Oral Side Effects in Patients with Metastatic Renal Cell Carcinoma Receiving the Antiangiogenic Agent Pazopanib—Report of Three Cases
Abstract
1. Introduction
2. Case Reports
2.1. Case 1
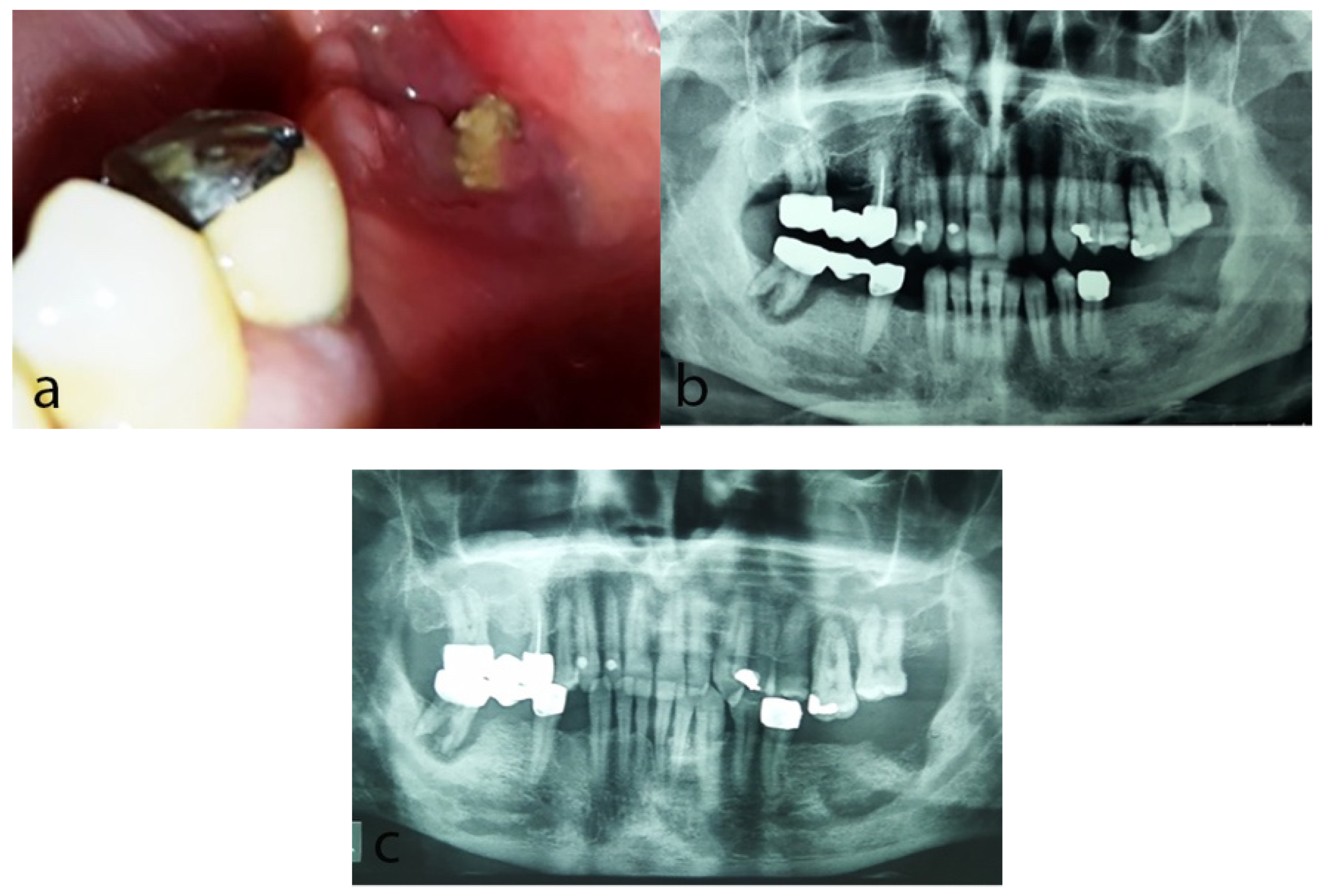
2.2. Case 2
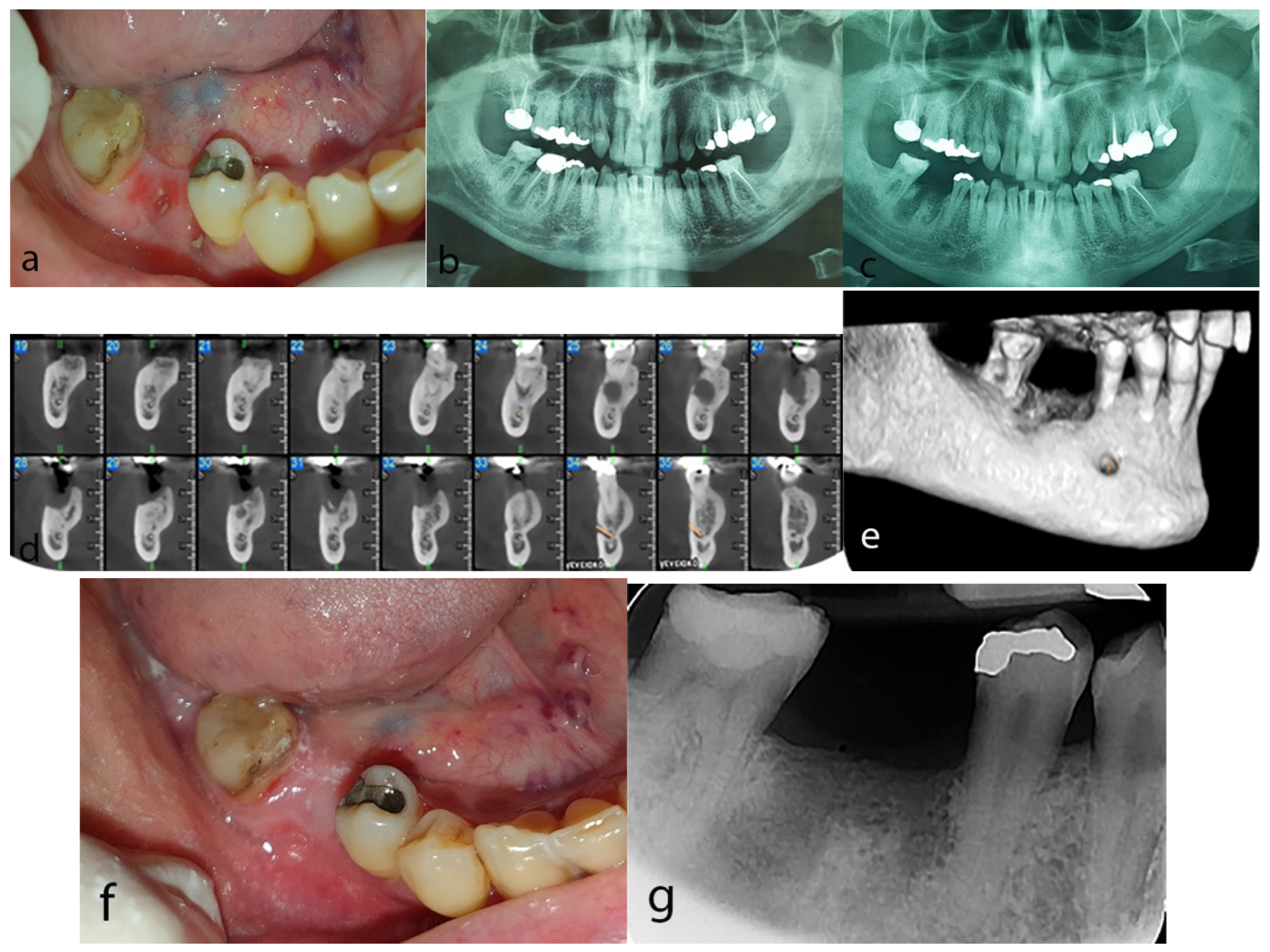
2.3. Case 3
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vigarios, E.; Epstein, J.B.; Sibaud, V. Oral mucosal changes induced by anticancer targeted therapies and immune checkpoint inhibitors. Support. Care Cancer 2017, 25, 1713–1739. [Google Scholar] [CrossRef] [PubMed]
- Chmieliauskaite, M.; Stojanov, I.; Saraghi, M.; Pinto, A. Oral adverse events associated with targeted cancer therapies. Gen. Dent. 2018, 66, 26–31. [Google Scholar] [PubMed]
- Al-Ansari, S.; Zecha, J.A.; Barasch, A.; de Lange, J.; Rozema, F.R.; Raber-Durlacher, J.E. Oral Mucositis Induced by Anticancer Therapies. Curr. Oral Health Rep. 2015, 2, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Watters, A.L.; Epstein, J.B.; Agulnik, M. Oral complications of targeted cancer therapies: A narrative literature review. Oral Oncol. 2011, 47, 441–448. [Google Scholar] [CrossRef]
- Fallah, A.; Sadeghinia, A.; Kahroba, H.; Samadi, A.; Heidari, H.R.; Bradaran, B.; Zeinali, S.; Molavi, O. Therapeutic targeting of angiogenesis molecular pathways in angiogenesis-dependent diseases. Biomed. Pharmacother. 2019, 110, 775–785. [Google Scholar] [CrossRef]
- Comandone, A.; Vana, F.; Comandone, T.; Tucci, M. Antiangiogenic Therapy in Clear Cell Renal Carcinoma (CCRC): Pharmacological Basis and Clinical Results. Cancers 2021, 13, 5896. [Google Scholar] [CrossRef]
- Sacco, R.; Woolley, J.; Patel, G.; Calasans-Maia, M.D.; Yates, J. Systematic review of medication related osteonecrosis of the jaw (MRONJ) in patients undergoing only antiangiogenic drug therapy: Surgery or conservative therapy? Br. J. Oral Maxillofac. Surg. 2022, 60, 216–230. [Google Scholar] [CrossRef]
- Caminha, R.D.G.; Chicrala, G.M.; Soares Júnior, L.A.V.; Santos, P.S.D.S. Risk profile for antiangiogenic agent-related osteonecrosis of the jaws. Einstein 2019, 17, 1–7. [Google Scholar] [CrossRef]
- Jung, T.Y. Osteonecrosis of jaw after angiogenic agent administration in a renal cell carcinoma patient. Oral Maxillofac. Surg. Cases 2017, 3, 27–33. [Google Scholar] [CrossRef]
- Antonuzzo, L.; Lunghi, A.; Petreni, P.; Brugia, M.; Laffi, A.; Giommoni, E.; Mela, M.; Mazzoni, F.; Balestri, V.; Di Constanzo, F. Osteonecrosis of the jaw and angiogenesis inhibitors: A revival of a rare but serious side effect. Curr. Med. Chem. 2017, 24, 3068–3076. [Google Scholar] [CrossRef]
- Pimolbutr, K.; Porter, S.; Fedele, S. Osteonecrosis of the Jaw Associated with Antiangiogenics in Antiresorptive-Naïve Patient: A Comprehensive Review of the Literature. Biomed. Res. Int. 2018, 23, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Lacouture, M.; Sibaud, V. Toxic Side Effects of Targeted Therapies and Immunotherapies Affecting the Skin, Oral Mucosa, Hair, and Nails. Am. J. Clin. Dermatol. 2018, 19, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Marx, R.E. Pamidronate (Aredia) and zoledronate (Zometa) induced avascular necrosis of the jaws: A growing epidemic. J. Oral Maxillofac. Surg. 2003, 61, 1115–1117. [Google Scholar] [CrossRef] [PubMed]
- Ruggiero, S.L.; Dodson, T.B.; Fantasia, J.; Goodday, R.; Aghaloo, T.; Mehrotra, B.; O’Ryan, F. American Association of Oral and Maxillofacial Surgeons. American Association of Oral and Maxillofacial Surgeons position paper on medication-related osteonecrosis of the jaw--2014 update. J. Oral Maxillofac. Surg. 2014, 72, 1938–1956. [Google Scholar] [CrossRef] [PubMed]
- Schiodt, M.; Otto, S.; Fedele, S.; Bedogni, A.; Nicolatou-Galitis, O.; Guggenberger, R.; Herlofson, B.B.; Ristow, O.; Kofod, T. Workshop of European task force on medication-related osteonecrosis of the jaw-Current challenges. Oral Dis. 2019, 25, 1815–1821. [Google Scholar] [CrossRef]
- Patel, V.; Sproat, C.; Kwok, J.; Tanna, N. Axitinib-related osteonecrosis of the jaw. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2017, 124, 257–260. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, S.; Kakutani, S.; Sato, Y.; Hanashi, A.; Kinoshita, Y.; Ishikawa, A. Drug review: Pazopanib. Jpn. J. Clin. Oncol. 2018, 48, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Hutson, T.E. Targeted therapies for the treatment of metastatic renal cell carcinoma: Clinical evidence. Oncologist 2011, 16, 14–22. [Google Scholar] [CrossRef]
- Nicolatou-Galitis, O.; Kouri, M.; Papadopoulou, E.; Vardas, E.; Galiti, D.; Epstein, J.B.; Elad, S.; Campisi, G.; Tsoukalas, N.; Bektas-Kayhan, K.; et al. Osteonecrosis of the jaw related to non-antiresorptive medications: A systematic review. Support. Care Cancer 2019, 27, 383–394. [Google Scholar] [CrossRef]
- Crist, M.; Hansen, E.; Chablani, L.; Guancial, E. Examining the bleeding incidences associated with targeted therapies used in metastatic renal cell carcinoma. Crit. Rev. Oncol. Hematol. 2017, 120, 151–162. [Google Scholar] [CrossRef]
- Nicolatou-Galitis, O.; Migkou, M.; Psyrri, A.; Bamias, A.; Pectasides, D.; Economopoulos, T.; Raber-Durlacher, J.E.; Dimitriadis, G.; Dimopoulos, M.A. Gingival bleeding and jaw bone necrosis in patients with metastatic renal cell carcinoma receiving sunitinib: Report of 2 cases with clinical implications. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2012, 113, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulou, E.; Bafaloukos, D.; Linardou, H.; Gatou, E.; Sarri, T.; Vardas, E.; Nicolatou-Galitis, O. Gingival bleeding in a patient with cervical cancer receiving bevacizumab. Report of a case. MOJ 2012, 1, 54–56. [Google Scholar]
- Boers-Doets, C.B.; Epstein, J.B.; Raber-Durlacher, J.E.; Ouwerkerk, J.; Logan, R.M.; Brakenhoff, J.A.; Lacouture, M.E.; Gelderblom, H. Oral adverse events associated with tyrosine kinase and mammalian target of rapamycin inhibitors in renal cell carcinoma: A structured literature review. Oncologist 2012, 17, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Ripamonti, C.I.; Cislaghi, E.; Mariani, L.; Maniezzo, M. Efficacy and safety of medical ozone (O3) delivered in oil suspension applications for the treatment of osteonecrosis of the jaw in patients with bone metastases treated with bisphosphonates: Preliminary results of a phase I–II study. Oral Oncol. 2011, 47, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, J.K.; Shankar, E.; Gupta, S. Chamomile: A herbal medicine of the past with bright future. Mol. Med. Rep. 2010, 3, 895–901. [Google Scholar] [PubMed]
- Bhaskaran, N.; Shukla, S.; Srivastava, J.K.; Gupta, S. Chamomile: An anti-inflammatory agent inhibits inducible nitric oxide synthase expression by blocking RelA/p65 activity. Int. J. Mol. Med. 2010, 26, 935–940. [Google Scholar]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papadopoulou, E.; Vardas, E.; Tziveleka, S.; Georgaki, M.; Kouri, M.; Katoumas, K.; Piperi, E.; Nikitakis, N.G. Oral Side Effects in Patients with Metastatic Renal Cell Carcinoma Receiving the Antiangiogenic Agent Pazopanib—Report of Three Cases. Dent. J. 2022, 10, 232. https://doi.org/10.3390/dj10120232
Papadopoulou E, Vardas E, Tziveleka S, Georgaki M, Kouri M, Katoumas K, Piperi E, Nikitakis NG. Oral Side Effects in Patients with Metastatic Renal Cell Carcinoma Receiving the Antiangiogenic Agent Pazopanib—Report of Three Cases. Dentistry Journal. 2022; 10(12):232. https://doi.org/10.3390/dj10120232
Chicago/Turabian StylePapadopoulou, Erofili, Emmanouil Vardas, Styliani Tziveleka, Maria Georgaki, Maria Kouri, Konstantinos Katoumas, Evangelia Piperi, and Nikolaos G. Nikitakis. 2022. "Oral Side Effects in Patients with Metastatic Renal Cell Carcinoma Receiving the Antiangiogenic Agent Pazopanib—Report of Three Cases" Dentistry Journal 10, no. 12: 232. https://doi.org/10.3390/dj10120232
APA StylePapadopoulou, E., Vardas, E., Tziveleka, S., Georgaki, M., Kouri, M., Katoumas, K., Piperi, E., & Nikitakis, N. G. (2022). Oral Side Effects in Patients with Metastatic Renal Cell Carcinoma Receiving the Antiangiogenic Agent Pazopanib—Report of Three Cases. Dentistry Journal, 10(12), 232. https://doi.org/10.3390/dj10120232





