The Disease Process, Diagnosis and Treatment of Invasive Cervical Resorption: A Review
Abstract
1. Introduction
2. Literature Search and Scope of Review
3. Putative Mechanisms of the Disease Process of ICR
4. Predisposing Factors
5. Differences in the Pattern of ICR between Vital Teeth and Endodontically Treated Teeth
- Intensity of resorptionMore extensive resorption is observed in endodontically treated teeth than in vital teeth. This may be because, during endodontic therapy, part of PRRS is damaged mechanically or altered chemically [16]. Moreover, loss of pulp vitality in endodontically treated teeth may create hypoxic microenvironments conducive to continued osteoclastic activities [16,17];
- Presence of osseous tissueIngrowth of osseous tissue in the resorptive defect is more often observed in vital teeth than in endodontically treated teeth [16,17]. Hypoxic conditions induced by pulp tissue extirpation may contribute to the continued growth of granulomatous tissue formation by attenuating osteoblast growth and differentiation [40,41], and stimulating osteoclastic activities [37]. On the other hand, vital teeth maintain normoxic microenvironments around PRRS and allow for the osseous replacement of granulomatous tissue in the resorptive site during normal bone remodeling at the late phase of ICR [17]. Osseous tissue substituted for and integrated into part of PRRS may also function as the protective layer [17].
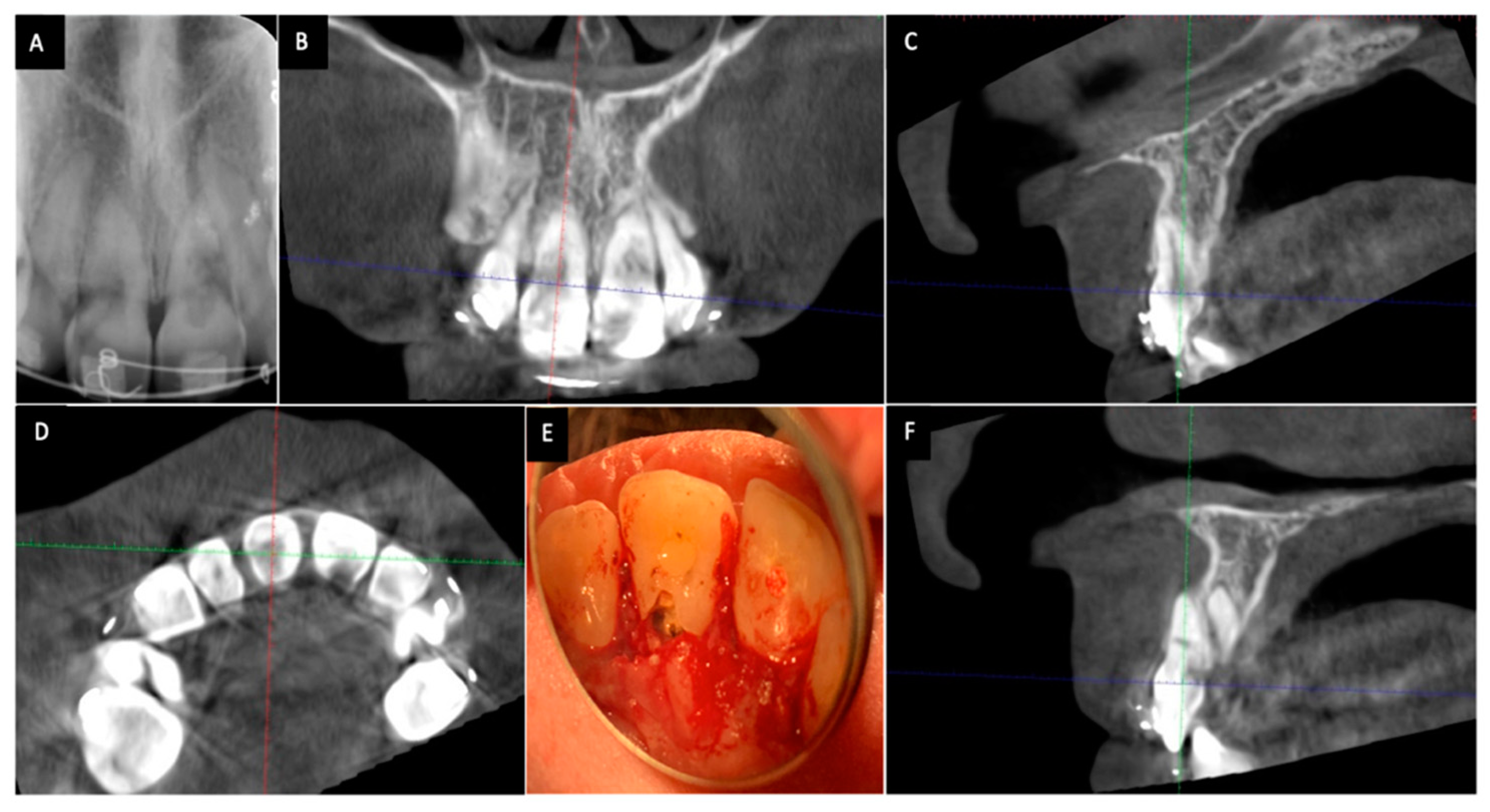
6. Diagnosis and Assessment of ICR
7. Classification of ICR
8. Strategies to Manage ICR Lesions
8.1. External Approach
8.2. Internal Approach
9. Prognosis
10. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Heithersay, G.S. Invasive cervical resorption: An analysis of potential predisposing factors. Quintessence Int. 1999, 30, 83–95. [Google Scholar] [PubMed]
- Gulsahi, A. Clinical and radiographic appearances of invasive cervical resorption. J. Oral Health Den. Manag. 2014, 13, 934–939. [Google Scholar]
- Irinakis, E.; Aleksejuniene, J.; Shen, Y.; Haapasalo, M. External cervical resorption—A retrospective case-control study. J. Endod. 2020. [Google Scholar] [CrossRef] [PubMed]
- Heithersay, G.S. Invasive cervical resorption. Endod. Top. 2004, 7, 73–92. [Google Scholar] [CrossRef]
- Frank, A.L. External–internal progressive resorption and its non-surgical correction. J. Endod. 1981, 7, 473–476. [Google Scholar] [CrossRef]
- Frank, A.L.; Blakland, L.K. Supra osseous extra-canal invasive resorption. J. Endod. 1987, 13, 348–387. [Google Scholar] [CrossRef]
- Patel, S.; Kanagasingam, S.; Pitt Ford, T. External cervical resorption: A review. J. Endod. 2009, 35, 616–625. [Google Scholar] [CrossRef]
- Gold, S.I.; Hasselgren, G. Peripheral inflammatory root resorption. A review of the literature with case reports. J. Clin. Periodontol. 1992, 19, 523–534. [Google Scholar] [CrossRef]
- Trope, M. Root resorption of dental and traumatic origin: Classification based on Etiology. Pr. Periodont. Aesthet. Dent. 1998, 10, 515–522. [Google Scholar]
- Fish, E.W. Begnign neoplasia of tooth and bone. Proc. R. Soc. Med. 1941, 34, 427–432. [Google Scholar]
- Parirokh, M.; Torabinejad, M. Mineral trioxide aggregate: A comprehensive literature review—Part III: Clinical applications, drawbacks, and mechanism of action. J. Endod. 2010, 36, 400–413. [Google Scholar] [CrossRef] [PubMed]
- Hakki, S.S.; Bozkurt, S.B.; Hakki, E.E.; Belli, S. Effects of mineral trioxide aggregate on cell survival, gene expression associated with mineralized tissues, and biomineralization of cementoblasts. J. Endod. 2009, 35, 513–519. [Google Scholar] [CrossRef]
- Prati, C.; Gandolfi, M.G. Calcium silicate bioactive cements: Biological perspectives and clinical applications. Dent. Mater. 2015, 31, 351–370. [Google Scholar] [CrossRef] [PubMed]
- Lovato, K.F.; Sedgley, C.M. Antibacterial activity of endosequence root repair material and proroot MTA against clinical isolates of Enterococcus faecalis. J. Endod. 2011, 37, 1542–1546. [Google Scholar] [CrossRef] [PubMed]
- Sipert, C.R.; Hussne, R.P.; Nishiyama, C.K.; Torres, S.A. In vitro antimicrobial activity of Fill Canal, Sealapex, Mineral Trioxide Aggregate, Portland cement and EndoRez. Int. Endod. J. 2005, 38, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Mavridou, A.M.; Hauben, E.; Wevers, M.; Schepers, E.; Bergmans, L.; Lambrechts, P. Understanding external cervical resorption patterns in endodontically treated teeth. Int. Endod. J. 2017, 50, 1116–1133. [Google Scholar] [CrossRef] [PubMed]
- Mavridou, A.M.; Hauben, E.; Wevers, M.; Schepers, E.; Bergmans, L.; Lambrechts, P. Understanding external cervical resorption in vital teeth. J. Endod. 2016, 42, 1737–1751. [Google Scholar] [CrossRef]
- Heithersay, G.S. Invasive cervical resorption following trauma. Aust. Endod. J. 1999, 25, 79–85. [Google Scholar] [CrossRef]
- Jeng, P.Y.; Lin, L.D.; Chang, S.H.; Lee, Y.L.; Wang, C.Y.; Jeng, J.H.; Tsai, Y.L. Invasive cervical resorption-distribution, potential predisposing factors, and clinical characteristics. J. Endod. 2020, 46, 475–482. [Google Scholar] [CrossRef]
- O’Mahony, A.; McNamara, C.; Ireland, A.; Sandy, J.; Puryer, J. Invasive cervical resorption and the oro-facial cleft patient: A review and case series. Br. Dent. J. 2017, 222, 677–681. [Google Scholar] [CrossRef][Green Version]
- Lo Giudice, G.; Matarese, G.; Lizio, A.; Lo Giudice, R.; Tumedei, M.; Zizzari, V.L.; Tete, S. Invasive cervical resorption: A case series with 3-year follow-up. Int. J. Periodontics Restor. Dent. 2016, 36, 103–109. [Google Scholar] [CrossRef][Green Version]
- Kim, Y.; Lee, C.Y.; Kim, E.; Roh, B.D. Invasive cervical resorption: Treatment challenges. Restor. Dent. Endod. 2012, 37, 228–231. [Google Scholar] [CrossRef] [PubMed]
- Discacciati, J.A.; de Souza, E.L.; Costa, S.C.; Sander, H.H.; Barros, V.M.; Vasconcellos, W.A. Invasive cervical resorption: Etiology, diagnosis, classification and treatment. J. Contemp. Dent. Pr. 2012, 13, 723–728. [Google Scholar] [CrossRef] [PubMed]
- Madison, S.; Walton, R. Cervical root resorption following bleaching of endodontically treated teeth. J. Endod. 1990, 16, 570–574. [Google Scholar] [CrossRef]
- Friedman, S.; Rotstein, I.; Libfeld, H.; Stabholz, A.; Heling, I. Incidence of external root resorption and esthetic restuls in 58 bleached pulpless teeth. Endod. Dent. Traumatol. 1988, 4, 23–26. [Google Scholar] [CrossRef] [PubMed]
- Heithersay, G.S.; Dahlstrom, S.W.; Marin, P.D. Incidence of invasive cervical resorption in bleached root-filled teeth. Aust. Dent. J. 1994, 39, 82–87. [Google Scholar] [CrossRef]
- Rotstein, I.; Friedman, S.; Mor, C.; Katznelson, J.; Sommer, M.; Bab, I. Histological characterization of bleaching-induced external root resorption in dogs. J. Endod. 1991, 17, 436–441. [Google Scholar] [CrossRef]
- Neuvald, L.; Consolaro, A. Cementoenamel junction; microscopic analysis and external cervical resorption. J. Endod. 2000, 26, 503–508. [Google Scholar] [CrossRef]
- Schroeder, H.E.; Scherle, W.F. Cemento-enamel junction—Revisted. J. Periodontal. Res. 1988, 23, 53–59. [Google Scholar] [CrossRef]
- Arroyo-Bote, S.; Bucchi, C.; Manzanares, M.C. External cervical resorption: A new oral manifestation of systemic sclerosis. J. Endod. 2017, 43, 1740–1743. [Google Scholar] [CrossRef]
- Liena-Puy, M.C.; Amengual-Lorenzo, J.; Forner-Navarro, L. Idiopathic external root resorption associated to hypercalciuria. Med. Oral. 2002, 7, 192–199. [Google Scholar]
- Von Arx, T.; Schawalder, P.; Ackermann, M.; Bosshardt, D.D. Human and feline invasive cervical resorption: The missing link?-Presentation of four cases. J. Endod. 2009, 35, 904–913. [Google Scholar] [CrossRef]
- Patel, K.; Schirru, E.; Niazi, S.; Mitchell, P.; Mannocci, F. Multiple apical radiolucencies and external cervical resorption associated with varicella zoster virus: A case report. J. Endod. 2016, 42, 978–983. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Chawla, A.; Kaur, A. Multiple idiopathic cervical root resorptions in patients with hepatitis B virus infection. J. Endod. 2018, 44, 1575–1577. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Saberi, N. External cervical resorption associated with the use of biosphosphonates: A case series. J. Endod. 2015, 742–748. [Google Scholar] [CrossRef] [PubMed]
- Mavridou, A.; Bergmans, L.; Barendregt, D.; Lambrechts, P. Descriptive analysis of factors associated with external cervical resorption. J. Endod. 2017, 43, 1602–1610. [Google Scholar] [CrossRef] [PubMed]
- Arnett, T.R.; Gibbons, D.C.; Utting, J.C.; Orriss, I.R.; Hoebertz, A.; Rosendaal, M.; Meghji, S. Hypoxia is a major stimulator of osteoclast formation and bone resorption. J. Cell. Physiol. 2003, 196, 2–8. [Google Scholar] [CrossRef]
- Mavridou, A.M.; Hikens, P.; Lambrichts, I.; Hauben, E.; Wevers, M.; Bergmans, L.; Lambrechts, P. Is hypoxia related to external cervical resorption? A case report. J. Endod. 2019, 45, 459–470. [Google Scholar] [CrossRef]
- Patel, S.; Mavridou, A.M.; Lambrechts, P.; Saberi, N. External cervical resorption—Part 1: Histopathology, distribution and presentation. Int. Endod. J. 2018, 51, 1205–1223. [Google Scholar] [CrossRef]
- Utting, J.C.; Robins, S.P.; Brandao-Burch, A.; Orriss, I.R.; Behar, J.; Arnett, T.R. Hypoxia inhibits the growth, differentiation and bone-forming capacity of rat osteoblasts. Exp. Cell. Res. 2006, 312, 1693–1702. [Google Scholar] [CrossRef]
- Arnett, T.R. Acidosis, hypoxia and bone. Arch. Biochem. Biophys. 2010, 503, 103–109. [Google Scholar] [CrossRef]
- Gunst, V.; Mavridou, A.; Huybrechts, B.; Van Gorp, G.; Bergmans, L.; Lambrechts, P. External cervical resorption: An analysis using cone beam and microfocus computed tomography and scanning electron microscopy. Int. Endod. J. 2013, 46, 877–887. [Google Scholar] [CrossRef]
- Patel, K.; Mannocci, F.; Patel, S. The assessment and management of external cervical resorption with periapical radiographs and cone-beam computed tomography: A clinical study. J. Endod. 2016, 42, 1435–1440. [Google Scholar] [CrossRef] [PubMed]
- Creanga, A.G.; Geha, H.; Sankar, V.; Teixeira, F.B.; McMahan, C.A.; Noujeim, M. Accuracy of digital periapical radiography and cone-beam computed tomography in detecting external root resorption. Imaging Sci. Dent. 2015, 45, 153–158. [Google Scholar] [CrossRef] [PubMed][Green Version]
- D’Addazio, P.; Campos, C.; Ozcan, M.; Teixeira, H.G.C.; Passoni, R.M.; Carvalho, A.C.P. A comparative study between cone-beam computed tomography and periapical radiographs in the diagnosis of simulated endodontic complications. Int. Endod. J. 2011, 44, 218–224. [Google Scholar] [CrossRef]
- Deliga Schroder, A.G.; Westphalen, F.H.; Schroder, J.C.; Fernandes, A.; Westphalen, V.P.D. Accuracy of digital periapical radiography and cone-beam computed tomography for diagnosis of natural and simulated external root resorption. J. Endod. 2018, 44, 1151–1158. [Google Scholar] [CrossRef]
- AAE/AAOMR Joint Position Statement. Available online: https://f3f142zs0k2w1kg84k5p9i1o-wpengine.netdna-ssl.com/specialty/wp-content/uploads/sites/2/2017/06/conebeamstatement.pdf (accessed on 1 June 2020).
- Patel, S.; Brown, J.; Semper, M.; Abella, F.; Mannocci, F. European Society of Endodontology position statement: Use of cone beam computed tomography in endodontics: European Society of Endodontology (ESE) developed by. Int. Endod. J. 2019, 52, 1675–1678. [Google Scholar] [CrossRef] [PubMed]
- Vaz de Souza, D.; Schirru, E.; Mannocci, F.; Foschi, F.; Patel, S. External cervical resorption: A comparison of the diagnostic efficacy using 2 different cone-beam computed tomographic units and periapical radiographs. J. Endod. 2017, 43, 121–125. [Google Scholar] [CrossRef]
- Patel, S.; Forschi, F.; Mannocci, F.; Patel, K. External cervical resorption: A three-dimensional classification. Int. Endod. J. 2018, 51, 206–214. [Google Scholar] [CrossRef]
- Heithersay, G.S. Treatment of invasive cervical resorption: Analysis of results using topical application of trichloracetic acid, curettage, and restoration. Quintessence Int. 1999, 30, 96–110. [Google Scholar]
- Karunakar, P.; Soloman, R.V.; Anusha, B.; Nagarjun, M. Endodontic management of invasive cervical resorption: Report of two cases. J. Coserv. Dent. 2018, 21, 578–681. [Google Scholar]
- Tsaousoglou, P.; Markou, E.; Efthimiades, N.; Vouros, I. Characteristics and treatment of invasive cervical resorption in vital teeth. A narrative review and a report of two cases. Br. Dent. J. 2017, 222, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Heithersay, G.S.; Wilson, D.F. Tissue responses in the rat to trichloracetic acid-an agent used in the treatment of invasive cervical resorption. Aust. Dent. J. 1988, 33, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, R.S.; Robbins, J.W.; Rindler, E. Management of invasive cervical resorption: Observations from three private practices and a report of three cases. J. Endod. 2010, 36, 1721–1730. [Google Scholar] [PubMed]
- Tantbirojn, D.; Feigal, R.J.; Ko, C.C.; Versluis, A. Remineralized dentin lesions induced by glass ionomer demonstrate increased resistance to subsequent acid challenge. Quintessence Int. 2006, 37, 273–281. [Google Scholar]
- Hasan, A.M.H.R.; Sidhu, S.K.; Nicholson, J.W. Fluoride release and uptake in enhanced bioactivity glass ionomer cememt (“glass carbomer™”) compared with conventional and resin-modified glass ionomer cements. J. Appl. Oral Sci. 2019, 27, e20180230. [Google Scholar]
- Sidhu, S.K.; Nicholson, J.W. A review of glass-ionomer cements for clinical dentistry. J. Funct. Biomater. 2016, 7, 16. [Google Scholar]
- Santamaria, M.P.; da Silva Feitosa, D.; Nociti, F.H.; Casati, M.Z.; Sallum, A.W.; Sallum, E.A. Cervical restoration and amount of soft tissue coverage achieved by coronally advanced flap. A 2-year-follow-up randomized controlled clinical trial. J. Clin. Periodontol. 2009, 36, 434–441. [Google Scholar] [CrossRef]
- Santamaria, M.P.; Ambrosano, G.M.; Casati, M.Z.; Nociti, F.H.; Sallum, A.W., Jr.; Sallum, E.A. Connective tissue graft plus resin-modified glass ionomer restoration for the treatment of gingival recession associated with non-carious cervical lesion: A randomized-controlled clinical trial. J. Clin. Periodontol. 2009, 36, 791–798. [Google Scholar] [CrossRef]
- Ikhar, A.; Thakur, N.; Patel, A.; Bhede, R.; Ptail, P.; Gupta, S. Management of external invasive cervical resorption tooth with mineral trioxide aggregate: A case report. Case. Rep. Med. 2013, 2013, 139801. [Google Scholar] [CrossRef]
- Eftekhar, L.; Ashraf, H.; Jabbari, S. Management of invasive cervical resorption lesions in a mandibular canine using Biodentine as a restorative material: A case report. Iran. Endod. J. 2017, 12, 386–389. [Google Scholar]
- Karypidou, A.; Chatzinkikolaou, I.D.; Kouros, P.; Koulaouzidou, E.; Econonomides, N. Management of bilateral invasive cervical resorption lesions in maxillary incirosrs using a novel calcium-silicate-based cement: A case report. Quintessence Int. 2016, 47, 637–642. [Google Scholar]
- Kumar, S.S.; Kumar, N.S.; Karunakaran, J.V.; Nagendran, S. Management of invasive cervical resorption in a maxillary central incisor. J. Pharm. Bioallied. Sci. 2015, 7, S712–S717. [Google Scholar] [CrossRef]
- Baranwal, A.K. Management of external invasive cervical resorption of tooth with Biodentine: A case report. J. Conserv. Dent. 2016, 19, 296–299. [Google Scholar] [CrossRef]
- Grech, L.; Mallia, B.; Camilleri, J. Invesitigation of the physical properties of tricalcium silicate cement-based root-end filling materials. Dent. Mater. 2013, 29, e20–e28. [Google Scholar] [CrossRef]
- Guo, Y.-J.; Du, T.-F.; Li, H.-B.; Shen, Y.; Mobuchon, C.; Hieawy, A.; Wang, Z.-J.; Yang, Y.; Ma, J.; Haapasalo, M. Physical properties and hydration behavior of a fast-setting bioceramic endodontic material. BMC Oral Health 2016, 16, 23. [Google Scholar] [CrossRef]
- Malkondu, O.; Karapinar Kaandag, M.; Kazazoglu, E. A review on biodentine, a contemporary dentine replacement and repair material. Biomed. Res. Int. 2014, 2014, 160951. [Google Scholar] [CrossRef] [PubMed]
- Camilleri, J. Investigation of Biodentine as dentine replacement material. J. Dent. 2013, 41, 600–610. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, V.; Singla, M.; Miglani, S.; Kohli, S. Comparative evaluation of push-out bond strength of ProRoot MTA, Biodentine, and MTA plus in furcation perforation repair. J. Conserv. Dent. 2013, 16, 462–465. [Google Scholar] [CrossRef] [PubMed]
- Guneser, M.B.; Akbulut, M.B.; Eldeniz, A.U. Effect of various endodontic irrigants on the push-out bond strength of Biodentine and conventional root perforation repair materials. J. Endod. 2013, 39, 380–384. [Google Scholar] [CrossRef]
- Gjorgievska, E.S.; Nicholson, J.W.; Apostolska, S.M.; Coleman, N.J.; Booth, S.E.; Slipper, I.J.; Miadenov, M.I. Interfacial properties of three different bioactive dentine substitutes. Microsc. Microanal. 2013, 19, 1450–1457. [Google Scholar] [CrossRef] [PubMed]
- Valles, M.; Roig, M.; Duran-Sindreu, F.; Martinez, S.; Mercade, M. Color stability of teeth restored with Biodentine: A 6-month in vitro study. J. Endod. 2015, 41, 1157–1160. [Google Scholar] [CrossRef] [PubMed]
- Kaur, M.; Singh, H.; Dhillon, J.S.; Batra, M.; Saini, M. MTA Versus Biodentine: Review of Literature With a Comparative Analysis. J. Clin. Diagn. Res. 2017, 11, ZG01–ZG05. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Shen, Y.; Stojicic, S.; Haapasalo, M. Biocompatibility of two novel root repair materials. J. Endod. 2011, 37, 793–798. [Google Scholar] [CrossRef]
- Smidt, A.; Nuni, E.; Keinan, D. Invasive cervical root resorption: Treatment rationale with an interdisciplinary approach. J. Endod. 2007, 33, 1383–1387. [Google Scholar] [CrossRef]
- Espona, J.; Roig, E.; Duran-Sindreu, F.; Abella, F.; Machado, M.; Roig, M. Invasive Cervical resorption: Clinical management in the anterior zone. J. Endod. 2018, 44, 1749–1754. [Google Scholar] [CrossRef]
- Salzano, S.; Tirone, F. Conservative nonsurgical treatment of class 4 invasive cervical resorption: A case series. J. Endod. 2015, 41, 1907–1912. [Google Scholar] [CrossRef]
- Baratto-Filho, F.; Limongi, O.; Araujo, C.D.J.R.; Neto, M.D.; Maia, S.M.; Santana, D. Treatment of invasive cervical resorption with MTA: Case report. Aust. Endod. J. 2005, 31, 76–80. [Google Scholar] [CrossRef]
- Park, J.B.; Lee, J.H. Use of mineral trioxide aggregrate in the non-surgical repair of perforating invasive cervical resorption. Med. Oral Patol. Oral Cir. Bucal. 2008, 1, E678–E680. [Google Scholar]
- Pace, R.; Giuliani, V.; Pagavino, G. Mineral trioxide aggregate in the treatment of external invasive resorption: A case report. Int. Endod. J. 2008, 41, 258–266. [Google Scholar] [CrossRef]
- Shemesh, A.; Ben Itzhak, J.; Solomonov, M. Minimally invasive treatment of class 4 invasive cervical resorption with internal approach: A case series. J. Endod. 2017, 43, 1901–1908. [Google Scholar] [CrossRef] [PubMed]
- Asgary, S.; Nosrat, A. Conservative management of class 4 invasive cervical root resorption using calcium-enriched mixture cement. J. Endod. 2016, 42, 1291–1294. [Google Scholar] [CrossRef] [PubMed]
- Patni, P.M.; Jain, P.; Jain, S.; Hiremath, H.; Agarwal, R.; Patni, M.J. Internal tunneling resorption associated with invasive cervical resorption. J. Conserv. Dent. 2018, 21, 105–108. [Google Scholar] [PubMed]
- Asgary, S.; Nourzadeh, M.; Verma, P.; Lamar Hicks, M.; Nosrat, A. Vital pulp therapy as a conservative approach for management of invasive cervical resorption: A case series. J. Endod. 2019, 45, 1161–1167. [Google Scholar] [CrossRef]
- Pierce, A. Experimental basis for the management of dental resorption. Endod. Dent. Traumatol. 1989, 5, 255–265. [Google Scholar] [CrossRef]
| Materials | Chemical Composition |
|---|---|
| ProRootMTA [74] | Tricalcium silicate, Dicalcium silicate, Tricalcium aluminate, Tetracalciumaluminoferrite, Calcium sulfate, Bismuth oxide, Calcium oxide, Silicon oxide, Aluminum oxide |
| Biodentine [74] | Tricalcium silicate, Dicalcium silicate, Calcium Carbonate, zirconium oxide, Iron oxide |
| Endosequence Root Repair Material [75] | Calcium silicate, Calcium phosphate monobasic, Zirconium oxide, Tantalum Oxide, filler and thickening agents |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rotondi, O.; Waldon, P.; Kim, S.G. The Disease Process, Diagnosis and Treatment of Invasive Cervical Resorption: A Review. Dent. J. 2020, 8, 64. https://doi.org/10.3390/dj8030064
Rotondi O, Waldon P, Kim SG. The Disease Process, Diagnosis and Treatment of Invasive Cervical Resorption: A Review. Dentistry Journal. 2020; 8(3):64. https://doi.org/10.3390/dj8030064
Chicago/Turabian StyleRotondi, Olivia, PhiAnh Waldon, and Sahng G. Kim. 2020. "The Disease Process, Diagnosis and Treatment of Invasive Cervical Resorption: A Review" Dentistry Journal 8, no. 3: 64. https://doi.org/10.3390/dj8030064
APA StyleRotondi, O., Waldon, P., & Kim, S. G. (2020). The Disease Process, Diagnosis and Treatment of Invasive Cervical Resorption: A Review. Dentistry Journal, 8(3), 64. https://doi.org/10.3390/dj8030064




