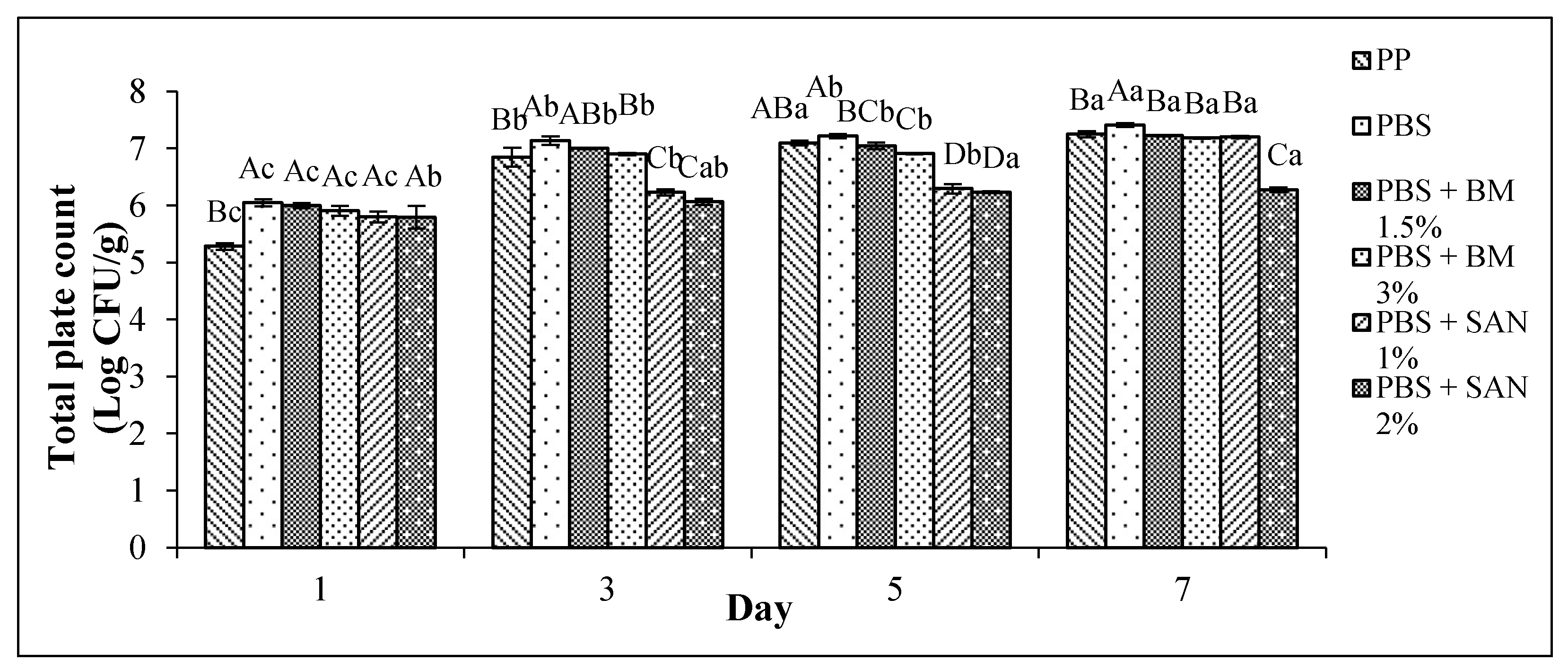
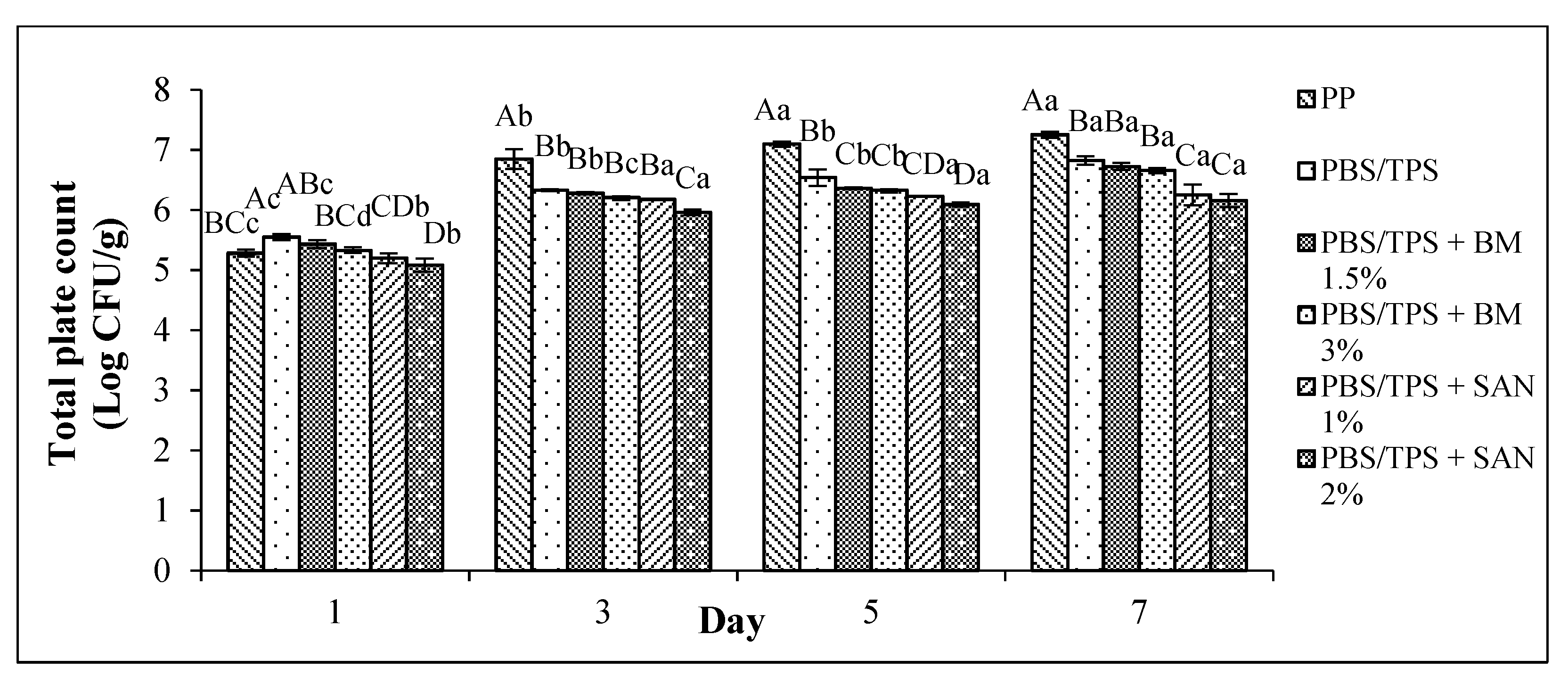
3.1. Total Plate Count (TPC)
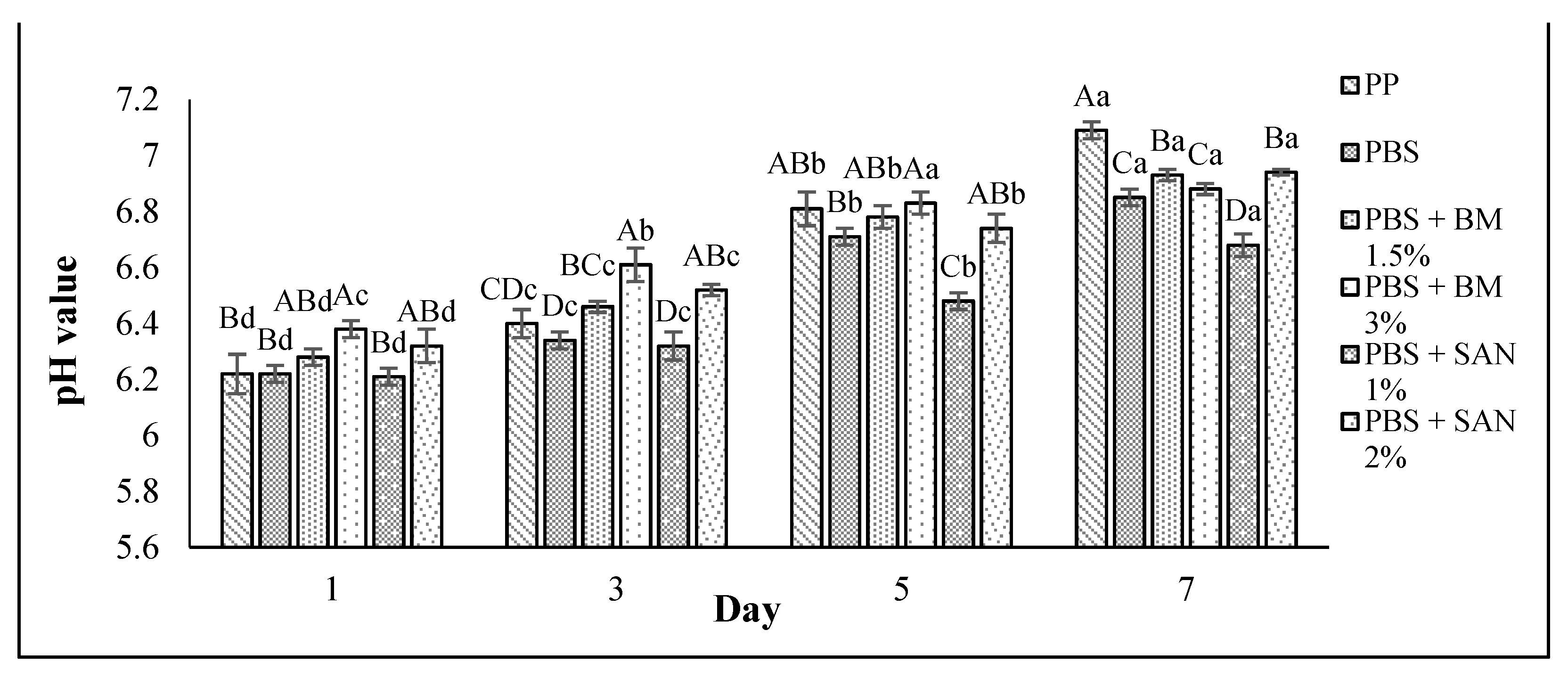
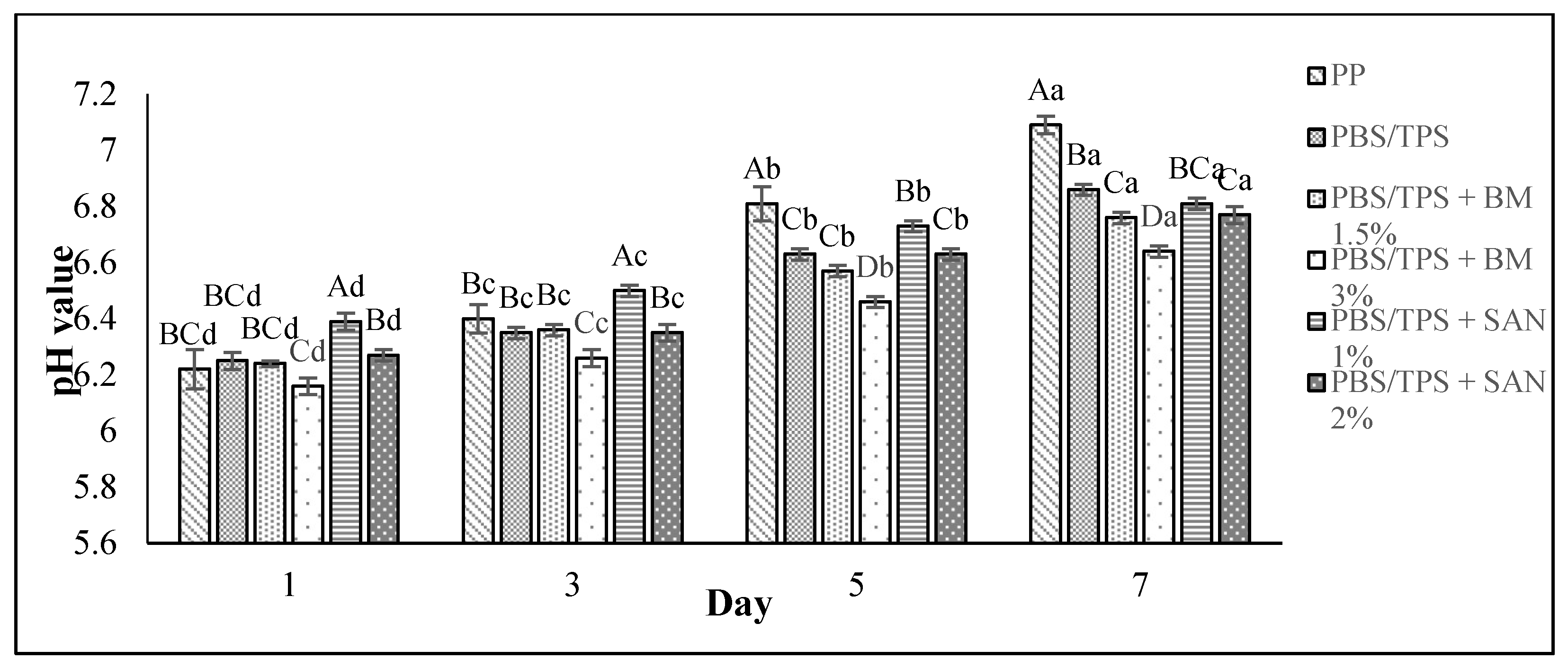
3.2. pH Value
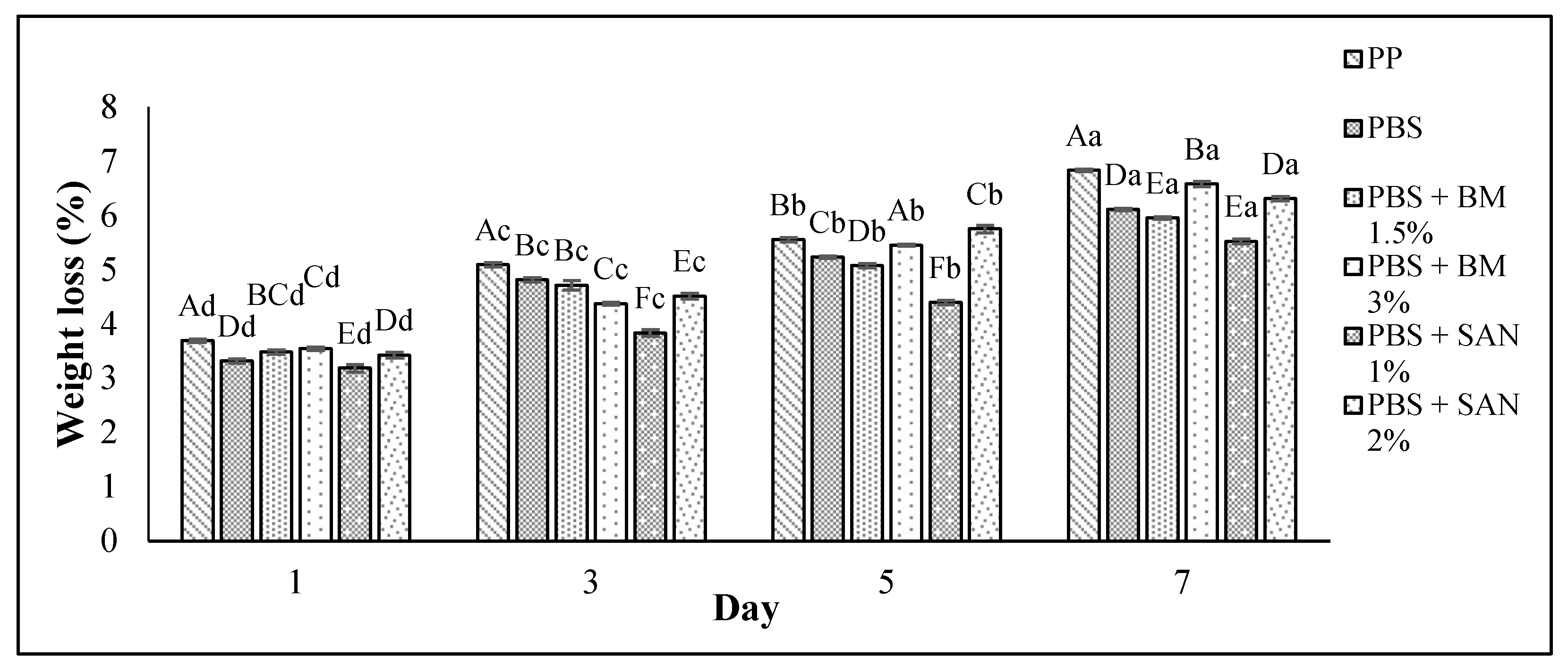
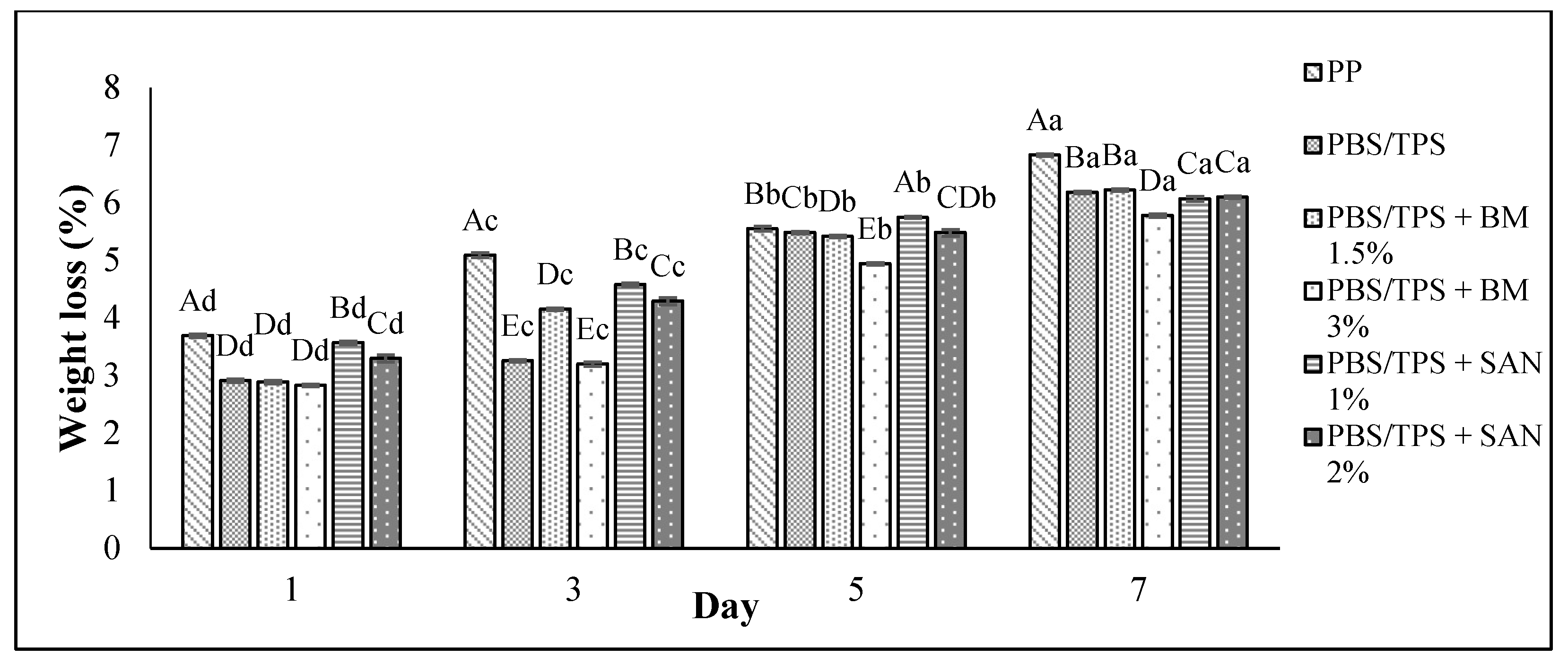
3.3. Weight Loss
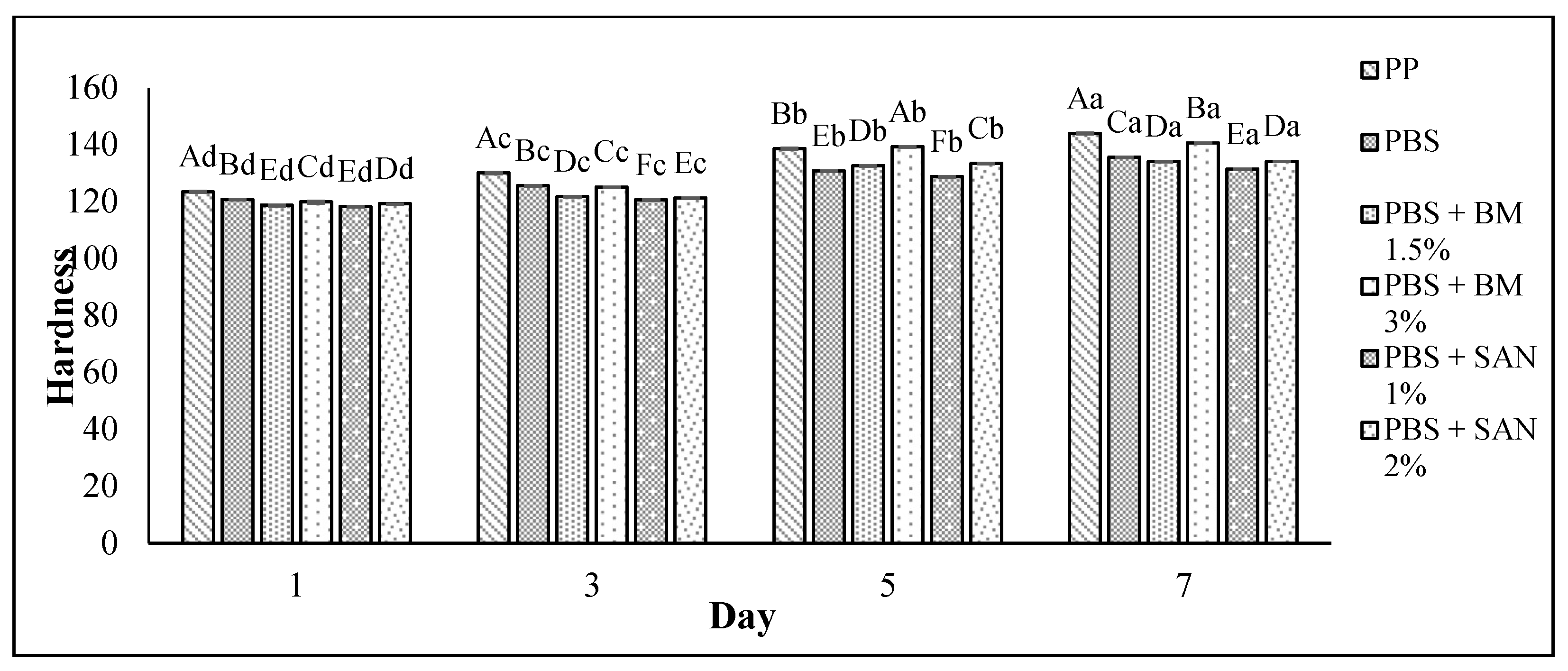
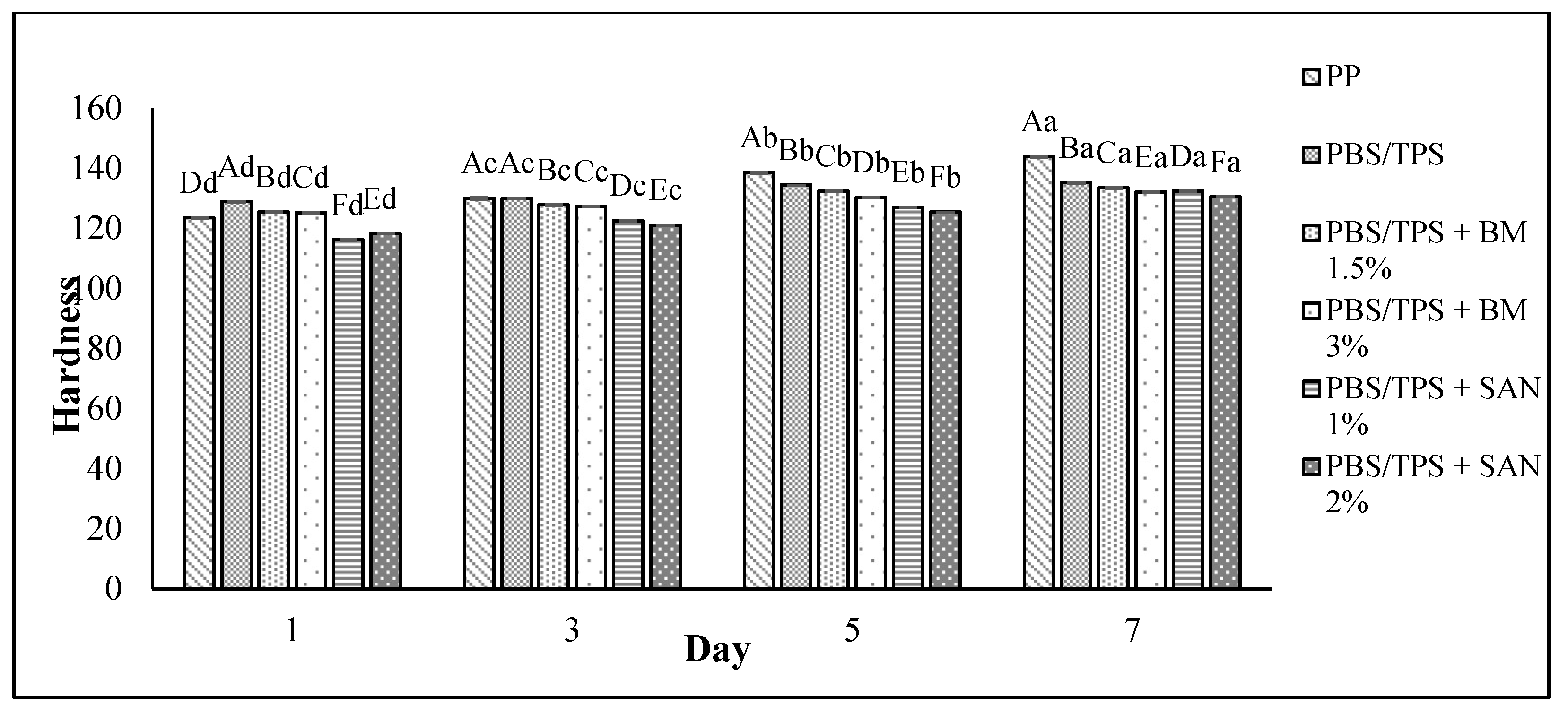
3.4. Texture Analysis
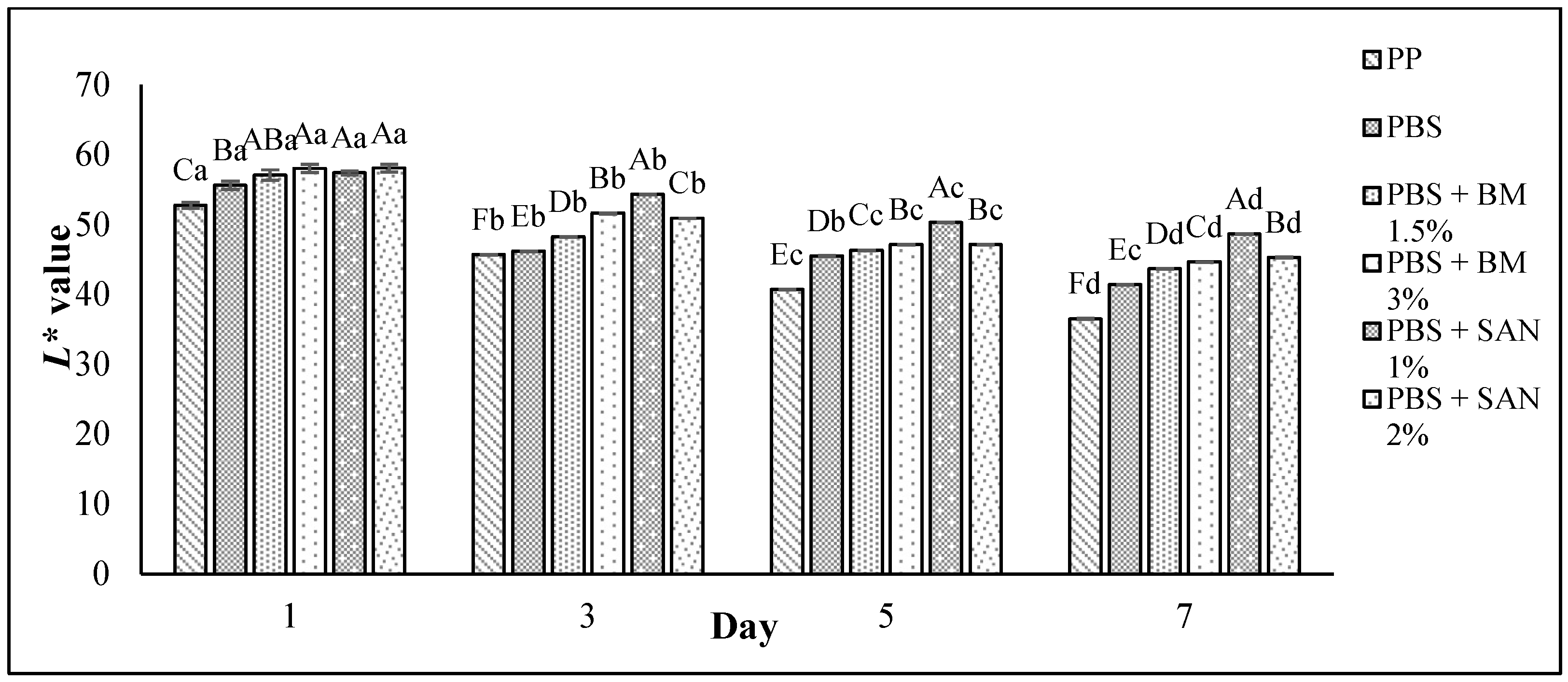
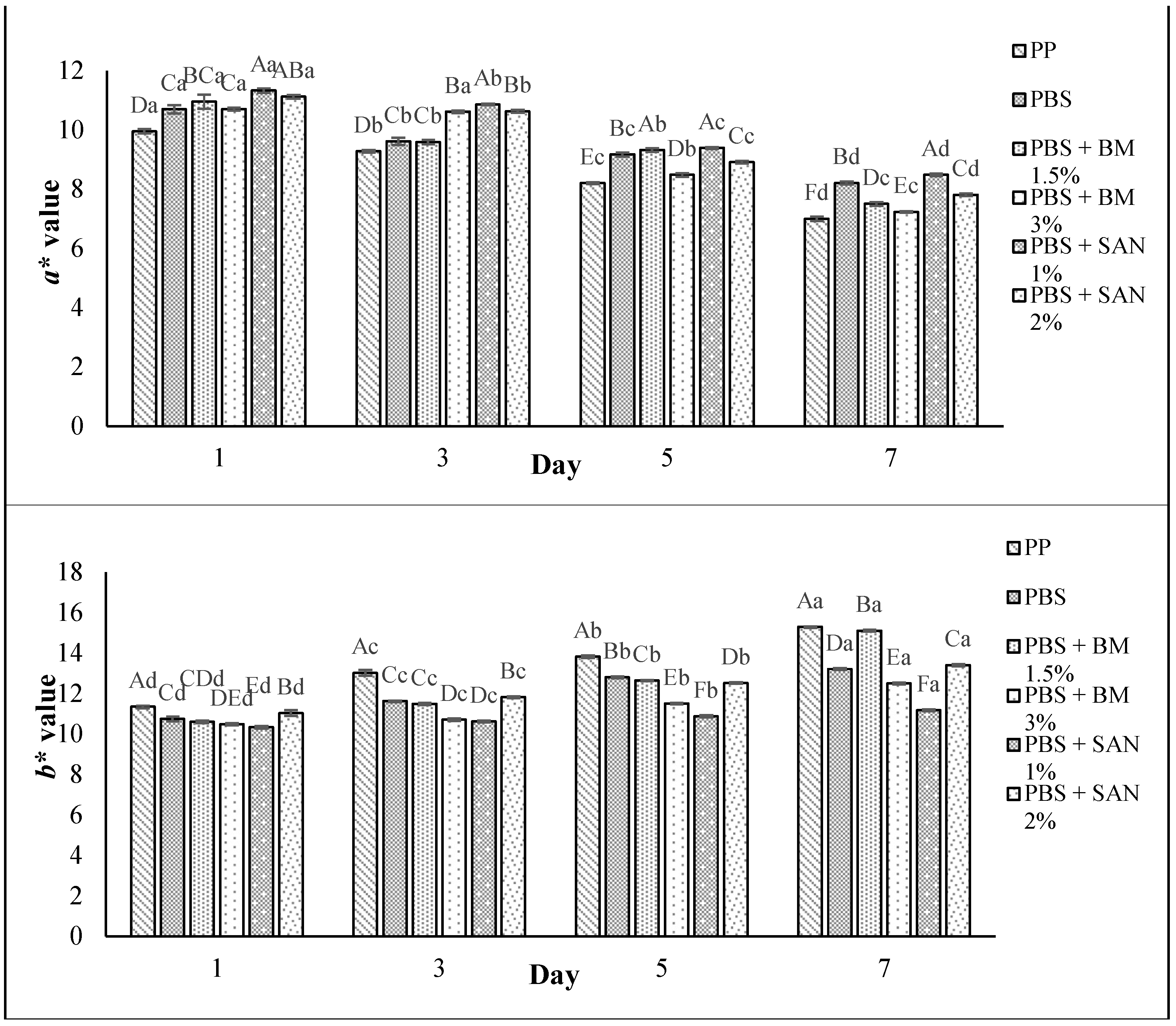
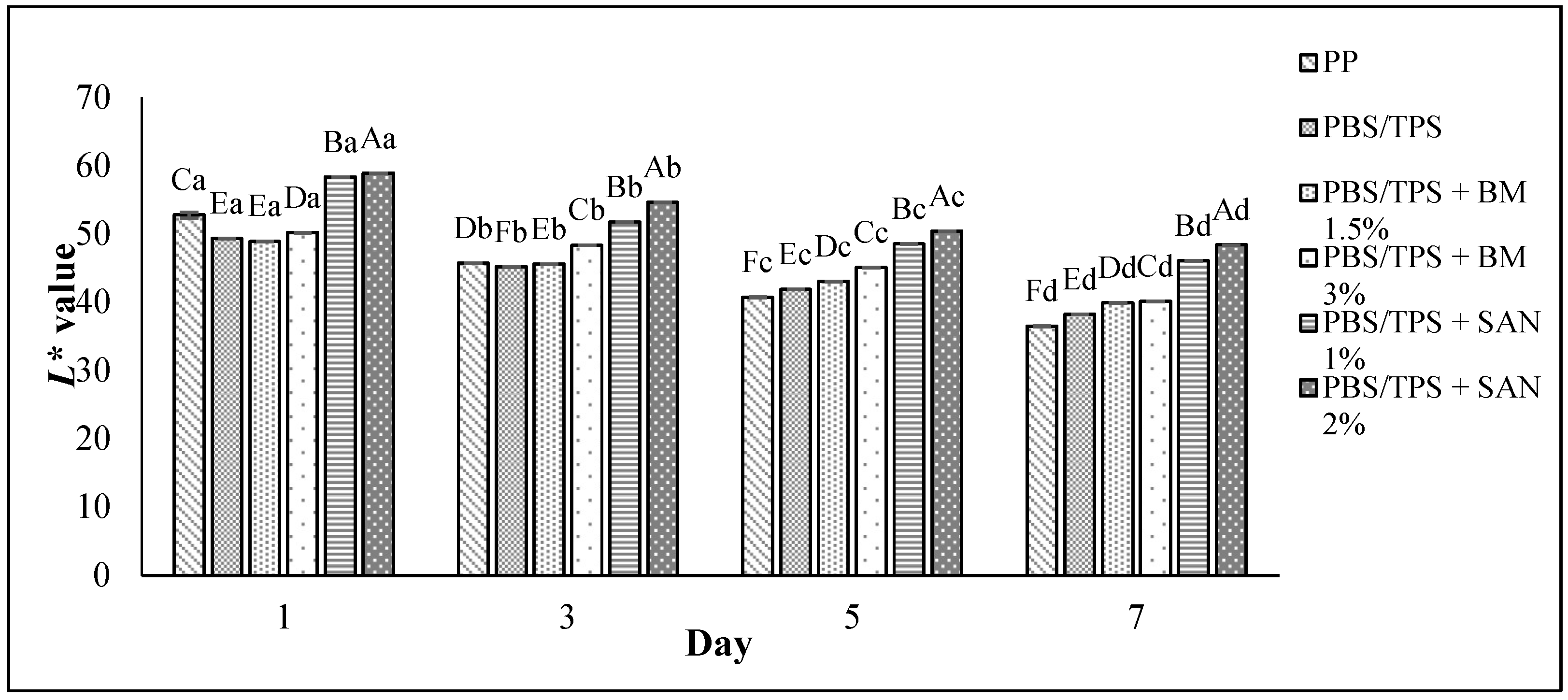
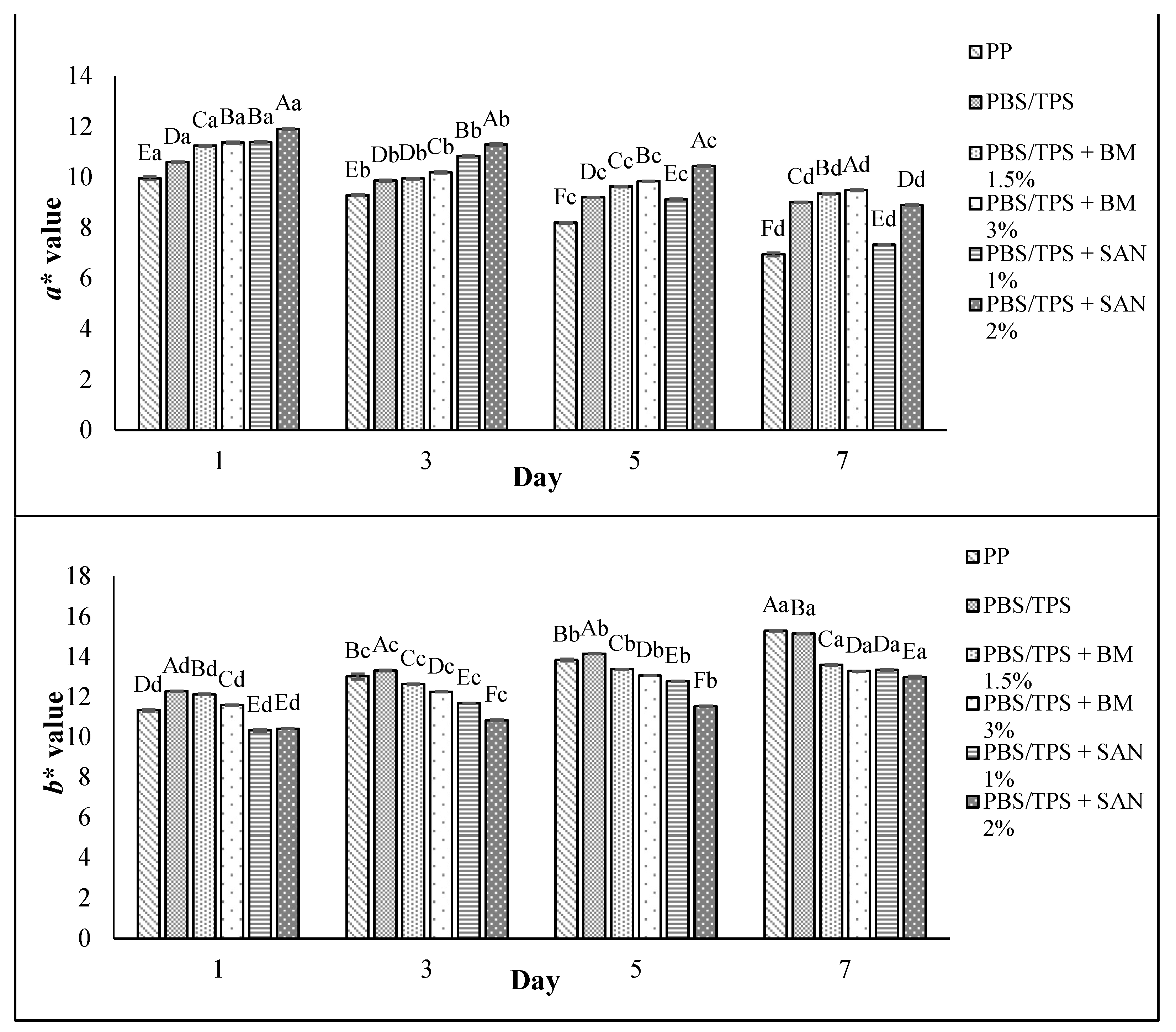
3.5. Color Changes and Overall Visual Quality
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Malhotra, B.; Keshwani, A.; Kharkwal, H. Antimicrobial food packaging: Potential and pitfalls. Front. Microbiol. 2015, 6, 611. [Google Scholar] [CrossRef] [Green Version]
- Quintavalla, S.; Vicini, L. Antimicrobial food packaging in meat industry. Meat Sci. 2002, 62, 373–380. [Google Scholar] [CrossRef]
- Vaithiyanathan, S.; Naveena, B.M.; Muthukumar, M.; Girish, P.S.; Kondaiah, N. Effect of dipping in pomegranate (Punica granatum) fruit juice phenolic solution on the shelf life of chicken meat under refrigerated storage (4 °C). Meat Sci. 2011, 88, 409–414. [Google Scholar] [CrossRef]
- Konuk, D.T.; Korel, F. Active packaging films as a carrier of black cumin essential oil: Development and effect on quality and shelf-life of chicken breast meat. Food Packag. Shelf Life 2019, 19, 210–217. [Google Scholar] [CrossRef]
- Díaz-Galindo, E.P.; Nesic, A.; Cabrera-Barjas, G.; Mardones, C.; von Baer, D.; Bautista-Baños, S.; Dublan Garcia, O. Physical-chemical evaluation of active food packaging material based on thermoplastic starch loaded with grape cane extract. Molecules 2020, 25, 1306. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.G.; Zhang, X.C.; Li, H.Y.; Liu, Z. Effect of surface modification with 3-aminopropyltriethyloxy silane on mechanical and crystallization performances of ZnO/poly(butylenesuccinate) composites. Compos. Part B Eng. 2012, 43, 2209–2216. [Google Scholar] [CrossRef]
- Pandey, J.K.; Reddy, K.R.; Kumar, A.P.; Singh, R.P. An overview on the degradability of polymer nanocomposites. Polym. Degrad. Stab. 2005, 88, 234–250. [Google Scholar] [CrossRef]
- Nam, T.H.; Ogihara, S.; Tung, N.H.; Kobayashi, S. Effect of alkali treatment on interfacial and mechanical properties of coir fiber reinforced poly(butylene succinate) biodegradable composites. Compos. Part B Eng. 2011, 42, 1648–1656. [Google Scholar] [CrossRef]
- Khan, B.; Niazi, M.B.K.; Samin, G.; Jahan, Z. Thermoplastic Starch: A Possible Biodegradable Food Packaging Material—A Review. J. Food Process Eng. 2017, 40, e12447. [Google Scholar] [CrossRef]
- Fahrngruber, B.; Fortea-Verdejo, M.; Wimmer, R.; Mundigler, N. Starch/Poly(butylene succinate) compatibilizers: Effect of different reaction-approaches on the properties of thermoplastic starch-based compostable films. J. Polym. Environ. 2020, 28, 257–270. [Google Scholar] [CrossRef]
- Yin, Q.J.; Chen, F.P.; Zhang, H.; Liu, C.S. Mechanical properties and thermal behavior of TPS/PBS Blends with maleated PBS as a compatibilizer. Adv. Mater. Res. 2015, 1119, 306–309. [Google Scholar] [CrossRef]
- Petchwattana, N.; Covavisaruch, S.; Wibooranawong, S.; Naknaen, P. Antimicrobial food packaging prepared from poly(butylene succinate) and zinc oxide. Measurement 2016, 93, 442–448. [Google Scholar] [CrossRef]
- Llorens, A.; Lloret, E.; Picouet, P.A.; Trbojevich, R.; Fernandez, A. Metallic-based micro and nanocomposites in food contact materials and active food packaging. Trends Food Sci. Technol. 2012, 24, 19–29. [Google Scholar] [CrossRef]
- Zhao, S.; Yao, J.; Fei, X.; Shao, Z.; Chen, X. An antimicrobial film by embedding in situ synthesized silver nanoparticles in soy protein isolate. Mater. Lett. 2013, 95, 142–144. [Google Scholar] [CrossRef]
- Theivendran, S.; Hettiarachchy, N.S.; Johnson, M.G. Inhibition of Listeria monocytogenes by nisin combined with grape seed extract or green tea extract in soy protein film coated on Turkey Frankfurters. J. Food Sci. 2006, 71, 39–44. [Google Scholar] [CrossRef]
- Mauriello, G.; De Luca, E.; La Storia, A.; Villani, F.; Ercolini, D. Antimicrobial activity of a nisin-activated plastic film for food packaging. Lett. Appl. Microbiol. 2005, 41, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Sivarooban, T.; Hettiarachchy, N.S.; Johnson, M.G. Physical and antimicrobial properties of grape seed extract, nisin, and EDTA incorporated soy protein edible films. Food Res. Int. 2008, 41, 781–785. [Google Scholar] [CrossRef]
- Calatayud, M.; López-de-Dicastillo, C.; López-Carballo, G.; Vélez, D.; Hernández Muñoz, P.; Gavara, R. Active films based on cocoa extract with antioxidant, antimicrobial and biological applications. Food Chem. 2013, 139, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Graü, M.A.; Avena-Bustillos, R.J.; Olsen, C.; Friedman, M.; Henika, P.R.; Martín-Belloso, O.; Pan, Z.; McHugh, T.H. Effects of plant essential oils and oil compounds on mechanical, barrier and antimicrobial properties of alginate–apple puree edible films. J. Food Eng. 2007, 81, 634–641. [Google Scholar] [CrossRef]
- Holt, K.B.; Bard, A.J. Interaction of silver(i) ions with the respiratory chain of Escherichia coli: An electrochemical and scanning electrochemical microscopy study of the antimicrobial mechanism of micromolar Ag+. Biochemistry 2005, 44, 13214–13223. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Qu, F.; Xu, H.; Lai, W.; Wang, Y.A.; Aguilar, Z.P.; Wei, H. Role of reactive oxygen species in the antibacterial mechanism of silver nanoparticles on Escherichia coli O157: H7. Biometals 2012, 25, 45–53. [Google Scholar] [CrossRef]
- Matés, J.M.; Sánchez-Jiménez, F.M. Role of reactive oxygen species in apoptosis: Implications for cancer therapy. Int. J. Biochem. Cell Biol. 2000, 32, 157–170. [Google Scholar] [CrossRef]
- Masoumi, B.; Abbasi, A.; Mazloomi, S.M. The effect of saffron on microbial, physicochemical and texture profile of chicken (breast) meat stored in refrigerator. Int. J. Nutr. Sci. 2018, 3, 164–170. [Google Scholar]
- Wang, J.; Zhuang, H.; Hinton, A.; Zhang, J. Influence of in-package cold plasma treatment on microbiological shelf life and appearance of fresh chicken breast fillets. Food Microbiol. 2016, 60, 142–146. [Google Scholar] [CrossRef]
- Mohammadi, H.; Kamkar, A.; Misaghi, A.; Zunabovic-Pichler, M.; Fatehi, S. Nanocomposite films with CMC, okra mucilage, and ZnO nanoparticles: Extending the shelf-life of chicken breast meat. Food Packag. Shelf Life 2019, 21, 100330. [Google Scholar] [CrossRef]
- Senter, S.D.; Arnold, J.W.; Chew, V. APC values and volatile compounds formed in commercially processed, raw chicken parts during storage at 4 and 13 °C and under simulated temperature abuse conditions. J. Sci. Food Agric. 2000, 80, 1559–1564. [Google Scholar] [CrossRef]
- Bian, J.; Han, L.; Wang, X.; Wen, X.; Han, C.; Wang, S.; Dong, L. Nonisothermal crystallization behavior and mechanical properties of poly(butylene succinate)/silica nanocomposites. J. Appl. Polym. Sci. 2010, 116, 902–912. [Google Scholar] [CrossRef]
- Appendini, P.; Hotchkiss, J.H. Review of antimicrobial food packaging. Innov. Food Sci. Emerg. Technol. 2002, 3, 113–126. [Google Scholar] [CrossRef]
- Warsiki, E.; Bawardi, J.T. Assessing mechanical properties and antimicrobial activity of zinc oxide-starch biofilm. IOP Confer. Ser. Earth Environ. Sci. 2018, 209, 012003. [Google Scholar] [CrossRef]
- Cardoso, L.G.; Pereira Santos, J.C.; Camilloto, G.P.; Miranda, A.L.; Druzian, J.I.; Guimarães, A.G. Development of active films poly (butylene adipate co-terephthalate)—PBAT incorporated with oregano essential oil and application in fish fillet preservation. Ind. Crop. Prod. 2017, 108, 388–397. [Google Scholar] [CrossRef]
- Ahmed, J.; Mulla, M.Z.; Arfat, Y.A. Thermo-mechanical, structural characterization and antibacterial performance of solvent casted polylactide/cinnamon oil composite films. Food Control 2016, 69, 196–204. [Google Scholar] [CrossRef]
- Shahbazi, Y.; Shavisi, N. A novel active food packaging film for shelf-life extension of minced beef meat. J. Food Saf. 2018, 38, 12569. [Google Scholar] [CrossRef]
- Emma, G.; Lesley, T. Using silver to help combat Campylobacter and other bacteria. Perspect. Public Health 2013, 133, 292–293. [Google Scholar]
- Kumar, R.; Ghoshal, G.; Goyal, M. Biodegradable composite films/coatings of modified corn starch/gelatin for shelf life improvement of cucumber. J. Food Sci. Technol. 2020, 58, 1227–1237. [Google Scholar] [CrossRef] [PubMed]
- Rashidaie, S.S.A.; Ariaii, P.; Charmchian Langerodi, M. Effects of encapsulated rosemary extract on oxidative and microbiological stability of beef meat during refrigerated storage. Food Sci. Nutr. 2019, 7, 3969–3978. [Google Scholar] [CrossRef] [PubMed]
- Katiyo, W.; de Kock, H.L.; Coorey, R.; Buys, E.M. Sensory implications of chicken meat spoilage in relation to microbial and physicochemical characteristics during refrigerated storage. LWT 2020, 128, 109468. [Google Scholar] [CrossRef]
- Ashrafi, A.; Jokar, M.; Mohammadi Nafchi, A. Preparation and characterization of biocomposite film based on chitosan and kombucha tea as active food packaging. Int. J. Biol. Macromol. 2018, 108, 444–454. [Google Scholar] [CrossRef]
- Souza, V.G.L.; Pires, J.R.A.; Vieira, É.T.; Coelhoso, I.M.; Duarte, M.P.; Fernando, A.L. Shelf life assessment of fresh poultry meat packaged in novel bionanocomposite of chitosan/montmorillonite incorporated with ginger essential oil. Coatings 2018, 8, 177. [Google Scholar] [CrossRef] [Green Version]
- Amjadi, S.; Emaminia, S.; Nazari, M.; Davudian, S.H.; Roufegarinejad, L.; Hamishehkar, H. Application of Reinforced ZnO Nanoparticle-Incorporated Gelatin Bionanocomposite Film with Chitosan Nanofiber for Packaging of Chicken Fillet and Cheese as Food Models. Food Bioprocess Technol. 2019, 12, 1205–1219. [Google Scholar] [CrossRef]
- Gazalli, H.; Malik, A.H.; Jalal, H.; Afshan, S.; Mir, A.; Ashraf, H. Packaging of meat. Int. J. Food Nutr. Saf. 2013, 4, 70–80. [Google Scholar]
| No. | PBS Film Code | Antimicrobial Agent | g/g (%) Amount of Antimicrobial Agent |
|---|---|---|---|
| 1 | PBS (untreated) | N/A | N/A |
| 2 | PBS + BM 1.5% | BM | 1.5 |
| 3 | PBS + BM 3% | BM | 3 |
| 4 | PBS + SAN 1% | SAN | 1 |
| 5 | PBS + SAN 2% | SAN | 2 |
| 4 | PBS/TPS (untreated) | N/A | N/A |
| 5 | PBS/TPS + BM 1.5% | BM | 1.5 |
| 6 | PBS/TPS + BM 3% | BM | 3 |
| 7 | PBS/TPS + SAN 1% | SAN | 1 |
| 8 | PBS/TPS + SAN 2% | SAN | 2 |
| Day | 0 | 1 | 3 | 5 | 7 |
|---|---|---|---|---|---|
| PP |  |  |  |  |  |
| PBS |  | 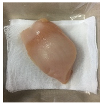 | 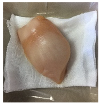 |  | 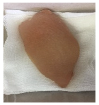 |
| PBS + BM 1.5% | 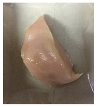 |  | 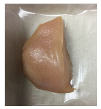 | 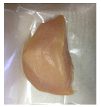 | 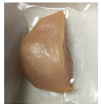 |
| PBS + BM 3% |  |  |  |  |  |
| PBS + SAN 1% | 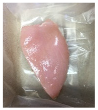 | 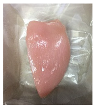 | 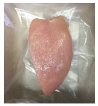 |  | 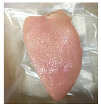 |
| PBS + SAN 2% | 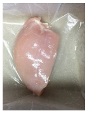 | 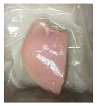 | 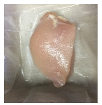 |  | 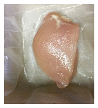 |
| Day | 0 | 1 | 3 | 5 | 7 |
|---|---|---|---|---|---|
| PP | 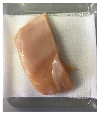 |  | 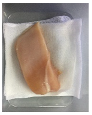 |  |  |
| PBS/TPS | 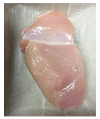 | 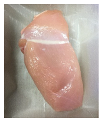 |  |  |  |
| PBS/TPS + BM 1.5% | 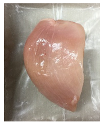 |  |  |  |  |
| PBS/TPS + BM 3% | 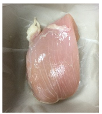 |  |  |  |  |
| PBS/TPS + SAN 1% | 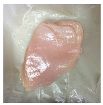 | 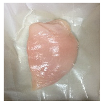 |  |  |  |
| PBS/TPS + SAN 2% | 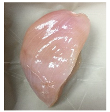 | 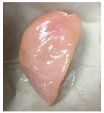 | 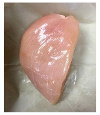 | 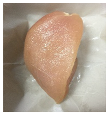 |  |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yusof, N.L.; Mutalib, N.-A.A.; Nazatul, U.K.; Nadrah, A.H.; Aziman, N.; Fouad, H.; Jawaid, M.; Ali, A.; Kian, L.K.; Sain, M. Efficacy of Biopolymer/Starch Based Antimicrobial Packaging for Chicken Breast Fillets. Foods 2021, 10, 2379. https://doi.org/10.3390/foods10102379
Yusof NL, Mutalib N-AA, Nazatul UK, Nadrah AH, Aziman N, Fouad H, Jawaid M, Ali A, Kian LK, Sain M. Efficacy of Biopolymer/Starch Based Antimicrobial Packaging for Chicken Breast Fillets. Foods. 2021; 10(10):2379. https://doi.org/10.3390/foods10102379
Chicago/Turabian StyleYusof, Noor L., Noor-Azira Abdul Mutalib, U. K. Nazatul, A. H. Nadrah, Nurain Aziman, Hassan Fouad, Mohammad Jawaid, Asgar Ali, Lau Kia Kian, and Mohini Sain. 2021. "Efficacy of Biopolymer/Starch Based Antimicrobial Packaging for Chicken Breast Fillets" Foods 10, no. 10: 2379. https://doi.org/10.3390/foods10102379
APA StyleYusof, N. L., Mutalib, N.-A. A., Nazatul, U. K., Nadrah, A. H., Aziman, N., Fouad, H., Jawaid, M., Ali, A., Kian, L. K., & Sain, M. (2021). Efficacy of Biopolymer/Starch Based Antimicrobial Packaging for Chicken Breast Fillets. Foods, 10(10), 2379. https://doi.org/10.3390/foods10102379