Moringa oleifera: An Unknown Crop in Developed Countries with Great Potential for Industry and Adapted to Climate Change
Abstract
1. Introduction
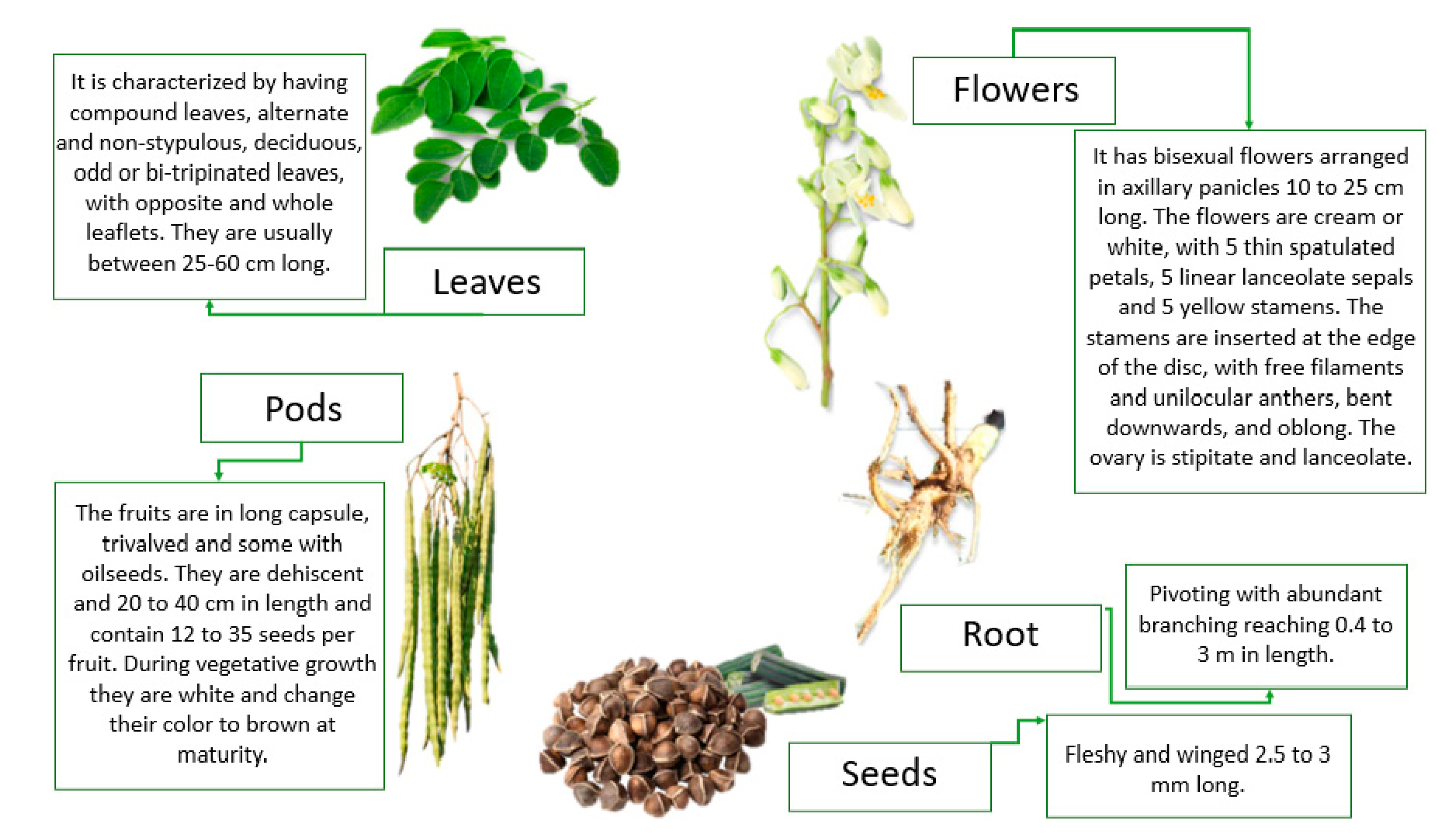
2. Botanical Description of the Crop and Its Growing Conditions
- Class Eudicotyledoneae
- Subclass Magnoliidae
- Clado Malvidae
- Order Brassicales
- Family Moringaceae
- Genus Moringa
- Species Moringa oleifera Lam
- -
- M. oleifera is especially sensitive to low temperatures. During the coldest months, it can withstand between −1 °C and 3 °C, tolerating short and low-intensity frosts. If the frost persists, the plant dies immediately. Consequently, low temperatures are considered to be the “exclusive” factor for the development of this plant. In the Iberian Peninsula and the Balearic Islands, December, January, and February are the months when the lowest temperatures are reached. The main mountain systems and most of the Douro Valley are unsuitable areas for the cultivation of the M. oleifera due to the low winter temperatures [24].
- -
- It does not survive temperatures above 48 °C [20]. No area in Spain reaches this average temperature during the summer months (June, July, and August).
- -
- If the average temperature exceeds 8 °C, the risk of light frost is low, so the plant could survive, although it would not begin growing [25].
- -
- The plant needs high daily average temperatures between 25–35 °C to have an optimal growth and a high production of pods and leaves, resulting in its most cost-effective cultivation [24].
- -
- For isohyets, the limit values are 300 mm and 500 mm [24].
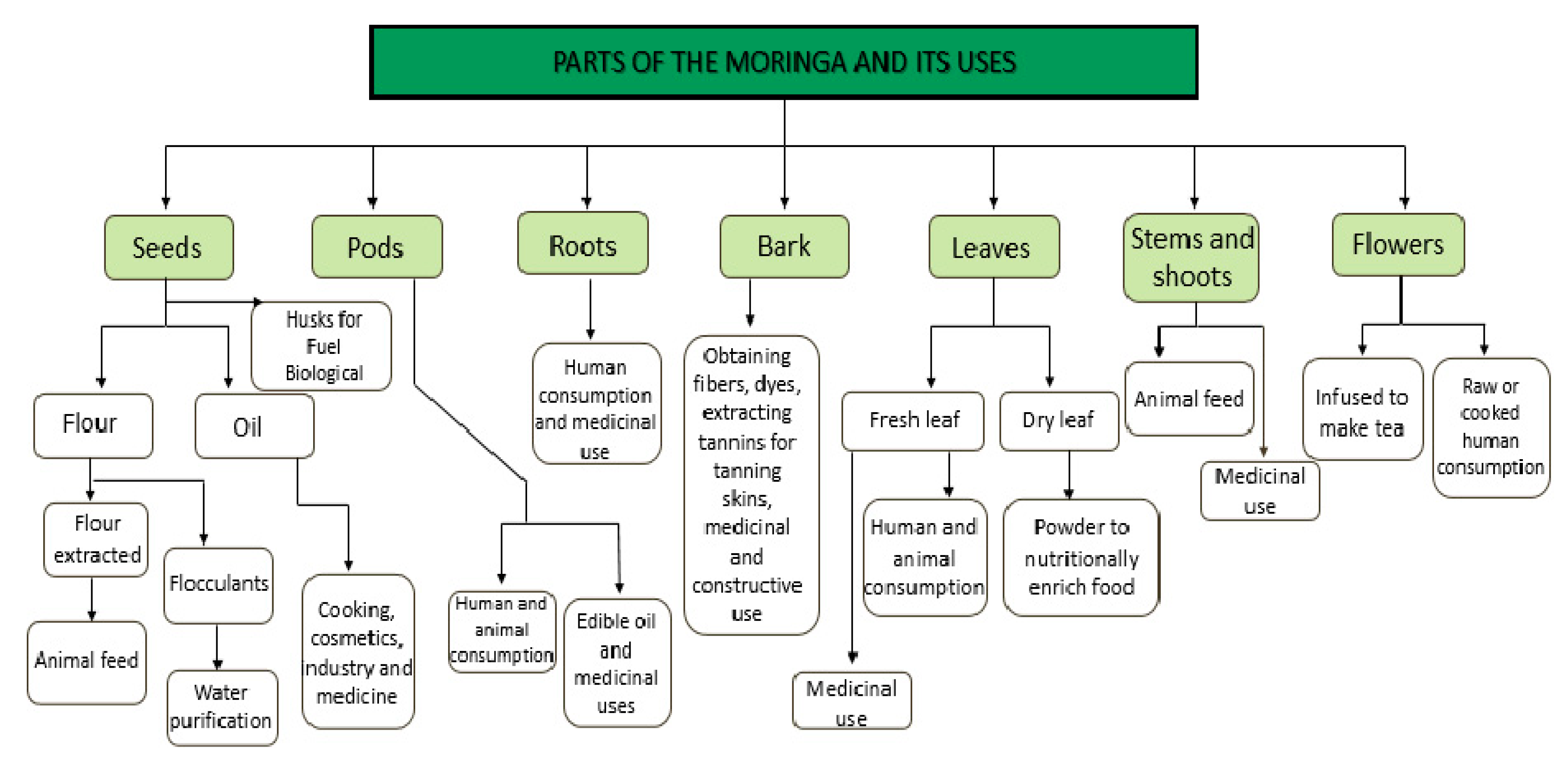
3. Parts of the M. oleifera and Their Composition
4. Uses of the Parts of the M. oleifera
4.1. Seeds
4.2. Pods
4.3. Root and Bark
4.4. Leaves
4.5. Stems, Shoots, and Flowers
5. Nutritional Aspects of Fresh and Dry Leaves of M. oleifera
6. Medicinal Properties
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Vaknin, Y.; Mishal, A. The potential of the tropical “miracle tree” Moringa oleifera and its desert relative Moringa peregrina as edible seed-oil and protein crops under Mediterranean conditions. Sci. Hortic. 2017, 225, 431–437. [Google Scholar] [CrossRef]
- Singh, A.K.; Rana, H.K.; Tshabalala, T.; Kumar, R.; Gupta, A.; Ndhlala, A.R.; Pandey, A.K. Phytochemical, nutraceutical and pharmacological attributes of a functional crop Moringa oleifera Lam: An overview. S. Afr. J. Bot. 2019, 129, 209–220. [Google Scholar] [CrossRef]
- Zainab, B.; Ayaz, Z.; Khan, S.; Rizwana, H.; Soliman, D.W.; Abbasi, A.M. In-silico elucidation of Moringa oleifera phytochemicals against diabetes mellitus. Saudi J. Biol. Sci. 2020, 27, 2299–2307. [Google Scholar] [CrossRef] [PubMed]
- Sujatha, B.K.; Patel, P. Moringa oleifera–Nature’s Gold. Imp. J. Interdiscip. Res. 2017, 3, 1175–1179. [Google Scholar]
- Němec, P.; Ungrová, M.; Alem, S.; Novák, J.; Habrová, H. Biomass production of a young plantation of Moringa stenopetala (Baker f.) Cufod. and Moringa oleifera Lam. in southern Ethiopia. S. Afr. J. Bot. 2020, 129, 463–470. [Google Scholar] [CrossRef]
- FAO FIDA Y PMA. El Estado de la Inseguridad Alimentaria en el Mundo 2012; Serie de la Organización de las Naciones Unidas Para la Alimentación y la Agricultura: Roma, Italy, 2012; p. 5. Available online: http://www.fao.org/3/i3027s/i3027s00.htm (accessed on 14 April 2020).
- Bancessi, A.; Bancessi, Q.; Baldé, A.; Catarino, L. Present and potential uses of Moringa oleifera as a multipurpose plant in Guinea-Bissau. S. Afr. J. Bot. 2020, 129, 206–208. [Google Scholar] [CrossRef]
- Solidaridad. Moringa Trees against Climate Change. Available online: https://www.solidaridadnetwork.org/news/Moringa-trees-against-climate-change (accessed on 7 December 2020).
- World Agroforestry (ICRAF). Addressing Climate Change and Food Security with the Moringa Tree. Available online: https://www.worldagroforestry.org/news/addressing-climate-change-and-food-security-Moringa-tree (accessed on 7 December 2020).
- Daba, M. Miracle tree: A review on multi-purposes of Moringa oleifera and its implication for climate change mitigation. J. Earth Sci. Clim. Chang. 2016, 7. [Google Scholar] [CrossRef]
- Villafuerte, L.R.; Villafurte-Abonal, L. Data Taken from the Forestry Agency of Japan in Moringa; Malunggay Phillippines, Apples of Gold Publishing: Singapore, 2009; p. 240. [Google Scholar]
- Manuwa, S.I.; Sedara, A.M.; Tola, F.A. Design, Fabrication and Performance Evaluation of Moringa (oleifera) Dried Leaves Pulverizer. J. Sci. Food Agric. 2020, 2, 100034. [Google Scholar] [CrossRef]
- Dev, S.R.S.; Geetha, P.; Orsat, V.; Gariépy, Y.; Raghavan, G.S.V. Effects of microwave-assisted hot air drying and conventional hot air drying on the drying kinetics, color, rehydration, and volatiles of Moringa oleifera. Dry. Technol. 2011, 29, 1452–1458. [Google Scholar] [CrossRef]
- Chase, M.W.; Christenhusz, M.J.M.; Fay, M.F.; Byng, J.W.; Judd, W.S.; Soltis, D.E.; Mabberley, D.J.; Sennikov, A.N.; Soltis, P.S.; Stevens, P.F. An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG IV. Bot. J. Linn. Soc. 2016, 181, 1–20. [Google Scholar]
- Olson, M.E.; Fahey, J.W. Moringa oleifera: Un árbol multiusos para las zonas tropicales secas. Rev. Mex. Biodivers. 2011, 82, 1071–1082. [Google Scholar] [CrossRef]
- Olson, M.E.; Palada, M.C.; Foidl, N.; Bates, R.M. Chapter 3: Botany and Propagation. In The Miracle Tree: Moringa oleifera, 2nd ed.; Xlibris: Bloomington, IN, USA, 2019. [Google Scholar]
- Chepote, M.A. Siembra del Cultivo de Moringa (Moringa oleífera) en la Pampa de Villacurí, Departamento de Ica 2018. Available online: http://190.119.243.88/bitstream/handle/UNALM/3223/F01-C44-T.pdf?sequence=4&isAllowed=y (accessed on 12 April 2020).
- Masih, L.P.; Singh, S.; Elamathi, S.; Anandhi, P.; Abraham, T. Moringa: A Multipurpose Potential Crop-A Review. Proc. Indian Natl. Sci. Acad. 2019, 85, 589–601. [Google Scholar] [CrossRef]
- James, A.; Zikankuba, V. Moringa oleifera a potential tree for nutrition security in sub-Sahara Africa. Am. J. Res. Commun. 2017, 5, 1–14. [Google Scholar]
- Palada, M.C.; Foidl, N.; Bates, R.M.; Ebert, A.W.; Amaglo, N. Chapter 5: Climate, Soil and Cultivation. In The Miracle Tree: Moringa oleifera, 2nd ed.; Xlibris: Bloomington, IN, USA, 2019. [Google Scholar]
- Nouman, W.; Basra, S.M.A.; Siddiqui, M.T.; Yasmeen, A.; Gull, T.; Alcayde, M.A.C. Potential of Moringa oleifera L. as livestock fodder crop: A review. Turk. J. Agric. For. 2014, 38, 1–14. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, X.Y.; Wei, X.M.; Gao, Z.T.; Han, J.P. Values, properties and utility of different parts of Moringa oleifera: An overview. Chin. Herb. Med. 2018, 10, 371–378. [Google Scholar] [CrossRef]
- Moringa oleífera: Árbol Multiusos de Interés Forestal Para el sur de la Península Ibérica 2016. Fichas de Transferencia Cajamar. Available online: https://www.cajamar.es/pdf/bd/agroalimentario/innovacion/investigacion/documentos-y-programas/020-Moringa-v3-1476963334.pdf (accessed on 11 March 2020).
- Godino, M.; Arias, C.; Izquierdo, M.I. Interés forestal de la Moringa oleifera y posibles zonas de implantación en España. In 6º Congreso Forestal Español: “Montes: Servicios y Desarrollo Rural”; Sociedad Española de Ciencias Forestales: Vitoria-Gasteiz, Spain, 2013; pp. 2–13. [Google Scholar]
- Balakumbahan, R.; Sathiyamurthy, V.A.; Janavi, G.J. Moringa Leaf–A Super Food. Biot. Res. Today 2020, 2, 438–440. [Google Scholar]
- Anwar, F.; Latif, S.; Ashraf, M.; Gilani, A.H. Moringa oleifera: A food plant with multiple medicinal uses. Phytother. Res. 2007, 21, 17–25. [Google Scholar] [CrossRef]
- Folorunso, A.E.; Akinwunmi, K.F.; Okonji, R.E. Comparative studies of the Biochemical parameters of the leaves and seeds of Moringa oleifera. J. Agric. Sci. Tech. B 2012, 2, 671–677. [Google Scholar]
- Taher, M.A.; Nyeem, M.A.B.; Ahammed, M.M.; Hossain, M.M.; Islam, M.N. Moringa oleifera (Shajna): The wonderful indigenous medicinal plant. Asian J. Med. Biol. Res. 2017, 3, 20–30. [Google Scholar] [CrossRef]
- Moringa—The Next Superfood? In Newsletter Moringa; University of California: Oakland, CA, USA, 2018; Available online: https://www.universityofcalifornia.edu/news/Moringa-next-superfood (accessed on 13 May 2020).
- Oyeyinka, A.; Oyeyinka, S. Moringa oleifera as a food fortificant: Recent trends and prospects. J. Saudi Soc. Agric. Sci. 2018, 17, 127–136. [Google Scholar] [CrossRef]
- Arias, C. Estudio de las Posibles Zonas de Introducción de la Moringa oleífera Lam. en la Península Ibérica, Islas Baleares e Islas Canarias; Universidad Politécnica de Madrid, Escuela Universitaria de Ingeniería Forestal: Madrid, Spain, 2014. [Google Scholar]
- Price, M.L. The Moringa tree. ECHO Tech. Note 2007, 17391, 1–19. [Google Scholar]
- Pontual, E.V.; Carvalho, B.E.; Bezerra, R.S.; Coelho, L.C.; Napoleão, T.H.; Paiva, P.M. Caseinolytic and milk-clotting activities from Moringa oleifera flowers. Food Chem. 2012, 135, 1848–1854. [Google Scholar] [CrossRef] [PubMed]
- Falowo, A.B.; Mukumbo, F.E.; Idamokoro, E.M.; Lorenzo, J.M.; Afolayan, A.J.; Muchenje, V. Multi-functional application of Moringa oleifera Lam. in nutrition and animal food products: A review. Food Res. Int. 2018, 106, 317–334. [Google Scholar] [CrossRef] [PubMed]
- Benítez, W.M. Aprovechamiento poscosecha de la Moringa (Moringa oleífera). Rev. Iberoam. Tecnol. Postcosecha 2012, 13, 171–174. [Google Scholar]
- Canett-Romero, R.; Arvayo-Mata, K.L.; Ruvalcaba-Garfias, N.V. Aspectos tóxicos más relevantes de Moringa oleifera y sus posibles daños. Biotecnia 2014, 16, 36–43. [Google Scholar] [CrossRef]
- Sandeep, G.; Anitha, T.; Vijayalatha, K.R.; Sadasakthi, A. Moringa for nutritional security (Moringa oleifera Lam.). Int. J. Bot. Stud. 2019, 4, 21–24. [Google Scholar]
- Toma, A.; Deyno, S. Phytochemistry and pharmacological activities of Moringa oleifera. Int. J. Pharmacol. 2014, 1, 222–231. [Google Scholar]
- Zaku, S.G.; Emmanuel, S.; Tukur, A.A.; Kabir, A. Moringa oleifera: An underutilized tree in Nigeria with amazing versatility: A review. Afr. J. Food Sci. 2015, 9, 456–461. [Google Scholar]
- Saa, R.W.; Fombang, E.N.; Ndjantou, E.B.; Njintang, N.Y. Treatments and uses of Moringa oleifera seeds in human nutrition: A review. Food Sci. Nutr. 2019, 7, 1911–1919. [Google Scholar] [CrossRef]
- Raman, J.K.; Alves, C.M.; Gnansounou, E. A review on Moringa tree and vetiver grass–Potential biorefinery feedstocks. Bioresour. Technol. 2018, 249, 1044–1051. [Google Scholar] [CrossRef]
- Fernandes, D.M.; Sousa, R.M.; de Oliveira, A.; Morais, S.A.; Richter, E.M.; Muñoz, R.A. Moringa oleifera: A potential source for production of biodiesel and antioxidant additives. Fuel 2015, 146, 75–80. [Google Scholar] [CrossRef]
- Foidl, N.; Makkar, H.P.S.; Becker, K. The potential of Moringa oleifera for agricultural and industrial uses. In The Miracle Tree: The Multiple Attributes of Moringa; CTA: Dar Es Salaam, Tanzania, 2001; pp. 45–76. [Google Scholar]
- Asensi, G.D. Moringa oleifera: Revisión sobre aplicaciones y usos en alimentos. Arch. Latinoam. Nutr. 2017, 67, 86–97. [Google Scholar]
- Gopalakrishnan, L.; Doriya, K.; Kumar, D.S. Moringa oleifera: A review on nutritive importance and its medicinal application. Food Sci. Hum. Wellness 2016, 5, 49–56. [Google Scholar] [CrossRef]
- Mahmood, K.T.; Mugal, T.; Haq, I.U. Moringa oleifera: A natural gift-A review. J. Pharm. Sci. Res. 2010, 2, 775. [Google Scholar]
- Paul, C.W.; Didia, B.C. The Effect of Methanolic Extract of Moringa oleifera Lam Roots on the Histology of Kidney and Liver of Guinea Pigs. Asian J. Med. Sci. 2012, 4, 55–66. [Google Scholar]
- Dhakar, R.C.; Maurya, S.D.; Pooniya, B.K.; Bairwa, N.; Gupta, M. Moringa: The herbal gold to combat malnutrition. Chron. Young Sci. 2011, 2, 119. [Google Scholar] [CrossRef]
- Brilhante, R.S.N.; Sales, J.A.; Pereira, V.S.; Castelo, D.D.S.C.M.; de Aguiar Cordeiro, R.; de Souza Sampaio, C.M.; Paiva, M.D.; dos Santos, J.B.; Sidrim, J.J.; Rocha, M.F.G. Research advances on the multiple uses of Moringa oleifera: A sustainable alternative for socially neglected population. Asian Pac. J. Trop. Med. 2017, 10, 621–630. [Google Scholar] [CrossRef]
- Quintanilla-Medina, J.; Garay-Martínez, J.; Alvarado-Ramirez, E.; Hernández-Meléndez, J.; Mendoza-Pedroza, S.; Rojas-Garcia, A.; Hernández-Garay, A. Tiempo y Temperatura sobre la pérdida de humedad y contenido de proteína en hojas de Moringa oleifera LAM. Agroproductividad 2018, 11, 88–92. [Google Scholar]
- Srinivasamurthy, S.; Yadav, U.; Sahay, S.; Singh, A. Development of muffin by incorporation of dried Moringa oleifera (Drumstick) leaf powder with enhanced micronutrient content. Int. J. Food Sci. Nutr. 2017, 2, 173–178. [Google Scholar]
- Makanjuola, B.A.; Obi, O.O.; Olorungbohunmi, T.O.; Morakinyo, O.A.; Oladele-Bukola, M.O.; Boladuro, B.A. Effect of Moringa oleifera leaf meal as a substitute for antibiotics on the performance and blood parameters of broiler chickens. Livest. Res. Rural Dev. 2014, 26, 144. [Google Scholar]
- Babiker, E.E.; Juhaimi, F.A.; Ghafoor, K.; Abdoun, K.A. Comparative study on feeding value of Moringa leaves as a partial replacement for alfalfa hay in ewes and goats. Livest. Sci. 2017, 195, 21–26. [Google Scholar] [CrossRef]
- Mukumbo, F.E.; Maphosa, V.; Hugo, A.; Nkukwana, T.T.; Mabusela, T.P.; Muchenje, V. Effect of Moringa oleifera leaf meal on finisher pig growth performance, meat quality, shelf life and fatty acid composition of pork. S. Afr. J. Anim. Sci. 2014, 44, 388–400. [Google Scholar] [CrossRef]
- Stohs, S.J.; Hartman, M.J. Review of the safety and efficacy of Moringa oleífera. Phytother. Res. 2015, 29, 796–804. [Google Scholar] [CrossRef] [PubMed]
- Bhargave, A.; Pandey, I.; Nama, K.S.; Pandey, M. Moringa oleifera Lam.—Sanjana (Horseradish Tree)—A miracle food plant with multipurpose uses in Rajasthan-India—An overview. Int. J. Pure Appl. Biosci. 2015, 3, 237–248. [Google Scholar]
- Amino-Acid Content of Foods and Biological Data on Proteins; Food and Nutrition Series nº 21–24; FAO: Roma, Italy, 1970; p. 19. Available online: http://www.fao.org/3/AC854T/AC854T43.htm (accessed on 11 March 2020).
- Xingú López, A.; González Huerta, A.; Cruz Torrez, E.D.L.; Sangerman-Jarquín, D.M.; Orozco de Rosas, G.; Rubí Arriaga, M. Chía (Salvia hispanica L.) situación actual y tendencias futuras. Rev. Mex. Cienc. Agríc. 2017, 8, 1619–1631. [Google Scholar] [CrossRef]
- Moyo, B.; Masika, P.J.; Hugo, A.; Muchenje, V. Nutritional characterization of Moringa (Moringa oleifera Lam.) leaves. Afr. J. Biotechnol. 2011, 10, 12925–12933. [Google Scholar]
- WHO. Grasas y Aceites en la Nutrición Humana; Estudio FAO Alimentación y Nutrición nº 57; FAO: Roma, Italy, 1997; p. 26. Available online: http://www.fao.org/3/V4700S/V4700S00.htm (accessed on 12 March 2020).
- Hayes, K.C. Dietary fat and heart health: In search of the ideal fat. Asia Pac. J. Clin. Nutr. 2002, 11, S394–S400. [Google Scholar] [CrossRef]
- Shahidi, F.; Ambigaipalan, P. Omega-3 polyunsaturated fatty acids and their health benefits. Annu. Rev. Food Sci. Technol. 2018, 9, 345–381. [Google Scholar] [CrossRef]
- Anta, O.; Rosa, M.; González-Rodríguez, L.G.; Villalobos Cruz, T.K.; Perea-Sánchez, J.M.; Aparicio Vizuete, A.; López-Sobaler, A.M. Fuentes alimentarias y adecuación de la ingesta de ácidos grasos omega-3 y omega-6 en una muestra representativa de adultos españoles. Nutr. Hosp. 2013, 28, 2236–2245. [Google Scholar]
- Sanhueza, J.; Durán, S.; Torres, J. Los ácidos grasos dietarios y su relación con la salud. Nutr. Hosp. 2015, 32, 1362–1375. [Google Scholar]
- Bernal-Estrada, J.; Cartagena-Valenzuela, J. Contenido de lípidos y composición relativa de los ácidos grasos en pulpa de aguacate ‘Hass’cosechados en el Trópico Andino del Departamento de Antioquia, Colombia. In Proceedings of the Memorias del V Congreso Latinoamericano del Aguacate, Ciudad Guzmán, Jalisco, Mexico, 4–7 September 2017. [Google Scholar]
- Ríos-Latorre, J.P.; Salazar Luna, D.E. Diseño y Construcción de un Equipo Para la Extracción de Aceite de Sésamo (Sesamum indicum) y Nuez (Juglans regia). Bachelor’s Thesis, Escuela Superior Politécnica de Chimborazo, Riobamba Canton, Ecuador, 2018. Available online: http://www.fao.org/3/V4700S/V4700S00.htm (accessed on 12 April 2020).
- Agudelo, L. Empleo del Polvo de Hojas de Moringa oleífera Lam Como Fortificante en un Alimento Enfocado a la Población Infantil Colombiana Menor de 4 Años. Ph.D. Thesis, Corporación Universitaria Lasallista, Caldas, Antioquia, Colombia, 2020. [Google Scholar]
- Pilay Malavé, M.V. Calidad Nutricional de la Moringa; Moringa oleifera Lam, en las Condiciones Ambientales de la Parroquia Manglaralto. Bachelor’s Thesis, Universidad Estatal Península de Santa Elena, La Libertad, Ecuador, 2019. [Google Scholar]
- García-Gabarra, A.; Castellà-Soley, M.; Calleja-Fernández, A. Ingestas de energía y nutrientes recomendadas en la Unión Europea: 2008–2016. Nutr. Hosp. 2017, 34, 490–498. [Google Scholar] [CrossRef] [PubMed]
- Villanueva-Flores, R.M. Fibra dietaria: Una alternativa para la alimentación. Ing. Ind. 2019, 37, 229–242. [Google Scholar] [CrossRef]
- Umar, S.A.; Mohammed, Z.; Nuhu, A.; Musa, K.Y.; Tanko, Y. Evaluation of Hypoglycaemic and Antioxidant Activity of Moringa oleifera Root in Normal and Alloxan-Induced Diabetic Rats. Trop. J. Nat. Prod. Res. 2018, 2, 401–408. [Google Scholar] [CrossRef]
- Batool, S.; Khan, S.; Basra, S.M.; Hussain, M.; Saddiq, M.S.; Iqbal, S.; Irshad, S.; Bashir, S.; Hafeez, M.B. Impact of natural and synthetic plant stimulants on Moringa seedlings grown under low-temperature conditions. Int. Lett. Nat. Sci. 2019, 76, 51. [Google Scholar] [CrossRef]
- Kumar, G.; Giri, A.; Arya, R.; Tyagi, R.; Mishra, S.; Mishra, A.K. Multifaceted applications of different parts of Moringa species: Review of present status and future potentials. Int. J. Chem. Stud. 2019, 7, 835–842. [Google Scholar]
- López-García, J.J. Moringa oleifera Lam.: Biología, Botánica, Propiedades Nutricionales y Medicinales; Depósito de Investigación; Universidad de Sevilla: Sevilla, Spain, 2016. [Google Scholar]
- Cáceres, A.; Saravia, A.; Rizzo, S.; Zabala, L.; De Leon, E.; Nave, F. Pharmacologie properties of Moringa oleifera. 2: Screening for antispasmodic, antiinflammatory and diuretic activity. J. Ethnopharmacol. 1992, 36, 233–237. [Google Scholar] [CrossRef]
- Padmarao, P.; Acharya, B.M.; Dennis, T.J. Pharmacognostic study on stembark of Moringa oleifera Lam. Bull. Med. Ethnobot. Res. 1996, 17, 141–151. [Google Scholar]
- Dahot, M.U. Vitamin contents of flowers and seeds of Moringa oleifera. Pak. J. Biochem. 1988, 21, 21–24. [Google Scholar]
- Dixit, S.; Tripathi, A.; Kumar, P. Medicinal properties of Moringa oleifera: A review. Int. J. Educ. Sci. Res. Rev. 2016, 3, 2–348. [Google Scholar]
- Pal, S.K.; Mukherjee, P.K.; Saha, B.P. Studies on the antiulcer activity of Moringa olifera leaf extract on gastric ulcer models in rats. Phytother. Res. 1995, 9, 463–465. [Google Scholar] [CrossRef]
- Jacques, A.S.; Arnaud, S.S.; Fréjus, O.O.; Jacques, D.T. Review on biological and immunomodulatory properties of Moringa oleifera in animal and human nutrition. J. Pharmacogn. Phytother. 2020, 12, 1–9. [Google Scholar]
| Content in (mg/100 g) | Fresh M. oleifera Leaves | Dry M. oleifera Leaves | Other Foods |
|---|---|---|---|
| Vitamin A | 7 | 18.9 | Carrot: 1.89 |
| Vitamin C | 220 | 17.3 | Orange: 30 |
| Calcium | 440 | 2003 | Cow’s milk: 120 |
| Iron | 085 | 28.2 | Spinach: 1.14 |
| Potassium | 259 | 1324 | Banana: 88 |
| Protein | 6700 | 27,100 | Yogurt: 3100 |
| Aminoacid Content (mg/100 g) | Dry M. oleifera Leaf | Soy | Beef |
|---|---|---|---|
| Arginine | 1325 | 380 | 1118 |
| Histidine *1 | 613 | 221 | 603 |
| Lysine * | 1325 | 233 | 1573 |
| Tryptophan * | 425 | 103 | --------- |
| Phenylalanine * | 1388 | 708 | 778 |
| Methionine * | 350 | 296 | 478 |
| Threonine * | 1188 | 328 | 812 |
| Leucine * | 1950 | 1764 | 1435 |
| Isoleucine * | 825 | 803 | 852 |
| Valine * | 1063 | 728 | 886 |
| Minerals (mg/100 g) | Fresh M. oleifera Leaf | Spinach | Dry M. oleifera Leaf | Chia |
|---|---|---|---|---|
| Calcium | 440 | 117 | 2003 | 631 |
| Iron | 0.85 | 2.7 | 28.2 | 0.01 |
| Copper | 0.07 | ----- | 0.57 | ----- |
| Magnesium | 42 | ----- | 368 | 335 |
| Phosphorus | 70 | 46 | 204 | 860 |
| Potassium | 259 | 554 | 1324 | 407 |
| Zinc | 0.16 | ----- | 3.29 | ----- |
| Fatty Acids (%) | Dried M. oleifera Leaves | Avocado | Walnut |
|---|---|---|---|
| Caprylic | 0.07 | ----- | 2.28 |
| Lauric | 0.58 | ----- | ----- |
| Myristic | 3.66 | 0.12–0.13 | ----- |
| Palmitic | 11.79 | 19.7 | 11.56 |
| Palmitoleic | 0.17 | 13.49 | ----- |
| Margaric | 3.19 | ----- | ----- |
| Stearic | 2.13 | 1.51–1.52 | 1.14 |
| Oleic | 3.96 | 49.55 | 11.37 |
| Linoleic | 7.44 | 14.01 | 18.91 |
| Linolenic | 44.57 | 1.26 | 3.41 |
| Total de Omega-6 | 7.64 | 14.01 | 18.91 |
| Total de Omega-3 | 44.57 | 1.26 | 3.41 |
| MUFA (total monounsaturated fatty acids) | 4.48 | 63.53 | 14.2 |
| PUFA (total polyunsaturated fatty acids) | 52.21 | 15.27 | 23.2 |
| Omega-6/Omega-3 Ratio | 0.17 | 11.12 | 5.54 |
| MUFA:PUFA Relationship | 0.09 | 4.16 | 0.61 |
| Nutritional Analysis | Fresh M. oleifera Leaves (per 100 g) | Dry M. oleifera Leaves (per 100 g) |
|---|---|---|
| Humidity (%) | 75–79.7 | 4.8–7.5 |
| Calories (Kcal) | 49.5–92 | 205–329 |
| Proteins (g) | 5.5–9.4 | 27.1–33.5 |
| Fats (g) | 1.4–2.1 | 2.3–9.75 |
| Carbohydrates (g) | 8.633–13.4 | 38.2–41.2 |
| Fiber (g) | 0.9 | 7.48–30.97 |
| Vitamin B1 (mg) | 0.06 | 2.02–2.64 |
| Vitamin B2 (mg) | 0.05 | 20.5–21.3 |
| Vitamin B3 (mg) | 0.8 | 7.6–8.2 |
| Vitamin E (mg) | 448 | 108–113 |
| Vitamin A (mg) | 7 | 17.3–91.8 |
| Vitamin C (mg) | 51.7–220 | 15.8–17.3 |
| Part of the Plant | Medicinal Use | References |
|---|---|---|
| Roots | Analgesic, anti-inflammatory, antitumor, antidiabetic, snake bite, antiulcer, antispasmodic, cholesterol-lowering effect, antibacterial, antiurolytic, antifungal, antidiuretic and antihypertensive. | [22,74,75,76,77,78] |
| Leaves | Anticatarrhal, antidiabetic, antiscurb, antihypertensive, antiproliferative, antioxidant, anxiolytic, diuretic, pharyngitis, cholesterol-lowering effect, hemorrhoids, glandular swellings, anti-inflammatory and anti-hyperthyroidism. | [22,74,78,79] |
| Flowers | Anti-inflammatory, antipsychotic and anti-tumor. | [22,74,78] |
| Seeds | Antidiuretic, antitumor, genitourinary, antituberculous, anti-asmatic, antibacterial and hepatoprotective. | [22,32,74,75,76] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trigo, C.; Castelló, M.L.; Ortolá, M.D.; García-Mares, F.J.; Desamparados Soriano, M. Moringa oleifera: An Unknown Crop in Developed Countries with Great Potential for Industry and Adapted to Climate Change. Foods 2021, 10, 31. https://doi.org/10.3390/foods10010031
Trigo C, Castelló ML, Ortolá MD, García-Mares FJ, Desamparados Soriano M. Moringa oleifera: An Unknown Crop in Developed Countries with Great Potential for Industry and Adapted to Climate Change. Foods. 2021; 10(1):31. https://doi.org/10.3390/foods10010031
Chicago/Turabian StyleTrigo, Carla, María Luisa Castelló, María Dolores Ortolá, Francisco José García-Mares, and María Desamparados Soriano. 2021. "Moringa oleifera: An Unknown Crop in Developed Countries with Great Potential for Industry and Adapted to Climate Change" Foods 10, no. 1: 31. https://doi.org/10.3390/foods10010031
APA StyleTrigo, C., Castelló, M. L., Ortolá, M. D., García-Mares, F. J., & Desamparados Soriano, M. (2021). Moringa oleifera: An Unknown Crop in Developed Countries with Great Potential for Industry and Adapted to Climate Change. Foods, 10(1), 31. https://doi.org/10.3390/foods10010031