Kinetic Study of Subcritical Water Extraction of Scopoletin, Alizarin, and Rutin from Morinda citrifolia
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Material and Chemicals
2.2. Methanol Extraction
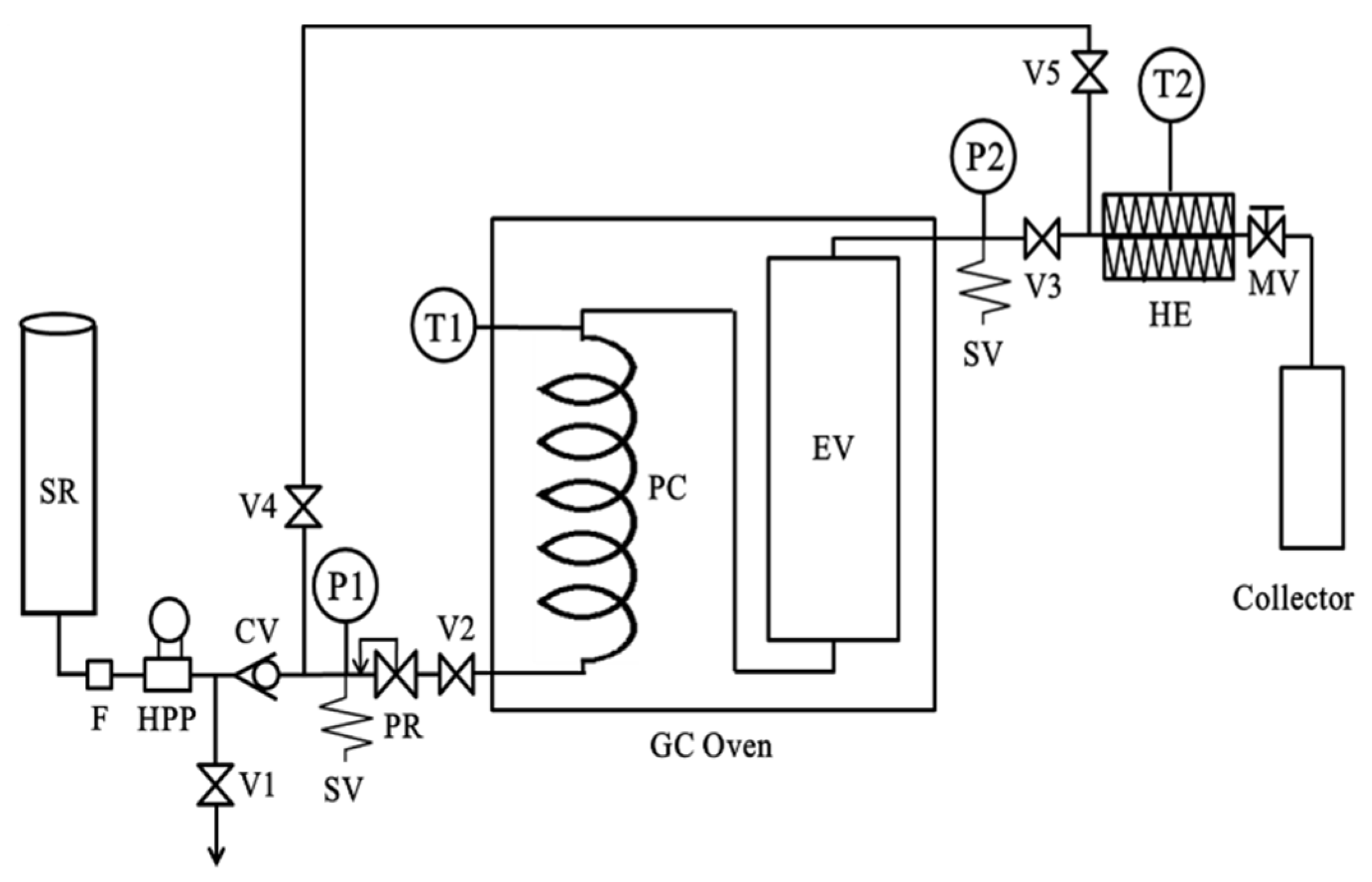
2.3. Subcritical Water Extraction
2.4. HPLC Analysis
2.5. Kinetic Modeling
3. Results and Discussion
3.1. Effect of Subcritical Water Temperature
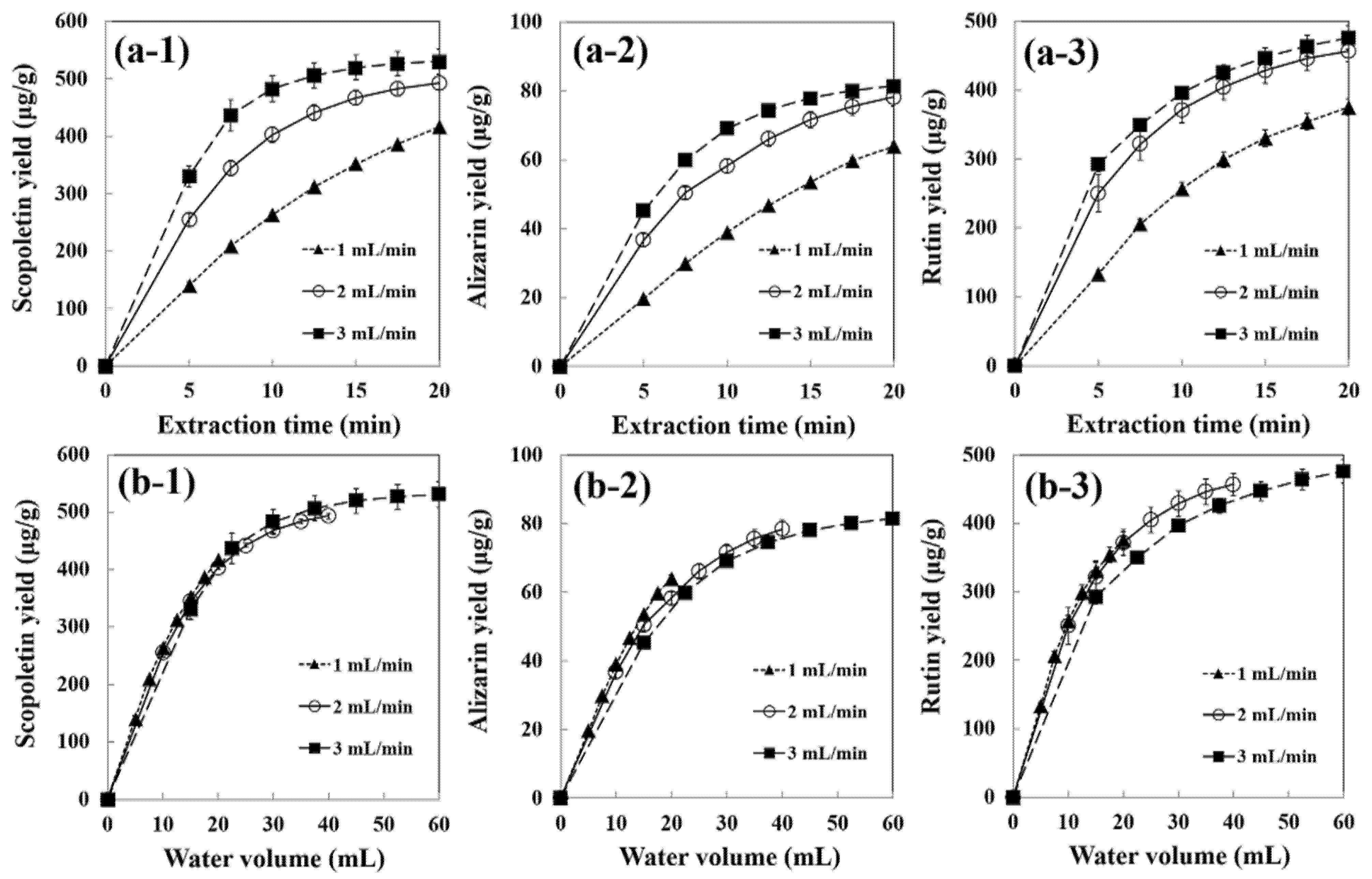
3.2. Effect of Water Flow Rate
3.3. Kinetic Modeling
3.4. Diffusion Coefficient and Activation Energy
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ali, M.; Kenganora, M.; Manjula, S.N. Health benefits of Morinda citrifolia (Noni): A review. Pharmacogn. J. 2016, 8, 321–334. [Google Scholar] [CrossRef] [Green Version]
- Torres, M.A.O.; de Fátima Braga Magalhães, I.; Mondêgo-Oliveira, R.; de Sá, J.C.; Rocha, A.L.; Abreu-Silva, A.L. One plant, many uses: A review of the pharmacological applications of Morinda citrifolia. Phyther. Res. 2017, 31, 971–979. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Yu, J.S.; Huang, P.; Qader, M.; Manavalan, A.; Wu, X.; Kim, J.C.; Pang, C.; Cao, S.; Kang, K.S.; et al. Identification of anti-inflammatory compounds from Hawaiian noni (Morinda citrifolia L.) fruit Juice. Molecules 2020, 25, 4968. [Google Scholar] [CrossRef] [PubMed]
- Chan-Blanco, Y.; Vaillant, F.; Mercedes Perez, A.; Reynes, M.; Brillouet, J.M.; Brat, P. The noni fruit (Morinda citrifolia L.): A review of agricultural research, nutritional and therapeutic properties. J. Food Comp. Anal. 2006, 19, 645–654. [Google Scholar] [CrossRef]
- Mahanthesh, M.C.; Manjappa, A.S.; Shindhe, M.V.; Jamkhandi, C.M.; Jalapure, S.S.; Patil, S.S. Morinda citrifolia Linn; a medicinal plant with diverse phytochemicals and its medicinal relevance. World J. Pharm. Res. 2013, 3, 215–232. [Google Scholar]
- Assi, R.A.; Darwis, T.; Abdulbaqi, I.M.; Khan, A.A.; Vuanghao, L.; Laghari, M.H. Morinda citrifolia (Noni): A comprehensive review on its industrial uses, pharmacological activities, and clinical trials. Arab. J. Chem. 2017, 10, 691–707. [Google Scholar] [CrossRef]
- Inada, A.C.; Figueiredo, P.S.; Santos-Eichler, R.A.; Freitas, K.C.; Hiane, P.A.; de Castro, A.P.; de Cássia Avellaneda Guimarães, R. Morinda citrifolia Linn. (noni) and its potential in obesity-related metabolic dysfunction. Nutrients 2017, 9, 540. [Google Scholar] [CrossRef]
- Choi, R.Y.; Ham, J.R.; Lee, H.I.; Cho, H.W.; Choi, M.S.; Park, S.K.; Lee, J.; Kim, M.J.; Seo, K.I.; Lee, M.K. Scopoletin supplementation ameliorates steatosis and inflammation in diabetic mice. Phyther. Res. 2017, 31, 1795–1804. [Google Scholar] [CrossRef]
- Ding, Z.; Dai, Y.; Hao, H.; Pan, R.; Yao, X.; Wang, Z. Anti-inflammatory effects of scopoletin and underlying mechanisms. Pharm. Biol. 2008, 46, 854–860. [Google Scholar] [CrossRef]
- Hornick, C.A.; Myers, A.; Sadowska, K.H.; Anthony, C.T.; Woltering, E.A. Inhibition of angiogenic initiation and disruption of newly established human vascular networks by juice from Morinda citrifolia (noni). Angiogenesis 2003, 6, 143–149. [Google Scholar] [CrossRef]
- Fotia, C.; Avnet, S.; Granchi, D.; Baldini, N. The natural compound alizarin as an osteotropic drug for the treatment of bone tumors. J. Orthop. Res. 2012, 30, 1486–1492. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pandy, V.; Narasingam, M.; Kunasegaran, T.; Murugan, D.D.; Mohamed, Z. Effect of noni (Morinda citrifolia Linn.) fruit and its bioactive principles scopoletin and rutin on rat vas deferens contractility: An ex vivo study. Sci. World J. 2014, 8, 909586. [Google Scholar]
- Huang, R.; Shi, Z.; Chen, L.; Zhang, Y.; Li, J.; An, Y. Rutin alleviates diabetic cardiomyopathy and improves cardiac function in diabetic ApoEknockout mice. Eur. J. Pharmacol. 2017, 814, 151–160. [Google Scholar] [CrossRef]
- Hawthorne, S.B.; Grabanski, C.B.; Martin, E.; Miller, D.J. Comparisons of Soxhlet extraction, pressurized liquid extraction, supercritical fluid extraction and subcritical water extraction for environmental solids: Recovery, selectivity and effects on sample matrix. J. Chromatogr. A 2000, 892, 421–433. [Google Scholar] [CrossRef]
- Barba, F.J.; Zhenzhou, Z.; Koubaa, M.; Sant’Ana, A.S.; Orlien, V. Green alternative methods for the extraction of antioxidant bioactive compounds from winery wastes and by-products: A review. Trends Food Sci. Technol. 2016, 49, 96–109. [Google Scholar] [CrossRef]
- Ameer, K.; Shahbaz, H.M.; Kwon, J.H. Green extraction methods for polyphenols from plant matrices and their byproducts: A review. Compr. Rev. Food Sci. Food Saf. 2017, 16, 295–315. [Google Scholar] [CrossRef] [Green Version]
- Mortazavi, S.V.; Eikani, M.H.; Mirzaei, H.; Jafari, M.; Golmohammad, F. Extraction of essential oils from Bunium persicum Boiss. using superheated water. Food Bioprod. Process. 2010, 88, 222–226. [Google Scholar] [CrossRef]
- Plaza, M.; Turner, C. Pressurized hot water extraction of bioactives. Trends Anal. Chem. 2015, 71, 39–54. [Google Scholar] [CrossRef] [Green Version]
- Castro-Puyana, M.; Marina, M.L.; Plaza, M. Water as green extraction solvent: Principles and reasons for its use. Curr. Opin. Green Sustain. Chem. 2017, 5, 31–36. [Google Scholar] [CrossRef]
- Sarfarazi, M.; Jafari, S.M.; Rajabzadeh, G.; Feizi, J. Development of an environmentally-friendly solvent-free extraction of saffron bioactives using subcritical water. LWT–Food Sci. Technol. 2019, 114, 108428. [Google Scholar] [CrossRef]
- Cheng, Y.; Xue, F.; Yu, S.; Du, S.; Yang, Y. Subcritical water extraction of natural products. Molecules 2021, 26, 4004. [Google Scholar] [CrossRef]
- Kim, J.W.; Mazza, G. Mass transfer during pressurized low-polarity water extraction of phenolics and carbohydrates from flax shives. Ind. Eng. Chem. Res. 2007, 46, 7221–7230. [Google Scholar] [CrossRef]
- Asl, A.H.; Khajenoori, M. Subcritical water extraction. In Mass Transfer-Advances in Sustainable Energy and Environment Oriented Numerical Modeling; Nakajima, H., Ed.; InTech: Rijeka, Croatia, 2013; pp. 464–465. [Google Scholar]
- Shotipruk, A.; Kiatsongserm, J.; Pavasant, P.; Goto, M.; Sasaki, M. Pressurized hot water extraction of anthraquinones from the roots of Morinda citrifolia. Biotechnol. Prog. 2004, 20, 1872–1875. [Google Scholar] [CrossRef] [PubMed]
- Anekpankul, T.; Goto, M.; Sasaki, M.; Pavasant, P.; Shotipruk, A. Extraction of anti-cancer damnacanthal from roots of Morinda citrifolia by subcritical water. Sep. Purif. Technol. 2007, 55, 343–349. [Google Scholar] [CrossRef]
- Barrera Vázquez, M.F.; Comini, L.R.; Milanesio, J.M.; Núnez Montoya, S.C.; Cabrera, J.L.; Bottini, S.; Martini, R.E. Pressurized hot water extraction of anthraquinones from Heterophyllaea pustulata Hook f. (Rubiaceae). J. Supercrit. Fluids 2015, 101, 170–175. [Google Scholar] [CrossRef]
- Duba, K.S.; Casazza, A.A.; Mohamed, H.B.; Perego, P.; Fiori, L. Extraction of polyphenols from grape skins and defatted grape seeds using subcritical water: Experiments and modeling. Food Bioprod. Process. 2015, 94, 29–38. [Google Scholar] [CrossRef]
- Kim, D.S.; Lim, S.B. Subcritical water extraction of rutin from the aerial parts of common buckwheat. J. Supercrit. Fluids 2019, 152, 104561. [Google Scholar] [CrossRef]
- Mufari, J.R.; Rodríguez-Ruiz, A.C.; Bergesse, A.E.; Miranda-Villa, P.P.; Nepote, V.; Velez, A.R. Bioactive compounds extraction from malted quinoa using water-ethanol mixtures under subcritical conditions. LWT–Food Sci. Technol. 2021, 138, 110574. [Google Scholar] [CrossRef]
- Islam, M.N.; Jo, Y.T.; Jung, S.K.; Park, J.H. Thermodynamic and kinetic study for subcritical water extraction of PAHs. J. Ind. Eng. Chem. 2013, 19, 29–136. [Google Scholar] [CrossRef]
- Ahmadian-Kouchaksaraie, Z.; Niazmand, R.; Najafi, M.N. Optimization of the subcritical water extraction of phenolic antioxidants from Crocus sativus petals of saffron industry residues: Box-Behnken design and principal component analysis. Innov. Food Sci. Emerg. Technol. 2016, 36, 234–244. [Google Scholar] [CrossRef]
- Munir, M.T.; Kheirkhah, H.; Baroutian, S.; Quek, S.Y.; Young, B.R. Subcritical water extraction of bioactive compounds from waste onion skin. J. Clean. Prod. 2018, 183, 487–494. [Google Scholar] [CrossRef]
- Tatke, P.; Rajan, M. Comparison of conventional and novel extraction techniques for the extraction of scopoletin from Convolvulus Pluricaulis. Ind. J. Pharm. Educ. Res. 2014, 48, 27–31. [Google Scholar] [CrossRef] [Green Version]
- Derksen, G.C.H.; Naayer, M.; van Beek, T.A.; Capelle, A.; Haaksman, I.K.; van Doren, H.A.; de Groot, A. Chemical and enzymatic hydrolysis of anthraquinone glycosides from Madder roots. Phytochem. Anal. 2003, 14, 137–144. [Google Scholar] [CrossRef]
- De Santis, D.; Moresi, M. Production of alizarin extracts from Rubia tinctorum and assessment of their dyeing properties. Ind. Crop. Prod. 2007, 26, 151–162. [Google Scholar] [CrossRef]
- Caro, Y.; Anamale, L.; Fouillaud, M.; Laurent, P.; Petit, T.; Dufosse, L. Natural hydroxyl anthraquinoid pigments as potent food grade colorants: An overview. Nat. Prod. Bioprospect. 2012, 2, 174–193. [Google Scholar] [CrossRef]
- Ford, L.; Rayner, C.M.; Blackburn, R.S. Isolation and extraction of ruberythric acid from Rubia tinctorum L. and crystal structure elucidation. Phytochemistry 2015, 117, 168–173. [Google Scholar] [CrossRef] [Green Version]
- Brunner, G. Hydrothermal and Supercritical Water Process; Kiran, D., Ed.; Elsevier: Amsterdam, The Netherlands, 2014; p. 77. [Google Scholar]
- Sengers, J.V.; Watson, J.T.R. Improved international formulations for the viscosity and thermal conductivity of water substance. J. Phys. Chem. Ref. Data 1986, 15, 1291–1314. [Google Scholar] [CrossRef] [Green Version]
- Pongnaravane, B.; Goto, M.; Sasaki, M.; Anekpankul, T.; Pavasant, P.; Shotipruk, A. Extraction of anthraquinones from roots of Morinda citrifolia by pressurized hot water: Antioxidant activity of extracts. J. Supercrit. Fluids 2006, 37, 390–396. [Google Scholar] [CrossRef]
- Shabkhiz, M.A.; Eikani, M.H.; Sadr, Z.B.; Golmohammad, F. Superheated water extraction of glycyrrhizic acid from licorice root. Food Chem. 2016, 210, 396–401. [Google Scholar] [CrossRef] [PubMed]
- Sovová, H. Steps of supercritical fluid extraction of natural products and their characteristic times. J. Supercrit. Fluids 2012, 66, 73–79. [Google Scholar] [CrossRef]
- Ho, C.H.L.; Cacace, J.E.; Mazza, G. Mass transfer during pressurized low polarity water extraction of lignans from flaxseed meal. J. Food Eng. 2008, 89, 64–71. [Google Scholar] [CrossRef]
- Kim, D.S.; Lim, S.B. Kinetic study of subcritical water extraction of flavonoids from citrus unshiu peel. Sep. Purif. Technol. 2020, 250, 117259. [Google Scholar] [CrossRef]
- Al-Hamimi, S.; Mayoral, A.A.; Cunico, L.P.; Turner, C. Carbon dioxide expanded ethanol extraction: Solubility and extraction kinetics of α-pinene and cis-verbenol. Anal. Chem. 2016, 88, 4336–4345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mottahedin, P.; Asl, A.H.; Khajenoori, M. Extraction of curcumin and essential oil from Curcuma longa L. by subcritical water via response surface methodology. J. Food Process. Preserv. 2017, 41, e13095. [Google Scholar] [CrossRef]
- Lakhotiya, H.; Mondal, K.; Nagarale, R.K.; Sharma, A. Low voltage non-gassing electro-osmotic pump with zeta potential tuned aluminosilicate frits and organic dye electrodes. RSC Adv. 2014, 4, 28814–28821. [Google Scholar] [CrossRef]



| No. | Temperature (°C) | Flow Rate (mL/min) | Extraction Yield (µg/g Dry Sample) | ||
|---|---|---|---|---|---|
| Scopoletin | Alizarin | Rutin | |||
| 1 | 100 | 1 | 364.2 ± 11.7 | 15.0 ± 0.8 | 372.9 ± 11.7 |
| 2 | 2 | 435.3 ± 6.4 | 19.1 ± 0.3 | 429.6 ± 11.9 | |
| 3 | 3 | 444.8 ± 3.3 | 20.0 ± 0.6 | 437.3 ± 4.8 | |
| 4 | 120 | 1 | 382.1 ± 9.5 | 28.8 ± 1.1 | 375.5 ± 12.0 |
| 5 | 2 | 441.3 ± 4.9 | 34.5 ± 2.3 | 457.0 ± 15.7 | |
| 6 | 3 | 492.6 ± 11.9 | 34.7 ± 1.8 | 476.2 ± 16.6 | |
| 7 | 140 | 1 | 416.4 ± 3.2 | 64.0 ± 1.0 | 368.8 ± 2.6 |
| 8 | 2 | 493.3 ± 11.5 | 78.3 ± 2.8 | 385.2 ± 12.1 | |
| 9 | 3 | 530.6 ± 22.1 | 81.5 ± 0.8 | 452.0 ± 16.2 | |
| Temperature (°C) | Flow Rate (mL/min) | Partitioning Coefficients (KD) | ||
|---|---|---|---|---|
| Scopoletin | Alizarin | Rutin | ||
| 100 | 1 | 9.0 ± 0.6 a | 71.6 ± 2.7 a | 9.0 ± 0.5 a |
| 2 | 9.1 ± 0.5 a | 70.2 ± 1.9 a | 9.2 ± 0.4 a | |
| 3 | 9.3 ± 0.6 a | 74.2 ± 4.5 a | 9.3 ± 0.4 a | |
| 120 | 1 | 8.1 ± 0.4 b | 51.2 ± 2.5 b | 6.7 ± 0.2 c |
| 2 | 8.1 ± 0.4 b | 52.5 ± 2.4 b | 6.5 ± 0.4 c | |
| 3 | 7.5 ± 0.5 bc | 53.9 ± 2.5 b | 6.7 ± 0.2 c | |
| 140 | 1 | 7.2 ± 0.1 c | 9.6 ± 0.3 c | 7.8 ± 0.1 b |
| 2 | 7.5 ± 0.3 bc | 9.2 ± 0.8 c | 7.9 ± 0.6 b | |
| 3 | 7.7 ± 0.0 bc | 9.1 ± 0.5 c | 7.5 ± 0.3 b | |
| Temperature (°C) | Flow Rate (mL/min) | Diffusion Coefficient (×10−9 m2/s) | |
|---|---|---|---|
| Scopoletin | Alizarin | ||
| 100 | 1 | 0.51 ± 0.03 f | 0.09 ± 0.01 f |
| 2 | 0.71 ± 0.03 de | 0.14 ± 0.00 ef | |
| 3 | 0.96 ± 0.02 c | 0.19 ± 0.01 de | |
| 120 | 1 | 0.55 ± 0.02 ef | 0.18 ± 0.01 de |
| 2 | 0.86 ± 0.02 cd | 0.22 ± 0.02 d | |
| 3 | 1.20 ± 0.03 b | 0.25 ± 0.03 d | |
| 140 | 1 | 0.64 ± 0.01 ef | 0.61 ± 0.01 c |
| 2 | 1.14 ± 0.13 b | 1.02 ± 0.07 b | |
| 3 | 1.93 ± 0.26 a | 1.46 ± 0.11 a | |
| Flow Rate (mL/min) | Activation Energy (kJ/mol) | |
|---|---|---|
| Scopoletin | Alizarin | |
| 1 | 7.3 ± 1.3 b | 62.7 ± 2.0 a |
| 2 | 15.0 ± 4.9 ab | 63.6 ± 1.8 a |
| 3 | 22.0 ± 4.6 a | 64.5 ± 1.8 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jamaludin, R.; Kim, D.-S.; Salleh, L.M.; Lim, S.-B. Kinetic Study of Subcritical Water Extraction of Scopoletin, Alizarin, and Rutin from Morinda citrifolia. Foods 2021, 10, 2260. https://doi.org/10.3390/foods10102260
Jamaludin R, Kim D-S, Salleh LM, Lim S-B. Kinetic Study of Subcritical Water Extraction of Scopoletin, Alizarin, and Rutin from Morinda citrifolia. Foods. 2021; 10(10):2260. https://doi.org/10.3390/foods10102260
Chicago/Turabian StyleJamaludin, Roslina, Dong-Shin Kim, Liza Md Salleh, and Sang-Bin Lim. 2021. "Kinetic Study of Subcritical Water Extraction of Scopoletin, Alizarin, and Rutin from Morinda citrifolia" Foods 10, no. 10: 2260. https://doi.org/10.3390/foods10102260






