Polyphenols Inhibit Candida albicans and Streptococcus mutans Biofilm Formation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Biofilm Growth
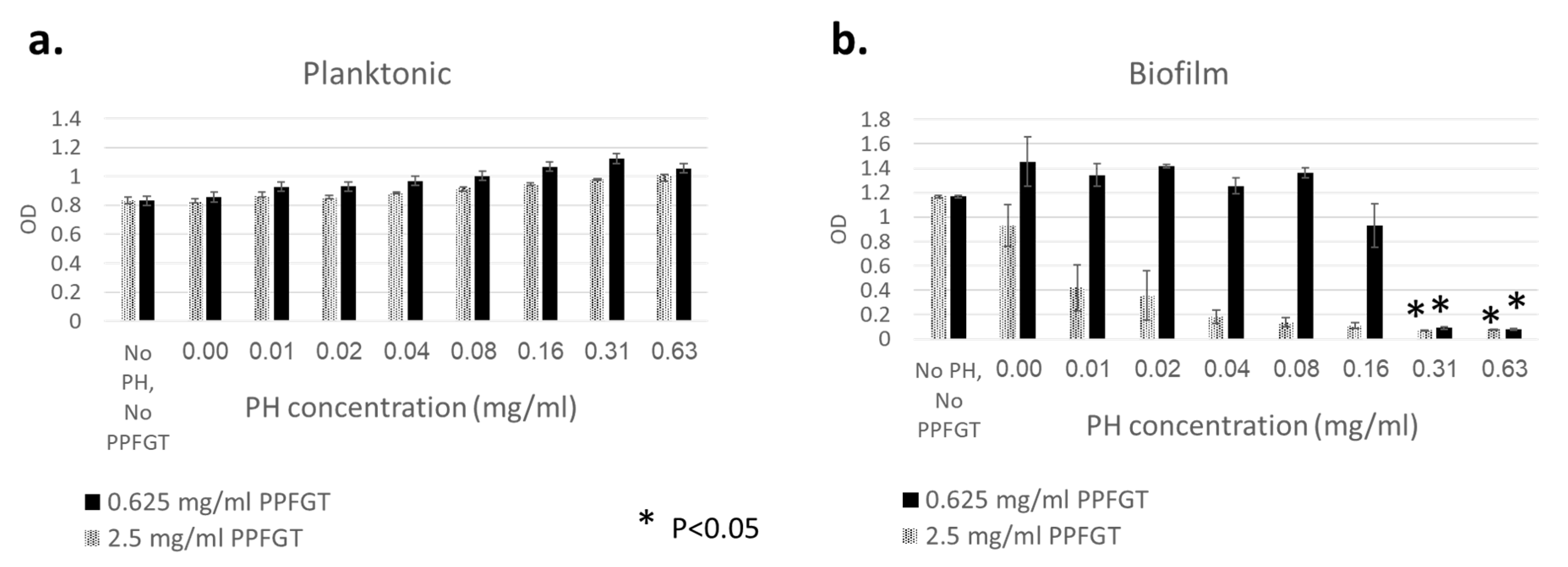
2.3. Biomass of Biofilms
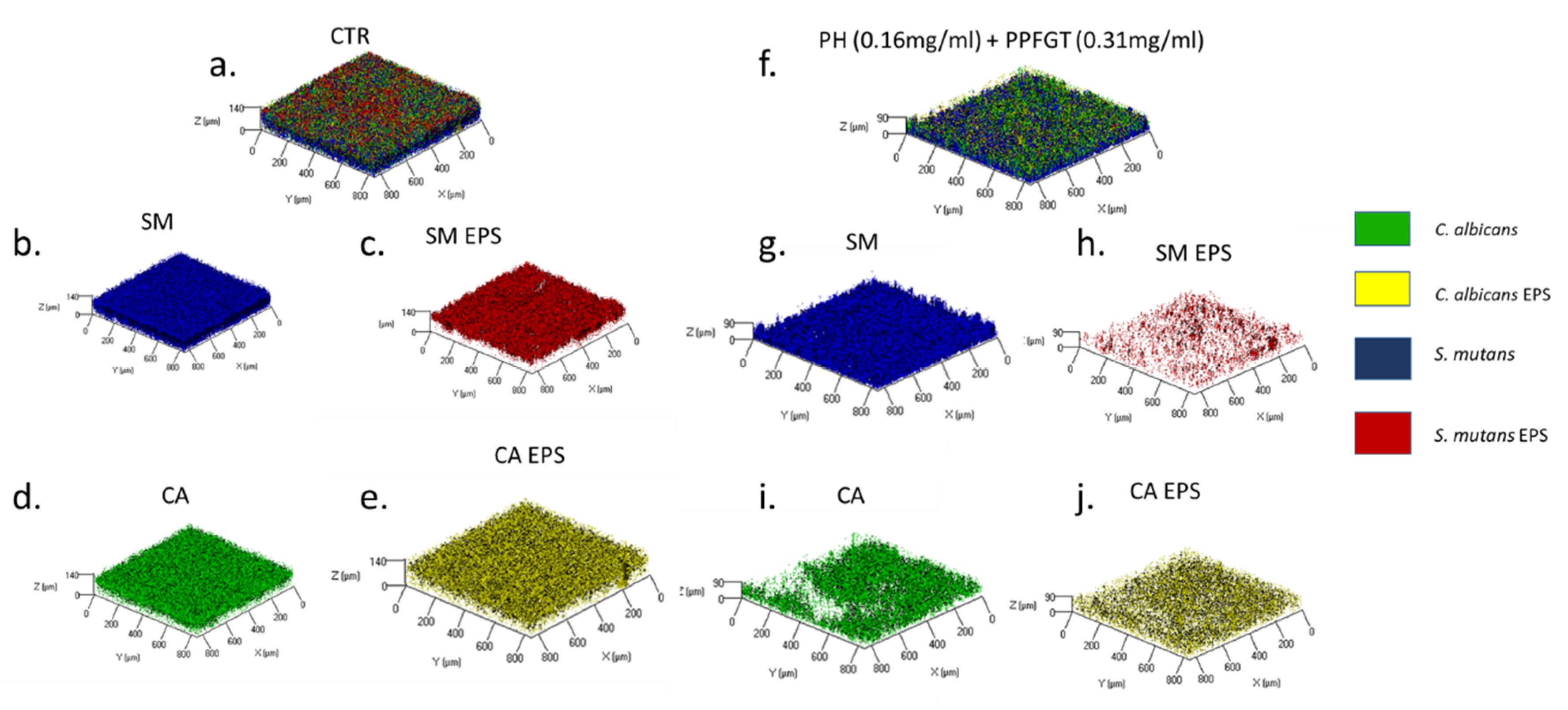
2.4. Confocal Laser Scanning Microscopy (CLSM)
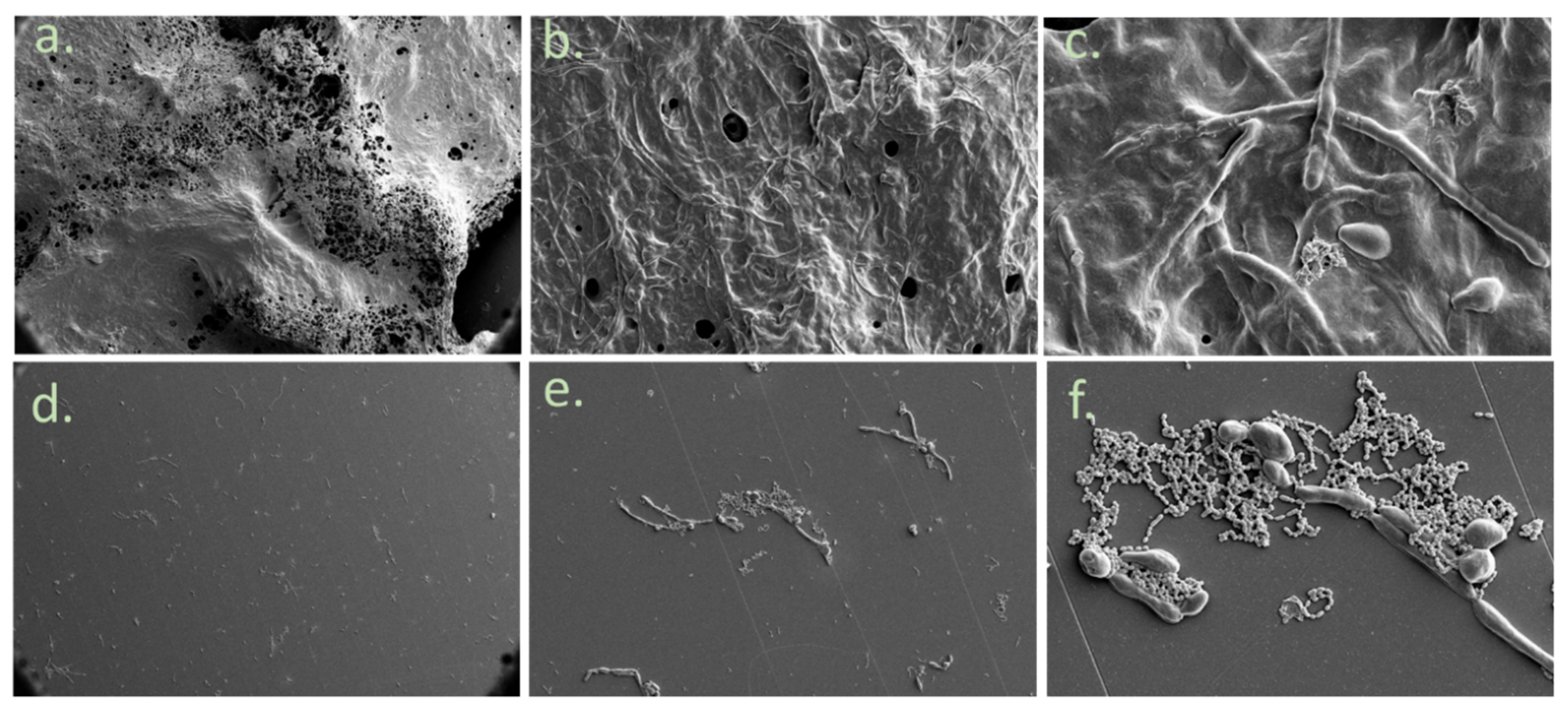
2.5. Morphology of the Biofilm
2.6. Statistical Analysis
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. Additional Controls
References
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial biofilms: A common cause of persistent infections. Science 1999, 284, 1318–1322. [Google Scholar] [CrossRef] [PubMed]
- Knoernschild, K.L.; Rogers, H.M.; Lefebvre, C.A.; Fortson, W.M.; Schuster, G.S. Endotoxin affinity for orthodontic brackets. Am. J. Orthod. Dentofac. Orthop. 1999, 115, 634–639. [Google Scholar] [CrossRef]
- Øgaard, B. Prevalence of white spot lesions in 19-near-olds: A study on untreated and orthodontically treated persons 5 years after treatment. Am. J. Orthod. Dentofac. Orthop. 1989, 96, 423–427. [Google Scholar] [CrossRef]
- Steinberg, D. Studying plaque biofilms on various dental surfaces. In Handbook of Bacterial Adhesion; Springer: Berlin, Germany, 2000; pp. 353–370. [Google Scholar]
- Koo, H.; Falsetta, M.; Klein, M. The exopolysaccharide matrix: A virulence determinant of cariogenic biofilm. J. Dent. Res. 2013, 92, 1065–1073. [Google Scholar] [CrossRef]
- Banas, J.A. Virulence properties of Streptococcus mutans. Front. Biosci. 2004, 9, 1267–1277. [Google Scholar] [CrossRef]
- Kim, J.; Sudbery, P. Candida albicans, a major human fungal pathogen. J. Microbiol. 2011, 49, 171. [Google Scholar] [CrossRef]
- Cuéllar-Cruz, M.; López-Romero, E.; Villagómez-Castro, J.C.; Ruiz-Baca, E. Candida species: New insights into biofilm formation. Future Microbiol. 2012, 7, 755–771. [Google Scholar] [CrossRef]
- Finkel, J.S.; Mitchell, A.P. Genetic control of Candida albicans biofilm development. Nat. Rev. Microbiol. 2011, 9, 109–118. [Google Scholar] [CrossRef]
- Klinke, T.; Kneist, S.; de Soet, J.J.; Kuhlisch, E.; Mauersberger, S.; Förster, A.; Klimm, W. Acid production by oral strains of Candida albicans and lactobacilli. Caries Res. 2009, 43, 83–91. [Google Scholar] [CrossRef]
- Samaranayake, L.; Hughes, A.; Weetman, D.; MacFarlane, T. Growth and acid production of Candida species in human saliva supplemented with glucose. J. Oral Pathol. Med. 1986, 15, 251–254. [Google Scholar] [CrossRef]
- Calderone, R.A.; Fonzi, W.A. Virulence factors of Candida albicans. Trends Microbiol. 2001, 9, 327–335. [Google Scholar] [CrossRef]
- Hägg, U.; Kaveewatcharanont, P.; Samaranayake, Y.; Samaranayake, L. The effect of fixed orthodontic appliances on the oral carriage of Candida species and Enterobacteriaceae. Eur. J. Orthod. 2004, 26, 623–629. [Google Scholar] [CrossRef]
- Arendorf, T.; Addy, M. Candidal carriage and plaque distribution before, during and after removable orthodontic appliance therapy. J. Clin. Periodontol. 1985, 12, 360–368. [Google Scholar] [CrossRef]
- Gündüz Arslan, S.; Akpolat, N.; Kama, J.D.; Özer, T.; Hamamcı, O. One-year follow-up of the effect of fixed orthodontic treatment on colonization by oral candida. J. Oral Pathol. Med. 2008, 37, 26–29. [Google Scholar] [CrossRef]
- Addy, M.; Shaw, W.; Hansford, P.; Hopkins, M. The effect of orthodontic appliances on the distribution of Candida and plaque in adolescents. Br. J. Orthod. 1982, 9, 158–163. [Google Scholar] [CrossRef]
- Brusca, M.; Chara, O.; Sterin-Borda, L.; Rosa, A. Influence of different orthodontic brackets on adherence of microorganisms in vitro. Angle Orthod. 2007, 77, 331–336. [Google Scholar] [CrossRef]
- Saloom, H.F.; Mohammed-Salih, H.S.; Rasheed, S.F. The influence of different types of fixed orthodontic appliance on the growth and adherence of microorganisms (in vitro study). J. Clin. Exp. Dent. 2013, 5, e36–e41. [Google Scholar] [CrossRef]
- Falsetta, M.L.; Klein, M.I.; Colonne, P.M.; Scott-Anne, K.; Gregoire, S.; Pai, C.-H.; Gonzalez, M.; Watson, G.; Krysan, D.J.; Bowen, W.H. Symbiotic relationship between Streptococcus mutans and Candida albicans synergizes the virulence of plaque-biofilms in vivo. Infect. Immun. 2014, 82, 1968–1981. [Google Scholar] [CrossRef]
- Ginsburg, I.; Koren, E.; Shalish, M.; Kanner, J.; Kohen, R. Saliva increases the availability of lipophilic polyphenols as antioxidants and enhances their retention in the oral cavity. Arch. Oral Biol. 2012, 57, 1327–1334. [Google Scholar] [CrossRef]
- Gorelik, S.; Ligumsky, M.; Kohen, R.; Kanner, J. A novel function of red wine polyphenols in humans: Prevention of absorption of cytotoxic lipid peroxidation products. FASEB J. 2008, 22, 41–46. [Google Scholar] [CrossRef]
- Dreosti, I.E. Bioactive ingredients: Antioxidants and polyphenols in tea. Nutr. Rev. 1996, 54, S51. [Google Scholar] [CrossRef]
- Khan, N.; Afaq, F.; Saleem, M.; Ahmad, N.; Mukhtar, H. Targeting multiple signaling pathways by green tea polyphenol (−)-epigallocatechin-3-gallate. Cancer Res. 2006, 66, 2500–2505. [Google Scholar] [CrossRef]
- Melok, A.; Lee, L.; Mohamed Yussof, S.; Chu, T. Green Tea Polyphenol Epigallocatechin-3-Gallate-Stearate Inhibits the Growth of Streptococcus mutans: A Promising New Approach in Caries Prevention. Dent. J. 2018, 6, 38. [Google Scholar] [CrossRef]
- Rice-evans, C.A.; Miller, N.J.; Bolwell, P.G.; Bramley, P.M.; Pridham, J.B. The relative antioxidant activities of plant-derived polyphenolic flavonoids. Free Radic. Res. 1995, 22, 375–383. [Google Scholar] [CrossRef]
- Choedon, T.; Kumar, V. Medicinal plants used in the practice of Tibetan medicine. Recent Prog. Med. Plants 2012, 34, 385–402. [Google Scholar]
- Ginsburg, I.; Koren, E.; Horani, A.; Mahamid, M.; Doron, S.; Muhanna, N.; Amer, J.; Safadi, R. Amelioration of hepatic fibrosis via Padma Hepaten is associated with altered natural killer T lymphocytes. Clin. Exp. Immunol. 2009, 157, 155–164. [Google Scholar] [CrossRef]
- Farkash, Y.; Feldman, M.; Ginsburg, I.; Steinberg, D.; Shalish, M. Green Tea Polyphenols and Padma Hepaten Inhibit Candida albicans Biofilm Formation. Evid. Based Complement. Alternat. Med. 2018, 2018. [Google Scholar] [CrossRef]
- Christensen, G.D.; Baldassarri, L.; Simpson, W.A. Methods for studying microbial colonization of plastics. Methods Enzymol. 1995, 253, 477–500. [Google Scholar]
- Chandra, J.; Kuhn, D.M.; Mukherjee, P.K.; Hoyer, L.L.; McCormick, T.; Ghannoum, M.A. Biofilm formation by the fungal pathogen Candida albicans: Development, architecture, and drug resistance. J. Bacteriol. 2001, 183, 5385–5394. [Google Scholar] [CrossRef]
- Assaf, D.; Steinberg, D.; Shemesh, M. Lactose triggers biofilm formation by Streptococcus mutans. Int. Dairy J. 2015, 42, 51–57. [Google Scholar] [CrossRef]
- Feldman, M.; Ginsburg, I.; Al-Quntar, A.; Steinberg, D. Thiazolidinedione-8 alters symbiotic relationship in C. albicans-S. mutans dual species biofilm. Front. Microbiol. 2016, 7, 140. [Google Scholar] [CrossRef]
- Xiao, J.; Grier, A.; Faustoferri, R.; Alzoubi, S.; Gill, A.; Feng, C.; Liu, Y.; Quivey, R.; Kopycka-Kedzierawski, D.; Koo, H. Association between Oral Candida and Bacteriome in Children with Severe ECC. J. Dent. Res. 2018, 97, 1468–1476. [Google Scholar] [CrossRef]
- Kim, D.; Liu, Y.; Benhamou, R.I.; Sanchez, H.; Simón-Soro, Á.; Li, Y.; Hwang, G.; Fridman, M.; Andes, D.R.; Koo, H. Bacterial-derived exopolysaccharides enhance antifungal drug tolerance in a cross-kingdom oral biofilm. ISME J. 2018, 12, 1427–1442. [Google Scholar] [CrossRef]
- Rautemaa, R.; Ramage, G. Oral candidosis–clinical challenges of a biofilm disease. Crit. Rev. Microbiol. 2011, 37, 328–336. [Google Scholar] [CrossRef]
- Krzyściak, W.; Jurczak, A.; Kościelniak, D.; Bystrowska, B.; Skalniak, A. The virulence of Streptococcus mutans and the ability to form biofilms. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 499–515. [Google Scholar] [CrossRef]
- Hernandez-Solis, S.E.; Rueda-Gordillo, F.; Flota-Alcocer, A.D.; Agullar-Ayala, F.; Rodríguez-Fernández, S.M.; Lama-Gonzalez, E.M. Influence of orthodontic appliances on the occurrence of Candida spp. in the oral cavity. Rev. Chil. Infectol. 2016, 33, 293–297. [Google Scholar] [CrossRef]
- Ogaard, B. White spot lesions during orthodontic treatment: Mechanisms and fluoride preventive aspects. Semin. Orthod. 2008, 14, 183–193. [Google Scholar] [CrossRef]
- Cildir, S.K.; Germec, D.; Sandalli, N.; Ozdemir, F.I.; Arun, T.; Twetman, S.; Caglar, E. Reduction of salivary mutans streptococci in orthodontic patients during daily consumption of yoghurt containing probiotic bacteria. Eur. J. Orthod. 2009, 31, 407–411. [Google Scholar] [CrossRef]
- Stecksén-Blicks, C.; Holgerson, P.L.; Olsson, M.; Bylund, B.; Sjöström, I.; Sköld-Larsson, K.; Kalfas, S.; Twetman, S. Effect of xylitol on mutans streptococci and lactic acid formation in saliva and plaque from adolescents and young adults with fixed orthodontic appliances. Eur. J. Oral Sci. 2004, 112, 244–248. [Google Scholar] [CrossRef]
- Beyth, N.; Redlich, M.; Harari, D.; Friedman, M.; Steinberg, D. Effect of sustained-release chlorhexidine varnish on Streptococcus mutans and Actinomyces viscosus in orthodontic patients. Am. J. Orthod. Dentofac. Orthop. 2003, 123, 345–348. [Google Scholar] [CrossRef]
- Watts, A.; Addy, M. Tooth discolouration and staining: Tooth discolouration and staining: A review of the literature. Br. Dent. J. 2001, 190, 309–316. [Google Scholar] [CrossRef]
- Matsumoto, M.; Tsuji, M.; Okuda, J.; Sasaki, H.; Nakano, K.; Osawa, K.; Shimura, S.; Ooshima, T. Inhibitory effects of cacao bean husk extract on plaque formation in vitro and in vivo. Eur. J. Oral Sci. 2004, 112, 249–252. [Google Scholar] [CrossRef]
- Lee, H.-J.; Park, H.-S.; Kim, K.-H.; Kwon, T.-Y.; Hong, S.-H. Effect of garlic on bacterial biofilm formation on orthodontic wire. Angle Orthod. 2011, 81, 895–900. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farkash, Y.; Feldman, M.; Ginsburg, I.; Steinberg, D.; Shalish, M. Polyphenols Inhibit Candida albicans and Streptococcus mutans Biofilm Formation. Dent. J. 2019, 7, 42. https://doi.org/10.3390/dj7020042
Farkash Y, Feldman M, Ginsburg I, Steinberg D, Shalish M. Polyphenols Inhibit Candida albicans and Streptococcus mutans Biofilm Formation. Dentistry Journal. 2019; 7(2):42. https://doi.org/10.3390/dj7020042
Chicago/Turabian StyleFarkash, Yosi, Mark Feldman, Isaac Ginsburg, Doron Steinberg, and Miriam Shalish. 2019. "Polyphenols Inhibit Candida albicans and Streptococcus mutans Biofilm Formation" Dentistry Journal 7, no. 2: 42. https://doi.org/10.3390/dj7020042
APA StyleFarkash, Y., Feldman, M., Ginsburg, I., Steinberg, D., & Shalish, M. (2019). Polyphenols Inhibit Candida albicans and Streptococcus mutans Biofilm Formation. Dentistry Journal, 7(2), 42. https://doi.org/10.3390/dj7020042




