Role of Brain Networks in Burning Mouth Syndrome: A Narrative Review
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Unraveling Brain Changes in Burning Mouth Syndrome
3.2. Brain Responses to Sensory Stimulation
3.3. Intrinsic Brain Activity and Structural Changes
4. Discussion
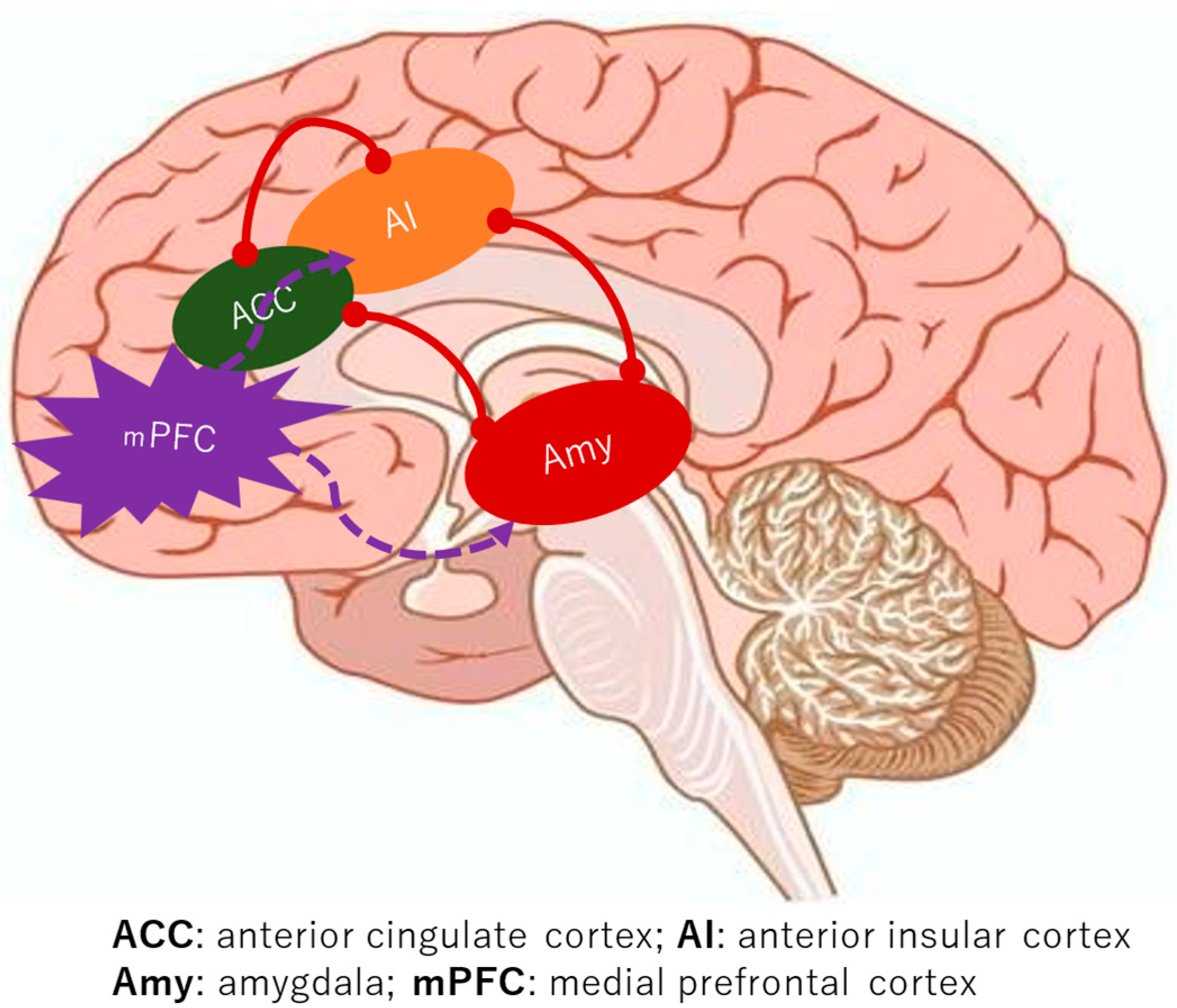
4.1. Brain Alterations in Burning Mouth Syndrome
4.2. Functional Impairment: Reconfigured Pain and Emotional Networks
4.3. Structural Abnormalities: Altered Brain Architecture
5. Limitations
6. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACC | Anterior cingulate cortex |
| AMY | Amygdala |
| BMS | Burning mouth syndrome |
| DTI | Diffusion tensor imaging |
| fMRI | Functional magnetic resonance imaging |
| Hc | Hippocampus |
| IC | Insular cortex |
| ICOP | International Classification of Orofacial Pain |
| mPFC | Medial prefrontal cortex |
| PET | Positron emission tomography |
References
- Aravindhan, R.; Vidyalakshmi, S.; Kumar, M.S.; Satheesh, C.; Balasubramanium, A.M.; Prasad, V.S. Burning mouth syndrome: A review on its diagnostic and therapeutic approach. J. Pharm. Bioallied Sci. 2014, 6 (Suppl. 1), S21–S25. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Yoo, T.; Han, P.; Liu, Y.; Inman, J.C. A pragmatic evidence-based clinical management algorithm for burning mouth syndrome. J. Clin. Exp. Dent. 2018, 10, e321–e326. [Google Scholar] [CrossRef]
- International Classification of Orofacial Pain, 1st edition (ICOP). Cephalalgia 2020, 40, 129–221. [CrossRef]
- Nagamine, T. Pathogenesis of Orofacial Pain Based on Brain Circuits. Oral Dis. 2025. [Google Scholar] [CrossRef]
- Albuquerque, R.J.C.; de Leeuw, R.; Carlson, C.R.; Okeson, J.P.; Miller, C.S.; Andersen, A.H. Cerebral activation during thermal stimulation of patients who have burning mouth disorder: An fMRI study. Pain 2006, 122, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Shinozaki, T.; Imamura, Y.; Kohashi, R.; Dezawa, K.; Nakaya, Y.; Sato, Y.; Watanabe, K.; Morimoto, Y.; Shizukuishi, T.; Abe, O.; et al. Spatial and Temporal Brain Responses to Noxious Heat Thermal Stimuli in Burning Mouth Syndrome. J. Dent. Res. 2016, 95, 1138–1146. [Google Scholar] [CrossRef] [PubMed]
- Kohashi, R.; Shinozaki, T.; Sekine, N.; Watanabe, K.; Takanezawa, D.; Nishihara, C.; Ozasa, K.; Ikeda, M.; Noma, N.; Okada-Ogawa, A.; et al. Time-dependent responses in brain activity to ongoing hot stimulation in burning mouth syndrome. J. Oral. Sci. 2020, 62, 170–174. [Google Scholar] [CrossRef]
- Yoshino, A.; Okamoto, Y.; Doi, M.; Okada, G.; Takamura, M.; Ichikawa, N.; Yamawaki, S. Functional Alterations of Postcentral Gyrus Modulated by Angry Facial Expressions during Intraoral Tactile Stimuli in Patients with Burning Mouth Syndrome: A Functional Magnetic Resonance Imaging Study. Front. Psychiatry 2017, 8, 224. [Google Scholar] [CrossRef]
- Khan, S.A.; Keaser, M.L.; Meiller, T.F.; Seminowicz, D.A. Altered structure and function in the hippocampus and medial prefrontal cortex in patients with burning mouth syndrome. Pain 2014, 155, 1472–1480. [Google Scholar] [CrossRef]
- Sinding, C.; Gransjøen, A.M.; Schlumberger, G.; Grushka, M.; Frasnelli, J.; Singh, P.B. Grey matter changes of the pain matrix in patients with burning mouth syndrome. Eur. J. Neurosci. 2016, 43, 997–1005. [Google Scholar] [CrossRef]
- Wada, A.; Shizukuishi, T.; Kikuta, J.; Yamada, H.; Watanabe, Y.; Imamura, Y.; Shinozaki, T.; Dezawa, K.; Haradome, H.; Abe, O. Altered structural connectivity of pain-related brain network in burning mouth syndrome-investigation by graph analysis of probabilistic tractography. Neuroradiology 2017, 59, 525–532. [Google Scholar] [CrossRef]
- Tan, Y.; Wu, X.; Chen, J.; Kong, L.; Qian, Z. Structural and Functional Connectivity Between the Amygdala and Orbital Frontal Cortex in Burning Mouth Syndrome: An fMRI Study. Front. Psychol. 2019, 10, 1700. [Google Scholar] [CrossRef] [PubMed]
- Kurokawa, R.; Kamiya, K.; Inui, S.; Kato, S.; Suzuki, F.; Amemiya, S.; Shinozaki, T.; Takanezawa, D.; Kohashi, R.; Abe, O. Structural connectivity changes in the cerebral pain matrix in burning mouth syndrome: A multi-shell, multi-tissue-constrained spherical deconvolution model analysis. Neuroradiology 2021, 63, 2005–2012. [Google Scholar] [CrossRef]
- Kato, S.; Kurokawa, R.; Suzuki, F.; Amemiya, S.; Shinozaki, T.; Takanezawa, D.; Kohashi, R.; Abe, O. White and Gray Matter Abnormality in Burning Mouth Syndrome Evaluated with Diffusion Tensor Imaging and Neurite Orientation Dispersion and Density Imaging. Magn. Reson. Med. Sci. 2024, 23, 204–213. [Google Scholar] [CrossRef]
- Nagamine, T. Estrogen-Mediated Neural Mechanisms of Sex Differences in Burning Mouth Syndrome. Neurol. Int. 2025, 17, 61. [Google Scholar] [CrossRef]
- Monteserín-Matesanz, M.; Domínguez-Gordillo, A.A.; Esparza-Gómez, G.C.; Jiménez-Ortega, L.; Cerero-Lapiedra, R. Central sensitization in burning mouth syndrome: A practical approach using questionnaires. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. 2022, 133, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Jaffal, S.M. Neuroplasticity in chronic pain: Insights into diagnosis and treatment. Korean J. Pain 2025, 38, 89–102. [Google Scholar] [CrossRef] [PubMed]
- Schimmelpfennig, J.; Topczewski, J.; Zajkowski, W.; Jankowiak-Siuda, K. The role of the salience network in cognitive and affective deficits. Front. Hum. Neurosci. 2023, 17, 1133367. [Google Scholar] [CrossRef] [PubMed]
- Murillo-Garcia, A.; Leon-Llamas, J.L.; Villafaina, S.; Gusi, N. Fibromyalgia impact in the prefrontal cortex subfields: An assessment with MRI. Clin. Neurol. Neurosurg. 2022, 219, 107344. [Google Scholar] [CrossRef]
- Tınastepe, N.; Oral, K. Neuropathic pain after dental treatment. Agri 2013, 25, 1–6. [Google Scholar] [CrossRef]
- Koren, T.; Yifa, R.; Amer, M.; Krot, M.; Boshnak, N.; Ben-Shaanan, T.L.; Azulay-Debby, H.; Zalayat, I.; Avishai, E.; Hajjo, H.; et al. Insular cortex neurons encode and retrieve specific immune responses. Cell 2021, 184, 5902–5915.e17. [Google Scholar] [CrossRef] [PubMed]
- Ong, W.Y.; Stohler, C.S.; Herr, D.R. Role of the Prefrontal Cortex in Pain Processing. Mol. Neurobiol. 2019, 56, 1137–1166. [Google Scholar] [CrossRef]
- Kang, D.; McAuley, J.H.; Kassem, M.S.; Gatt, J.M.; Gustin, S.M. What does the grey matter decrease in the medial prefrontal cortex reflect in people with chronic pain? Eur. J. Pain 2019, 23, 203–219. [Google Scholar] [CrossRef] [PubMed]
- Hagelberg, N.; Forssell, H.; Rinne, J.O.; Scheinin, H.; Taiminen, T.; Aalto, S.; Luutonen, S.; Någren, K.; Jääskeläinen, S. Striatal dopamine D1 and D2 receptors in burning mouth syndrome. Pain 2003, 101, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Noorani, A.; Hung, P.S.; Zhang, J.Y.; Sohng, K.; Laperriere, N.; Moayedi, M.; Hodaie, M. Pain Relief Reverses Hippocampal Abnormalities in Trigeminal Neuralgia. J. Pain 2022, 23, 141–155. [Google Scholar] [CrossRef]
- Bhatt, R.R.; Gupta, A.; Rapkin, A.; Kilpatrick, L.A.; Hamadani, K.; Pazmany, E.; Van Oudenhove, L.; Stains, J.; Aerts, L.; Enzlin, P.; et al. Altered gray matter volume in sensorimotor and thalamic regions associated with pain in localized provoked vulvodynia: A voxel-based morphometry study. Pain 2019, 160, 1529–1540. [Google Scholar] [CrossRef]
- Sugimoto, M.; Takahashi, Y.; Sugimura, Y.K.; Tokunaga, R.; Yajima, M.; Kato, F. Active role of the central amygdala in widespread mechanical sensitization in rats with facial inflammatory pain. Pain 2021, 162, 2273–2286. [Google Scholar] [CrossRef]

| P (Population) | Adults diagnosed with burning mouth syndrome |
| I (Intervention) | Studies utilizing functional magnetic resonance imaging (fMRI) to examine neural network activity |
| C (Comparison) | Healthy controls |
| O (Outcome) | Identification and description of altered neural network activity and associated brain regions implicated in the pathological mechanisms of burning mouth syndrome |
| Author (Year) | Sample Size | Study Method | Outcome Assessment | Conclusions |
|---|---|---|---|---|
| Albuquerque et al., 2006 [5] | BMS group: n = 8 (female: 8), mean age 49.1 y.o. Control group: n = 8 (female: 8), healthy subjects, mean age 50.3 y.o. | fMRI performed while applying thermal stimulation to the trigeminal nerve | In the BMS group, greater regional signal changes were observed in the right anterior cingulate cortex and bilateral precuneus during thermal stimulation of the trigeminal nerve. | The BMS group exhibited brain activation patterns analogous to those observed in patients with other types of chronic neuropathic pain disorders, with danger-detecting circuits, such as the anterior cingulate cortex, demonstrating heightened activation. |
| Shinozaki et al., 2016 [6] | BMS group: n = 16 (female: 16), postmenopausal/perimenopausal, under 65 years Control group: n = 15 (female: 15), healthy subjects, under 65 years | fMRI during repetition of noxious heat stimulus on the lower lip using Statistical Parametric Mapping 8 software | In the BMS group, activity in the anterior cingulate cortex and the para-hippocampal gyrus was connected compared to the control group. | In the BMS group, thermal stimulation of the lower lip resulted in the activation of the anterior cingulate cortex and its associated neural systems. This activation was achieved through input from the trigeminal nerve to the central nervous system. |
| Yoshino et al., 2017 [8] | BMS group: n = 27 (female: 21), mean age 44.8 y.o. Control group: 21 (female: 18), healthy subjects, mean age 46.3 y.o. | fMRI when subjects were shown pictures of angry and neutral faces to elicit emotions and were given tactile stimulation | Changes in touch-related activation in the postcentral gyrus evoked by angry facial expressions were significantly positively correlated with pain intensity in daily life in the BMS group. | In the BMS group, neural responses in the postcentral gyrus exhibited heightened sensitivity to angry facial expressions when compared with those observed in healthy subjects. This finding suggests the presence of an association between emotion and pain perception. |
| Kohashi et al., 2020 [7] | BMS group: n = 15 (female: 15), mean age 52.6 y.o. Control group: n = 15 (female: 15), healthy subjects, 49.0 y.o. | fMRI with continuous thermal stimulation | In the BMS group, the subjects responded very sensitively to stimulation of the trigeminal nerve, with the medial frontal cortex, insular cortex, and anterior cingulate cortex becoming significantly more activated than in healthy subjects. | The BMS group responded extremely sensitively to pain information generated by the trigeminal nervous system and showed coordinated activation of the so-called salience network and the medial frontal cortex. |
| Author (Year) | Sample Size | Study Method | Outcome Assessment | Conclusions |
|---|---|---|---|---|
| Khan et al., 2014 [9] | BMS group: n = 9 (female: 9), postmenopausal/perimenopausal Control group: n = 9 (female: 9), matched healthy subjects, postmenopausal/perimenopausal | Functional connectivity in resting-state fMRI using voxel-based morphometry and diffusion tensor imaging. | The BMS group exhibited an increase in gray matter volume in the hippocampus, accompanied by a decrease in gray matter volume in the medial prefrontal cortex. | A series of structural alterations were identified in the medial prefrontal cortex and the hippocampus. Concurrently, modifications in brain networks were also observed. These structural changes may have a role in the pathophysiology of mood and depressive symptoms. |
| Sinding et al., 2016 [10] | BMS group: n = 12 (female: 7), mean age 59.4 y.o. Control group 1: n = 17 (female:11), dysgeusic patients, mean age 58.4 y.o. Control group 2: n = 13 (female:10), healthy subjects, mean age 59 y.o. | Functional connectivity in fMRI using voxel-based morphometry. | In the BMS group, the severity of pain was found to be associated with alterations in gray matter density within several regions of the brain, including the anterior cingulate cortex, the cerebellar lobule, the inferior temporal lobe, and the prefrontal cortex. | A potential underlying factor in the development of BMS is the presence of inadequate pain management. This phenomenon is further compounded by the absence of a correlation between BMS and dysgeusia, suggesting that these conditions may not be attributable to analogous structural alterations within the brain. |
| Wada et al., 2017 [11] | BMS group: n = 14 (female: 14), mean age 50.9 y.o. Control group: n = 14 (female: 50.2 y.o. | Structural connectivity was calculated. The brain network of BMS brains was studied by using probabilistic tractography and graph analysis. | A substantial discrepancy in local brain connectivity was identified in the anterior cingulate cortex, medial orbitofrontal cortex, and pars orbitalis of the brain, which are components of the medial pain system. Conversely, no substantial discrepancy was identified in the lateral pain system, encompassing the somatic sensory cortex. | A structural brain network analysis was conducted, revealing alterations in the medial pain system of the pain-related brain network in patients with BMS. These alterations were found to be analogous to those observed in chronic pain patients. |
| Tan et al., 2019 [12] | BMS group: n = 26 (female: 21), mean age 52.12 y.o. Control group: n = 27 (female: 25), mean age 51.11 y.o. | Structural and functional connectivity between the amygdala and orbital frontal cortex via fMRI. | The degree of functional connectivity between the medial prefrontal cortex and the amygdala exhibited a positive correlation with the duration of illness in the BMS group. | The enhanced functional connectivity between the medial prefrontal cortex and amygdala, as observed in BMS patients, is associated with the severity of the disease. |
| Kurokawa et al., 2021 [13] | BMS group: n = 14 subjects, perimenopausal Control group: n = 11 age- and sex-matched healthy volunteers | Brain structural connectivity using probabilistic tractography and graph analysis. | The betweenness centrality exhibited a significant increase in the left insula, right amygdala, and right lateral orbitofrontal cortex, and a significant decrease in the right inferior temporal cortex in the BMS group compared to the healthy control group. | A graph analysis of brain probabilistic structural connectivity, based on diffusion imaging, revealed alterations in regions comprising the pain matrix and medial pain ascending pathway. |
| Kato et al., 2024 [14] | BMS group: n = 14 (female: 14), mean age 57.0 y.o. Control group: n = 11 (female: 11) healthy subjects, mean age 55.8 y.o. | Fractional anisotropy and volume fraction are used to examine the direction of neuronal connections. | In the BMS group, increased astrocyte volume and alterations in the microstructure of the amygdala were associated with pain. | The activation of emotional and affective neural circuits, along with structural and functional alterations, has been observed in patients diagnosed with BMS. These alterations are often associated with the presence of comorbid anxiety and depression. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagamine, T. Role of Brain Networks in Burning Mouth Syndrome: A Narrative Review. Dent. J. 2025, 13, 304. https://doi.org/10.3390/dj13070304
Nagamine T. Role of Brain Networks in Burning Mouth Syndrome: A Narrative Review. Dentistry Journal. 2025; 13(7):304. https://doi.org/10.3390/dj13070304
Chicago/Turabian StyleNagamine, Takahiko. 2025. "Role of Brain Networks in Burning Mouth Syndrome: A Narrative Review" Dentistry Journal 13, no. 7: 304. https://doi.org/10.3390/dj13070304
APA StyleNagamine, T. (2025). Role of Brain Networks in Burning Mouth Syndrome: A Narrative Review. Dentistry Journal, 13(7), 304. https://doi.org/10.3390/dj13070304







